| Review Article | ||
Open Vet. J.. 2025; 15(12): 6166-6189 Open Veterinary Journal, (2025), Vol. 15(12): 6166-6189 Review Article Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024)Pavol Švorc, Soňa Grešová and Viktória Novotná*1Department of Medical Physiology, Faculty of Medicine, Pavol Jozef Šafárik University, Košice, Slovakia *Corresponding Author: Viktória Novotná. Department of Medical Physiology, Faculty of Medicine, Pavol Jozef Šafárik University, Košice, Slovakia. Email: viktoria.novotna [at] student.upjs.sk Submitted: 23/07/2025 Revised: 20/10/2025 Accepted: 03/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractRats are frequently used in laboratory experiments and are suitable animal models for physiology, pharmacology, pathophysiology, or behavioral studies. Since rats are also used in experimental surgery, adequate anesthesia and analgesia must be ensured to comply with the principle of refinement within the “3R” rules. This study evaluated all available numerical blood pressure (BP) values reported in studies identified through a comprehensive search using PubMed, Web of Science, Scopus, ScienceDirect, and Google Scholar between 2021 and 2024 to examine whether control (baseline) BP values differ at the beginning of the experiment between various anesthetics and the site of invasive BP measurement in normotensive and hypertensive rats. Statistical analysis in normotensive male rats focused on conscious animals and those under the most commonly used anesthetics, with individual agents and their combinations categorized into four groups: urethane, ketamine, barbital, and isoflurane. No statistical comparison could be performed in hypertensive rats due to the insufficient number of data extracted. Our results suggest that the choice among these anesthetics is probably not a consistent determinant of control baseline BP values in normotensive rats when considering only the carotid and femoral arteries, which are the two most frequent sites of invasive measurement. These findings should be interpreted with caution because of several limitations, including the small number of extracted data in some groups, their uneven distribution, underpowered data in hypertensive rats, and variability related to experimental conditions, e.g., circadian rhythms, animal handling, omission of females, among others. However, we propose that most anesthetics may cause a slight initial decrease in control baseline BP at the beginning of the experiment compared to conscious rats, which warrants further investigation particularly concerning the invasive measurement site and aforementioned limitations. Keywords: Anesthesia, Blood pressure monitoring, Laboratory animals, Rats. IntroductionIn in vivo experiments, homeostatic regulatory mechanisms are neither completely eliminated nor deactivated, which means that insight into the investigated phenomenon is gained in the context of all its complexity. Because many in vivo experiments begin by inducing general anesthesia in the animals, the blood pressure (BP), an important cardiovascular parameter affecting the animal’s state, can be affected before the experimentation even begins. As such, the extent to which BP is altered after anesthetic administration may introduce a problem before assessing the phenomenon being studied. Most of the physiological and pharmacological knowledge regarding BP and its regulation is derived from studies performed on acutely prepared, anesthetized, or immobilized laboratory animals (Kramer and Remie, 2005). BP in small laboratory animals, such as rats or mice, can be measured using either invasive or noninvasive techniques, each with specific advantages and limitations that depend on the study design. Noninvasive methods, such as the tail cuff, are less expensive, effective, and do not require surgery (Kapsdorferová et al., 2024). Sun et al. (2025) highlighted the potential of contactless monitoring of respiratory rate variability under isoflurane as an additional indicator of hemodynamic stability. Such an approach reduces animal stress and improves data quality by providing a precise, non invasive, and reliable method for monitoring respiratory rates in experimental settings (Sun et al., 2025). Invasive methods, including telemetry and direct arterial catheterization, are generally considered the most accurate and physiologically relevant and are often regarded as the gold standard. However, these patients require surgical intervention and anesthesia, which can cause fluctuations and affect normal BP regulation (Kapsdorferová et al., 2024). Urethane or pentobarbitone are considered the most suitable anesthetics for performing invasive BP measurements in rats because they do not cause significant changes in basal hemodynamic parameters (De Wildt et al., 1983; Zorniak et al., 2010). As early as 1959, Vidt et al. (1959) reported differences in cardiac output, peripheral resistance, and regional blood flow between ether and pentobarbital anesthesia in rats. Cardiac output is 1.5 times greater under ether than under pentobarbital anesthesia, and peripheral resistance is approximately two-thirds higher than that with ether. Compared with pentobarbital, ether increases blood flow in the myocardium, brain, and liver, while decreasing blood flow in the kidneys, splanchnic, and skin regions (Vidt et al., 1959). Ketamine does not affect BP but potentiates it with chloralose and light pentobarbitone anesthesia (Brezenoff, 1973). During stable anesthesia, mean arterial pressure (MAP) has been reported to be reduced from 122 ± 2 to 69 ± 4 mmHg with halothane, to 70 ± 3 mmHg with enflurane, and to 103 ± 7 mmHg with ketamine (Miller et al., 1978). Although halothane decreases BP, it causes fewer electrocardiographic changes and lowers the heart rate compared with conscious or fentanyl anesthetized rats (MacLeod et al., 1983). Results of a study by Seyde and Longnecker (1984) indicated that ketamine most closely approximated the conscious state in normovolemic animals in terms of cardiac output and regional blood flow, whereas isoflurane anesthesia most closely resembled the conscious state after hemorrhage (Seyde and Longnecker, 1984). Secher et al. (1985) emphasized the importance of anesthesia in cardiac arrest experimental studies. Isoflurane increased coronary perfusion pressure during resuscitation, shortened the time to return to spontaneous circulation, and increased arterial BP in the post cardiac arrest period (Secher et al., 1985). These effects on key parameters in cardiac arrest research are important when interpreting the results of animal studies. Pentobarbital anesthesia leads to a decrease in HR and oxygen saturation in rats, whereas inhalation anesthesia leads to the desired HR, standard deviation of the normal-to-normal sinus node-initiated R-R interval (SD of the normal-to-normal sinus node-initiated R-R interval; a parameter of time analyses of HR variability presenting the total variability of the HR and oxygen saturation (Murakami et al., 2010). These results underscore the importance of choosing the appropriate anesthetic agent, which should be consistent with the experiment’s objective. Isoflurane inhalation anesthesia is a suitable candidate for general anesthesia in small animals (Murakami et al., 2010). Albrecht et al. (2014a) reported mild hypotension and tachycardia with isoflurane, whereas medetomidine-midazolam-fentanyl induced marked hypertension and a significant decrease in HR. The smallest changes in BP, HR, and pulse pressure were observed with ketamine/xylazine (Albrecht et al., 2014a). HR, BP, body temperature, duration of anesthetic intervals, and body weight were differentially affected by repeated anesthesia using isoflurane, ketamine/xylazine, or medetomidine-midazolam-fentanyl (Albrecht et al., 2014b). Isoflurane provided reproducible anesthesia and is, therefore, suitable for repeated use, although with reductions in HR and BP over six anesthesia sessions. Therefore, the use of isoflurane in experiments in which these parameters should not be affected is not recommended. The inability to ensure surgical tolerance, decreased sleep time, decreased body weight, and tissue necrosis are significant contraindications to the repeated use of ketamine/xylazine. Only mild changes in BP were observed with repeated anesthesia with medetomidine-midazolam-fentanyl; therefore, it appears to be suitable for serial use (Albrecht et al., 2014b). Arrhythmia scores and the incidence of ventricular fibrillation and tachycardia were significantly reduced in ketamine/xylazine-anesthetized rats compared with the thiopental-anesthetized group (mean arrhythmia score, 2.0 ± 2.1 vs. 3.7 ± 1.2; p < 0.05). Throughout the experiment, the ketamine/xylazine group had a significantly lower HR, while the pressure velocity product was also significantly lower at different time points of the ischemic and reperfusion periods (Gonca, 2015). Misak et al. (2023) also pointed out the difference in BP and HR in isoflurane and zoletil/xylazine anesthesia. Before administration of Acanthopanax senticosus extract, the mean systolic BP (SBP) using isoflurane anesthesia was 108 ± 2.4 mmHg, with a diastolic BP (DBP) of 83.6 ± 3.9 mmHg, and HR of 419 ± 36 beats/min. In zoletil/xylazine anesthesia, the mean SBP, DBP, and HR were 97 ± 4.6 mmHg, 62.5 ± 6 mmHg, and 268 ± 20 beats/min, respectively (Misak et al., 2023). Both anesthesia and surgery acutely affect cardiovascular autonomic function in the perioperative period (Vinik and Ziegler, 2007). Anesthesia reduces BP and HR (Altholtz et al., 2006) and impairs baroreflex responsiveness (Yoshimoto et al., 2011). In contrast, surgical interventions increase BP and HR (Gemes et al., 2009; Charlet et al., 2011). The marked differences in the cardiovascular effects of various anesthetics, the need for caution in the interpretation of experimental results in which cardiovascular measurements are performed while the animals are under general anesthesia. Based on these data and other studies, general anesthesia can alter BP in rats, which can lead to misinterpretation of results from studies using these animals. Questions related to the correct interpretation of the final results arise. For example, is it safe to assume that the BP value was of a certain value at the beginning of the experiments performed with the animals under general anesthesia? Can the initial changes in BP also affect other organ systems that are the subject of the experiment? Can anesthesia significantly affect BP? Which method (invasive vs. non-invasive) or site (e.g., artery) of BP measurement is most suitable? The extent to which BP values differ from “normal” is debatable, and the absence of reference values for comparison is a problem. Therefore, the effect of anesthesia and the measurement site of BP in the anesthetized rat must be considered before the experiment even begins. Because different medications for anesthesia are used in experiments, the present study aimed to evaluate whether there are differences in control (baseline) BP values in rats depending on the measurement site and anesthesia medication. Understanding how anesthetics and measurement sites affect baseline BP values are crucial for strengthening the validity of preclinical studies and supporting their translation into clinical practice. Due to the limited number of extracted mean BP values for some groups, we focused only on the most commonly used medications for anesthesia, where individual anesthetics and their combinations were categorized into broader groups (urethane, ketamine, barbital, and isoflurane), together with conscious rats with invasive BP measurement as a control group, and on the most frequently reported measurement sites (carotid and femoral artery), as detailed in the methodology section. The carotid and femoral arteries were selected because they were the most commonly reported measurement sites in the literature, whereas other sites, e.g., tail or brachial artery were rarely used and thus unsuitable for statistical comparison. We focused on studies published between 2021 and 2024, assuming that modern techniques and validated protocols are more likely to be employed, thereby improving the reliability and comparability of our analysis. Data were underpowered in hypertensive rats; therefore, statistical analysis could not be performed. Databases, search strategy, and data extractionAll available numerical BP values reported between 2021 and 2024 in PubMed, Web of Science, Scopus, ScienceDirect, and Google Scholar were evaluated. A manual search of studies performed by 1 of the authors (P.Š.) using the keyword "BP in rat" was conducted in April 2024, with no language restrictions. Refined search strategies using Boolean operators, MeSH terms, or combinations of more specific keywords (e.g., (“BP” OR BP) AND rat* AND (anesthesia OR anaesthetic) AND (invasive OR arterial OR carotid OR femoral)) were not applied in the current screening. The search relied on a broader single keyword approach to maximize the inclusion of potentially relevant studies. Studies retrieved through gray literature searches or reference chaining were not included. Preferred Reporting Items for Systematic Reviews and Meta-Analyses registration and a formal risk of bias assessment were not performed, which represents a limitation of this study. Due to the considerable variability of published data regarding BP measurement in male rats, the primary emphasis was mainly on the evaluation of differences across individual medications for anesthesia, while also considering the measurement site. Because this review focused exclusively on the statistical analysis of extracted BP data, we did not evaluate the methodological quality of the included studies and the risk of bias. One of the authors (P.Š.) performed the initial data extraction using Word software (Microsoft Corporation, Redmond, WA). More specifically, the extracted data included the following: BP (SBP, DBP, and MAP); author; year of publication; rat strain; number of rats used to measure baseline BP values; animal weight; medications for anesthesia; and BP measurement site. In the next phase of data extraction, the authors (P.Š., V.N., and S.G.) independently verified the data and its suitability for final inclusion in the study (Fig. 1). For the control group (conscious rats with invasively measured BP), BP data were obtained from our previous study (Kapsdorferová et al., 2024).
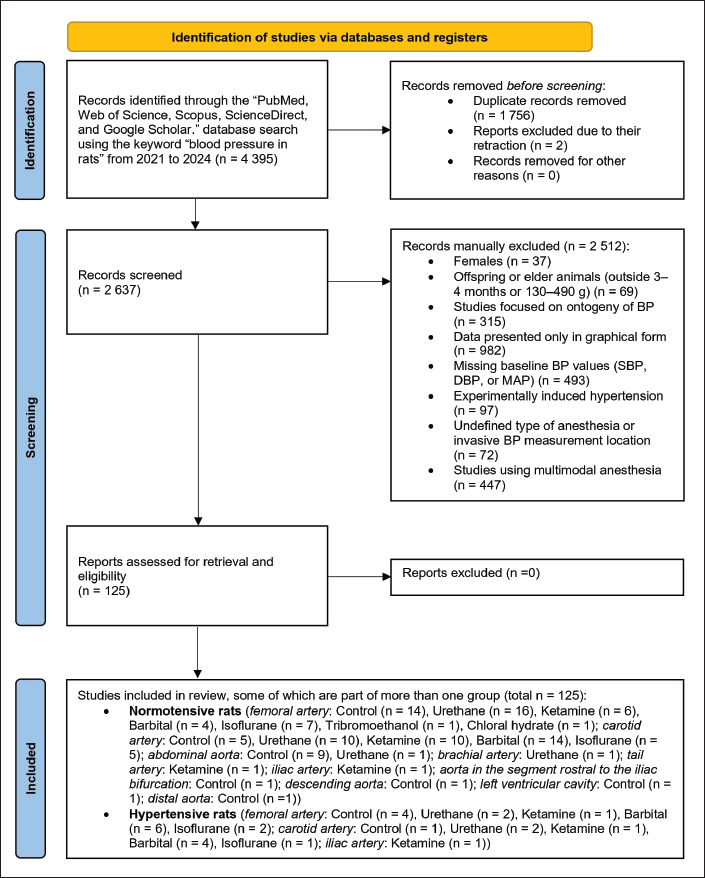
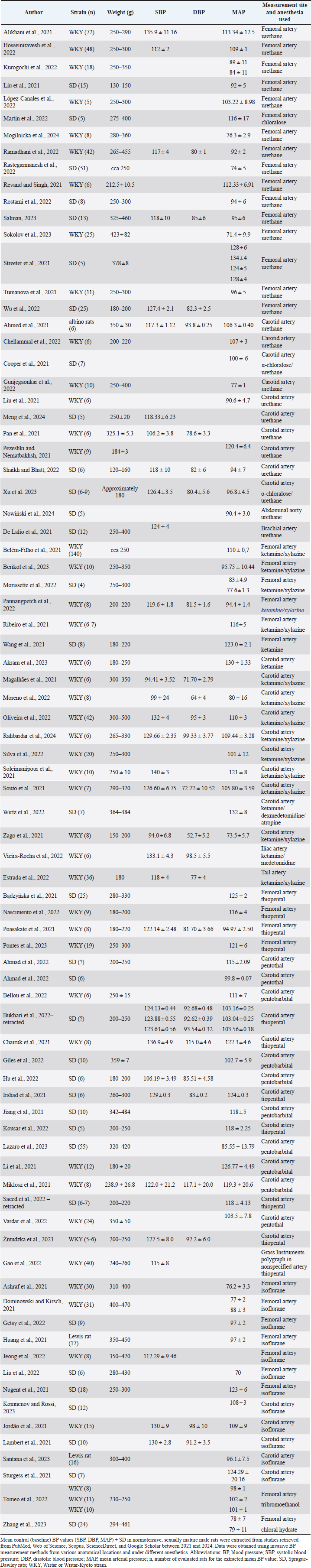
Fig. 1. Flow-diagram according to Page et al., 2021. BP – blood pressure; DBP – diastolic blood pressure; MAP – mean arterial pressure; SBP – systolic blood pressure. Article screening, inclusion, and exclusion criteriaPotentially relevant studies were independently screened by three authors (P.Š., V.N., and S.G.). Data regarding SBP, DBP, and MAP were extracted from studies that fulfilled the following criteria: sexually mature male rats (age 3–4 months, or weight 130–490 g); reported BP status, numerically reported baseline values of BP in normotensive and hypertensive male rats before the experiment itself in general anesthesia; determined medications for anesthesia and BP measurement site. Hypertension was defined as SBP > 140 mmHg, and only baseline or control BP values from experimental studies were analyzed, ensuring that no external interventions influenced the BP values. Therefore, hypertension was not induced, and the hypertensive rats used in the included studies were mostly genetically predisposed strains, such as spontaneously hypertensive rat rats, which exhibit naturally elevated BP. The circadian variation of BP was not analyzed because many experimental studies insufficiently describe or omit light/dark cycle details, limiting the potential for standardized circadian interpretation. Several exclusion criteria were applied to ensure methodological consistency and relevance. To ensure methodological consistency and relevance, we excluded studies outside the 2021–2024 period. This timeframe was chosen because publications from this period are more likely to apply validated BP measurement protocols and modern experimental standards, increasing the reliability and comparability across studies. Studies involving female rats, neonates, aged animals, or those reporting BP data only in graphical form were also excluded. BP ontogeny was not monitored. The exclusion of female rats represents a major limitation of this study. However, including females in this dataset could reduce statistical power and complicate interpretation due to additional variability. This highlights the need for future studies to incorporate more female rats to improve the generalizability and robustness of the findings. Studies employing multimodal anesthesia, i.e., combinations of different anesthetic classes were not analyzed as a separate experimental group, as they may confound comparisons and make it difficult to attribute BP effects to a specific agent. For statistical analysis, anesthetics were therefore categorized into broader groups (urethane, including α-chloralose/urethane; ketamine, including its commonly used combinations such as ketamine/xylazine; barbital derivatives; and isoflurane) to ensure sufficient sample sizes and comparability across studies. Although this grouping strategy allowed us to capture the most frequently used anesthetics, it represents a limitation because combining single agents with their common adjuncts (e.g., ketamine with xylazine) may obscure some drug-specific effects. Finally, inconsistent reporting of animal age and weight further limited inclusion, as this affected interpretability given the age-dependent nature of cardiovascular values. Statistical analysisThis study used study-level mean BP values because individual animal-level data were not consistently reported across the included studies. Two-way analysis of variance was then applied to assess the influence of two independent factors (medications used for anesthesia and invasive measurement site) on BP values and whether there is an interaction between them (e.g., whether a certain medication for anesthesia has a different effect on BP depending on where BP was measured). Subsequently, Sidak s post hoc test was used to compare individual pairs of average values to determine how the average BP values for a particular anesthetic or control group differ between the femoral and carotid arteries. The column factor represents the location of BP measurement, whereas the row factor represents the specific medication for anesthesia. The mean of each cell was compared with the mean of the other cells in that row. For incomplete repeated-measures data (i.e., missing BP values in specific anesthetic–artery combinations), a mixed-effects model with restricted maximum likelihood estimation was used. In this model, matched subjects were treated as a random effect to account for repeated measurements. This approach allowed the handling of missing values without discarding the affected datasets while preserving the overall experimental design. Formal power calculations and tests for normality and homoscedasticity were not feasible due to heterogeneous study designs and varying numbers of extracted values per study. Thus, the reported results should be interpreted in this context. Differences with p < 0.05 were considered statistically significant. Data analysis and plot construction were performed by one author (V.N.) using Prism version 8 (GraphPad Inc., San Diego, CA). Study selectionFrom the initial number of studies retrieved (n=4,395), 1,756 duplicates and two retracted studies were removed, and no records were excluded for other reasons. Based on the exclusion criteria, 2,512 studies were subsequently excluded. The remaining studies (n=125) were screened and evaluated for suitability, with some belonging to more than one group (see Fig. 1 for details). For the control group, the mean BP values were extracted from studies with invasive BP measurements in conscious normotensive or hypertensive rats. In the case of invasive BP measurement under anesthesia, mean BP values were extracted from studies using the following anesthetics: urethane–urethane anesthetics or α-chloralose/urethane combination; ketamine—anesthetics ketamine or a combination of ketamine/xylazine, ketamine/dexmedetomidine/atropine, ketamine/medetomidine; barbital—anesthetics pentobarbital, thiopental, thiobutabarbital, pentothal; isoflurane—anesthetic isoflurane; tribromoethanol—anesthetic tribromoethanol; chloral hydrate—anesthetic chloral hydrate. In the case of normotensive rats, baseline BP values obtained by invasive measurement from conscious animals were included in the study, specifically from the femoral artery (Barretto-de-Souza et al., 2021; Cruz et al., 2021; Santos et al., 2021; Flahault et al., 2021; Kirillov et al., 2021; Sedighi et al., 2021; Sharma et al., 2021; dos Santos et al., 2022; Fioretti et al., 2022; Lopes et al., 2022; Luz et al., 2022; Oliveira et al., 2022a; Gomes et al., 2023a; Toczek et al., 2023), carotid artery (Zicha et al., 2021; Komnenov and Rossi, 2023; Olatoye et al., 2023; Raji et al., 2023; Wu et al., 2023), abdominal aorta (Ayaz et al., 2021; Barrera et al., 2021; Costa-Ferreira et al., 2021; Oliveira et al., 2021; Potter et al., 2021; Silva et al., 2021; Abdulsalam et al., 2023; Gomes et al., 2023b; Mohamed et al., 2024), and by (radio)telemetry in the aorta in the segment rostral to the iliac bifurcation (Zhang et al., 2023a), and in the descending aorta (Griffiths et al., 2021), left ventricular cavity (Selejan et al., 2022), and distal aorta (Das et al., 2022). Furthermore, in normotensive rats, BP data were obtained invasively measured under anesthesia, specifically under urethane anesthesia, from the femoral artery (Alikhani et al., 2021; Liu et al., 2021a; Revand and Singh, 2021; Streeter et al., 2021; Tumanova et al., 2021; Hosseiniravesh et al., 2022; Kurogochi et al., 2022; López-Canales et al., 2022; Martin et al., 2022; Rostami et al., 2022; Wu et al., 2022; Ramadhani et al., 2023; Rastegarmanesh et al., 2023; Salman, 2023; Sokolov et al., 2023; Mogilnicka et al., 2024), carotid artery (Ahmed, 2021; Cooper et al., 2021; Liu et al., 2021b; Pan et al., 2021; Pezeshki and Nematbakhsh, 2021; Chellammal et al., 2022; Gunjegaonkar et al., 2022; Shaikh and Bhatt, 2022; Xu et al., 2023; Meng et al., 2024), abdominal aorta (Nowiński et al., 2024), and brachial artery (De Lalio and Stocker, 2021). BP values were obtained from the femoral artery in normotensive rats under ketamine anesthesia (Belém-Filho et al., 2021; Ribeiro et al., 2021; Wang et al., 2021; Morissette et al., 2022; Pannangpetch et al., 2022; Berikol et al., 2023), carotid artery (Magalhães et al., 2021; Soleimanipour et al., 2021; Souto et al., 2021; Zago et al., 2021; Moreno et al., 2022; Oliveira et al., 2022b; Silva et al., 2022; Wirtz et al., 2022; Akram et al., 2023; Rahbardar et al., 2024), as well as from the iliac artery (Vieira-Rocha et al., 2022) and tail artery (Estrada et al., 2022). Under barbiturate anesthesia, BP values were measured from the femoral artery (Bądzyńska et al., 2021; Poasakate et al., 2021; Nascimento et al., 2022; Pontes et al., 2023), carotid (Chairuk et al., 2021; Irshad et al., 2021; Jiang et al., 2021; Li et al., 2021; Miklosz et al., 2021; Ahmad et al., 2022a ; Ahmad et al., 2022b; Bellou et al., 2022; Giles et al., 2022; Hu et al., 2022; Kousar et al., 2022; Vardar et al., 2022; Lazaro et al., 2023; Żmudzka et al., 2023), and polygraph (Grass Instruments) in a nonspecified artery (Gao et al., 2022). In isoflurane anesthesia, also from the femoral artery (Ashraf et al., 2021; Dominowski et al., 2021; Huang et al., 2021; Nugent et al., 2021; Getsy et al., 2022; Jeong et al., 2022; Liu et al., 2022) and carotid artery (Jordão et al., 2021; Lambert et al., 2021; Sturgess et al., 2021; Komnenov and Rossi, 2023; Santana et al., 2023). BP data for tribromoethanol (Tomeo et al., 2022) and chloral hydrate (Zhang et al., 2023b) anesthesia were also extracted in the case of the femoral artery in normotensive rats. In hypertensive rats, control BP values were obtained by invasive measurement from conscious animals from the femoral artery (Bandoni et al., 2021; Gardim et al., 2021; Marc et al., 2021; Toczek et al., 2023), carotid artery (Oleksa et al., 2021), and (radio) telemetry from the abdominal aorta (Geraldes et al., 2023; Fan et al., 2024; Sun et al., 2024), descending aorta (Griffiths et al., 2021), and in the left ventricular cavity (Selejan et al., 2022). In the case of urethane, these measurements were performed in the femoral artery (Melo et al., 2022; Mogilnicka et al., 2024) and carotid artery (Liu et al., 2021b; Pan et al., 2021). Under ketamine anesthesia in hypertensive rats, BP values from the femoral artery (Ribeiro et al., 2021), carotid artery (Moreno et al., 2022), and iliac artery (Vieira-Rocha et al., 2022) were measured. In the case of barbiturate anesthesia, from the femoral artery (Bądzyńska et al., 2021; Salman et al., 2022; Yen et al., 2022; Salman et al., 2023; De Souza et al., 2023; Honetschlägerova et al., 2024) and carotid artery (Li et al., 2021; Wang and Zhang, 2021; Ahmad et al., 2022a; Kousar et al., 2022). In the case of isoflurane, the measurements were performed in the femoral artery (Liu et al., 2022; Mensah et al., 2022) and carotid artery (Sainsily et al., 2021). In both normotensive and hypertensive rats, urethane, ketamine, barbital, and isoflurane were the most commonly used anesthetics. Femoral and carotid arteries were the most common invasive BP measurement sites. Details on extracted mean BP values included in the statistical analysis, as well as information on less commonly used anesthetic agents or measurement sites, strain, body weight, and number of animals per study, are provided in Supplementary Files 1 and 2. Effect of anesthesia and measurement site on baseline BP in normotensive ratsDue to the limited number of extracted mean BP values for some groups, the statistical analysis in normotensive rats was restricted to the most frequently reported medications for anesthesia. The individual agents and their combinations were categorized into four groups: urethane, ketamine, barbital, and isoflurane. Conscious animals served as the control group. Invasive BP measurements were performed in the femoral or carotid artery (Table 1; Fig. 2).
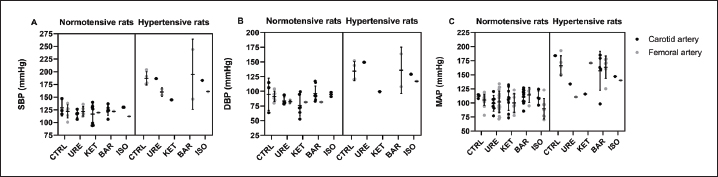
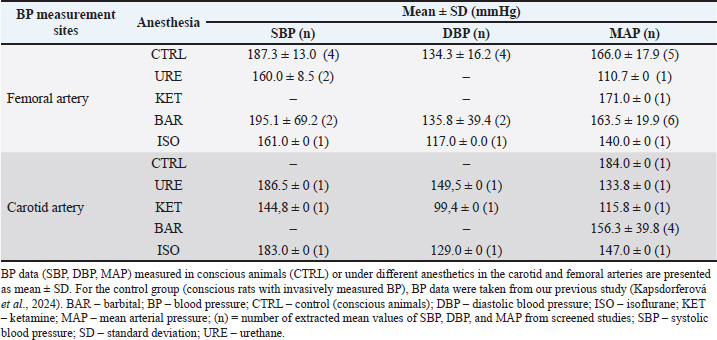
Fig. 2. BP differences between the most frequently reported medications for anesthesia and measurement sites in normotensive and hypertensive male rats. Differences in (A) SBP, (B) DBP, and (C) MAP are presented as individual data points, representing the mean BP values extracted from studies (black points – BP measured in the carotid artery; gray points – BP measured in the femoral artery), with a straight line representing the mean of the individual group ± SD. In each graph, data for normotensive rats are shown on the left, and data for hypertensive rats are shown on the right, separated by a vertical line. Some extracted values are descriptive (n=1), based on a small number of studies, as indicated in Table 1. For the control group (conscious rats with invasively measured BP), BP data were taken from our previous study (Kapsdorferová et al., 2024). BAR – barbital; BP – blood pressure; DBP – diastolic blood pressure; CTRL – control (conscious animals); ISO – isoflurane; KET – ketamine; MAP – mean arterial pressure; SBP – systolic blood pressure; SD – standard deviation; URE – urethane. Table 1. Control (baseline) BP values in normotensive male rats with the most common medications for anesthesia and BP measurement sites1.
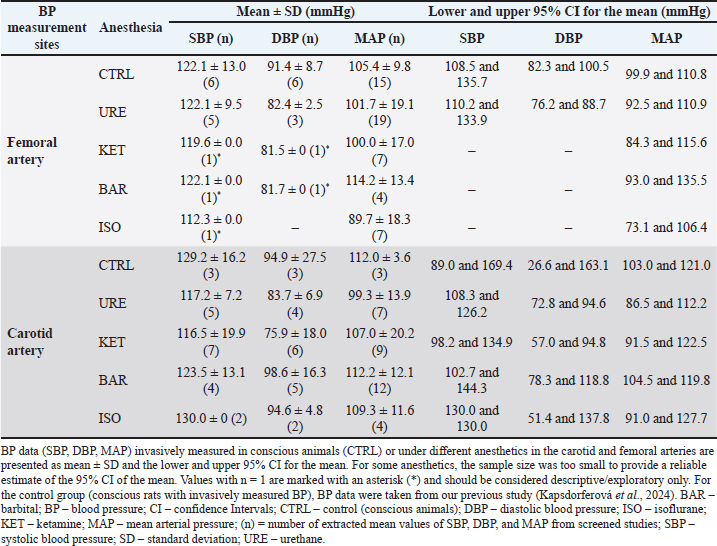
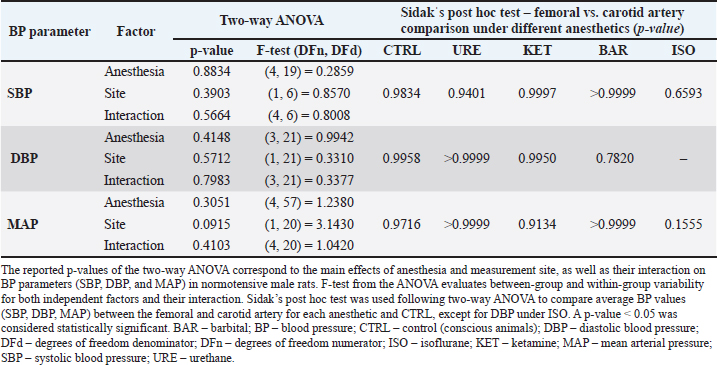
Mean SBP values ranged from 112.3 to 130 mmHg, depending on the measurement site and anesthesia used. Femoral SBP was relatively consistent among control animals and rats under urethane, ketamine, and barbital, whereas isoflurane caused a slight decrease. However, ketamine, barbital, and isoflurane were less frequently represented in femoral artery studies (n=1 for ketamine and barbital, n=0 for isoflurane). In contrast, carotid SBP showed greater variability than femoral measurements, being relatively stable in conscious rats and those under isoflurane or barbital and slightly reduced under ketamine. The DBP varied from 81.5 to 98.6 mmHg. In the femoral artery, DBP decreased in all anesthetic groups except isoflurane, for which no femoral measurements were available. In contrast, DBP was generally more variable in the carotid artery than in the femoral artery. Conscious rats and rats treated with isoflurane showed relatively stable carotid DBP, whereas barbital slightly increased it, and urethane and ketamine caused a slight decrease. These observations should be interpreted cautiously due to the uneven distribution of data across groups and the small number of extracted means [e.g., ketamine (n=1), barbital (n=1), and isoflurane (n=0)] in the femoral artery. The MAP values also exhibited variability, ranging from 89.7 to 114.2 mmHg. In the femoral artery, most anesthetics decreased MAP, with the largest reduction observed for isoflurane, whereas barbital increased MAP compared to conscious rats. A similar trend was observed in the carotid artery, where MAP was more consistent in the barbital and conscious animals, and the reduction under isoflurane was less pronounced than in the femoral artery. Despite these observations, no significant effects were found (Table 2). These findings suggest that the choice among these anesthetics has only minor effects on baseline BP in normotensive rats. However, this should be interpreted with caution, given the limitations discussed below. Table 2. Results of the statistical analysis in normotensive male rats1.
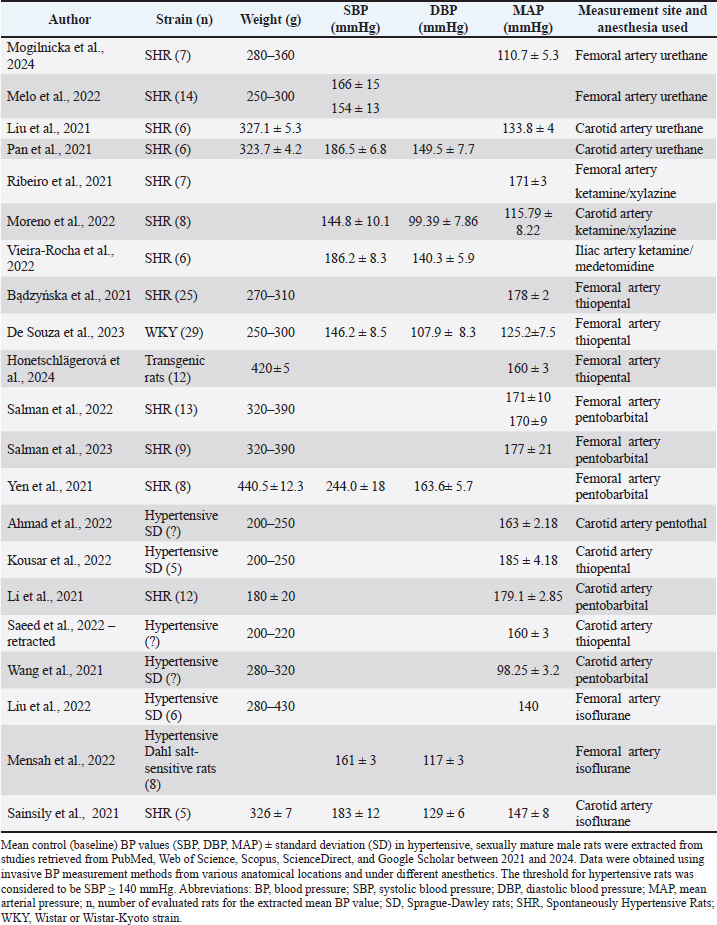
Effect of anesthesia and measurement site on baseline BP in hypertensive ratsNo statistical comparison could be performed in hypertensive rats due to the insufficient number of extracted BP data and the uneven distribution of data between groups. However, the average BP values were extracted (Fig. 2; Table 3). Table 3. Control (baseline) BP values in hypertensive male rats with the most common medications for anesthesia and invasive measurement sites1.
Hypertensive rats showed higher BP values compared with normotensive animals. The SBP ranged from 144.8 to 195.1 mmHg, depending on the anesthetic and measurement site. The femoral SBP was relatively high in control animals and under the barbital. Under urethane and isoflurane, the SBP was slightly lower. Carotid SBP was reported in only a few studies (urethane, ketamine, isoflurane; n=1 each) and varied from 144.8 to 186.5 mmHg. The DBP ranged from 99.4 to 149.5 mmHg. In the femoral artery, barbital showed slightly higher variability. DBP under isoflurane was slightly lower compared with conscious animals. Carotid DBP data were sparse, indicating high variability and a limited sample size. The MAP values varied from 110.7 to 184.0 mmHg. Femoral MAP decreased under all medications for anesthesia compared with conscious animals. Carotid MAP values have been reported in very few studies. However, these results showed a trend similar to that of the femoral artery. Overall, these observations suggest that hypertensive rats exhibit higher baseline BP than normotensive animals. Considerable variability exists across medications for anesthesia and measurement sites. No firm conclusions can be drawn due to the limited and uneven data. The extracted values provide a descriptive overview of the reported trends. DisscussionBased on the statistical comparison, it can be hypothesized that the choice of the most commonly used anesthetic is not a consistent factor for changes in control (baseline) BP values when focusing on the two most common invasive BP measurement sites—the femoral and carotid arteries. Our descriptive data also suggest that both femoral and carotid access may provide comparable BP measurements under the most commonly reported anesthetics. However, our analysis highlights several reporting gaps that limit reproducibility and interpretation, including limited information on the circadian timing of BP measurements, the sex of animals, or detailed anesthetic protocols. Future studies addressing these gaps are essential to improve the comparability and reliability of BP measurements across laboratories. Additionally, the small sample sizes for some anesthetics and the uneven distribution of data warrant caution when generalizing these findings. Nevertheless, based on the extracted mean BP values, we expect a slight initial decrease in control (baseline) BP at the onset of the experiment under most medications for anesthesia compared to conscious rats. A decrease in MAP following anesthesia administration has also been reported by Bencze et al. (2013), supporting the trend observed in our extracted data. In Wistar and SHR rats, the administration of pentobarbital, isoflurane, or chloralose-urethane led to a reduction in baseline MAP compared to conscious animals in nearly all cases, except for pentobarbital in SHR rats. Pentobarbital was identified as the anesthetic with the least impact on BP. In contrast, isoflurane and chloralose-urethane can be recommended only when changes in BP, renin-angiotensin system activity, sympathetic nervous system activity, or nitric oxide (NO) do not influence the monitored parameters. Moreover, isoflurane activates K+ channels and induces membrane hyperpolarization, which may distort the experimental outcomes. The ketamine-xylazine combination is not suitable for functional cardiovascular studies. Therefore, the choice of an anesthetic should consider its specific cardiovascular effects to avoid misinterpretation of the results (Bencze et al., 2013). Notably, the authors did not account for sex differences, circadian variability of the measured parameters, or the potential influence of different BP measurement sites. Sano et al. (2016) highlighted the absence of baseline data on hemodynamics, cardiac function, and glucose and lipid metabolism in conscious rats. Obtaining such data from conscious animals is not always ethically acceptable and may be prone to methodological errors, which complicates the accurate assessment of anesthetic effects. Based on their findings, pentobarbital appears to have the least impact on metabolic parameters and is recommended for studies focused on metabolic outcomes. The ketamine/xylazine combination exhibited a pronounced bradycardic effect, suggesting its suitability for Doppler-based assessment of left ventricular diastolic function. The relatively easy control of anesthetic depth makes isoflurane a suitable option for experiments that require manipulation over extended periods of data collection (Sano et al., 2016). The biological mechanisms underlying the decrease in BP under general anesthesia remain a subject of discussion. According to Bencze et al. (2013), the decline in BP observed in both normotensive and hypertensive rats is largely attributable to reduced sympathetic nervous system activity in maintaining BP, with the exception of pentobarbital, which induces smaller hemodynamic changes compared with other anesthetics (Bencze et al., 2013). A pronounced BP reduction in hypertensive rats receiving ketamine–xylazine anesthesia has also been linked to marked decreases in HR and cardiac output (Saha et al., 2007). Another important mechanism involves vasodilation and a consequent reduction in venous return, which can contribute to hypotension. For example, propofol alters the balance between the vasoconstrictor thromboxane A2 (TxA2) and the vasodilator NO, leading to vasodilation and a decrease in BP. Beyond vasoactive mediators, hypotension may also result from complex neurohumoral interactions (Tsikas et al., 2015). Saugel et al. (2022) further suggested that hypotension primarily reflects arterial dilation with reduced systemic vascular resistance rather than venous dilation or impaired myocardial contractility. In addition, some anesthetics not only suppress sympathetic activity but also attenuate baroreflex sensitivity, thereby reducing the organism’s capacity to compensate for the decreased BP (Bencze et al., 2013; Tsikas et al., 2015). Anesthetic agents may also modulate the renin–angiotensin system, further contributing to BP reduction (Bencze et al., 2013). Finally, some anesthetics, such as isoflurane and propofol, directly decrease myocardial contractility, resulting in reduced cardiac output and subsequent BP decline (Meng et al., 2019). Taken together, these findings suggest that the anesthetic-induced hypotension is multifactorial, involving neural, vascular, humoral, and cardiac mechanisms, and depends on the specific anesthetic agent used. In our review, it is not possible to reliably recommend specific anesthetics as suitable for cardiovascular studies in terms of the invasive BP measurement site due to non-significant results, limited data in certain groups, and methodological inconsistencies across the included studies. However, in the carotid artery of normotensive rats, the mean SBP values were slightly, but nonsignificantly, lower in almost all cases with the most commonly used anesthetics (urethane, ketamine, and barbital) compared with conscious rats, except for isoflurane. In the femoral artery of normotensive rats, the mean SBP values were slightly lower only under isoflurane. A similar decrease in the mean SBP values was observed in the femoral artery of hypertensive rats under urethane and isoflurane, whereas barbital increased mean SBP values. If the SBP results from the contraction force of the left ventricle, which primarily depends on end-diastolic filling (preload), then, hypothetically, the contraction force may decrease in rats under general anesthesia. The question arises whether these changes are significant enough to affect other monitored organ functions that are influenced by or primarily dependent on SBP. Because MAP is the main determinant of organ perfusion pressure, even a slight reduction in MAP may theoretically increase the risk of inadequate tissue perfusion. When MAP is not adequately maintained, vital organs may not receive sufficient blood supply, potentially leading to hypotensive shock and organ failure. Therefore, apart from barbital anesthesia, other anesthetics could create conditions associated with a higher cardiovascular risk in normotensive rats. However, further investigation is required to determine whether these changes have significant functional consequences. The decrease in mean DBP values in normotensive rats except under barbital and isoflurane may, in turn, indicate reduced peripheral resistance. However, in hypertensive rats, the lack of sufficient data makes it difficult to draw any reliable comparisons. When assessing the effects of individual medications for anesthesia on BP measured in the femoral or carotid arteries, the majority of the extracted numerical data referred to MAP. The greatest variability in BP values among normotensive rats was observed under ketamine anesthesia. Substantial variability in BP values was noted in hypertensive rats under barbital anesthesia. In general, if we compare the SBP from the femoral artery in normotensive rats with that in conscious rats, there are minimal differences in the average values, in contrast to the more variable average values of SBP measured in the carotid artery. The average DBP values of normotensive rats measured in the femoral artery are slightly lower compared to non-anesthetized rats, except under isoflurane. In contrast, DBP values obtained from the carotid artery, as well as MAP values measured in both the carotid and femoral arteries of normotensive rats, show fluctuations depending on the medications for anesthesia. Unfortunately, no high-quality reference BP ranges are available; as such, assessing whether the baseline BP values from the screened studies are in the physiological ranges at the beginning of the experiment(s) is problematic. The most significant limitation of this study is the small number of extracted average values in some groups, the uneven number between groups, and the variability of the obtained data, which can potentially weaken statistical power. One possible reason for the variability may be the reliability of the BP measurements. Invasive BP measurements typically favor MAP as the most reliable parameter compared with the noninvasive tail-cuff method, which favors SBP (Kapsdorferová et al., 2024). This may explain why the extracted mean values of MAP are more numerous in our study focused on invasive methods than in the case of SBP and DBP. Another important factor is the influence of the light/dark cycle. As shown in our previous work, the precise specification of the timing of the experimental intervention is often neglected in study methodologies, which makes it difficult—sometimes impossible—to interpret possible circadian variability in the observed parameters (Kapsdorferová et al., 2024). However, we assume that the BP values screened in our study were predominantly measured during the light period of the day, which corresponds to the inactive period of the rat regimen day. It is well established that several cardiovascular parameters exhibit approximately 24-hour oscillations, including BP and HR (van den Buuse, 1999). Nevertheless, the interaction between anesthesia and circadian rhythmicity represents an often overlooked but crucial aspect in experimental design. Without considering the underlying circadian variation in the autonomic nervous system, the extent of this effect cannot be fully understood. Studies in rats have demonstrated that the autonomic nervous system exhibits circadian rhythmicity, modulating cardiovascular parameters throughout the light/dark cycle (Makino et al., 1997). Since anesthesia reduces sympathetic activity (Bencze et al., 2013), its impact on BP is likely to differ depending on whether the intervention occurs during the active or inactive period of the light/dark cycle. Therefore, accurate reporting of experimental timing and light/dark conditions is essential for reproducibility and correct interpretation of the findings. The potential impact of anesthesia on animal welfare warrants discussion. Anesthesia is essential for adherence to the 3Rs principles by minimizing stress and pain during experimental procedures. However, it can also affect multiple physiological parameters, including HR and BP (Orts-Sebastian et al., 2019). Our results, together with previous experiments (Bencze et al., 2013), highlight the hypotensive effects of most anesthetics, which may influence both the validity of experimental outcomes and animal welfare. These considerations emphasize the dual role of anesthesia in supporting ethical standards while introducing potential confounding factors. Relatively large BP variances may also depend on other factors that can influence cardiovascular parameters. These include breeding conditions, sex (Maris et al., 2005), age (Chaves et al., 2003), animal handling prior to the experiment or stress itself (Layton et al., 2023), strain (Konopelski and Ufnal, 2016), body weight (Mutiso et al., 2014), anesthetic administration method (intraperitoneal, intravenous, subcutaneous, or intramuscular), type of anesthetic, depth of anesthesia, body temperature (Redfors et al., 2014), and many others. The reason for this decision is the lack of detailed information on some of these factors, which could lead to inaccurate or incomplete conclusions. Moreover, the inclusion of these factors would further complicate the analysis, especially given the limitations of our study, such as the small number of extracted data in some groups and their uneven distribution between the groups. Therefore, all the aforementioned limitations should be addressed in future experimental research. ConclusionOur synthesis did not detect consistent, significant differences between the most commonly used anesthetic agents—urethane, ketamine, barbital, and isoflurane—when measured invasively via the femoral or carotid arteries. However, we hypothesize a slight initial decrease in baseline (control) BP at the onset of the experiment under most of these anesthetic agents compared to conscious animals. This observation, along with other limitations—such as sex, circadian variability, age, body weight, anesthetic type and depth, and others—warrants further investigation, particularly concerning the invasive site of BP measurement. AcknowledgmentsNot applicable. Conflict of interestThe authors declare no conflict of interest. FundingThis study received no specific grant. Author contributionsConceptualization: P.Š., S.G.; Study search: P.Š.; Study screening: P.Š., V.N., S.G.; Data extraction: P.Š.; Data verification: P.Š., V.N., S.G.; Data analysis and plot construction: V.N.; Manuscript writing: P.Š., V.N.; Manuscript review: P.Š., V.N., S.G.; Study supervision: P.Š. All authors have approved the final version of the manuscript. Data availabilitySupplementary Files 1 and 2 provide details on the extracted mean BP values included in the statistical analysis, as well as information on less commonly used anesthetic agents or measurement sites, strain, body weight, and number of animals per study. For the control group (conscious rats with invasively measured BP), BP data are available in the following published article and its supplementary materials: Kapsdorferová, V., Grešová, S. and Švorc, P. 2024. Measurement of blood pressure in rats: Invasive or noninvasive methods? Physiol. Rep. 12, e70041. ReferencesAhmad, T., Javed, A., Khan, T., Althobaiti, Y.S., Ullah, A., Almutairi, F.M. and Shah, A.J. 2022b. Investigation into the antihypertensive effects of diosmetin and its underlying vascular mechanisms using rat model. Pharmaceuticals 15, 951. Ahmad, T., Qayyum, R., Khan, T., Mahnashi, M.H., Jalal, M.M., Altayar, M.A., Alshehri, O.M. and Shah, A.J. 2022a. Vasorelaxant and antihypertensive effects of bergenin on isolated rat aorta and high salt-induced hypertensive rats. Evid. Based Complement. Alternat. Med. 2022(1), 4886193. Ahmed, F. 2021. Ficus benghalensis bark extract shows blood pressure lowering effect in normotensive and angiotensin II-induced hypertensive rats. Pharmacophore 12(7), 7–10. Akram, A., Jamshed, A., Anwaar, M., Rasheed, H.M.F., Haider, S.I., Aslam, N. and Jabeen, Q. 2023. Evaluation of Caralluma edulis for its potential against obesity, atherosclerosis and hypertension. Dose-Response 21(1), 15593258231152112. Albrecht, M., Henke, J., Tacke, S., Markert, M. and Guth, B. 2014a. Effects of isoflurane, ketamine-xylazine and a combination of medetomidine, midazolam and fentanyl on physiological variables continuously measured by telemetry in Wistar rats. BMC. Vet. Res. 10, 198. Albrecht, M., Henke, J., Tacke, S., Markert, M. and Guth, B. 2014b. Influence of repeated anaesthesia on physiological parameters in male Wistar rats: a telemetric study about isoflurane, ketamine-xylazine and a combination of medetomidine, midazolam and fentanyl. BMC Vet. Res. 10, 310. Alikhani, V., Mohebbati, R., Hosseini, M., Khajavirad, A. and Shafei, M.N. 2021. Role of the glutamatergic system of ventrolateral periaqueductal gray (vlPAG) in the cardiovascular responses in normal and hemorrhagic conditions in rats. Iran. J. Basic Med. Sci. 24, 586–594. Altholtz, L.Y., Fowler, K.A., Badura, L.L. and Kovacs, M.S. 2006. Comparison of the stress response in rats to repeated isoflurane or CO2: o2 anesthesia used for restraint during serial blood collection via the jugular vein. J. Am. Assoc. Lab. Anim. Sci. 45, 17–22. Ashraf, O., Huynh, T., Purnell, B.S., Murugan, M., Fedele, D.E., Chitravanshi, V. and Boison, D. 2021. Suppression of phrenic nerve activity as a potential predictor of imminent sudden unexpected death in epilepsy (SUDEP). Neuropharmacology 184, 108405. Bądzyńska, B., Vaneckova, I., Sadowski, J., Hojná, S. and Kompanowska-Jezierska, E. 2021. Effects of systemic and renal intramedullary endothelin-1 receptor blockade on tissue NO and intrarenal hemodynamics in normotensive and hypertensive rats. Eur. J. Pharmacol. 910, 174445. Bandoni, R.L., Bricher Choque, P.N., Dellê, H., De Moraes, T.L., Porter, M.H.M., Da Silva, B.D., Neves, G.A., Irigoyen, M.C., De Angelis, K., Pavlov, V.A., Ulloa, L. and Consolim-Colombo, F.M. 2021. Cholinergic stimulation with pyridostigmine modulates a heart-spleen axis after acute myocardial infarction in spontaneous hypertensive rats. Sci. Rep. 11(1), 9563. Barrera, A., Morales-Loredo, H., Garcia, J.M., Fregoso, G., Pace, C.E., Mendiola, P.J., Naik, J.S., Gonzalez Bosc, L.V. and Kanagy, N.L. 2021. Simulated sleep apnea alters hydrogen sulfide regulation of blood flow and pressure. Am. J. Physiol. Heart. Circ. Physiol. 320, H511–H519. Barretto-De-Souza, L., Benini, R., Reis-Silva, L.L. and Crestani, C.C. 2021. Corticotropin-releasing factor neurotransmission in the lateral hypothalamus modulates the tachycardiac response during acute emotional stress in rats. Brain. Res. Bull. 166, 102–109. Belém-Filho, I.J.A., Brasil, T.F.S., Fortaleza, E.A.T., Antunes-Rodrigues, J. and Corrêa, F.M.A. 2021. A functional selective effect of oxytocin secreted under restraint stress in rats. Eur. J. Pharmacol. 904, 174182. Bellou, A., Sennoun, N., Aburawi, E.H., Jayaraj, R.L., Alper, S.L., Alfaki, I.A., Yasin, J., Sekar, S., Shafiuallah, M., Al-Salam, S., Nemmar, A., Kazzam, E., Mertes, P.M. and Al-Hammadi, S. 2022. Combined treatment with KV channel inhibitor 4-aminopyridine and either γ-cystathionine lyase inhibitor β-cyanoalanine or epinephrine restores blood pressure, and improves survival in the Wistar rat model of anaphylactic shock. Biology 11(10), 1455. Bencze, M., Behuliak, M. and Zicha, J. 2013. The impact of four different classes of anesthetics on the mechanisms of blood pressure regulation in normotensive and spontaneously hypertensive rats. Physiol. Res. 62(5), 471–478. Bozdereli Berikol, G., Berikol, G., Ayrik, C., Kose, A., Bozkurt Babus, S., Tamer Gumus, L., Bozdogan Arpaci, R., Dag, A., Ayan, E. and Gorur, A. 2023. Antioxidant and neuroprotective effects of L-arginine administration after traumatic brain injury and hemorrhagic shock in rats. Turk. Neurosurg. 33, 379–385. Brezenoff, H.E. 1973. Cardiovascular responses to noradrenaline in the rat before and after administration of various anaesthetics. Br. J. Pharmacol. 49, 565–572. Buuse, V.D. 1999. Circadian rhythms of blood pressure and heart rate in conscious rats: effects of light cycle shift and timed feeding. Physiol. Behav. 68(9), 10–15. Chairuk, P., Zaman, R.U., Naphatthalung, J. and Jansakul, C. 2021. Effect of consumption of whole egg and egg fractions on cardiovascular disease factors in adult rats. J. Sci. Food. Agric. 101(9), 3942–3951. Charlet, A., Rodeau, J.L. and Poisbeau, P. 2011. Radiotelemetric and symptomatic evaluation of pain in the rat after laparotomy: long-term benefits of perioperative ropivacaine care. J. Pain 12, 246–256. Chaves, A.A., Dech, S.J., Nakayama, T., Hamlin, R.L., Bauer, J.A. and Carnes, C.A. 2003. Age and anesthetic effects on murine electrocardiography. Life. Sci. 72(21), 2401–2412. Chellammal, H.S.J., Hasan, M.H., Kshirsagar, R.P., Musukula, V.K.R., Ramachandran, D. and Diwan, P.V. 2022. Metformin inhibits cardiometabolic syndrome associated cognitive deficits in high fat diet rats. J. Diabetes. Metab. Disord. 21, 1415–1426. Cooper, I.R., Liu, S. and Delorey, D.S. 2021. Effects of sex and exercise training on β-adrenoreceptor-mediated opposition of evoked sympathetic vasoconstriction in resting and contracting muscle of rats. J. Appl. Physiol. 130, 114–123. Costa-Ferreira, W., Gomes-De-Souza, L. and Crestani, C.C. 2021. Role of angiotensin receptors in the medial amygdaloid nucleus in autonomic, baroreflex and cardiovascular changes evoked by chronic stress in rats. Eur. J. Neurosci. 53, 763–777. Cruz, P.L., Moraes-Silva, I.C., Ribeiro, A.A., Machi, J.F., De Melo, M.D.T., Dos Santos, F., Da Silva, M.B., Strunz, C.M.C., Caldini, E.G. and Irigoyen, M.C. 2021. Nicotinamide attenuates streptozotocin-induced diabetes complications and increases survival rate in rats: role of autonomic nervous system. BMC. Endocr. Disord. 21, 133. Das, S., Komnenov, D., Newhouse, L., Rishi, A.K. and Rossi, N.F. 2022. Paraventricular nucleus V1a receptor knockdown blunts neurocardiovascular responses to acute stress in male rats after chronic mild unpredictable stress. Physiol. Behav. 253, 113867. De Moura Magalhães, B.A.B., Rodrigues, L.F., De Oliveira, T.F., Vassallo, D.V. and Simões, M.R. 2021. Lead and mercury 28 day exposure at small concentrations reduces smooth muscle relaxation by decreasing cGMP. Toxicol. Appl. Pharmacol. 413, 115405. De Souza, F.D., Fidale, T.M., Pereira, T.C.R., Mantovani, M.M., Deconte, S.R., Moreira-Silva, D., De Moura, F.B.R., Martins, L.D.Q., Alex Dos Santos, L., Medeiros, R.D.S., Neto, M.L.F. and Resende, E.S. 2023. Effects of hyperprotein diet on anxiety, haemodynamics and morphofunctional aspects of the heart of Wistar rats. Exp. Physiol. 108(6), 818–826. De Wildt, D.J., Hillen, F.C., Rauws, A.G. and Sangster, B. 1983. Etomidate-anaesthesia, with and without fentanyl, compared with urethane-anaesthesia in the rat. Br. J. Pharmacol. 79, 461–469. DeLalio, L.J. and Stocker, S.D. 2021. Impact of anesthesia, sex, and circadian cycle on renal afferent nerve sensitivity. Am. J. Physiol. Heart. Circ. Physiol. 320, H117–H132. Dominowski, L. and Kirsch, M. 2021. Synergistic effect of β-alanine and aprotinin on mesenteric ischemia. J. Surg. Res. 263, 78–88. Donertas Ayaz, B., Oliveira, A.C., Malphurs, W.L., Redler, T., De Araujo, A.M., Sharma, R.K., Sirmagul, B. and Zubcevic, J. 2021. Central administration of hydrogen sulfide donor NaHS reduces Iba1-positive cells in the PVN and attenuates rodent angiotensin II hypertension. Front. Neurosci. 15, 690919. Dos Santos, R.B., De C. Oliveira, L.V., Sena, E.P., De Sousa, D.P., Maia Filho, A.L.M., Soriano, R.N., Da S. Lopes, L., Branco, L.G.S., De Oliveira, A.P., Salgado, H.C. and Sabino, J.P.J. 2021. Acute autonomic effects of rose oxide on cardiovascular parameters of Wistar and spontaneously hypertensive rats. Life. Sci. 287, 120107. Estrada, J.E.C., Rodrigues, K.E., Maciel, A., Bannwart, C.M., Dias, W.F., Hamoy, M., Zingali, R.B., Soares, A.M., Ribeiro, C.H.M.A., Gerlach, R.F., Monteiro, M.C. and Prado, A.F. 2022. BmooMPα-I, a metalloproteinase isolated from Bothrops moojeni venom, reduces blood pressure, reverses left ventricular remodeling and improves cardiac electrical conduction in rats with renovascular hypertension. Toxins 14, 766. Fan, H., Shang, N., Davidge, S.T. and Wu, J. 2024. Chicken muscle-derived ACE2-upregulating peptide VVHPKESF reduces blood pressure associated with the ACE2/Ang (1-7)/MasR axis in spontaneously hypertensive rats. Mol. Nutr. Food. Res. 68(5), 2300524. Fioretti, A.C., Dsouki, N.A., Do Vale, B., De Carvalho, R.P., Dias, D.P.M., Venancio, D.P., Fonseca, F.L.A. and Sato, M.A. 2022. Vitamin D supplementation at different doses affects the vagal component of the baroreceptor reflex and the Bezold-Jarisch reflex in eutrophic rats. Front. Physiol. 13, 2022. Flahault, A., Keck, M., Girault-Sotias, P.E., Esteoulle, L., De Mota, N., Bonnet, D. and Llorens-Cortes, C. 2021. LIT01-196, a metabolically stable apelin-17 analog, normalizes blood pressure in hypertensive DOCA-salt rats via a NO synthase-dependent mechanism. Front. Pharmacol. 12, 715095. Gao, J., Akbari, A. and Wang, T. 2022. Green tea could improve elderly hypertension by modulating arterial stiffness, the activity of the renin/angiotensin/aldosterone axis, and the sodium–potassium pumps in old male rats. J. Food Biochem. 46, 14398; e14398 Gardim, C.B., Veiga, A.C., Aguilar, B.A., Philbois, S.V. and Souza, H.C.D. 2021. Effects of chronic cholinergic stimulation associated with aerobic physical training on cardiac morphofunctional and autonomic parameters in spontaneously hypertensive rats. Sci. Rep. 11(1), 17141. Gemes, G., Rigaud, M., Dean, C., Hopp, F.A., Hogan, Q.H. and Seagard, J. 2009. Baroreceptor reflex is suppressed in rats that develop hyperalgesia behavior after nerve injury. Pain 146, 293–300. Geraldes, V., Laranjo, S., Nunes, C. and Rocha, I. 2023. Central autonomic network regions and hypertension: unveiling sympathetic activation and genetic therapeutic perspectives. Biology 12(8), 1153. Getsy, P.M., Young, A.P., Bates, J.N., Baby, S.M., Seckler, J.M., Grossfield, A., Hsieh, Y.H., Lewis, T.H.J., Jenkins, T.W., Gaston, B. and Lewis, S.J. 2022. S-nitroso-L-cysteine stereoselectively blunts the adverse effects of morphine on breathing and arterial blood gas chemistry while promoting analgesia. Biomed. Pharmacother. 153, 113436. Giles, G.I., Erickson, J.R. and Bussey, C.T. 2022. Photoactivation of tDodSNO induces localized vasodilation in rats: metabolically stable S-nitrosothiols can act as targeted nitric oxide donors in vivo. Nitric. Oxide. 129, 53–62. Gomes, M.F.P., De Moura, E.D.O.C., Cardoso, N.M., Da Silva, G.A., Dos Santos, A.C.C., De Souza, F.S., Estadella, D., Lambertucci, R.H., Lago, J.H.G. and Medeiros, A. 2023. Supplementation with okra combined or not with exercise training is able to protect the heart of animals with metabolic syndrome. Sci. Rep. 13(1), 1468. Gomes, P.M., Batista, J.S., Sá, R.W.M. and Antunes, V.R. 2023. Short exposure to high salt in drinking solution leads to a cardiovascular phenotype of hypertension without changes in the blood volume of rats. Exp. Physiol. 108, 361–370. Gonca, E. 2015. Comparison of thiopental and ketamine+xylazine anesthesia in ischemia/reperfusion-induced arrhythmias in rats. Turk. J. Med. Sci. 45, 1413–1420. Griffiths, P.R., Lolait, S.J., Paton, J.F.R. and O’Carroll, A.M. 2021. Circumventricular organ apelin receptor knockdown decreases blood pressure and sympathetic drive responses in the spontaneously hypertensive rat. Front. Physiol. 12, e5827. Gunjegaonkar, S.M., Khakal, V.R., Joshi, A.A., Nargund, S.L., Shinde, S.D. and Khan, N.S. 2022. Cardio-protective effect of Cocculus hirsutus in experimental hypertension in rats. J. Ayurveda Integr. Med. 13, 100652. Honetschlägerová, Z., Husková, Z., Kikerlová, S., Sadowski, J., Kompanowska-Jezierska, E., Táborský, M., Vaňourková, Z., Kujal, P. and Červenka, L. 2024. Renal sympathetic denervation improves pressure-natriuresis relationship in cardiorenal syndrome: insight from studies with Ren2 transgenic hypertensive rats with volume overload induced using aorto-caval fistula. Hypertens. Res. 47(4), 998–1016. Hosseiniravesh, M.R., Hojati, V., Mohebbati, R., Khajavirad, A., Shajiee, H. and Shafei, M.N. 2022. Effect of the non-N-Methyl-D-aspartate receptor of the glutamatergic system of the pedunculopontine tegmental nucleus on cardiovascular responses in normotensive and hydralazine-induced hypotensive rats. Adv. Biomed. Res. 11, 78. Hu, Y., Wu, T., Liu, X. and Qiao, D. 2022. Effects of exercise on the cardiovascular function of rats in a sulfur dioxide polluted environment. Acad. Bras. Ciênc. 94(2), e20211180. Huang, X.H., Hamza, S.M., Zhuang, W.Q., Cupples, W.A. and Braam, B. 2021. Angiotensin II and the renal hemodynamic response to an isolated increased renal venous pressure in rats. Front. Physiol. 12, 753355. Irshad, N., Khan, A., Khan, A. and Iqbal, M.S. 2021. Antihypertensive potential of selected pyrimidine derivatives: explanation of underlying mechanistic pathways. Biomed. Pharmacother. 139, 111567. Jeong, H., Cho, A., Ay, I. and Bonmassar, G. 2022. Short-pulsed micro-magnetic stimulation of the vagus nerve. Front. Physiol. 13, 938101. Jiang, S.Y., Shen, Y.H., Rao, T.W. and Zhao, X.G. 2021. Therapeutic role of atrial natriuretic peptide in early treatment of traumatic hemorrhagic shock. Eur. J. Inflamm. 19, 20587392211004402. Jordão, M.R., Pessoa, F.G., Fonseca, K.C.B., Zanoni, F., Salemi, V.M.C., Souza, L.E., Ribeiro, O.N., Fernandes, F., Irigoyen, M.C., Moreira, L.F.P., Mady, C. and Ramires, F.J.A. 2021. Effects of sympathectomy on myocardium remodeling and function. Clinics 76, e1958. Kapsdorferová, V., Grešová, S. and Švorc, P. 2024. Measurement of blood pressure in rats: invasive or noninvasive methods?. Physiol. Rep. 12(17), e70041. Kirillov, Y., Makartseva, L., Kozlova, M., Chernov, I., Shtemplevskaya, Y. and Areshidze, D. 2021. Effect of chronic alcohol intoxication and constant lighting on cardiovascular parameters in male rats. Arch. Euromedica. 11, 43–47. Komnenov, D. and Rossi, N.F. 2023. Fructose-induced salt-sensitive blood pressure differentially affects sympathetically mediated aortic stiffness in male and female Sprague-Dawley rats. Physiol. Rep. 11, e15687. Konopelski, P. and Ufnal, M. 2016. Electrocardiography in rats: a comparison to human. Physiol. Res. 65, 717–725. Kousar, M., Salma, U., Khan, T. and Shah, A.J. 2022. Antihypertensive potential of tartaric acid and exploration of underlying mechanistic pathways. Dose-Response 20(4), 15593258221135728. Kramer, K. and Remie, R. 2005. Measuring blood pressure in small laboratory animals. Methods. Mol. Med. 108, 51–62. Kurogochi, K., Uechi, M. and Orito, K. 2022. Involvement of neurokinin-1 receptors in the autonomic nervous system in colorectal distension-induced cardiovascular suppression in rats. Front. Pharmacol. 13, 1020685. Lambert, K., Demion, M., Lagacé, J.C., Hokayem, M., Dass, M., Virsolvy, A., Jover, B., Bourret, A. and Bisbal, C. 2021. Grape polyphenols and exercise training have distinct molecular effects on cardiac hypertrophy in a model of obese insulin-resistant rats. J. Nutr. Biochem. 87, 108522. Layton, R., Layton, D., Beggs, D., Fisher, A., Mansell, P. and Stanger, K.J. 2023. The impact of stress and anesthesia on animal models of infectious disease. Front. Vet. Sci. 10, 1086003. Lazaro, A., Stoll, P., Von Elverfeldt, D., Kreisel, W. and Deibert, P. 2023. Close relationship between systemic arterial and portal venous pressure in an animal model with healthy liver. Int. J. Mol. Sci. 24, 9963. Li, X., Yang, H.F., Chen, Y., Pei, L.J. and Jiang, R. 2021. Effect of the icariin on endothelial microparticles, endothelial progenitor cells, platelets, and erectile function in spontaneously hypertensive rats. Andrology 10(3), 576–584. Liu, S., Bilston, L.E., Rodriguez, N.F., Wright, C., McMullan, S., Lloyd, R., Stoodley, M.A. and Hemley, S.J. 2022. Changes in intrathoracic pressure, not arterial pulsations, exert the greatest effect on tracer influx in the spinal cord. Fluids. Barriers. CNS. 19(1), 14. Liu, X.F., Patel, K.P. and Zheng, H. 2021a. Role of renal sympathetic nerves in GLP-1 (glucagon-like peptide-1) receptor agonist Exendin-4-mediated diuresis and natriuresis in diet-induced obese rats. J. Am. Heart Assoc. 10, 22542. Liu, X.X., Wang, X.X., Pan, Y., Zhao, L., Sun, S., Luo, A., Bao, C.G., Tang, H.A. and Han, Y. 2021b. Artemisinin improves acetylcholine-induced vasodilatation in rats with primary hypertension. Drug Des. Dev. Ther. 2021, 4489. Lopes, F.N.C., Da Cunha, N.V., De Campos, B.H., Fattori, V., Panis, C., Cecchini, R., Verri Jr, W.A., Pinge-Filho, P. and Martins-Pinge, M.C. 2022. Antioxidant therapy reverses sympathetic dysfunction, oxidative stress, and hypertension in male hyperadipose rats. Life. Sci. 295, 120405. López-Canales, O.A., Pavón, N., Ubaldo-Reyes, L.M., Juárez-Oropeza, M.A., Torres-Durán, P.V., Regla, I. and Paredes-Carbajal, M.C. 2022. Characterization of hypotensive and vasorelaxant effects of PHAR-DBH-Me a new cannabinoid receptor agonist. Korean. J. Physiol. Pharmacol. 26, 77–86. Luz, M.S., Gadelha, D.D.A., Andrade, K.J.S., Travassos, R.A., Ribeiro, J.D., Carvalho-Galvão, A., Cruz, J.C., Balarini, C.M., Braga, V.A. and França-Falcão, M.S. 2022. Borneol reduces sympathetic vasomotor hyperactivity and restores depressed baroreflex sensitivity in rats with renovascular hypertension. Hypertens. Res. 45, 802–813. Macleod, B.A., Augereau, P. and Walker, M.J.A. 1983. Effects of halothane anesthesia compared with fentanyl anesthesia and no anesthesia during coronary ligation in rats. Anesthesiology 58, 44–52. Makino, M., Hayashi, H., Takezawa, H., Hirai, M., Saito, H. and Ebihara, S. 1997. Circadian rhythms of cardiovascular functions are modulated by the baroreflex and the autonomic nervous system in the rat. Circulation 96, 1667–1674. Marc, Y., Hmazzou, R., De Mota, N., Balavoine, F. and Llorens-Cortes, C. 2021. Effects of firibastat in combination with enalapril and hydrochlorothiazide on blood pressure and vasopressin release in hypertensive DOCA-salt rats. Biomed. Pharmacother. 140, 111682. Maris, M.E., Melchert, R.B., Joseph, J. and Kennedy, R.H. 2005. Gender differences in blood pressure and heart rate in spontaneously hypertensive and Wistar-Kyoto rats. Clin. Exp. Pharmacol. Physiol. 32(1-2), 35–39. Martin, D., Reihe, C., Drummer, S., Roessler, K., Boomer, S. and Nelson, M. 2022. Venoconstrictor responses to activation of bradykinin-sensitive pericardial afferents involve the region of the hypothalamic paraventricular nucleus. Physiol. Rep. 10, e15221. Melo, M.R., Voger, J.J., Connelly, A.A., Bassi, J.K., Colombari, E. and Allen, A.M. 2022. Chemogenetic inhibition of Phox2-expressing neurons in the commissural NTS decreases blood pressure in anesthetized spontaneously hypertensive rats. Neurosci. Lett. 787, 136817. Meng, T., Ren, X., Chen, X., Yu, J., Agrimi, J., Paolocci, N. and Gao, W.D. 2019. Anesthetic agents isoflurane and propofol decrease maximal Ca2+-activated force and thus contractility in the failing myocardium. J. Pharmacol. Exp. Ther. 371, 615–623. Meng, X.X., Nan, G.J., Hu, H., He, J.Y., Li, Y.Z. and Yang, G.D. 2024. Effects of vitamin C on the pharmacokinetics and pharmacodynamics of nimodipine in rats. Biomed. Chromatogr. 38(4), 703–717. Mensah, E.A., Daneshtalab, N. and Tabrizchi, R. 2022. Differential biomechanics in resistance arteries of male compared with female Dahl hypertensive rats. J. Hyperten. 40(3), 596–605. Miklosz, J., Kalaska, B., Zajaczkowski, S., Pawlak, D. and Mogielnicki, A. 2021. Monitoring of cardiorespiratory parameters in rats—validation based on pharmacological stimulation. Pharmaceuticals 14(12), 1223. Miller, E.D., Ackerly, J.A. and Peach, M.J. 1978. Blood pressure support during general anesthesia in a renin-dependent state in the rat. Anesthesiology 48, 404–408. Misak, A., Grman, M., Tomasova, L., Makara, O., Rostakova, Z., Waczulikova, I. and Ondrias, K. 2023. Use of a rat model to characterize 35 arterial pulse wave parameters in a comparative study of isoflurane and Zoletil/xylazine anesthesia and the effect of Acanthopanax senticosus extract. Anim. Model. Exp. Med. 6, 474–488. Mogilnicka, I., Jaworska, K., Koper, M., Maksymiuk, K., Szudzik, M., Radkiewicz, M., Chabowski, D. and Ufnal, M. 2024. Hypertensive rats show increased renal excretion and decreased tissue concentrations of glycine betaine, a protective osmolyte with diuretic properties. PLos One 19, 294926. Mohamed, R.H., Abdel Hay, N.H., Fawzy, N.M., Tamim, Y.M., Doaa Karem, M.M., Yehia, D.A.Y., Abdel Maksoud, O.M. and Abdelrahim, D.S. 2024. Targeting mevalonate pathway by zoledronate ameliorated pulmonary fibrosis in a rat model: promising therapy against post-COVID-19 pulmonary fibrosis. Fundam. Clin. Pharmacol. 38(4), 703–717. Mohammed Abdulsalam, T., Hasanin, A.H., Hussein Mohamed, R., Khairy, E., Mahmoud, D., Habib, E.K. and Badawy, A.E.S. 2023. A comparative study between angiotensin receptor neprilysin inhibitor (thiorphan/irbesartan) with each of nitrate and carvedilol in a rat model of myocardial ischemic reperfusion injury. Fundam. Clin. Pharmacol. 37, 753–768. Moreno, K.G.T., Marques, A.A.M., Da Silva, G.P., Lourençone, B.R., Fortini, C.S., Leite, P.R.T., Dos Santos, A.C., Souza, R.I.C., Da Siva, L.I. and Gasparotto Junior, A. 2022. A new approach for the development of multiple cardiovascular risk factors in two rat models of hypertension. Pharmaceuticals 15(7), 853. Morissette, P., Li, N., Ballard, J.E., Vavrek, M., Adams, G.L., Regan, C., Regan, H., Lee, K.J., Wang, W., Burton, A., Chen, F., Gerenser, P., Li, Y., Kraus, R.L., Tellers, D., Palani, A., Zhu, Y., Sun, C., Bianchi, E., Colarusso, S., De Simone, D., Frattarelli, T., Pasquini, N.M. and Amin, R.P. 2022. Guiding chemically synthesized peptide drug lead optimization by derisking mast cell degranulation-related toxicities of a NaV1.7 peptide inhibitor. Toxicol. Sci. 185(2), 170–183. Murakami, M., Niwa, H., Kushikata, T., Watanabe, H., Hirota, K., Ono, K. and Ohba, T. 2014. Inhalation anesthesia is preferable for recording rat cardiac function using an electrocardiogram. Biol. Pharm. Bull. 37(5), 834–839. Mutiso, S.K., Rono, D.K. and Bukachi, F. 2014. Relationship between anthropometric measures and early electrocardiographic changes in obese rats. BMC. Res. Notes. 7, 931. Nascimento, M.M., Bernardo, D.R.D., De Bragança, A.C., Shimizu, M.H.M., Seguro, A.C., Volpini, R.A. and Canale, D. 2022. Treatment with β-blocker nebivolol ameliorates oxidative stress and endothelial dysfunction in tenofovir-induced nephrotoxicity in rats. Front. Med. - Sec. Nephrol. 9, 953749. Nowiński, A., Chabowski, D., Giebułtowicz, J., Aleksandrowicz, M. and Ufnal, M. 2024. Deoxycholic acid, a secondary bile acid, increases cardiac output and blood pressure in rats. Nutrients 16, 32. Nugent, W.H., Carr, D.A., Macbryde, R., Bruce, E.D. and Song, B.K. 2021. Gavage approach to oxygen supplementation with oxygen therapeutic Ox66™ in a hypoventilation rodent model of respiratory distress. Artif. Cells Nanomed. Biotechnol. 49, 709–716. Olatoye, F.J., Akindele, A.J., Balogun, O.E., Awodele, O. and Adejare, A.A. 2023. Antihypertensive effect of kolaviron, a bioflavonoid from Garcinia kola, in L-NAME induced hypertension in rats. Nat. Prod. Commun. 18(3), 1934578X221148608. Oleksa, V., Bernátová, I., Patsula, V., Líšková, S., Bališ, P., Radošinská, J., Mičurová, A., Kluknavský, M., Jasenovec, T., Radošinská, D., Macková, H. and Horák, D. 2021. Poly(ethylene glycol)-alendronate-coated magnetite nanoparticles do not alter cardiovascular functions and red blood cells’ properties in hypertensive rats. Nanomaterials 11(5), 1238. Oliveira, G.M., Dionísio, T.J., Fleury, C.A., Calvo, A.M., Santos, C.F. and Faria, F.A.C. 2022b. Is it safe to use vasoconstrictors in association treated with amitriptyline or can it potentiate cardiovascular effects? In vivo animal study. Appl. Sci. 12, 11998. Oliveira, L.A., Carvalho, I.I., Kurokawa, R.Y., Duarte, J.D.O., Busnardo, C. and Crestani, C.C. 2022a. Differential roles of prelimbic and infralimbic cholinergic neurotransmissions in control of cardiovascular responses to restraint stress in rats. Brain. Res. Bull. 181, 175–182. Oliveira, L.A., Pollo, T.R.S., Rosa, E.A., Duarte, J.O., Xavier, C.H. and Crestani, C.C. 2021. Both prelimbic and infralimbic noradrenergic neurotransmissions modulate cardiovascular responses to restraint stress in rats. Front. Physiol. 12, 700540. Orts-Sebastian, A., Ludin, N.M., Pawley, M.D.M., Cheeseman, J.F. and Warman, G.R. 2019. Impact of anaesthesia on circadian rhythms and implications for laboratory experiments. Exp. Neurol. 311, 318–322. Page, M.J., McKenzie, J.E., Bossuyt, P.M., Boutron, I., Hoffmann, T.C., Mulrow, C.D., Shamseer, L., Tetzlaff, J.M., Akl, E.A., Brennan, S.E., Chou, R., Glanville, J., Grimshaw, J.M., Hróbjartsson, A., Lalu, M.M., Li, T., Loder, E.W., Mayo-Wilson, E., McDonald, S., McGuinness, L.A., Stewart, L.A., Thomas, J., Tricco, A.C., Welch, V.A., Whiting, P. and Moher, D. 2021. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372, 71. Pan, Y., Sun, S., Wang, X.X., Chen, A.D., Fei, X.J., Wang, W. and Han, Y. 2021. Improvement of vascular function by knockdown of salusin-β in hypertensive rats via nitric oxide and reactive oxygen species signaling pathway. Front. Physiol. - Sec. Vascular Physiol. 12, 622954. Pannangpetch, P., Tangsucharit, P., Thanaruksa, R., Proongkhong, T., Srisuwan, S. and Aekthammarat, D. 2022. Antihypertensive effect of Mali-Nil surin rice bran hydrolysate and its mechanisms related to the EDHF-mediated vasorelaxation and L-type Ca2+ channel-mediated vasoconstriction in L-NAME hypertensive rats. Biomed. Pharmacother. 150, 113003. Pezeshki, Z. and Nematbakhsh, M. 2021. Sex differences in the renal vascular responses of AT1 and Mas receptors in two-kidney-one-clip hypertension. Int. J. Hypertens. 2021, 8820646. Poasakate, A., Maneesai, P., Rattanakanokchai, S., Bunbupha, S., Tong-Un, T. and Pakdeechote, P. 2021. Genistein prevents nitric oxide deficiency-induced cardiac dysfunction and remodeling in rats. Antioxidants 10(2), 237. Pontes, R.B., Nishi, E.E., Crajoinas, R.O., Milanez, M.I.O., Girardi, A.C.C., Campos, R.R. and Bergamaschi, C.T. 2023. Relative contribution of blood pressure and renal sympathetic nerve activity to proximal tubular sodium reabsorption via NHE3 activity. Int. J. Mol. Sci. 24, 349. Potter, J.C., Whiles, S.A., Miles, C.B., Whiles, J.B., Mitchell, M.A., Biederman, B.E., Dawoud, F.M., Breuel, K.F., Williamson, G.A., Picken, M.M. and Polichnowski, A.J. 2021. Salt-sensitive hypertension, renal injury, and renal vasodysfunction associated with Dahl salt-sensitive rats are abolished in consomic SS.BN1 rats. J. Amer. Heart Assoc. 10, 20261. Rahbardar, M.G., Eisvand, F., Rameshrad, M., Razavi, B.M., Yazdi, A.T. and Hosseinzadeh, H. 2024. Carnosic acid mitigates doxorubicin-induced cardiac toxicity: evidence from animal and cell model investigations. Iran. J. Basic. Med. Sci. 27, 425–438. Raji, V., Loganathan, C., Ramesh, T. and Thayumanavan, P. 2023. Dual antidiabetic and antihypertensive activity of fucoxanthin isolated from Sargassum wightii Greville in in vivo rat model. Food Sci. Hum. Wellness 12, 1693–1700. Ramadhani, R., Sato, T., Okada, Y., Ohke, H. and Ishii, H. 2023. Differences in the regulatory mechanism of blood flow in the orofacial area mediated by neural and humoral systems. J. Comp. Physiol. B 193, 109–124. Rastegarmanesh, A., Rostami, B., Nasimi, A. and Hatam, M. 2023. In the parvocellular part of paraventricular nucleus, glutamatergic and GABAergic neurons mediate cardiovascular responses to AngII. Synapse 77, e22259. Redfors, B., Shao, Y. and Omerovic, E. 2014. Influence of anesthetic agent, depth of anesthesia and body temperature on cardiovascular functional parameters in the rat. Lab. Anim. 48(1), 6–14. Revand, R. and Singh, S.K. 2021. Ipsilateral somatic nerves mediate histamine-induced vasosensory reflex responses involving perivascular afferents in rat models. Sci. Rep. 11, 14648. Ribeiro, A.B., Da Silva, T.M., Santos-Júnior, N.N., Castania, J.A., Fazan, R. and Salgado, H.C. 2021. Short-term effect of ligature-induced periodontitis on cardiovascular variability and inflammatory response in spontaneously hypertensive rats. BMC Oral Health 21, 515. Rostami, B., Nasimi, A. and Hatam, M. 2022. Hypothalamic paraventricular nucleus augments baroreflex sensitivity, role of angiotensin II. Brain Res. 1802, 148218. Saha, D.C., Saha, A.C., Malik, G., Astiz, M.E. and Rackow, E.C. 2007. Comparison of cardiovascular effects of tiletamine-zolazepam, pentobarbital, and ketamine-xylazine in male rats. J. Am. Assoc. Lab. Anim. Sci. 46, 74–80. Sainsily, X., Coquerel, D., Giguere, H., Dumont, L., Tran, K., Noll, C., Ionescu, A.L., Côté, J., Longpré, J.M., Carpentier, A., Marsault, E., Lesur, O., Sarret, P. and Auger-Messier, M. 2021. Elabela protects spontaneously hypertensive rats from hypertension and cardiorenal dysfunctions exacerbated by dietary high-salt intake. Front. Pharmacol. -. Sec. Cardiovasc. Smooth. Muscle. Pharmacol. 12, 709467. Salman, I.M. 2023. Differential frequency-dependent reflex summation of the aortic baroreceptor afferent input. Pflugers. Archiv - Eur. J. Physiol. 475, 933–944. Salman, I.M., Ameer, O.Z., McMurray, S., Hassan, S.F., Sridhar, A., Lewis, S.I. and Hsieh, Y.H. 2023. Differential central integration of left versus right baroreceptor afferent input in spontaneously hypertensive rats. J. Hypertens. 41(7), 1191–1200. Salman, I.M., Ameer, O.Z., Mcmurray, S., Hassan, S.F., Sridhar, A., Lewis, S.J. and Hsieh, Y.H. 2022. Low intensity stimulation of aortic baroreceptor afferent fibers as a potential therapeutic alternative for hypertension treatment. Sci. Rep. 12(1), 12242. Sano, Y., Ito, S., Yoneda, M., Nagasawa, K., Matsuura, N., Yamada, Y., Uchinaka, A., Bando, Y.K., Murohara, T. and Nagata, K. 2016. Effects of various types of anesthesia on hemodynamics, cardiac function, and glucose and lipid metabolism in rats. Am. J. Physiol. Heart. Circ. Physiol. 311(6), H1360–H1366. Santana, A.C., Andraus, W., Zimelewicz Oberman, D., Rabelo, N.N., Silva, F.M.O., Dellê, H., Pepineli, R., De Moraes, E.L., Scavone, C., De Sá Lima, L., Degaspari, S., Brasil, S., Solla, D.J.F., Ruiz, L.M., De Oliveira-braga, K.A., Nepomuceno, N.A., Pêgo-Fernandes, P.M., Tullius, S.G. and Figueiredo, E.G. 2023. Immunomodulatory response in an experimental model of brain death. Sci. Rep. 13(1), 10524. Santos, C.V.D., Kerkhoff, J., Tomazelli, C.A., Wenceslau, C.F., Sinhorin, A.P., De Jesus Rodrigues, D., Carneiro, F.S. and Bomfim, G.F. 2022. Vasoconstrictor and hemodynamic effects of a methanolic extract from Rhinella marina toad poison. Toxicon 218, 57–65. Saugel, B., Bebert, E.J., Briesenick, L., Hoppe, P., Greiwe, G., Yang, D., Ma, C., Mascha, E.J., Sessler, D.I. and Rogge, D.E. 2022. Mechanisms contributing to hypotension after anesthetic induction with sufentanil, propofol, and rocuronium: a prospective observational study. J. Clin. Monit. Comput. 36, 341–347. Secher, N., Malte, C.L., Tønnesen, E., Østergaard, L. and Granfeldt, A. 1985. Comparing anesthesia with isoflurane and fentanyl/fluanisone/midazolam in a rat model of cardiac arrest. J. Appl. Physiol. 123, 867–875. Sedighi, M., Ghorbanzadeh, V., Abaszadeh, S., Karimi, A., Cheraghi, M., Rafieian-Kopaei, M., Moghimian, M., Mohammadi, A., Veiskarami, S., Mokhayeri, Y. and Nazari, A. 2021. Up-regulation of chemokine receptor type 4 expression in the ischemic reperfused heart by alcoholic extract of Cichorium intybus rescue the heart from ischemia injury in male rat. J. Pharm. Pharmacol. 73, 1351–1360. Selejan, S.R., Linz, D., Mauz, M., Hohl, M., Huynh, A.K.D., Speer, T., Wintrich, J., Kazakov, A., Werner, C., Mahfoud, F. and Böhm, M. 2022. Renal denervation reduces atrial remodeling in hypertensive rats with metabolic syndrome. Basic. Res. Cardiol. 117, 36. Seyde, W.C. and Longnecker, D.E. 1984. Anesthetic influences on regional hemodynamics in normal and hemorrhaged rats. Anesthesiology 61, 686–698. Shaikh, F. and Bhatt, L.K. 2022. Cardioprotective effect of polymyxin-B and dantrolene combination on isoproterenol-induced hypertrophic cardiomyopathy in rats, via attenuation of calmodulin-dependent protein kinase II. Chem. &. Biodiversity. 19(10), e202200309. Sharma, N.M., Haibara, A.S., Katsurada, K., Nandi, S.S., Liu, X., Zheng, H. and Patel, K.P. 2021. Central Ang II (Angiotensin II)-mediated sympathoexcitation. Role for HIF-1α (hypoxia-inducible Factor-1α) facilitated glutamatergic tone in the paraventricular nucleus of the hypothalamus. Hypertension 77(1), 147–157. Silva, E.A.P., Carvalho, J.S., Dos Santos, D.M., Oliveira, A.M.S., De Souza Araújo, A.A., Serafini, M.R., Oliveira Santos, L.A.B., Batista, M.V.D.A., Viana Santos, M.R., Siqueira Quintans, J.D.S., Quintans-Júnior, L.J. and Barreto, A.S. 2021. Cardiovascular effects of farnesol and its β-cyclodextrin complex in normotensive and hypertensive rats. Eur. J. Pharmacol. 901, 174060. Silva, E.D., Conde, C., Machado, D.I., Ventura, S., Couto, S.M.F. and Vattimo, M.F.F. 2022. Effect of curcumin on acute chronic kidney disease due to ischemia-reperfusion syndrome. Rev. Esc. Enferm. 56(spe), e20210440. Sokolov, A.Y., Volynsky, M.A., Potapenko, A.V., Iurkova, P.M., Zaytsev, V.V., Nippolainen, E. and Kamshilin, A.A. 2023. Duality in response of intracranial vessels to nitroglycerin revealed in rats by imaging photoplethysmography. Sci. Rep. 13, 11928. Soleimanipour, S., Kian, M., Hamedeyazdan, S., Movahhedin, N., Ghaderi, F. and Soraya, H. 2021. The effects of hydroalcoholic extract of Arum orientale on CLP-induced sepsis in rats. Pharm. Sci. 27, 162–169. Souto, C.G.R.G., Lorençone, B.R., Marques, A.A.M., Palozi, R.A.C., Romão, P.V.M., Guarnier, L.P., Tirloni, C.A.S., Dos Santos, A.C., Souza, R.I.C., Zago, P.M.J.J., Dos Reis Lívero, F.A., Lourenço, R.L.B. and et al. 2021. Cardioprotective effects of Talinum paniculatum (Jacq.) Gaertn. in doxorubicin-induced cardiotoxicity in hypertensive rats. J. Ethnopharmacol. 5, 114568. Streeter, K.A., Sunshine, M.D., Davenport, P.W. and Fuller, D.D. 2021. Phrenic afferent activation modulates cardiorespiratory output in the adult rat. J. Neurophysiol. 126, 2091–2103. Sturgess, D.J., Morrison, S., Haluska, B., Gobe, G.C., Jones, M.A., Volante, S. and Venkatesh, B. 2021. Left ventricular impaired relaxation and interstitial myocarditis identified in sepsis-associated cardiac dysfunction: use of a rodent model. Med. Sci. Monit. 27, e929512. Sun, G., Kurosawa, M., Ninomiya, Y., Baba, K., Son, N.H., Yen, H.T., Suzuki, S. and Kano, Y. 2025. Contactless monitoring of respiratory rate variability in rats under anesthesia with a compact 24GHz microwave radar sensor. Front. Vet. Sci. 12, 1518140. Sun, J., Xiao, Y., Xu, W., Xing, W., Du, F., Tian, M., Xu, D., Ren, Y. and Fang, X. 2024. Anti-hypertensive effect of a novel angiotensin II receptor neprilysin inhibitor (ARNi)-S086 in DSS rat model. Front. Cardiovasc. Med. 11, 1348897. Toczek, M., Ryszkiewicz, P., Remiszewski, P., Schlicker, E., Krzyżewska, A., Kozłowska, H. and Malinowska, B. 2023. Weak hypotensive effect of chronic administration of the dual FAAH/MAGL inhibitor JZL195 in spontaneously hypertensive rats as revealed by area under the curve analysis. Int. J. Mol. Sci. 24(13), 10942. Tomeo, R.A., Gomes-de-Souza, L., Benini, R., Reis-Silva, L.L. and Crestani, C.C. 2022. Site-specific regulation of stress responses along the rostrocaudal axis of the insular cortex in rats. Front. Neurosci. 16, 878927. Tsikas, D., Jordan, J. and Engeli, S. 2015. Blood pressure-lowering effects of propofol or sevoflurane anaesthesia are not due to enhanced nitric oxide formation or bioavailability. Br. J. Clin. Pharmacol. 79, 1030–1033. Tumanova, T.S., Kokurina, T.N., Rybakova, G.I. and Aleksandrov, V.G. 2021. Increased systemic level of endotoxin attenuates baroreflex and cardiovascular effects of infralimbic cortex electrostimulation in anesthetized rats. J. Evol. Biochem. Physiol. 57, 1471–1479. Vardar, K., Can, K. and Aksu, U. 2022. Fluid resuscitation aggravates the cellular injury in a hemorrhagic shock model. Dubai Med. J. 5, 141–150. Vidt, D.G., Bredemeyer, A., Sapirstein, E. and Sapirstein, L.A. 1959. Effect of ether anesthesia on the cardiac output, blood pressure, and distribution of blood flow in the albino rat. Circ. Res. 7, 759–764. Vieira-Rocha, M.S., Sousa, J.B., Rodríguez-Rodríguez, P., Arribas, S.M. and Diniz, C. 2022. Elevated vascular sympathetic neurotransmission and remodelling is a common feature in a rat model of foetal programming of hypertension and SHR. Biomedicines 10, 1902. Vinik, A.I. and Ziegler, D. 2007. Diabetic cardiovascular autonomic neuropathy. Circulation 115, 387–839. Wang, W. and Zhang, X. 2021. Antihypertensive effect of galegine from Biebersteinia heterostemon in rats. Molecules 26(16), 4830. Wang, X., Gu, H., Li, K., Lin, J., Zhu, Y. and Deng, W. 2021. DPP4 inhibitor reduces portal hypertension in cirrhotic rats by normalizing arterial hypocontractility. Life Sci. 284, 119895. Wirtz, M.R., Roelofs, J.J., Goslings, J.C. and Juffermans, N.P. 2022. Treatment with ddAVP improves platelet-based coagulation in a rat model of traumatic hemorrhagic shock. Trauma. Surg. Acute. Care. Open. 7, 852. Wu, H., Jiang, L., Shum, T.F. and Chiou, J. 2022. Elucidation of anti-hypertensive mechanism by a novel Lactobacillus rhamnosus AC1 fermented soymilk in the deoxycorticosterone acetate-salt hypertensive rats. Nutrients 14, 3174. Wu, Y., Yu, Q., Dai, M., Wang, J., Shu, L., Dai, D., Chen, J., Zhang, J. and Bao, M. 2023. Carotid baroreceptor stimulation prevents oxidative stress by inhibiting MAO-A and prevents cardiac remodeling in obese rats. Obesity 31, 1620–1633. Xu, Y., Fei, X., Fu, H., Chen, A., Zhu, X., Zhang, F. and Han, Y. 2023. Upregulated expression of a TOR2A gene product-salusin-β in the paraventricular nucleus enhances sympathetic activity and cardiac sympathetic afferent reflex in rats with chronic heart failure induced by coronary artery ligation. Acta Physiol. 238, e13987. Yen, P.S.Y., Liu, Y.C., Chu, C.H. and Chen, S.L. 2022. Upregulation of glutamatergic receptors in hippocampus and locomotor hyperactivity in aged spontaneous hypertensive rat. Cell. Mol. Neurobiol. 42(7), 2205–2217. Yoshimoto, T., Eguchi, K., Sakurai, H., Ohmichi, Y., Hashimoto, T., Ohmichi, M., Morimoto, A., Yamaguchi, Y., Ushida, T., Iwase, S., Sugenoya, J. and Kumazawa, T. 2011. Frequency components of systolic blood pressure variability reflect vasomotor and cardiac sympathetic functions in conscious rats. J. Physiol. Sci. 61, 373–383. Zago, P.M.J.J., Silva, G.R.D., Amaral, E.C., Barboza, L.N., Braga, F.D.A., Lorençone, B.R., Marques, A.A.M., Moreno, K.G.T., Leite, P.R.T., Veiga, A.D.A., Souza, L.M.D., Souza, R.I.C., Santos, A.C.D., Ribeiro-Paes, J.T., Gasparotto Junior, A. and Lívero, F.A.D.R. 2021. Multiple risk factors for heart disease: a challenge to the ethnopharmacological use of Croton urucurana Baill. Evidence-Based Complementary Alternative Med. 2021, 6580458. Zhang, Q., Shen, Y., Niloy, S.I., O'Rourke, S.T. and Sun, C. 2023. Chronic effects of apelin on cardiovascular regulation and angiotensin II-induced hypertension. Pharmaceuticals 16, 600. Zhang, X.L., Cheng, Y., Xing, C.L., Ying, J.Y., Yang, X., Cai, X.D. and Lu, G.P. 2023. Establishment of a rat model of capillary leakage syndrome induced by cardiopulmonary resuscitation after cardiac arrest. Curr. Med. Sci. 43(4), 708–715. Zicha, J., Behuliak, M., Vavřínová, A., Dobešová, Z., Kuneš, J., Rauchová, H. and Vaněčková, I. 2021. Cooperation of augmented calcium sensitization and increased calcium entry contributes to high blood pressure in salt-sensitive Dahl rats. Hypertens. Res. 44, 1067–1078. Żmudzka, E., Lustyk, K., Siwek, A., Wolak, M., Gałuszka, A., Jaśkowska, J., Kołaczkowski, M., Sapa, J. and Pytka, K. 2023. Novel arylpiperazine derivatives of salicylamide with α1-adrenolytic properties showed antiarrhythmic and hypotensive properties in rats. Int. J. Mol. Sci. 24(1), 293. Zorniak, M., Mitrega, K., Bialka, S., Porc, M. and Krzeminski, T.F. 2010. Comparison of thiopental, urethane, and pentobarbital in the study of experimental cardiology in rats in vivo. J. Cardiovasc. Pharmacol. 56, 38–44. Supplementary MaterialSupplementary File 1. BP data in normotensive rats.
Supplementary File 2. BP data in hypertensive rats.
| ||
| How to Cite this Article |
| Pubmed Style Švorc P, Grešová S, Novotná V. Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Vet. J.. 2025; 15(12): 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 Web Style Švorc P, Grešová S, Novotná V. Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). https://www.openveterinaryjournal.com/?mno=272716 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.3 AMA (American Medical Association) Style Švorc P, Grešová S, Novotná V. Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Vet. J.. 2025; 15(12): 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 Vancouver/ICMJE Style Švorc P, Grešová S, Novotná V. Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 Harvard Style Švorc, P., Grešová, . S. & Novotná, . V. (2025) Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Vet. J., 15 (12), 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 Turabian Style Švorc, Pavol, Soňa Grešová, and Viktória Novotná. 2025. Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Veterinary Journal, 15 (12), 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 Chicago Style Švorc, Pavol, Soňa Grešová, and Viktória Novotná. "Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024)." Open Veterinary Journal 15 (2025), 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 MLA (The Modern Language Association) Style Švorc, Pavol, Soňa Grešová, and Viktória Novotná. "Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024)." Open Veterinary Journal 15.12 (2025), 6166-6189. Print. doi:10.5455/OVJ.2025.v15.i12.3 APA (American Psychological Association) Style Švorc, P., Grešová, . S. & Novotná, . V. (2025) Effect of anesthesia and measurement site on baseline blood pressure in rats (2021–2024). Open Veterinary Journal, 15 (12), 6166-6189. doi:10.5455/OVJ.2025.v15.i12.3 |