| Research Article | ||
Open Vet. J.. 2025; 15(12): 6480-6486 Open Veterinary Journal, (2025), Vol. 15(12): 6480-6486 Research Article Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, IndonesiaHesti Rahayu1, Teguh Rianda1, Dwi Priyowidodo2*, Irkham Widiyono3, Vika Ichsania Ninditya21Postgraduate Student, Master of Veterinary Science Program, Faculty of Veterinary Medicine, Gadjah Mada University, Yogyakarta, Indonesia 2Department of Parasitology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 3Department of Internal Medicine, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Dwi Priyowidodo. Department of Parasitology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. Email: priyo [at] ugm.ac.id Submitted:28/07/2025 Revised: 22/10/2025 Accepted: 03/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
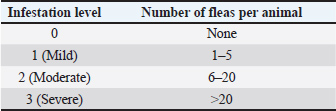
AbstractBackground: Mycoplasma haemofelis infection is considered an important feline disease and is suspected to be transmitted via the ectoparasitic vector Ctenocephalides felis, particularly in tropical areas. Aim: This study aimed to detect M. haemofelis at the molecular level in cats naturally infested with C. felis in Padang City, West Sumatra, Indonesia. Methods: A total of 61 cats were examined to assess their clinical condition and flea infestation levels, followed by blood sampling for polymerase chain reaction analysis targeting the 16S rRNA gene of M. haemofelis. Results: The PCR results revealed a high prevalence of infection (72.13%; 44/61). The level of flea infestation was positively associated with the frequency of M. haemofelis infection, with rates of 40.0%, 60.0%, and 83.3% in mild, moderate, and severe infestations, respectively. Statistical analysis revealed a significant association (p < 0.05) between flea infestation level and infection status. Cats kept in multi-cat households were more frequently infected than those kept in single-cat households. No significant associations were found between infection occurrence and age, sex, or maintenance pattern. Conclusion: A high prevalence of M. haemofelis was detected in cats infested with C. felis in Padang City, with a significant association with flea infestation level and housing pattern. Keywords: Mycoplasma haemofelis, Ctenocephalides felis, Cat, PCR, Padang. IntroductionArthropod-borne vector-borne diseases represent a global health issue because they involve various important pathogens, including bacteria, viruses, and protozoa. Ctenocephalides felis is one of the arthropod vectors that play a significant role in transmitting infectious agents in animals (Otranto et al., 2025; Páez-Triana et al., 2025). This species is recognized as a primary vector for Bartonella henselae and Rickettsia felis, both of which are important zoonotic pathogens that can cause disease in humans and animals (Moore et al., 2024a). Ctenocephalides felis is a hematophagous ectoparasite belonging to the family Pulicidae and is widely distributed worldwide. Heavy infestations in cats can result in clinical manifestations, such as flea allergic dermatitis, secondary skin lesions due to scratching, opportunistic infections, and physiological stress that can lead to immunosuppression (Pereira et al., 2025). In addition to its role as an ectoparasite, C. felis also harbors the pathogenic bacterium Mycoplasma haemofelis (Zarea et al., 2023; Razgūnaitė et al., 2024). Mycoplasma haemofelis is a hemotropic bacterium belonging to the genus Mycoplasma, previously known as Haemobartonella felis. This bacterium lacks a cell wall, making it relatively resistant to various antimicrobial agents and capable of surviving under diverse environmental conditions (Cortés Sánchez, 2022). Morphologically, M. haemofelis adheres to the outer surface of erythrocytes without penetrating the cell membrane and is recognized as the most pathogenic hemoplasma species in cats (Yasmin et al., 2022). The clinical manifestations of M. haemofelis infection in cats can range from subclinical to severe hemolytic anemia, particularly in cats with compromised immune systems, which are often accompanied by lethargy, pale mucous membranes, anorexia, and weight loss (Lima, 2025). Although the transmission routes of M. haemofelis are not yet fully understood, several mechanisms are suspected to be involved, including blood transfusion, aggressive interactions between individuals, and the role of ectoparasite vectors, such as C. felis (Tasker et al., 2018; Pimpjong et al., 2025; Shi et al., 2025). Although M. haemofelis DNA was detected within fleas in recent molecular studies, its prevalence was low, and some results may represent nonspecific amplification (Moore et al., 2024b). The detection of M. haemofelis DNA in fleas further supports its potential role as a mechanical vector for the transmission of this pathogen (Mifsud et al., 2020; Razgūnaitė et al., 2024). Flea infestations tend to increase in warm and humid tropical environments, accelerating the life cycle of C. felis and increasing the risk of pathogen transmission (Pereira et al., 2025). Padang City, which has a tropical climate with an average temperature of 25 °C and humidity of 80% (BPS (Statistics Indonesia), 2024), provides an optimal environment for C. felis development, potentially increasing the risk of M. haemofelis transmission among the local cat population. To date, no data are available regarding the occurrence of M. haemofelis infection in cats infested with C. felis in Padang City. Therefore, this study aimed to molecularly detect M. haemofelis in cats infested with C. felis in Padang, West Sumatra, Indonesia. The results of this study are expected to contribute to vector-borne disease control efforts and to improve overall animal health. Materials and MethodsThis study included 61 cats naturally infested with C. felis, originating from Padang City, West Sumatra. Each cat underwent a thorough physical examination to assess its clinical condition and determine the degree of flea infestation. The study population comprised 57 owned cats and 4 cats from local shelters. Ctenocephalides felis infestation was assessed using the thumb counting method, in which fleas were counted through visual inspection within 3 minutes by manually parting the hair at predetermined predilection sites, including the dorsal midline, inguinal region, axillae, flanks, and tail base (Fox et al., 1969; Gregory et al., 1995). According to Marchiondo et al. (2007), the infestation level was classified as mild, moderate, or severe based on the total number of fleas found on the animal’s body. The flea infestation levels based on flea counts are presented in Table 1. Table 1. Levels of flea infestation (Marchiondo et al., 2007).
Blood samples (0.5 ml) were collected from the cephalic vein of each cat in ethylenediaminetetraacetic acid (EDTA) tubes following the standard procedures described by Reynolds et al. (2007). Genomic DNA was extracted using the Genomic DNA Mini Kit (Blood/Cultured Cell) (Geneaid Biotech Ltd., Taiwan) according to the manufacturer’s instructions. The extracted DNA was used as a template for polymerase chain reaction (PCR), employing specific primers designed based on the 16S rRNA gene sequence of H. felis from GenBank accession number U95297.1, yielding an amplicon of approximately 590 bp. PCR amplification consisted of an initial denaturation at 95°C for 5 minutes, followed by 30 cycles of denaturation at 94°C for 1 minutes, annealing at 56°C for 30 seconds, extension at 72°C for 30 seconds, and a final extension at 72°C for 10 minutes. PCR was performed using a Biorad PTC Tempo Thermal Cycler, California, USA. The PCR products were electrophoresed on a 1.5% agarose gel and visualized using a UV transilluminator (UVP, Upland, CA, USA). This protocol follows previously published molecular detection methods for M. haemofelis (Imre et al., 2020). Statistical analysisThe data obtained were analyzed descriptively to assess flea infestation levels and the frequency of cats infected with M. haemofelis based on polymerase chain reaction results. Distribution analysis between PCR-positive and PCR-negative cats for M. haemofelis was performed using a chi-square χ² test, considering flea infestation level C. felis sex, age, housing system, and number of cats per household. In addition, logistic regression analysis was performed to evaluate the strength of association between these risk factors and M. haemofelis infection. All analyses were conducted at a 95% confidence level (p<0.05). Using the IBM SPSS Statistics software version 26.0. Ethical approvalThis study was approved by the Ethics Committee of the Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia approval number: 39/EC-FKH/int./2025). ResultsThis study included 61 cats naturally infested with C. felis. The 16S rRNA gene of M. haemofelis was detected in 44 of 61 blood samples (72.13%) using conventional PCR. Figure 1 shows the results of the conventional PCR electrophoresis for M. haemofelis.
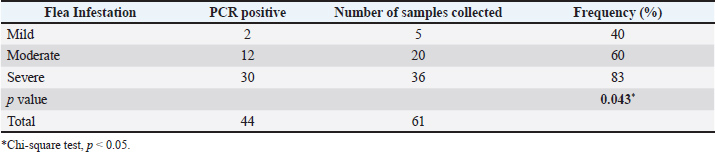
Fig. 1. Conventional PCR electrophoresis on 1.5% agarose gel. The distribution of flea infestation levels was predominant in the severe category (59.0%; 36/61), followed by the moderate (32.8%; 20/61) and mild (8.2%; 5/61) categories. Accordingly, the prevalence of M. haemofelis infection increased progressively in the mild (40%; 2/5), moderate (60%; 12/20), and severe (83.3%; 30/36) categories. No infections were observed in cats without flea infestation (Table 2). Table 2. Frequency of M. haemofelis infection based on flea infestation levels (n=61).
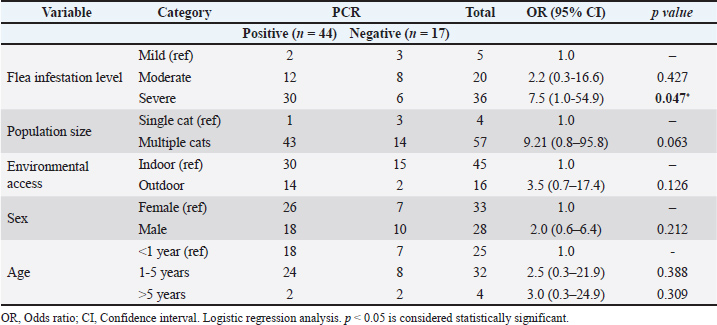
Analysis of the general characteristics showed that the samples included flea infestation level, various age groups, sexes, housing systems, and household cat populations, which were categorized as single-cat and multi-cat households. Table 3 presents the association analysis between M. haemofelis incidence and sample characteristics. Cats with severe infestation had higher odds of infection than those with mild infestation (OR=7.50, 95% CI: 1.02–54.99, p=0.047). Sex, age, and housing system were not significantly associated with M. haemofelis infection with the occurrence of infection (p > 0.05). In contrast, the household cat population showed a tendency toward increased risk, with cats from many-cat households having higher odds than those from few-cat households (OR=9.21, 95% CI: 0.89–95.86, p=0.063). Table 3. Logistic regression analysis of characteristics of cats associated with M. haemofelis infection.
DiscussionThe present study revealed a high prevalence (72.13%) of M. haemofelis infection among cats naturally infested with C. felis. This prevalence is considerably higher than that in China (52.5%) (Shi et al., 2025), Romania (23.7%) (Imre et al., 2020), the Czech Republic (12.7%) (Lobo et al., 2021), Brazil (8.4%) (Melo TB de et al., 2023), Iran (2.2%) (Hoseinpoor et al., 2024), and Türkiye (1.74%) (Altay et al., 2025). The high prevalence observed in this study is most likely influenced by homogeneous population characteristics, as all sampled cats were infested with C. felis, which are suspected to act as potential vectors for M. haemofelis (Abdullah et al., 2019). A significant association was observed between the severity of flea infestation and M. haemofelis infection (p=0.043). The infection rate increased from 40% in mild infestations to 83.3% in severe infestations. This pattern may be associated with the tropical humid climate of Padang City, which is characterized by high rainfall and relatively stable humidity throughout the year, which supports the survival of C. felis and increases the risk of infestation in cats (Pereira et al., 2025). A notable finding of this study was that cats with severe flea infestations were more likely to be infected with M. haemofelis than those with mild infestations (OR=7.50, 95% CI: 1.02–54.99, p=0.047). These results support the concept that a higher ectoparasite burden elevates the likelihood of hemotropic pathogen transmission (Altay et al., 2025; Herrera León, 2025) and are consistent with previous studies highlighting the role of fleas as mechanical vectors of M. haemofelis (Abdullah et al., 2019; Mifsud et al., 2020; Razgūnaitė et al., 2024). Berzina et al. (2021) reported that most infected cats did not receive routine antiparasitic treatment. Lappin (2018) noted that heavy infestations can cause skin lesions and immunosuppression, thereby increasing the risk of secondary infections. Although flea-mediated transmission is possible, its efficiency is low (Woods et al., 2005). While direct blood transmission is the primary route, flea infestation remains an important indicator of infection risk (Tasker et al., 2018). No significant associations were found between infection and sex, age, or housing system of cats. This finding is consistent with the report by Berzina et al. (2021) but differs from other studies that reported an increased risk of aggressive behavior in male cats (Pekel and Duru, 2022; Herrera León, 2025) and adult cats (Lobová et al., 2021; Lima, 2025). Interestingly, cats living in multi-cat households showed a tendency toward increased (OR=9.21, 95% CI: 0.89–95.86, p=0.063), possibly due to higher ectoparasite infestation and pathogen transmission potential in dense populations (Tasker et al., 2018; Kamyingkird et al., 2021). Studies on M. haemofelis infection in Indonesia remain limited and are generally restricted to individual case reports without population-based incidence data. Several reports have included cases in Bandung (Satriawan and Octaviani, 2021), two cases in Bali (Tjokorda Istri Agung Pradnya Dewi Pemayun et al., 2024; Billa et al., 2024), and a case of hypoproteinemia in Yogyakarta (Jayanti et al., 2023). Most of these reports relied on microscopic examination of blood smears for diagnosis, which has limited sensitivity and specificity compared with molecular methods. Therefore, the use of 16S rRNA gene-based PCR in this study provides significant diagnostic advantages and serves as an initial contribution to population-based prevalence data on cats infested with C. felis in Indonesia. The diagnostic accuracy strongly depends on the method used. Several studies have demonstrated that PCR has significant advantages over blood smear examination. Sagare et al. (2024) reported a prevalence of M. haemofelis of 5.4% using blood smears, which increased to 13.5% when PCR was used. In Brazil, a difference was observed between conventional PCR (15.6%) and quantitative PCR (28.6%) (Herrera León, 2025). Therefore, molecular methods are essential tools for detecting subclinical cases that are often missed by microscopic examinations (Cortés Sanchez, 2022). The prevalence of M. haemofelis infection exceeding 72.1% in cats infested with C. felis highlights the importance of ectoparasite control and routine molecular screening, even in animals without clinical signs. These findings support the need for population-based preventive strategies and environmental management to control M. haemofelis infections. The cross-sectional design did not allow for assessing the causal relationship between flea infestation and M. haemofelis infection. The identification focused solely on one hemoplasma species without detecting possible coinfections with Candidatus M. haemominutum or Candidatus M. turicensis. Immunological status, presence of other viruses such as FIV, FeLV, and environmental factors were not further analyzed due to data limitations. Longitudinal studies and multigenus molecular approaches are highly recommended to improve our understanding of hemoplasma epidemiology and transmission dynamics. Despite these limitations, this study provides the first molecular evidence of M. haemofelis infection in naturally infected cats with C. felis in Indonesia. Previous national reports were restricted to microscopic findings based on individual cases (Satriawan and Octaviani, 2021; Billa et al., 2024), whereas molecular data have been lacking. Therefore, the present findings offer important baseline information for Southeast Asia and emphasize the role of ectoparasite control in tropical regions, consistent with earlier detection of M. haemofelis DNA in cat fleas (Abdullah et al., 2019). ConclusionThis study detected a high prevalence of M. haemofelis in cats infested with C. felis in Padang City. A significant association was observed between infection status and flea infestation level and housing pattern. AcknowledgmentsThe authors extend their sincere gratitude to the Master of Veterinary Science Program, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, and the Department of Parasitology and Internal Medicine for academic guidance and support. Appreciation is also given to the Provincial Animal Hospital of West Sumatra, Balai Veteriner Bukittinggi, Paw’s Vet Padang, Platinum Vet Care, Aurora Pet Care, and Dunia Pet Care for their valuable assistance and for providing access to clinical cases and sample collection during this study. FundingThis study was self-funded by the authors. Authors' contributionsHesti Rahayu: Original draft writing, conceptualization, data curation, formal analysis, and visualization. Teguh Rianda: conceptualization, funding acquisition, supervision, and project administration. Dwi Priyowidodo: investigation, methodology, supervision, and validation. Irkham Widiyono: Data curation, investigation, supervision, and validation. Vika Ichsania Ninditya: Preparation of the manuscript’s initial draft. Conflict of interestThe authors have no conflicts of interest to declare. Data availabilityAll references are open access, so data can be obtained from the internet. ReferencesAbdullah, S., Helps, C. and Tasker, S. 2019. Pathogens in fleas collected from cats and dogs: distribution and prevalence in the United Kingdom. Vectors 12(1), 71; doi:10.1186/s13071-019-3326-x Altay, K., Coskun, A., Erol, U., Sahin, O.F. and Turk, S. 2025. Development of a novel triplex-PCR assay for the identification of feline hemoplasma species and survey of hemoplasma species in cats in Türkiye. Parasitol. Int. 104, 102969; doi:10.1016/j.parint.2024.102969 Berzina, I., Capligina, V., Namina, A., Visocka, A. and Ranka, R. 2021. Haemotropic Mycoplasma species in pet cats in Latvia: a study, phylogenetic analysis and clinical case report. JFMS. Open Rep. 7(2), 20551169211028088; doi:10.1177/20551169211028088 Billa, A.T.S., Batan, I.W. and Suartha, I.N. 2024. Case report: management of stomatitis and Mycoplasma haemofelis infecting erythrocytes in a pet cat. Indo. Med. Vet. 13(3), 284–295; doi:10.19087/imv.2024.13.3.284 BPS (Statistics Indonesia). 2024. Temperature and humidity of Padang City in 2024. Meteorology, Climatology, and Geophysics Agency, Padang, Indonesia. Cortés Sánchez. 2022. Hemoplasmosis en gates domésticos y la importance de un been diagnóstico en la clinica veterinaria Thesis, Universidad Autónoma Metropolitana, Unidad Xochimilco. Repositorio Institucional UAM. Available via https://repositorio.xoc.uam.mx/jspui/handle/123456789/251709 Fox, I., Rivera, G.A. and Bayona, I.G. 1969. Controlling cat fleas with dichlorvos-impregnated collars. J. Econom. Entomol. 62(5), 1246–1249; doi:10.1093/jee/62.5.1246a Herrera León, J.T. 2025. Micoplasmosis hemotrópica felina: terapéuticas y métodos diagnósticos de la enfermedad Revisión Sistemática de Literatura Thesis, Universidad Cooperativa de Colombia, Facultad de Medicina Veterinaria y Zootecnia, Bucaramanga. Available via https://hdl.handle.net/20.500.12494/59326 Hoseinpoor, E., Goudarztalejerdi, A. and Sazmand, A. 2024. Molecular prevalence and phylogenetic analysis of hemotropic Mycoplasma species in cats in different regions of Iran. BMC. Microbiol. 24(1), 1-8. doi: 10.1186/s12866-024-03356-8 Imre, M., Văduva, C., Dărăbuș, G., Morariu, S., Herman, V., Plutzer, J., Suici, T., Lait, P.J.P. and Imre, K. 2020. Molecular detection of hemotropic mycoplasmas (hemoplasmas) in domestic cats (Felis catus) in Romania. BMC Vet. Res. 16(1), 1-8. doi:10.1186/s12917-020-02626-7 Jayanti, P.D., Gunawan, I.W.N.F. and Indarjulianto, S. 2023. Haemoplasmosis in A Hypoalbuminemic Cat: a Case Report. J. Med. Vet. 6(2), 288–296. Kamyingkird, K., Jiyipong, T., Amavisit, P., Stich, R.W. and Jittapalapong, S. 2021. Molecular detection of Mycoplasma haemofelis, ‘Candidatus Mycoplasma haemominutum’ and ‘Candidatus Mycoplasma turicensis in stray cats residing in Bangkok monasteries, Thailand. Agricul. Natur. Res. 55(3), 423–430; doi:10.34044/j.anres.2021.55.3.12 Lappin, M.R. 2018. Update on flea and tick associated diseases of cats. Parasitol 254, 26–29; doi:10.1016/j.vetpar.2018.02.022 Lima, E.F. 2025. Diagnóstico molecular de M. hemotrópicos em felinos (Felis catus) no semiárido da Paraíba, Brasil: Prevalência e fatores associados. Instituto Federal de Educação, Ciência e Tecnologia da Paraíba, Campus Sousa, Brazil Repositorio Institucional IFPB. Available via https://repositorio.ifpb.edu.br/handle/177683/4330 Lobová, D., Konvalinová, J., Bedáňová, I., Filipejová, Z. and Molinková, D. 2021. Infections of cats with blood mycoplasmas in various contexts. Acta. Vet. Brno. 90(2), 211–219; doi:10.2754/avb202190020211 Marchiondo, A.A., Holdsworth, P.A., Green, P., Blagburn, B.L. and Jacobs, D.E. 2007. World Association for the Advancement of Veterinary Parasitology (W.A.A.V.P.) guidelines for evaluating the efficacy of parasiticides for the treatment, prevention, and control of flea and tick infestation on dogs and cats. Vet 58(3-4), 59–68; doi:10.1016/j.vetpar.2006.10.028 Melo TB de., Silva, T.R.M., Almeida TLAC de., Tutija, J.F., Silva AO da., Lira M da., Amorim, D., Giannelli, A., Ramos CA do., Alves, L.C., Carvalho GA de. and Ramos, R.A.N. 2023. Molecular detection of vector borne pathogens in FIV- and FeLV-positive cats. Vet. Parasitol. 40, 100857; doi:10.1016/j.vprsr.2023.100857 Mifsud, M., Takács, N., Gyurkovszky, M., Solymosi, N. and Farkas, R. 2020. Detection of Flea Borne Pathogens from Cats and Fleas in a Maltese Shelter. Zoonotic. Dis. 20(7), 529–534; doi:10.1089/vbz.2019. Moore, C.O., André, M.R., Šlapeta, J. and Breitschwerdt, E.B. 2024. Vector biology of the cat flea Ctenocephalides felis. Trends. Parasitol. 40(4), 324–337; doi:10.1016/j.pt.2024.02.006 Moore, C.O., Lashnits, E., Lappin, M., Hawley, J. and Breitschwerdt, E.B. 2024. A case of mistaken identity: a systematic review, meta analysis, and reinvestigation of hemotropic Mycoplasma spp. infection in Ctenocephalides felis cat flea. Parasites. Vectors. 17(1), 209; doi:10.1186/s13071-024-06292-8 Otranto, D., Carbonara, M., Baneth, G., Dantas-Torres, F., Lappin, M.R. and Barrs, V.R. 2025. Feline vector borne diseases: from local risks to global concerns. Trends Parasitology 41(6), 486–503; doi:10.1016/j.pt.2025.04.009 Páez-Triana, L., Martinez, D., Patiño, L.H., Muñoz, M., Sandoval-Ramírez, C.M., Pinilla León, J.C. and Ramirez, J.D. 2025. Exploring endosymbionts and pathogens in Rhipicephalus sanguineus and Ctenocephalides felis felis with Oxford Nanopore Technology. Res. Vet. Sci. 185, 105562; doi:10.1016/j.rvsc.2025.105562 Pekel, B. and Duru, Y.Y. 2022. Determination of the Incidence of Mycoplasma haemofelis in Cats Visiting Veterinary Clinics in Kırıkkale. Intern. J. Vet. Animl. Res. 5(2), 40–46; doi:10.5281/zenodo.7020233 Pereira, A., Cruz, A., Novo, T. and Maia, C. 2025. C. felis (cat flea) Trends. Parasitol 41(3), 249–250; doi:10.1016/j.pt.2024.12.016 Pimpjong, K., Ratyotha, K., Rungruang, T., Thongpriam, N., Vaisusuk, K. and Piratae, S. 2025. Molecular Investigation and Characterization of Mycoplasma spp. infections in Naturally Infected Cats in Thailand. Acta Parasitol. 70(2), 62; doi:10.1007/s11686-025-01002-5 Razgūnaitė, M., Lipatova I Paulauskas., Snegiriovaitė, J., Karvelienė, B., Zamokas, G., Laukutė, M. and Radzijevskaja, J. 2024. Prevalence and Diversity of Haemotropic Mycoplasma Species in Cats and Their Ectoparasites. Vet. Sci. 11(2), 81; doi:10.3390/vetsci11020081 Reynolds, B.S., Boudet, K.G., Faucher, M.R., Germain, C., Geffre, A. and Lefebvre, H.P. 2007. Comparison of a new device for blood sampling in cats with a vacuum tube collection system plasma biochemistry, hematology and practical usage assessment. Surg 9(5), 382–386; doi:10.1016/j.jfms.2007.03.006 Sagare, R., Gowri, B., Senthil, N.R., Jeyaraja, K. and Arunprasad, A. 2024. Incidence of Feline Haemotropic Mycoplasmosis in Chennai, Tamil Nadu. Indian. Vet. J. 101(5), 41–44; doi: 10.62757/IVA.2024.101.5.41-44 Satriawan, I. and Octaviani, D.I.D.A. 2021. Haemobartonellosis in a domestic cat in Indonesia: a case study. Vet. Bio. Clin. J. 3(2), 23–26. Shi, H., Li, G., Li, D., Zhai, H., Ji, S., Hu, Y., Wang, L. and Yao, L. 2025. Molecular investigation reveals three hemotropic mycoplasmas in cats and three tick species in China. Front. Vet. Sci. 12, 1522904; doi:10.3389/fvets.2025.1522904 Tasker, S., Hofmann Lehmann, R., Belák, S., Frymus, T., Addie, D.D., Pennisi, M.G., Boucraut Baralon, C., Egberink, H., Hartmann, K., Hosie, M.J., Lloret, A., Marsilio, F., Radford, A.D., Thiry, E., Truyen, U. and Möstl, K. 2018. Haemoplasmosis in cats: European guidelines from the ABCD on prevention and management. Surg 20(3), 256–261; doi:10.1177/1098612X18758594 Tjokorda Istri Agung Pradnya Dewi Pemayun., Sri Kayati Widyastuti., I Nyoman Suartha. and Ni Putu Trisna Asih. 2024. Haemobartonellosis in domestic short hair cats. Vet. Sci. Med. J. 6(04), 364–377; doi:10.24843/vsmj.2024.v06.i04.p05 Woods, J.E., Brewer, M.M., Hawley, J.R., Wisnewski, N. and Lappin, M.R. 2005. Evaluation of experimental transmission of Candidatus Mycoplasma haemominutum and Mycoplasma haemofelis by Ctenocephalides felis to cats. Am. J. Vet. Res. 66(6), 1008–1012; doi:10.2460/ajvr.2005.66.1008 Yasmin, A.R., Peng, T.L., Abdul-Azeez, I.O., Nur Atikah, H., Salma, C.W.Z., Hamdan, R.H. and Loong, S.K.2022. Retrospective prevalence and associated risk factors of Mycoplasma haemofelis infection in owned cats. Trop. Biomed. 39(3), 444–450; doi:10.47665/tb.39.3.015 Zarea, A.A.K., Tempesta, M., Fouad, E.A., Ndiana, L.A., Mahmoud, M.S., Mrenoshki, D., Martella, V., Decaro, N., Chomel, B. and Greco, G. 2023. Prevalence of Bartonella spp., haemotropic Mycoplasma spp. and others vector-borne pathogens in private-owned dogs and cats, Egypt. Acta Trop. 240, 106857; doi:10.1016/j.actatropica.2023.106857/ | ||
| How to Cite this Article |
| Pubmed Style Rahayu H, Rianda T, Priyowidodo D, Widiyono I, Ninditya VI. Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Vet. J.. 2025; 15(12): 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 Web Style Rahayu H, Rianda T, Priyowidodo D, Widiyono I, Ninditya VI. Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. https://www.openveterinaryjournal.com/?mno=273674 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.32 AMA (American Medical Association) Style Rahayu H, Rianda T, Priyowidodo D, Widiyono I, Ninditya VI. Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Vet. J.. 2025; 15(12): 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 Vancouver/ICMJE Style Rahayu H, Rianda T, Priyowidodo D, Widiyono I, Ninditya VI. Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 Harvard Style Rahayu, H., Rianda, . T., Priyowidodo, . D., Widiyono, . I. & Ninditya, . V. I. (2025) Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Vet. J., 15 (12), 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 Turabian Style Rahayu, Hesti, Teguh Rianda, Dwi Priyowidodo, Irkham Widiyono, and Vika Ichsania Ninditya. 2025. Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Veterinary Journal, 15 (12), 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 Chicago Style Rahayu, Hesti, Teguh Rianda, Dwi Priyowidodo, Irkham Widiyono, and Vika Ichsania Ninditya. "Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia." Open Veterinary Journal 15 (2025), 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 MLA (The Modern Language Association) Style Rahayu, Hesti, Teguh Rianda, Dwi Priyowidodo, Irkham Widiyono, and Vika Ichsania Ninditya. "Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia." Open Veterinary Journal 15.12 (2025), 6480-6486. Print. doi:10.5455/OVJ.2025.v15.i12.32 APA (American Psychological Association) Style Rahayu, H., Rianda, . T., Priyowidodo, . D., Widiyono, . I. & Ninditya, . V. I. (2025) Molecular detection of Mycoplasma haemofelis in cats infested with Ctenocephalides felis in Padang, West Sumatra, Indonesia. Open Veterinary Journal, 15 (12), 6480-6486. doi:10.5455/OVJ.2025.v15.i12.32 |