| Research Article | ||
Open Vet. J.. 2026; 16(1): 577-588 Open Veterinary Journal, (2026), Vol. 16(1): 577-588 Research Article Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacyNuohan Xia1†, Jian Sun2†, Ge Hu1, Qian Zhang1 and Tao Zhang1*1Beijing Key Laboratory of Traditional Chinese Veterinary Medicine, Animal Science and Technology College, Beijing University of Agriculture, Beijing, China 2School of Animal Science and Technology, Beijing Vocational College of Agriculture, Beijing, China *Corresponding Author: Tao Zhang. Beijing Key Laboratory of Traditional Chinese Veterinary Medicine, Animal Science and Technology College, Beijing University of Agriculture, Beijing, China. Email: zhangtao [at] bua.edu.cn Submitted: 31/07/2025 Revised: 15/11/2025 Accepted: 07/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Pulmonary microvascular endothelial cells (MVECs) are not only targets of the highly pathogenic porcine reproductive and respiratory syndrome virus (HP-PRRSV) but also the pivotal hub of inflammation, interacting with alveolar macrophages to amplify the inflammatory response and determine the extent of tissue damage. Glycocalyx disruption in MVECs is often the initiating factor of numerous functional disorders, and astragalus polysaccharide (APS) has been shown to improve glycocalyx integrity in MVECs. Aim: This study aimed to investigate the mediating role of glycocalyx disruption in HP-PRRSV-induced pulmonary microvascular endothelial dysfunction and the glycocalyx contribution to the anti-HP-PRRSV efficacy of APS. Methods: In vitro porcine pulmonary MVECs were infected with the HP-PRRSV JXA1 strain and treated with APS with or without prior glycocalyx degradation mediated by heparinase (HPA) III. The expression of glycocalyx components, PRRSV N protein, and adhesion molecules was detected using western blotting or flow cytometry. The barrier function was analyzed by measuring trans-endothelial electric resistance (TEER) and permeability of horseradish peroxidase-labeled streptavidin (HRP-SA). Furthermore, neutrophil TEM and its bactericidal capacity were assayed using Transwell plates. Results: HP-PRRSV infection reduced the expression of heparan sulfate proteoglycan 2 (HSPG-2), glypican-1, and syndecan-1 proteoglycans. It also lowered labeled Lens culinaris agglutinin, Datura stramonium agglutinin, and Phaseolus vulgaris agglutinin-E -reactive glycan ligand levels in porcine pulmonary MVECs. However, APS significantly alleviated this damage. Furthermore, its infection decreased TEER, increased HRP-SA permeability, elevated vascular cell adhesion molecule-1 and intercellular adhesion molecule-1 expression, promoted neutrophil TEM, and impaired their bactericidal activity. APS markedly suppressed HP-PRRSV replication and restored these virus-induced impairments. Conversely, enzymatic glycocalyx removal with HPA III intensified microvascular endothelial injury and largely weakened the APS’s restorative effect. Conclusion: Glycocalyx disruption is a critical pathogenic event that orchestrates pulmonary microvascular endothelial dysfunction during HP-PRRSV infection. More importantly, we delineated a novel mechanism by which APS exerts its therapeutic effects. Keywords: Porcine reproductive and respiratory syndrome virus, Astragalus polysaccharides, Microvascular endothelial cells, Glycocalyx, Endothelial dysfunction. IntroductionPorcine reproductive and respiratory syndrome virus (PRRSV) causes reproductive disorders in sows and respiratory issues in young pigs, with a high degree of variability. Currently, highly pathogenic PRRSV (HP-PRRSV) has become the primary variant strain in many regions, including the JXA1 strain in China, due to its high pathogenicity and mortality rate (Wang et al., 2018; Jiang et al., 2023). It is more challenging to control and has a devastating impact on the pig industry, while its pathogenic mechanism has not yet been fully elucidated. In piglets, interstitial pneumonia is considered the primary lesion caused by PRRSV, with alveolar macrophages identified as the main target cells. However, it cannot account for all aspects of its pathogenic mechanisms, relying only on alveolar macrophages, such as pulmonary edema and inflammatory cell infiltration. Microvascular endothelial cells (MVECs) are the pivotal “inflammation-immunity-coagulation” hub in lung viral infection. Their dysfunction rapidly dismantles the alveolo-capillary barrier, ignites cytokine storms, and recruits leukocytes through their interaction with alveolar macrophages (Osorio-Valencia et al., 2024; Sun et al., 2022), thereby driving the progression to acute lung injury and multi-organ failure. Our previous research has shown that HP-PRRSV can infect porcine pulmonary MVECs (Li et al., 2020), leading to glycocalyx degradation and glycan component changes (Song et al., 2022). The glycocalyx, a villous or reticular structure covering the surface of all MVECs, comprises glycoproteins, proteoglycans, and other polysaccharide-rich components (Jedlicka et al., 2020). It not only participates in numerous biological functions of MVECs (Liu et al., 2020; Weinbaum et al., 2021) but also plays a role in viral infections, becoming the primary target of damage (Yamaoka-Tojo, 2020; Taghavi et al., 2022). Disruption of the surface glycocalyx layer is often the initial event that leads to functional alterations in MVECs (Zhang et al., 2018). The study of viral pathogenesis through endothelial glycocalyx damage and the exploration of therapeutic strategies by targeting the endothelial glycocalyx are both receiving increasing attention (Cao et al., 2019; Machin et al., 2023). Chinese herbal polysaccharides are natural active macromolecules extracted from traditional Chinese medicines. Although they are generally recognized to possess broad bioactivities, including immunomodulation, antioxidation, and antitumor effects, their precise mechanisms of action remain unclear. Astragalus polysaccharide (APS), extracted from the traditional Chinese medicine astragalus membranaceus, is the most widely used and the most intensively studied herbal polysaccharide. Interestingly, Chinese herbal polysaccharides share numerous similarities with the endothelial glycocalyx in both chemical composition and biological properties. Our existing findings demonstrated that both APS and Pulsatilla Decoction polysaccharides can improve the structure and function of the glycocalyx in MVECs (Zhang et al., 2014; Zhang et al., 2021). Therefore, we hypothesized that the anti-HP-PRRSV effect of APS may be related to its actions on the endothelial glycocalyx. In this study, to investigate the mediating role of glycocalyx disruption in HP-PRRSV-induced pulmonary microvascular endothelial dysfunction and the glycocalyx contribution to the anti-HP-PRRSV efficacy of APS, we analyzed the microvascular endothelial barrier function, adhesion molecule expression, and neutrophil transendothelial migration. Our findings would enrich the understanding of the pathogenic mechanism of HP-PRRSV and explore the innovative mechanism of APS in counteracting HP-PRRSV-induced damage to MVECs from the perspective of glycocalyx. Materials and MethodsVirusThe HP-PRRSV JXA1 strain was kindly provided by Dr. Zhanzhong Zhao at the Chinese Academy of Agricultural Sciences and was propagated in Marc-145 cells. When approximately 80% of the cells exhibited an obvious cytopathic effect (CPE), the cells were subjected to three freeze-thaw cycles (−20°C/room temperature) to release the virus, followed by centrifugation at 3,000 rpm for 20 minutes. The supernatant was used as the virus solution. Similarly, normal Marc-145 cells were repeatedly frozen, thawed, and centrifuged, and the supernatant was used as the negative control solution. The virus titer was determined using the Reed–Muench method (Lei et al., 2021), and the TCID50 of the virus solution was 10−5.64/100 μl. The virus and negative control solutions were stored at −80°C. Bacterial strainThe E. coli K99 strain preserved at −80°C in our laboratory was thawed and inoculated into Luria–Bertani broth (Qingdao Haibo, #HB0128) overnight at 37°C with shaking. The bacterial solution was then inoculated onto MacConkey agar plates and incubated at 37°C for approximately 16 hours. Single colonies were picked and cultured overnight in Luria–Bertani broth again. The bacterial concentration was determined to be 4 × 1010 CFU/ml using the plate count method. The bacterial solution was stored at 4°C and used within a week. Isolation of neutrophils from porcine peripheral bloodBlood from clinically healthy pigs was collected and anticoagulated with heparin. Pig peripheral blood neutrophils were separated using a pig peripheral blood neutrophil separation kit (Solarbio, #P4140) according to the manufacturer’s instructions. Cells were resuspended in RPMI 1640 medium (Solarbio, #11875) containing 10% fetal bovine serum (Gibco, #10099141), adjusted to a cell density of 1 × 106 cells/ml, and incubated in a cell culture incubator until further use. Isolation and culture of porcine pulmonary MVECsPeripheral lung tissue from an approximately 10-day-old SPF piglet (Beijing SPF Pig Breeding Management Center) was collected for separating MVECs as described previously (Song et al., 2022). The piglet was anesthetized by an intraperitoneal injection of pentobarbital sodium (40 mg/kg), followed by an intraperitoneal injection of potassium chloride (75 mg/kg) for euthanasia. The Animal Care and Protection Committee of Beijing University of Agriculture approved all procedures (No. BUA_ZT2022012). The purified cells were cultured in DMEM/F12 medium (Gibco, #2105324) containing 10% fetal bovine serum, and passages 4–6, which maintain stable endothelial characteristics, were used for the experiments. Flow cytometryFlow cytometry was performed to verify the expression of heparan sulfate proteoglycan 2 (HSPG-2) and sugar ligands of the three lectins. Cells were grown to subconfluence and divided into three groups: control, JXA1, and JXA1+APS. MVECs of the control group were treated with the negative control solution for 1 hour and incubated in the maintenance medium containing 2% fetal bovine serum for 24 hours. MVECs of the JXA1 group were treated with the HP-PRRSV JXA1 strain solution for 1 hour and incubated in the maintenance medium for 24 hours. MVECs of the JXA1+APS group were treated with the HP-PRRSV JXA1 strain solution for 1 hour and incubated in the maintenance medium containing 100 μg/ml APS (purity: 90%, Solarbio, #SA9790) for 24 hours. The concentration of APS was determined based on preliminary experiments and literature (Zhou et al., 2018). After detachment with trypsin-EDTA solution (Solarbio, #T1300) and centrifugation at 1,000 rpm for 5 minutes, the cells were incubated with FITC-labeled Lens culinaris agglutinin (LCA, Vector, #FL-1041, 1:200), FITC-labeled Datura stramonium agglutinin (DSA, Vector, #FL-1181, 1:200), FITC-labeled Phaseolus vulgaris agglutinin-E (PHA-E, Vector, #FL-1211, 1:200), and the HSPG-2 antibody (Bioss, #bs5072R, 1:100), followed by incubation with FITC-labeled goat anti-rabbit IgG. In the negative control group, PBS was used to replace the antibody or lectin. After incubation at 37°C for 1 hour, the cells were washed with phosphate-buffered saline (PBS), resuspended in 500 μl PBS, and analyzed using a flow cytometer (Becton Dickinson, FACS Calibur). Western blotThe MVECs were grouped and treated according to Section “2.5. Flow cytometry” for the detection of glypican 1 (GPC-1, Bioss, #bs2426R, 1:100) and syndecan 1 (SDC-1, Abcam, #ab128936, 1:100). MVECs were divided into five groups according to treatment strategy to detect vascular cell adhesion molecule-1 (VCAM-1), intercellular adhesion molecule-1 (ICAM-1), and PRRSV N protein (Fig. 3A): control, JXA1, JXA1+APS, HPA III +JXA1, and HPA III +JXA1+APS, each in triplicate. Cells were sampled 24 hours after the APS treatment. The concentration of HPA III (R&D, #6145-GH-010) was optimized from 0, 10, 20, 30, and 50 mU/ml, which were used to remove SDC-1, HPSG-2, and GPC-1 by immunofluorescence staining (Supplementary Fig. 1).
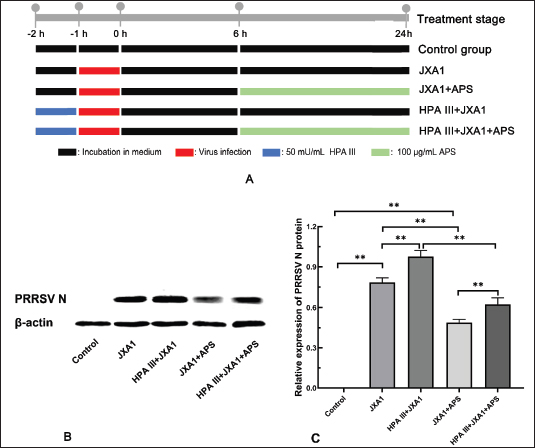
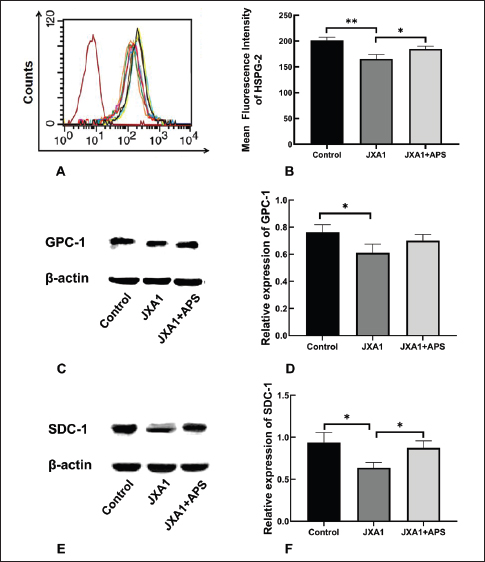
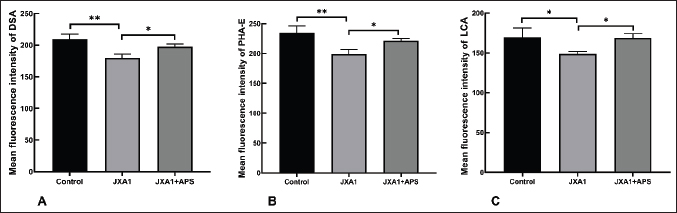
Fig. 3. Inhibition of astragalus polysaccharide on HP-PRRSV JXA1 strain replication in porcine pulmonary microvascular endothelial cells. (A) Schematic illustration of experimental groups and treatment procedures. (B) Western blot quantification of PRRSV N protein levels. (C) Statistical analysis of PRRSV N protein levels. “*” indicates P < 0.05, “**” indicates P < 0.01. MVECs were lysed using RIPA buffer (Beyotime, #P0013B) and centrifuged at 14,000 g for 10 minutes to collect supernatants. Protein concentrations were measured using the BCA assay kit (Beyotime, #P0011) and adjusted to the same level for each group using sample loading buffer, followed by boiling to denature the proteins. Electrophoresis was performed for approximately 2 hours at a constant voltage of 75 V, followed by wet transfer at a constant current of 400 mA for 25 minutes. After blocking with a skim milk solution and washing with Tris-buffered saline and Tween-20 buffer (Solarbio, #T1081), the PVDF membranes (Solarbio, #YA1691) were incubated in antibodies against β-actin (1:20,000), SDC-1 (1:3,000), GPC-1 (1:1,000), VCAM-1 (1:500, Santa Cruz, #sc-18864), ICAM-1 (1:10000, Proteintech, #10831-AP), and PRRSV N protein (1:1,000, Bioss, #bs23941R) overnight at 4°C. After washing, the PVDF membranes were incubated in HRP-conjugated IgG (1:1000, Beyotime, #A0208) at room temperature with shaking for 1 hour. The PVDF membranes were developed using an ECL chemiluminescence kit (Beyotime, #P0018S) and imaged using a gel imaging system. Measurement of trans-endothelial electric resistance (TEER)MVECs were passaged into Transwell culture inserts (Corning, #3421), and cell monolayers were considered to have formed when the TEER no longer increased within 12 hours. Cells were divided into five groups according to treatment strategy (Fig. 3A), each with three replicates. TEER was measured at 0, 12, 24, 36, 48, 60, and 72 hours using an epithelial cell resistance meter (Millipore, Millicell® ERS-2). Each well was measured thrice at different points, and the average value was calculated. The TEER value of each well was calculated using the formula below, where the blank resistance value was the resistance measured in wells without cells, and the area of the Transwell insert membrane used was 0.33 cm². The TEER values of each group were expressed as “mean ± SD” and analyzed using GraphPad Prism 8 software, with p < 0.05 considered statistically significant. TEER=(measured resistance value of each well - blank resistance value) × 0.33 Evaluation of the permeability of horseradish peroxidase-streptavidin (HRP-SA)The material permeability of the cell monolayer was determined using the leakage ratio of HRP-SA, as previously described (Chen and Yeh, 2017). Cells were subcultured and grouped as described in Section “2.7. Measurement of the TEER.” At 12, 24, 36, 48, 60, and 72 hours, 0.1 ml of 1 mg/ml HRP-SA solution (Merck, #06102022) was added to each insert well. After a 30-min incubation, the media in the lower chamber were sampled and replaced with new media. The OD values were measured at 492 nm using the TMB color development kit (Bioss, #C04-03001) according to the manufacturer’s instructions. Neutrophil migration assayMVECs were seeded in Transwell inserts (Corning, #3421) and grouped according to Fig. 3A. Twenty-four hours after APS treatment, 0.1 ml of 1 × 106 cells/ml neutrophils was added to the Transwell inserts. After a 4-hour incubation, neutrophils were collected from the lower chambers and counted using a counting chamber. The neutrophil migration rates for each group were calculated using the following formula: Neutrophil migration ratio (100%)=number of neutrophils in the lower chamber/number of neutrophils added in the upper chamber × 100%. Neutrophil bactericidal assayNeutrophils were collected from the lower chambers of each group according to Section “2.9. Neutrophil migration assay” and were centrifuged and resuspended in a maintenance medium without antibiotics. Their densities were adjusted to be the same. Dilute the logarithmic phase bacteria 105 folds and added to the neutrophil suspension at a 1:10 volume ratio. After incubation for 3 hours, 0.1 ml of the medium was sampled and plated in triplicate onto bacterial culture plates. The plates were cultured for approximately 16 hours, and the bacterial colonies were counted in each group. Data analysisWestern blot results were analyzed using ImageJ software to calculate the grayscale values of the protein bands, with the relative protein expression defined as the ratio of the β-actin band. Flow cytometry was used to assess the relative fluorescence intensity compared to the control. All experiments were performed using three independent replicates (n=3), and each replicate was derived from a distinct cell culture preparation. Data were presented as mean ± SD and analyzed using GraphPad Prism 8.0 software. Specifically, two-way ANOVA (factors: treatment and time) followed by Tukey’s post hoc test was applied to the TEER and HRP-SA leakage data, whereas one-way ANOVA with Tukey’s test was used for all other datasets. Statistical analysis used one-way or two-way ANOVA with Tukey’s post hoc test in GraphPad Prism 8.0 software, with Statistical significance set at P < 0.05 or P < 0.01. Ethical approvalNot needed for this study. ResultsGlycocalyx component expression in HP-PRRSV-infected MVECsFlow cytometry and Western blot results indicated that HP-PRRSV infection led to a decrease in the expression of proteoglycans HSPG-2, GPC-1, and SDC-1 (Fig. 1) and reduced the LCA, DSA, and PHA-E-reactive glycan ligands (Fig. 2) in porcine lung MVECs, whereas APS treatment significantly alleviated this damage. These data indicate that APS significantly repaired the damage to glycocalyx components caused by HP-PRRSV.
Fig. 1. Effects of astragalus polysaccharide on glycocalyx proteoglycan components in HP-PRRSV-infected porcine pulmonary microvascular endothelial cells. (A) HSPG-2 levels were quantified by flow cytometry. (B) Statistical analysis of HSPG-2 levels. (C) GPC-1 levels were quantified by Western blot. (D) Statistical analysis of GPC-1 levels. (E) SDC-1 levels were quantified by Western blot. (F) Statistical analysis of the levels of SDC-1. “*” indicates P < 0.05, “**” indicates P < 0.01.
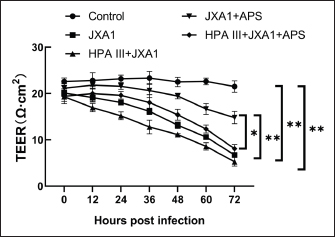
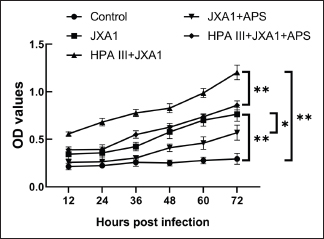
Fig. 2. Effects of astragalus polysaccharide on glycocalyx sugar chain components in porcine pulmonary microvascular endothelial cells. (A) Quantitative analysis of DSA-reactive glycan ligand levels. (B) Quantitative analysis of PHA-E-reactive glycan ligand levels. (C) Quantitative analysis of LCA-reactive glycan ligand levels. “*” indicates P < 0.05, “**” indicates P < 0.01. HP-PRRSV replication in porcine pulmonary MVECsGiven the crucial role of glycocalyx in virus-cell interactions, its status would affect viral infection and replication in host cells. By western blot assay (Fig. 3), there was a notable PRRSV N protein expression in MVECs after exposure to HP-PRRSV JXA1 for 30 hours, and pre-treatment with HPA III increased that. Intervention with 100 μg/ml APS significantly reduced PRRSV N protein expression, whereas the HPA III+APS group showed significantly higher expression than the JXA1+APS group. These results indicate that APS can reduce HP-PRRSV replication in infected MVECs, whereas glycocalyx degradation significantly inhibits its intervention effect. HP-PRRSV-induced TEERThe decreased expression of glycocalyx components indicates the possible disruption of the microvascular endothelial barrier. TEER across the monolayer of porcine pulmonary MVECs was measured In vitro using the Transwell model. The results showed that HP-PRRSV infection significantly reduced TEER, with a gradual decrease observed with increased infection time (P < 0.01), which was significantly upregulated by APS treatment (P < 0.01). Partial degradation of the glycocalyx with HPA III further promoted the HP-PRRSV-induced decrease in TEER (P < 0.01), and it significantly weakened the effect of APS treatment (P < 0.05) (Fig. 4).
Fig. 4. Effects of astragalus polysaccharide on TEER across the monolayer of porcine pulmonary microvascular endothelial cells infected with the HP-PRRSV JXA1 strain. “*” indicates P < 0.05, “**” indicates P < 0.01. HP-PRRSV-induced HRP-SA leakageTo confirm the regulatory role of APS in barrier function damage caused by HP-PRRSV infection in porcine lung MVECs, we assessed solute permeability. HP-PRRSV infection significantly increased HRP-SA leakage, with an increasing amount of leakage observed with prolonged infection time (P < 0.01), which was significantly attenuated by the APS treatment (P < 0.05) (Fig. 5). After pretreatment with HPA III, the HP-PRRSV-induced HRP-SA leakage was further aggravated, and the APS inhibitory effect was also significantly weakened (P < 0.01).
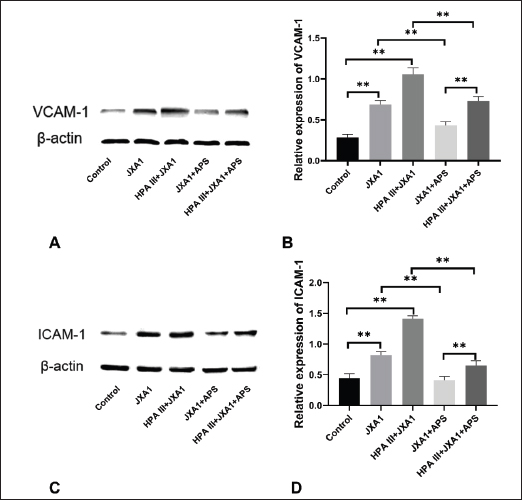
Fig. 5. Effects of astragalus polysaccharide on the permeability of horseradish peroxidase-labeled streptavidin across the monolayer of porcine pulmonary microvascular endothelial cells infected with HP-PRRSV JXA1. “*” indicates P < 0.05, “**” indicates P < 0.01. VCAM-1 and ICAM-1 expression in HP-PRRSV-infected MVECsThe shedding of glycocalyx would bring out adhesion molecules on cell surfaces and affect their expression, recruiting and activating inflammatory cells. Based on previous transcriptome sequencing results (Wu et al., 2023), the expression of two adhesion molecules was investigated in this experiment. The results showed that normal porcine lung MVECs had low VCAM-1 and ICAM-1 expression, and HP-PRRSV infection significantly increased the expression of both adhesion molecules (P < 0.01), and HPA III treatment further promoted their expression (Fig. 6). Moreover, APS significantly downregulated the expression of VCAM-1 and ICAM-1 (P < 0.01), whereas treatment with HPA III significantly attenuated the downregulation effect of APS (P < 0.01).
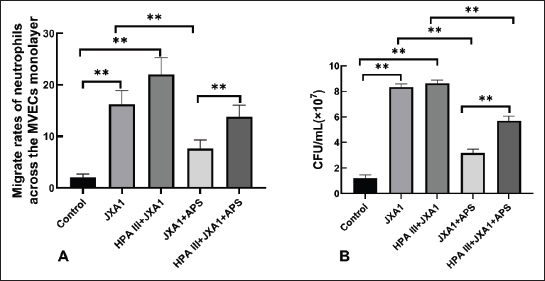
Fig. 6. Effects of astragalus polysaccharide on VCAM-1 and ICAM-1 expression in HP-PRRSV-infected porcine pulmonary microvascular endothelial cells. (A) VCAM-1 levels were quantified by western blot. (B) Statistical analysis of VCAM-1 levels. (C) ICAM-1 levels were quantified by Western blot. (D) Statistical analysis of ICAM-1 levels. “**” indicates P < 0.01. Neutrophil migration across the MVEC monolayer during HP-PRRSV infectionWe examined the transendothelial migration rate of neutrophils and the bactericidal activity of migrated neutrophils to further understand the recruitment and activation of inflammatory cells due to glycocalyx damage. We found that HP-PRRSV infection promoted neutrophil migration across the MVEC monolayer (P < 0.01), which was significantly inhibited by the APS intervention (P < 0.01), and that the partial degradation of the glycocalyx by HPA III not only further increased the HP-PRRSV-induced increase in neutrophil migration rate but also significantly weakened the inhibitory effect of APS on HP-PRRSV-induced neutrophil migration (Fig. 7A). After treating E. coli with neutrophils that had migrated through the MVECs monolayer, the number of bacterial colonies in each group was similar to the migration rate results (fig. 7b), indicating that the bactericidal activity of neutrophils in each group is inversely correlated with their migration rates. In other words, the glycocalyx disruption reduced the bactericidal activity of neutrophils that migrated across the endothelial monolayer, while APS could restore the bactericidal activity of neutrophils by improving the endothelial glycocalyx.
Fig. 7. Effects of astragalus polysaccharide on neutrophil migration across the monolayer of porcine pulmonary microvascular endothelial cells and neutrophil bactericidal activities during HP-PRRSV infection. (A) Neutrophil migration rate across the monolayer of porcine pulmonary MVECs. (B) Bacterial colonies grown after treatment with the migrated neutrophil across the monolayer of porcine pulmonary MVECs. HP-PRRSV infection, HP-PRRSV infection. “**” indicates P < 0.01. DiscussionMVECs not only serve as a barrier between the blood and tissues but also produce inflammatory factors and antiviral proteins that play essential roles in immune and inflammatory responses, with the glycocalyx being a key player. Thus, glycocalyx is a potential therapeutic target for mitigating antiviral damage (Zha et al., 2022). In this study, we used an in vitro model of HP-PRRSV-infected porcine pulmonary MVECs and discovered that APS has a significant regulatory effect on the barrier function of MVECs, the expression of adhesion molecules, and the mediation of neutrophil trans-endothelial migration and bactericidal functions disrupted by HP-PRRSV infection. Furthermore, APS inhibited the replication of HP-PRRSV within porcine pulmonary MVECs; however, glycocalyx degradation significantly weakened the effects of APS. As a natural biological barrier, the glycocalyx’s selective permeability is crucial for maintaining homeostasis between microvessels and tissues. Glycocalyx damage caused by viral infection increases microvascular permeability, leading to the leakage of solutes, plasma proteins, and fluids, which in turn causes inflammation and edema (Wang et al., 2024; Zhan et al., 2025). Proteoglycans and their attached glycosaminoglycan chains are major components of the glycocalyx, and changes in their composition can affect the glycocalyx’s barrier function. For example, the plasma levels of SDC-1, hyaluronan, and heparan sulfate were significantly higher in patients with severe H1N1 influenza, which has been demonstrated in infections of several respiratory viruses, such as the influenza virus (Huang et al., 2022). In this study, HSPG-2, GPC-1, and SDC-1 were selected as representative markers of the basement membrane, GPI-anchored, and transmembrane proteoglycan families, respectively, and constitute the most recognized endothelial glycocalyx research indicators. The determination of the three glycan ligands was based on previous results from the lectin chip (Song et al., 2022). Our results showed significant changes in the expression of proteoglycans HSPG-2, GPC-1, SDC-1, and the LCA, DSA, and PHA-E-reactive glycan ligands, indicating that HP-PRRSV disrupts the glycocalyx components of porcine pulmonary MVECs. SDC-1 shedding from the surface of pulmonary MVECs may be the reason for its elevated plasma level. The impact of PRRSV infection on cell surface monosaccharides has been reported for the first time. TEER measurements and HRP-SA permeability assays further indicated that the barrier function of porcine pulmonary MVECs was compromised. APS treatment significantly reduced the changes in glycocalyx components caused by HP-PRRSV and markedly restored MVEC monolayer barrier function. Glycoproteins are another important glycocalyx component. Carbohydrate chains play a crucial role in their function, although they primarily exhibit the characteristics of proteins due to their generally lower carbohydrate content compared to proteoglycans (Sprovieri and Martino, 2018). The adhesion molecules VCAM-1 and ICAM-1 are glycoprotein components of the endothelial glycocalyx that regulate leukocyte adhesion and trans-endothelial migration through MVECs and play essential roles in inflammatory responses (Singh et al., 2023). Considering that interstitial pneumonia is the primary pathological change in piglets infected with HP-PRRSV, we investigated VCAM-1 and ICAM-1 expression in porcine pulmonary MVECs during HP-PRRSV infection. Our results showed that glycocalyx degradation promoted the upregulation of VCAM-1 and ICAM-1 induced by HP-PRRSV and facilitated neutrophil trans-endothelial migration, while also inhibiting the regulatory effects of APS on these processes. PRRSV infection in piglets leads to neutrophil infiltration in the lungs (Yuan et al., 2022a,b), and the increased trans-endothelial migration of neutrophils by glycocalyx disruption in our research supports this finding. However, the bactericidal ability of neutrophils undergoing trans-endothelial migration was significantly reduced. This indicates that the integrity of the glycocalyx influences not only neutrophil recruitment but also their bactericidal function, potentially contributing to adverse outcomes. Previous studies have shown that piglets infected with HP-PRRSV frequently suffer from secondary bacterial infections (Xiangjinyan et al., 2020). If the increased number of neutrophils entering the lung tissue is unable to effectively eliminate the bacteria responsible for secondary infections, but instead releases a large number of inflammatory factors, a severe inflammatory response will occur. Our findings may partially explain the causes of severe lung injury resulting from secondary bacterial infection. Therefore, protecting the integrity and function of the endothelial glycocalyx is crucial not only for preventing pathological damage caused by HP-PRRSV but also for controlling secondary bacterial infections-induced severe lung damage. The critical role of the pulmonary MVEC glycocalyx in the pathogenesis of several respiratory viruses has been well documented. The shedding of the hyaluronan component of the endothelial glycocalyx significantly increases in the acute respiratory distress syndrome after flu syndrome and is considered to be positively correlated with the 28-day mortality rate and elevated neutrophils in the alveolar septum (Benatti et al., 2020). In SARS-CoV-2 infection, the intact endothelial glycocalyx prevents viruses from invading MVECs, while the virus spike protein binds with ACE2 and enters MVECs with an incomplete glycocalyx for replication (Lv et al., 2023). However, porcine pulmonary MVECs have not yet been widely recognized as primary target cells for HP-PRRSV, and studies on its pathogenic mechanism and therapeutic strategies from the endothelial glycocalyx perspective are rare. This study reveals the glycocalyx damage caused by HP-PRRSV in porcine pulmonary MVECs, which not only increases the permeability of the pulmonary microvascular endothelial barrier but also enhances the infiltration of neutrophils with weakened bactericidal function, thereby enriching our understanding of its pathogenic mechanisms. APS, a major component of astragalus membranaceus, a traditional Chinese medicine, has significant antiviral activity. Traditional views suggest that its antiviral mechanisms include protecting cells and tissues, enhancing immunity, anti-inflammatory effects, antioxidant properties (Zheng et al., 2020), and inhibiting viral replication (Lee et al., 2023). In recent years, endothelial cells have increasingly become important target cells of APS, which show significant regulatory effects on endothelial dysfunction, such as promoting endothelial cell migration and tube formation (Zhang et al., 2022) and swine fever virus-induced upregulated cytokines associated with immune responses (Zhuge et al., 2017). Several studies have investigated the effects of APS on HP-PRRSV infection, including improving the survival rate of infected pigs (Song and W, 2021), reducing oxidative stress and apoptosis induced by PRRSV, and protecting porcine alveolar macrophages (Tuo et al., 2024). Unlike these studies, our research focuses on the glycocalyx in MVECs and demonstrates that APS inhibits HP-PRRSV-induced endothelial dysfunction by protecting the glycocalyx. Protecting and restoring damaged glycocalyx in pulmonary MVECs is closely related to improved survival rates in patients with COVID-19 and severe influenza (Benatti et al., 2020; Machin et al., 2023). Therefore, we believe that APS protection of the glycocalyx in porcine pulmonary MVECs could also be a potential therapeutic strategy for HP-PRRSV infection, effectively reducing the severity of lung injury and controlling possible secondary bacterial infections. Our previous in vivo study showed that polysaccharides from Pulsatilla Decoction can boost the glycocalyx in intestinal MVECs in mice (Zhang et al., 2014). However, we have not yet investigated how HP-PRRSV damages the endothelial glycocalyx or how APS affects it in vivo. Thus, further in vivo studies are required to validate the protective role of APS against PRRSV-induced injury in porcine pulmonary MVECs and the glycocalyx’s pivotal role in this process. We targeted the glycocalyx to investigate the mechanism of APS creatively based on the commonalities of their structures and functions. The glycoproteins and proteoglycans of the glycocalyx contain abundant polysaccharide chains, and APS is a polysaccharide component extracted from Astragalus. The glycocalyx on the surface of MVECs protects them from pathogen invasion and participates in immune responses by regulating immune cell recognition and adhesion. Similarly, APS exerts broad antioxidant, anti-inflammatory, and immunomodulatory effects, protecting tissue cells from external damage and regulating immune responses. We believe that just as exogenous calcium supplementation can address calcium deficiency in the body, providing exogenous polysaccharides should also help improve virus-induced damage to the endothelial glycocalyx, which has been demonstrated in several studies. Fucoidan restored the endothelial glycocalyx and protected against dysfunction induced by serum of COVID-19 patients (Yuan et al., 2022a,b). Sulodexide, a heparin sulfate-like compound, accelerated endothelial glycocalyx regeneration in sepsis in vitro and in vivo and improved animal survival (Song et al., 2017). Rhamnan sulfate improves the glycocalyx to enhance endothelial barrier function and reduce vascular inflammation (Patil et al., 2022). However, APS has not yet been used to target the endothelial glycocalyx to counteract viral infection. In this study, APS was used primarily based on its status as the most intensively investigated and clinically utilized polysaccharide derived from Chinese herbs for immunomodulatory, anti-inflammatory, antiviral, and antitumor applications. Nevertheless, Chinese herbal polysaccharides are heteropolysaccharides with complicated structures, whose bioactivities are influenced by multiple factors, including molecular weight, glycosidic linkage, and monosaccharide composition. Owing to the absence of parallel controls involving other polysaccharides or monosaccharides in the experimental design, this study cannot conclusively determine whether similar bioactivities would be observed with other polysaccharides. Future research will focus on comparative investigations of additional polysaccharides alongside APS to elucidate the structure-activity relationships underlying their functions. This study aims to establish a foundational framework for the precise modulation of glycocalyx function. Owing to the relatively slow progress in glycobiology research, studying the endothelial glycocalyx mechanism remains challenging. In addition to acting through known signaling pathways, such as Rho/ROCK and TLRs/NF κB/TNF-α, we speculate that APS might also directly bind to certain glycocalyx components, thereby altering the function of these proteoglycans or glycoproteins. Further research is needed to clarify the mechanisms by which APS protects and regulates the endothelial glycocalyx. ConclusionThis study provides compelling evidence that glycocalyx disruption is a critical pathogenic event that orchestrates pulmonary microvascular endothelial dysfunction during HP-PRRSV infection. More importantly, we delineated a novel mechanism by which APS exerts its therapeutic effects. APS significantly restores glycocalyx components and alleviates PRRSV-induced microvascular endothelial dysfunction in a glycocalyx-dependent manner. AcknowledgmentWe sincerely thank Dr. Zhanzhong Zhao from the Chinese Academy of Agricultural Sciences for providing the PRRSV strain JXA1 and gratefully acknowledge the support from our respective institutions. FundingThe work was financially supported by the National Natural Science Foundation of China (No. 32273050) and the Beijing Natural Science Foundation (No. 6242008). Authors’ contributionStudy design and planning: Tao Zhang and Nuohan Xia. Experiments conducting and data processing, and analysis: Jian Sun, Nuohan Xia. Drafting of the manuscript: Jian Sun and Tao Zhang; Method development and experimental supervision: Qian Zhang and Ge Hu. All authors have read, edited, and approved the manuscript. Conflicts of interestThe authors declare no conflicts of interest. Data availabilityThe data that support the findings of this study are available from the corresponding author upon reasonable request. ReferencesBenatti, M.N., Fabro, A.T. and Miranda, C.H. 2020. Endothelial glycocalyx shedding in the acute respiratory distress syndrome after flu syndrome. J. Intensive Care 8, 72. Cao, R.N., Tang, L., Xia, Z.Y. and Xia, R. 2019. Endothelial glycocalyx as a potential therapeutic target in organ injuries. Chin. Med. J. 132, 963–975. Chen, H.R. and Yeh, T.M. 2017. In vitro assays for measuring endothelial permeability by transwells and electrical impedance systems. Bio. Protoc. 7, 2273. Huang, X., Lu, F., Tian, H., Hu, H., Ning, F., Shang, Q., Hao, D., Zhu, W., Kong, G., Ma, X., Feng, J., Wang, T. and Wang, X. 2022. Association between plasma glycocalyx component levels and poor prognosis in severe influenza type A (H1N1). Sci. Rep. 12, 163. Jedlicka, J., Becker, B.F. and Chappell, D. 2020. Endothelial glycocalyx. Crit. Care Clin. 36, 217–232. <AQ4> Jiang, D., Tu, T., Zhou, Y., Li, Y., Luo, Y., Yao, X., Yang, Z., Ren, M. and Wang, Y. 2023. Epidemiological investigation and pathogenicity of porcine reproductive and respiratory syndrome virus in Sichuan, China. Front. Microbiol. 14, 1241354. Lee, C.Y., Nguyen, A.T., Doan, L.H., Chu, L.W., Chang, C.H., Liu, H.K., Lee, I.L., Wang, T.H., Lai, J.M., Tsao, S.M., Liao, H.J., Ping, Y.H. and Huang, C.Y.F. 2023. Repurposing astragalus polysaccharide PG2 for inhibiting ACE2 and SARS-CoV-2 spike syncytial formation and anti-inflammatory effects. Viruses 15, 641. Lei, C., Yang, J., Hu, J. and Sun, X. 2021. On the calculation of TCID50 for quantitation of virus infectivity. Virol. Sin. 36, 141–144. Li, P., Yang, Z., Ma, S., Hu, G., Dong, H. and Zhang, T. 2020. Susceptibility of porcine pulmonary microvascular endothelial cells to porcine reproductive and respiratory syndrome virus. J. Vet. Med. Sci. 82, 1404–1409. Liu, H.Q., Li, J., Xuan, C.L. and Ma, H.C. 2020. A review on the physiological and pathophysiological role of endothelial glycocalyx. J. Biochem. Mol. Toxicol. 34, e22571. Lv, B., Huang, S., Huang, H., Niu, N. and Liu, J. 2023. Endothelial glycocalyx injury in SARS-CoV-2 infection: molecular mechanisms and potential targeted therapy. Mediators Inflamm. 2023, 6685251. Machin, D.R., Sabouri, M., Zheng, X. and Donato, A.J. 2023. Therapeutic strategies targeting the endothelial glycocalyx. Curr. Opin. Clin. Nutr. Metab. Care 26, 543–550. Osorio-Valencia, S. and Zhou, B. 2024. Roles of macrophages and endothelial cells and their crosstalk in acute lung injury. Biomedicines 12, 632. Patil, N.P., Gómez-Hernández, A., Zhang, F., Cancel, L., Feng, X., Yan, L., Xia, K., Takematsu, E., Yang, E.Y., Le, V., Fisher, M.E., Gonzalez-Rodriguez, A., Garcia-Monzon, C., Tunnell, J., Tarbell, J., Linhardt, R.J. and Baker, A.B. 2022. Rhamnan sulfate reduces atherosclerotic plaque formation and vascular inflammation. Biomaterials 291, 121865. Singh, V., Kaur, R., Kumari, P., Pasricha, C. and Singh, R. 2023. ICAM-1 and VCAM-1: gatekeepers in various inflammatory and cardiovascular disorders. Clin. Chim. Acta. 548, 117487. Song, J.W., Zullo, J.A., Liveris, D., Dragovich, M., Zhang, X.F. and Goligorsky, M.S. 2017. Therapeutic restoration of endothelial glycocalyx in sepsis. J. Pharmacol. Exp. Ther. 361, 115–121. Song, W. 2021. Diagnosis and control of porcine reproductive and respiratory syndrome in a pig farm in hunan province. Master’s thesis, Agricultural University of Hunan, China. Song, X., Wu, Y., Wu, X., Hu, G. and Zhang, T. 2022. Effects of highly pathogenic porcine reproductive and respiratory syndrome virus infection on the surface glycoprofiling of porcine pulmonary microvascular endothelial cells. Viruses 14, 2569. Sprovieri, P. and Martino, G. 2018. The role of the carbohydrates in plasmatic membrane. Physiol. Res. 67, 1–11. Sun, W., Wu, W., Jiang, N., Ge, X., Zhang, Y., Han, J., Guo, X., Zhou, L. and Yang, H. 2022. Highly pathogenic PRRSV-infected alveolar macrophages impair the function of pulmonary microvascular endothelial cells. Viruses 14, 452. Taghavi, S., Abdullah, S., Shaheen, F., Mueller, L., Gagen, B., Duchesne, J., Steele, C., Pociask, D., Kolls, J. and Jackson-Weaver, O. 2022. Glycocalyx degradation and the endotheliopathy of viral infection. PLos One. 17, 276232. Tuo, T., Chen, D., Wang, L., Zhang, Y., Zhou, L., Ge, X., Han, J., Guo, X. and Yang, H. 2024. Infection of PRRSV inhibits CSFV C-strain replication by inducing macrophages polarization to M1. Vet. Microbiol. 289, 109957. Wang, D., Wang, K., Liu, Q., Liu, M., Zhang, G., Feng, K., Wang, K., Ding, X., Zhu, H., Yang, S., Liu, Y., Li, T., Gong, P., Wang, M., Wang, P.G., Jin, H., Zhao, W. and Yu, F. 2024. A novel drug candidate for sepsis targeting heparanase by inhibiting cytokine storm. Adv. Sci. (Weinh), 11, e2403337. Wang, H.M., Liu, Y.G., Tang, Y.D., Liu, T.X., Zheng, L.L., Wang, T.Y., Liu, S.G., Wang, G. and Cai, X.H. 2018. A natural recombinant PRRSV between HP-PRRSV JXA1-like and NADC30-like strains. Transbound. Emerg. Dis. 65, 10780–11086. Weinbaum, S., Cancel, L.M., Fu, B.M. and Tarbell, J.M. 2021. The glycocalyx and its role in vascular physiology and vascular related diseases. Cardiovasc. Eng. Technol. 12, 37–71. Wu, Y., Song, X., Li, P., Wang, Z., Zhao, Z. and Zhang, T. 2023. Highly pathogenic porcine reproductive and respiratory syndrome virus-induced inflammatory response in porcine pulmonary microvascular endothelial cells and effects of herbal ingredients on main inflammatory molecules. Int. Immunopharmacol. 118, 110012. Xiangjinyan., Zeng, J., Li, X., Zhang, Z., Din, A.U., Zhao, K. and Zhou, Y. 2020. High incidence and characteristic of PRRSV and resistant bacterial Co-Infection in pig farms. Microb. Pathog. 149, 104536. Yamaoka-Tojo, M. 2020. Vascular endothelial glycocalyx damage in COVID-19. Int. J. Mol. Sci. 21, 9712. Yuan, L., Cheng, S., Sol, W.M.P.J., Van Der Velden, A.I.M., Vink, H., Rabelink, T.J. and Van Den Berg, B.M. 2022. Heparan sulfate mimetic fucoidan restores the endothelial glycocalyx and protects against dysfunction induced by serum of COVID-19 patients in the intensive care unit. ERJ. Open Res. 8, 652. Yuan, L., Zhu, Z., Fan, J., Liu, P., Li, Y., Li, Q., Sun, Z., Yu, X., Lee, H.S., Tian, K. and Li, X. 2022. High pathogenicity of a Chinese NADC34-like PRRSV on Pigs. Microbiol. Spectr. 10, 154122. Zha, D., Fu, M. and Qian, Y. 2022. Vascular endothelial glycocalyx damage and potential targeted therapy in COVID-19. Cells 11, 1972. Zhan, J.H., Wei, J., Liu, Y.J., Wang, P.X. and Zhu, X.Y. 2025. Sepsis-associated endothelial glycocalyx damage: a review of animal models, clinical evidence, and molecular mechanisms. Int. J. Bio. Macromol. 295, 139548. Zhang, G., Huang, J., Hao, S., Zhang, J. and Zhou, N. 2022. Radix astragalus polysaccharide accelerates angiogenesis by activating AKT/eNOS to promote nerve regeneration and functional recovery. Front. Pharmacol. 13, 838647. Zhang, T., Wu, S., Sun, X., Duan, H.Q. and Mu, X. 2014. Effects of polysaccharides from pulsatilla decoction on the microvascular endothelial glycocalyx. J. Integr. Agric. 13, 2558–2561. Zhang, X., Sun, D., Song, J.W., Zullo, J., Lipphardt, M., Coneh-Gould, L. and Goligorsky, M.S. 2018. Endothelial cell dysfunction and glycocalyx - A vicious circle. Matrix. Biol. 71-72(72), 421–431. Zhang, Z., Sun, X., Wang, Z., Wang, Q. and Zhang, T. 2021. Effects of the endothelial glycocalyx injury on regulating bioactive compound secretion of microvascular endothelial cells regulated by astragalus polysaccharide. J. Beijing. Univ. Agr. 36, 78–82. Zheng, Y., Ren, W., Zhang, L., Zhang, Y., Liu, D. and Liu, Y. 2020. A review of the pharmacological action of astragalus polysaccharide. Front. Pharmacol. 11, 349. Zhou, Q., Meng, G., Teng, F., Sun, Q. and Zhang, Y. 2018. Effects of astragalus polysaccharide on apoptosis of myocardial microvascular endothelial cells in rats undergoing hypoxia/reoxygenation by mediation of the PI3K/Akt/eNOS signaling pathway. J. Cell. Biochem. 119, 806–816. Zhuge, Z., Dong, Y., Li, L. and Jin, T. 2017. Effects of astragalus polysaccharide on the adhesion-related immune response of endothelial cells stimulated with CSFV in vitro. PeerJ 5, e3862. Supplementary Material
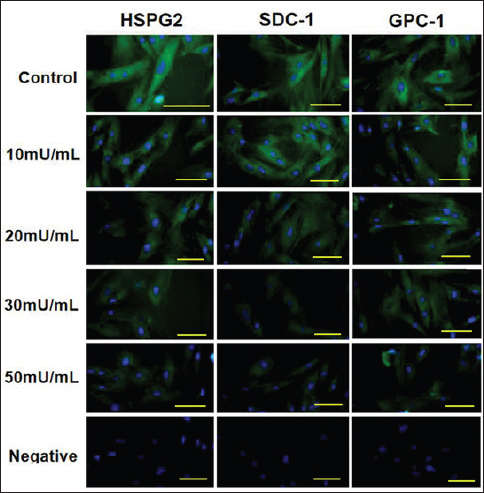
Supplementary Fig. 1. Immunofluorescence staining of proteoglycans HSPG-2, SDC-1, and GPC-1 in porcine pulmonary microvascular endothelial cells treated with heparinase (HPA) III at different concentrations. Microvascular endothelial cells were treated with 0, 10, 20, 30, and 50 mU/ml HPA III for 2 hours, then incubated in fresh medium for a further 6 hours, fixed with 4 % paraformaldehyde, and processed for immunofluorescence. Negative cells without HPA III treatment were incubated with PBS instead of the primary proteoglycan antibody. Bar=40 μm. | ||
| How to Cite this Article |
| Pubmed Style Xia N, Sun J, Hu G, Zhang Q, Zhang T. Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Vet. J.. 2026; 16(1): 577-588. doi:10.5455/OVJ.2026.v16.i1.54 Web Style Xia N, Sun J, Hu G, Zhang Q, Zhang T. Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. https://www.openveterinaryjournal.com/?mno=274537 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.54 AMA (American Medical Association) Style Xia N, Sun J, Hu G, Zhang Q, Zhang T. Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Vet. J.. 2026; 16(1): 577-588. doi:10.5455/OVJ.2026.v16.i1.54 Vancouver/ICMJE Style Xia N, Sun J, Hu G, Zhang Q, Zhang T. Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 577-588. doi:10.5455/OVJ.2026.v16.i1.54 Harvard Style Xia, N., Sun, . J., Hu, . G., Zhang, . Q. & Zhang, . T. (2026) Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Vet. J., 16 (1), 577-588. doi:10.5455/OVJ.2026.v16.i1.54 Turabian Style Xia, Nuohan, Jian Sun, Ge Hu, Qian Zhang, and Tao Zhang. 2026. Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Veterinary Journal, 16 (1), 577-588. doi:10.5455/OVJ.2026.v16.i1.54 Chicago Style Xia, Nuohan, Jian Sun, Ge Hu, Qian Zhang, and Tao Zhang. "Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy." Open Veterinary Journal 16 (2026), 577-588. doi:10.5455/OVJ.2026.v16.i1.54 MLA (The Modern Language Association) Style Xia, Nuohan, Jian Sun, Ge Hu, Qian Zhang, and Tao Zhang. "Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy." Open Veterinary Journal 16.1 (2026), 577-588. Print. doi:10.5455/OVJ.2026.v16.i1.54 APA (American Psychological Association) Style Xia, N., Sun, . J., Hu, . G., Zhang, . Q. & Zhang, . T. (2026) Glycocalyx disruption mediates HP-PRRSV-induced microvascular endothelial dysfunction and underlies astragalus polysaccharide’s antiviral efficacy. Open Veterinary Journal, 16 (1), 577-588. doi:10.5455/OVJ.2026.v16.i1.54 |