| Research Article | ||
Open Vet. J.. 2025; 15(12): 6497-6505 Open Veterinary Journal, (2025), Vol. 15(12): 6497-6505 Research Article Toxicity and potential of propolis resin to boost embryo growth of broilersMaslichah Mafruchati 1*, Lailatul Fadliyah2, Akhmad Kusuma Wardhana3, Intan Idiana Hassan4 and Joni Susanto51Department of Veterinary Anatomy, Faculty of Veterinary Medicine (60115), Universitas Airlangga, Mulyorejo, Surabaya, Indonesia 2Faculty of Vocational Studies, Airlangga University, Indonesia 3Department of Environmental Management, Prince of Songkla University, Hatyai, Surabaya, Thailand 4School of Health Sciences, Universiti Sains Malaysia, George Town, Malaysia 5Faculty of Medicine, Department of Anatomy, Histology, Universitas Airlangga Campus A, Surabaya, Indonesia *Corresponding Author: Maslichah Mafruchati. Department of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: maslichah-m [at] fkh.unair.ac.id Submitted: 28/07/2025 Revised: 29/10/2025 Accepted: 12/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Propolis resin could be used as a therapeutic agent for chicken embryos, but its toxicity must be evaluated before application in livestock. Aim: This study aimed to investigate the toxicity and growth-promoting properties of propolis resin on the embryo of the chicken. Methods: Fertilized broiler eggs were used as experimental samples by being injected with propolis resin. The solution volume was predetermined to be 1 ml per egg for each treatment dose. Results: The result showed that the morphology of chicken eggs was viable after propolis resin injection on days 13–21 after inoculation. Moreover, 62.5 mg/egg was the effective dose, 125 mg/egg was the Toxic dose 250 mg/egg was the lethal dose. A mid-range dose (62.5–125 mg/egg) was associated with higher embryo weight and wing bone length compared to controls, though differences require further confirmation. Conclusion: Propolis resin at intermediate concentration can boost embryonic growth, but higher doses rapidly induce toxicity. These findings emphasize the importance of dose optimization and chemical profiling before in-ovo implementation. Further studies across poultry species are needed to evaluate antiviral and growth-promoting effects while ensuring safety. Keywords: Chicken embryo, Food production, Propolis resin, Toxicity. IntroductionPropolis resin, a substance collected from plant exudates, is a biodegradable substance. Propolis resin was a bit cheaper to obtain in the sample of the study than honey or royal jelly. Moreover, the study conducted by Aygun (2016) stated that propolis resin has many active compounds that could give health benefits, forming a complex substance that is suitable to be used as a promising candidate for new drugs. Even though it is cheaper than honey or royal jelly, the antimicrobial effect was as significant as theirs, but it needed a different dosage. Its antimicrobial, anti-inflammatory, antioxidant, and antiviral properties have been documented in a variety of livestock applications, including dietary supplementation in laying hens and in vitro antiviral assays against herpes and influenza viruses (Belmehdi et al., 2023). Despite this wealth of data in adult birds and cell culture systems, the in-ovo use of propolis resin remains poorly defined, particularly concerning safe dosing thresholds and its capacity to enhance embryonic development (Batkowska et al., 2018). Previous studies have demonstrated that low-dose propolis resin can support immunomodulation when co-administered with vaccines, while higher concentrations may impart toxicity to embryonic tissues (Saeed et al., 2017; Belmehdi et al., 2023). However, no systematic investigation has yet delineated the dose-dependent window between growth-promoting and toxic or lethal effects in broiler embryos. Without this critical information, attempts to harness propolis resin for in-ovo immunization or growth enhancement risk suboptimal outcomes or unintended embryotoxicity. This study addresses that gap by administering graded doses of standardized ethanolic propolis resin extract directly into the allantoic cavity of 12-day-old broiler embryos. This study tries to evaluate three key endpoints: embryo viability, morphological integrity, and quantitative measures of body weight and skeletal development, to identify the effective dose (ED), toxic dose (TD), and lethal dose (LD) of propolis resin in ovo. By establishing these parameters, this study aims to provide the first comprehensive dosing framework for safely leveraging propolis resin’s bioactive properties during avian embryogenesis. Few studies have been done in the past about the effects of propolis resin treatment on chicken embryos still in their eggs. One of these studies was a study conducted by Saeed et al. (2017), which stated that flavonoids are important substances used as adjuvants in the prevention of viruses in chickens vaccinated with active or inactive vaccines. On the other hand, a trial conducted by Belmehdi et al. (2023) stated that propolis resin has a toxic substance if it is given to the embryo of a chicken in a certain dose. The phenol, flavonoids, and alkaloids in propolis resin could harm the embryo of a chicken at a certain dose (Aygun 2016). The correct dosage of propolis resin for the embryo of broiler chicken to avoid being intoxicated, as well as to boost its growth, could become a good research gap for this study. The primary purpose of this investigation is to determine the safe and effective in-ovo dose range of propolis resin for broiler embryos, balancing its potential growth-promoting effects with the risk of embryotoxicity. Propolis resin is a complex resinous mixture produced by honeybees from plant exudates, containing over 300 identified compounds, including flavonoids (e.g., pinocembrin, galangin, chrysin), phenolic acids (e.g., caffeic acid, ferulic acid), terpenes, and aromatic aldehydes (Belmehdi et al., 2023). These bioactives are known for antimicrobial, antioxidant, and immunomodulatory properties, but at high concentrations, some, particularly caffeic acid phenethyl ester (CAPE) and certain flavonoids, could induce oxidative stress or interfere with embryonic development. The chemical composition of propolis resin varies with botanical origin and season (Bertolucci et al., 2025). For example, temperate-zone propolis resin is often rich in flavonoids and phenolic acids, while tropical propolis resin may contain higher levels of prenylated compounds. Seasonal variation can alter the concentration of active constituents by up to 40%, potentially influencing both efficacy and toxicity (Saeed et al., 2017). Therefore, this study incorporates chemical profiling of the propolis resin batch used to contextualize the observed biological effects. Based on that research gap, this study has a purpose to investigate the correct dosage of propolis resin. Materials and MethodsSource and preparationThe propolis resin used was a commercial ethanolic extract (70% ethanol) sourced from East Java, Indonesia, harvested in the dry season (July). The extract was filtered, evaporated to dryness, and reconstituted in sterile phosphate-buffered saline (PBS) with 1% ethanol as a carrier. Concentrations were verified via High-Performance Liquid Chromatography against standards for pinocembrin, galangin, chrysin, caffeic acid, and CAPE. To determine the effect of Propolis resin products on the development of chicken embryos, an experiment was designed by administering a series of propolis resin product doses to several chicken eggs that were incubated (Gniewosz et al., 2022). The propolis resin dose and embryo mortality rate could be measured by using a logistic sigmoidal dose. This study evaluated six dosages of propolis resin to be injected into each of the eggs of the broiler. This equation allows this study to predict the level of embryonic mortality at various propolis resin doses within the tested range, as well as help to determine a safe and effective dose range. The equation is as follows:
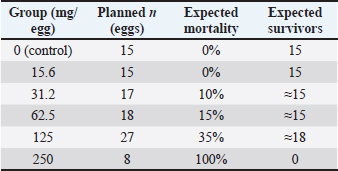
D=propolis resin dose (mg/egg) M (D)=predicted percentage of embryo mortality Mmax=100 (maximum mortality) D50 ≈ 180 (estimated dose that causes 50% embryo mortality) k ≈ 0.04 (slope factor of the curve) The six dosages of the propolis resin are as follows: D=15.6 mg, M (D) ≈ 0% D=31.2 mg, M (D) ≈ 10% D=125 mg, M (D) ≈ 35% D=250 mg, M (D) ≈ 100% According to a study by Aygun (2016), a wide dose range of 15.6–250 mg allows researchers to identify the therapeutic window of propolis resin in broiler chicken embryos, namely the lowest dose that provides a positive growth-promoting effect without causing toxic effects. The dose of 15.6–250 mg/broiler egg is an amount that begins to cause tissue damage in egg embryos and the risk of embryo death. The initial dose using this dose range refers to in-ovo studies by Batkowska et al. (2018) stated that natural bioactives are reported to be safe in poultry. Medium doses (62.5–125 mg/broiler egg) are used to test the potential for increasing growth while detecting early toxicity. The highest dose (250 mg/egg) is used to determine the lethal dose point as a safety reference. This study observes 100 eggs of the broiler with different weights, ranging from 54.8 to 64.5 gm from local poultry of Kediri, Indonesia. A total of 100 fertile eggs were selected, weighing between 54.8 g and 64.5 g. This range was deliberately chosen to maintain uniformity in the size and volume of the allantoic cavity, allowing the in-ovo dose of propolis resin to be distributed proportionally to the egg mass. The sampling process was conducted using a stratified randomized design based on weight class, so that each treatment group received eggs with an equal weight distribution. This approach ensured that any observed differences in results were primarily due to the dose treatment, rather than variations in egg size or quality. Each egg was injected with propolis resin at six dosages for 21 days (Ma et al., 2022). After 21 days of incubation, the embryos were carefully removed from eggshells, euthanized, and measured. They were preserved using alcohol and inserted into a bottle containing 1% potassium hydroxide solution. They were renamed in the bottle until the skeleton and muscles became transparent. Subsequently, the embryos were immersed in a dish containing Alizarin Red dye solution. After approximately 3 hours, the skeleton developed a distinct dark red to purple coloration. The number of eggs administered to each group of treatment is shown in Table 1. Table 1. The number of eggs administered to each group of treatment.
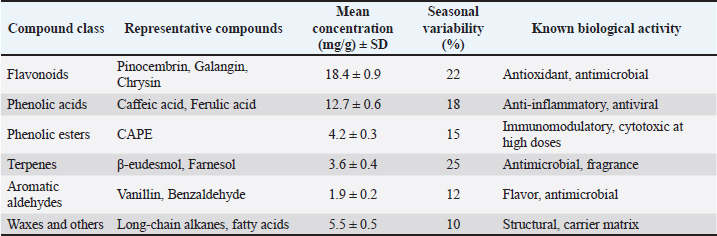
The embryos were then injected with a potassium hydroxide solution at a 2% concentration. The embryos underwent this treatment for a while. All of the muscle tissue turned transparent after about 30 minutes. The inward movement was made with the embryos. The following hour will be spent using the clarification solution. The embryos were then submerged in a pure glycerin solution that had been treated with thymol, watched, and their development was compared to the embryonic skeletal system (Przybyłek and Karpiński, 2019). This study followed ethical conduct of research and passed the ethical clearance test through the Airlangga University Faculty of Dentistry Laboratory. The certificate of ethical test was 1.KE.102.08.2022. The certificate of ethical clearance has been uploaded to the Zenodo repository. Chemical compositionChemical composition of the propolis resin batch used is shown in Table 2. Table 2. Chemical composition of the propolis resin batch used.
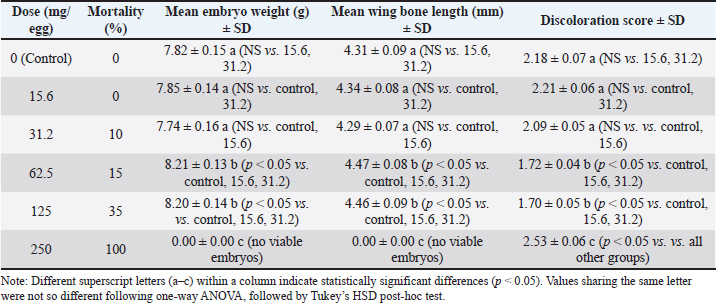
Dilution mediumAll dilutions were prepared in PBS + 1% ethanol to ensure solubility of hydrophobic compounds. The control group received PBS + 1% ethanol only. Paraffin justificationLiquid paraffin was applied to seal the injection site, preventing microbial contamination and dehydration during incubation. Incubation parameters Eggs were incubated at 37.8°C, with a relative humidity of 55%–60%, and were turned automatically every 2 hours. All eggs were sourced from a single Specific Pathogen Free (SPF)-certified broiler breeder flock. Dosage rationaleDoses were selected based on prior in-ovo studies of plant-derived bioactives, adjusted for egg mass, and spanning a range from sub-physiological to potentially toxic levels. For clarity, the ED was defined as the lowest concentration of propolis resin that produced significant improvements in embryonic weight and wing bone length without histopathological damage (62.5 mg/egg). The TD was defined as the concentration associated with histopathological lesions in liver and heart tissues and elevated mortality (125 mg/egg). The LD was defined as the concentration that resulted in 100% embryo mortality with severe necrosis and organ failure (250 mg/egg). These definitions were based on observed outcomes in this study and aligned with prior in-ovo bioactive compound research. Statistical analysisData were analyzed using one-way ANOVA with Tukey’s HSD post-hoc test. Embryonic weight was included as a covariate in Analysis of Covariance models. Mortality data were analyzed using logistic regression to estimate LD50. Significance was set at p < 0.05. Data were presented using the percentage of embryonic mortality. This study also presented body weight gain and the length of the wing bone of broiler embryos after being treated with propolis resin. The data analysis presented in this study involved approximately 20 eggs of varying weights. The weight range shown, 54.8–64.5 gm, reflects the standard weight of commercial broiler eggs used in in-ovo studies. This range ensures a consistent allantoic cavity volume for a 1 ml propolis resin injection, thus maintaining a proportional propolis resin dose per gram of egg. Of the 110 eggs, 20 were selected for analytical sampling, which measured morphology, embryo weight, wing bone length, and color change scores. This was in accordance with the principle of stratified random design sampling, where the subset displayed for in-depth analysis results was drawn from a homogeneous population. This study used a total of 100 fertile broiler eggs allocated across six treatment groups (n=15–18/group), except n=8 at the highest dose. Group sizes were chosen to balance ethical reduction of animal use with sufficient replication for dose-response modeling. Post-hoc power analysis at α=0.05 indicated that the study was well powered (>0.95) to detect medium to large effects in embryo weight and wing bone length at the effective (62.5 mg/egg) and toxic (125 mg/egg) doses (Cohen’s d=1.95–2.8, n=15/group). Mortality comparison showed moderate power (0.6–0.75) for detecting 15% differences and high power (0.85–0.95) for detecting 35% differences. Logistic regression for LD50 estimation was supported by six dose levels spanning 0%–100% mortality, ensuring stable parameter estimation. Stratified randomization by egg weight and inclusion of egg mass as a covariate in ANCOVA further increased statistical efficiency. Discoloration was scored on a 0–5 ordinal scale by two independent observers who were blinded to treatment allocation. The scoring criteria were predefined based on three aspects. First, the intensity of the color change. Second, the extent of the affected area. Third, distribution of pigmentation. When the two observers’ scores differed by more than one point, a consensus score was reached after joint review. This approach ensured consistency and minimized subjective bias in the evaluation of tissue discoloration (Altun and Turkyilmaz, 2024). To explore the relationship between key variables, correlation analysis was performed using Pearson’s or Spearman’s method, depending on data distribution. Specifically, correlations were tested between embryo weight, wing bone length, discoloration core, and histopathology score across treatment groups. This analysis helped identify dose-dependent trends and associations between morphological and tissue-level outcomes (Aygun 2016). Ethical approvalThis paper has not used any animals or humans as a sample for the study. This study used 100 broiler eggs from local farms of Kediri, East Java, Indonesia. The eggs used in this study came from a broiler farm in Kediri, East Java, which holds SPF certification to ensure they are free from certain pathogens and have a uniform genetic background. This source selection aimed to minimize biological variation that could affect the experimental results, ensuring that each egg had an equivalent initial condition. This study followed the ethical principle of research, which was to avoid any harm to humans and animals. This study conducted an ethical test of using animals as samples through the Airlangga University Faculty of Dentistry Laboratory. The certificate of ethical test was 1.KE.102.08.2022. ResultsEmbryo growth and mortalityThe proportion of viable chicken eggs that sprouted following propolis resin injections is shown in Table 2 of the study's findings. Table 2 summarizes the effects of increasing propolis resin doses on mortality, embryo weight, wing bone length, and discoloration score. The chemical composition of the propolis resin batch is summarized in Table 2, showing flavonoids as the dominant class (18.4 ± 0.9 mg/g), followed by phenolic acid and esters. Statistical summary of key outcomes is shown in Table 3. Table 3. Statistical summary of key outcomes.
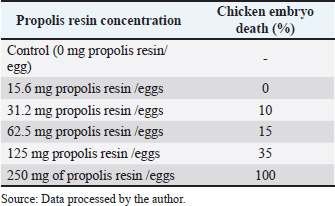
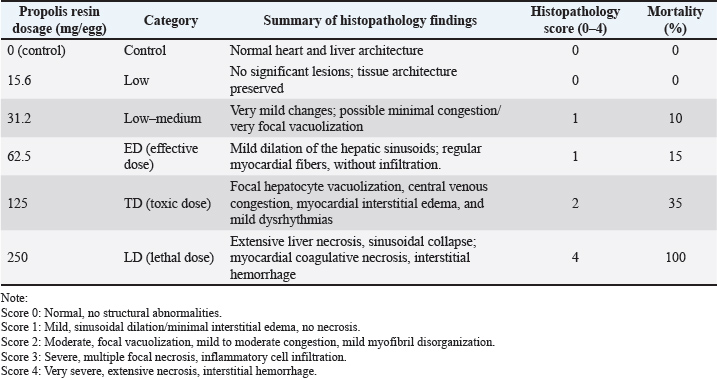
Different superscript letters within a column indicate significant differences at p < 0.05. Histology findingsLiver and heart tissues from embryos in the 0, 62.5, 125, and 250 mg/egg groups showed dose-dependent changes. At 62.5 mg/egg, hepatocytes appeared normal with mild sinusoidal dilation. At 125 mg/egg, focal hepatocellular vacuolation and mild congestion were observed. At 250 mg/egg, severe hepatocyte necrosis, sinusoidal collapse, and myocardial fiber degeneration were evident. Chicken embryo death rate after propolis resin injection on day 21 is shown in Table 4. Table 4. Chicken embryo death rate after propolis resin injection on day 21.
Observation of embryo morphology in each treatment is shown in Figure 1.
Fig. 1. Observation of embryo morphology in each treatment. Source: Data processed by the author. A: control (0 mg of propolis resin /egg). B: 15.6 mg of propolis resin /egg. C: 31.2 mg of propolis resin /egg. D: 62.5 mg propolis resin /egg. E:125 mg propolis resin /egg. F: 250 mg propolis resin /egg. Based on the study conducted by Chaves-Hernández et al. (2024), the score of discoloration was 0=no discoloration, up to 5=severe discoloration. Table 3 shows no significant discoloration after being injected with propolis resin. The "discoloration" score in 21-day-old broiler embryos after 20 eggs, which were used in this study, were injected with propolis resin to assess the extent to which tissue color changed visually. This color change was scored on an ordinal scale (0–5), with low scores indicating barely any color change, while high scores indicated the appearance of dark, pale, or distinct spots. The discoloration score encompasses three aspects: the strength of the color change, the extent of the affected area, and the distribution of pigmentation. This provides a comprehensive overview of the compound's impact on the tissue (Belmehdi et al., 2023). The result of the histopathology scale is shown in Table 5. Table 5. Result of histopathology scale.
DiscussionToxicity and mortality dose of propolis resin to embryo of broilerThe result in Table 3 shows that discoloration scores for the 15.6 mg propolis resin dose were 2.21 ± 0.06 at 15.6 mg/egg, indicating mild to moderate discoloration. Meanwhile, Tables 4 and 5 demonstrate the progressive increase in mortality and histopathological severity at higher doses. A score around 2 indicates that the embryo is experiencing discoloration concentrated in the wing or thorax area. It is usually due to a slight decrease in superficial blood vessel saturation or a local reaction to the bioactive compound. At the 31.2 mg dose, scores increased slightly (1.76–2.13), indicating a consistent shift to fainter pigmentation or small bluish patches. This likely correlates with an increased microinflammatory response in the embryonic tissue. The mid-range dose (62.5 mg/egg) optimized growth without histological damage, suggesting a hormetic effect. Toxicity at higher doses is likely mediated by CAPE and certain flavonoids, which can induce mitochondrial dysfunction and ROS generation in embryonic tissues. As shown in Table 2, flavonoids and phenolic acids were the most abundant compounds, supporting the observed antioxidant and immunomodulatory effects at intermediate doses. Seasonal variability in propolis resin composition underscores the need for batch-specific chemical profiling before in-ovo application. The inclusion of histology strengthens the evidence for dose-dependent toxicity mechanisms. Administration of propolis resin via in ovo injection demonstrated a dose-dependent biological response, with significant differences appearing in the 62.5 mg/egg and 125 mg/egg groups. Both doses resulted in statistically significant increases in embryo weight and wing bone length compared to the control and low-dose groups, while simultaneously reducing tissue color change scores. These findings indicate that propolis at intermediate concentrations can improve embryo morphological quality without drastically increasing the risk of mortality. On the other hand, administration of propolis resin at a dose of 250 mg/egg caused all embryos to die before reaching 21 days of incubation. Although morphometric parameters could not be assessed, color change scores were recorded through visual observation before death. The visual observation result showed the highest color change scores on the embryo at a dose of 250 mg/egg compared to all other groups. It means that this dose is systematically toxic and causes acute physiological disturbances. It should be emphasized that non-significant differences between groups are explicitly stated in the results table, in accordance with transparent statistical reporting principles. The control, 15.6 mg/egg, and 31.2 mg/egg groups showed similar values for all parameters, and there were no significant differences between them. The "NS" designation in the table ensures that readers do not misinterpret the results as statistically significant. Overall, these results indicate that propolis resin has potential as a bioactive agent in the embryonic phase, but its effectiveness is highly dependent on the dose used. Doses below 62.5 mg/egg had no significant effect, while doses above 125 mg/egg pose a high risk to embryo survival. Therefore, selecting the optimal dose is key to implementing propolis as a nutritional or immunomodulatory intervention in the early stages of broiler chicken development. The research results presented in (Xu et al., 2022) served as the basis for the molecular research findings in this study. The study demonstrated that flavonoids, the primary components of propolis, including pinocembrin, galangin, and chrysin, can activate the Nuclear Factor Erythroid 2–Related Factor 2 pathway, leading to an increase in the production of endogenous antioxidant enzymes such as Superoxide Dismutase, Catalase, and glutathione peroxidase (GPx). This increase in enzymes can neutralize free radicals and maintain normal cell membranes, thereby reducing oxidative damage during incubation. Furthermore, flavonoids can also reduce the production of pro-inflammatory cytokines and prevent impaired embryonic tissue differentiation. The mechanism in this study aligns with the results, where treatment of chicken eggs with propolis resin at a dose of 62.5 mg/egg significantly increased embryo weight and wing bone length without causing significant histological damage. The color change score in the embryos was also lower than in the high-dose group. However, it should be emphasized that the study conducted by Xu et al. (2022) stated that the protective effects of Nrf2 and inhibition of NF-κB have limits to the biological tolerance of embryos. At very high doses of flavonoids, some compounds can become pro-oxidants through redox cycling mechanisms, thus triggering increased ROS, mitochondrial dysfunction, decreased ATP production, and activation of the apoptosis pathway. This also occurred in this paper's study, where doses of propolis resin of 125 mg/egg and 250 mg/egg caused a significant increase in mortality, characterized by severe histological damage to the liver and heart, as well as the highest color change scores. The success of in-ovo application of propolis resin depends on achieving a dose that maximizes the activation of the protective Nrf2 pathway and suppression of NF-κB, while avoiding the concentration threshold that triggers toxic effects. Based on the study conducted by Sadekuzzaman et al. (2024), discoloration of the embryo of poultry occurs due to impaired microcirculation, methemoglobin accumulation, or oxidation of heme pigments in embryonic tissue. Propolis resin, although rich in flavonoids and phenolics, at certain doses can trigger the accumulation of free radicals and reduce capillary membrane resistance (Altun and Turkyilmaz, 2024). Discoloration scores help researchers quantify the sublethal toxicity threshold and design optimal doses. By analyzing the pattern of scores across doses, we can determine the therapeutic window of propolis resin, where maximal growth effects are achieved without triggering abnormal color changes, and formulate recommendations for safer in-ovo use. This is in line with the study conducted by Batkowska et al. (2018), who reported that Japanese quail eggs sprayed with 15% alcohol propolis resin extract as a natural disinfectant showed a higher survival rate of quail chicks during the first 2 weeks than other groups. In this study, the treatment group of 600 Japanese quail eggs was divided into 4 groups, namely negative control or without propolis resin injection, formaldehyde control (C), using formalin fumigation, alcohol (A), by spraying 96% alcohol on the eggs, and propolis resin (P), by spraying 15% alcohol propolis resin. The group treated with 15% propolis resin showed a safe disinfectant effect on Japanese quail embryos, compared to disinfectants containing toxic formaldehyde. Histopathology scale of propolis resin to the organ of broiler embryoTable 5 shows that increasing the dose of propolis resin in ovo causes progressive structural changes in the liver and heart of broiler embryos. At the effective dose of approximately 62.5 mg/egg, the only visible changes are mild dilation of the liver sinusoids, while the heart muscle fibers remain regular without signs of inflammatory cell infiltration. However, at a high dose of 125 mg/egg, focal hepatocyte vacuolization, central venous congestion, and interstitial edema of the myocardium begin to appear. These conditions peak at a lethal dose of 250 mg/egg, with extensive liver necrosis, sinusoidal collapse, and coagulative necrosis of the heart muscle fibers accompanied by interstitial hemorrhage. A study conducted by Kurek-Górecka et al. (2024) stated that propolis resin molecules contain a complex mixture of bioactive compounds, particularly flavonoids such as pinocembrin, galangin, chrysin, and phenolic acids. These compounds play a crucial role in protecting cells through antioxidant mechanisms, neutralizing free radicals, and preventing lipid peroxidation in cell membranes (Oliveira et al., 2021). This activity is also associated with increased activity of the NrF2 signaling pathway, which triggers the production of endogenous antioxidant enzymes such as superoxide dismutase and (GPx). In line with this, another study conducted by Kurek-Górecka et al. (2024) shows that CAPE and other phenolics have anti-inflammatory effects by inhibiting the activation of the Nuclear Factor Kappa – Light Chain Enhancer of Activated B Cells pathway. Thus, it can reduce the production of pro-inflammatory cytokines that can disrupt embryonic tissue differentiation (Shin et al., 2023). However, at high doses, some flavonoids can act as pro-oxidants through redox cycling mechanisms, triggering oxidative stress, impaired mitochondrial function, decreased Adenosine Triphosphate production, and even apoptosis. Another study by Daza-Leon et al. (2022) shows that CAPE can protect heart tissue from damage caused by oxidative stress and inflammation. This protective mechanism involves suppressing the production of pro-inflammatory cytokines, inhibiting NF-κB activation, and increasing the activity of the Nrf2 pathway, which triggers the expression of endogenous antioxidant enzymes. At medium doses, this effect is sufficient to maintain myocardial integrity and prevent muscle fiber disorganization, as seen in the 62.5 mg/egg group in Table 4. Conversely, at high doses, the accumulation of phenolic compounds can reverse to pro-oxidants, triggering a surge in ROS and mitochondrial dysfunction, so that the protective effect of CAPE is no longer able to offset the damage, as reflected in the highest histopathological scores in the 250 mg/egg group. ConclusionAccording to the above-mentioned result, it can be concluded that the lethal dose of propolis resin is 250 mg/egg, the toxic dose of propolis resin is 125 mg/egg, and the effective dose of propolis resin is 62.5 mg/egg. That exact toxic dose of propolis resin was obtained after the leg bone of the chicken's embryo was injected with propolis resin for 21 days using a different dose. Propolis resin exhibits a narrow therapeutic window in broiler embryo development. While moderate doses can enhance growth, higher doses induce significant mortality and tissue damage (Soares et al., 2022). Future studies should incorporate chemical profiling, histology, and covariate-adjusted statistical models to ensure reproducibility and safety in potential applications. It can be concluded that at the ED=62.5 mg/egg, there is a growth promotion on the embryos and no damage. At TD=125 mg/egg, there is a lesion and mortality in the embryos. At LD=250 mg/egg, there is a 100% mortality in embryos. On the other hand, the result showed that chicken eggs that sprouted after being injected with propolis resin were viable at days 13–21 after inoculation, as well as their shapes. It was mentioned that propolis resin could boost the growth of embryos. Further studies are needed using various types of embryos of poultry cell embryos to observe the toxicity, antiviral, and growth-boosting effects of propolis resin. Future study should refine dose spacing near the effective to toxic boundary, incorporate molecular biomarkers of oxidative stress and apoptosis, and extend testing to other poultry species and propolis chemotypes. By integrating histological, biochemical, and developmental endpoints, subsequent studies can better define the implementation potential of propolis resin as a safe, sustainable intervention in poultry health and productivity. AcknowledgmentThank you to the rector of Universitas Airlangga for approving the proposal of this study to be funded. We are also thankful to PASINBIS Universitas Airlangga for providing space to work on this paper. Conflict of interestThis study has no conflict of interest. FundingThis study was funded by the Universitas Airlangga as a Grant research through LPPM of Universitas Airlangga, Indonesia. Authors' contributionMM was done with funding acquisition, data curation, supervising, writing the draft of the manuscript, and methodology. LF was responsible for data curation, data visualization, and methodology. AKW was responsible for translating, data analysis, proofreading, reviewing, and editing. IIH proofread and reviewed the draft. JS was responsible for methodology and data curation. Data availabilityThe raw data used for this study were available online at Zenodo. The raw data were embryo development, appendices of tools and goods used in the laboratory, and supplementary data on germinating eggs. Link to the raw data: https://zenodo.org/record/7796694#.ZCuVFXZBx1s ReferencesAltun, N. and Turkyilmaz, A. 2024. Potential crown discoloration induced by the combination of various intracanal medicaments and scaffolds applied in regenerative endodontic therapy. Nigerian J. Clin. Pract. 27(7), 897–904. Aygun, A. 2016. The effects of in-ovo injection of propolis on egg hatchability and starter live performance of Japanese quails. Braz. J. Poultry. Sci. 18, 83–89. Batkowska, J., Al-Shammari, K.I.A., Lukasz, W., Nowakowicz-Debek, B. and Gryzinska, M. 2018. Evaluation of propolis extract as a disinfectant of Japanese quail (Coturnix coturnix japonica) hatching eggs. Poultry Sci. 97(7), 2372–2377. Belmehdi, O., El Menyiy, N., Bouyahya, A., El Baaboua, A., El Omari, N., Gallo, M., Montesano, D., Naviglio, D., Zengin, G., Skali Senhaji, N., Goh, B.H. and Abrini, J. 2023. Recent advances in the chemical composition and biological activities of propolis. Food. Rev. Int. 39(9), 6078–6128. Bertolucci, V., Ninomiya, A.F., Longato, G.B., Kaneko, L.O., Nonose, N., Scariot, P.P.M. and Messias, L.H.D. 2025. Bioactive compounds from propolis on bone homeostasis: a narrative review. Antioxidants 14(1), 81. Chaves-Hernández, A.J., Barquero-Calvo, E., Quesada-Vasquez, D. and Chacón-Díaz, C. 2024. Comamonas testosteroni as the cause of mortality in embryonated chicken eggs of breeding broiler hens in Costa Rica. Avian Pathol. 53(2), 124–133. Daza-Leon, C., Gomez, A.P., Álvarez-Mira, D., Carvajal-Diaz, L., Ramirez-Nieto, G., Sanchez, A., Vargas, J.I. and Betancourt, L. 2022. Characterization and evaluation of Colombian propolis on the intestinal integrity of broilers. Poultry. Sci. 101(12), 102159. Gniewosz, M., Pobiega, K., Olbryś, N., Kraśniewska, K. and Synowiec, A. 2022. The Effect of Ethanol Propolis Extracts on Inhibition of Growth of Fusarium solani on Hen Eggs. Appl. Sci. 13(1), 315. Kurek-Górecka, A., Kłósek, M., Pietsz, G., Balwierz, R., Olczyk, P. and Czuba, Z.P. 2024. Ethanolic extract of propolis and CAPE as cardioprotective agents against LPS and IFN-α stressed cardiovascular injury. Nutrients 16(5), 627. Ma, H., Liang, S., Wu, H., Du, C., Ren, Z., Yang, X. and Yang, X. 2022. Effects of in ovo feeding and dietary addition oils on growth performance and immune function of broiler chickens. Poultry Sci. 101(5), 101815. Oliveira, J.M.D.S., Cavalcanti, T.F.S., Leite, I.F., Dos Santos, D.M.R.C., Porto, I.C.C.D.M., De Aquino, F.L.T., Sonsin, A.F., Lins, R.M.L., Vitti, R.P., De Freitas, J.D., Barreto, E.D.O., De Souza, S.T., Kamiya, R.U., Do Nascimento, T.G. and Tonholo, J. 2021. Propolis in oral healthcare: antibacterial activity of a composite resin enriched with brazilian red propolis. Front. Pharmacol. 12, 787633. Przybyłek, I. and Karpiński, T.M. 2019. Antibacterial properties of propolis. Molecules 24(11), 2047. Sadekuzzaman, M., Miah, M.S., Parvin, R., Haque, M.E., Islam, T.R., Sigma, S.H., Hossain, M.G., Hayat, S., Hossain, M.T. and Islam, M.A. 2024. Pathological investigation, molecular characterization and first-time isolation of the predominant serotypes of fowl adenovirus (FAdV-D and E) from commercial poultry in Bangladesh. Front. Microbiol. 15, 1490255. Saeed, M., Arain, M.A., Kamboh, A.A., Memon, S.A., Umar, M., Rashid, M., Babazadeh, D., Abd El-hack, M.E. and Alagawany, M. 2017. Raw propolis as a promising feed additive in poultry nutrition: trends and advances. J. Anim. Health Prod. 5(4), 132–142. Shin, S.B., Lee, J.K. and Ko, M.J. 2023. Enhanced extraction of bioactive compounds from propolis (Apis mellifera L.) using subcritical water. Scientific Rep. 13(1), 15038. Soares, A.C.B., Brito, D.A.P., Soares, S.C.P., Gomes, K.S., Saldanha, G.K.M.S. and Soares, V.D.S. 2022. Maintenance of quality of eggs submitted to treatment with propolis extract and sanitizers. Acta. Scientiarum. Anim. Sci. 44. Xu, W., Lu, H., Yuan, Y., Deng, Z., Zheng, L. and Li, H. 2022. The antioxidant and anti-inflammatory effects of flavonoids from propolis via Nrf2 and NF-κB pathways. Foods 11(16), 2439. | ||
| How to Cite this Article |
| Pubmed Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II, Susanto J. Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Vet. J.. 2025; 15(12): 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 Web Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II, Susanto J. Toxicity and potential of propolis resin to boost embryo growth of broilers. https://www.openveterinaryjournal.com/?mno=273732 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.34 AMA (American Medical Association) Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II, Susanto J. Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Vet. J.. 2025; 15(12): 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 Vancouver/ICMJE Style Mafruchati M, Fadliyah L, Wardhana AK, Hassan II, Susanto J. Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 Harvard Style Mafruchati, M., Fadliyah, . L., Wardhana, . A. K., Hassan, . I. I. & Susanto, . J. (2025) Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Vet. J., 15 (12), 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 Turabian Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, Intan Idiana Hassan, and Joni Susanto. 2025. Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Veterinary Journal, 15 (12), 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 Chicago Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, Intan Idiana Hassan, and Joni Susanto. "Toxicity and potential of propolis resin to boost embryo growth of broilers." Open Veterinary Journal 15 (2025), 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 MLA (The Modern Language Association) Style Mafruchati, Maslichah, Lailatul Fadliyah, Akhmad Kusuma Wardhana, Intan Idiana Hassan, and Joni Susanto. "Toxicity and potential of propolis resin to boost embryo growth of broilers." Open Veterinary Journal 15.12 (2025), 6497-6505. Print. doi:10.5455/OVJ.2025.v15.i12.34 APA (American Psychological Association) Style Mafruchati, M., Fadliyah, . L., Wardhana, . A. K., Hassan, . I. I. & Susanto, . J. (2025) Toxicity and potential of propolis resin to boost embryo growth of broilers. Open Veterinary Journal, 15 (12), 6497-6505. doi:10.5455/OVJ.2025.v15.i12.34 |