| Research Article | ||
Open Vet. J.. 2025; 15(12): 6461-6469 Open Veterinary Journal, (2025), Vol. 15(12): 6461-6469 Research Article Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessmentsMuhammad Yasir Shah1, Shakeeb Ullah1,2*, Ali Zaman1, Faiqah Ramzan1 and Saifur Rehman21Faculty of Veterinary and Animal Sciences, Gomal University Dera Ismail Khan, Pakistan 2Faculty of Veterinary Sciences, University of Veterinary and Animal Sciences, Swat, Pakistan *Corresponding Author: Shakeeb Ullah, Faculty of Veterinary and Animal Sciences, Gomal University Dera Ismail Khan, Pakistan. Email: shakeebullah [at] gmail.com Submitted: 31/07/2025 Revised: 22/10/2025 Accepted: 05/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
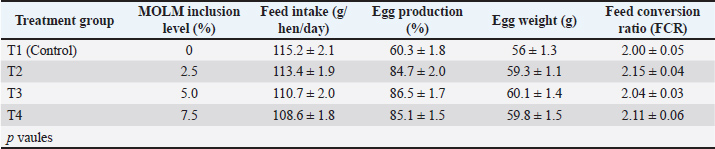
AbstractBackground: Moringa oleifera is a nutrient-rich plant widely explored for its potential as a natural feed additive in poultry due to its antioxidant, antimicrobial, and immunomodulatory properties. Incorporating Moringa leaf meal into laying hen diets may influence feed efficiency (FCR), egg quality, and immune-metabolic responses, warranting comprehensive in vivo and ex vivo investigations. Aim: This study investigated the impact of incorporating M. oleifera leaf meal (MOLM) into the diets of laying hens on egg productivity, egg characteristics, antioxidant capacity, immune response, and tissue morphology. Methods: Over an 8-week period, 40 30-week-old Hy-Line Brown layers were randomly distributed into four dietary groups (10 birds per group) and received nutritionally balanced diets with MOLM supplementation levels of 0%, 2.5%, 5.0%, or 7.5%. The key performance indicators, including feed intake, egg production, egg weight, and FCR, were recorded weekly. Egg quality traits and antioxidant parameters in liver and intestinal tissues were assessed along with pro-inflammatory cytokine levels and histopathological scoring. Results: Inclusion of 7.5% MOLM significantly improved egg production (86.5%), egg weight (60.1 g ± desert), and FCR (2.04), compared with the control group (p < 0.05). Egg quality traits, including shell thickness, yolk pigmentation, Haugh unit, and albumen height, were enhanced in the MOLM-supplemented groups, particularly at 7.5.0% inclusion. A marked increase (p < 0.01) in the activity of key antioxidant enzymes (superoxide dismutase, catalase, and glutathione peroxidase) was observed in liver and intestinal tissues, accompanied by a decline in malondialdehyde levels, reflecting an improved oxidative state. Furthermore, MOLM supplementation resulted in a dose-dependent decrease in the levels of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α). The greatest reduction was observed at the 5.0% inclusion level. Histopathological evaluations corroborated these findings, revealing significantly reduced inflammation and oxidative damage scores, particularly in the group with 7.5% MOLM. Conclusion: Incorporating MOLM into the diet significantly improved laying hens’ performance, enhanced egg quality, boosted antioxidant capacity, and positively influenced immune responses. These findings highlight the value of MOLM as a natural phytogenic additive with potential benefits for enhancing poultry productivity and overall health. Keywords: Moringa oleifera leaf meal, Laying hens, Antioxidant enzymes, Cytokines, Immune modulation. IntroductionThe egg industry plays a vital role in supplying high-quality animal protein, and poultry production is a key component of global food security (Bebas et al., 2023). However, rising feed costs and increasing concerns about antibiotic residues and synthetic feed additives have prompted the poultry industry to explore sustainable, natural alternatives (Abou-Elkhair et al., 2014; Yamin et al., 2025). Plant-based supplements with functional bioactive properties offer a promising solution to enhance productivity, feed efficiency (FCR), and safeguard poultry health (Abou-Elkhair et al., 2014; Tufarelli et al., 2016). Moringa oleifera, a rapidly growing tree indigenous to South Asia and Africa, has attracted considerable interest as a natural feed supplement in animal nutrition (Ike et al., 2025). Its leaves are rich in vitamins (A, C, and E), minerals, essential amino acids, flavonoids, and phenolic compounds (Rivas et al., 2024), which collectively contribute to its antioxidant, anti-inflammatory, antimicrobial, and immunomodulatory activities (Leone et al., 2015; Gopalakrishnan et al., 2016; Abdelkader et al., 2025). These properties make MOLM a strong candidate for enhancing poultry growth performance, immune resilience, and egg quality parameters (Pebas et al., 2023). Moringa oleifera leaves are a nutrient-dense feed component, containing over 25% crude protein, essential fatty acids, vitamins, and potent antioxidants such as quercetin, kaempferol, and chlorogenic acid (Leone et al., 2015). These bioactive compounds are known to modulate cellular redox status and inflammatory pathways, which are critical for maintaining laying hen health and productivity (Verma et al., 2020). MOLM supplementation can improve laying performance, egg weight, shell thickness, and yolk color due to its high carotenoid and micronutrient content (Lu et al., 2016; Ayodele et al., 2020). FCR has also been reported to improve with the inclusion of MOLM, particularly at supplementation levels ranging from 1% to 5% (Nkukwana et al., 2014). In poultry, oxidative stress and inflammation pose significant physiological challenges, often leading to reduced performance and productivity (Celeste et al., 2011; Al-Suwailem et al., 2024). MOLM, rich in phenolic and flavonoid compounds, has demonstrated strong antioxidant potential by enhancing the activity of vital enzymes such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) within poultry tissues (Akinyemi et al., 2017). Ex vivo studies have depicted that MOLM suppresses the production of critical inflammatory mediators, including TNF-α, IL-6, and IL-1β (Shedeed et al., 2022), indicating its immunomodulatory potential. While in vivo performance studies help determine practical feed benefits, ex vivo assays provide mechanistic insights into cellular and tissue-level responses (Gul et al., 2024). The combined approach allows for a more robust understanding of how MOLM supplementation modulates metabolic, oxidative, and immunological pathways in laying hens (Abdel-Moneim et al., 2020). The current study aimed to determine the in vivo and ex vivo effects of MOLM on FCR, egg production traits, and tissue-level antioxidant and inflammatory markers in laying hens (Sooksawat et al., 2024). By integrating both performance and physiological outcomes, this study offers a comprehensive understanding of MOLM as a functional dietary intervention in modern poultry systems. The novelty of this study lies in its integrated evaluation of M. oleifera leaf meal (MOLM) supplementation on laying hens by simultaneously investigating FCR, egg quality traits, and immunometabolic responses through both in vivo and ex vivo assessments. While previous research has primarily focused on production performance or egg traits in isolation, our work uniquely combines nutritional, reproductive, and immune-metabolic perspectives to provide a holistic understanding of Moringa’s potential as a functional feed additive (Haryati et al., 2025). Moringa oleifera is chosen based on its exceptional nutritional and phytochemical profile. It is rich in proteins, essential amino acids, vitamins, minerals, and bioactive compounds with antioxidant and immunomodulatory properties, making it a promising natural alternative to synthetic feed additives and antibiotics. The novel aspect of our study compared to earlier works is the dual approach: not only assessing direct effects on feed conversion and egg production under practical conditions, but also employing ex vivo analyses to explore the underlying immunometabolic mechanisms. This comprehensive approach provides new insights into how Moringa simultaneously influences metabolic pathways, immune competence, and egg quality, positioning the study as a significant step forward in the development of sustainable and health-promoting poultry nutrition strategies. Materials and MethodsPreparation of MOLMFresh M. oleifera leaves were harvested from locally available trees, thoroughly washed with clean water to remove dust and impurities, and then air-dried under ambient temperature to preserve their nutritional and bioactive components. Once completely dried, the leaves were ground into a fine powder using a hammer mill and stored in airtight containers until incorporation into the experimental diets. Dried leaf meal was used for dietary supplementation. Experimental design and the birdsIn this study, 40 healthy 30-week-old Hy-Line Brown laying hens were randomly assigned to four dietary treatment groups with 10 birds per group. At the beginning of the trial, the hens had an average body weight of 1.55 ± 0.12 kg, indicating uniformity in the experimental stock. The narrow SD reflected minimal variation among birds, ensuring comparability across treatment groups. The hens were housed in environmentally controlled cages under standardized management conditions (temperature of 22°C–25°C, relative humidity of 55%–65%, and photoperiod of 16 hours light and 8 hours dark) with unrestricted access to feed and water for the duration of the experiment. Dietary treatmentsThe experimental diets were isonitrogenous and isoenergetic and were formulated based on the NRC (1994) nutrient recommendations for layers. The experimental groups received the following treatments: T1 (Control): Basal diet (Maize (corn) 58.00, soybean meal (44% CP) 22.00, wheat bran 5.00, fish meal (60% CP) 3.00, dicalcium phosphate 1.80, Limestone (CaCO3) 8.50, vegetable oil 1.50, salt (NaCl) 0.30) without MOLM T2: Basal diet + 2.5% MOLM T3: Basal diet + 5.0% MOLM T4: Basal diet + 7.5% MOLM Data collectionPerformance parametersFeed intake (g/hen/day): Measured weekly by recording the difference between the feed offered and the feed refused. Egg production (%): calculated daily as hen-day egg production. Egg weight (g): measured using a digital balance. Feed conversion ratio (FCR): calculated as feed intake (g)/egg mass (g). Egg quality analysisAt the end of the trial, five eggs from each replicate were randomly selected for analysis. Shell thickness (mm) was measured at three different points on each egg using a digital micrometer, and the average of these measurements was recorded. The Roche Yolk Color Fan was used to assess yolk color, which scores yolk pigmentation on a scale from 1 to 15. Albumen height (mm) was measured using a tripod micrometer, and the Haugh unit, an indicator of egg quality, was calculated based on the albumen height and egg weight. Ex vivo tissue collection and analysisFollowing the experiment, tissue samples were collected from two birds per replicate after euthanasia via cervical dislocation. Antioxidant enzyme assaysLiver and intestinal tissues were excised, rinsed in phosphate-buffered saline, homogenized, and centrifuged at 10,000×g for 10 minutes at 4°C to obtain the supernatants. Subsequently, these were used to determine oxidative stress and antioxidant enzyme activities. The activity of SOD was assayed according to the method of Marklund and Marklund (1974), while that of catalase (CAT) was measured following the procedure described by Aebi (1984) (Akbar et al., 2024). Glutathione peroxidase (GPx) activity was determined using the method described by Paglia and Valentine (1967). Lipid peroxidation was evaluated by estimating malondialdehyde (MDA) levels through thiobarbituric acid reactive substances measurement. Pro-inflammatory cytokinesLiver and intestinal homogenates were analyzed for the inflammatory cytokines Interleukin-1β (IL-1β), Interleukin-6 (IL-6), and Tumor Necrosis Factor-alpha (TNF-α). Quantification of these biomarkers was performed using commercial enzyme-linked immunosorbent assay kits (MyBioSource, USA) following the manufacturer’s instructions. Histopathological evaluationLiver and intestinal tissue samples were fixed in 10% buffered formalin and embedded in paraffin according to the manufacturer’s protocol. Paraffin blocks were sectioned at a thickness of 5 µm and stained with hematoxylin and eosin (H&E) for histopathological examination. The stained slides were examined under a light microscope (Olympus BX53, Tokyo, Japan) equipped with a digital camera system. Images were captured and analyzed using ImageJ software (NIH, Bethesda, MD, USA). Microscopic evaluation was performed at different magnifications (typically 10×, 20×, and 40× objectives) to assess general morphology and specific pathological alterations. An unbiased veterinary pathologist, blinded to the experimental groups, scored the microscopic tissue changes (Bancroft and Gamble, 1998). Histopathological alterations in the liver and intestinal tissues were assessed using a semiquantitative scoring system adapted from previously published protocols (Suvarna et al., 2018). The two main parameters were evaluated: inflammation and oxidative damage, each scored on a 0–3 scale (0=none, 1=mild, 2=moderate, and 3=severe). The degree of leukocyte infiltration, vascular congestion, and presence of necrotic foci were used to assess inflammation. Oxidative damage was evaluated by observing cytoplasmic vacuolization, nuclear pyknosis, loss of cellular integrity, and tissue degeneration related to lipid peroxidation. Specific features, such as hepatocellular degeneration, necrosis, sinusoidal dilatation, and periportal inflammatory cell infiltration, were scored for liver sections. Villi architecture, mucosal erosion, epithelial necrosis, and inflammatory cell infiltration in the lamina propria were evaluated in intestinal sections. The scores were calculated by examining at least five random, non-overlapping microscopic fields per section at magnifications of 10×, 20×, and 40×. All evaluations were performed by an unbiased veterinary pathologist, blinded to the experimental groups. Data analysisPrior to analysis, the raw data were checked for normality using the Shapiro–Wilk test and for homogeneity of variances using Levene’s test to ensure compliance with the assumptions of parametric testing. For normally distributed data, a one-way analysis of variance was performed to determine the effect of dietary treatments on the studied parameters. Tukey’s post hoc test was applied for multiple comparisons among treatment means. In cases where data did not meet the assumption of normality, appropriate non-parametric tests (such as the Kruskal–Wallis test followed by Dunn’s multiple comparison test) were considered. The statistical model used for the one-way ANOVA was as follows: Yij=μ + Ti + ϵij where Yij_ is the observation of the jth replicate of the ith treatment, μ is the overall mean, Ti is the effect of the ith treatment, and ϵij is the random error term. Histopathological tissue scores (ordinal data) were analyzed using non-parametric methods, specifically the Kruskal–Wallis test, followed by pairwise comparisons with the Mann–Whitney U test (with Bonferroni correction) to identify differences between groups. All analyses were performed using SPSS statistical software (version 25.0; IBM Corp., Armonk, NY, USA). A p-value < 0.05 was considered statistically significant, whereas p < 0.01 indicated high significance. Data are expressed as mean ± SD. Ethical approvalThe feeding trial lasted for 8 weeks and was conducted in compliance with the ethical standards approved by the Ethical Review Board of Gomal University, Dera Ismail Khan (Approval No. 831/ERB/GU). ResultsImpact of MOLM on egg productivity and feed conversionMOLM inclusion in laying hen diets improved FCR and egg production compared with the control. The 5.0% inclusion level (T3) demonstrated the best FCR (2.04), along with improvements in egg weight and egg output. The inclusion level of 7.5% (T4) also showed enhanced performance, with an FCR of 2.11, which remained better than the control and T2, although not significantly superior to T3. Interestingly, the feed intake decreased at the 7.5% level, a potential threshold beyond which no additional productivity gains were achieved. Overall, 5.0% MOLM can be identified as the optimal level for enhancing laying hen productivity, while higher inclusion (7.5%) maintained improved efficiency without further significant benefits (Table 1). Table 1. Influence of MOLM supplementation on feed conversion ratio and egg production in laying hens (8-week trial).
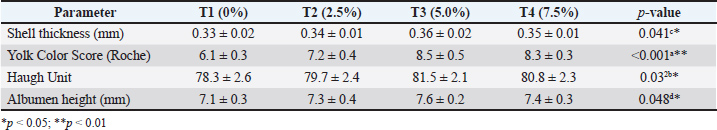
MOLM impact on egg quality traits in laying hensTable 2 shows that shell thickness showed a modest but statistically significant increase, particularly at 5.0% MOLM inclusion (p=0.041), indicating enhanced calcium metabolism or shell gland function. Yolk color was markedly improved, with the highest scores observed at 5.0% and 7.5% inclusion levels (p < 0.001), likely due to the high carotenoid content of MOLM contributing to deeper yolk pigmentation. Similarly, internal egg quality improved as evidenced by significant increases in both Haugh unit and albumen height (p=0.032 and p=0.048, respectively), indicating better albumen viscosity and freshness (Table 2). Table 2. Effect of MOLM supplementation on egg quality traits in laying hens.
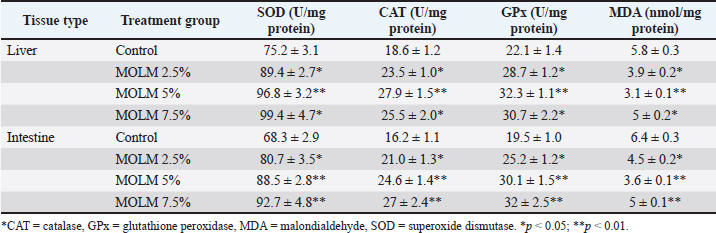
Impact of MOLM on antioxidant enzyme activityEx vivo analysis of antioxidant enzyme activity in liver and intestinal tissues of laying hens revealed a dose-dependent improvement in oxidative status following MOLM supplementation. MOLM administration led to a notable increase in the activity of SOD, CAT, and GPx antioxidant enzymes across all treated groups compared with the control, indicating enhanced enzymatic protection against oxidative stress. Notably, the 5% MOLM group showed the most pronounced effects, with liver SOD, CAT, and GPx levels increasing by approximately 29%, 50%, and 46%, respectively, while MDA levels, a marker of lipid peroxidation, were significantly reduced by 46%, reflecting reduced oxidative damage. A similar trend was observed in intestinal tissues, further supporting the systemic antioxidative role of MOLM. However, a slight elevation in MDA was noted in both tissues at the 7.5% inclusion level, a potential threshold beyond which oxidative balance may not improve or could even decline. These findings underscore the capacity of MOLM to enhance antioxidant defense mechanisms in poultry tissues, particularly at inclusion levels of 2.5%–5% (Table 3). Table 3. Antioxidant enzyme activity in poultry tissues supplemented with MOLM (ex vivo).
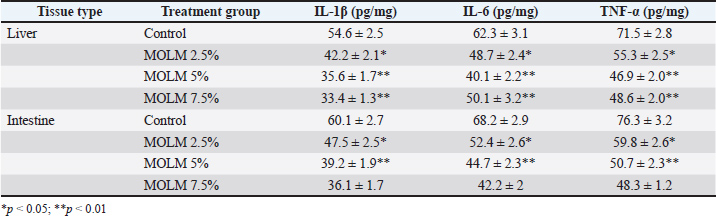
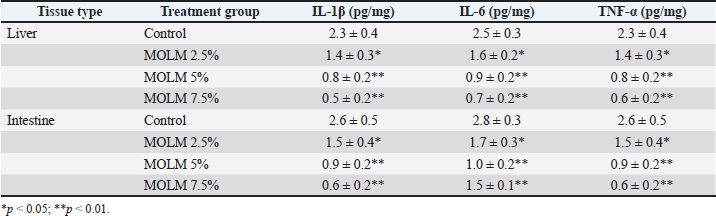
Effect of MOLM on pro-inflammatory cytokine levelsEx vivo analysis showed that MOLM reduced pro-inflammatory cytokine levels (IL-1β, IL-6, and TNF-α) in a dose-dependent manner, with the 5% inclusion level providing the most significant anti-inflammatory effect. Both liver and intestinal tissues exhibited reduced cytokine expression, indicating the strong immunomodulatory potential of MOLM. However, the 7.5% dose showed a slight decline in effectiveness, optimal threshold of 5%. These results highlight the potential of MOLM to enhance gut and liver health in laying hens through its anti-inflammatory properties (Table 4). Table 4. Proinflammatory cytokine levels in poultry tissues supplemented with MOLM (ex vivo).
The impact of MOLM on liver and intestinal tissuesThe histopathological scores of liver and intestinal tissues revealed a clear, dose-dependent improvement in tissue health following MOLM dietary supplementation. The control group exhibited moderate inflammation and oxidative damage (scores > 2.3) in both liver and intestinal samples, indicative of basal inflammatory stress. However, MOLM supplementation at 2.5%, 5%, and 7.5% significantly reduced these scores (p < 0.05, p < 0.01), with the most pronounced effect observed at the 7.5% inclusion level. At the highest dose, liver and intestinal tissues exhibited near-normal histological architecture with minimal inflammatory infiltration and oxidative lesions (scores ≤ 0.7). These findings strongly support the anti-inflammatory and antioxidative potential of MOLM, which is likely mediated through the downregulation of cytokine-driven pathways and the mitigation of tissue-level oxidative stress. This reinforces the utility of MOLM as a functional feed additive for improving gut and hepatic integrity in laying hens (Table 5). Table 5. Histopathological scores of liver and intestinal tissues after MOLM supplementation.
DiscussionThis study evaluated the impact of MOLM supplementation in egg-laying hens by assessing dietary impacts through in vivo performance metrics and examining tissue responses using ex vivo biochemical, immunological, and histological analyses. The results consistently demonstrated that MOLM, particularly at a 5% inclusion level, enhances productive performance, improves egg quality traits, and modulates antioxidant and anti-inflammatory responses in layer poultry. MOLM incorporation into layer diets improved egg characteristics and FCR, with the optimal response observed at the 5% inclusion level (T3). This enhancement may be attributed to the rich nutritional profile of M. oleifera, including its high protein content, essential amino acids, vitamins, and minerals, which support efficient nutrient utilization and reproductive performance (Nkukwana et al., 2014; Gopalakrishnan et al., 2016). The decline in feed intake at the 7.5% level without a corresponding improvement in performance suggests a saturation point or possible negative effects on palatability or nutrient absorption (Abdel-Moneim et al., 2020). Comparable dose-dependent improvements in laying performance associated with MOLM have been documented in previous studies (Olugbemi et al., 2010; Kakati et al., 2015; Khoramabadi et al., 2022). MOLM significantly enhanced egg quality (Amad et al., 2022) traits, including yolk color, albumen height, Haugh unit, and shell thickness. The observed increased yolk pigmentation is most likely due to the high concentration of carotenoids and xanthophylls in Moringa leaves, which are directly deposited into the yolk (Melesse et al., 2011; Leone et al., 2015). Improvements in albumen quality and shell thickness may reflect enhanced metabolic support for egg formation through better mineral utilization, particularly calcium and magnesium, which are abundant in MOLM (Tufarelli et al., 2016; Elwan et al., 2019; Mabrok et al., 2022). These enhancements are of considerable commercial interest, given the consumer preference for richly pigmented yolks and stronger shells. Ex vivo analyses demonstrated a significant increase in the activity of the endogenous antioxidant enzymes SOD, CAT, and GPx in the liver and intestinal tissues of MOLM-treated birds, particularly at 5% inclusion. This was associated with a significant decrease in MDA levels, reflecting reduced lipid peroxidation and oxidative stress (Abd El-hack et al., 2021). The antioxidant action of Moringa has been associated with polyphenols, flavonoids (quercetin and kaempferol), and vitamins A, C, and E. These nutrients participate in the cellular redox system in addition to reactive oxygen scavenging (Tari et al., 2023). The results that have been obtained on increased antioxidant enzyme activities clearly indicate that MOLM builds up the defense system of birds against oxidative stress. This is very important to keep birds productive and maintain their physiological functions when they are under any kind of intensive rearing condition. MOLM supplementation led to a significant downregulation of pro-inflammatory cytokines IL-1β, IL-6, and TNF-α in both liver and intestinal tissues. This anti-inflammatory response is likely mediated by the bioactive phytochemicals in MOLM that inhibit nuclear factor-kappa B (NF-κB) signaling and reduce cytokine expression (Hanan et al., 2020; Arshad et al., 2022; Shedeed et al., 2022; Al-Maiman et al., 2023). Notably, the 5% inclusion level produced the strongest anti-inflammatory effects, aligning with the antioxidant trends and highlighting a critical threshold for efficacy. Although 7.5% MOLM also reduced cytokine levels, the benefits plateaued, that higher doses may not provide additional immunological advantage and should be cautiously evaluated (Abbas et al., 2021). The histological findings further validated the biochemical results, with significant reductions in inflammation and oxidative damage scores in the liver and intestinal tissues among the MOLM-supplemented groups. Birds fed 5% and 7.5% MOLM displayed near-normal histological architecture, minimal inflammatory cell infiltration, and preserved tissue morphology. Interestingly, although the 7.5% group continued to show improved histopathological scores, biochemical markers such as MDA indicated a slight increase in lipid peroxidation compared with the 5% group. This depicted that while antioxidant defenses were still functionally protective at the tissue level, the biochemical system may have reached a threshold where higher MOLM inclusion no longer conferred proportional benefits (Varna et al., 2018). Such findings align with the concept of a biphasic or hormetic response to phytogenics, where moderate supplementation optimally stimulates endogenous antioxidant pathways, but excessive inclusion can lead to diminishing or even counteractive effects due to metabolic adaptation or potential pro-oxidant activity of certain phytochemicals at higher concentrations. Therefore, while both 5% and 7.5% MOLM improved tissue integrity, the plateau or slight divergence between biochemical and histological outcomes highlights the importance of identifying an optimal inclusion level to balance efficacy and avoid potential over-supplementation. These findings reinforce the immunoprotective and cytoprotective effects of MOLM and align with its traditional use in ethnomedicine for liver and gastrointestinal health (Gopalakrishnan et al., 2016; Al-Juhaimi and Özcan, 2017; Azzam et al., 2021; Rehman et al., 2023). ConclusionThe findings of this study support the use of M. oleifera leaf meal as a functional feed additive in laying hens. A 5% inclusion level emerged as the optimal dose, promoting FCR, improving egg production and quality, enhancing antioxidant defense systems, and reducing tissue inflammation and oxidative damage. MOLM demonstrates a dual function by enhancing both production performance and physiological health, making it a promising natural substitute for synthetic additives in the nutrition of laying hens. Further long-term studies, including molecular assays and reproductive evaluations, are recommended to fully elucidate its mechanistic pathways and commercial applicability. AcknowledgmentWe sincerely acknowledge the invaluable support and cooperation of the Faculty of Veterinary and Animal Sciences, Gomal University, and the University of Veterinary and Animal Sciences, Swat, Pakistan, for their assistance in facilitating the data collection process for this study. Conflict of interestThe authors declare no competing interests or conflicts of interest associated with this study. FundingThe current study was supported by the Faculty of Veterinary and Animal Sciences, Gomal University, and University of Veterinary and Animal Sciences, Swat. Authors’ contributionsAll authors were actively involved in data analysis, manuscript preparation, and critical revisions. The authors collectively take full responsibility for the integrity and accuracy of the work presented in this study. Data availabilityData were available to the primary author and will be provided on special request. ReferencesAbbas, R.Z., Liu, J., Iqbal, Z. and Akhtar, M. 2021. Antioxidant and anti-inflammatory potential of Moringa oleifera leaf extract in poultry: a review. World. Poultry. Sci. J. 77(4), 891–903; doi:10.1080/00439339.2021.1925332 Abd El-hack, M.E., Alagawany, M., Farag, M.R., Tiwari, R., Karthik, K., Dhama, K. and Zorriehzahra, J. 2021. Nutritional, antioxidant, and antibacterial efficacy of Moringa oleifera leaves as a feed additive in poultry nutrition. Poultry Sci. 100(5), 100875; doi:10.1016/j.psj.2021.100875 Abdelkader, G.S., El-Naenaeey, E.S.Y., Abdallah, H.M., Abu-Zeid, E.H., Rehan, I.F., Zigo, F. and Elmowalid, G.A. 2025. Immune enhancement and disease resistance against Aeromonas hydrophila infection by dietary Lactobacillus plantarum-fermented Moringa oleifera leaves in Oreochromis niloticus. Front. Vet. Sci. 12, 1557671; doi:10.3389/fvets.2025.1557671 Abdel-Moneim, A. M. E., Shehata, A. M., Alzahrani, S. O., Shafi, M. E., Mesalam, N. M., Taha, A. E., and Abd El-Hack, M. E. 2020. The role of polyphenols in poultry nutrition. J. Animal Physiol. Anim. Nutr. 104(6), 1851-1866. Abou-Elkhair, R., Ahmed, H.A. and Selim, S. 2014. Effects of black pepper (Piper nigrum), turmeric powder (Curcuma longa), and coriander seeds (Coriandrum sativum) and their combinations as feed additives on broiler chicken growth performance, carcass traits, blood parameters, and humoral immune response. Asian-Australasian. J. Anim. Sci. 27(6), 847–854; doi:10.5713/ajas.2013.13644 Aebi, H. 1984. Catalase in vitro. In Methods in enzymology (Vol. 105, pp. 121-126). Academic press. Akbar, M.A., Siregar, A.R. and Asnawi, A. 2024. The effects of relationship marketing on customer loyalty through customer retention in layer feed customers in South Sulawesi, Indonesia. Int. J. Agriculture. Biosciences. 13(3), 378–389; doi:10.47278/journal.ijab/2024.129 Akinyemi, F.T., Adedapo. and Afolayan. 2017. Phytochemical evaluation and antioxidant activities of Moringa oleifera leaf aqueous extract. MedPlants–International. J. Phytomedicines Rel. Industries 9(2), 89–96; doi:10.5958/0975-6892.2017.00014.3 Al-Juhaimi, F.Y. and Özcan, M.M. 2017. Phytochemical components and antioxidant and mineral contents of seven Moringa accessions. Arabian. J. Chem. 10(4), 503–508; doi:10.1016/j.arabjc.2014.12.005 Al-Maiman, S.A., Ahmed, M.M., Shehata, A.M. and Alghamdi, A.A. 2023. Dietary phytobiotics modulate the expression of inflammatory genes and gut histomorphology in broilers exposed to immune challenge. Poultry Sci. 102(5), 102563; doi:10.1016/j.psj.2023.102563 Al-Suwailem, N.K., Kamel, N.N., Abbas, A.O., Nassar, F.S., Mohamed, H.S., Gouda, G.F. and Safaa, H.M. 2024. Impact of dietary M. oleifera leaf supplementation on stress markers, immune responses, and productivity in heat-stressed broilers. Int. J. Vet. Sci. 13(6), 980–987; doi:10.47278/journal.ijvs/2024.210 Arshad, M.I., Ashraf, S., Aslam, B. and Saeed, M. 2022. Therapeutic potential of M. oleifera phytochemicals in inflammatory disorders in animals. Vet. Quart. 42(1), 1–11; doi:10.1080/01652176.2021.1984432 Amad, A.A., and Zentek, J. 2022. Moringa (M. oleifera) leaf meal in diets for broilers and laying hens: A review. J. Agr. Sci, 14(10), 12-33. Azzam, M.M.M., Zou, X.T., Dong, X.Y., Xie, P. and Sun, Z. 2021. Effects of dietary M. oleifera leaf meal on poultry performance, carcass characteristics, and health status. Trop. Anim. Health. Prod. 53(4), 403; doi:10.1007/s11250-021-02827-4 Bancroft, J.D. and Gamble, M. (Eds.). 2008. Theory and practice of histological techniques. Elsevier Health Sciences. Bebas, W., Gorda, I. W., Agustina, K. K., and Merdana, I. M. 2023. Effects of Adding Glutathione to AndroMed Diluent on Intact Plasma and Acrosome Membranes and Progressive Motility of Cattle Spermatozoa During Freezing Processes. World's Vet. J. 13(4), 561–570. Celeste, A., Bulang, M. and Kluth, H. 2011. Assessing the egg quality of eggs from chickens fed with different amounts of dried Moringa oleifera leaves. J. Anim. Physiol. Anim. Nutr. 95(4), 481–487; doi:10.1111/j.1439-0396.2010.01078.x Elwan, H.A.M., Elnesr, S.S. and El-Saadony, M.T. 2019. Comparative evaluation of the effects of natural antioxidant sources on performance, egg quality, and egg antioxidant capacity in laying hens. Poultry Sci. J. 7(1), 61–75. Gopalakrishnan, L., Doriya, K. and Kumar, D.S. 2016. Moringa oleifera: a review of its nutritive importance and medicinal application. Food Sci. Hum. Wellness 5(2), 49–56; doi:10.1016/j.fshw.2016.04.001 Gul, S.T., Raza, R., Hannan, A., Khaliq, S., Waheed, N. and Aderibigbe, A. 2024. Potential of the medicinal plant Urtica dioica (Stinging nettle) as a feed additive for animals and birds: a review. Agrobiological Records 17, 110–118; doi:10.47278/journal.abr/2024.029 Hanan, A.M., El-Mandrawy, S.A. and El-Edel, M.A. 2020. Dietary M. oleifera and inflammation-related gene expression in broilers. Vet. World 13(11), 2424–2431; doi:10.14202/vetworld.2020.2424-2431 Hayati, T., Herliatika, A., Sinurat, A.P., Wina, E., Purba, M. and Puastuti, W. 2025. Efficacy of clove leaf extract, mangosteen peel extract, and liquid smoke as feed additives for native chickens. Int. J. Vet. Sci. 14(1), 107–112; doi:10.47278/journal.ijvs/2024.214 Kakati, L.N., Das, A. and Kalita, N. 2015. Effect of M. oleifera leaf meal on egg production, egg quality, and biochemical parameters in laying hens. Indian. J. Anim. Nutr. 32(4), 401–406; doi:10.1016/j.ijani.2013.08.010 Khoramabadi, V., Kermanshahi, H. and Moghaddam, H.N. 2022. Effects of dietary Moringa oleifera on performance, antioxidant status, and immune response in poultry: a meta-analysis. Anim. Feed Sci. Technol. 292, 115441; doi:10.1016/j.anifeedsci.2022.115441 Leone, A., Spada, A., Battezzati, A., Schiraldi, A., Aristil, J. and Bertoli, S. 2015. Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: an overview. Plant Mol. Biol. Int. J. Mol. Sci. 16(6), 12791–12835; doi:10.3390/ijms160612791 Lu, W., Li, S., Sun, Z., Tang, Y. and Qian, Q. 2016. Effects of M. oleifera leaf meal on laying performance, egg quality, and antioxidant activity of laying hens. Poultry Sci. 95(10), 1–7; doi:10.3382/ps/pew321 Mabrok, H.B., Khafaga, A.F. and Mohamed, E.A. 2022. Nutritional role of Moringa oleifera as a natural feed additive for improving egg quality and reproductive performance in poultry. BMC. Vet. Res. 18, 273; doi:10.1186/s12917-022-03351-0 Marklund, S. and Marklund, G. 1974. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 47(3), 469–474. National Research Council (NRC), 1994. Nutrient requirements of poultry. 9th Edition. National Academy Press, Washington, DC. Nkukwana, T.T., Muchenje PJ Masika., Hoffman K Dzama. and Descalzo. 2014. Effect of replacing soybean meal with Moringa oleifera leaf meal on broiler chicken growth performance, apparent digestibility, and carcass characteristics. Livestock Sci. 161, 139–146; doi:10.1016/j.livsci.2013.12.020 Olugbemi, T. S., Mutayoba, S. K. and Lekule, F. P. 2010. Effect of Moringa (Moringa oleifera) inclusion in cassava based diets fed to broiler chickens. Int. J. Poultry Sci. 9(4), 363–367. Paglia, D.E. and Valentine, W.N. 1967. Quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70(1), 158–169. Pebas, W., Gorda, I.W. and Agustina, K.K. 2023. Spermatozoa quality of Kintamani dogs in coconut water-egg yolk diluent with Moringa leaf and carrot extract addition. Int. J. Vet. Sci. 12(3), 333–340; doi:10.47278/journal.ijvs/2022.197 Rehman, M.S.U., Zahra, N. and Shehzad, A. 2023. Dietary M. oleifera supplementation improves gut integrity and liver function in poultry. Front. Vet. Sci. 10, 1136123; doi:10.3389/fvets.2023.1136123 Rivas-Caceres, R.R., Khazaei, R., Ponce-Covarrubias, J.L., Di Rosa, A.R., Ogbuagu, N.E., Estrada, G.T., Zigo, F., Gorlov, I.F., Slozhenkina, M.I., Mosolov, A.A., Lackner, M. and Elghandour, M.M.M.Y. 2024. Effects of dietary Silybum marianum powder on growth performance, egg and carcass characteristics, immune response, intestinal microbial population, haemato-biochemical parameters and sensory meat quality of laying quails. Poultry. Sci.103, 104036; doi: 10.1016/j.psj.2024.104036 Shedeed, H.A., El-Wardany, I., El-Sayed, A. and Younis, M. 2022. Dietary inclusion of M. oleifera leaf meal modulates immune and inflammatory responses in broilers under oxidative stress. J. Anim. Physiol. Anim. Nutr. 106(1), 1–10; doi:10.1111/jpn.13637 Sooksawat, N., Chittawanij, A., Olanratmanee, P., Insoongnern, H., Wongsaeng, P., Kumproa, K., Chinaworn, S., Ruanpan, W., Ruanpan, N., Inthorn, D. and Vangnai, A. 2024. Potential use of sunn hemp as green manure and biostimulant for enhancement of animal feed corn crop and fertilized soil properties. Int. J. Agriculture Biosciences 13(3), 419–428; doi:10.47278/journal.ijab/2024.138 Suvarna, S., Dsouza, J., Ragavan, M. L. and Das, N. 2018. Potential probiotic characterization and effect of encapsulation of probiotic yeast strains on survival in simulated gastrointestinal tract condition. Food Sci. Biotechnol. 27(3), 745–753. Tari, A., Warly, L., Hermon, S. and Evitayani. 2023. Metabolic response and meat quality of goats fed with A. heterophyllus and M. oleifera. Int. J. Vet. Sci. 12(4), 498–503; doi:10.47278/journal.ijvs/2022.216 Varna, S.K., Layton, C. and Bancroft, J.D. 2018. Bancroft’s theory and practice of histological techniques (8th ed.). New York: Routledge. Tufarelli, V., Casalino, E., D'Alessandro, A.G. and Laudadio, V. 2016. Dietary administration of phytogenic compounds improves the productive performance and oxidative status of laying hens South African J. Anim. Sci. 46(3), 295–301. Doi: 10.4314/sajas.v46i3.3 Ike OO, Ekugba CU, Ezenyilimba BN, Onwumelu IJ, Okonkwo AP, Ejivade OM, Ezejesi HC, Okonkwo JC and Nwankwo CA, 2025. Strain effect on hematological indices of broiler chicks fed graded levels of Phyllanthus amarus leaf extract. Agrobiological Records 19, 50-55. doi: 10.47278/journal.abr/2025.006 Verma, A.R., Vijayakumar, M., Mathela, C.S. and Rao, C.V. 2020. In vitro and in vivo antioxidant properties of Moringa oleifera leaf fractions. Food. Chem. Toxicol. 44(5), 1044–1049; doi:10.1016/j.fct.2006.11.007 Yamin, A.A., Ridwan, M., Purwanti, S. and Syamsu, J.A. 2025. Analysis of the sustainability of small-scale feed mills in supporting the poultry industry in Sidenreng Rappang Regency. Int. J. Agriculture Biosciences 14(1), 153–163; doi:10.47278/journal.ijab/2024.192 | ||
| How to Cite this Article |
| Pubmed Style Shah MY, Ullah S, Zaman A, Ramzan F, Rehman S. Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Vet. J.. 2025; 15(12): 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 Web Style Shah MY, Ullah S, Zaman A, Ramzan F, Rehman S. Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. https://www.openveterinaryjournal.com/?mno=274416 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.30 AMA (American Medical Association) Style Shah MY, Ullah S, Zaman A, Ramzan F, Rehman S. Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Vet. J.. 2025; 15(12): 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 Vancouver/ICMJE Style Shah MY, Ullah S, Zaman A, Ramzan F, Rehman S. Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 Harvard Style Shah, M. Y., Ullah, . S., Zaman, . A., Ramzan, . F. & Rehman, . S. (2025) Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Vet. J., 15 (12), 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 Turabian Style Shah, Muhammad Yasir, Shakeeb Ullah, Ali Zaman, Faiqah Ramzan, and Saifur Rehman. 2025. Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Veterinary Journal, 15 (12), 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 Chicago Style Shah, Muhammad Yasir, Shakeeb Ullah, Ali Zaman, Faiqah Ramzan, and Saifur Rehman. "Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments." Open Veterinary Journal 15 (2025), 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 MLA (The Modern Language Association) Style Shah, Muhammad Yasir, Shakeeb Ullah, Ali Zaman, Faiqah Ramzan, and Saifur Rehman. "Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments." Open Veterinary Journal 15.12 (2025), 6461-6469. Print. doi:10.5455/OVJ.2025.v15.i12.30 APA (American Psychological Association) Style Shah, M. Y., Ullah, . S., Zaman, . A., Ramzan, . F. & Rehman, . S. (2025) Effect of dietary Moringa oleifera leaf meal on feed conversion, egg traits, and immunometabolic responses in laying hens: In vivo and ex vivo assessments. Open Veterinary Journal, 15 (12), 6461-6469. doi:10.5455/OVJ.2025.v15.i12.30 |