| Research Article | ||
Open Vet. J.. 2026; 16(2): 1038-1047 Open Veterinary Journal, (2026), Vol. 16(2): 1038-1047 Research Article The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive ratsPutut Har Riyadi1* , Eko Susanto1, Tri Winarni Agustini1, Apri Dwi Anggo1 and Mochammad Fitri Atho’illah21Department of Fishery Product Technology, Faculty of Fisheries and Marine Science, Diponegoro University, Semarang, Indonesia 2Department of Biology, Faculty of Mathematics and Natural Sciences, Brawijaya University, Malang, Indonesia *Corresponding Author: Putut Har Riyadi. Department of Fishery Product Technology, Faculty of Fisheries and Marine Science, Diponegoro University, Semarang, Indonesia. Email: putut.riyadi [at] live.undip.ac.id Submitted: 03/08/2025 Revised: 05/01/2026 Accepted: 21/01/2026 Published: 28/02/2016 © 2026 Open Veterinary Journal
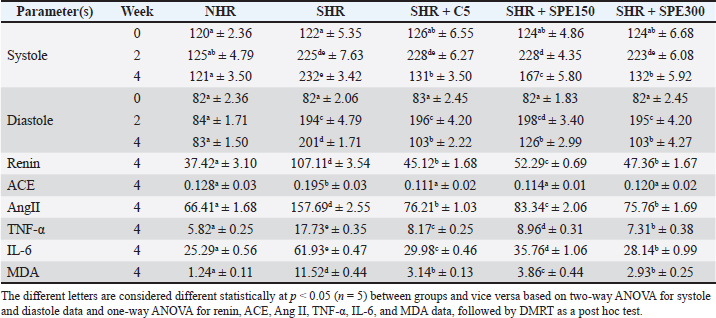
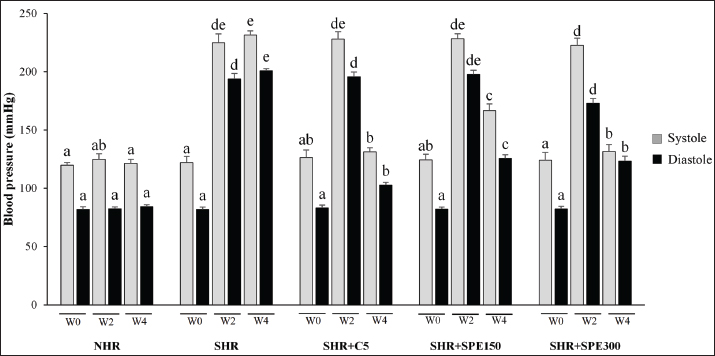
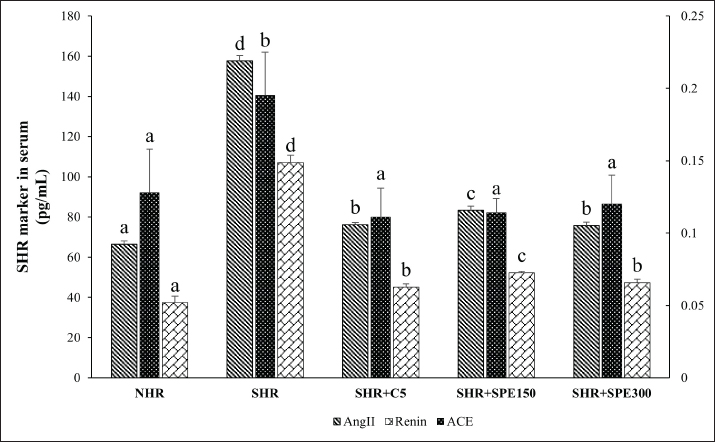
AbstractBackground: Spirulina platensis is a blue-green microalga known for its health benefits, including treating cardiovascular disease. However, research on the functional properties of S. platensis extracted via sonication remains limited. Aim: To investigate the efficacy of S. platensis hydromethanolic extract (SPE) on controlling blood pressure, renin–angiotensin–aldosterone system hormones, including renin, angiotensin-converting enzyme (ACE), and angiotensin II (Ang II), and pro-inflammatory cytokines in the spontaneously hypertensive rats (SHRs) model. Methods: Twenty-five male Wistar rats were divided into five groups (n=5), and the 20 rats were administered deoxycorticosterone acetate-salt for 5 weeks to induce SHR and treated orally with SPE for 28 days. Blood pressure was evaluated three times: initial, middle, and final treatment. The rats were then sacrificed, and the blood serum was collected. The serum was then analyzed for renin, Ang II, ACE, tumor necrosis factor-α, and interleukin (IL)-6 using enzyme-linked immunosorbent assay. Malondialdehyde was determined based on a colorimetric assay. Results: Our results demonstrated that SPE could reduce hypertensive, inflammatory, and lipid peroxidation markers significantly (p < 0.05). Conclusion: Our finding suggested that SPE improves hypertension by maintaining inflammation and antioxidants. Spirulina platensis may be regarded as a potential nutraceutical supplement S. platensis may be regarded as a potential nutraceutical supplement from fishery product for managing hypertension and other diseases associated with inflammation disturbances. Keywords: Angiotensin II, Angiotensin-converting enzyme, Hypertension, Renin, Spirulina platensis. IntroductionHypertension is a major global health issue affecting a significant portion of the world's population. Global statistics recorded that approximately 1.28 billion adults aged 30–79 years worldwide have hypertension, with about 46% population in the latent phase (Farhadi et al., 2023). Asia has a high prevalence of hypertension and has increased drastically over the past few decades (Loo et al., 2024; Rahman et al., 2024). Approximately one-third of the adult population in many Asian countries is affected by hypertension. Indonesia has the greatest prevalence of hypertension in Southeast Asia, with around 42.7% of men and 39.2% of women affected, representing a growth rate of 29% over four years (Mashuri et al., 2022; Shadiqa et al., 2025). Several factors contribute to the development of hypertension, including age, genetic predisposition, obesity, lack of physical activity, poor diet (high in salt and low in potassium), excessive alcohol consumption, and stress (Rios et al., 2023). Hypertension is a primary risk factor for cardiovascular diseases such as stroke, myocardial infarction, and heart failure and is also linked to chronic kidney disease and dementia (Song et al., 2021). Hypertension is associated with oxidative stress, an imbalance between free radicals and antioxidants in the body. Malondialdehyde (MDA) is generated during lipid peroxidation and is often utilized as a biomarker to assess oxidative stress and oxidative damage in different tissues. MDA contributed to the pathophysiology of hypertension. Elevated MDA levels increase oxidative stress, impair endothelial function, and may lead to the progression of hypertension (Verma et al., 2019). Several studies have shown that patients with hypertension have higher serum concentrations of MDA compared to normotensive individuals, suggesting an increased oxidative stress burden in these patients (Yu et al., 2018; Bastawy et al., 2025). Furthermore, oxidative stress promotes inflammation within the vascular system that involves pro-inflammatory cytokines, such as tumor necrosis factor-α (TNF-α) and interleukin (IL)-6 (Zhang et al., 2022). TNF-α increases the expression of adhesion molecules on endothelial cells, which supports the recruitment of inflammatory cells to the vessel wall and is associated with vascular inflammation (Griffin et al., 2012; Dinh et al., 2014). This inflammation can lead to vascular remodeling characterized by increased stiffness, fibrosis, and reduced elasticity, further contributing to elevated blood pressure (Kim, 2023). On the other hand, hypertension is associated with a complex interplay of various physiological factors, including the renin–angiotensin–aldosterone system (RAAS) hormones. Chronic inflammation induces the formation of reactive oxygen species (ROS), leading to oxidative stress, with both conditions complementing each other in a detrimental cycle. Further, if these conditions are unresolved, it might enhance the activity of the RAAS, including increased angiotensin II (Ang II) levels, which plays a role in vasoconstriction in the blood vessels (Zheng et al., 2020; Triebel and Castrop, 2024). The mechanism of action of RAAS, particularly through Ang II, is pivotal in promoting oxidative stress and inflammation, which in turn are involved in vascular remodeling and the pathogenesis of diseases like atherosclerosis (Koh et al., 2008; Zhao et al., 2025). Various medications have been developed to treat hypertension, including angiotensin-converting enzyme (ACE) inhibitors, Ang II receptor blockers, renin inhibitors, aldosterone antagonists, and others (Riet et al., 2015). Unfortunately, medications using synthetic drugs contribute to long-term side effects, including dry cough, hyperkalemia (Al-Janabi et al., 2025), renal dysfunction, and angioedema (Ameer et al., 2023). Nowadays, herbal medications are used as an alternative safe therapy, including for hypertension (Lai et al., 2022; Choi et al., 2024). Spirulina platensis is a blue-green microalga known for its rich nutritional profile and various bioactive compounds. Spirulina platensis contains 60%–70% of protein, including essential amino acids (Ramírez-Amaro et al., 2020). Spirulina platensis contains phycocyanin pigment, which is reported to have strong antioxidant content. Moreover, S. platensis is also rich in various active compounds, including chlorophyll, beta-carotene, vitamin E, phenolic compounds, and gamma-linolenic acid, commonly consumed as processed fishery food products for improving health and well-being (Athiyappan et al., 2024). Spirulina platensis has been studied extensively for its health benefits, including its potential for lowering blood pressure. A previous study reported that phycocyanin, the abundance pigment in S. platensis, improved endothelial nitric oxide synthase (eNOS) expression levels (Ichimura et al., 2013). Antihypertensive activity of S. platensis is assumed from antioxidants, anti-inflammatory, or its angiostatic effect (Ali et al., 2015; Brito et al., 2020). Interestingly, different geographical locations of harvest are influenced by the total phenolics, antioxidants, or chlorophyll of S. platensis (Ramon-Mascarell et al., 2022). However, there is limited information on in vivo S. platensis hydromethanolic extract (SPE) studies on the local Spirulina, which has been harvested in Indonesian waters by sonication methods for enhancing its functional properties. This study aimed to examine the efficacy of SPE on controlling blood pressure, RAAS hormones, including renin, ACE, and Ang II, and pro-inflammatory cytokines in the spontaneously hypertensive rats (SHRs) model. Materials and MethodsCollection and extraction of S. platensisSpirulina platensis powder was collected from PT Alga Bioteknologi, Indonesia. Briefly, 4.5 g of S. platensis powder was dissolved in 50% methanol (w/v), followed by continuous orbital agitation at 120 rpm at ambient temperature under low-light conditions. The mixture was then sonicated in a bath (Branson 1800) at 50 kHz of frequency in the dark, at room temperature for 30 minutes. The result was then centrifuged at 8,000 rpm for 15 minutes. The supernatant was collected and filtered using Whatman paper no. 1. The filtered supernatant was then evaporated using a rotary evaporator (Buchi Rotavapor® R-100) at 60°C to remove the solvent. SPE was then collected and stored at 4°C for further analysis. Animal experimental designTwenty-five male Wistar (Rattus norvegicus) with 200 ± 10 g body weight (BW) were divided into five groups (n=5). Twenty rats were induced into SHR using deoxycorticosterone acetate (DOCA)-salt. DOCA salt was dissolved in corn oil and administered twice a week orally for 5 weeks. In the first five injections, rats were injected with a dosage of 20 mg/kg BW and then 10 mg/kg BW for the rest of the five times (Riyadi et al., 2020). Rats were measured for systolic blood pressure (SBP) and diastolic blood pressure (DBP) using the tail-cuff method (Blood Pressure Analyzer, Kent Scientific, USA) (Riyadi et al., 2020) before injection, second week of treatment, and fourth week of treatment. Rats were then separated into five groups: normal rats, SHR, SHR treated with Captopril 5 mg/kgBW (SHR + C5), SHR treated with SPE 150 mg/kg BW (SHR + SPE150), and SHR treated with SPE 300 mg/kg BW (SHR + SPE300). The treatment was given orally for 28 days (Riyadi et al., 2020). After 28 days, the rats were then sacrificed using a combination of ketamine and xylazine. The blood was drawn from the cardiac puncture and collected in a gel clot activator tube. The blood was then centrifuged at 3,000 rpm at 10°C for 10 minutes to obtain the serum. The collected serum was then stored at 4°C for further analysis. Hypertension marker analysisHypertension marker analysis, including renin, ACE, and Ang II, was performed in the serum using enzyme-linked immunosorbent assay (ELISA). Sandwich ELISA was done by using an ELISA kit and following the procedure according to the manufacturer. The ELISA kit used was purchased from Bioassay Technology Laboratory, Shanghai, China, such as rat renin with catalogue number E0548Ra, rat angiotensin 2 with catalogue number E0655Ra, and rat ACE2 with catalogue number E0968Ra. Level of pro-inflammatory cytokine measurementPro-inflammatory cytokines, including TNF-α and IL-6, were evaluated using ELISA. TNF-α ELISA kit used rat TNF-α with catalogue number ER1393 (FineTest, Wuhan, China), and the IL-6 ELISA kit used rat IL-6 with catalogue number ER0042 (FineTest, Wuhan, China). The ELISA procedure followed the manufacturer’s protocol. Statistical analysisThe sample size of the present study was estimated by using Federer’s formula as follows: (t – 1) (r – 1) ≥ 15 (1) t=number of treatment groups, r=sample size per group The data were assessed for normality using the Kolmogorov-Smirnov test. Differences between groups were analyzed with a one-way analysis of variance (ANOVA), followed by the Duncan’s multiple range test Duncans multiple range test (DMRT) as a post hoc analysis. A p-value of <0.05 was considered statistically significant. Ethical approvalBioethics Committee for Medical/Health Research of The Faculty of Medicine, Sultan Agung Islamic University, Semarang, approved the procedure and animal handling in this experiment with an ethical clearance number 203/V/2023/KomisiBioetik. ResultsSPE lowers systolic and DBPThe results are summarized in Table 1. Our result demonstrated that the blood pressure of normal healthy rats’ groups was stable at 120–125 mmHg of SBP and 82–84 mmHg of DBP for 4 weeks. In comparison, the SHR groups exhibited a significant increase (0.00 < 0.05) in second and fourth weeks, ranging from 225 to 232 mmHg of SBP and 194 to 201 mmHg of diastole (Fig. 1). Our present results demonstrated that SPE administration for 2 weeks did not decrease blood pressure compared to the SHR groups. Interestingly, the lowering of blood pressure effect by SPE was observed at 4 weeks after treatment (Fig. 1). SPE at 300 mg/kg BW showed better results than SPE 150 mg/kg BW, and similar results with captopril 5 mg/kg BW to ameliorate both SBP and DBP in SHR (Fig. 1). SPE decreases the hypertension markerThe hypertension markers, including renin, ACE, and Ang II, significantly increased in the SHR group compared with the normal group. After 28 days of treatment, there are decreased of renin (0.00 < 0.05), ACE (0.00 < 0.05), and Ang II (0.00 < 0.05) significantly compared to the SHR group and close to the result from the SHR treated with captopril group (Fig. 2). SPE ameliorated the level of pro-inflammatory cytokineThe data analysis showed that the serum pro-inflammatory cytokines TNF-α and IL-6 in the SHR group are at the highest level between groups (Fig. 3). Administration of SPE decreased the level of TNF-α and IL-6 significantly compared with the SHR group (0.00 < 0.05). SPE 150 mg/kg BW also has results on decreasing TNF-α and IL-6. Table 1. Results summary of the present study.
Fig. 1. Systolic and DBP in normal and SHR groups. NHR, normal healthy rats (non-SHR); SHR, spontaneously hypertensive rats; SHR + C5, SHR treated with Captopril 5 mg/kg BW; SHR + SPE150, SHR treated with S. platensis extract dosage 150 mg/kg BW; SHR + SPE300, SHR treated with S. platensis extract dosage 300 mg/kg BW. The different letters are considered different statistically at p < 0.05 (n=5) between groups and vice versa based on two-way ANOVA followed by DMRT as a post hoc test.
Fig. 2. The level of renin, ACE, and Ang II in the normal and SHR groups. NHR, normal healthy rats (non-SHR); SHR, spontaneously hypertensive rats; SHR + C5, SHR treated with Captopril 5 mg/kg BW; SHR + SPE150, SHR treated with S. platensis extract dosage 150 mg/kg BW; SHR + SPE300, SHR treated with S. platensis extract dosage 300 mg/kg BW. The different letters are considered different statistically at p < 0.05 (n=5) between groups and vice versa based on one-way ANOVA followed by DMRT as a post hoc test.
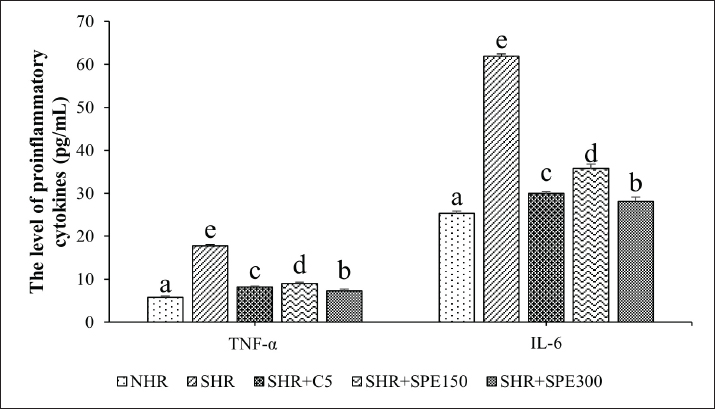
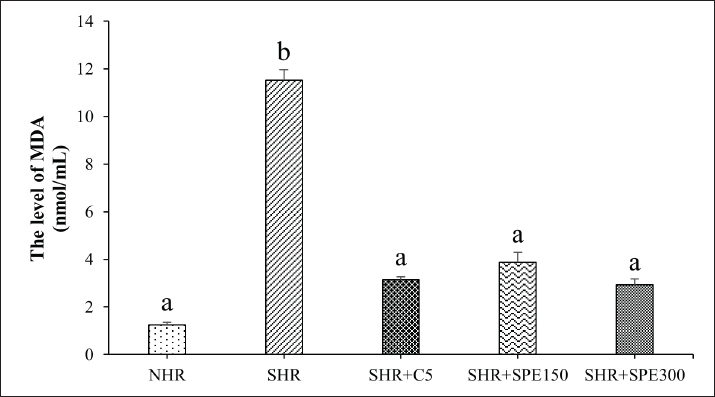
Fig. 3. The level of pro-inflammatory cytokines TNFα and IL6 in normal and SHR groups. NHR, normal healthy rats (non-SHR); SHR, spontaneously hypertensive rats; SHR + C5, SHR treated with Captopril 5 mg/kg BW; SHR + SPE150, SHR treated with S. platensis extract dosage 150 mg/kg BW; SHR + SPE300, SHR treated with S. platensis extract dosage 300 mg/kg BW. The different letters are considered different statistically at p < 0.05 (n=5) between groups and vice versa based on one-way ANOVA followed by DMRT as a post hoc test. SPE decreased the stress oxidative status MDAThe MDA analysis resulted in the SHR group has higher level of MDA compared with the normal group (0.00 < 0.05). The MDA level decreased significantly after treatment in both Captopril and SPE groups (Fig. 4). The MDA levels in the SHR group treated with Captopril and SPE decreased significantly compared with the SHR group. DiscussionSpirulina platensis contains potassium that is assumed to have a lowering effect on blood pressure (Lee et al., 2008). The lipophilic components of Spirulina are assumed to activate the potassium channels and change the activity of KATP channels and modulate the vasodilator response during hypertension (Villalpando et al., 2020). The blue pigment, phycocyanin, is recognized to have antioxidant activity and might contribute to decreasing blood pressure through elevate the eNOS in the aorta (Bobescu et al., 2020; Moradi et al., 2021). Other studies reported that Spirulina maxima supplementation reduced the endothelial marker dysfunction, such as sVCAM-1, E-selectin, and endothelin-1, and improved the arterial stiffness index (Szulinska et al., 2017; Martínez-Sámano et al., 2018). Spirulina platensis demonstrated a promising effect on blood pressure through enhancing the endothelial nitric oxide production and improving vascular function. Spirulina platensis-derived peptides induce direct endothelium-dependent vasodilatation in ex vivo vessels and have hemodynamic effects for reducing blood pressure through a PI3K/Akt signaling pathway (Carrizzo et al., 2019). A previous study reported that S. platensis peptides Ile-Gln-Pro (IQP), Val-Glu-Pro (VEP), or S. platensis hydrolysates (SH) could modulate the renin angiotensin system in SHR. IQP, VEP, and SH downregulate ACE, Ang II, and angiotensin type-1 receptor (Zheng et al., 2017). Spirulina platensis also contains phenolic acid, phycocyanin, and polysaccharides, which exhibited antihypertensive properties (Finamore et al., 2017). Moreover, S. platensis supplementation influences the gut microbiota abundance, which in turn regulates the ACE activity through the gut-heart axis (Hua et al., 2018). Spirulina platensis supplementation can significantly reduce the production of pro-inflammatory cytokines, including TNF-α, in various experimental settings (Abu-Taweel et al., 2019; Bondar et al., 2023). Similar to the previous study, Spirulina extract decreased TNF-α, along with other inflammatory markers, such as IL-6 (Nasirian et al., 2018; Abouzed et al., 2022). Phycocyanins and phenolic compounds from S. platensis inhibited nuclear factor-kappa B activation, which in turn decreased the expression of inducible Nitric Oxide Synthase (iNOS) and reduced production of NO, contributing to vascular inflammation and dysfunction (Lee et al., 2017). In contrast, the decrease of pro-inflammatory markers is followed by the increase of anti-inflammatory markers, such as IL-10 (Santos et al., 2020). This dual action might contribute to the beneficial effects of Spirulina as an anti-hypertensive Click or tap here to enter text.Click or tap here to enter text.agent.
Fig. 4. The level of MDA in normal and SHR groups. NHR, normal healthy rats (non-SHR); SHR, spontaneously hypertensive rats; SHR + C5, SHR treated with Captopril 5 mg/kg BW; SHR + SPE150, SHR treated with S. platensis extract dosage 150 mg/kg BW; SHR + SPE300, SHR treated with S. platensis extract dosage 300 mg/kg BW. The different letters are considered different statistically at p < 0.05 (n=5) between groups and vice versa based on one-way ANOVA followed by DMRT as a post hoc test. Our next result indicated that Spirulina administration significantly reduced MDA. MDA is a common marker of oxidative stress and lipid peroxidation, which is well-known to be elevated during hypertension (Riyadi et al., 2020). In line with our result, a previous work reported that S. platensis administration for 8 weeks could significantly reduce the MDA levels in plasma and tissues (Mazloomi et al., 2022). The antioxidant properties of Spirulina might contribute to modulating MDA levels. Spirulina could stimulate endogenous enzymatic and non-enzymatic antioxidants, such as glutathione peroxidase and superoxide dismutase (Mahmoud and Abd El-Ghffar, 2019). Further, the endogenous antioxidants neutralize ROS and prevent lipid peroxidation, thereby reducing MDA formation. Current research reported that C-phycocyanin in Spirulina displayed high scavenging activity, along with β-carotene, diadinoxanthin, and diatoxanthin to prevent cardiovascular diseases (Deng and Chow, 2010; Sommella et al., 2018; Grover et al., 2021). Spirulina extracts scavenge ROS and inhibit lipid peroxidation, leading to a reduction in oxidative stress-induced damage by neutralizing free radicals in a dose-dependent manner, restoring redox balance, and reducing vascular inflammation without cytotoxic effects (Gabr et al., 2020; Alves et al., 2025). Additionally, by lowering oxidative stress, Spirulina can improve vascular function and reduce hypertension-related complications (Abdel-Daim et al., 2013). In conclusion, our results demonstrated that local S. platensis with sonication methods has beneficial effects on spontaneous hypertensive rats. The administration of SPE reduces the blood pressure, TNF-α, IL-6, and MDA. The limitations of the present study are that we did not examine the urinary sodium and potassium excretion amounts and ratios, blood potassium levels, and kidney function parameters. Additionally, the changes in the gene expression related to inflammatory cytokines are not observed in the present study. Further investigations are still required to explain more details of the SPE mechanism for improving hypertension. Spirulina platensis from Indonesian waters might have the potential to be considered as a nutraceutical supplement to mitigate hypertension and other diseases related to inflammation disruption. SPE could serve as a natural, well-tolerated supplement with relatively few side effects compared to conventional antihypertensive drugs, which might be particularly beneficial in populations with mild to moderate hypertension or where pharmacotherapy side effects are a concern. Further high-quality clinical trials are necessary to clarify optimal clinical protocols and confirm their efficacy and safety in diverse patient populations for future translational applications. AcknowledgmentsThe authors thank the Faculty of Marine and Fisheries Sciences, Diponegoro University, for facilitating this research. Conflicts of interestThe authors declare no conflict of interest. FundingThis research was funded by the Ministry of Education, Culture, Research, and Technology with grant number 047/ES/PG.02.00.PL/2024 and 601-35/UN7.D2/PP/VI/2024. Authors' contributionsP.H.R. was responsible for conceptualization, funding acquisition, supervision, writing original draft, and writing-review and editing, E.S. contributed in conducted investigation, methodology, validation, writing original draft, and writing-review and editing, T.W.A. made contributions in formal analysis, validation, and writing original draft, A.D.A. was responsible for investigation, visualization, and writing original draft, and M.F.A. assisted in investigation, validation, and writing original draft. All these authors have substantial contributions to the final manuscript and have approved this submission. Data availabilityThe data that support the findings of this study are available from the corresponding author upon reasonable request under the Project funding. ReferencesAbdel-Daim, M.M., Abuzead, S.M.M. and Halawa, S.M. 2013. Protective role of Spirulina platensis against acute deltamethrin-induced toxicity in rats. PLos One. 8, e72991; doi:10.1371/journal.pone.0072991 Abouzed, T.K., Soliman, M.M., Khatab, S.A., Gouda, W.M., Eldomany, E.B. and Dorghamm, D.A. 2022. The protective impacts of Spirulina platensis against cisplatin-induced renal injury through the regulation of oxidative stress, pro-inflammatory cytokines and Bax/Bcl2. Toxicol. Res. 11, 169–178; doi:10.1093/toxres/tfab128 Abu-Taweel, G.M., Mohsen, G.A., -M.., Antonisamy, P., Arokiyaraj, S., Kim, H.,-J.., Kim, S.-J.., Park, K.H. and Kim, Y.O. 2019. Spirulina consumption effectively reduces anti-inflammatory and pain related infectious diseases. J. Infect. Public Health 12, 777–782; doi:10.1016/j.jiph.2019.04.014 Ali, E.A.I., Barakat, B.M. and Hassan, R. 2015. Antioxidant and angiostatic effect of Spirulina platensis suspension in complete freund’s adjuvant-induced arthritis in rats. PLos One. 10, 121523; doi:10.1371/journal.pone.0121523 Al-Janabi, F.L.M., Moussa, F., Taleb, S., Leutscher, P.D.C., Søndergaard, M.M., Melgaard, D., Søgaard, P., Torp-Pedersen, C., Kragholm, K. and Krogager, M.L. 2025. Risk of developing hyperkalemia in patients with hypertension treated with combination antihypertensive therapy – a retrospective register-based study. Hypertens. Res. 48, 378–387; doi:10.1038/s41440-024-01894-2 Alves, J.L.D.B., Costa, P.C.T.D., Sales, L.C.S.D., Silva Luis, C.C., Bezerra, T.P.T., Souza, M.L.A., Costa, B.A. and De Souza, E.L. 2025. Shedding light on the impacts of Spirulina platensis on gut microbiota and related health benefits. Crit. Rev. Food Sci. Nutr. 65, 2062–2075; doi:10.1080/10408398.2024.2323112 Ameer, M.A., Mushtaq, J., Chaudhry, H., Patel, N. and Khan, S.I. 2023. Angiotensin-converting enzyme inhibitor-induced angioedema: a case report with a review of management options. Cureus 15, e40320; doi:10.7759/cureus.40320 Athiyappan, K.D., Routray, W. and Paramasivan, B. 2024. Phycocyanin from Spirulina: a comprehensive review on cultivation, extraction, purification, and its application in food and allied industries. Food. Humanity. 2, 100235; doi:10.1016/j.foohum.2024.100235 Bastawy, N., El-Mosallamy, A.E.M.K., Rasheed, R.A., Sadek, A.S., Khattab, R.T., Ali, E., Zaghloul, R.A., Ghaly, W.B.A. and Boushra, A.F. 2025. Renoprotective effect of dulaglutide in L-NAME-induced hypertensive nephropathy in rats: insight into the roles of PPAR-gamma and VEGF. Hypertens. Res. 1-13; doi: 10.1038/s41440-025-02403-9 Bobescu, E., Bălan, A., Moga, M.A., Teodorescu, A., Mitrică, M. and Dima, L. 2020. Are there any beneficial effects of Spirulina supplementation for metabolic syndrome components in postmenopausal women? Mar. Drugs. 18, 651; doi:10.3390/md18120651 Bondar, A., Horodincu, L., Solcan, G. and Solcan, C. 2023. Use of Spirulina platensis and Curcuma longa as nutraceuticals in poultry. Agriculture 13, 1553; doi:10.3390/agriculture13081553 Brito, A.D.F., Silva, A.S., De Oliveira, C.V.C., De Souza, A.A., Ferreira, P.B., De Souza, I.L.L., Da Cunha Araujo, L.C., Da Silva Félix, G., De Souza Sampaio, R., Tavares, R.L., De Andrade Pereira, R., Neto, M.M. and Da Silva, B.A. 2020. Spirulina platensis prevents oxidative stress and inflammation promoted by strength training in rats: dose-response relation study. Sci. Rep. 10, 6382; doi:10.1038/s41598-020-63272-5 Carrizzo, A., Conte, G.M., Sommella, E., Damato, A., Ambrosio, M., Sala, M., Scala, M.C., Aquino, R.P., De Lucia, M., Madonna, M., Sansone, F., Ostacolo, C., Capunzo, M., Migliarino, S., Sciarretta, S., Frati, G., Campiglia, P. and Vecchione, C. 2019. Novel potent decameric peptide of Spirulina platensis reduces blood pressure levels through a PI3K/AKT/eNOS-dependent mechanism. Hypertension 73, 449–457; doi:10.1161/HYPERTENSIONAHA.118.11801 Choi, D., Im, H.B., Choi, S.J. and Han, D. 2024. Safety classification of herbal medicine use among hypertensive patients: a systematic review and meta-analysis. Front. Pharmacol. 15, 1321523; doi:10.3389/fphar.2024.1321523 Deng, R. and Chow, T.J. 2010. Hypolipidemic, antioxidant, and antiinflammatory activities of microalgae Spirulina. Cardiovasc. Ther. 28, e33-e45; doi: 10.1111/j.1755-5922.2010.00200.x Dinh, Q.N., Drummond, G.R., Sobey, C.G. and Chrissobolis, S. 2014. Roles of inflammation, oxidative stress, and vascular dysfunction in hypertension. BioMed Res. Int. 2014, 406960; doi:10.1155/2014/406960 Farhadi, F., Aliyari, R., Ebrahimi, H., Hashemi, H., Emamian, M.H. and Fotouhi, A. 2023. Prevalence of uncontrolled hypertension and its associated factors in 50–74 years old Iranian adults: a population-based study. BMC Cardiovasc. Disord. 23, 318; doi:10.1186/s12872-023-03357-x Finamore, A., Palmery, M., Bensehaila, S. and Peluso, I. 2017. Antioxidant, immunomodulating, and microbial‐modulating activities of the sustainable and ecofriendly Spirulina. Oxidative. Med. Cellular. Longevity. 2017, 3247528; doi:10.1155/2017/3247528 Gabr, G.A., El-Sayed, S.M. and Hikal, M.S. 2020. Antioxidant activities of phycocyanin: a bioactive compound from Spirulina platensis. J. Pharm. Res. Int. 32(2), 73-85; doi: 10.9734/jpri/2020/v32i230407 Griffin, G.K., Newton, G., Tarrio, M.L., Bu, D., Maganto-Garcia, E., Azcutia, V., Alcaide, P., Grabie, N., Luscinskas, F.W., Croce, K.J. and Lichtman, A.H. 2012. IL-17 and TNF-α sustain neutrophil recruitment during inflammation through synergistic effects on endothelial activation. J. Immunol. 188, 6287–6299; doi:10.4049/jimmunol.1200385 Grover, P., Bhatnagar, A., Kumari, N., Narayan Bhatt, A., Kumar Nishad, D. and Purkayastha, J. 2021. C-Phycocyanin-a novel protein from Spirulina platensis- In vivo toxicity, antioxidant and immunomodulatory studies. Saudi J. Biol. Sci. 28, 1853–1859; doi:10.1016/j.sjbs.2020.12.037 Hua, P., Yu, Z., Xiong, Y., Liu, B. and Zhao, L. 2018. Regulatory efficacy of Spirulina platensis protease hydrolyzate on lipid metabolism and gut microbiota in high-fat diet-fed rats. Int. J. Mol. Sci. 19, 4023; doi:10.3390/ijms19124023 Ichimura, M., Kato, S., Tsuneyama, K., Matsutake, S., Kamogawa, M., Hirao, E., Miyata, A., Mori, S., Yamaguchi, N., Suruga, K. and Omagari, K. 2013. Phycocyanin prevents hypertension and low serum adiponectin level in a rat model of metabolic syndrome. Nutr. Res. 33, 397–405; doi:10.1016/j.nutres.2013.03.006 Kim, H.L. 2023. Arterial stiffness and hypertension. Clin. Hypertens. 29, 31; doi:10.1186/s40885-023-00258-1 Koh, K.K., Oh, P.C. and Quon, M.J. 2008. Does reversal of oxidative stress and inflammation provide vascular protection? Cardiovasc. Res. 81, 649–659; doi:10.1093/cvr/cvn354 Lai, X., Dong, Z., Wu, S., Zhou, X., Zhang, G., Xiong, S., Wu, W., Cao, R., Wang, X., Hua, Q., Du, J., Fan, J., Mao, J., Jiang, W., Yuan, H., Chen, Y., Xu, Y., Li, Z., Zhang, J., Dong, G., Zhen, H., Ding, R., Wu, Z. and Gao, Y. 2022. Efficacy and safety of Chinese herbal medicine compared with losartan for mild essential hypertension: a randomized, multicenter, double-blind, noninferiority trial. Circ. Cardiovasc. Qual. Outcomes 15, 7923; doi:10.1161/CIRCOUTCOMES.121.007923 Lee, E.H., Park, J.E., Choi, Y.J., Huh, K.B. and Kim, W.Y. 2008. A randomized study to establish the effects of spirulina in type 2 diabetes mellitus patients. Nutr. Res. Pract. 2(4), 295; doi:10.4162/nrp.2008.2.4.295 Lee, J., Park, A., Kim, M., Lim, H.J., Rha, Y.A. and Kang, H.G. 2017. Spirulina extract enhanced a protective effect in type 1 diabetes by anti-apoptosis and anti-ROS production. Nutrients 9, 1363; doi:10.3390/nu9121363 Loo, G., Puar, T., Foo, R., Ong, T.K., Wang, T., -D.., Nguyen, Q.N., Chin, C.T. and Chin, C.W.L. 2024. Unique characteristics of Asians with hypertension: what is known and what can be done? J. Hypertens. 42, 1482; doi:10.1097/HJH.0000000000003706 Mahmoud, Y.I. and Abd El-ghffar, E.A. 2019. Spirulina ameliorates aspirin-induced gastric ulcer in albino mice by alleviating oxidative stress and inflammation. Biomed. Pharmacother. 109, 314–321; doi:10.1016/j.biopha.2018.10.118 Martínez-Sámano, J., Torres-montes De Oca, A., Luqueño-Bocardo, O.I., Torres-Durán, P.V. and Juárez-Oropeza, M.A. 2018. Spirulina maxima decreases endothelial damage and oxidative stress indicators in patients with systemic arterial hypertension: results from exploratory controlled clinical trial. Mar. Drugs. 16, 496; doi:10.3390/md16120496 Mashuri, Y.A., Ng, N. and Santosa, A. 2022. Socioeconomic disparities in the burden of hypertension among Indonesian adults - a multilevel analysis. Glob. Health Action 15, 2129131; doi:10.1080/16549716.2022.2129131 Mazloomi, S.M., Samadi, M., Davarpanah, H., Babajafari, S., Clark, C.C.T., Ghaemfar, Z., Rezaiyan, M., Mosallanezhad, A., Shafiee, M. and Rostami, H. 2022. The effect of Spirulina sauce, as a functional food, on cardiometabolic risk factors, oxidative stress biomarkers, glycemic profile, and liver enzymes in nonalcoholic fatty liver disease patients: a randomized double‐blinded clinical trial. Food. Sci. &. Nutr. 10, 317–328; doi:10.1002/fsn3.2368 Moradi, S., Zobeiri, M., Feizi, A., Clark, C.C.T. and Entezari, M.H. 2021. The effects of spirulina (Arthrospira platensis) supplementation on anthropometric indices, blood pressure, sleep quality, mental health, fatigue status and quality of life in patients with ulcerative colitis: a randomised, double‐blinded, placebo‐controlled trial. Int. J. Clin. Pract. 75(10), e14472; doi:10.1111/ijcp.14472 Nasirian, F., Dadkhah, M., Moradi-Kor, N. and Obeidavi, Z. 2018. Effects of Spirulina platensis microalgae on antioxidant and anti-inflammatory factors in diabetic rats. Diabetes Metab. Syndr. Obes. Targets Ther. 11, 375–380; doi:10.2147/DMSO.S172104 Rahman, A.R.A., Magno, J.D.A., Cai, J., Han, M., Lee, H.-Y., Nair, T., Narayan, O., Panyapat, J., Van Minh, H. and Khurana, R. 2024. Management of hypertension in the Asia-Pacific region: a structured review. Am. J. Cardiovasc. Drugs 24, 141–170; doi:10.1007/s40256-023-00625-1 Ramírez-Amaro, S., Ordines, F., Esteban, A., García, C., Guijarro, B., Salmerón, F., Terrasa, B. and Massutí, E. 2020. The diversity of recent trends for chondrichthyans in the Mediterranean reflects fishing exploitation and a potential evolutionary pressure towards early maturation. Sci. Rep. 10, 547; doi:10.1038/s41598-019-56818-9 Ramon-Mascarell, F., Martí-Quijal, F.J., Castagnini, J.M., Phimolsiripol, Y., Ruksiriwanich, W., Rajoka, M.S.R., Mehwish, H.M. and Barba, F.J. 2022. Influence of geographical location of spirulina (Arthrospira platensis) on the recovery of bioactive compounds assisted by pulsed electric fields. Separations 9, 257; doi:10.3390/separations9090257 Riet, L.T., Van Esch, J.H.M., Roks, A.J.M., Van Den Meiracker, A.H. and Danser, A.H.J. 2015. Hypertension: renin-angiotensin-aldosterone system alterations. Circ. Res. 116, 960–975; doi:10.1161/CIRCRESAHA.116.303587 Rios, F.J., Montezano, A.C., Camargo, L.L. and Touyz, R.M. 2023. Impact of environmental factors on hypertension and associated cardiovascular disease. Can. J. Cardiol. 39, 1229–1243; doi:10.1016/j.cjca.2023.07.002 Riyadi, P.H., Atho'Illah, M.F., Tanod, W.A. and Rahmawati, I.S. 2020. Tilapia viscera hydrolysate extract alleviates oxidative stress and renal damage in deoxycorticosterone acetate-salt-induced hypertension rats. Vet. World. 13, 2477–2483; doi:10.14202/vetworld.2020.2477-2483 Santos, D.S., Lauria, P.S.S., Evangelista, A.F., Azeredo, F.J., Costa, J.A.V., Soares, M.B.P., Druzian, J.I. and Villarreal, C.F. 2020. Beyond inflammation: centrally mediated antinociceptive properties of Spirulina platensis LEB-18 biomass via the opioid system. J. Funct. Foods 72, 104083; doi:10.1016/j.jff.2020.104083 Shadiqa, S., Puspawikan, P., Farabi, F. and Tiksnadi, B.B. 2025. Hypertension population growth in Indonesia: insights from BPJS-Kesehatan Big Data. J. Hypertens. 43, 12; doi:10.1097/01.hjh.0001118672.05821.93 Sommella, E., Conte, G.M., Salviati, E., Pepe, G., Bertamino, A., Ostacolo, C., Sansone, F., Prete, F.D., Aquino, R.P. and Campiglia, P. 2018. Fast profiling of natural pigments in different Spirulina (Arthrospira platensis) dietary supplements by DI-FT-ICR and evaluation of their antioxidant potential by pre-column DPPH-UHPLC assay. Molecules 23, 1152; doi:10.3390/molecules23051152 Song, M.-S., Choi, Y.J., Kim, H., Nam, M.J., Lee, C., Han, K., Jung, J.-H., Park, Y.-G., Kim, D.-H. and Park, J.-H., 2021. Relationship between blood pressure levels and ischemic stroke, myocardial infarction, and mortality in very elderly patients taking antihypertensives: a nationwide population-based cohort study. BMC Geriatr. 21, 620; doi10.1186/s12877-021-02570-7 Szulinska, M., Gibas-Dorna, M., Miller-Kasprzak, E., Suliburska, J., Miczke, A., Walczak-Gałezewska, M., Stelmach-Mardas, M., Walkowiak, J. and Bogdanski, P. 2017. Spirulina maxima improves insulin sensitivity, lipid profile, and total antioxidant status in obese patients with well-treated hypertension: a randomized double-blind placebo-controlled study. Eur. Rev. Med. Pharmacol. Sci. 21, 2473–2481. Triebel, H. and Castrop, H. 2024. The renin angiotensin aldosterone system. Pflüg. Arch. - Eur. J. Physiol. 476, 705–713; doi:10.1007/s00424-024-02908-1 Verma, M.K., Jaiswal, A., Sharma, P., Kumar, P., Narayan Singh, A. and Singh, A.N. 2019. Oxidative stress and biomarker of TNF-α, MDA and FRAP in hypertension. J. Med. Life. 12, 253–259; doi:10.25122/jml-2019-0031 Villalpando, D.M., Verdasco-Martín, C.M., Plaza, I., Gómez-Rivas, J., R De Bethencourt, F., Villarroel, M., García, J.L., Otero, C. and Ferrer, M. 2020. Beneficial effects of Spirulina aqueous extract on vasodilator function of arteries from hypertensive rats. Int. J. Vasc. Med. 2020, 1–9; doi:10.1155/2020/6657077 Yu, Y., Gao, Q., Xia, W., Zhang, L., Hu, Z., Wu, X. and Jia, X. 2018. Association between physical exercise and biomarkers of oxidative stress among middle-aged and elderly community residents with essential hypertension in China. BioMed. Res. Int. 2018, 1–11; doi:10.1155/2018/4135104 Zhang, Z., Zhao, L., Zhou, X., Meng, X. and Zhou, X. 2022. Role of inflammation, immunity, and oxidative stress in hypertension: new insights and potential therapeutic targets. Front. Immunol. 13, 1098725; doi:10.3389/fimmu.2022.1098725 Zhao, G., Wang, T., Luan, Y., Xu, Y., Qin, N., Chen, W. and Wang, Q. 2025. Research progress on the correlation between renin-angiotensin-aldosterone system (RAAS) and cardiovascular and cerebrovascular diseases. J. Renin. Angiotensin. Aldosterone. Syst. 26, 14703203251351194; doi:10.1177/14703203251351194 Zheng, J., Wang, J., Pan, H., Wu, H., Ren, D. and Lu, J. 2017. Effects of IQP, VEP and Spirulina platensis hydrolysates on the local kidney renin angiotensin system in spontaneously hypertensive rats. Mol. Med. Rep. 16, 8485–8492; doi:10.3892/mmr.2017.7602 Zheng, M., -H.., Li, F., -X.., -Z.., Xu, F., Lin, X., Wang, Y., Xu, Q., -S.., Guo, B., Yuan, L.Q. 2020. The interplay between the renin-angiotensin-aldosterone system and parathyroid hormone. Front. Endocrinol. 11, 539; doi:10.3389/fendo.2020.00539 | ||
| How to Cite this Article |
| Pubmed Style Riyadi PH, Susanto E, Agustini TW, Anggo AD, Atho'illah MF. The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Vet. J.. 2026; 16(2): 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 Web Style Riyadi PH, Susanto E, Agustini TW, Anggo AD, Atho'illah MF. The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. https://www.openveterinaryjournal.com/?mno=275103 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.24 AMA (American Medical Association) Style Riyadi PH, Susanto E, Agustini TW, Anggo AD, Atho'illah MF. The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Vet. J.. 2026; 16(2): 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 Vancouver/ICMJE Style Riyadi PH, Susanto E, Agustini TW, Anggo AD, Atho'illah MF. The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 Harvard Style Riyadi, P. H., Susanto, . E., Agustini, . T. W., Anggo, . A. D. & Atho'illah, . M. F. (2026) The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Vet. J., 16 (2), 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 Turabian Style Riyadi, Putut Har, Eko Susanto, Tri Winarni Agustini, Apri Dwi Anggo, and Mochammad Fitri Atho'illah. 2026. The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Veterinary Journal, 16 (2), 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 Chicago Style Riyadi, Putut Har, Eko Susanto, Tri Winarni Agustini, Apri Dwi Anggo, and Mochammad Fitri Atho'illah. "The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats." Open Veterinary Journal 16 (2026), 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 MLA (The Modern Language Association) Style Riyadi, Putut Har, Eko Susanto, Tri Winarni Agustini, Apri Dwi Anggo, and Mochammad Fitri Atho'illah. "The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats." Open Veterinary Journal 16.2 (2026), 1038-1047. Print. doi:10.5455/OVJ.2026.v16.i2.24 APA (American Psychological Association) Style Riyadi, P. H., Susanto, . E., Agustini, . T. W., Anggo, . A. D. & Atho'illah, . M. F. (2026) The protective effects of Spirulina platensis hydromethanolic extract against deoxycorticosterone acetate-salt-induced spontaneously hypertensive rats. Open Veterinary Journal, 16 (2), 1038-1047. doi:10.5455/OVJ.2026.v16.i2.24 |