Open Veterinary Journal, (2025), Vol. 15(12): 6413-6422
Research Article
10.5455/OVJ.2025.v15.i12.26
Serological detection of Neospora caninum-specific antibodies in dogs of rural and urban areas within the Valdivia Province, Southern Chile
Karla Alvarez1,2, Marcos Rogério André3, Rosangela Zacarias Machado3, Pedro Bittencourt4,
Ananda Müller4, Carlos Hermosilla5, Anja Taubert5 and Marcelo A. Gómez6*
1Facultad de Ciencias Veterinarias, Universidad Austral de Chile, Campus Isla Teja, Valdivia, Chile
2VETERTEST, Laboratorio Clínico Veterinario, Puerto Montt, Chile
3Faculdade de Ciencias Agrarias e Veterinarias, Universidade Estadual Paulista,
(FCAV/UNESP), São Paulo, Brazil
4Department of Biomedical Sciences, Ross University of Veterinary Medicine, Basseterre, Saint Kitts and Nevis
5Institute of Parasitology, Justus Liebig University, Giessen, Germany
6Facultad de Ciencias Veterinarias, Universidad Austral de Chile, Chile
*Corresponding Author: Marcelo A. Gómez. Facultad de Ciencias Veterinarias, Universidad Austral de Chile,
Campus Isla Teja, Valdivia, Chile. Tel.: 56-63-2221072. Email: marcelogomez [at] uach.cl
Submitted: 23/07/2025 Revised: 20/10/2025 Accepted: 02/11/2025 Published: 31/12/2025
© 2025 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Neospora caninum belongs to the subphylum Apicomplexa (phylum Alveolata) and is an obligate intracellular protozoan parasite that has been described as a causative agent of neuromuscular paralysis in dogs and as a major cause of bovine abortion worldwide. However, knowledge regarding the presence of N. caninum-infected dogs in Chile is limited.
Aim: This study investigated N. caninum seroprevalence and risk factors in dogs from Valdivia Province, Los Ríos Region, Southern Chile.
Methods: For the study, 164 serum samples from dogs were collected, comprising 108 samples from dogs inhabiting rural areas and 56 from urban areas. The samples were analyzed using the indirect fluorescent antibody test to detect N. caninum-specific antibodies.
Results: In total, 24.4% (40/164) of the dogs were seropositive with a titer of 1:50. Rural dogs had higher seropositivity for N. caninum than urban dogs, with a seroprevalence of 28.7% (31/108) and 16.1% (9/56), respectively. Age was a risk factor for N. caninum seropositivity, affecting dogs ≥3 years old (p < 0.05).
Conclusion: To the best of our knowledge, this is the first study to report the seroprevalence of N. caninum in rural and urban dogs from Valdivia Province in Southern Chile, while also examining potential risk factors associated with N. caninum infections.
Keywords: Canine neosporosis, Parasite, IFAT, Risk factors, Dogs.
Introduction
Neospora caninum belongs to the subphylum Apicomplexa (phylum Alveolata) and is an obligate intracellular coccidian parasite that causes multisystemic and neuromuscular paralysis in dogs and is a major cause of infertility, bovine abortion, and neonatal calf mortality worldwide (Dubey, 2003). The dog (Canis lupus familiaris), Australian dingo (Canis lupus dingo), coyote (Canis latrans), and gray wolf (Canis lupus) have been confirmed as definitive hosts as well as intermediate hosts and play a crucial role in the horizontal transmission of this protozoan parasite to other domestic and wild animal species (Mcallister et al., 1998; Gondim et al., 2004; King et al., 2010). Natural intermediate hosts include cattle, horses, water buffalo, bison, goats, sheep, and marine mammals (Dubey, 2003; Villagra-Blanco et al., 2017). Natural intermediate hosts became infected after ingestion of N. caninum oocysts shed in the feces of the definitive hosts, which can contaminate food and water (Dubey, 2003). Dogs can acquire the infection horizontally via the ingestion of infected tissues containing N. caninum cysts or sporulated oocyst-contaminated water and food sources. Following the ingestion of sporulated oocysts by mammalian intermediate hosts, sporozoites are liberated from the oocysts, infect intestinal epithelial cells, and transform into tachyzoites, which then spread during acute N. caninum infection in vivo (Dubey, 1999; Dubey and Schares, 2011). Non-sporulated oocysts are shed by the definite host for 5–17 days after infection with tissue cysts containing bradyzoites. In addition, N. caninum tachyzoite can be vertically transmitted (congenital, transplacental) from bitches to fetuses in canid species during pregnancy (Dubey et al., 2007).
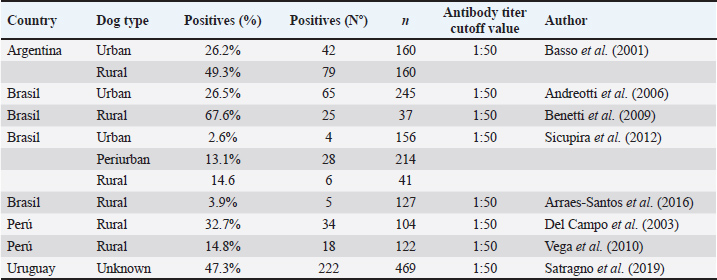
Neospora caninum is distributed globally and causes a variety of clinical signs in dogs, including pelvic limb ataxia and progressive pelvic limb paralysis caused by polymyositis and polyradiculoneuritis, resulting in diffuse neurological signs (Dubey and Lindsay, 1996). Other alterations include pulmonary, myocardial, dermatological, and reproductive disorders (Dubey and Lindsay, 1996; Dubey, 2003). Canine neosporosis in dogs is difficult to diagnose based on clinical signs because most of them are rather nonspecific; however, diagnostic methods such as serology via the indirect fluorescent antibody test (IFAT) can be helpful (Dubey and Lindsay, 1996; Björkman and Uggla, 1999). Oocysts are rarely detected in dog feces; thus, serological surveys are the primary method for estimating the prevalence of canine neosporosis (Schares et al., 2005; Villagra-Blanco et al., 2018). The IFAT is considered the gold standard for the diagnosis of canine neosporosis (Björkman and Uggla, 1999). A meta-analysis study estimated the pooled seroprevalence of N. caninum in dogs based on the World Health Organization regions: Americas: 15.15%, Africa: 26.59%, Asia: 19.01%, Europe: 17.72%, and Oceania: 21.35% (Anvari et al., 2020). Based on the IFAT method, the pooled seroprevalence of N. caninum in dogs worldwide was 15.45% (Anvari et al., 2020). Studies on the seroprevalence of N. caninum in dogs using the IFAT have been conducted in South America in the last 20 years (Basso et al., 2001; Del Campo et al., 2003; Andreotti et al., 2006; Benetti et al., 2009; Vega et al., 2010; Sicupira et al., 2012; Arraes-Santos et al., 2016; Satragno et al., 2019) (Table 1). Although numerous recent epidemiological studies have been published on the prevalence of N. caninum in dogs, a substantial knowledge gap persists in several countries and territories, including Chile. In Chile, N. caninum has been detected in dairy cattle (Meléndez et al., 1999; Patitucci et al., 1999; Patitucci et al., 2000; Hervé-Claude et al., 2017; Tuemmers et al., 2017; Moroni et al., 2018), dogs (Patitucci et al., 1999; Patitucci et al., 2000; Patitucci et al., 2001; Hervé-Claude et al., 2017; Tuemmers et al., 2017; Moroni et al., 2018), horses (Patitucci et al., 2004), and native foxes (Pseudalopex fulvipes) (Patitucci et al., 2001). In dogs, the first study on N. caninum was conducted in La Araucanía Region, Southern Chile, demonstrating the presence of the parasite in four out of seven (57.1%) rural dogs using the IFAT (Patitucci et al., 2000). Later, in the same region and using the same diagnostic method, authors reported for the first time a seroprevalence of 18% in dogs from Chile, showing differences between rural and urban dog populations with varying seroprevalences of 26% (21/81) and 12.5% (15/120), respectively, for N. caninum (Patitucci et al., 2001). These results confirm the presence of N. caninum-specific antibodies in Chilean dogs; however, the seroprevalence of N. caninum has not been reported in other areas of Southern Chile, such as Valdivia Province, Los Ríos Region. This region has a high percentage of dairy cattle and contributes to approximately 31% of Chile´s milk production (ODEPA and Oficina de Estudios y Políticas Agrarias, 2019). Dairy cows are highly susceptible intermediate hosts for N. caninum infections, and their interaction with rural or farm dogs is recognized as a relevant risk factor for cattle abortion due to neosporosis (Antony and Williamson, 2003). The total economic impact of neosporosis on domestic livestock in Chile has not yet been determined, but should be rather high as reported for other countries (Reichel et al., 2013). In addition, based on a published human-to-dog ratio of 5.1:1, this region has an estimated owned dog population of 79,776 dogs (Garde et al., 2022).
Table 1. Prevalence of Neospora caninum in dogs from South America diagnosed by indirect immunofluorescence (IFAT).
This study aimed to determine the prevalence of serum antibodies to N. caninum in rural and urban dogs from Valdivia Province, Los Ríos Region, Chile, and to identify seropositivity-associated risk factors for canine neosporosis.
Materials and Methods
Animals and study area
A total of 164 dogs were sampled from December 2014 to November 2015 from Valdivia Province, Los Ríos Region, one of the main livestock areas in Southern Chile, including the dairy cattle industry. The sampling type was non-probabilistic by convenience. Blood samples were obtained from 108 domestic dogs from rural areas in Valdivia Province via cephalic venipuncture. Dogs were classified as “rural dogs” when they inhabit sparsely populated areas with extensive open spaces and agricultural settings. The rural areas included the Chaihuín (39°56’15“S, 73°35’19“W), Cadillal Alto (39°59’18“S, 73°31’11“W), Huiro (39°57’59“S, 73°38’52“W), and Huape (39°54’51“S, 73°30’55“W) localities (Fig. 1). Dogs were classified as “urban dogs” if they resided in densely populated areas with significant human infrastructure, including residential buildings, commercial establishments, and transportation systems. A total of 56 samples were obtained from domestic dogs from Valdivia city (39°48’30” S, 73°14’30” W) (Fig. 1). In rural areas, samples were obtained in the frame of the local distemper vaccination program, and samples from the urban area were collected during home visits to pet-owning households by local veterinarians, dogs attending the Veterinary Hospital of Universidad Austral de Chile (UACH, Valdivia, Chile), and dogs attending the Valdivia city sterilization program. Dogs were sampled regardless of age, sex, breed, health, or reproductive status.
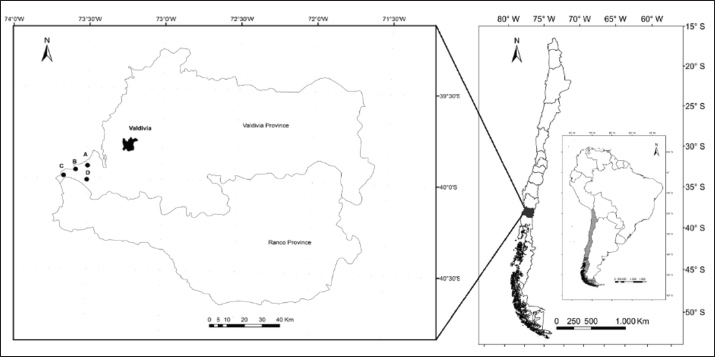
Fig. 1. Map of Los Ríos Region, Chile, and Valdivia Province, including Valdivia city (urban area) and the rural localities of Huape (A), Chaihuín (B), Huiro (C), and Cadillal Alto (D), where the samples were collected.
Owners signed a consent form before blood samples were collected. Dog owners were provided with a questionnaire to identify possible risk factors associated with N. caninum infections. Data obtained from the questionnaire included demographic factors, such as origin, sex, age, and reproductive status, and pet ownership factors, such as veterinary control.
Blood collection and preparation of serum samples
Blood samples were aseptically obtained from each dog’s cephalic vein. All samples were collected in a sterile plastic tube (Vacutainer®) without anticoagulant and immediately sent to the Veterinary Clinical Pathology Laboratory of Universidad Austral de Chile (UACH), Valdivia, Chile. Samples were centrifuged at 5,000 rpm for 5 min to obtain serum and then stored at −20°C until further analysis. Samples were then sent to the Immunoparasitology Laboratory, Faculdade de Ciências Agrárias e Veterinárias of the Universidade Estadual Paulista, Jaboticabal, Brazil, for application of the IFAT for detection of N. caninum-specific antibodies.
IFAT
The presence of N. caninum antibodies in the serum of each animal was detected by the IFAT in the Immunoparasitology Laboratory, Faculdade de Ciências Agrárias e Veterinárias of the Universidade Estadual Paulista, Jaboticabal, Brazil. Tachyzoites of N. caninum strain NC-1 were used as antigens (Dubey, 2003). The antigen slides were removed from storage at -20ºC and allowed to thaw at room temperature (RT) for 30 min. Subsequently, 10 μl of 2-fold dilutions of sera of 1:50 (cut-off for N. caninum) in PBS were placed in wells on antigen slides. Positive and negative canine sera were used as controls. Slides were incubated in a moist chamber at 37°C for 45 minutes, washed three times in phosphate-buffered saline (pH 7.2) for 5 minutes, and air-dried at RT. Immunoglobulin G (IgG) anti-dog conjugate (whole molecule with fluorescein isothiocyanate, dilution of 1:32; Sigma®, St. Louis, Missouri) was added to each well and diluted according to the manufacturer’s instructions. Slides were incubated again, washed, dried, and overlaid with buffered glycerin (pH 8.7), covered with glass coverslips, and examined using a fluorescence microscope (Olympus, Japan). Only a bright, linear peripheral fluorescence of the tachyzoite dog (taDog) sera showing a fluorescent signal at a titer of 1:50 was considered seropositive.
Statistical analysis
To determine the overall seroprevalence of N. caninum in dogs from rural and urban areas of Valdivia Province, the serologically positive dogs were divided by the total number of tested animals, multiplied by 100, and expressed as a percentage (%). Information about potential risk factors obtained from the questionnaire was converted into categorical data. Age was categorized according to the median of the sample because this variable did not have a normal distribution. Then, demographic factors such as age were divided into ≥3 years versus <3 years, origin into rural versus urban, sex into female versus male, and neutered into no versus yes. Pet ownership factors, such as veterinary control, were divided into no versus yes. Dogs with incomplete demographic or ownership information were excluded from the study. Logistic regression models were used to evaluate the relationships between risk factors and N. caninum serological positivity. First, univariate logistic regression models were used. All variables reaching a significance level of p ≤ 0.25 (measured using the likelihood-ratio test, LRT) were selected for incorporation into the multivariate logistic regression model. The model was built using a forward selection method in which only variables with a p-value ≤ 0.05 (calculated using the LRT) were considered. Hosmer–Lemeshow goodness-of-fit tests were used to assess the model’s fit. Logistic regression analysis was conducted using R software (version 3.6.1; R Foundation for Statistical Computing, Vienna, Austria).
Ethical approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Committee for Animal Care and Use of the Universidad Austral de Chile. Resolution number 250/2016.
Results
Seroprevalence of Neospora caninum in dogs
In this study, 164 dogs were analyzed using IFAT to detect antibodies against N. caninum. A total of 40 samples were positive, indicating a seroprevalence of 24.4% in dogs from Valdivia Province. The seropositivity of dogs in rural areas was 28.7% (31/108), whereas it was 16.1% (9/56) in urban areas.
Analysis of risk factors
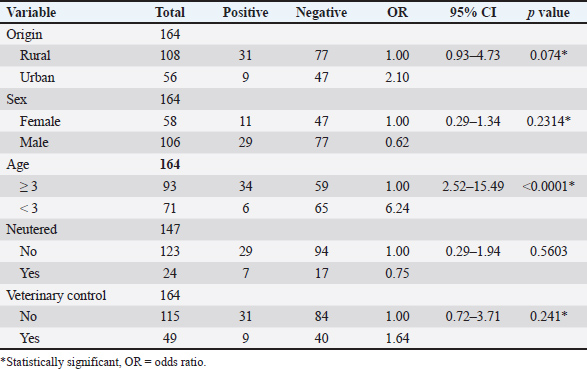
Of the analyzed risk factors, data regarding origin, sex, age, and veterinary control were obtained for 164 dogs, and data regarding reproductive status were collected for 147 dogs. From the five potential risk factors analyzed in the univariate logistic regression model for N. caninum, four were evaluated using the multivariate test: origin, sex, age, and veterinary control (Table 2). In the multivariate logistic regression model, only age remained significantly associated with N. caninum infection (odds ratio: 6.24, 95% confidence interval: 2.45–15.93, p < 0.0001*).
Table 2. Univariate logistic regression model of risk factors associated with Neospora caninum infection in dogs from the Valdivia province, Los Ríos region, southern Chile.
Discussion
This study constitutes the first report of neosporosis seroprevalence in dogs from Valdivia Province, southern Chile, with a seroprevalence of 24.4% (40/164) detected by IFAT (1:50). The results corroborate the presence of N. caninum-specific antibodies in the country and indicate an increased seroprevalence in dogs compared with a previous study that revealed a seroprevalence of 18% (36/201) in dogs from La Araucanía Region, southern Chile (Patitucci et al., 2001).
In the current study, a greater seropositivity for N. caninum was observed in rural dogs (28.7%) than in urban dogs (16.1%). Although the logistic regression analysis did not detect the origin variable as a risk factor for N. caninum seropositivity, it evidenced a tendency for higher exposure in rural dogs. These findings confirm earlier observations in the country that rural dogs present a higher risk of being seropositive for N. caninum (26%) (21/81) than urban dogs (12.5%) (Patitucci et al., 2001). The highest seropositivity in Valdivia Province can be associated with the presence of dairy cattle herds (ODEPA and Oficina de Estudios y Políticas Agrarias, 2019). An epidemiological relationship exists between the seropositivity of N. caninum in dogs and their contact with cattle (Antony and Williamson, 2003; Hornok et al., 2006; Malmasi et al., 2007; Da Cunha Filho et al., 2008; Robbe et al., 2016; Gao and Wang, 2019; Garde et al., 2022). Previous research in Southern Chile reported a prevalence of antibodies against N. caninum in cattle of 36.4% and 22.4%, respectively, suggesting a possible risk of infection for dogs through consumption of N. caninum-infected bovine meat or abortive material, as previously reported (Patitucci et al., 2001). Therefore, we hypothesize that horizontal transmission of neosporosis between cattle and dogs may be taking place at the affected farms. For each additional dog on a farm, the probability of a cow being seropositive for N. caninum increases by a factor of 1.13 (Corbellini et al., 2006).
The seroprevalence of N. caninum in dogs varies according to the country and locality (rural or urban) (Anvari et al., 2020). Several studies worldwide have reported a higher number of anti-N. caninum antibodies in rural dogs than in urban dogs (Hornok et al., 2006; Malmasi et al., 2007; Da Cunha Filho et al., 2008; Robbe et al., 2016; Gao and Wang, 2019). Brazil is the country with the most N. caninum seroprevalence studies (Anvari et al., 2020). Our results are similar to those already obtained in some Brazilian localities, with seroprevalences for rural dogs ranging from 25.3% (50/197) (Fridlund-Plugge et al., 2008) to 29% (7/24) (Romanelli et al., 2007). However, N. caninum IFAT prevalence in rural dogs in Argentina was higher at 49.3%, considered among the highest reported worldwide (Basso et al., 2001). The seroprevalences obtained in this study are higher than those obtained in some European countries, such as Austria, where the prevalence in rural dogs is 5.3% (23/433) (Wanha et al., 2005), Hungary, with 6% (15/249) (Hornok et al., 2006), and Italy, with 15% (58/391) (Machacova et al., 2016). It is also higher than that reported in Asian countries, namely, China with a seroprevalence of 20% (95/476) (Gao and Wang, 2019), South Korea with 21.6% (11/51) (Kim et al., 2003), and Iran with 28% (14/50) (Haddadzadeh et al., 2007). Several studies across South American countries have documented a high prevalence of bovine neosporosis, with rates of 20.3% in Argentina (Fort et al., 2015), 21.6% in Brazil (Bruhn et al., 2013), and 28.3% in Colombia (Llano et al., 2018), suggesting a potential risk of infection in rural dogs.
The higher seroprevalence observed in dogs from rural areas in the South of Chile could also be related to their diet, as dogs in these areas are commonly fed with raw bovine meat (Patitucci et al., 2000; Patitucci et al., 2001). In Argentina, a neighboring country with among the highest N. caninum-seroprevalences in rural and urban dogs, dogs are commonly fed raw or undercooked beef (Campero et al., 2023). Diets that include raw bovine meat are a proven risk factor for neosporosis in rural dogs in Chile (Patitucci et al., 2001). This is in accordance with the literature, which established a relationship between the type of diet and the presentation of N. caninum, mainly when dogs consume uncooked meat, placenta, and aborted bovine fetuses (or other intermediary hosts) (Patitucci et al., 2001; Da Cunha Filho et al., 2008; Gao and Wang, 2019). The horizontal transmission of N. caninum (feeding infected tissues) is supported by an increase in seroprevalence in rural dogs (Malmasi et al., 2007). Dogs originating from dairy farms, regardless of whether these farms have a history of cattle abortion, exhibited a higher seroprevalence than those from urban regions (Dubey and Schares, 2011). This disparity may be due to the increased likelihood of cattle fetuses or placentas, or uterine discharges infected with either tachyzoites or cysts containing bradyzoites of N. caninum (Dubey, 2003; Dijkstra et al., 2002). Variations in the seroprevalence of N. caninum in dogs may reflect differing attitudes and management practices of dog owners regarding their dogs’ diets.
Small mammals, such as rodents, have been proposed as additional potential sources of N. caninum infection in dogs and foxes (Ferroglio et al., 2001). In China, 5.8% of wild brown rats (Rattus norvegicus) tested positive for N. caninum by PCR, and in the UK, 4.4% of wild brown rats and 3% of free-ranging mice (Mus musculus) tested positive for N. caninum (Huang et al., 2004; Hughes et al., 2006). Rural and farm dogs frequently prey on small mammals; therefore, these animals may represent another potential source of infection. Future research should explore the impact of different dietary practices in the rural areas of this particular region and investigate the role of small mammals as natural reservoir hosts for N. caninum.
This study also found that urban dogs from the city of Valdivia presented positive serology for N. caninum, with a seroprevalence of 16.1% (9/56). This result showed that dogs living in the urban areas of Valdivia Province were also exposed to N. caninum. The observed values were higher than those reported in Brazil, where the seroprevalence of urban dogs was 2.6% (4/156) (Sicupira et al., 2012). In addition, the high indices of seropositivity obtained in urban dogs from Valdivia city were greater than those reported in European- and Asian countries, such as Hungary with a seroprevalence of 1% (4/402) (Hornok et al., 2006), Austria with 2.1% (8/381) (Wanha et al., 2005), the Czech Republic with 2.6% (5/195) (Václavek et al., 2007), Spain with 2.9% (3/102) (Collantes-Fernández et al., 2008), and South Korea with 8.3% (24/289) (Machacova et al., 2016). The high seropositivity rates obtained in urban areas in our study could be related to both horizontal and vertical transmission routes. Horizontal transmission is mainly associated with the feed type offered by the owners. The marked increase in N. caninum seroprevalence in urban dogs over the last 20 years has been associated with an increased habit of enriching the commercial diet of domestic dogs with raw meat (Collantes-Fernández et al., 2008; Sloan et al., 2017). Chile does not diverge from this international trend, and according to previous studies, 52% of southern Chilean dogs are fed with uncooked meat (Patitucci et al., 2000; Patitucci et al., 2001; Tuemmers et al., 2017; Hervé-Claude et al., 2017; Moroni et al., 2018), which could explain the high levels of seropositivity in urban dogs in this study. This fact is consistent with other studies that highlight that the consumption of homemade meals with raw or undercooked meat that also contains N. caninum cysts significantly increases the risk of canine neosporosis (Goździk et al., 2011). A study on beef meat detected 2% of samples as PCR-positive to N. caninum (Wyss et al., 2000), and a study conducted in Iran found that 8% of raw and undercooked chicken meat tested positive for N. caninum-DNA, suggesting it may serve as a potential risk factor for canine neosporosis (Barimani et al., 2023). Additionally, dogs from peripheral neighborhoods in urban areas near forest areas might become infected due to proximity to other animal species, including wildlife, which play an important role in the transmission of this parasitic infection (Souza et al., 2019).
It is estimated that 48% (65/136) of dogs in the South of Chile are fed with a “no raw” diet (Patitucci et al., 2001); in addition to this, other authors stated that the likelihood of postnatal transmission in pet dogs is relatively low (Souza et al., 2019). In this case, vertical transmission as a possible route of infection in urban dogs from Valdivia Province should be considered. It has been reported that N. caninum can be transmitted congenitally from infected bitches to their offspring, especially when the bitches have high antibody titers before gestation (Barber and Trees, 1998; Dubey et al., 2005; Goździk et al., 2011). Other authors have already associated this route of infection with the increase in anti-N. caninum antibodies in urban dogs (Dubey et al., 2005; Hughes et al., 2006).
Age has been a controversial risk factor for N. caninum seropositivity in dogs due to the lack of a significant relationship among research results (Gavrea et al., 2012; Prandini Da Costa Reis et al., 2016; Kwok et al., 2018). However, of all the variables analyzed in this study, age was the only significant risk factor for N. caninum seropositivity. Dogs ≥3 years old were at increased risk of N. caninum infection. This result differs from that previously reported in Chile (Patitucci et al., 2001), which found no relationship between seropositivity for N. caninum and age. However, it coincides with other studies conducted in Brazil and the Czech Republic, where the prevalence of anti-N. caninum antibodies were higher in dogs aged >3 years (Václavek et al., 2007; Da Cunha Filho et al., 2008) and between 3 and 3.5 years of age (Hughes et al., 2006). Other authors also studied age as a risk factor, but employed different ranges to determine how this variable affects the presence of anti-N. caninum antibodies, confirming that seropositivity increased with age (Haddadzadeh et al., 2007; Václavek et al., 2007). The higher odds of seropositivity in older dogs (≥3 years) suggest that horizontal transmission is more important in dogs from Southern Chile. Vertical transmission of N. caninum in dogs is highly variable because the probability of transmission is directly related to the level of antibodies from the bitch. In addition, infection in puppies is unlikely to persist in the absence of horizontal transmission (Wyss et al., 2000; Goździk et al., 2011; Souza et al., 2019; Barimani et al., 2023). Conversely, adult dogs have a more developed carnivorous or hunting behavior and are therefore more likely to consume infected tissue. As the literature shows, this hunting behavior of dogs has a higher relevance in acquiring N. caninum infections (Dubey et al., 2007). However, another study indicates that adult dogs shed fewer oocysts than puppies following primary exposure, and puppies may also re-excrete oocysts after new challenges, suggesting that age can affect oocyst production (Kwok et al., 2018). Therefore, future studies are necessary to elucidate the effect of age in relation to N. caninum infection in canines.
The other analyzed risk factors, such as sex, reproductive state, and veterinary inspections, were not significantly associated with canine neosporosis in this study. Similar results have been reported worldwide, indicating that there are no differences between males and females at the moment of acquiring N. caninum infections (Basso et al., 2001; Wanha et al., 2005; Haddadzadeh et al., 2007). Another study also failed to find a relationship between the reproductive status of dogs and the presence of the parasite (Garde et al., 2022). Pet ownership-related factors, such as veterinary control, were not significant variables for N. caninum seropositivity. However, this variable is important to consider to detect the clinical presentation of the diseases; of 56 dogs seropositive for N. caninum as assessed by the ELISA test from a study conducted in Poland, 12 had clinical symptoms that were compatible with canine neosporosis (Goździk et al., 2011). Therefore, regular health controls might prevent the underdiagnosis of this parasitic disease in dog populations.
According to what was previously described, the seroprevalence and risk factors for N. caninum seropositivity in dogs are consistent with those reported worldwide. Nevertheless, further studies are needed to determine with more certainty the sources of N. caninum infections in dogs in southern Chile. For example, by analyzing the kind of feedstuff offered to rural and urban dogs, the level of contact with suitable wild intermediate hosts (e.g., mice, rats, rabbits, and hares), and the level of transmission to offspring would be of merit. Finally, clinical cases of canine neosporosis have not been reported in Chile, which can be associated with a lack of awareness of the disease, consequently leading to its underdiagnosis. Nevertheless, due to the high prevalence detected in this study in rural and urban areas, researchers should continue to investigate N. caninum and consider the clinical presentation of neosporosis in dogs. This study demonstrates for the first time anti-N. caninum antibodies in dogs from the Valdivia Province, Los Ríos Region, Southern Chile. The elevated rate of infection in rural and urban dogs highlights the need to implement control measures targeting both dogs and cattle to prevent the adverse economic impact of neosporosis not only in dairy cattle but also in sheep, goats, horses, and endemic New World Camelids.
The limitations of this investigation include the sampling in a restricted geographical area and the limited epidemiological information obtained from dogs. The selection method used may have resulted in a population sample that is not entirely representative of the entire dog population of the analyzed region. Therefore, the overall prevalence of rural and urban dogs may have been underestimated. The conduction of a massive sampling, including extensive information on the animals, especially focusing on dogs ≥3 years, is recommended in rural and urban dog populations in Los Ríos Region to obtain a more representative picture of the disease prevalence. Moreover, further studies should include a broader geographical range, a larger sample size to obtain a more representative population, and the molecular detection of tachyzoites/bradyzoites to confirm active infection and generate genetic diversity data on N. caninum circulating in Southern Chile, as reported for other countries (Regidor-Cerrillo et al., 2011).
Conclusion
In this study, the seroprevalence of N. caninum in dogs from Valdivia Province was higher than that previously reported in Chile and other parts of the world. In addition, rural dogs exhibited greater seropositivity than urban dogs, and dogs aged 3 years or older were identified as being at a higher risk of infection. This study demonstrates for the first time the presence of anti-N. caninum antibodies in rural and urban dogs from Valdivia Province, Los Ríos Region, Southern Chile, and calls for larger dog-related monitoring studies, including bitches and their offspring, to better understand the role of vertical transmission in Chile.
Acknowledgments
The authors would like to thank Drs. Carla Freschi and F. Soto from FCAV/UNESP, São Paulo, Brazil, for their assistance in the analysis of samples using the IFAT test.
Conflict of interest
The authors declare no conflict of interest.
Funding
None.
Authors’ contributions
All authors participated in the drafting of the manuscript. KA, AM, PB, and MG participated in the study design and conception. KA, MRA, RZM, PB, and AM performed the tests and collected the required data. The collected data were analyzed by KA, CH, AT, and MG. All authors have revised and approved the final version of the manuscript.
Data availability
All data supporting this study’s findings are available in the manuscript.
References
Andreotti, R., Oliveira, J.M., Silva, E.A.E., Oshiro, L.M. and Matos, M.D.F.C. 2006. Occurrence of Neospora caninum in dogs and its correlation with visceral leishmaniasis in the urban area of campo grande, Mato Grosso Do Sul, Brazil. Vet. Parasitol. 135, 375–379; doi:10.1016/j.vetpar.2005.10.011
Antony, A. and Williamson, N. 2003. Prevalence of Antibodies to Neospora caninum in dogs of rural or urban origin in central New Zealand. N. Z. Vet. J. 51, 232–237; doi:10.1080/00480169.2003.36372
Anvari, D., Saberi, R., Sharif, M., Sarvi, S., Hosseini, S.A., Moosazadeh, M., Hosseininejad, Z., Chegeni, T.N. and Daryani, A. 2020. Seroprevalence of Neospora caninum infection in dog population worldwide: a systematic review and meta-analysis. Acta Parasitol. 65, 1–18; doi:10.2478/s11686-019-00163-4
Arraes-Santos, A.I., Araújo, A., Guimarães, M., Santos, J., Pena, H., Gennari, S., Azevedo, S., Labruna, M. and Horta, M. 2016. Seroprevalence of anti-Toxoplasma gondii and anti-Neospora caninum antibodies in domestic mammals from two distinct regions in the semi-arid region of northeastern Brazil. Vet. Parasitology 135, 375–379; doi:10.1016/j.vprsr.2016.08.007
Barber, J.S. and Trees, A.J. 1998. Naturally occurring vertical transmission of Neospora caninum in dogs. Int. J. Parasitol. 28, 57–64; doi:10.1016/s0020-7519(97)00171-9
Barimani, S., Rassouli, M. and Emadi Chashmi, S.H. 2023. Molecular detection of Neospora caninum in chicken meat and eggs in Iran. Vet. Parasitol. Reg. Stud. Rep. 40, 100862; doi:10.1016/j.vprsr.2023.100862
Basso, W., Venturini, L., Venturini, M.C., Moore, P., Rambeau, M., Unzaga, J.M., Campero, C., Bacigalupe, D. and Dubey, J.R. 2001. Prevalence of Neospora caninum infection in dogs from beef-cattle farms, dairy farms, and from urban areas of Argentina. J. Parasitol. 87(2001), 906–907.
Benetti, A.H., Schein, F.B., Santos, T.R.D., Toniollo, G.H., Costa, A.J.D., Mineo, J.R., Lobato, J., Silva, D.A.D.O. and Gennari, S.M. 2009. Pesquisa de anticorpos anti-Neospora caninum em bovinos leiteiros, cães e trabalhadores rurais da região sudoeste do estado de Mato Grosso. Rev. Bras. Parasitol. Vet. 18, 29–33; doi:10.4322/rbpv.018e1005
Björkman, C. and Uggla, A. 1999. Serological diagnosis of Neospora caninum infection. Int. J. Parasitol. 29, 1497–1507; doi:10.1016/S0020-7519(99)00115-0
Bruhn, F.R.P., Daher, D.O., Lopes, E., Barbieri, J.M., Da Rocha, C.M.B.M. and Guimarães, A.M. 2013. Factors associated with seroprevalence of Neospora caninum in dairy cattle in southeastern Brazil. Trop. Anim. Health. Prod. 45, 1093–1098; doi:10.1007/s11250-012-0330-y
Campero, L.M., Basso, W., Moré, G., Fiorani, F., Hecker, Y.P., Echaide, I., Cantón, G.J., Cirone, K.M., Campero, C.M., Venturini, M.C. and Moore, D.P. 2023. Neosporosis in Argentina: past, present and future perspectives. Vet. Parasitol. Reg. Stud. Rep. 41, 100882. doi:10.1016/j.vprsr.2023.100882
Collantes-Fernández, E., Gómez-Bautista, M., Miró, G., Álvarez-García, G., Pereira-Bueno, J., Frisuelos, C. and Ortega-Mora, L.M. 2008. Seroprevalence and risk factors associated with Neospora caninum infection in different dog populations in Spain. Vet. Parasitol. 152(1-2), 148–151; doi:10.1016/j.vetpar.2007.12.005
Corbellini, L.G., Smith, D.R., Pescador, C.A., Schmitz, M., Correa, A., Steffen, D.J. and Driemeier, D. 2006. Herd-level risk factors for Neospora caninum seroprevalence in dairy farms in southern Brazil. Prev. Vet. Med. 74, 130–141.
Da Cunha Filho, N.A., Lucas Ada, S., Pappen, F.G., Ragozo, A.M., Gennari, S.M., Junior, T.L. and Farias, N.A. 2008. Fatores de risco e prevalência de anticorpos anti-Neospora caninum em cães urbanos e rurais do Rio Grande do Sul, Brasil. Rev. Bras. Parasitol. Vet. 17, 301–306.
Del Campo S., J., Chávez V., A., Delgado C., A., Falcón P., N., Ornelas A., A., Casas A., E. and Serrano M., E. 2003. Frecuencia de Neospora caninum en perros de establos lecheros del Valle de Lima. Rev. Inv. Vet. Perú. 14(2), 145–149; doi:10.15381/RIVEP.V14I2.1619
Dijkstra, T., Barkema, H.W., Eysker, M., Hesselink, J.W. and Wouda, W. 2002. Natural transmission routes of Neospora caninum between farm dogs and cattle. Vet. Parasitol. 30, 99–104; doi:10.1016/s0304-4017(02)00010-9
Dubey, J.P. 1999. Recent advances in Neospora caninum and neosporosis. Vet. Parasitol. 84, 349–367; doi:10.1016/S0304-4017(99)00044-8
Dubey, J.P. 2003. Review of Neospora caninum and neosporosis in animals. Korean. J. Parasitol. 41, 1–16; doi:10.3347/kjp.2003.41.1.1
Dubey, J.P. and Lindsay, D.S. 1996. A Review of Neospora caninum and neosporosis. Vet. Parasitol. 67, 1–59; doi:10.1016/S0304-4017(96)01035-7
Dubey, J.P. and Schares, G. 2011. Neosporosis in animals-the last five years. Vet. Parasitol. 180, 90–108; doi:10.1016/j.vetpar.2011.05.031
Dubey, J.P., Knickman, E. and Greene, C.E. 2005. Neonatal Neospora caninum infections in dogs. Acta. Parasitol. 50, 176–179; doi:10.2460/javma.1988.193.10.1259
Dubey, J.P., Schares, G. and Ortega-Mora, L.M. 2007. Epidemiology and control of neosporosis and Neospora caninum. Clin. Microbiol. Rev. 20, 323–367; doi:10.1128/CMR.00031-06
Ferroglio, E., Bassano, B., Trisciuoglio, A. and Rossi, L. 2001. Antibodies to Neospora caninum in Alpine ibex from the Italian Alps. Z. Jagdwiss. 47, 226–228; doi:10.1007/BF02241552
Fort, M., Edelsten, M., Maley, S. and Innes, E. 2015. Seroepidemiological study of Neospora caninum in beef and dairy cattle in La Pampa, Argentina. Acta Parasitol. 60(2), 275–282; doi:10.1515/ap-2015-0039
Fridlund-Plugge, N., Montiani-Ferreira, F., Richartz, R.R.T.B., Dal Pizzol, J., Machado Jr, P.C., Patrício, L.F.L., Rosinelli, A.S. and Locatelli-Dittrich, R. 2008. Frequency of antibodies against Neospora caninum in stray and domiciled dogs from urban, periurban and rural areas from Paraná State, Southern Brazil. Rev. Bras. Parasitol. Vet. 17, 222–226; doi:10.1590/S1984-29612008000400010
Gao, X. and Wang, H. 2019. Seroprevalence and risk factors for Neospora caninum infection in dogs in rural northeastern Mainland China. Parasite 26, 32. doi:10.1051/parasite/2019034
Garde, E., Marín-Vial, P., Pérez, G.E. and Sandvig, E.M. 2022. A Review and analysis of the national dog population management program in Chile. Animals 12, 228; doi:10.3390/ani12030228
Gavrea, R., Mircean, V., Pastiu, A. and Cozma, V. 2012. Epidemiological survey of Neospora caninum infection in dogs from Romania. Vet. Parasitol. 188, 382–385; doi:10.1016/j.vetpar.2012.03.044
Gondim, L.F.P., Mcallister, M.M., Pitt, W.C. and Zemlicka, D.E. 2004. Coyotes (Canis Latrans) are definitive hosts of Neospora caninum. Int. J. Parasitol. 34, 159–161; doi:10.1016/j.ijpara.2004.01.001
Goździk, K., Wrzesień, R., Wielgosz-Ostolska, A., Bień, J., Kozak-Ljunggren, M. and Cabaj, W. 2011. Prevalence of antibodies against Neospora caninum in dogs from urban areas in Central Poland. Parasitol. Res. 108, 991–996; doi:10.1007/s00436-010-2143-0
Haddadzadeh, H.R., Sadrebazzaz, A., Malmasi, A., Talei Ardakani, H., Khazraii Nia, P. and Sadreshirazi, N. 2007. Seroprevalence of Neospora caninum infection in dogs from rural and urban environments in Tehran, Iran. Parasitol. Res. 101, 1563–1565; doi:10.1007/s00436-007-0678-5
Hervé-Claude, L., Alvarado, A., Rivera, D., Navarrete-Talloni, M. and Hamilton-West, C. 2017. Seroprevalence and risk factors for Neospora caninum in small dairy farms in central Chile. Rev. MVZ. Córdoba. 22, 5666–5673; doi:10.21897/rmvz.926
Hornok, S., Edelhofer, R., Fok, E., Berta, K., Fejes, P., Répási, A. and Farkas, R. 2006. Canine neosporosis in Hungary: screening for seroconversion of household, herding and stray dogs. Vet. Parasitol. 137, 197–201; doi:10.1016/j.vetpar.2006.01.030
Huang, C.C., Yang, C.H., Watanabe, Y., Liao, Y.K. and Ooi, H.K. 2004. Finding of Neospora caninum in the wild brown rat (Rattus norvegicus). Vet. Res. 35, 283–290; doi:10.1051/vetres:2004010
Hughes, J.M., Williams, R.H., Morley, E.K., Cook, D.A.N., Terry, R.S., Murphy, R.G., Smith, J.E. and Hide, G. 2006. The prevalence of Neospora caninum and co-infection with Toxoplasma gondii by PCR analysis in naturally occurring mammal populations. Parasitology 132, 29–36; doi:10.1017/S0031182005008784
Kim, J.H., Kang, M.S., Lee, B.C., Hwang, W.S., Lee, C.W., So, B.J., Dubey, J.P. and Kim, D.Y. 2003. Seroprevalence of antibodies to Neospora caninum in dogs and Raccoon dogs in Korea. Korean. J. Parasitol. 41, 243–245; doi:10.3347/kjp.2003.41.4.243
King, J.S., Šlapeta, J., Jenkins, D.J., Al-Qassab, S.E., Ellis, J.T. and Windsor, P.A. 2010. Australian dingoes are definitive hosts of Neospora caninum. Int. J. Parasitol. 40, 945–950; doi:10.1016/j.ijpara.2010.01.008
Kwok, B., Crisman, R., Malik, R. and Šlapeta, J. 2018. Presumptive vertical transmission of Neospora caninum in related Bernese Mountain dogs. Vet. Parasitol. Reg. Stud. Rep. 14(7), 7–10; doi:10.1016/j.vprsr.2018.07.011
Llano, H.A.B., Guimarães, M.S., Soares, R.M., Polo, G. and Da Silva, A.C.S. 2018. Eroprevalence and risk factors for Neospora caninum infection in cattle from the eastern Antioquia, Colombia. Vet. Anim. Sci. 6, 69–74; doi:10.1016/j.vas.2018.03.001
Machacova, T., Bartova, E., Sedlak, K., Slezakova, R., Budikova, M., Piantedosi, D. and Veneziano, V. 2016. Seroprevalence and risk factors of infections with Neospora caninum and Toxoplasma gondii in hunting dogs from Campania region, southern Italy. Folia Parasitol. 63(1), 1–5; doi:10.14411/fp.2016.012
Malmasi, A., Hosseininejad, M., Haddadzadeh, H., Badii, A. and Bahonar, A. 2007. Serologic study of anti-Neospora caninum antibodies in household dogs and dogs living in dairy and beef cattle farms in Tehran, Iran. Parasitol. Res. 100, 1143–1145; doi:10.1007/s00436-006-0385-7
Mcallister, M.M., Dubey, J.P., Lindsay, D.S., Jolley, W.R., Wills, R.A. and Mcguire, A.M. 1998. Dogs are definitive hosts of Neospora caninum. Int. J. Parasitol. 28, 1473–1478; doi:10.1016/S0020-7519(98)00138-6
Meléndez, P., Concha, C., Donovan, A. and Bjorkman, C. 1999. Evidencia serológica de Neospora caninum en un rebaño lechero de la zona central de Chile. Av. Cs. Vet. 14, 13–16; doi:10.5354/acv.v14i1-2.9022
Moroni, M., Navarro, M., Paredes, E., Romero, A., Alberdi, A., Lischinsky, T., Moore, D., Campero, C. and Uzal, F. 2018. Identification of Neospora caninum in aborted bovine fetuses of Southern Chile. Braz. J. Vet. Pathol. 11, 37–41; doi:10.24070/bjvp.1983-0246.v11i2p37-41
ODEPA, Oficina de Estudios y Políticas Agrarias, Chile. 2019. Descripción de la Cadena Láctea en Chile
Patitucci, A.N., Pérez, M.J., Cárcamo, C.M. and Baeza, L. 2004. Presencia de anticuerpos séricos contra Neospora caninum en equinos de Chile. Arch. Med. Vet. 36, 203–206; doi:10.4067/S0301-732X2004000200011
Patitucci, A.N., Perez, M.J., Israel, K.F. and Rozas, M.A. 2000. Prevalencia de anticuerpos séricos contra Neospora caninum en dos rebaños lecheros de la IX Región de Chile. Archivos Médicos Veterinarios 36, 203–206; doi:10.4067/S0301-732X2000000200008
Patitucci, A.N., Pérez, M.J., Luders, C.F., Ratto, M.H. and Dumont, A.G. 1999. Evidencia serológica de infección por Neospora caninum en rebaños lecheros del sur de Chile. Arch. Med. Vet. 31, 215-218. http://dx.doi.org/10.4067/S0301-732X1999000200009.
Patitucci, A.N., Pérez, M.J., Rozas, M.A. and Israel, K.F. 2001. Neosporosis canina: presencia de anticuerpos séricos en poblaciones caninas rurales y urbanas de Chile. Arch. Med. Vet. 33, 227–232; doi:10.4067/S0301-732X2001000200011
Prandini Da Costa Reis, R., Crisman, R., Roser, M., Malik, R. and Šlapeta, J. 2016. Neonatal neosporosis in a 2-week-old Bernese Mountain dog infected with multiple Neospora caninum strains based on MS10 microsatellite analysis. Vet. Parasitology. 221, 134–138; doi:10.1016/j.vetpar.2016.03.023
Regidor-Cerrillo, J., Gómez-Bautista, M., Sodupe, I., Aduriz, G., Álvarez-García, G., Del Pozo, I. and Ortega-Mora, L. 2011. In vitro invasion efficiency and intracellular proliferation rate comprise virulence-related phenotypic traits of Neospora caninum. Vet. Res. 42, 41; doi:10.1186/1297-9716-42-41
Reichel, M.P., Alejandra Ayanegui-alcérreca, M., Gondim, L.F.P. and Ellis, J.T. 2013. What is the global economic impact of Neospora caninum in cattle - the billion dollar question. Int. J. Parasitol. 43, 133–142; doi:10.1016/j.ijpara.2012.10.022
Robbe, D., Passarelli, A., Gloria, A., Di Cesare, A., Capelli, G., Iorio, R. and Traversa, D. 2016. Neospora caninum seropositivity and reproductive risk factors in dogs. Exp. Parasitol. 164, 31–35; doi:10.1016/j.exppara.2016.02.003
Romanelli, P.R., Freire, R.L., Vidotto, O., Marana, E.R.M., Ogawa, L., De Paula, V.S.O., Garcia, J.L. and Navarro, I.T. 2007. Prevalence of Neospora caninum and Toxoplasma gondii in sheep and dogs from Guarapuava farms, Paraná State, Brazil. Res. Vet. Sci. 82(2), 202–207; doi:10.1016/j.rvsc.2006.04.001
Satragno, D., Pavón-Rocha, A.J., Rábago-Castro, J.L. and La Cruz-hernández, N.I.D. 2019. Alta seroprevalencia de Neospora caninum en perros con sospecha clínica de neosporosis en Montevideo, Uruguay. Rev. Argent. Microbiol. 52, 165–166; doi:10.1016/j.ram.2019.03.007
Schares, G., Pantchev, N., Barutzki, D., Heydorn, A.O., Bauer, C. and Conraths, F.J. 2005. Oocysts of Neospora caninum, Hammondia heydorni, Toxoplasma gondii and Hammondia hammondi in faeces collected from dogs in Germany. Int. J. Parasitol. 35, 1525–1537; doi:10.1016/j.ijpara.2005.08.008
Sicupira, P.M.L., De Magalhães, V.C.S., Da Silva Galvão, G., Pereira, M.J.S., Gondim, L.F.P. and Munhoz, A.D. 2012. Factors associated with infection by Neospora caninum in dogs in Brazil. Vet. Parasitol. 185, 305–308; doi:10.1016/j.vetpar.2011.09.029
Sloan, S., Šlapeta, J., Jabbar, A., Hunnam, J., De Groef, B., Rawlin, G. and McCowan, C. 2017. High seroprevalance of Neospora caninum in dogs Victoria, Australia, compared to 20 years ago. Parasites Vectors 10, 1–9; doi:10.1186/s13071-017-2464-2
Souza, I.B.D., Fernandes, P.R., Silva, T.R.M., Santos, C.V.B., Silva, N.M.M.D., Ubirajara Filho, C.R.C., Carvalho, G.A.D., Alves, L.C., Mota, R.A. and Ramos, R.A.N. 2019. Seroprevalence of Neospora caninum and Toxoplasma gondii in dogs from an urban area of North-eastern Brazil: a spatial approach. Rev. Soc. Bras. Med. Trop. 52, e20180440; doi:10.1590/0037-8682-0440-2018
Tuemmers, C., Valenzuela, G., Nuñez, C., De La Cruz, R., Meyer, J., Andaur, M., Leyan, P. and Mora, C. 2017. Seroprevalencia de Neospora caninum en bovinos de una feria ganadera de la región de La Araucanía, Chile. Rev. Investig. Vet. Perú 28(3), 629–635; doi:10.15381/rivep.v28i3.12680
Václavek, P., Sedlák, K., Hůrková, L., Vodrážka, P., Šebesta, R. and Koudela, B. 2007. Serological survey of Neospora caninum in dogs in the Czech Republic and a long-term study of dynamics of antibodies. Vet. Parasitology 143, 35–41; doi:10.1016/j.vetpar.2006.07.020
Vega O., L., Chávez V., A., Falcón P., N., Casas A., E. and Puray Ch., N. 2010. Prevalencia de Neospora caninum en perros pastores de una empresa ganadera de la sierra sur del Perú. Rev. Inv. Vet. Perú 21(1), 80–86; doi:10.15381/rivep.v21i1.311
Villagra-Blanco, R., Angelova, L., Conze, T., Schares, G., Bärwald, A., Taubert, A., Hermosilla, C. and Wehrend, A. 2018. Seroprevalence of Neospora caninum-specific antibodies in German breeding bitches. Parasit. Vectors 11, 96; doi:10.1016/j.ijppaw.2017.09.002
Villagra-Blanco, R., Silva, L.M.R., Aguilella-Segura, A., Arcenillas-Hernández, I., Martínez-Carrasco, C., Seipp, A., Gärtner, U., Ruiz De Ybañez, R., Taubert, A. and Hermosilla, C. 2017. Bottlenose dolphins (Tursiops truncatus) do also cast neutrophil extracellular traps against the apicomplexan parasite Neospora caninum. Int. J. Parasitol. Parasites Wildl. 6, 287–294; doi:10.1186/s13071-018-2683-1
Wanha, K., Edelhofer, R., Gabler-Eduardo, C. and Prosl, H. 2005. Prevalence of antibodies against Neospora caninum and Toxoplasma gondii in dogs and foxes in Austria. Vet. Parasitol. 28, 189–1893; doi:10.1016/j.vetpar.2004.11.027
Wyss, R., Sager, W.R., Muller, N., Inderbitzin, F., Konig, M., Audige, L. and Gottstein, B. 2000. Untersuchungen zum Vorkommen von Toxoplasma gondii und Neospora caninum unter fleischhygienischen Aspekten. Schweiz. Archiv. Für. Tierheilkunde. 142, 95–108; doi:10.5169/seals-590222











