| Research Article | ||
Open Vet. J.. 2025; 15(12): 6434-6441 Open Veterinary Journal, (2025), Vol. 15(12): 6434-6441 Research Article Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in ratsAmal Umran Mosa1* , Moayed Mijbil Ubaid2 , Sarah Najm Abed1 and Shatha Hussein Kadhim11Department of Pharmacology and Toxicology, College of Pharmacy, University of Kerbala, Karbala, Iraq 2Department of Biology, College of Basic Education, University of Sumer, Rifai, Iraq *Corresponding Author: Amal Umran Mosa. Department of Pharmacology and Toxicology, College of Pharmacy, University of Kerbala, Karbala, Iraq. Email: amal.imran [at] uokerbala.edu.iq Submitted: 27/07/2025 Revised: 29/10/2025 Accepted: 12/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
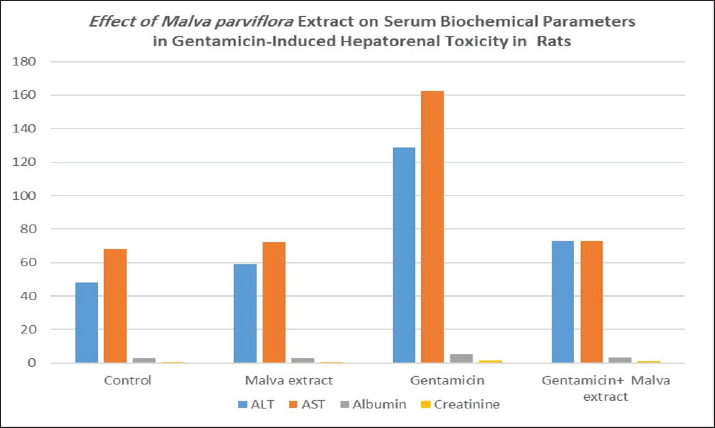
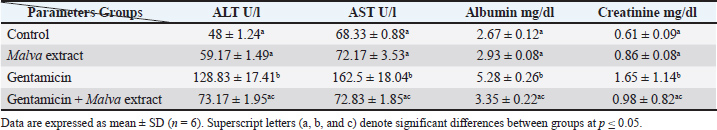
AbstractBackground: Gentamicin is a widespread aminoglycoside antibiotic associated with liver and renal toxicity. To evaluate the protective role of the bioactive constituents of Malva parviflora through their antioxidant and anti-inflammatory actions. Aim: The aim of this study was to assess the influence of M. parviflora extract on liver and renal toxicity in male albino rats after intramuscular injection of gentamicin. Methods: Twenty-four albino rats were partitioned arbitrarily into 4 groups: control group, gentamicin-treated group for 30 days, M. parviflora extract-treated group for 30 days, and gentamicin and Malva combined-treated group. Blood biomarkers, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), creatinine, and oxidative stress biomarkers, malondialdehyde (MDA), glutathione (GSH), and Glutathione-S-transferase (GST), were analyzed, and histopathological alterations in the liver and kidney were evaluated. A p-value ≤ 0.05 is rated as significant statistically. Results: Significant increase in ALT, AST, and creatinine levels, and liver and renal histology examinations revealed marked alterations in gentamicin use. These alterations were attenuated by combining M. parviflora extract and preserving tissue structures and organ functions near normal. Malva parviflora reverses the oxidative stress of gentamicin by lowering the MDA concentration and recovers the GSH and GST levels seen in the group treated with gentamicin plus M. parviflora. Conclusion: Gentamicin-induced renal and hepatic toxicities can be significantly reduced by M. parviflora extract and protect organs. Keywords: Malva parviflora, Gentamicin, Hepatotoxicity, Nephrotoxicity, Oxidative stress. IntroductionThe Malva parviflora Malvaceae family, named Sunchal in Pakistan, includes the well-known plant M. parviflora L. Its leaves are used as food in green salad and in helminth infection treatment, and in gastrointestinal diseases such as gastritis, diarrhea, constipation, abdominal pain, hair loss, and wound (Altyar et al., 2022). This plant has many pharmacological properties, including anti-inflammatory, antidiabetic, antibacterial, antioxidant, analgesic, neuroprotective, and ulcer healing properties. These effects are related to phenolic acids and flavonoids, which are secondary metabolites abundant in M. parviflora, where mucilaginous cells produce mucilage that is potentially therapeutic (Munir et al., 2021). Malva parviflora gains its pharmacological preferences from its abundant bioactive ingredients like phenolic acids, flavonoids, and polysaccharides. Its prospective mechanism of action includes anti-inflammatory effects that compounds in M. parviflora may inhibit inflammatory mediators like cytokines, lipoxygenase, and cyclooxygenase, and it also includes antioxidant activity, and the reactive oxygen species (ROS) and free radicals are scavenged by the plant’s constituents and then decreasing the stress of oxidation. The immunomodulatory effects of bioactive molecules may influence immune cell function and cytokine production. Some studies suggest antimicrobial potential. Impressively, extracts promote wound healing through cell proliferation, collagen synthesis, and tissue regeneration (Medrano-Jiménez et al., 2019). Malva parviflora have small white or pale pink flowers and lobed leaves. Flavonoids, phenolic acids, polysaccharides, and many phyto-constituents are part of its chemical composition, granting its therapeutic characteristics (Naser et al., 2022 ). Malva parviflora is applied medically, and it has been used traditionally in a variety of cases, such as anti-inflammation, as it reduces swelling and pain and has a demulcent effect, where it relieves sore throats and coughing by its mucilage content that treats mucous membranes. Its extracts are also used as respiratory support, such as in bronchitis, asthma, and cough treatment, due to their expectorant properties. Furthermore, it is used for wound healing as it is topically applied to reduce skin irritation and encourage healing. Moreover, digestive aid was described as reducing bloating and indigestion. Last but not least, diuretic and antioxidant: decreases oxidative damage and encourages urine excretion (Garg et al., 2024). Gentamicin, an aminoglycoside antibiotic, was discovered in 1963 by Weinstein and colleagues through soil sampling at Schering Corporation in Bloomfield, New Jersey. It was later purified and developed as a broad-spectrum injectable antibiotic. Gentamicin is extracted from Micromonospora purpurea and is composed primarily of multiple gentamicin members, C1, C1a, and C2 that form 80% of its active compound, with the remainder being minor analogs (Wei et al., 2019). Gentamicin mechanism of action reveals that the drug coheres to the 30S subunit of bacterial ribosomes, interfering with tRNA selection and resulting in non-functional misfolded proteins. It primarily targets the 16S rRNA component, causing lethal errors in protein synthesis (Dao et al., 2018). A secondary mechanism involves binding to rRNA site 69, inhibiting proper ribosome recycling and causing extended, non-terminating protein chains (Borovinskaya et al., 2007). The extent of gram-negative bacterial infections is effectively treated with gentamicin and is used for treating urinary tract infections, bone infections, sepsis, and certain eye conditions. Despite the emergence of newer antibiotics, it remains in clinical use due to its potent activity (Burchum and Rosenthal, 2014). In regards to its adverse effects, prolonged use of gentamicin can cause nephrotoxicity, ototoxicity, and neurotoxicity. The drug accumulates in renal tubular cells and inner ear hair cells, leading to irreversible damage in some patients. Risk factors include age, existing renal impairment, and co-administration with other nephrotoxic agents (Lopez-Novoa et al., 2011). Interestingly, many studies have proven that antibiotics have many side effects under the title of hepatotoxicity and nephrotoxicity, especially long-term use, which may result in intense injury to the liver and kidneys (Ubaid, 2019). Gentamicin-induced nephrotoxicity is marked by acute tubular necrosis, free radical generation, lipid peroxidation, and reduced glomerular filtration. Hepatic injury, although less common, manifests as elevated liver enzymes and occasional cholestatic hepatitis, often immunoallergic in origin (Moafa et al., 2023). Materials and MethodsAnimalsThis study was proceeded in June 2024 and included 24 healthy adult male albino rats (Rattus norvegicus). These animals weighing 225–250 gm ± 5 g and were aged 10–12 weeks. Animals were acquired from the animal house of the pharmacy college/Kerbala University and kept at a temperature between 25°C and 28°C. The changing room’s air was changed in a continuous pattern using a ventilation vacuum, and light-dark cycles (12/12) hour per day were maintained. The litters of cages were changed every 3 days. They were given a supplemented rodent diet. MaterialsGentamicinGentamicin 40 mg/ml intramuscular injection, each 2 ml ampoule contains 80 mg of gentamicin. Malva extract preparationNine hundred ml of ethanol were used to extract M. parviflora leaves to produce 300 g at room temperature for 48 hours. Followed by filtering of the extracts, concentrated then sterile dark bottles were used to storing extracts at four°C (El-Naggar et al., 2020). Experimental designThe experimental rats were subdivided into four groups: • Group 1: The control group was fed with standardized water and diet. • Group 2: Gentamicin 120 mg per kg intramuscularly for 30 days. • Group 3: Malva parviflora extract 170 mg per kg orally for 30 days. • Group 4: Gentamicin 120 mg/kg intramuscularly, with M. parviflora 170 mg/kg orally for 30 days. All animals were drenched with drugs at 9 am daily during the experiment period. Biochemical measurementobtaining blood samples via the orbital venous plexus. Then separating the serum by centrifuging the blood samples at 4,500 rpm for 10 minutes. The obtained serum samples were kept at 20°C until the time of performing biochemical measurements. Samples were assessed for (aspartate aminotransferase (AST), alanine aminotransferase (ALT), albumin, and creatinine levels, and oxidative stress biomarkers; malondialdehyde (MDA), Glutathione-S-transferase (GST), and reduced glutathione (GSH) (Kadhim et al., 2022). Histopathological assessmentThe liver and kidney of each rat were divided into small pieces and preserved in 10% formalin saline for 24 hours after sacrificed the animals under euthanasia. The samples were further processed and dehydrated using different ethanol concentrations (70%,90%, and 100%) in xylene, and they were cleaned for 2 hours. Hard paraffin wax was impregnated for 1 hour at 60°C, and soft paraffin wax for 3 hours at 45–50 after paraffin block were made 5 mm slices then staining with hematoxylin and eosin to estimate fibrosis and hematoxylin and fusing for standard pathology evaluation (Fischer et al., 2008). Data analysisAnalyzing biochemical measurements’ results were used by one-way variance analysis using SPSS version 27, followed by a post-hoc test (Turkey’s HSD) to determine the significance of differences between groups. Data are expressed as mean ± standard deviation. Different superscript letters (a, b) indicate statistically significant differences between groups at p < 0.05. Ethical approvalThe Ethics Committee of the College of Pharmacy, University of Kerbala, convened and approved our study on June 12, 2024 and registered it (Ref:2024). The Declaration of Helsinki was followed and performed to litigate this study. ResultsBiochemical parametersGentamicin-treated rats showed significant elevations in ALT, AST, creatinine, and albumin levels. Co-administration of M. parviflora extract reduced these levels closer to control values, indicating a protective effect. Table 1 shows the significant positive effect of the plant extract in reducing the toxic effect of the drug on liver and kidney parameters. The results of ALT, AST, albumin, and creatinine revealed a significant increase in the gentamicin group (128.83, 162.5, 5.28, and 1.65), respectively, while these results reduced significantly at p ≤ 0.05 in Malva and gentamicin groups (73.17, 72.83, 3.35, and 0.98), respectively, which also significantly differed from the control group (48, 68.33, 2.67, and 0.61), respectively (Fig. 1).
Fig. 1. Malva parviflora extract mitigated gentamicin-induced elevations in ALT, AST, albumin, and creatinine levels, restoring normal values. <AQ3> Table 1. Effect of Malva parviflora extract on serum biochemical parameters in gentamicin-induced hepatorenal toxicity in rats.
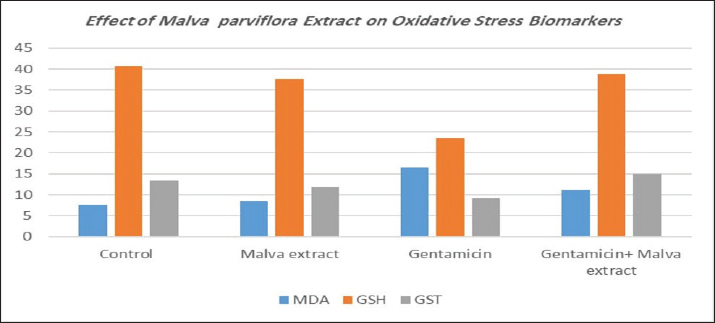
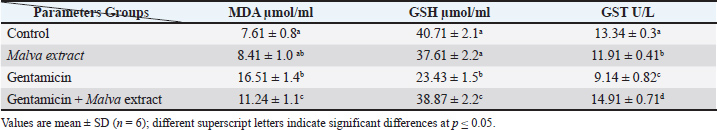
Table 2 shows the significant positive effect of the plant extract by reducing the toxic effect of the drug on oxidative stress biomarkers. The results of GSH and GST revealed a significant decrease in the gentamicin group (23.43 and 9.14, respectively) (Fig. 2), while these results increased significantly at p ≤ 0.05 in Malva and gentamicin groups (38.87 and 14.91, respectively), which also significantly differed from the control group (40.71 and 13.34, respectively). On the other hand, MDA revealed a significant increase in the gentamicin group (16.51), while this result decreased significantly at p ≤ 0.05 in Malva and gentamicin groups (11.24), which also significantly differed from the control group (7.61).
Fig. 2. Malva parviflora extract reduced MDA and restored GSH and GST levels in gentamicin-treated rats, indicating strong antioxidant protection. Table 2. Effect of Malva parviflora extract on oxidative stress biomarkers.
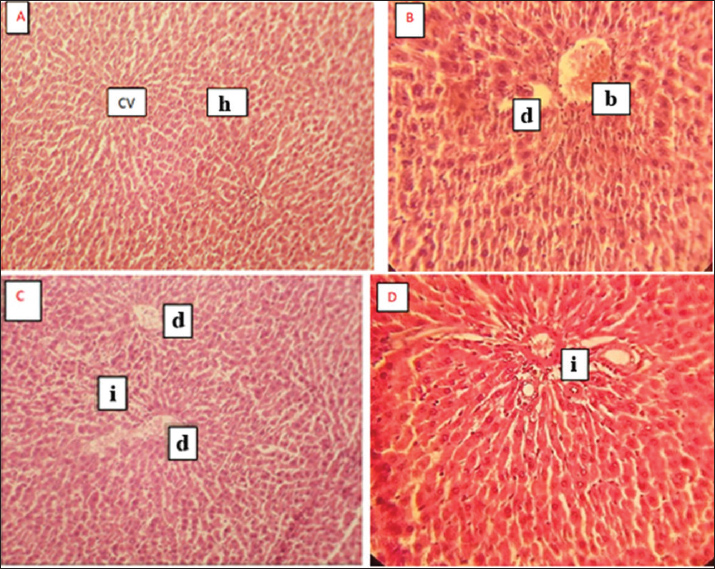
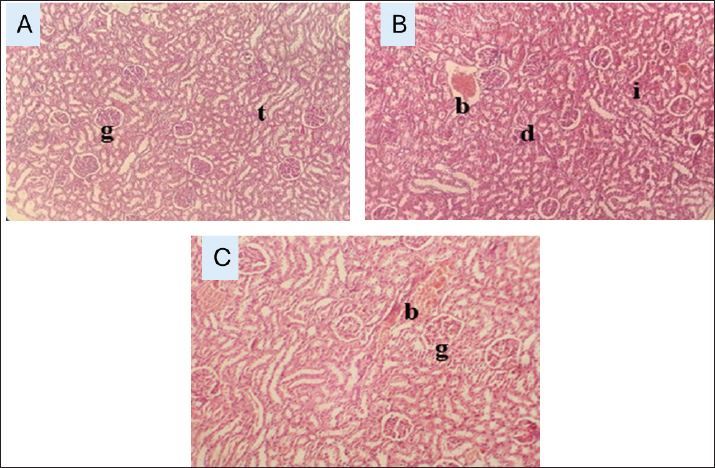
Histological findingsGentamicin induced hepatic and renal damage characterized by cellular degeneration congestion and inflammation. In contrast, co-treatment with M. parviflora mitigated these effects, preserving tissue structure (Figs. 3 and 4).
Fig. 3. A/ cross section in liver tissue of control group shows central vein (cv) and normal hepatocytes (h), (H&E400x) B/ cross section in liver tissue of gentamicin group shows degeneration (d), (H&E400x) C/ Cross section of liver of group treated with gentamicin 120 mg/kg intramuscular injection for 30 days shows inflammation (i) and Degeneration(d)( H&E400x) D/ Cross section of liver of (gentamicin 120 mg/kg intramuscular injection for 30 days & Malva 170 mg/kg for 30 days) group shows inflammation (i) and normal hepatocytes (H&E400x).
Fig. 4. A/cross section in the kidney tissue of the control group shows normal glomeruli (g) and normal tubules (t) (H&E400x). B/cross section in the kidney tissue of the gentamicin group shows degeneration (d), bleeding (b), and inflammation (i) (H&E400x). C Cross section of the kidney tissue of the gentamicin & Malva group shows less bleeding (b) and normal glomeruli (g) (H&E400x). DiscussionGentamicin, a commonly used aminoglycoside antibiotic, is notorious for its nephrotoxic and hepatotoxic effects, particularly at high doses or prolonged administration periods (Hussein et al., 2024). Our study revealed a considerable rise in serum AST, ALT, creatinine, and albumin levels in gentamicin-treated rats, indicating hepatic and renal damage. These findings are consistent with previous research demonstrating that gentamicin disrupts organ function through oxidative stress and inflammation (Gumbar et al., 2023). Procedurally, ROS is excessively produced from gentamicin treatment, ending with oxidation injury to lipids, proteins, nucleic acids, and mitochondrial dysfunction in hepatic and renal tissues. When cell membrane integrity is compromised by this oxidative stress, intracellular enzymes ALT and AST seep into the bloodstream (Martínez-Salgado et al., 2002). In the kidneys, gentamicin accumulates in proximal tubular epithelial cells, triggering oxidative stress, apoptosis, tubular necrosis, and glomerular filtration defects (Choi et al., 2000). Moreover, ROS generation exacerbates inflammation by drawing monocytes and macrophages to injured tissues, promoting necrosis and cell death. Our histological results, which revealed necrosis, inflammatory infiltration, and architectural disarray in the liver and kidney tissues of rats treated with gentamicin, demonstrated these pathological processes (Mittal et al., 2014). Ten to twenty-five percent of patients undergoing long-term treatment experience nephrotoxicity, which is still a major clinical concern during gentamicin therapy. In several cases, irreversible damage has been documented (Patzer, 2008). In regards to M. parviflora Extract's Protective Properties, the biochemical and histological changes caused by gentamicin are extremely lowered when M. parviflora extract was administered concurrently, suggesting that it may provide hepatorenal protection. The extract's high concentration of polyphenols, flavonoids, and other bioactive phytochemicals with strong anti-inflammatory and antioxidant qualities likely underlie its medicinal properties. (Dugani et al., 2016). In the gentamicin group, elevated serum albumin levels could be caused by dehydration or changes in protein metabolism resulting from acute renal injury (Huang et al., 2020). Cyclooxygenase and lipoxygenase enzymes downregulate and combat ROS, preventing lipid peroxidation, protecting cell membranes, and reducing the release of pro-inflammatory cytokines, which is probably the cause of the normalization of liver and renal indicators (Jomova et al., 2023). Moreover, the diuretic and antimicrobial properties of plants augment toxin excretion in urine and decrease microbial infection, lowering renal load and enhancing nephroprotective outcomes (Alobaidi 2024). Histopathological exams result supports the above outcome that inflammation and necrosis in treated rats are reduced with retaining tissue architecture when compared to only gentamicin-treated rats. Malva parviflora regulate calcium influx that may protect renal tubular cells from calcium overload, inducing tubular necrosis (Gumbar et al., 2023). The former mechanism is consistent with the effects of renal protection of calcium channel blockers against gentamicin nephrotoxicity (Stojiljković et al., 2008). Gentamicin treatment causes a great decline in GSH and GST levels in addition to a great increase in MDA, that is major sign in peroxidation of lipid. Moreover, gentamicin treatment results in producing numerous ROS and ends with damage to the cell antioxidant process, resulting in tissue necrosis and organ dysfunction (Hassanien, 2021). Oxidative stress caused by gentamicin is abolished by M. parviflora by decreasing the MDA concentration and recovering the levels of GST and GSH, which is observed in the group that is treated with combined gentamicin and Malva through struggling against antioxidant enzyme reduction. An imbalance between antioxidants and pro-oxidants can increase the production of reactive oxygen species that cause oxidative stress by means of retrieving endogenous antioxidants (GSH, GST) and scavenging reactive oxygen species, as well as likely anti-inflammatory and cytoprotective effects (Bouriche et al., 2011). Malva parviflora extracts extremely decreasing gentamicin oxidation and protect liver and renal tissues through anti-oxidation properties (Mohamadi Yarijani et al., 2019). ConclusionMalva parviflora extract significantly protects hepatic and renal toxicity in rats by recovering biochemical, oxidative stress, and histological specifications. These feedbacks call attention to its potential therapeutic function; nevertheless, corroboration by additional clinical and preclinical works is needed. Conflict of interestThe authors declare no conflicts of interest. FundingThe research is a self-funding study. Authors contributionsAmal Umran Mosa presided over the study’s conception and design as research; Sarah Najm Abed was responsible for data collection and organization. Statistical assessment was conducted by Moayed Mijbil Ubaid and Shatha Hussein Kadhim. Amal Umran Mosa provided supervision throughout the project and proofread the manuscript. Data availabilityThe manuscript includes the entire data that supports the findings of this study. ReferencesAlobaidi, S. 2024. Renal health benefits and therapeutic effects of parsley (Petroselinum crispum): a review. Front. Med. (Lausanne) 11, 1494740; doi:10.3389/fmed.2024.1494740 Altyar, A.E., Munir, A., Ishtiaq, S., Rizwan, M., Abbas, K., Kensara, O., Elhady, S.S., Rizg, W.Y., Youssef, F.S. and Ashour, M.L. 2022. Malva parviflora leaves and fruits mucilage as natural sources of anti-inflammatory, antitussive and gastro-protective agents: a comparative study using rat models and gas chromatography. Pharm 15(4), 427; doi:10.3390/ph15040427 Borovinskaya, M.A., Pai, R.D., Zhang, W., Schuwirth, B.S., Holton, J.M., Hirokawa, G., Kaji, H., Kaji, A. and Cate, J.H.D. 2007. Structural basis for aminoglycoside inhibition of bacterial ribosome recycling. Nat. Struct. Mol. Biol. 14(8), 727–732. Bouriche, H., Meziti, H., Senator, A. and Arnhold, J. 2011. Anti-inflammatory, free radical-scavenging, and metal-chelating activities of Malva parviflora. Pharm. Biol. 49(9), 942–946; doi:10.3109/13880209.2011.558102 Burchum, J.R. and Rosenthal, L.D. 2014. Lehne’s pharmacology for nursing care. Choi, K.H., Kim, T.I., Chong, D.L., Lee, H.Y. and Han, D.S. 2000. Gentamicin induced apoptosis of renal tubular epithelial (LLC-PK1) cells. Korean J. Intern. Med. 15(3), 218–223; doi:10.3904/kjim.2000.15.3.218 Dao, E.H., Poitevin, F., Sierra, R.G., Gati, C., Rao, Y., Ciftci, H.I., Akşit, F., Mcgurk, A., Obrinski, T., Mgbam, P., Hayes, B., De Lichtenberg, C., Pardo-Avila, F., Corsepius, N., Zhang, L., Seaberg, M.H., Hunter, M.S., Liang, M., Koglin, J.E., Wakatsuki, S. and Demirci, H. 2018. Structure of the 30S ribosomal decoding complex at ambient temperature. RNA 24(12), 1667–1676. Dugani, A., Dakhil, B. and Treesh, S. 2016. Protective effect of the methanolic extract of malva parviflora l. leaves on acetic acid-induced ulcerative colitis in rats. Saudi J. Gastroenterol. 22(3), 226–233; doi:10.4103/1319-3767.182459 El-Naggar, M.E., Hussein, J., El-Sayed, S.M., Youssef, A.M., El Bana, M., Latif, Y.A. and Medhat, D. 2020. Protective effect of yogurt based on Malva parviflora leaves extract nanoemulsion on acetic acid-induced ulcerative colitis in rats. J. Mater. Res. Technol. 9(6), 14500–14508. Fischer, A.H., Jacobson, K.A., Rose, J. and Zeller, R. 2008. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harb Protoc 2008(5): pdb. prot4986. doi: 10.1101/pdb.prot4986 Garg, R., Dobhal, K. and Singh, A. 2024. Utilization of medicinal herbal plants in the management of respiratory conditions. London: IntechOpen. doi: 10.5772/intechopen.1008176 Gumbar, S., Bhardwaj, S., Mehan, S., Khan, Z., Narula, A.S., Kalfin, R., Tabrez, S., Zughaibi, T.A. and Wasi, S. 2023. Renal mitochondrial restoration by gymnemic acid in gentamicin-mediated experimental nephrotoxicity: evidence from serum, kidney and histopathological alterations. Front. Pharmacol. 14, 1218506; doi:10.3389/fphar.2023.1218506 Hassanien, M.A. 2021. Antioxidant effects of gum arabic on gentamicin-induced hepatotoxicity in rats. Tanta Med. J. 49(2), 146–154; doi:10.4103/tmj.tmj_41_ Huang, H., Jin, W.W., Huang, M., Ji, H., Capen, D.E., Xia, Y., Yuan, J., Păunescu, T.G. and Lu, H.A.J. 2020. Gentamicin-Induced Acute Kidney Injury in an Animal Model Involves Programmed Necrosis of the Collecting Duct. J. Am. Soc. Nephrol. 31(9), 2097–2115; doi:10.1681/ASN.2019020204 Hussein, U.A., -R.., Mohammed, M.N., Al-Saedi, H.F. and Ramadhan, M.A. 2024. Nephroprotective effect of dapsone against gentamicin-induced acute nephrotoxicity in rats. Iraqi. J. Pharm. Sci. 33(1), 89–93; doi:10.31351/vol33iss1pp89-93 Jomova, K., Raptova, R., Alomar, S.Y., Alwasel, S.H., Nepovimova, E., Kuca, K. and Valko, M. 2023. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: chronic diseases and aging. Arch. Toxicol. 2499(10), 2499–2574; doi:10.1007/s00204-023-03562-9 Kadhim, S.H., Mosa, A.U. and Ubaid, M.M. 2022. Hepatorenal protective activity of Artemisia against diclofenac toxicity in male rats. Pan Afr. Med. J. 43, 192; doi:10.11604/pamj.2022.43.192.36160 Lopez-Novoa, J.M., Quiros, Y., Vicente, L., Morales, A.I. and Lopez-Hernandez, F.J. 2011. New insights into the mechanism of aminoglycoside nephrotoxicity: an integrative point of view. Kidney. Int. 79(1), 33–45; doi:10.1038/ki.2010.337 Martínez-Salgado, C., Eleno, N., Tavares, P., Rodríguez-Barbero, A., García-Criado, J., Bolaños, J.P. and López-Novoa, J.M. 2002. Involvement of reactive oxygen species on gentamicin-induced mesangial cell activation. Kidney Int. 62(5), 1682–1692; doi:10.1046/j.1523-1755.2002.00635.x Medrano-Jiménez, E., Jiménez-Ferrer, I., Pedraza-Escalona, M., Ramírez-Serrano, C.E., Álvarez-Arellano, L., Cortés-Mendoza, J., Herrera-Ruiz, M., Jiménez-Ferrer, E., Zamilpa, A., Tortoriello, J., Pedraza-Alva, G. and Pérez-Martínez, L. 2019. Malva parviflora extract ameliorates cognitive deficit in a mouse model of Alzheimer’s disease by restoring microglial function via a PPAR-γ-dependent mechanism. J. Neuroinflammation. 16(1), 143; doi:10.1186/s12974-019-1515-3 Mittal, M., Siddiqui, M.R., Tran, K., Reddy, S.P. and Malik, A.B. 2014. Reactive oxygen species in inflammation and tissue injury. Antioxid. Redox Signal. 20(7), 1126–1167; doi:10.1089/ars.2012.5149 Moafa, A., Aldossary, S.A., Al Mohaini, M. and Alsalman, A.J. 2023. Protective effect of aspirin against gentamicin-induced hepatotoxicity in a rat model. Biomed. Pharmacol. J. 16(4), 1923–1931. Mohamadi Yarijani, Z., Najafi, H., Shackebaei, D., Madani, S.H., Modarresi, M. and Jassemi, S.V. 2019. Amelioration of renal and hepatic function, oxidative stress, inflammation and histopathologic damages by Malva sylvestris extract in gentamicin induced renal toxicity. Biomed. Pharmacother. 112, 108635; doi:10.1016/j.biopha.2019.108635 Munir, A., Youssef, F.S., Ishtiaq, S., Kamran, S.H., Sirwi, A., Ahmed, S.A., Ashour, M.L. and Elhady, S.S. 2021. Malva parviflora leaves mucilage: an eco-friendly and sustainable biopolymer with antioxidant properties. Polym 13(23), 4251; doi:10.3390/polym13234251 Naser, E.H., Mahdi, L.S. and Alasadi, R.T. 2022. Phytochemical constituents and pharmacological activity of Malva parviflora plant: A review. Sci. J. Med. Res. 6(23), 35–44. Patzer, L. 2008. Nephrotoxicity as a cause of acute kidney injury in children. Pediatr. Nephrol. 23(12), 2159–2173; doi:10.1007/s00467-007-0721-x Stojiljković, N., Veljković, S., Mihailović, D., Stoiljković, M., Radovanović, D. and Randelović, P. 2008. The effect of calcium channel blocker verapamil on gentamicin nephrotoxicity in rats. Bosn. J. Basic Med. Sci. 8(2), 170–176; doi:10.17305/bjbms.2008.2977 Ubaid, M.M. 2019. Using of omega 3 to reduce the toxic effect of antituberculosis therapy on hepatorenal function and some blood parameters of albino male rats. Indian. J. Public. Health. Res. Dev. 10(11), 2101–2105; doi:10.37506/ijphrd.v10i11.2128 Wei, Z., Shi, X., Lian, R., Wang, W., Hong, W. and Guo, S. 2019. Exclusive Production of Gentamicin C1a from Micromonospora purpurea by Metabolic Engineering. Antibiotics 8(4), 267; doi:10.3390/antibiotics8040267 | ||
| How to Cite this Article |
| Pubmed Style Mosa AU, Ubaid MM, Abed SN, Kadhim SH. Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Vet. J.. 2025; 15(12): 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 Web Style Mosa AU, Ubaid MM, Abed SN, Kadhim SH. Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. https://www.openveterinaryjournal.com/?mno=273597 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.28 AMA (American Medical Association) Style Mosa AU, Ubaid MM, Abed SN, Kadhim SH. Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Vet. J.. 2025; 15(12): 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 Vancouver/ICMJE Style Mosa AU, Ubaid MM, Abed SN, Kadhim SH. Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 Harvard Style Mosa, A. U., Ubaid, . M. M., Abed, . S. N. & Kadhim, . S. H. (2025) Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Vet. J., 15 (12), 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 Turabian Style Mosa, Amal Umran, Moayed Mijbil Ubaid, Sarah Najm Abed, and Shatha Hussein Kadhim. 2025. Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Veterinary Journal, 15 (12), 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 Chicago Style Mosa, Amal Umran, Moayed Mijbil Ubaid, Sarah Najm Abed, and Shatha Hussein Kadhim. "Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats." Open Veterinary Journal 15 (2025), 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 MLA (The Modern Language Association) Style Mosa, Amal Umran, Moayed Mijbil Ubaid, Sarah Najm Abed, and Shatha Hussein Kadhim. "Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats." Open Veterinary Journal 15.12 (2025), 6434-6441. Print. doi:10.5455/OVJ.2025.v15.i12.28 APA (American Psychological Association) Style Mosa, A. U., Ubaid, . M. M., Abed, . S. N. & Kadhim, . S. H. (2025) Evaluation of the protective role of Malva parviflora extract in gentamicin-induced hepato-nephrotoxicity in rats. Open Veterinary Journal, 15 (12), 6434-6441. doi:10.5455/OVJ.2025.v15.i12.28 |