| Research Article | ||
Open Vet. J.. 2025; 15(12): 6851-6855 Open Veterinary Journal, (2025), Vol. 15(12): 6851-6855 Research Article Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss)Mohamed Omar Abdalla Salem1* and Masoud A. S. Lakwani21Department of Biology, Faculty of Education, Bani Waleed University, Bani Waleed, Libya 2Department of Zoology, Faculty of Science, University of Derna, El-Gubbh, Libya *Corresponding Author: Mohamed O. A. Salem. Department of Biology, Faculty of Education, Bani Waleed University, Bani Waleed, Libya. Email: MohamedSalem [at] bwu.edu.ly Submitted: 29/07/2025 Revised: 30/10/2025 Accepted: 14/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
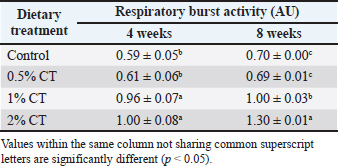
AbstractBackground: The exploration of natural dietary supplements to enhance immune function and disease resistance in aquaculture is of significant interest. Aim: This study aimed to investigate the capacity of dietary Vitex agnus-castus L. (Chaste Tree; CT) extract to modulate immune function in rainbow trout (Oncorhynchus mykiss). Methods: Fish were divided into four cohorts and maintained on isocaloric diets containing 0% (control), 0.5%, 1%, or 2% (w/w) CT extract for 60 days. Key innate immune markers, including blood respiratory burst activity and serum lysozyme activity, were quantified at weeks 4 and 8. Results: Supplementation with CT significantly increased (p < 0.05) respiratory burst activity in groups receiving 1% and 2% CT relative to controls. After 8 weeks of feeding, serum lysozyme activity was markedly elevated in fish fed 0.5% and 1% CT. Conclusion: These findings indicate that V. agnus-castus extract effectively potentiates crucial innate immune responses in rainbow trout, supporting its prospective application as a functional dietary supplement to enhance health status and disease resistance in aquaculture. Keywords: Vitex agnus-castus L., Immunostimulant, Respiratory burst, Oncorhynchus mykiss, Aquaculture. IntroductionThe rainbow trout (Oncorhynchus mykiss) is a cornerstone species in global aquaculture production (Salem and Moammer, 2024). Ensuring optimal health and welfare of farmed fish is critical for sustainable industry development, as they frequently encounter stressors such as environmental shifts, disease pressures, and nutritional imbalances that can impair physiological function (Bilen et al., 2019; Salem and Lakwani, 2024). Robust innate immune responses, which provide the first line of protection against pathogens, are a vital component of their defense against such challenges (Yrüten Özdemir et al., 2018; Salem et al., 2022) Consequently, strategies to enhance immunocompetence are essential for improving aquaculture sustainability (Salem et al., 2021). Vitex agnus-castus L. (Chaste Tree), a member of the Lamiaceae family, is a medicinal plant with antioxidant, anti-inflammatory, and immunomodulatory properties attributed to bioactive constituents such as flavonoids and rosmarinic acid (Taştan and Salem, 2021; Salem and Mohamed, 2025). Plant-derived extracts are increasingly recognized for their potential to boost immunity in fish (Reverter et al., 2014; Yılmaz et al., 2019; Kadak and Salem, 2020; Kuebutornye et al., 2024; Salem et al., 2025a,b,c; Salem and Mohamed, 2025) . However, the specific immunomodulatory effects of Chaste Tree extract in rainbow trout remain poorly characterized. This study seeks to fill this knowledge gap by providing a detailed dose-response analysis of the effects of Chaste Tree (CT) on key innate immune parameters. Therefore, this study aimed to evaluate the impact of dietary CT supplementation on specific innate immune parameters, such as respiratory burst activity and lysozyme activity, in this economically important fish species. Materials and MethodsExperimental designJuvenile rainbow trout (mean initial weight 50 ± 5 g) were acclimated for 14 days and randomly distributed into four treatment groups (n=30/group). The groups were fed isonitrogenous, isocaloric diets formulated to meet trout nutritional requirements, supplemented with 0% (control), 0.5%, 1%, or 2% (w/w) CT extract for 60 days. No signs of toxicity were observed at any dose level during the experiment. Extract preparationChaste tree seeds were dried, ground, and extracted using 70% aqueous ethanol (100 g powder in 500 ml solvent) via maceration at 25°C for 72 hours. The filtrate was concentrated under vacuum using rotary evaporation and stored at −20°C until diet incorporation (Salem, 2024a,b,c). SamplingFish were sampled after 4 and 8 weeks following a 24-hour fast. Individuals were anesthetized (MS-222, 100 mg/l) and euthanized, and blood was collected via the caudal vein into heparinized syringes (n=3/group/time point). The sample size was selected based on previous studies in fish immunology where these specific assays have demonstrated low intra-group variability and high reproducibility, allowing for the detection of significant differences despite the small sample size. Serum was separated by centrifugation (3,000 × g, 15 minutes, 4°C) and stored at −80°C. Immune assaysRespiratory burstMeasured using the nitroblue tetrazolium (NBT) reduction assay. Blood was incubated with NBT and phorbol myristate acetate; formazan production was quantified spectrophotometrically at 570 nm (Absorbance Units, AU). Lysozyme activityDetermined turbidimetrically (Sigma-Aldrich kit) by monitoring the lysis rate of Micrococcus lysodeikticus cells. Activity was reported as U/ml (1 unit=ΔA450 of 0.001/minute). Statistical analysisData were analyzed using SPSS v25. After testing for normality and homogeneity of variance, data that met parametric assumptions were analyzed using one-way analysis of variance. For data that violated these assumptions, the non-parametric Kruskal-Wallis test was used. Group differences were assessed via one-way ANOVA. Significant effects (p < 0.05) were further probed using Duncan's multiple range test. Results are expressed as mean ± SEM. Ethical approvalAll experimental procedures were conducted in accordance with the ethical guidelines for the use of animals in research and were approved by the Kastamonu University Institutional Animal Care and Use Committee. (Reference number: KUHADYEK-2023/118, Date: 09/10/2023). ResultsRespiratory burst activityDietary supplementation with V. agnus-castus CT extract exerted was associated with a statistically significant modulatory influence on respiratory burst activity in rainbow trout (Table 1). A pronounced and dose-dependent augmentation of this critical phagocyte function was observed. Specifically, the cohorts receiving diets expressed with 1% and 2% CT inclusion demonstrated a marked elevation in respiratory burst activity relative to the unsupplemented control group. This significant enhancement was consistently observed across both experimental sampling intervals, indicating a sustained immunostimulatory effect. Furthermore, the magnitude of the response exhibited a clear dose relationship, culminating in the maximal observed respiratory burst activity within the 2% CT diet-administered group. This peak phagocyte activation was recorded at the terminal sampling point, specifically after continuous dietary intervention for 8 weeks. Table 1. Respiratory burst activity (AU) in the serum of rainbow trout fed with diets supplemented with different concentrations of CT extract.
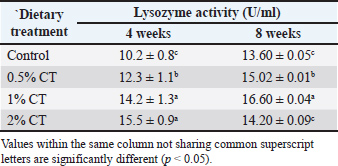
Lysozyme activityDietary inclusion of V. agnus-castus extract elicited a significant modulatory effect on serum lysozyme activity in rainbow trout, as quantified and presented in Table 2. A pronounced and dose-dependent augmentation of this key humoral immune parameter was observed after the 8-week experimental period. Specifically, the cohorts receiving diets supplemented with 0.5% and 1% CT demonstrated statistically significant elevations in serum lysozyme concentration relative to the non-supplemented control group. This enhancement signifies a potentiation of constitutive antibacterial defense mechanisms at these intermediate inclusion levels. Conversely, and of particular note, lysozyme activity measured in the cohort administered the highest dietary concentration (2% CT) at the 8-week interval exhibited no statistically significant divergence from control levels. This absence of enhancement at the supra-optimal dose further corroborates the non-linear, concentration-dependent response dynamic characterizing the immunomodulatory action of CT on this specific parameter, suggesting the existence of a bounded efficacy window for lysozyme induction. Table 2. Lysozyme activity (U/ml) in the serum of rainbow trout fed with diets supplemented with different concentrations of CT extract.
DiscussionThis investigation provides evidence that dietary supplementation utilizing an extract derived from V. agnus-castus (CT) elicits a significant modulatory effect on specific facets of the innate immune system in rainbow trout. The observed, statistically significant enhancement of respiratory burst activity in cohorts administered diets containing 1% and 2% CT extract, coupled with the concomitant elevation in serum lysozyme activity in fish receiving dietary inclusions of 0.5% and 1% CT, collectively underscores the potent immunostimulatory properties conferred by this phytogenic intervention. Respiratory burst activity is a critical functional indicator that directly reflects the phagocytic cellular capacity to generate microbicidal reactive oxygen species essential for the intracellular destruction of pathogenic invaders (Salem, 2017; Lakwani et al., 2022; Salem et al., 2023; Lakwani and Salem, 2024). The pronounced increase in this activity observed in the present study suggests that CT extract supplementation effectively potentiates the phagocyte-mediated defensive capability of rainbow trout against microbial challenges. The serum-based assay used here serves as an indicator of systemic oxidative potential, and future studies should include cellular assays to confirm these findings at the leukocyte level. Similarly, the documented increase in lysozyme activity—a key enzymatic component of the humoral innate immune response known for its ability to hydrolyze peptidoglycan bonds within bacterial cell walls—signifies an augmentation of constitutive humoral immunity within CT-supplemented fish. This enhancement was demonstrated to be effective at intermediate inclusion levels (0.5%, 1%). The lack of enhancement at the 2% dose may be attributed to a potential biphasic (hormetic) response, where supra-optimal concentrations lead to receptor saturation, negative feedback mechanisms, or compensatory metabolic pathway induction that attenuates the stimulatory effect. The observed immunomodulatory effects are principally ascribed to the chemically diverse spectrum of bioactive constituents inherent within V. agnus-castus CT, with flavonoids and polyphenolic compounds representing prominent candidate molecules. In diverse biological models, these phytogenic agents have been extensively documented to exhibit inherent immunostimulatory properties, capable of modulating key immune pathways (Mahmoud and Ibrahim, 2013; Dezfuly 2017; Hoseini et al., 2021; Hamad et al., 2024; Salem and Barkah, 2025). Future research should focus on identifying the specific bioactive compounds within CT responsible for these effects. The absence of a pathogen challenge test to directly correlate the observed immune enhancements with improved disease resistance is a limitation of this study. Furthermore, no signs of toxicity (e.g., mortality, altered behavior) were observed at any dose during the experiment, suggesting that these concentrations are safe for short-term use. However, comprehensive toxicity studies are recommended. ConclusionDietary V. agnus-castus extract effectively stimulates key innate immune responses in rainbow trout. Based on our findings, dietary inclusion of 0.5%–1% CT extract is recommended for enhancing innate immunity without adverse effects. These results support its potential as a natural immunostimulant feed additive to enhance disease resistance and overall health in aquaculture settings. AcknowledgmentsNone. Conflict of interestThe authors have no conflicts of interest to declare. FundingNine. Authors’ contributionsMohamed Omar Abdalla Salem: Plant collection, preparation of the extraction, during experimental period fish care, laboratory studies, statistical analyses, and writing. Masoud A. S. Lakwani: preparation of experimental design, laboratory studies, and writing. Data availabilityAll data were provided in the manuscript. ReferencesBilen, S., Altief, T.A.S., Özdemir, K.Y., Salem, M.O.A., Terzi, E. and Güney, K. 2019. Effect of lemon balm (Melissa officinalis) extract on growth performance, digestive and antioxidant enzyme activities, and immune responses in rainbow trout (Oncorhynchus mykiss). Fish Physiol. Biochem. 45(4), 1053–1062; doi:10.1007/s10695-019-00661-2 Dezfuly, T. 2017. Effects of Aloe vera crude extract on growth performance and some hemato-immunological indices of Oncorhynchus mykiss in farm scale. Iran. J. Vet. Med. 11(4), 383–393; doi:10.22059/ijvm.2017.231790.1004806 Hamad, M., Mohamed, T., Lakwani, M.A.S., Hatim, I.E.I., Kamal, E., Salem, M.O.A., Attia, A. and Almansory, R. 2024. Effect of Adding Olive (Olea europaea L.) Oil to Broiler Feed on Growth Performance and Mortality Rate. North Afr. J. Sci. Publ. 2(4), 194–200; https://najsp.com/index.php/home/article/view/315 Hoseini, S.M., Taheri Mirghaed, A., Iri, Y., Hoseinifar, S.H., Van Doan, H. and Reverter, M. 2021. Effects of dietary Russian olive, Elaeagnus angustifolia, leaf extract on growth, hematological, immunological, and antioxidant parameters in common carp, Cyprinus carpio. Aquaculture 536, 736461; doi:10.1016/j.aquaculture.2020.736461 Kadak, A.E. and Salem, M.O.A. Antibacterial activity of chitosan, some plant seed extracts and oils against Escherichia coli and Staphylococcus aureus. Alınteri J. Agric. Sci. 35(2), 144–150; doi:10.47059/alinteri/V35I2/AJAS20086 Kuebutornye, F.K.A., Roy, K., Folorunso, E.A. and Mraz, J. 2024. Plant-based feed additives in Cyprinus carpio aquaculture. Rev. Aquac. 16(1), 309–336; doi:10.1111/raq.12840 Lakwani, M.A.S. and Salem, M.O.A. 2024. Effects of using olive tree (Olea europaea L.) derivatives as feed additives on growth efficiency, immunological response, and oxidative status in finfish: a Review. Afr-Asian. J. Sci. Res. 2(3), 204–216; https://aajsr.com/index.php/aajsr/article/view/183 Lakwani, M.A.S., Kenanoğlu, O.N., Taştan, Y. and Bilen, S. 2022. Effects of black mustard (Brassica nigra) seed oil on growth performance, digestive enzyme activities and immune responses in rainbow trout (Oncorhynchus mykiss). Aquac. Res. 53(1), 300–313; doi:10.1111/are.15583 Mahmoud, R.E. and Ibrahim, D. 2013. The effect of aloe gel and dried Camphora leaves on growth performance, body composition, immunity, some blood parameters and economic efficiency in Nile Tilapia (Oreochromis niloticus). Glob. J. Fish. Aqua. Res. 6(6), 149–164. Reverter, M., Bontemps, N., Lecchini, D., Banaigs, B. and Sasal, P. 2014. Use of plant extracts in fish aquaculture as an alternative to chemotherapy: current status and future perspectives. Aquaculture 433, 50–61; doi:10.1016/j.aquaculture.2014.05.048 Salem, M.O.A. 2017. Determination of antioxidant enzyme activities in rainbow trout (Oncorhynchus mykiss) fry fed with feeds containing dandelion (Taraxacum officinale) and beard lichen (Usnea barbata). Karahindiba (Taraxacum officinalis) ve sakal yosunu (Usnea barbata) içeren yemlerle beslenen gökkuşağı alabalığı yavrularında (Oncorhynchus mykiss) antioksidan enzim aktivitelerinin belirlenmesi. Institute of Science and Technology, Kastamonu University. Availablle via https://hdl.handle.net/20.500.12597/16793 Salem, M.O.A. 2024. Antimicrobial Activity of Aqueous Methanolic Extract of Lichen Usnea barbata Against E. coli and S. aureus. Libyan J. Ecol. Environ. Sci. Technol. 6(1), 19–23; doi:10.5281/zenodo.12638654 Salem, M.O.A. and Barkah, A.D.A. 2025. The Effect of Dietary Supplementation with Chaste Tree (Vitex agnus-castus L.) Seed Extract on the Haematological Indices of Rainbow Trout (Oncorhynchus mykiss). Afr. J. Adv. Pure. Appl. Sci. 4(2), 389–393; https://www.aaasjournals.com/index.php/ajapas/article/view/1283 Salem, M.O.A. and Lakwani, M.A.S. 2024. Determination of chemical composition and biological activity of flaxseed (Linum usitatissimum) essential oil. J. Biom. Stud. 4(2), 91–96; doi:10.61326/jofbs.v4i2.05 Salem, M.O.A. and Moammer, E.M.E. 2024. Potential benefits of Aloe vera derivative in aquaculture. Bani. Waleed. Univ. J. Humanit. Appl. Sci. 9(2), 379–389; doi:10.58916/jhas.v9i2.269 Salem, M.O.A. and Mohamed, N.M. 2025. Investigating the Effects of Aqueous-Methanol Extract from Chaste Tree (Vitex agnus-castus L.) on Growth Enhancement and Digestive Enzyme Activity in Rainbow Trout (Oncorhynchus mykiss). Libyan. J. Contemp. Acad. Stud. 3(1), 21–27; https://ljcas.ly/index.php/ljcas/article/view/31 Salem, M.O.A., Abuamoud, M.M.M., Rezgalla, R.M. and Ahmed, G.S. 2025. Antimicrobial Activity of Extracts of Dandelion (Taraxacum officinale) Against Escherichia coli and Staphylococcus aureus: mechanisms, Modern Insights, and Therapeutic Potential. Libyan. J. Med. Appl. Sci. 3(2), 37–40; doi:10.64943/ljmas.v3i2.52 Salem, M.O.A., Salem, T.A., Yürüten Özdemir, K., Sönmez, A.Y., Bilen, S. and Güney, K. 2021. Antioxidant enzyme activities and immune responses in rainbow trout (Oncorhynchus mykiss) juveniles fed diets supplemented with dandelion (Taraxacum officinale) and lichen (Usnea barbata) extracts. Fish. Physiol. Biochem. 47(4), 1053–1062; doi:10.1007/s10695-021-00952-7 Salem, M.O.A., Taştan, Y., Bilen, S., Terzi, E. and Sönmez, A.Y. 2022. Effects of white mustard (Sinapis alba) Oil on growth performance, immune response, blood parameters, digestive and antioxidant enzyme activities in rainbow trout (Oncorhynchus mykiss). Fish. Shellfish. Immunol. 131, 108–118; doi:10.1016/j.fsi.2022.09.065 Salem, M.O.A., Taştan, Y., Bilen, S., Terzi, E. and Sönmez, A.Y. 2023. Dietary flaxseed (Linum usitatissimum) oil supplementation affects growth, oxidative stress, immune response, and diseases resistance in rainbow trout (Oncorhynchus mykiss). Fish. Shellfish. Immunol. 138, 108798; doi:10.1016/j.fsi.2023.108798 Taştan, Y. and Salem, M.O.A. 2021. Use of Phytochemicals as Feed Supplements in Aquaculture: a Review on Their Effects on Growth, Immune Response, and Antioxidant Status of Finfish. J. Agric. Prod. 2(1), 32–43; doi:10.29329/agripro.2021.344.5 Yılmaz, E., Çoban, D., Kırım, B. and Güler, M. 2019. Effects of Extracts of Feed Additives Including Rosemary (Rosmarinus officinalis) and Aloe Vera (Aloe barbadensis) on the Growth Performance and Feed Utility of Nile Tilapia (Oreochromis niloticus). Turk. J. Agric. -. Food. Sci. Technol. 7(6), 866–870; doi:10.24925/turjaf.v7i6.866-870.2438 Yrüten Özdemir, K., Aras, S., Salem, M.O.A., Altief, T.A.S. and Bilen, S. Effects of Marshmallow (Althaea officinalis) Methanolic Extracts on Growth Performance and Antioxidant Enzyme Activities of Rainbow Trout (Oncorhynchus mykiss). In Proceedings of the 1st International Congress on Engineering and Life Science,2018 Kastamonu, https://hdl.handle.net/20.500.12597/31412 | ||
| How to Cite this Article |
| Pubmed Style Salem MOA, Lakwani MAS. Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Vet. J.. 2025; 15(12): 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 Web Style Salem MOA, Lakwani MAS. Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). https://www.openveterinaryjournal.com/?mno=274084 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.65 AMA (American Medical Association) Style Salem MOA, Lakwani MAS. Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Vet. J.. 2025; 15(12): 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 Vancouver/ICMJE Style Salem MOA, Lakwani MAS. Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 Harvard Style Salem, M. O. A. & Lakwani, . M. A. S. (2025) Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Vet. J., 15 (12), 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 Turabian Style Salem, Mohamed Omar Abdalla, and Masoud A. S. Lakwani. 2025. Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Veterinary Journal, 15 (12), 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 Chicago Style Salem, Mohamed Omar Abdalla, and Masoud A. S. Lakwani. "Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss)." Open Veterinary Journal 15 (2025), 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 MLA (The Modern Language Association) Style Salem, Mohamed Omar Abdalla, and Masoud A. S. Lakwani. "Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss)." Open Veterinary Journal 15.12 (2025), 6851-6855. Print. doi:10.5455/OVJ.2025.v15.i12.65 APA (American Psychological Association) Style Salem, M. O. A. & Lakwani, . M. A. S. (2025) Immunomodulatory effects of dietary chaste tree (Vitex agnus-castus L.) extract in rainbow trout (Oncorhynchus mykiss). Open Veterinary Journal, 15 (12), 6851-6855. doi:10.5455/OVJ.2025.v15.i12.65 |