| Review Article | ||
Open Vet. J.. 2025; 15(12): 6146-6165 Open Veterinary Journal, (2025), Vol. 15(12): 6146-6165 Review Article Review of avian influenza outbreaks in Nigeria 2006–2025Olatunde B. Akanbi1* and Tobia Lakes21Department of Veterinary Pathology, Faculty of Veterinary Medicine, University of Ilorin, Ilorin, Nigeria 2Geography Department, Humboldt-Universität zu Berlin, Berlin, Germany *Corresponding Author: Olatunde B. Akanbi. Department of Veterinary Pathology, Faculty of Veterinary Medicine, University of Ilorin, Ilorin, Nigeria. Email: akanbi.ob [at] unilorin.edu.ng Submitted: 01/08/2025 Revised: 15/11/2025 Accepted: 25/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
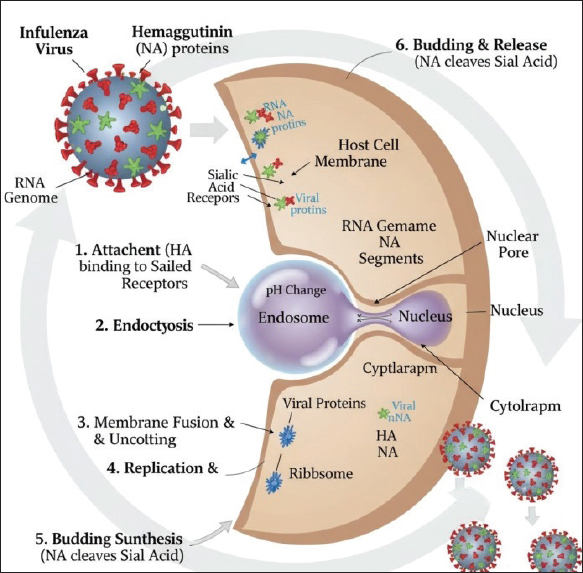
AbstractAvian influenza (AI), particularly highly pathogenic AI (HPAI) subtype H5N1, emerged as a significant global threat in the mid-2000s, causing widespread outbreaks in poultry and sporadic human infections. HPAI has persisted in Nigeria since the initial A/H5N1 incursion in 2006, leading to a chronic, cyclical, and devastating crisis with major outbreaks recurring in significant waves in 2015, 2017, and a prolonged crisis from late 2020–2025. This review synthesizes published peer-reviewed literature on AI in Nigerian poultry, encompassing wild birds and other avian species, from 2006 to 2025. It examines the epidemiology, molecular characteristics, control strategies, and socio-economic impacts of the disease outbreaks, with a specific focus on HPAI A/H5N1 and other circulating strains such as H5N2, H5N6, H5N8, and H9N2. This research reveals that AI in Nigeria is a persistent, cyclical outbreak driven not by isolated events but by systemic vulnerabilities and complex transmission pathways involving commercial and backyard poultry, as well as wild and exotic birds that facilitate viral persistence and recurrence in Nigeria. Also, it reveals Nigeria as a dynamic epicenter for viral evolution, marked by multiple introductions of A/H5N1 clades and frequent reassortment events, which increase the risk of novel, virulent strains. Epidemiologically, the triad of host, agent, and environment, including the physical and contextual factors that influence AI disease ecology as seen in Live Bird Markets (LBMs) scenarios and ubiquitous backyard poultry—which accounts for 60% of the national flock—act as critical mixing and dissemination hubs, which is pivotal to the control of the disease. Control efforts, primarily relying on stamping-out policies and biosecurity, have faced significant challenges due to implementation gaps, delayed or non-reporting, and the dynamic nature of the virus. This research demonstrates the urgent need for sustained, adaptive, and integrated "One Health" approaches, including enhanced surveillance, targeted biosecurity, and a re-evaluation of the policy on no-vaccination, to mitigate the on-going threat to animal health, food security, and public and environmental health in Nigeria. Keywords: Avian influenza viruses; Live bird markets; Epidemiology; Nigeria; One Health. IntroductionBackground on Avian influenza (AI): Global and regional contextAI, particularly highly pathogenic AI (HPAI) subtype H5N1, emerged as a significant global threat in the mid-2000s, causing widespread outbreaks in poultry and sporadic human infections (Webby and Uyeki, 2024). The H5N1 lineage, originating from A/goose/Guangdong/96, has undergone numerous evolutionary changes and extensive global dissemination, leading to a complex and evolving epidemiological landscape (WOAH, 2025). The global impact has been substantial, with HPAI causing the death and mass slaughter of over 633 million poultry between 2005 and 2024, peaking at an unprecedented 146 million in 2022 (WOAH, 2025) as a result of the on-going outbreaks in the United States of America. Furthermore, in the current seasonal wave, over 5.6 million poultry birds were culled in March 2025 alone (WOAH, 2025). The arrival of H5N1 in Nigeria in 2006 was significant with the initial number of birds lost exceeding 40,000 between January to March 2006 (Joannis et al., 2006) compared with the global total already in the millions, but the first entry point of the virus into the African continent created a "worst fears" scenario due to the nature of the structure of the Nigerian poultry industry—where 60% of production occurs in small backyard flocks living in close contact with humans—greatly complicated containment efforts and raised the risk of human exposure, contrasting with more industrialized systems elsewhere. Outbreaks trigger trade restrictions and poultry market scares, causing downstream losses for farmers and supply chains. Socioeconomic assessments revealed that compensation costs alone exceeded $11M and that the Nigerian government allocated large funds for relief (Adegboye and Kotze, 2013). The HPAI A/H5N1 clade 2.3.4.4b, which experienced a resurgence in late 2020, demonstrating the capacity to cross species barriers, thereby causing panzootic (Webby and Uyeki, 2024), among bird populations worldwide, subsequently affecting both poultry and a diverse range of terrestrial and marine mammal species, including dairy cattle (Webby and Uyeki, 2024). The reassortment and adaptation of internal genes are considered likely contributors to the virus's increased infectivity and expanded host range (Webby and Uyeki, 2024). Human infections with various AI subtypes, including H5N1, H7N9, H5N6, and H9N2, have been reported globally (WHO, 2025). Historically, from 2003 to April 2025, H5N1 alone accounted for 973 human cases, resulting in 470 deaths, a case fatality rate of 48% [WHO, 2025; Pan American Health Organization (PAHO), 2025]. While widespread human-to-human transmission has not been extensively documented, the continuous mutation and reassortment of H5N1, particularly with seasonal or swine influenza viruses, raise significant concerns regarding its potential to trigger a human pandemic [WHO, 2025; Pan American Health Organization (PAHO), 2025]. The global context highlights the persistent and evolving nature of AI, making regional analyses, such as this review focusing on Nigeria, crucial for understanding localized dynamics and informing targeted interventions. Since the initial HPAI H5N1 incursion in 2006, Nigeria has not achieved sustained eradication, resulting in persistent, devastating, and cyclical outbreaks. This crisis is driven by the complex interaction between: 1) the dynamic molecular evolution and co-circulation of multiple virulent strains (H5N1, H5N8, H9N2) within Nigeria; 2) systemic policy inadequacies (policy paradoxes, inadequate biosecurity implementation); and 3) the pivotal role of unregulated Live Bird Markets (LBMs) and backyard flocks as persistent viral reservoirs and dissemination hubs, creating an on-going, unmitigated threat to food security and public health. AI virus infection of the host cellInfluenza infection begins with attachment of the virus on the host cell, where the viral hemagglutinin (HA) protein binds to sialic acid receptors on the host cell surface. The virus then enters via endocytosis. Viral proteins are synthesized and processed in the cytoplasm, and assembly and release occurs at the cell membrane. The new virion buds off, and the Neuraminidase (NA) enzyme cleaves the bond to the host cell, freeing the infectious particle to spread to neighbouring cells, completing the cycle (Carter and Iqbal, 2024) as illustrated in Figure 1.
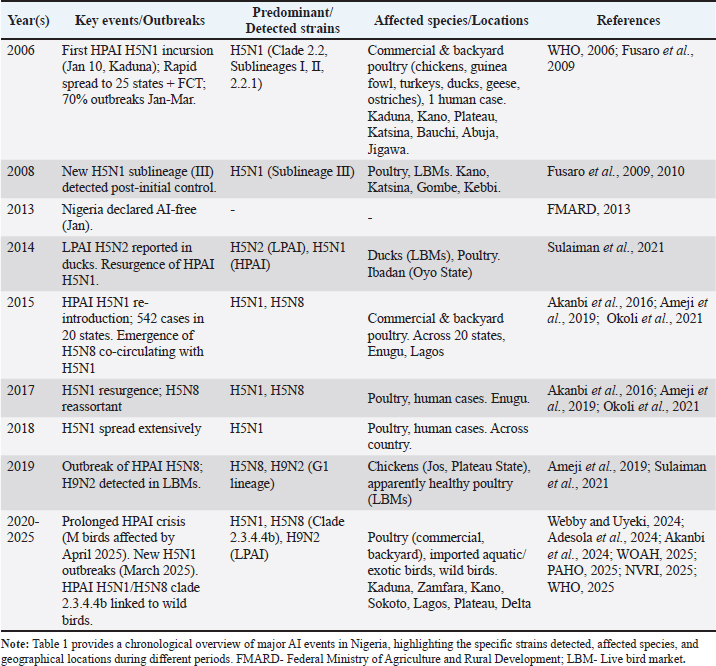
Fig. 1. Infection of the host cell by AI virus. Overview of AI in Nigeria (2006–2025)Nigeria recorded one of the earliest outbreaks of HPAI H5N1 in Africa, with the first confirmed case occurring on the 16th of January 2006 in Kaduna State. This initial incursion rapidly disseminated across the country, affecting 25 of Nigeria's 36 states and the Federal Capital Territory (FCT) within a few weeks (Akanbi et al., 2017), and the virus persisted for approximately 21 months (Fusaro et al., 2009), with 299 isolates (233 from poultry farms (153 commercial, 80 local backyard); and 66 from LBMs/feral birds) (Akanbi and Taiwo, 2014a,b). This first wave led to the loss of approximately one million birds by June 2006 alone, with a total of 1,264,191 birds depopulated by 2008, resulting in a substantial economic toll estimated at over US$5.4 million (Obayelu, 2007). Since this initial first wave of epidemics, the disease has recurred in multiple significant waves, notably in 2015 (Akanbi et al., 2017, 2018; Alhaji et al., 2023) and a prolonged crisis that began in December 2020 and continues into 2025. These recurrent outbreaks have imposed devastating socio-economic consequences on the Nigerian poultry industry and the livelihoods of numerous households dependent on poultry farming (Odita et al., 2022). The repeated nature of these events underscores the complex challenges associated with AI control in the Nigerian context. Purpose and scope of the reviewThe purpose of this review is to synthesize published peer-reviewed literature on AI in Nigeria from 2006 to 2025, with a primary focus on analyzing its epidemiological patterns, molecular characteristics, implemented control and prevention strategies, and socioeconomic impacts on domestic poultry (both commercial and backyard flocks), wild birds, and other avian species. This overview focuses on the highly pathogenic H5N1 strain, which triggered the crisis, as well as other common AI strains detected in the nation, such as H5N2, H5N6, H5N8, and H9N2 (Ameji et al., 2019; Akanbi et al., 2024). By consolidating information from this critical period, this review seeks to provide an up-to-date, holistic understanding of AI dynamics in Nigeria while identifying and highlighting key areas for future research and intervention. Materials and MethodsSearch strategy and inclusion criteriaThe review was conducted by synthesizing information from a comprehensive set of published peer-reviewed papers on AI in Nigeria, with a focus on the period from 2006 to 2025 in PubMed and Scopus databases. The search strategy employed a combination of keywords to identify relevant literature, including "AI in Nigeria," "HPAI Nigeria," "H5N1 Nigeria," "H9N2 Nigeria," "poultry Nigeria," and "wild birds Nigeria." The search and selection of publications were guided by specific inclusion criteria to ensure the comprehensiveness and relevance of the review. Papers were included if they provided information on one or more of the following aspects of AI in the Nigerian context and excluded if not. These criteria include: Epidemiology and outbreak dynamicsStudies detailing the spread, geographical distribution, and temporal patterns of outbreaks. Molecular characteristics and viral evolutionResearch on the genetic characterization, clades, sublineages, and reassortment events of circulating strains. Control and prevention strategiesAnalyses of biosecurity measures, vaccination policies, surveillance systems, and stamping-out protocols. Socio-economic impactsResearch on the economic consequences for the poultry industry and the effects on the livelihoods of farmers. The review specifically focused on HPAI H5N1 as the primary subject, but also incorporated search results on other circulating strains, including H5N2, H5N6, H5N8, and H9N2, to provide a holistic understanding of the viral landscape in Nigeria. The selected literature collectively informed the detailed discussions on the chosen aspects of the disease presented in this manuscript. Epidemiology and outbreak dynamics (2006–2025)Initial incursion and spread of HPAI H5N1 (2006–2008)The first confirmed outbreak of HPAI H5N1 in Nigeria occurred on January 16, 2006, affecting a commercial farm in Kaduna State (Adene et al., 2006; Joannis et al., 2006; NVRI, 2006; Akanbi et al., 2017). This event marked the initial detection of H5N1 on the African continent. Following this index case, the virus rapidly disseminated, impacting commercial poultry farms in several contiguous states, including Kano, Plateau, Katsina, Bauchi, and the Abuja area. In a few weeks, the virus disseminated throughout 25 of Nigeria's 36 states and the FCT (Akanbi et al., 2017; Fusaro et al., 2009). The early phase of the HPAI epidemic saw a high concentration of outbreaks, with approximately 70% of the initial 145 reported incidents occurring between January and March 2006. The rapid spread was notably linked to areas with high concentrations of poultry farms, such as the Kano metropolis, particularly along Gwarzo road, where the disease was first observed (Saidu et al., 2008) (Table 1). By the end of 2007, official reports indicated that control measures, including stamping-out and enhanced surveillance, had successfully contained the outbreaks. Table 1. Timeline of major AI outbreaks and detected strains in Nigeria (2006–2025).
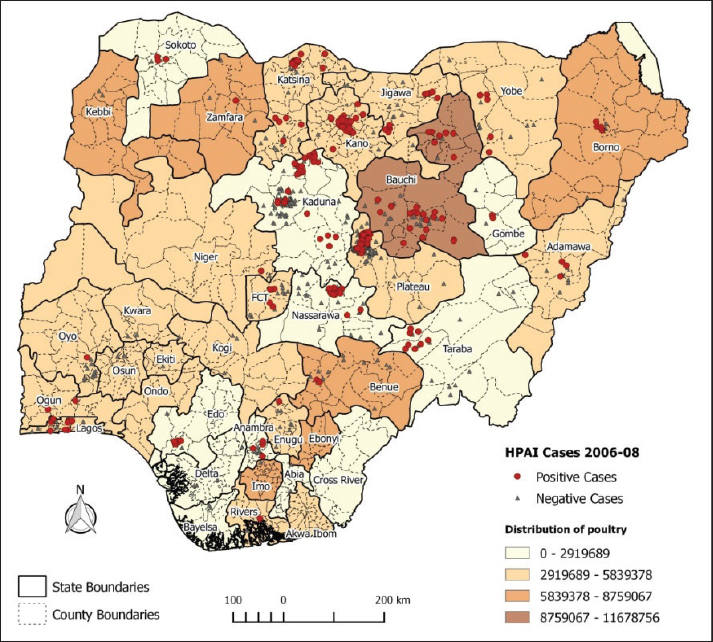
However, the notion of "control" during this period appears to have been a temporary suppression rather than a complete eradication of the virus. Evidence suggests that new cases of H5N1, originating from a previously undetected sublineage (sublineage III), were reported (Fusaro et al., 2009) in Kano and Katsina states, as well as in LBMs in Gombe and Kebbi states (Fusaro et al., 2009). This re-emergence, even after initial containment efforts, indicates that underlying vulnerabilities within the poultry sector and broader ecosystem, such as porous borders, unregulated trade, and gaps in biosecurity, likely remained unaddressed. These factors might have inadvertently led to subsequent re-introductions and recurrent waves of infection. Figure 2 shows the spatial distribution of the 2006–2008 AI outbreaks in Nigeria.
Fig. 2. Spatial distribution of HPAI H5N1 outbreaks in poultry, Nigeria, January 2006 to July 2008. Adapted and modified from Akanbi and Taiwo, 2015. Recurrent outbreaks and resurgences (2015 onwards, including 2020–2025)The HPAI H5N1 virus re-emerged in commercial poultry in Nigeria in 2015, signifying a major incursion into the country after a long period of no official reports. This resurgence was significant, with over 542 Influenza A subtype H5N1 cases confirmed from a total number of 1,137 suspicion across 20 states between January and December 2015. Several sporadic outbreaks occurred between 2017 and 2025 with various subtypes of the virus. Investigations revealed that the heightened transmission and severity of the 2015–2016 resurgence (Fig. 3) were directly attributable to a decline in surveillance activities and inadequate biosecurity measures within poultry farms.
Fig. 3. Spatial distribution of HPAI H5N1 outbreaks in poultry, Nigeria, January to December, 2015. Adapted from Akanbi et al., 2016. From December 2020, Nigeria has been grappling with a prolonged HPAI crisis, and diagnosed infections in December 2024 led to seven confirmed HPAI cases by January 2025 (NVRI, 2025). As of January 2025, Kano State alone reported six confirmed HPAI cases and 4,470 suspected cases. New H5N1 outbreaks were identified in Kaduna, Zamfara, and Kano states in March 2025. By April 2025, it was reported that 475 outbreaks recorded, impacted over 2.47 million birds nationwide. Reports as recent as November 2025 confirm the continuation of this crisis. This persistent pattern demonstrates that the virus continues to resurface despite the culling and containment efforts. The outbreak figures are already going up as December 2025 draws near and wintering birds begin to arrive from Europe. Germany and other European countries have begun to detect AI virus in wild birds [Freidrich Loeffler Institute (FLI), 2025], while cases are still being reported from the USA. The repeated re-emergence of HPAI H5N1 in Nigeria, observed in distinct waves in 2015, suggests a deeply entrenched, cyclical pattern rather than a series of isolated events (Odita et al., 2022), and the detection of H9N2 in Ibadan Oyo State (Akanbi et al., 2024) (Fig. 4), and in Kaduna and Kano (Mkpuma et al., 2024), both in commercial poultry raises additional concerns. Factors such as the relaxation of surveillance and biosecurity protocols during inter-epidemic periods and continuous viral introductions via migratory birds exacerbate this cyclical nature. The implication of this recurring pattern is that reactive, episodic control policies, often characterized by "stamping out" measures followed by a relaxation of vigilance, are insufficient to achieve long-term disease suppression or eradication. A more effective approach would necessitate a sustained, adaptive strategy that integrates continuous, robust surveillance, stringent biosecurity protocols, and potentially targeted vaccination programs. Furthermore, effective community engagement is essential to foster adherence to preventive measures and break this cycle of recurrent outbreaks, thereby safeguarding both animal and public health.
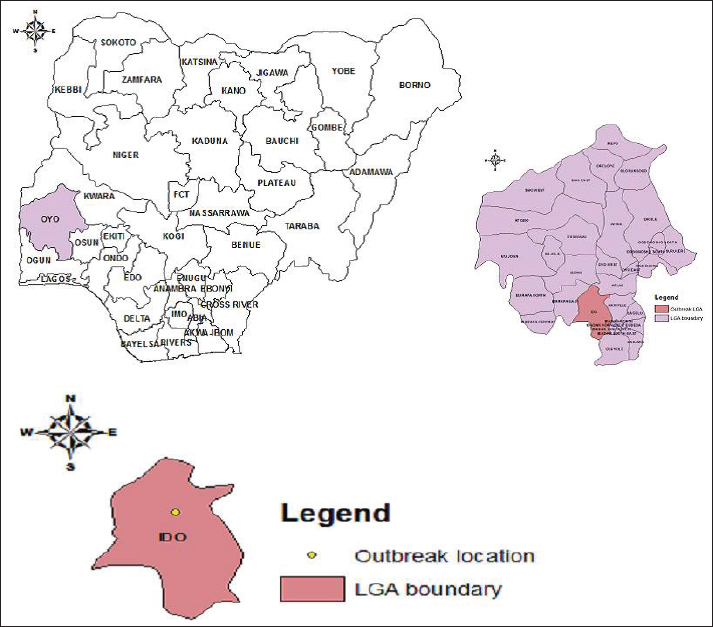
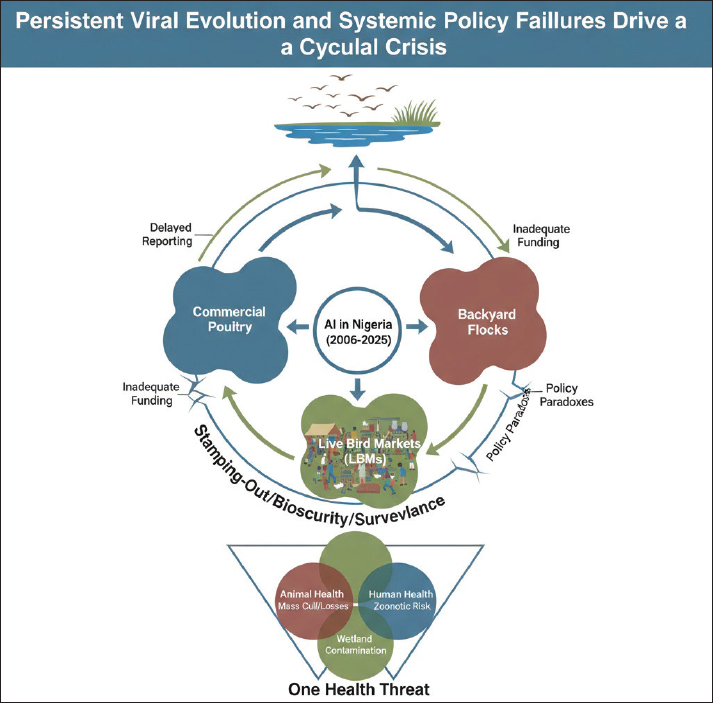
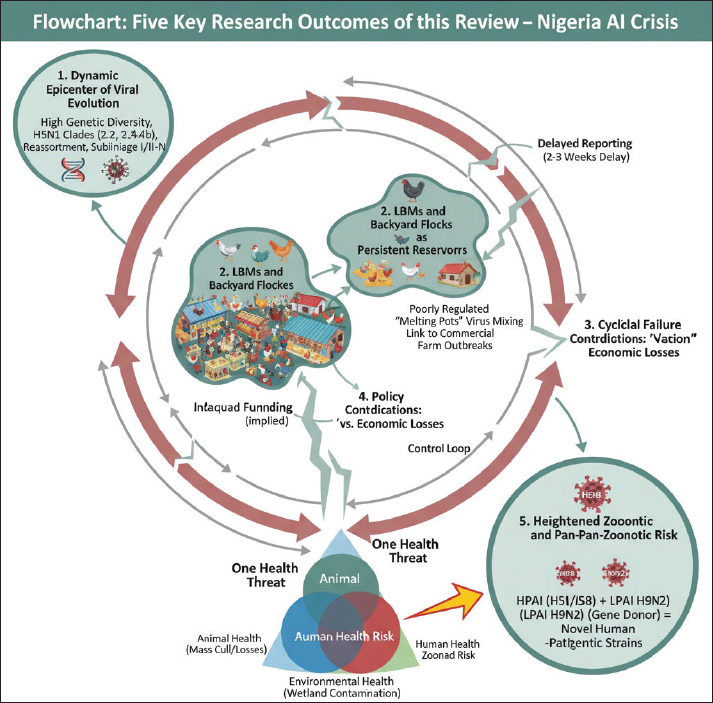
Fig. 4. Map of the location of the detected A/H9N2 virus in commercial poultry in Ido LGA Ibadan, Oyo State, Nigeria. Adapted from Akanbi et al. (2024). Geographical distribution and temporal patternsThe initial 2006 HPAI outbreak in Nigeria showed a distinct geographical distribution, with a high percentage of cases reported in Plateau State (29.7%), followed by Kaduna (12.4%), Kano (11.7%), and Bauchi (11.7%). The virus rapidly spread from the North-Western part of the country to the central regions within weeks (Adegboye and Kotze, 2013). Phylogenetic analysis of viral isolates revealed widespread viral gene flow across Nigeria; however, Bayesian MCMC analysis indicated clear clustering by geographic region, confirming localized in situ evolution of the virus. A parsimony-based migration analysis identified specific "source" and "sink" regions for viral spread (Fusaro et al., 2010). Viral migration predominantly occurred from north-central states (e.g., Katsina, Jigawa, Kano, Yobe, Kaduna, Bauchi, and Gombe) and southwestern states (e.g., Kwara, Ekiti, Osun, Lagos, Ogun, and Oyo) to northeastern, southeastern, and south-central regions. This suggests that the north-central and south-western regions serve as critical points for virus dissemination, while other regions act as ecological sinks where the virus might establish localized populations (Fusaro et al., 2010). The identified source regions align with areas of high poultry farm density (southwestern region) and areas with frequent contact between wild birds and backyard poultry (north-central region, particularly the Hadejia-Nguru wetlands). A notable temporal pattern in HPAI introductions into Nigeria appears to be linked to the migration of wild waterfowls from Asia and Europe, which typically occurs between September and January each year. This seasonal influx of migratory birds often precedes the detection of AI cases in Nigeria, indicating a recurrent pathway for viral introduction. Furthermore, preliminary epidemiological analysis indicated an increase in H5N1 incidence at farms located at high altitudes, and the climatic conditions prevalent in the North Central region were found to favour the virus's survival and transmission (Adegboye and Kotze, 2013). Role of domestic poultry (commercial and backyard flocks)Nigeria possesses a substantial poultry population, estimated at around 140 million birds (FAO, 2008), with approximately 60% of this production occurring in small backyard flocks. Both commercial and backyard poultry systems have been significantly impacted by AI. During the 2006–2008 epidemics, an estimated 939,620 commercial chickens and 14,512 local chickens from backyard farms were lost to the disease (Akanbi and Taiwo, 2014a; Akanbi and Taiwo, 2014a,b). In terms of outbreak frequency, backyard flocks accounted for a larger proportion of cases (77.1%) compared to commercial poultry flocks (22.9%) (Akanbi et al., 2016a). Mortality rates varied among different poultry species and farm types. Commercial flocks generally experienced higher absolute mortality rates due to their larger flock sizes. However, backyard local chickens recorded the highest number of deaths relative to their total flock size (89.4%), and ducks within these settings showed the highest mortality rate at 87.1% (Akanbi and Taiwo, 2014b; Akanbi et al., 2016a). The management of backyard poultry farming has been identified as a significant challenge to epidemic control. This is often due to lower levels of knowledge and awareness regarding AI and its risks, coupled with suboptimal hygiene practices among backyard poultry keepers. The extensive close human contact with free-ranging poultry in village households further amplifies the risk of human exposure and infection (Fasina et al., 2009). The differential vulnerability and risk associated with various poultry systems warrant tailored control strategies. While commercial farms face substantial economic losses from high mortality in large flocks, backyard and small-scale operations present unique epidemiological challenges (Akanbi et al., 2016a). The high proportion of backyard flocks in Nigeria's total poultry population and their significant involvement in outbreaks suggest that these systems can act as persistent reservoirs and amplifiers of the virus (Akanbi et al., 2016a). This is particularly concerning given the extensive human-animal interface and often lowers biosecurity standards prevalent in these settings (Alhaji et al., 2023). The observation that free-range flocks might exhibit a lower risk due to constant exposure to low pathogenic AI (LPAI) strains, potentially leading to some level of flock immunity, presents a complex dynamic. This indicates that a nuanced understanding of immunity, exposure, and management practices across different poultry systems is necessary for developing effective and targeted interventions that address the specific epidemiological roles and vulnerabilities of each sector. Role of wild birds and other avian speciesWild birds, particularly waterfowl belonging to the orders Anseriformes and Charadriiformes, are recognized as the natural reservoir for all AI viruses (AIV) subtypes and play a fundamental role in the global spread and maintenance of these viruses (Webster et al., 1992). Migratory birds are particularly significant contributors to the wide geographic dissemination of HPAI H5 viruses. Nigeria's geographical location along major migratory routes for wild birds from Asia and Europe renders it highly vulnerable to recurrent introductions of AIVs. The presence of HPAI in Northern Nigeria is plausibly linked to wild bird sanctuaries in the region, serving as potential points of entry for new strains. Evidence from Nigeria indicates the circulation of HPAI in apparently healthy waterfowls, highlighting their importance in the maintenance and transmission of the virus within the country (Akanbi et al., 2016a). Waterfowls are generally less susceptible to HPAI infection compared to chickens, allowing them to shed the virus as asymptomatic carriers in backyard farms and LBMs. Beyond migratory wild birds, exotic and zoological birds also contribute to the AI epidemiology in Nigeria. HPAI H5N1 and H5N8 strains have been detected in imported aquatic birds (such as geese and ducks) and exotic birds (like the yellow golden pheasant) in Nigeria (Atuman et al., 2025). The introduction of captured wild birds into parks has also been linked to H5N1-related deaths in domestic birds. The illegal trade and inadequate surveillance of these exotic birds are considered significant factors contributing to the ecology and epidemiology of AIVs within the country (Atuman et al., 2025). The expanding spectrum of avian reservoirs and transmission pathways for AI in Nigeria is a critical observation. The detection of HPAI in exotic and zoological birds, including imported aquatic species, broadens the traditional focus on migratory wild birds and domestic poultry. This indicates that the concept of "bridge species" is more complex than previously assumed, encompassing a wider range of avian hosts and transmission routes, including illegal trade and introduction into captive environments (Columba Teru et al., 2012). This necessitates a more comprehensive surveillance approach that extends beyond conventional poultry farms and established wild bird flyways to include LBMs, zoos, and points of entry for exotic birds. A holistic understanding of these diverse reservoirs and pathways is essential for developing effective prevention and control strategies (Columba Teru et al., 2012). Transmission factors and risk pathwaysNigeria's key transmission multipliers—the "X" factors—can be theoretically categorized using the spatial definitions of density (Lakes et al., 2025). The unregulated nature of LBMs, described as viral melting pots, fits the concept of "density as numbers gathering at sites" , representing high-frequency interaction points crucial for viral mixing and rapid dissemination to commercial farms. Conversely, the significant involvement of backyard flocks, which account for 60% of Nigeria's poultry population and involve extensive human-animal contact, aligns with "density as numbers living in a house" (often termed crowding). This classification clarifies the heightened transmission risk within socio-economically vulnerable communities and explains why containment measures designed for industrial farms often fail in these unstructured, high-density settings (Lakes et al., 2025). Several key factors and pathways contribute to the transmission and spread of AI in Nigeria. At the farm level, identified risk factors for HPAI A/H5N1 infection include the frequent reception of visitors (egg buyers, debeakers, etc.) on farm premises, the purchase of live poultry or poultry products, farm workers residing outside the farm premises (Fasina et al., 2011), and introduction of poultry from LBMs (Akanbi and Taiwo, 2015). These factors facilitate the introduction and movement of the virus into and between farms. Broader systemic factors also play a significant role. The spread of H5N1 within Nigeria and across Africa has been attributed to both international and inter-state poultry trade, as well as the regional influx of wild birds (Fasina et al., 2011). Within farm settings, the clustering of poultry pens in farm settlements has been shown to contribute significantly to the rapid dissemination of the disease once an outbreak occurs. LBMs are particularly implicated, described as "proverbial melting pots" that play a vital role in the dissemination of the AI virus to commercial farms and serve as hotspots for the introduction and spread of emerging strains. The poorly regulated poultry value chain and unstructured nature of many LBMs were significant contributors to the 2015 resurgence of HPAI (Akanbi et al., 2016a; Sulaiman et al., 2021). Environmental and ecological factors also influence transmission intensity. Studies indicate that higher wind speed can increase the intensity of virus transmission, while higher temperatures may decrease it (Adegboye and Kotze, 2013). Proximity to the initial outbreak site also correlates with increased transmission intensity. Furthermore, the presence of nearby water bodies, which facilitate contact between domestic poultry and wild birds, significantly influences the resurgence of the virus (Alhaji et al., 2023). From a public health perspective, zoonotic risk pathways include the consumption of undercooked poultry meat and products, as well as direct contact with infected birds and contaminated fomites (Alhaji et al., 2023). Poor biosecurity practices are a pervasive issue, encompassing inadequate cleaning and disinfection, insufficient control over the movement of birds, people, and vehicles, all of which significantly contribute to viral resurgence (Ameji et al., 2019). An assessment of HPAI resurgence by Alhaji et al. (2023) revealed a complex, interconnected web of transmission drivers for AI in Nigeria and highlights HPAI transmission as the deep interconnection with socio-economic practices, environmental conditions, and regulatory frameworks. The identified risk factors range from direct farm-level biosecurity breaches, such as uncontrolled visitor access and the introduction of purchased birds or products, to broader systemic issues like unregulated poultry trade and the role of LBMs as viral dissemination hubs. This interconnectedness implies that effective control of AI in Nigeria requires a multi-layered approach that transcends simple farm-level interventions. It necessitates systemic changes in poultry trade regulations, the restructuring of market environments, and comprehensive public awareness campaigns to address behavioural and cultural factors that contribute to viral spread. Persistent viral evolution and migrating wild birds that contaminate the environment drive the transmission pathway of AI in Nigeria. The virus then spreads to backyard flocks and moves through LBMs—which act as interconnected hubs for mixing and dissemination—and other contact sources before entering commercial poultry flocks. Due to control Loop gaps in the stamping-out/biosecurity/surveillance measures, delayed reporting, inadequate funding, and policy paradoxes (e.g., the conflict over vaccination). This systemic breakdown ensures the continuous cycle, leading to a One Health threat involving overlapping risks to Animal Health (mass cull/losses), Human Health (zoonotic risk), and Environmental Health (wetland and environment contamination). This is shown below in Figure 5.
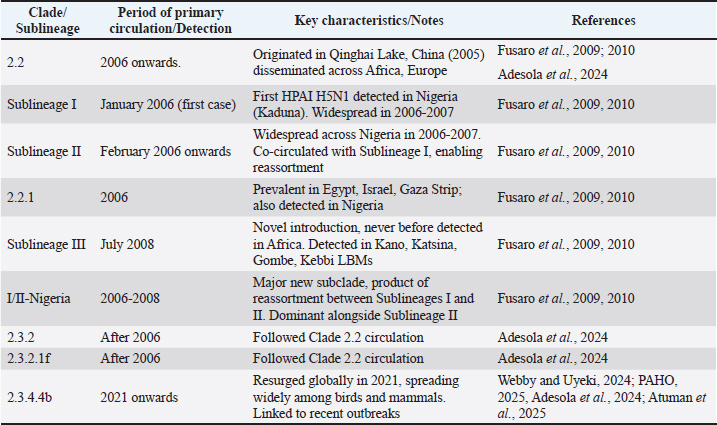
Fig. 5. Persistent viral evolution, transmission factors and policy inadequacy drive a cyclical crisis. Molecular epidemiology and viral evolutionGenetic characterization of H5N1 clades and sublineagesNigeria has experienced multiple introductions of HPAI A/H5N1 viruses, resulting in a highly complex and dynamic genetic landscape. The A/H5N1 lineage circulating across Africa, Europe, and Asia is primarily identified as clade 2.2, which originated from a significant wild bird outbreak at Qinghai Lake in western China in 2005 (Fusaro et al., 2009). Early outbreaks in Nigeria, particularly between 2006 and 2007, were characterized by the co-circulation of three distinct sublineages: I, II, and IV (Fusaro et al., 2009). Sublineages I and II were widely distributed across the country, creating conditions conducive to reassortment events, with the first such event identified as early as June 2006. A notable development occurred in July 2008 with the introduction of a novel sublineage, sublineage III, which had not been previously detected in Africa (Fusaro et al., 2009). This makes Nigeria unique as the only African country where clade 2.2.1 and three different sublineages (I, II, and III) of clade 2.2 have all been identified (Fusaro et al., 2010). Phylogenetic analysis of A/H5N1 strains spanning 2006–2021 reveals a clear evolutionary progression of clades. A/H5N1 Clade 2.2 was predominant in 2006, subsequently giving way to the emergence of Clades 2.3.2, 2.3.2.1f, and most recently, 2.3.4.4b in 2021 (Adesola et al., 2024). The clade 2.3.4.4b, in particular, experienced a global resurgence in 2021, causing widespread outbreaks in various avian and mammalian species (Webby and Uyeki, 2024). In addition, a major new sub-clade, designated sublineage I/II-Nigeria ("I/II-N"), was identified [Fusaro et al., 2010; Pan American Health Organization (PAHO), 2025]. This sub-clade is phylogenetically distinct and appears to have originated from a significant reassortment event between clades related to sublineages I and II (Fusaro et al., 2010). This novel sublineage, along with sublineage II, constituted the majority of A/H5N1 isolates collected in Nigeria between 2006 and 2008 (Fusaro et al., 2010). The presence of multiple A/H5N1 clades and sublineages (I, II, III, IV, 2.2.1, 2.3.2, 2.3.2.1f, 2.3.4.4b) and the identification of novel reassortment events, including a unique Nigerian subclade (I/II-N), strongly indicate that Nigeria is not merely a passive recipient of introduced viruses (Adesola et al., 2024). Instead, it functions as a dynamic environment for viral evolution (Fusaro et al., 2010). This high genetic diversity and frequent reassortment potential are likely driven by the co-circulation of multiple strains within dense poultry populations and LBMs (Fusaro et al., 2010). This complex evolutionary landscape poses a significant challenge for the development of effective vaccines and the implementation of sustained control measures, as newly emerging variants may exhibit altered pathogenicity or transmissibility, potentially rendering existing interventions less effective. Circulation of other AI strains (e.g., H5N2, H5N6, H5N8, and H9N2)Beyond the dominant A/H5N1 strain, Nigeria has experienced the circulation of other HPAI and LPAI subtypes. These strains were detected during active and passive surveillance with the use of the Reverse transcriptase polymerase reaction. Since the initial outbreak in 2006, a total of five subtypes, including H5N1, H5N2, H5N6, H5N8, and H9N2, have been identified in the country (Ameji et al., 2019; Akanbi et al., 2024; Atuman et al., 2025). H5N2This LPAI subtype was reported in 2014 from a pool of ducks at an LBM in Ibadan, Oyo State (Coker et al., 2014). Further evidence of its circulation comes from the detection of antibodies to the H5 subtype (likely H5N2) in dogs (2.2% prevalence) and chickens (6.7% prevalence) in Kaduna metropolis, with a higher prevalence observed in railway station LBMs (13%) (Assam and Shehu, 2023). H5N6This HPAI subtype has also been detected in LBMs across Nigeria in recent years (Shittu et al., 2020). H5N8HPAI H5N8 emerged and co-circulated with H5N1 in the Nigerian poultry sector in Kano in 2016. A distinct outbreak of HPAI H5N8 was confirmed in two multi-age chicken flocks in Jos, Plateau State, in February 2019, leading to significant mortality (Ameji et al., 2019). H5N8 has also been detected in imported aquatic birds (geese and ducks) and exotic birds (yellow golden pheasant) in Nigeria, with links to trade from Libya (Atuman et al., 2025). Globally, H5N8 has caused widespread outbreaks and is recognized for its zoonotic potential, maintaining a sustained prevalence in Africa. H9N2Although not widely reported in Nigeria prior to 2019, surveillance efforts in that year revealed the presence of H9N2 in LBMs, with 19 viruses detected from apparently healthy poultry (Sulaiman et al., 2021). These strains belong to the G1 lineage, which is known for its zoonotic potential and clusters genetically with contemporary African H9N2 isolates. In 2024, the first reported H9N2 outbreak in commercial poultry in Southern Nigeria exhibited high pathogenicity (Akanbi et al., 2024). LPAI H9N2 appears to be endemic in Nigeria as it was also detected in commercial poultry in Northern states of Kaduna and Kano in 2024 (Mkpuma et al., 2024), and occasionally leads to morbidity and mortality when co-occurring with other infections or under unfavourable husbandry conditions. Globally, H9N2 is the most widespread AIV subtype in poultry, frequently co-circulating with HPAIVs and notably contributing internal gene segments to other AIVs that have caused fatal human infections (Li et al., 2020). Reassortment events and genetic diversityThe co-circulation of multiple distinct H5N1 sublineages (I, II, and IV) within Nigeria created an environment conducive to frequent genetic reassortment events, with the first documented reassortant virus identified as early as June 2006 (Fusaro et al., 2009). Additional reassortant viruses were subsequently identified in early 2007. A significant example of this genetic mixing is the novel sublineage I/II-N, which itself is a product of a major reassortment event between sublineages I and II (Fusaro et al., 2010). Specific reassortment events, such as R2 and R4, were identified in isolates from 2006 to 2007, further illustrating the dynamic genetic exchange occurring within the viral population. The isolation of a reassortant H5N8 strain in 2017 fuelled concerns about the potential emergence of novel influenza strains with altered characteristics. The ability of the HPAI virus to evolve into other subtypes, such as the emergence of A/H5N8 from A/H5N1 lineages, contributes significantly to its persistence and the on-going challenge of disease control (Table 2). Table 2. Key H5N1 clades/ sublineages and their periods of circulation in Nigeria.
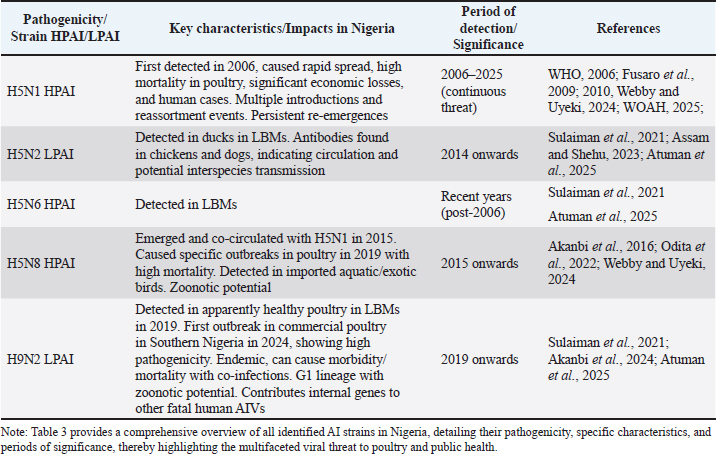
Implications of viral evolution for disease controlThe continuous evolution, clade switching, and frequent reassortment events observed in Nigerian AIVs underscore a critical challenge for disease control efforts. This genetic dynamism necessitates continuous genomics surveillance to accurately track antigenic variation (Adesola et al., 2024). The inherent viral plasticity creates an on-going "arms race" where control measures must constantly adapt to the evolving pathogen. For instance, a vaccine developed against an older viral clade may become less effective against newly circulating or reassorted strains due to antigenic drift (Nagy et al., 2017). Furthermore, the co-circulation of multiple subtypes, including LPAI strains like H9N2, complicates surveillance and intervention strategies (Sulaiman et al., 2021). H9N2 can act as a "silent" carrier or a donor of internal genes for more pathogenic viruses, potentially masking the high mortality rates typically associated with HPAIVs and thus hindering early detection and rapid response (Peakcock et al., 2019) (Table 3). The detection of specific virulent amino acid changes, such as lysine at PB2 position 627, in Nigerian isolates further highlights the potential for increased viral virulence (Nagy et al., 2017). This complex evolutionary landscape demands highly sophisticated surveillance systems that incorporate real-time genomic sequencing and antigenic characterization (Fusaro et al., 2009). Such advanced capabilities are crucial for informing adaptive vaccine strategies and for continuously refining biosecurity protocols to effectively counter the evolving viral threat. Table 3. Summary of AI strains (H5N1, H5N2, H5N6, H5N8, H9N2) and their impact in Nigeria.
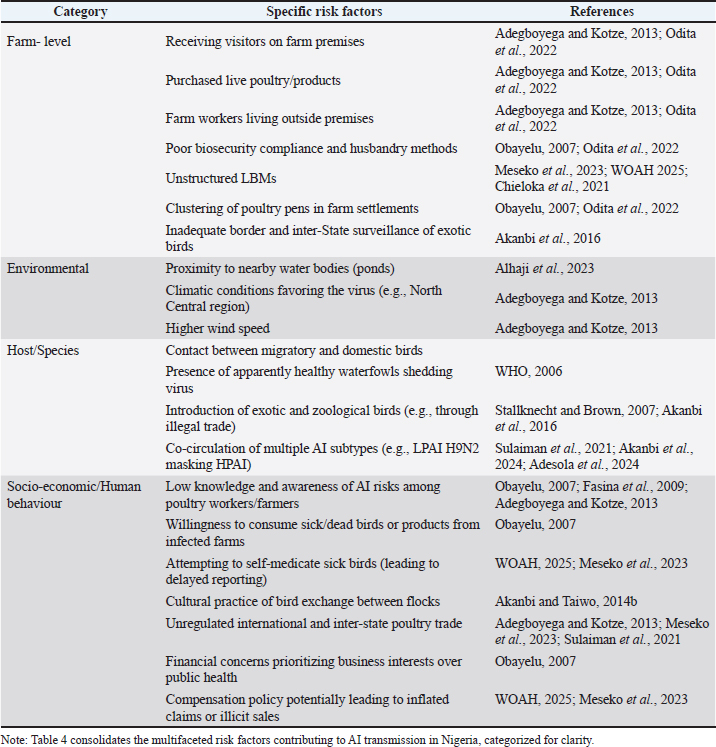
Control and prevention strategiesBiosecurity measures and implementation challengesImproving farm hygiene and biosecurity is consistently recommended as a primary measure to reduce the risk of AI infection (Fasina et al., 2011). Indeed, strict biosecurity implementation has been demonstrated to significantly reduce viral transmission in both backyard and commercial farm settings (Akanbi and Taiwo, 2014a; Akanbi et al., 2024). Key biosecurity practices include the separation of sick birds from healthy ones, frequent cleaning and disinfection of equipment and premises, strict control over the movement of birds, consistent use of personal protective equipment by workers, and regulated movement of persons and vehicles onto and off farm sites. Restricting visitors to poultry farms is also considered a crucial preventive measure (Table 4). Table 4. Identified risk factors for AI transmission in Nigeria.
Despite these clear recommendations and demonstrated effectiveness, the implementation of biosecurity measures in Nigeria faces significant challenges (Alhaji et al., 2023). Poor biosecurity compliance and suboptimal husbandry methods among stakeholders have been identified as contributing factors to the persistence of HPAI in the country (Akanbi and Taiwo, 2014a; Odita et al., 2022). The resurgence of H5N1 during 2015–2017 was directly attributed to a decline in surveillance coupled with inadequate biosecurity measures (Alhaji and Odetokun, 2011; Ameji et al., 2019). The challenges in implementing biosecurity are often nuanced and vary by farm type (WAOH, 2025). A significant challenge lies in the gap between biosecurity knowledge and actual practice (Obayelu, 2007). While poultry workers have reported knowing some risk factors for AI (61%), a smaller proportion (56%) could correctly describe these factors, and a considerable percentage exhibited poor food safety knowledge or a willingness to consume sick birds (Obayelu, 2007). This discrepancy between awareness and consistent application of biosecurity principles is critical. The continued occurrence of outbreaks despite recommendations suggests that knowledge transfer alone is insufficient (Obayelu, 2007). Socio-economic factors, such as poverty, limited access to resources, prioritizing financial preservation over public health risks, and deeply ingrained cultural practices like bird exchange, likely act as substantial barriers to effective biosecurity implementation, particularly in informal backyard settings (Adegboyega and Kotze, 2013). Addressing these underlying socio-economic and cultural determinants is essential for improving biosecurity adherence (Obayelu, 2007; Alhaji et al., 2023). Depopulation, disinfection, and quarantine protocolsNigeria's "modified AI Control policy," implemented since the initial outbreaks in 2006, primarily relies on a set of core measures including stamping-out of infected birds, thorough disinfection and decontamination of farms and equipment, and compensation for affected farmers (Chieloka et al., 2021). Depopulation procedures are carried out in accordance with international guidelines, such as EU guideline 93/119/EC. In practice, humane slaughter methods like decapitation and dislocation of the neck are preferred in states like Enugu. Following depopulation, affected carcasses and non-biodegradable materials are buried in deep pits (at least 6 feet deep) and covered with layers of calcium hydroxide and earth (Chieloka et al., 2021). Disinfection protocols are rigorous, involving the physical removal of disease-causing agents and contaminated organic/inorganic material from farm equipment and tools to ensure proper penetration of disinfectants. Cleaning methods include dry cleaning (scraping, sweeping) and wet cleaning using detergents and pressure washers. All production units, including hatcheries, egg storage rooms, packaging areas, and transportation vehicles, undergo disinfection. Walls, floors, ceilings, metal cages, and structures are disinfected, with hot water treatment for metal components. Drinkers, food hoppers, water reservoirs, and silos are also thoroughly cleaned and disinfected (Chieloka et al., 2021). Quarantine measures are initiated upon suspicion of an outbreak. These include placing notices at farm entrances to prohibit the movement of poultry and poultry products, collecting human samples (nasal swabs) from exposed farm workers, and establishing defined control zones around outbreak foci. A 3 km Protection Zone is established around the outbreak, extending to a buffer zone, and a 10 km Surveillance Zone (SZ) is set up for contact tracing in surrounding areas (Chieloka et al., 2021). While depopulation and disinfection are effective in containing immediate outbreaks, their implementation in Nigeria has faced significant challenges and unintended consequences. A major issue is the delay in early disease reporting by farmers, often ranging from 2 to 3 weeks post-suspicion. This delay is frequently attributed to farmers attempting to self-medicate their flocks or to avoid the financial implications of culling (Chieloka et al., 2021). Coupled with subsequent delays in the veterinary authority’s response and the activation of control measures (3–4 days), the overall effective control period for an outbreak in some states can extend to 3–5 weeks, which often exceeds the virus's maximum incubation period of 3 weeks (21 days). This extended delay allows for further viral spread to contiguous farms and LBMs. Furthermore, the compensation policy, intended to incentivize reporting, has been observed to create perverse incentives (Chieloka et al., 2021). Farmers have reportedly attempted to inflate poultry flock sizes to claim higher compensation, and there have been instances of infected poultry being sold to unsuspecting buyers before official culling (Atuman et al., 2025). These trades-offs and unintended consequences highlight a critical policy paradox: a well-intentioned measure designed to encourage compliance can, in practice, undermine the very goal of disease control by fostering delays and illicit practices. This complex interplay necessitates a re-evaluation of policy design to ensure that incentives align with public health and animal health objectives (Chieloka et al., 2021). Surveillance systems: design, effectiveness, and gapsSince the initial HPAI A/H5N1 incursion in 2006, Nigeria has made efforts to enhance its surveillance and laboratory capacities for influenza diagnostics (Assam and Shehu, 2023). LBMs are recognized as primary targets for surveillance activities due to their role as "melting pots" where diverse poultry species from various sources converge, making them critical points for virus mixing and dissemination (Sulaiman et al., 2021). The surveillance system is designed to involve detection and immediate notification of suspected outbreaks by AI desk officers at the local government area (LGA) level. Samples, typically whole carcasses, are collected from affected poultry for confirmatory diagnosis at the National Veterinary Research Institute (NVRI) in Jos (Fusaro et al., 2009). Upon laboratory confirmation of AI, control zones are established, including a 3-kilometer control zone around the infected farm and a 10-kilometer SZ for contact tracing. The World Health Organization and the Nigeria Centre for Disease Control have supported the establishment of 10 National Influenza Sentinel Surveillance sites across the country, including major teaching hospitals (Chieloka et al., 2021). An evaluation of the AI surveillance system in Enugu State between 2015 and 2017 found it to be useful, simple, flexible, and acceptable to stakeholders. However, this evaluation also identified significant operational gaps. These included poor data quality, instability of the surveillance system, and persistent delays in reporting, with timeliness of reports declining from 51% in 2015 to 47% in 2017. Additionally, the data collected were often not representative of the broader epidemiological situation (Chieloka et al., 2021). The decline in surveillance measures has been directly implicated in the increased transmission and severity of the HPAI A/H5N1 resurgence observed from 2015 to 2017 (Oladokun et al., 2012; Tassoni et al., 2016). This indicates that despite initial enhancements and a functional design, the Nigerian surveillance system exhibits critical fragilities, particularly in its sustained operational effectiveness. The system's reliance on external funding (donor-driven) may have contributed to its instability to function when financial support fluctuates (Oladokun et al., 2012; Tassoni et al., 2016). To build a resilient surveillance system, it must be integrated into routine veterinary public health programs with sustainable domestic funding. Continuous monitoring of LBMs and molecular characterization of AIVs are strongly advocated to mitigate economic losses and public health threats, especially given the co-circulation of multiple viral subtypes (Oladokun et al., 2012). Active seasonal and sentinel surveillance in high-risk zones, such as wetlands and LBMs, is recommended for early detection and rapid response, among other solutions, as shown in Figure 6. In addition, leveraging technological advances like mathematical modelling and artificial intelligence (AI) is essential for improving forecasting, hotspot detection, and overall response strategies.
Fig. 6. Flow chart of the Infection cycle of AI in Nigeria. Vaccination policies and considerations in NigeriaSince 2006, HPAI H5N1 subtypes have significantly impacted poultry production in Nigeria (Akanbi et al., 2016a), leading to growing concerns about the sole reliance on biosecurity and calls for the adoption of vaccination as an additional control measure. However, the Federal Government of Nigeria has, as of April 2023, maintained a "no vaccination" policy for HPAI (FMARD, 2023 ). The rationale provided for this policy includes the desire to save costs associated with vaccine imports and to demonstrate that Nigeria's surveillance system is robust and capable of containing the disease through stringent non-vaccination measures. Experts have also expressed concerns that abandoning the "no vaccination" policy could lead to difficulties in disease control, an influx of substandard vaccines, issues with virus mutation, and the emergence of resistant strains. Despite this official stance, the NVRI has recently recommended "consideration of HPAI vaccination options," indicating a potential shift in perspective or an acknowledgment of the evolving challenges. A rational approach to vaccination in Nigeria necessitates careful consideration of several factors. The adoption of vaccination is often viewed as an admission of an endemic situation, which could impact Nigeria's access to international poultry markets due to World Organisation for Animal Health (WOAH) trade regulations. Should vaccination be implemented, a controlled approach would be essential. This would involve rigorous monitoring of vaccine application, establishing the capacity to differentiate vaccinated from infected (DIVA) flocks, and assessing sero-conversion and other immune correlates of protection. Furthermore, continuous surveillance for circulating AIV strains and analysis of any antigenic mismatch with vaccine strains would be critical to ensure vaccine effectiveness. For countries with substantial commercial poultry investments, like Nigeria, zoning territories for targeted "ring vaccination" in high-risk areas could be a viable strategy. Nigeria's "no vaccination" policy reflects a strategic decision to prioritize perceived trade advantages and confidence in non-vaccine control measures. However, the persistent outbreaks and significant economic losses sustained under this policy, coupled with the NVRI's recent call to reconsider vaccination, highlight a growing tension between policy and epidemiological reality. The central dilemma is whether the substantial economic and public health costs associated with recurrent outbreaks outweigh the perceived benefits of maintaining a "vaccine-free" status for international trade. A shift towards vaccination would require considerable investment in DIVA capabilities, robust surveillance to detect antigenic drift, and careful diplomatic engagement to navigate international trade implications. This represents a complex, multi-faceted policy challenge that demands a comprehensive risk-benefit analysis based on the latest scientific evidence and a forward-looking strategy for disease management. The "One Health" approach in AI managementThe resurgence and persistent circulation of HPAI in Nigeria are increasingly recognized as a significant threat to the achievement of the "One Health" concept, given the global scale of viral spread at the poultry-human-environment interface (Alhaji et al., 2023). The "One Health" framework provides a holistic model for assessing the intricate inter-links among biophysical, environmental, and sociocultural activities that drive the emergence and resurgence of diseases like AI (Alhaji et al., 2023). Effective mitigation of AI necessitates multidisciplinary and multisectoral collaborations spanning human, animal, and shared environmental domains (Alhaji et al., 2023). The WOAH strongly advocates for a One Health approach to AI management, emphasizing the importance of continued surveillance in both wild and domestic species, stringent biosecurity measures at the farm level, and timely reporting of all AI outbreaks. Furthermore, the integration of technological advances, such as mathematical modelling and AI, into a One Health framework is considered essential for improving surveillance, forecasting, and response strategies to mitigate the impacts of the on-going AI outbreaks. These tools can enhance the ability to predict disease spread, identify hotspots, and provide early warning systems. Despite the recognized importance of the One Health approach, there remains a notable dearth of research evidence specifically on the implementation of One Health interventions against HPAI emergence and re-emergence in sub-Saharan Africa (Alhaji et al., 2023). This research gap hinders the ability to effectively evaluate the progress of control and prevention efforts in developing economies like Nigeria. Addressing this gap is crucial for developing and implementing integrated, sustainable strategies that account for the complex interactions at the human-animal-environment interface (Alhaji et al., 2023). This research gap is further compounded by a general lack of awareness and integration of the One Health concept among essential medical professionals, as was revealed in a study in Brazil (Rosa Meurer et al., 2024), which found that 100% of surveyed physicians were unaware of the One Health approach. Conclusions and recommendationsAI, particularly HPAI H5N1, has posed a persistent and evolving threat to Nigeria's poultry industry and public health since its initial incursion in 2006. The period from 2006 to 2025 has been characterized by recurrent outbreaks, which have been driven by multiple viral introductions from diverse clades (e.g., 2.2, 2.3.2, 2.3.4.4b) and frequent reassortment events, thereby establishing Nigeria as a dynamic epicentre for viral evolution. The co-circulation of other strains, including H5N2, H5N6, H5N8, and the increasingly significant H9N2, further complicates the epidemiological landscape. Transmission is facilitated by a complex interplay of factors, including inadequate farm-level biosecurity, the central role of unregulated LBMs, environmental conditions, and socio-economic behaviours that often prioritize immediate financial concerns over long-term disease control. While initial control measures, such as stamping-out and disinfection, achieved temporary suppression, their long-term effectiveness has been hampered by delayed reporting, challenges in consistent biosecurity implementation, and unintended consequences of compensation policies. Despite enhancements, the surveillance system still shows weaknesses in data quality and timeliness. The continuous outbreaks and significant economic losses are placing increasing pressure on Nigeria's "no vaccination" policy, which aims to maintain trade status. This review’s finding that control measures are hampered by "policy paradoxes" is reinforced by critical spatial analysis, which highlights the difficulty in enforcing policies in complex urban settings. Just as COVID-19 studies warned, policies relying on spatially aggregated data risk overlooking the dynamic nature of human (and poultry) mobility and interactions (Lakes et al., 2025). The endemic challenge of delayed reporting driven by compensation issues is a consequence of failing to align financial incentives with localized spatial behaviour. Also, researchers and policymakers must heed the warning against making political statements regarding "infectious spaces" or neighbourhoods. To move forward, policies aimed at reforming LBMs or increasing biosecurity must distinguish carefully between statistical associations and causal inferences to avoid stigmatization and to develop interventions that genuinely integrate animal, human, and environmental realities within a robust One Health framework. To effectively mitigate the on-going threat of AI in Nigeria, the following recommendations are put forth: Strengthen and sustain surveillance systemsIt is important and crucial to implement robust, continuous, and integrated surveillance programs that extends beyond commercial farms to actively monitor backyard flocks, LBMs, and wild bird populations, particularly along migratory routes and in wetlands. This requires consistent funding, improved data quality, and timely reporting mechanisms. Leveraging advanced technologies, like genomic sequencing, mathematical modeling, and AI, for real-time risk assessment, forecasting, and hotspot detection is crucial. Enhance and tailor biosecurity measuresIt is crucial to develop and enforce context-specific biosecurity protocols for both commercial and backyard poultry systems, taking into account their unique operational realities and vulnerabilities. This includes promoting comprehensive training and education for farmers, addressing socio-economic barriers to compliance, and fostering community-led sanitary measures in free-range settings. Strict regulation of poultry movement and trade, particularly within and from LBMs, is paramount. Re-evaluate vaccination policyConduct a comprehensive, science-based risk-benefit analysis of HPAI vaccination, considering the persistent economic losses from recurrent outbreaks versus potential trade implications. If vaccination is adopted, it should be part of a controlled strategy that includes robust DIVA capabilities, continuous monitoring of circulating strains for antigenic match, and potential zoning for targeted application in high-risk areas. Adopt a comprehensive "One Health" approachFully integrate human, animal, and environmental health sectors in AI prevention and control efforts. This requires sustained multidisciplinary and multi-sectoral collaboration, joint research initiatives, and coordinated policy development to address the complex drivers of disease emergence and spread at the interface. Investment in research on the effectiveness of One Health interventions in the Nigerian context is vital. Review and reform compensation policiesRe-evaluate the current compensation framework to ensure it effectively incentivizes early reporting and compliance with culling, without creating perverse incentives for illicit practices or inflated claims. Alternative financial support mechanisms or insurance schemes could be explored to alleviate farmers' losses and encourage adherence to control measures. By implementing these integrated and adaptive strategies, Nigeria can move towards a more resilient and effective AI control program, safeguarding its poultry industry, ensuring food security, and protecting public health from this evolving zoonotic threat. This review shifts the focus from treating Nigerian outbreaks as isolated episodes (e.g., 2006) to recognizing a chronic, cyclical pattern. The findings show that disease resurgence is fundamentally linked to a relaxation of surveillance and biosecurity protocols during the periods between major epidemics, allowing the virus to re-establish and re-amplify. The analysis goes beyond the dominant H5N1 strain to detail the simultaneous presence of other subtypes, including the emerging H5N8 and the endemic LPAI H9N2. This co-circulation increases the potential for genetic reassortment, positioning Nigeria as an active environment for the evolution of new, potentially more virulent or pandemic strains. The review scrutinizes control measures, revealing a significant policy paradox. Specifically, the compensation policy, designed to encourage reporting, often leads farmers to delay notification or engage in illicit sales of infected birds to inflate compensation claims. This behavioural response undermines the entire culling strategy and directly contributes to viral dissemination. Instead of attributing outbreak persistence merely to inadequate biosecurity, this manuscript identifies the deeper socio-economic drivers. These drivers, such as farmer poverty, the cultural practice of bird exchange between flocks, and the financial imperative to consume or sell sick birds, act as profound barriers that prevent the consistent application of control measures. Conflict of interestThe authors declare that there is no conflict of interest that could influence or bias this work. FundingOlatunde Babatunde Akanbi received the DAAD sponsorship to visit Germany for this work. Authors contributionsThe authors conceptualized, drafted the scope, and agreed on the content of this review. ReferencesAdegboye, O.A. and Kotze, D. 2013. Epidemiological analysis of spatially misaligned data: a case of highly pathogenic avian influenza virus outbreak in Nigeria. Epidemiol. Infect. 142(5), 940–949. https://doi.org/10.1017/S0950268813002136 Adene, D.F., Wakawa, A.M. and Abdu, P.A. 2006. Clinico-pathological and husbandry features associated with the maiden diagnosis of avian influenza in Nigeria. Nig. Vet. J. 27(1), 32–38. Adesola, R.O., Onoja, B.A., Adamu, A.M., Agbaje, S.T., Abdulazeez, M.D., Akinsulie, O.C., Bakre, A. and Adegboye, O.A. 2024. Molecular epidemiology and genetic evolution of avian influenza H5N1 subtype in Nigeria, 2006 to 2021. Virus Genes 60(5), 501–509; doi:10.1007/s11262-024-02080-9 Akanbi, B.O. 2014. Characterization and comparative susceptibility of highly pathogenic avian influenza H5N1 virus infections in chickens and ducks. Ph.D. thesis, University of Ibadan, Veterinary Pathology Department, Ibadan, Nigeria. Akanbi, B.O., Meseko, C.A., Odita, C.I., Shittu, I., Rimfa, A.G., Ugbe, D., Pam, L., Gado, D.A., Olawuyi, K.A., Mohammed, S.B., Kyauta, I.I., Bankole, N.O., Ndahi, M., Joannis, T.M., Ahmed, M.S., Shamaki, D. and Okewole, P.A. 2016. Epidemiology and clinicopathological manifestation of resurgent highly pathogenic avian influenza (H5N1) virus in Nigeria. Nig. Vet. J. 37(3), 175–186; https://www.ajol.info/index.php/nvj/article/view/147399 Akanbi, B.O., Fereidouni, S., Taiwo, V.O., Starick, E., Okewole, P.A., Binder, A. Heenemann, K. and Teifke, J.P. 2017. Formalin fixed and paraffin embedded tissues of chickens are useful for retrospective studies on pathology of H5N1 highly pathogenic Avian Influenza Virus Outbreaks in Nigeria. Nig. Vet. J. 38(3), 1–10. Akanbi, O.B. and Taiwo, V.O. 2014a. Mortality and pathology associated with highly pathogenic avian influenza H5N1 outbreaks in commercial poultry production systems in Nigeria. Int. Schol. Res. Notices. https://doi.org/10.1155/2014/415418. Akanbi, O.B. and Taiwo, V.O. 2014b. Backyard poultry mortality associated with highly pathogenic avian influenza (HPAI) H5N1 outbreaks in Nigeria. IOSR. J. Agric. Vet. Sci. 7(9), 23–27. Akanbi, O.B. and Taiwo, V.O. 2015. Husbandry practices and outbreak features of natural highly pathogenic avian influenza (H5N1) in turkey flocks in Nigeria, 2006–2008. J. World’s. Poult. Res. 5(4), 109–114. Akanbi, O.B., Alaka, O.O., Olaifa, O.S., Meseko, C.A., Inuwa, B., Ohore, O.G., Tijani, M., Jarikre, T., Ola, O., Odita, C., Ahmed, J.S., Fagbohun, O., Oluwayelu, D., Daodu, O.B., Oladele, O., Olapade, J., Taiwo, O. and Muhammad, M. 2024. Pathology and molecular detection of influenza A subtype H9N2 virus in commercial poultry in Nigeria. Open. Vet. J. 14(9), 2381–2391; doi:10.5455/OVJ.2024.v14.i9.26 Akanbi, O.B., Ekong, P.S., Odita, C.I. and Taiwo, V.O. 2016. The effect of highly pathogenic avian influenza (HPAI) H5N1 outbreaks on mixed species poultry farms in Nigeria. J. World’s. Poult. Res. 6(3), 147–152. Alhaji, N.B., Adeiza, A.M., Godwin, E.A., Haruna, A.E., Aliyu, M.B. and Odetokun, I.A. An assessment of the highly pathogenic avian influenza resurgence at human-poultry-environment interface in North-central Nigeria: Sociocultural determinants and One Health implications. 2023. One Health, 1(16),1–9; doi: 10.1016/j.onehlt.2023.100574 Alhaji, N.B. and Odetokun, I.A. (2011) Assessment of biosecurity measures against highly pathogenic avian influenza risks in small-scale commercial farms and free-range poultry flocks in the Northcentral Nigeria. Transbound Emerg. Dis. 58(2), 157–161; doi: 10.1111/j.1865-1682.2010.01195.x Ameji, N.O., Oladele, O.O., Meseko, C.A., Mshelia, G.D. and Lombin, L.H. 2019. Outbreak of highly pathogenic avian influenza subtype H5N8 in two multi-age chicken farms in Jos, Plateau State, Nigeria. Sokoto. J. Vet. Sci. 17(3), 60–65. Assam, A. and Shehu, D. 2023. Avian influenza surveillance in pigs, dogs, chickens and slaughter slabs biosecurity in Kaduna Metropolis, Nigeria. J. Appl. Life Sci. Int. 26(5), 17–26; doi:10.9734/jalsi/2023/v26i5615 Atuman, Y.A., Inuwa, B., Bakam, J., Orioko, E., Emmanuel, H.S. and Meseko, C. 2025. Exotic and zoological birds resident and imported into Nigeria harbour highly pathogenic avian influenza virus: Threat to poultry production, food security and public health. BioRxiv, 1; HYPERLINK "https://doi.org/10.1101/2025.01.22.634354" doi:10.1101/2025.01.22.634354 (Accessed 20 July 2025). Carter, T. and Iqbal, M. 2024. The influenza A virus replication cycle: a comprehensive review. Viruses 16(2), 316; doi:10.3390/v16020316 Chieloka, O.S., Ameh, C. and Stephen, A.A. 2021. Evaluation of the avian influenza surveillance system in Enugu State, Nigeria, 2015–2017. PAMJ-One. Health 4(6), 1–7; doi: 10.11604/pamj-oh.2021.4.6.27496 Coker, T., Meseko, C., Odaibo, G. and Olaleye, D. 2014. Circulation of the low pathogenic avian influenza subtype H5N2 virus in ducks at a live bird market in Ibadan, Nigeria. Infect. Dis. Poverty. 3(1), 38; doi:10.1186/2049-9957-3-38 Columba Teru, V., Manu, S.A., Ahmed, G.I., Junaidu, K., Newman, S., Nyager, J., Iwar, V.N., Mshelbwala, G.M., Joannis, T., Maina, J.A. and Apeverga, P.T. 2012. Situation-based survey of avian influenza viruses in possible “bridge” species of wild and domestic birds in Nigeria. Influenza Res. Treat. 2012, 567601; doi:10.1155/2012/567601 Food and Agriculture Organization (FAO). 2008. Poultry sector country review—Nigeria 1-95. Geneva, Switzerland, Food and Agriculture Organization of the United Nations. Available via https://openknowledge.fao.org/server/api/core/bitstreams/c49d8c0c-fa4a-4ea5-84ca-b7fc17fa1313/content (Accessed 20 July 2025) Fasina, F.O., Bisschop, S.P.R., Ibironke, A.A. and Meseko, C.A. 2009. Avian influenza risk perception among poultry workers, Nigeria. Emerg. Infect. Dis. 15(4), 616–617; doi:10.3201/eid1504.070159 Fasina, F.O., Rivas, A.L., Bisschop, S.P.R., Stegeman, A.J. and Hernandez, J.A. 2011. Identification of risk factors associated with highly pathogenic avian influenza H5N1 virus infection in poultry farms in Nigeria during the epidemic of 2006–2007. Prev. Vet. Med. 98(2–3), 204–208; doi:10.1016/j.prevetmed.2010.11.007 Federal Ministry of Agriculture and Rural Development (FMARD). 2013. Self-declaration from Nigeria on its disease-free status from notifiable avian influenza. Abuja, Nigeria, Federal Ministry of Agriculture and Rural Development. Available via https://doc.woah.org/dyn/portal/digidoc.xhtml?statelessToken=Rea8-c6iokriuzElsjraIowR9xD0deZq8ndUH3gFQT8=&actionMethod=dyn%2Fportal%2Fdigidoc.xhtml%3AdownloadAttachment.openStateless (Accessed 20 July 2025) Food and Agriculture Organization (FAO). 2008. Poultry sector country review—Nigeria. Food and Agriculture Organization of the United Nations, Geneva, Switzerland. Freidrich Loeffler Institute (FLI). 2025. Avian influenza: The risk of outbreaks in poultry is increasing, with cranes among wild birds being particularly affected for the first time. Available via https://www.fli.de/en/press/press-releases/press-singleview/avian-influenza-the-risk-of-outbreaks-in-poultry-is-increasing-with-cranes-among-wild-birds-being-particularly-affected-for-the-first-time/ (Accessed 20 October 2025). Fusaro, A., Joannis, T., Monne, I., Salviato, A., Yakubu, B., Meseko, C., Oladokun, T., Fassina, S., Capua, I. and Cattoli, G. 2009. Introduction into Nigeria of a distinct genotype of avian influenza virus (H5N1). Emerg. Infect. Dis. 15(3), 445–447; doi:10.3201/eid1503.081161 Fusaro, A., Nelson, M.I., Joannis, T., Bertolotti, L., Monne, I., Salviato, A., Olaleye, O., Shittu, I., Sulaiman, L., Lombin, L.H., Capua, I., Holmes, E.C. and Cattoli, G. 2010. Evolutionary dynamics of multiple sublineages of H5N1 influenza viruses in Nigeria from 2006 to 2008. J. Virol. 84, 3239–3247; doi:10.1128/jvi.02385-09 Joannis, T., Lombin, L.H., De Benedictis, P., Cattoli, G. and Capua, I. 2006. Confirmation of H5N1 avian influenza in Africa. Vet. Rec. 158(9), 309–310. Lakes, T., Schmitz, T. and Füller, H. 2025. Pathogenic built environment? Reflections on modeling spatial determinants of health in urban settings considering the example of COVID-19 studies. Front. Public. Health. 13, 1502897; doi:10.3389/fpubh.2025.1502897 Li, X., Lv, X., Li, Y., Xie, L., Peng, P., An, Q., Fu, T., Qin, S., Cui, Y., Zhang, C., Qin, R., Qu, F., Zhao, Z., Wang, M., Xu, Q., Li, Y., Yang, G., Chen, G., Zhang, J., Zheng, H., Ma, E., Zhou, R., Zeng, X., Wang, Y., Hou, Z., Wang, Y., Chu, D., Li, Y. and Chai, H. 2020. Emergence, prevalence, and evolution of H5N8 avian influenza viruses in central China, 2020. Emerg. Microbes Infect. 11(1), 73–82; doi:10.1080/22221751.2021.2011622 Meseko, C., Milani, A., Inuwa, B., Chinyere, C., Shittu, I., Ahmed, J., Giussani, E., Palumbo, E., Zecchin, B., Bonfante, F., Maniero, S., Angot, A., Niang, M., Fusaro, A., Gobbo, F., Terregino, C., Olasoju, T., Monne, I. and Muhammad, M. 2023. The evolution of highly pathogenic avian influenza A (H5) in poultry in Nigeria, 2021–2022. Viruses 15(6), 1387; doi:10.3390/v15061387 Mkpuma, N., Oyedeji, L.M., Akintule, A.O., Chinyere, C.N., Agbato, A.O., Inuwa, B., Shehu, S., Olawuyi, K.A., Tige, D.B., Gado, D.A. and Meseko, C.A. 2024. Serological and molecular detection of low pathogenic avian influenza subtype H9N2 in commercial poultry flocks in Kaduna and Kano States, Nigeria. Nig. Vet. J. 54(2), 26–38. Nagy, A., Mettenleiter, T.C. and Abdelwhab, E.M. 2017. A brief summary of the epidemiology and genetic relatedness of avian influenza H9N2 virus in birds and mammals in the Middle East and North Africa. Epidemiol. Infect. 145(16), 3320–3333; doi:10.1017/S0950268817002576 National Veterinary Research Institute (NVRI). 2006. Unpublished Case database. Vom, Nigeria. National Veterinary Research Institute (NVRI). 2025. Recurrent highly pathogenic avian influenza (HPAI) H5N1. https://nvri.gov.ng/single/RE-CURRENT-HIGHLY-PATHOGENIC-AVIAN-INFLUENZA-%28HPAI%29-H5N1. National Veterinary Research Institute (NVRI). 2025. Report. Vom, Nigeria. Availble via https://nvri.gov.ng/single/RE-CURRENT-HIGHLY-PATHOGENIC-AVIAN-INFLUENZA-%28HPAI%29-H5N1 (Accessed 20 July 2025). Obayelu, A.E. 2007. Socio-economic analysis of the impacts of avian influenza epidemic on households’ poultry consumption and poultry industry in Nigeria: empirical investigation of Kwara State. Livest. Res. Rural. Dev. 19(1), 1–13. Available via https://lrrd.cipav.org.co/lrrd19/1/obay19004.htm. Odita, C. I., Hambolu, M. A., Olasoju, T. I., Makanjuola, O. A., Sito, O. and Meseko, C. A. 2022. Cluster poultry farming, the bane of highly pathogenic avian influenza spread in Lagos State Nigeria. Niger. Vet. J. 43(4), 1-9; doi: 10.4314/nvj.v43i4.1 Oladokun, A.T., Meseko, C.A., Ighodalo, E., John, B. and Ekong, P.S. 2012. Effect of intervention on the control of highly pathogenic avian influenza in Nigeria. Pan Afr. Med. J. 13, 14. Pan American Health Organization (PAHO). 2025. Epidemiological update: Avian influenza A (H5N1) in the Americas region – 15 May 2025. https://www.paho.org/en/documents/epidemiological-update-avian-influenza-ah5n1-americas-region-15-may-2025. Peacock, T.H.P., James, J., Sealy, J.E. and Iqbal, M. 2019. A global perspective on H9N2 avian influenza virus. Viruses 5(11), 620; doi: 10.3390/v11070620 Rosa Meurer, I., Silva, M.R., Kleinsorge Roland, R., Do Amaral Corrêa, J.O. and Soares Coimbra, E. 2024. Evaluation of medical professionals’ knowledge about Q fever. Sci. Med. 34(1), e45474; doi:10.15448/1980-6108.2024.1.45474 Saidu, L., Wakawa, A.M., Abdu, P.A., Adene, D.F., Kazeem, H.M., Ladan, K.C., Abdu, M., Miko, R.B., Fatihu, M.Y., Adamu, J. and Mamman, P.H. 2008. Impact of avian influenza in some states of Nigeria. Int. J. Poult. Sci. 7(9), 913–916. Shittu, I., Bianco, A., Gado, D., Mkpuma, N., Sulaiman, L., Laleye, A., Gobbo, F., Bortolami, A., Bonfante, F., Vakuru, C., Meseko, C., Fusaro, A., Shamaki, D., Alabi, O., Terregino, C. and Joannis, T. 2020. First detection of highly pathogenic H5N6 avian influenza virus on the African continent. Emerg. Microbes. Infect. 9(1), 886–888. Stallknecht, D.E. and Brown, J.D. 2007. Wild birds and the epidemiology of avian influenza. J. Wildl. Dis. 43(3), S15–S20. Sulaiman, L., Shittu, I., Fusaro, A., Inuwa, B., Zecchin, B., Gado, D., Schivo, A., Bianco, A., Laleye, A., Gobbo, F., Vakuru, C., Joannis, T., Monne, I. and Meseko, C. 2021. Live bird markets in Nigeria: a potential reservoir for H9N2 avian influenza viruses. Viruses 13(8), 1–8; doi:10.3390/v13081445 Tassoni, L., Fusaro, A., Milani, A., Lemey, P., Awuni, J.A., Sedor, V.B., Dogbey, O., Commey, A.N.O., Meseko, C., Joannis, T., Minoungou, G.L., Ouattara, L., Haido, A.M., Cisse-Aman, D., Couacy-Hymann, E., Dauphin, G., Cattoli, G. and Monne, I. 2016. Genetically different highly pathogenic avian influenza A(H5N1) viruses in West Africa, 2015. Emerg. Infect. Dis. 22(12), 2132–2136; doi:10.3201/eid2212.160578 Webby, R.J. and Uyeki, T.M. 2024. An update on highly pathogenic avian influenza A(H5N1) virus, clade 2.3.4.4b. J. Infect. Dis. 230(3), 533–542; doi:10.1093/infdis/jiae379 Webster, R.G., Bean, W.J., Gorman, O.T., Chambers, T.M. and Kawaoka, Y. 1992. Evolution and ecology of influenza A viruses. Microbiol. Rev. 56, 152–179. World Organisation for Animal Health (WOAH). 2025. Highly pathogenic avian influenza (HPAI) situation report 67, 1–12. Available via at https://www.woah.org/app/uploads/2025/02/hpai-report-67.pdf (Accessed 20 July 2025) World Health Organization (WHO). 2025. Cumulative number of confirmed human cases for avian influenza A (H5N1) reported to WHO, 2003–2025, 22 April 2025. Geneva: WHO. Available via https://www.who.int/publications/m/item/cumulative-number-of-confirmed-human-cases-for-avian-influenza-a(H5N1)-reported-to-who--2003-2025--22-april-2025 (Accessed 30 July 2025) | ||
| How to Cite this Article |
| Pubmed Style Akanbi OB, Lakes T. Review of avian influenza outbreaks in Nigeria 2006–2025. Open Vet. J.. 2025; 15(12): 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 Web Style Akanbi OB, Lakes T. Review of avian influenza outbreaks in Nigeria 2006–2025. https://www.openveterinaryjournal.com/?mno=274830 [Access: January 24, 2026]. doi:10.5455/OVJ.2025.v15.i12.2 AMA (American Medical Association) Style Akanbi OB, Lakes T. Review of avian influenza outbreaks in Nigeria 2006–2025. Open Vet. J.. 2025; 15(12): 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 Vancouver/ICMJE Style Akanbi OB, Lakes T. Review of avian influenza outbreaks in Nigeria 2006–2025. Open Vet. J.. (2025), [cited January 24, 2026]; 15(12): 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 Harvard Style Akanbi, O. B. & Lakes, . T. (2025) Review of avian influenza outbreaks in Nigeria 2006–2025. Open Vet. J., 15 (12), 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 Turabian Style Akanbi, Olatunde B., and Tobia Lakes. 2025. Review of avian influenza outbreaks in Nigeria 2006–2025. Open Veterinary Journal, 15 (12), 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 Chicago Style Akanbi, Olatunde B., and Tobia Lakes. "Review of avian influenza outbreaks in Nigeria 2006–2025." Open Veterinary Journal 15 (2025), 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 MLA (The Modern Language Association) Style Akanbi, Olatunde B., and Tobia Lakes. "Review of avian influenza outbreaks in Nigeria 2006–2025." Open Veterinary Journal 15.12 (2025), 6146-6165. Print. doi:10.5455/OVJ.2025.v15.i12.2 APA (American Psychological Association) Style Akanbi, O. B. & Lakes, . T. (2025) Review of avian influenza outbreaks in Nigeria 2006–2025. Open Veterinary Journal, 15 (12), 6146-6165. doi:10.5455/OVJ.2025.v15.i12.2 |