| Research Article | ||
Open Vet. J.. 2026; 16(1): 452-457 Open Veterinary Journal, (2026), Vol. 16(1): 452-457 Research Article Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars)Luay O. Hamza*Department of Anatomy, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq *Corresponding Author: Luay Obed Hamza, Department of Anatomy, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq. Email: Luay.o [at] covm.uobaghdad.edu.iq Submitted: 04/08/2025 Revised: 27/11/2025 Accepted: 13/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: The fundamental histoarchitecture of the gastric glands in neonatal canines is critical for understanding the digestive physiology and its adaptation to their diet. Aim:This study was conducted to examine the histological and histochemical features of the gastric gland to document the qualitative changes occurring at this age. Methods: Gastric tissue samples were collected from six puppies and sectioned into cardiac, fundic, and pyloric regions for standard histological processing. Results: The analysis revealed a stomach wall comprising the mucosa, submucosa, muscularis, and serosa, which was lined with simple columnar epithelium but lacked a stratum compactum. The gastric glands were universally branched and simple tubular, with the cardiac region dominated by mucous secretory cells, the fundic region characterised by oxyntic and peptic cells that increased slightly with age, and the pyloric region containing primarily mucous-secreting cells. Conclusion: The study concludes that these core histological characteristics in neonatal dogs are phylogenetically conserved and share significant similarity with those of other neonatal carnivores. Keywords: Histological, Gastric glands, Neonatal, Canine. IntroductionThe domestic dog (Canis familiaris) is a domesticated large carnivore of the Canidae family (Wang, 2008; Hammodi and Hamza, 2022). Its digestive system processes and absorbs nutrients from food (Hill et al., 2008). Compared to herbivores, carnivores like dogs have a shorter digestive tract, an adaptation to their diet (Barone, 1986; Dyce et al., 2010). A dog’s stomach is a simple, monogastric organ. In newborns, the wall contains the four standard tunicae: the mucosa, submucosa, muscularis, and serosa. Its mucosal layer is glandular and features numerous gastric folds opening into gastric pits lined by simple columnar epithelium (Bacha Jr and Wood, 1990; Cavalcante de Sousa et al., 2021). Research in rats has shown that gastric morphogenesis can occur rapidly, with undifferentiated epithelium forming structured glands containing differentiated cells within 3 days of pregnancy (Alvares, 1994). In carnivores like dogs, the stomach serves essential secretory and motor functions in digestion, such as glycaemic maintenance, especially during the nutritionally vulnerable neonatal period that lasts until the second week of life (Hoskins, 2001; Grundy, 2006). This neonatal stage, the focus of neonatology, is one of significant mortality—between 17% and 30% of puppies die within the first 15 days—and is characterized by underdeveloped neurological, immunological, and behavioral systems (Crespilho, 2007; Münnich, 2008; Peterson and Kutzler, 2010; Mila et al., 2021). A recent study was conducted to examine the histological and histochemical features of the gastric gland to document the qualitative changes occurring at this age. Materials and MethodsAnimals of the studySix newborn dogs (puppies) were collected post-parturition and humanely euthanized via a deep intramuscular injection of a ketamine and xylazine mixture. Sample preparationFollowing a rinse in cold normal saline, the samples were dissected into small fragments, then fixed for 48–72 hours in 10% formalin. The tissue after fixation was processed by histological standard procedures, embedded into paraffin, and sectioned at a thickness of 4–6 µm using an electric microtome (Hamza and Balash, 2005; Bancroft and Gamble, 2008; Al-Saffar and Al-Samawy, 2016; Mohammed and Hamza, 2023). The resulting sections were then stained with Harris’s Hematoxylin and Eosin (H&E) for routine histological examination (Luna, 1968; Mohammed et al., 2017; Gharban et al., 2019; Hasan and Hamza, 2021; Al-Araji and Mohammed, 2023; Obead and Hamza, 2025). Statistical evaluationAnalysis was done using Analysis of Variance in Statistical Package for the Social Sciences to compare the thickness of tunics and the glands, parietal cells, and chief cells in the cardiac, fundic, and pyloric stomach locations. A p-value of ≤ 0.05 was employed as the statistical cut-off (Gharban, 2022). Ethical approvalThis study received ethical approval (Ref: P.G/820) from the Animal Care and Use Committee of the College of Veterinary Medicine, Baghdad University, on 7 April 2025 (Ref: P.G/820) before its initiation. ResultsIn puppies, the mucosa lining the internal stomach surface varies into a variety of regions depending on the dominant glandular structures, i.e., the cardiac, fundic, and pyloric regions. Microscopically, the gastric wall is made up of four diverse layers or tunics: mucosa, submucosa, muscularis, and serosa, all adding up to the overall structural and functional integrity of the organ (Figs. 1, 4, and 7).
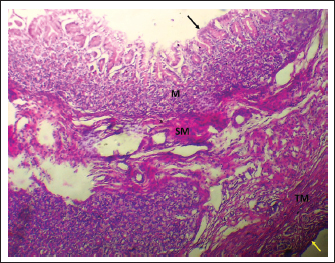
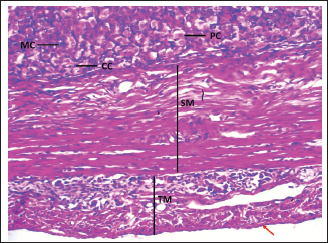
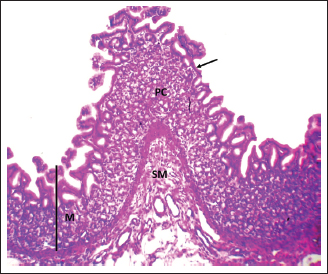
Fig. 1. Shows the four tunics of the cardiac zone in the neonatal dog: Mucosa (M), epithelium (black arrow), submucosa (SM), muscularis (TM), and serosa (red arrow). H&E stain X40.
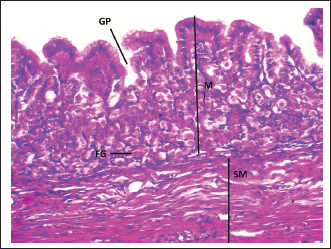
Fig. 4. Shows the fundic zone in a neonatal dog: Mucosa (M), Gastric pit (GP), Fundic Gland (FG), Submucosa (SM). H&E stain X40.
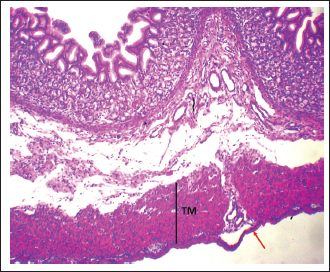
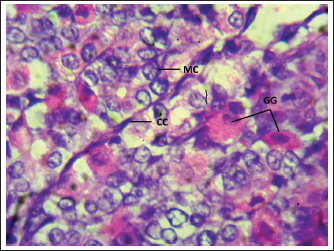
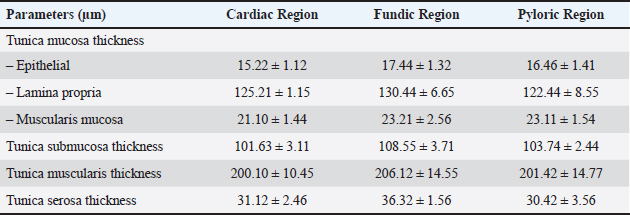
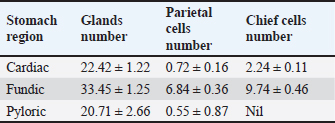
Fig. 7. Shows the pyloric zone in a neonatal dog: Tunica Muscularis (TM), and serosa (red arrow). H&E stain X40. Cardiac zoneThe cardiac glands were located exclusively near the cardiac orifice. Histologically, they were simple, branched, and tubular, forming shallow, slit-like gastric pits. The stomach wall consisted of the standard layers: a mucosa with simple columnar epithelium, a two-layered muscularis mucosa, a loose connective tissue submucosa, a two-layered muscularis with Auerbach’s plexus, and an outer serosa. In this region, the glands were composed mainly of mucous cells with low columnar shapes and basal nuclei; parietal cells were rare, and chief cells were absent (Figs. 1 and 2). The parietal cells, which had centrally placed nuclei, appeared as spherical or almost spherical bodies. The secretory units of the cardiac glands had an oval or spherical form, and their overall number was lower than that found in other stomach regions (Figs. 2 and 3). This investigation found that, in the cardiac gland area of puppies, the average measurements for epithelial height, lamina propria thickness, and the muscularis mucosa, submucosa, muscularis, and serosa layers were around (15.22 ± 1.12 µm, 125.21 ± 1.15 µm, 21.10 ± 1.44 µm, 101.63 ± 3.11 µm, 200.10 ± 10.45 µm, and 31.12 ± 2.46 µm) in that order (Table 1). For neonatal dogs, the calculated number of glands in this area reached 22.42 ± 1.22, while the mean counts for the cells of parietal and chief about (0.72 ± 0.16; 2.24 ± 0.11), respectively.
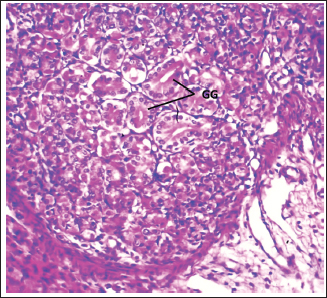
Fig. 2. Shows the cardiac gland (CG) in neonatal dog. H&E stain X100.
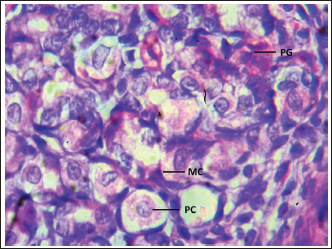
Fig. 3. Shows the cells of the cardiac zone in the neonatal dog: mucous cell (MC), Parietal cell (PC), and Chief cell (Cc). H&E stain X400. Table 1. Thickness of stomach tunics in the cardiac, fundic, and pyloric regions of neonatal dogs (µm).
Fundic zoneThe interior surface of the fundic region displayed prominent rugae that subdivided into shorter, wider secondary folds extending between the gastric crypts. Lined by a simple columnar epithelium averaging 17.44 µm in height, the mucosa featured comparatively shallow gastric pits (Fig. 4, Table 1). Long, branched tubular glands of varying size opened into the pit bases, with a mean count of 33.45 per area, and were surrounded by a loose connective tissue lamina propria (Figs. 4 and 5; Table 2). The underlying layers—the lamina propria, muscularis mucosa, and submucosa—had specific thicknesses (Table 1). The core of the submucosal rugae contained loose connective tissue along with blood vessels, lymph vessels, and scattered immune cells. The muscularis was composed of inner circular and outer longitudinal smooth muscle layers, separated by Auerbach’s plexus (Figs. 2 and 4). The thickness of the tunica muscularis was around (206.12 ± 14.55 µm), which was much greater compared to the cardiac and pyloric regions. The serosa was a layer of loose connective tissue with an average thickness of 36.32 ± 1.56 µm. A diverse mix of glandular cells, including parietal, mucous, and chief cells, was present in the fundic region. Parietal cells had large, rounded bodies, plentiful eosinophilic cytoplasm, and spherical, centrally placed nuclei. These cells were distributed throughout the fundic glands, predominantly in their upper and middle parts (Fig. 5). Average parietal and chief cell counts were (6.84 ± 0.36 and 9.74 ± 0.46), respectively.
Fig. 5. Shows the fundic zone in a neonatal dog: Mucous cell (MC), parietal cell (PC), chief cell (CC), tunica submucosa (SM), tunica muscularis (TM), and serosa (red arrow). H&E stain X100. Table 2. Number of glands, parietal cells, and chief cells in the cardiac, fundic, and pyloric regions of the stomach in neonatal dogs.
Pyloric zoneThe pyloric region was characterized by short, straight, unbifurcating folds that overlapped invaginations of the gastric crypts. Pits in this region were deeper than those in the cardiac and fundic regions. The lining was made up of simple columnar epithelium with an epithelial height of 16.46 ± 1.41 µm (Fig. 6) (Table 1). The lamina propria contained vascular connective tissue disseminated with pyloric glands (Fig. 8). The lamina propria thickness was 122.44 ± 8.55 µm, and the mean number of pyloric glands was 20.71 ± 2.66 (Tables 1 and 2). Beneath the lamina propria, the muscularis mucosa was composed of smooth muscle fibers organized into inner circular and outer longitudinal layers, with a mean thickness of 23.11 ± 1.54 µm (Fig. 7). Tunica submucosa was constituted of loose connective tissue containing blood and lymphatic vessels, collagen fibers, fibroblasts, and adipocytes. The muscularis externa contained two layers of smooth muscle fibers: an inner thick circular layer and an outer thinner longitudinal layer. The tunica submucosa and muscularis externa thicknesses were 103.74 ± 2.44 µm and 201.42 ± 14.77 µm, respectively (Table 1). The outermost layer was the serosa, which was composed of mesothelium and loose connective tissue with a mean thickness of 30.42 ± 3.56 µm (Table 1). Mucous-secreting cells formed the majority of this cell population, and there were few parietal cells, averaging 0.55 ± 0.87, and chief cells were not present (Table 2). Mucous-secreting cells were pyramidal in shape with basally placed nuclei (Fig. 8).
Fig. 6. Shows the pyloric zone in a neonatal dog: Mucosa (M), epithelial (black arrow), plica circularis (PC), submucosa (SM). H&E stain X40.
Fig. 8. Shows the pyloric glands in a neonatal dog: Pyloric glands (PG), mucous cell (MC), and parietal cell (PC). H&E stain X400. DiscussionIn newborn dogs, the gastrointestinal tract is simple and monogastric and resembles that of other carnivores such as cats (Salih and Hamza, 2022). Compared to that, the stomachs of horses, pigs, and rats both possess glandular and non-glandular regions (Brewer and Cruise, 1994). Neonatal canine stomach mucosal lining possesses longitudinal folds in all regions, which facilitate gastric distension when large amounts of food are ingested. The folds are also more pronounced when the stomach is in a fasting state, similar to that mentioned in the case of the Grey Mongoose (Hussein and Kadhim, 2019). In neonatal dogs, simple columnar epithelium lined the gastric wall of the three parts of the stomach. Such epithelium is characteristic of carnivorous animals, as reported by Zahariev et al. (2010) in the adult dog. The stomach in herbivorous animals is typically multilocular and glandular, with glandular portions lined by simple columnar epithelium and non-glandular regions covered with stratified squamous epithelium (Frandson and Spurgeon, 1992). Mucous and parietal cells appeared in the cardiac and pyloric regions of the newborn dog stomach, but not chief cells. On the other hand, the fundic zone contained mucous, parietal, and chief cells, which is a pattern characteristic of this development stage from a dietary physiology point of view. This is concordant with those occurring in neonatal cats (Salih and Hamza, 2022). The gastric glands were also discovered to be present at varying depths in the lamina propria, and in the cardiac area, they were present in the form of short, branched tubular structures with related elongated crypts, a feature comparable to that described in cat and dog by Imai et al. (1990). The fundic glands presented as simple, branched tubular structures containing chief cells and parietal cells. The presence of these types of cells signals the ongoing processes of digestion that occur in this part of the stomach and which form its largest functional area. This parallels with findings of Beasley et al. (2015), which gastric acidity is usually stronger in omnivorous and carnivorous animals than in herbivores. In the pyloric area, gastric pits were considerably deeper compared to what was observed in domestic animals’ cardiac and fundic areas. However, this structural aspect differs from Hussein and Kadhim (2019) description of the Grey Mongoose’s stomach. ConclusionThis study elucidates the histological organization of the neonatal dog stomach, with special focus on the unique structural and cellular features dividing the cardiac, fundic, and pyloric areas. Each region was seen to possess specific gland patterns and epithelial compositions in keeping with its primary functional role in digestion. The dominance of mucous-secreting cells in the cardiac and pyloric areas suggests a protective and lubricating role, and the richness of differentiated parietal and chief cells in the fundic area suggests initiation of secretory activity needed for protein digestion. Regional variations of quantitative differences in mucosal and muscular thickness further underscore mechanical and secretory-related specialization. In conclusion, the neonatal dog’s stomach histological organization is that of all young carnivores and reflects an adaptive maturation of structure to accommodate the progressing development of autonomous digestive function. The results provide a required anatomical and developmental foundation for the ensuing comparative analysis of carnivore mammals’ gastrointestinal physiology. AcknowledgmentThe author extends sincere appreciation to all members of the Anatomy and Histology Department staff of the College of Veterinary Medicine, University of Baghdad, for their kind help. Conflict of interestI declare that there are no competing interests. FundingNo specific grant was obtained from a particular funding agency for this research. Authors’ contributionsAll authors have read and approved the final draft of the manuscript. BPM collected and analyzed the data. HLS verified and interpreted the experimental results. APB supervised the experiment. BPM and NDD conceptualized the manuscript. ZZ developed the study design. BA, RM, CH, and RR validated the experimental design and contributed to manuscript revision. Data availabilityThe data supporting this research is not publicly available as it is sensitive in nature, but can be made available by the corresponding author on reasonable request. ReferencesAl-Araji, H.M. and Mohammed, H.H. 2023. Morphologic and morphometric study of lungs in adult indigenous Peafowl (Pavo cristatus). Iraqi. J. Vet. Med. 47(1), 74–79. Al-Saffar, F.J. and Al-Samawy, E.R.M. 2016. Histomorphological and histochemical study of the stomach of domestic pigeon (Columba livia domestica). Iraqi J. Vet. Med. 40(1), 89–96. Alvares, E.P. 1994. Extensive networks of TMPase positive basal lysosomes are present in fetal rat gastric epithelium before overt differentiation. J. Submicrosc. Cytol. Pathol. 26(4), 515–523. Bacha Jr, W. and Wood, L.M. 1990. Color atlas of digestive system. veterinary histology. Bacha, AP: Williams and Wilkins Scientific Publishers, pp. 111–150. Bancroft, J.D. and Gamble, M. eds., 2008. Theory and practice of histological techniques. Elsevier health sciences. Barone, R. 1986. Anatomie comparée des mammifères domestiques–Tome 1: Ostéologie–. Vigot Freres, Paris, pp. 342–356. Beasley, D.E., Koltz, A.M., Lambert, J.E., Fierer, N. and Dunn, R.R. 2015. The evolution of stomach acidity and its relevance to the human microbiome. PLos One 10(7), e0134116. Brewer, N.R. and Cruise, L.J. 1994. The Guinea pig heart--some comparative aspects. Contemp. Top. Lab. Anim. Sci. 33(6), 64–67. Cavalcante De Sousa, M.M.A., Fonseca, C.M.B., Mendonça, T.G.S. and Júnior, A.M.C. 2021. Structure of the stomach in the canine neonate. J. Interdiscip. Biociências. 6(2), 9–13. Crespilho. and A. 2007. Therapeutic approach of the canine and feline newborns: 2. Intensive care, anti-parasites and antimicrobial therapy. Rev. Bras. Reprod. Anim. 31(4), 425–432. Dyce, K. M., Sack, W. O. and Wensing, C. J. G. 2010. Veterinary Anatomy. 4th edn, pp. 680–684. University of Edinburgh, Edinburgh, Scotland. Frandson, R.D. and Spurgeon, T.L. 1992. Anatomy and physiology of farm animals. Evaluation 3, 4. Gharban, H.A. 2022. Clinic al and serological diagnosis of bovine hypodermosis in Wasit Province. Rev Electron Vet. 23(3), 457–466. Gharban, H.A.J., Al-Shaeli, S.J.J., Al-Fattli, H.H.H. and Altaee, M.N.K. 2019. Molecular and histopathological confirmation of clinically diagnosed lumpy skin disease in cattle, Baghdad Province of Iraq. Vet. World. 12(11), 1826. Grundy, S.A. 2006. Clinically relevant physiology of the neonate. Vet. Clin. Small. Anim. Pract. 36(3), 443–459. Hammodi, M.I. and Hamza, L.O. 2022. Histological and histochemical observations of the prostate gland at resting and stimulating status in adult local dog (Canis familiaris). Iraqi J. Vet. Sci. 36(3), 605–610. Hamza, L.O. and Khawla J Balash. 2005. Histological study of the quail spleen (Coturnix coturnix coturnix). Iraqi. J. Vet. Med. 29(1), 98–110. Hasan, A.M. and Hamza, L.O. 2021. Histological and histochemical observations of the vesicular gland in adult indigenous bull, Bos indicus. Biochem. Cell. Arch. 21(1), 1635–1640. Hill, R.W., Wyse, G.A. and Anderson, M. 2008 Animal Physiology. 2nd Ed., Sinauer Associates, Inc., p. 762. Sunderland, England. Hoskins, J. D., 2009. Puppy and kitten losses. Veterinary Pediatrics, 15, 57–61. Hussein, B.M. and Kadhim, K.K. 2019. Histomorphology and histochemical study of esophagus and stomach in grey mongoose (Herpestes edwardsii) in Iraq. Indian J. Nat. Sci. 9(52), 16458–11675. Imai, M., Shibata, T., Moriguchi, K., Yamamoto, M. and Kinbara, M. 1990. Do the cardiac glands exist? 6. The cat. Okajimas. Folia. Anat. Jpn. 67(1), 31–36. Luna, L.G. 1966. Manual of histological staining. Methods of Armed Forces Institute of Pathology, London, pp. 1–31. Mila, H., Guerard, C. and Raymond-Letron, I. 2021. Guidelines for postmortem examination of newborn dogs. Anim. Health Res. Rev. 22(2), 109–119. Mohammed, H.H. and Hamza, L.O. 2023. Histomorphological study of tongue in Indigenous peacock (Pavo cristatus). Revis. Bionatura. 8(3), 87. Mohammed, H.H., Abd Ali, I.K. and Reshag, A.F. 2017. Histological changes in the liver, kidney and spleen of white albino rat after aluminum chloride administration. Iraqi J. Vet. Med. 41(2), 1–6. Münnich, A. 2008. The pathological newborn in small animals: the neonate is not a small adult. Vet. Res. Commun. 32(Suppl 1), 81–85. Obead, S.S. and Hamza, L.O. 2025. Histomorphological study of the lymphoglandular complex in adult local breed dog (Canis familiaris). Iraqi. J. Vet. Sci. 39(3), 431–438. Peterson, M.E. and Kutzler, M., 2010. Small animal pediatrics: the first 12 months of life. Elsevier Health Sciences. Corvallis, Oregon. Salih, A.N. and Hamza, L.O. 2022. Histological and histochemical study of stomach in neonatal Cats. Ann. For. Res. 65(1), 6441–6452. Wang, X. 2008. Dogs: their fossil relatives and evolutionary history. Columbia University Press. Columbia University Press, New York, NY Zahariev, P., Sapundzhiev, E., Pupaki, D., Rashev, P., Palov, A. and Todorov, T. 2010. Morphological characteristics of the canine and feline stomach mucosa. Anat. Histol. Embryol. 39(6), 563–568. | ||
| How to Cite this Article |
| Pubmed Style Luay O. Hamza. Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Vet. J.. 2026; 16(1): 452-457. doi:10.5455/OVJ.2026.v16.i1.42 Web Style Luay O. Hamza. Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). https://www.openveterinaryjournal.com/?mno=275360 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.42 AMA (American Medical Association) Style Luay O. Hamza. Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Vet. J.. 2026; 16(1): 452-457. doi:10.5455/OVJ.2026.v16.i1.42 Vancouver/ICMJE Style Luay O. Hamza. Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 452-457. doi:10.5455/OVJ.2026.v16.i1.42 Harvard Style Luay O. Hamza (2026) Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Vet. J., 16 (1), 452-457. doi:10.5455/OVJ.2026.v16.i1.42 Turabian Style Luay O. Hamza. 2026. Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Veterinary Journal, 16 (1), 452-457. doi:10.5455/OVJ.2026.v16.i1.42 Chicago Style Luay O. Hamza. "Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars)." Open Veterinary Journal 16 (2026), 452-457. doi:10.5455/OVJ.2026.v16.i1.42 MLA (The Modern Language Association) Style Luay O. Hamza. "Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars)." Open Veterinary Journal 16.1 (2026), 452-457. Print. doi:10.5455/OVJ.2026.v16.i1.42 APA (American Psychological Association) Style Luay O. Hamza (2026) Histological and histochemical detection for the gastric gland in neonatal dogs (Canis familiars). Open Veterinary Journal, 16 (1), 452-457. doi:10.5455/OVJ.2026.v16.i1.42 |