| Research Article | ||
Open Vet. J.. 2025; 15(12): 6856-6871 Open Veterinary Journal, (2025), Vol. 15(12): 6856-6871 Research Article Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysisAde Erma Suryani1,2, Khoirun Nisa1, Sri Handayani3, Sri Handyani1, Anastasia Wheni Indrianingsih1, Wafda Naufi Marva Nuzulia4, Hastari Wuryastuty5* and Yanuartono Yanuartono51Research Center for Food Technology and Processing, National Research and Innovation Agency, Yogyakarta, Indonesia 2Doctor of Veterinary Science Program, Faculty of Veterinary Medicine, Gadjah Mada University, Yogyakarta, Indonesia 3Research Center for Pharmaceutical Ingredients and Traditional Medicine, National Research and Innovation Agency, Bogor, Indonesia 4Biology Study Program, Faculty of Mathematics and Natural Sciences, Sebelas Maret University, Surakarta, Indonesia 5Departement of Veterinary Internal Medicine, Faculty of Veterinary Medicine, Gadjah Mada University, Yogyakarta, Indonesia *Corresponding Author: Hastari Wuryastuty. Department of Veterinary Internal Medicine, Faculty of Veterinary Medicine, Gadjah Mada University, Yogyakarta, Indonesia. Email: hastari [at] ugm.ac.id Submitted: 23/08/2025 Revised: 14/11/2025 Accepted: 26/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
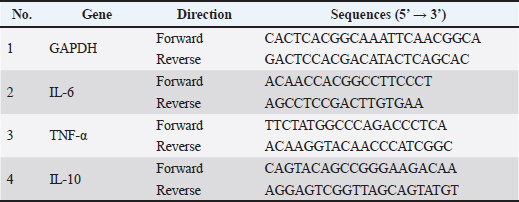
AbstractBackground: Peronema canescens Jack is a botanical with immunostimulant and immunosuppressive properties that is capable of modulating the immune system. Aim: The immunomodulatory properties of fractions from et leaves, specifically the fraction made from ethyl acetate (PcEA), n-butanol solvent (PcB), and water solvent (PcW) solvents, were evaluated in vitro and then characterized using Fourier transform infrared. Gas chromatography-mass spectrometry (GC-MS) and gene expression were used to analyze the fraction with the best immunomodulatory activity. Methods: Peronema canescens was extracted in 96% ethanol. A liquid-liquid partitioning method yielded PcEA, PcB, and PcW. After testing RAW 264.7 cells for immunomodulation, FT-IR characterization was performed. The fractions with the highest immunomodulatory activity metabolites were identified using GC-MS and gene expression profiling. Three replicates were prepared for each parameter assay. Results: All fractions at 3.1 to 50 µg/ml concentrations exhibited no toxicity to RAW 264.7 cells in the viability assay. The phagocytic capacity assay showed that all fractions significantly improved phagocytosis compared with the control group (p < 0.05). Three fractions at 1.56 µg/ml significantly enhanced nitric oxide (NO) release relative to the untreated group (p < 0.05). In contrast, PcEA at 3.13 µg/ml tended to lower NO production than the control (p > 0.05). Tumor necrosis factor-alpha (TNF-α) production revealed that all fractions at all doses significantly increased in comparison to the control group (p < 0.05). Interleukin-6 (IL-6) production of PcEA at 25 and 50 µg/ml concentrations did not differ substantially from the control (p > 0.05). Interleukin-10 (IL-10) production of PcEA at 25 and 50 µg/ml concentrations, and PcB at 12.5 µg/ml, was significantly elevated compared to the control (p < 0.05). The GC-MS analysis of PcEA revealed eight chemicals, including terpenoids, steroids, fatty acids, and siloxane. TNF-α, IL-6, and IL-10 gene expression demonstrate that PcEA modulates the immune system. Conclusion: PcEA has potential as an immunomodulatory agent in vitro. Future research should focus on the isolation and purification of the identified compound and in vivo and mechanistic studies to confirm its therapeutic potential. Keywords: Cytokine, FT-IR analysis, GC-MS analysis, Immunomodulatory, Peronema canescens. IntroductionInfectious diseases and lifestyle-related stress can weaken the immune system, thereby increasing the risk of immune-related disorders. Chemical compounds known as immunomodulators help regulate immune responses (Sujono et al., 2021) . Biological or synthetic immunomodulators function by stimulating, inhibiting, or modulating various body defense mechanisms, including innate and adaptive immunity. In addition to pharmaceutical agents, natural sources, such as phytochemicals or secondary metabolites found in plants, can also have immunomodulatory effects (Bunga et al., 2024). Peronema canescens Jack., commonly known as the Sungkai plant, is one such plant with recognized immunomodulatory potential (Dillasamola et al., 2021). This plant is predominantly found in West and South Sumatra, Jambi, Bengkulu, West Java, and Kalimantan (Noviarni, 2023). Traditionally, the leaves of P. canescens have been used by Indonesian communities to treat wounds, coughs, colds, fevers, and intestinal worms and as a mouthwash for relieving toothaches and sore throats (Dillasamola et al., 2023). The traditional use of boiled Sungkai or P. canescens leaf water as a mouthwash for toothaches and sore throats, and its use in the treatment of wounds, indicates that the plant possesses anti-inflammatory properties. Latief et al. (2021) indicated that 15% extract of P. canescens leaves exhibited up to 50% inhibition of inflammation. Similarly, Versita and Setiani (2023) demonstrated that ethanol extracts of P. canescens leaves at concentrations of 2%, 4%, and 6% showed anti-inflammatory activity in male rabbits (Oryctolagus cuniculus), as reflected by average reductions in burn wound size of 64%, 66%, and 78.67%, respectively, by the eighth day of treatment. Tarigan et al. (2022) found that the ethanol fraction of P. canescens leaves contains apigenin and squalene compounds. Both isolated compounds demonstrated anti-inflammatory activity, with inhibition rates of 58.12% and 56.59% for apigenin and squalene, respectively. Several studies have also highlighted the immunostimulatory potential of P. canescens leaf extract beyond its anti-inflammatory effects. Dillasamola et al. (2021) showed the immunostimulatory potential of P. canescens leaf extract through enhanced macrophage activity and phagocytic capacity, increased total leukocyte counts and lymphocyte percentages, decreased segmented neutrophil counts, and boosted the release of cytokines, including interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) in RAW 264.7 cell cultures. Murine macrophage-like cells, known as RAW 264.7 cells, are frequently used as models for the in vitro assessment of immunomodulatory activity. Dillasamola et al. (2022) revealed that administration of P. canescens leaf fractions prepared with n-butanol solvent (PcB) increased the concentration of natural killer (NK) cells, and the water fraction increased the levels of CD8+ T-cell proteins in the serum of male white mice exposed to the SARS-CoV-2 virus antigen. Some of the findings above indicate that P. canescens extracts and fractions possess properties that influence the immune response by modulating cytokine release and regulating immune cell responses, including those of macrophages, T cells, and leukocytes. In comparison to other solvent-based extracts and fractions, the maximum levels of total phenolic and flavonoid compounds were found in P. canescens leaf fractions prepared using ethyl acetate (PcEA), n-butanol, and water solvents (PcWs), as demonstrated by our previous research. These fractions also exhibited vigorous antioxidant activity. Further analysis by liquid chromatography/high-resolution mass spectrometry identified a diverse array of phenolic and flavonoid compounds in the extracts and fractions, including glycitein, diosmetin, rutin, isoprunetin 7-O-glucose, quercetin 3,7-dimethyl ether, 4′,5,7-trihydroxy-3-methoxyflavone-7-O-β-D-glucopyranoside, pinoquercetin, tricin 7-O-glucose, naringenin 7-O-β-D-glucose, and acteoside (Suryani et al., 2025). Flavonoids can modulate inflammation markers and reduce the production of the proinflammatory mediator IL-6 by regulating the mitogen-activated protein kinase (MAPK) and nuclear factor-κB (NF-κB) signaling pathways (Spagnuolo et al., 2018). The immunomodulatory roles of plant-derived metabolites have been widely reported, acting through various mechanisms, such as the modulation of macrophage, T cell, B cell, neutrophil, and mast cell activities, as well as cytokine production (Behl et al., 2021). Building on this foundation, we hypothesized that distinct solvent fractions of P. canescens leaves differentially modulate macrophage immune responses via cytokine and nitric oxide (NO) regulation. The immunomodulatory properties of PcEA, n-butanol, and water fractions from P. canescens leaves were evaluated using RAW 264.7 cell cultures. Characterization of functional groups via Fourier transform infrared (FT-IR) spectroscopy complemented this evaluation. Furthermore, for the fraction exhibiting the best immunomodulatory activity, metabolite compounds will be identified using gas chromatography-mass spectrometry (GC-MS) and gene expression analysis to provide comprehensive scientific information about P. canescens and support its use as an herbal preparation for immune system enhancement. Materials and MethodsRaw materials, chemicals, and instrumentsThe P. canescens leaves were obtained from Bengkulu Province, Indonesia. Sigma-Aldrich (Germany) was the source of 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT), Dulbecco’s Modified Eagle Medium (DMEM), antibiotic antimycotic solution, trypsin-Ethylenediaminetetraacetic acid solution, fetal bovine serum (FBS), lipopolysaccharide (LPS), neutral red solution, and phosphate-buffered saline (PBS). The Griess Reagent System was purchased from Promega Corporation, and RAW 264.7 murine macrophage cells were purchased from ATCC (ATCC TIB-71). An enzyme-linked immunosorbent assay reagent for mouse IL-6, mouse interleukin-10 (IL-10), and TNF-α was provided by Elabsciences Biotechnology Co., Ltd., Wuhan, China. All other compounds and solvents used in this study were procured from Sigma-Aldrich, Germany. Vertex 80 (Bruker, Germany) was used to conduct FT-IR analysis, and a GC-MS instrument (Thermo Fisher Scientific, USA) was used to conduct GC-MS analysis. Preparation of fractions from P. canescens leaf extractThe solids-to-solvent ratio of 96% ethanol was 1:6 (w/v), and a 90-minute extraction duration was employed to extract P. canescens using an ultrasonic-assisted extraction method. The extraction technique was conducted three times at ambient temperature, as described by Suryani et al. (2025). After acquiring the freeze-dried extract, liquid-liquid fractionation was conducted in accordance with Saake et al. (2025a, 2025b) methodology. The fractionation process produced three different fractions: the first fraction, soluble in PcEA, the second fraction with PcB, and the third fraction with PcW. The percent yield of each freeze-dried fraction was calculated after freeze-drying. The PcEA, PcB, and PcW yields were 8.13%, 18.94%, and 5%, respectively. Preparation of the RAW 264.7 cell lineThe RAW 264.7 cell line was cultured at 1 × 105 cells/ml in a DMEM solution enriched with 10% FBS and 1% antibiotic antimycotic solution. The prepared cell mixture was incubated at 37°C in a 5% carbon dioxide (CO2) atmosphere until 70%–80% confluence was attained. The growth media were routinely exchanged every 2–3 days. Subsequently, the cells were washed and harvested using trypsin-EDTA solution. Cells were cultured according to the protocols described by Hossen et al. (2023). Viability/cytotoxicity assay of RAW 264.7 cellsThe cytotoxic effect of the P. canescens leaf fraction on RAW 264.7 cells was assessed using the MTT assay. Cells were cultivated during the logarithmic phase to perform the assay. A 200 µl suspension of cell cultures with a concentration of 1 × 105 cells/ml was pipetted into 96-well plates and incubated at 37°C with 5% CO2 for 24 hours. All of the cell culture mediums were discarded, and cells were treated with 0 (control), 3.1, 6.25, 12.5, 25, and 50 μg/ml of fractions from P. canescens leaves (three repetitions) and incubated at 37°C for 24 hours. Each well was incubated with 20 ml of MTT solution (5 mg/ml) at 37°C for 4 hours. Dimethyl sulfoxide (DMSO) was then added to the plate and mixed for 10 minutes. The Multiskan Go microplate reader, manufactured by Thermo ScientificTM, was employed to assess absorbance values at an effective wavelength of 450 nm. Cell viability was measured as the percentage of treated cells relative to the untreated control cells (Park et al., 2018). Phagocytic capacity assay of RAW 264.7 cellsThe phagocytic ability of RAW 264.7 cells was evaluated using the modification technique described by Lee et al. (2022). Cultivated RAW 264.7 cells were harvested during the logarithmic growth phase, resulting in a cell suspension with a concentration of 5 × 105 cells/ml. Subsequently, the suspension was placed into 96-well plates, with each well containing a volume of 200 μl, and placed in an incubator at 37°C with 5% CO2 for 24 hours. Different concentrations of P. canescens leaf fractions, including 1.56, 3.13, 6.25, 12.5, and 25 µg/ml, were prepared from an initial stock solution with a concentration of 100,000 µg/ml in DMSO. The serial concentrations were obtained by stepwise dilution, in which each subsequent concentration was prepared by mixing half of the previous solution with an equal volume of culture medium. These working concentrations were then dispensed onto 96-well plates containing LPS-free cell culture media. Cell cultures supplemented with 1 μg/ml LPS were used as a positive control. Meanwhile, the control medium consisted of the cell culture maintained in fraction-free medium. After fraction treatment, the cells were incubated for 24 hours. The supernatant was discarded after the incubation process, and the plate was rinsed three times with PBS. After washing, 100 µl of a 0.1% neutral red solution was added, followed by another hour of incubation. The neutral red, which was not phagocytosed by RAW 264.7 cells, was eliminated by rinsing the 96-well plate three times with PBS. Two hundred microliters of cell lysis solution were added, and the plate was incubated for 2 hours. Following the incubation period, the amount of absorption was determined by employing the Multiskan Go equipment manufactured by Thermo ScientificTM at a wavelength of 540 nm. Analysis of NO production in RAW 264.7 cellsRAW 264.7 cells were cultivated in 24-well plates at a density of 1 × 105 cells per well and then incubated for 24 hours. Several P. canescens fraction samples were added to the plates. These concentrations included 1.56, 3.13, 6.25, 12.5, and 25 μg/ml. Additionally, LPS was administered at doses of 1.25 and 2.5 μg/ml to serve as positive controls. The control sample consisted of complete medium. Subsequently, the cells were grown for 24 hours in a 5% CO2 incubator at 37°C. After collecting the supernatant from the culture medium, it was centrifuged for 10 minutes at a speed of 1,500 rpm at a temperature of 4°C. Next, 100 ml of the centrifuged supernatant was dispensed into a 96-well plate, and 100 μl of Griess reagent was added. After incubation at room temperature for 10 minutes and protection from light, the absorbance of the 96-well plates was measured at 540 nm using a microplate reader. NO production by macrophages was determined using the standard curve obtained with sodium nitrite (Park et al., 2018). Analysis of cytokine production in RAW 264.7 cellsRAW 264.7 cells were cultured at a concentration of 5 × 105 cells/mL at 37°C for 20 hours with varying doses of P. canescens fraction samples, which included 12.5, 25, and 50 µg/ml, while LPS (1 and 10 µg/ml) was used separately as a positive control to stimulate cytokine production. The supernatants were then collected, and cytokine levels (TNF-α, IL-6, IL-10) were measured using enzyme-linked immunosorbent assay kits according to the manufacturer’s instructions (Zhou et al., 2021). FT-IR spectral analysisThe functional groups of molecules in the P. canescens fraction samples were identified using an attenuated total reflectance (ATR)-FT-IR spectrophotometer (Indrianingsih et al., 2019). The sample was positioned on an ATR crystal and analyzed within the wavelength range of 4,000–600 cm−1 to determine the sample spectrum. GC-MS analysis of the P. canescens fraction in PcEAA 1-mg sample of PcEA was solubilized in the same amount of solvent. After passing through a syringe filter, the sample solution was subsequently placed into a 1.5 ml GC-MS vial. The prepared sample is then injected into the GC-MS instrument. The sample solution was analyzed using GC-MS (ThermoScientific GC Trace 1300 coupled with ISQ 7000 and TriPlus RSH, USA) along with a TG-5MS capillary column (30 m × 0.25 mm and 0.25 μm film thickness). The analysis started at 50°C for 5 minutes, then increased to 80°C for 10 minutes and held at 2°C/min. Helium served as the transport gas, maintaining a steady flow rate of 1.00 ml/min. A volume of 1 μl of the sample was injected into the column at a temperature of 110°C using a split ratio of 20. The ion source and mass spectrometer transfer line temperatures were kept at 300°C and 240°C, respectively. The acquisition rate for the mass spectral data was two samples per second, with an m/z ratio ranging from 50 to 550 amu. Chemical compounds in the PcEA samples were identified by comparing the spectra and retention times of each compound with those of the validated reference compounds documented in the mass spectral database (Sari et al., 2023). Gene expression analysisThis study examined gene expression in RAW 264.7 cell lines exposed to PcEA fractions derived from P. canescens leaves. Following the cultivation of RAW 264.7 cells (1 × 106 cells/well) in the flask for 24 hours, the culture medium was switched to a medium that contained 1 µg/ml LPS or PcEA samples at 25 µg/ml at a concentration and incubated for 6 hours. Cellular RNA was extracted from PcEA-treated RAW 264.7 cells using an RNA extraction kit provided by Promega Corporation, USA. Gene expression analysis was conducted using the Real-Time Cycler Rotor-Gene Q 5 Plex Hrm System (Qiagen) and a one-step RT-qPCR system (Promega, USA) in accordance with the manufacturer’s guidelines. The primers were implemented following Liu et al. (2021). Table 1 displays the primer oligonucleotide sequences employed. The RNA was converted into cDNA (Promega Corporation, USA) at 37°C for 15 minutes, followed by inactivation at 95°C for 10 minutes. The cDNA was amplified for 35 cycles consisting of denaturation at 95°C for 10 seconds, annealing at 55°C for 30 seconds, and extension at 72°C for 30 seconds. All reactions were performed three times. The relative mRNA levels of TNF-α, IL-6, and IL-10 were adjusted to those of Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) genes, and relative expression was determined using the 2-ΔΔCq method (Liu et al., 2021), where Ct values were determined after normalization to β-actin. Table 1. The primer sequences used in the RT-qPCR analysis.
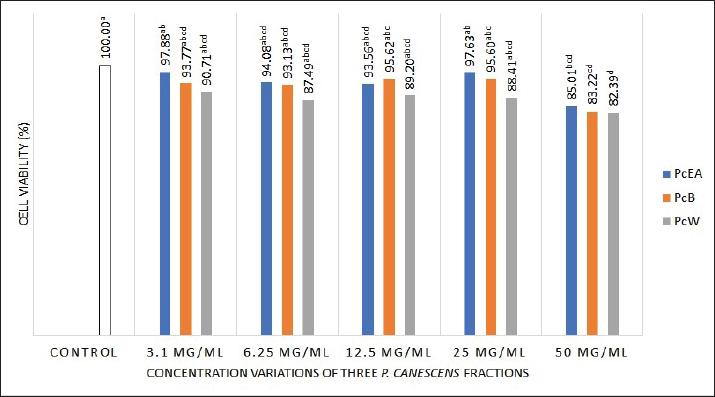
Statistical analysisThe mean ± SD is used to express the data. Significant differences between the treatment and control groups were evaluated using one-way analysis of variance and the post hoc Duncan test, employing SPSS 20.0 software. Each experiment consisted of three replicates. A p value of 0.05 indicates statistical significance (Bewick et al., 2004). The FT-IR spectra were interpreted and analyzed using OPUS 8.5 software (Bruker, Germany). Ethical approvalNot needed for this study. ResultsEffect of P. canescens leaf fractions on RAW 264.7 cell viabilityThe present study demonstrated that the viability of normal control group cells was set at 100%, demonstrating the absence of dead cells (no toxic effects). Following treatment of RAW 264.7 cells with three P. canescens fractions at dosages of 3.1, 6.25, 12.5, 25, and 50 µg/ml, these cells exhibited viability ranging from 97.88% to 82.39 % (Fig. 1).
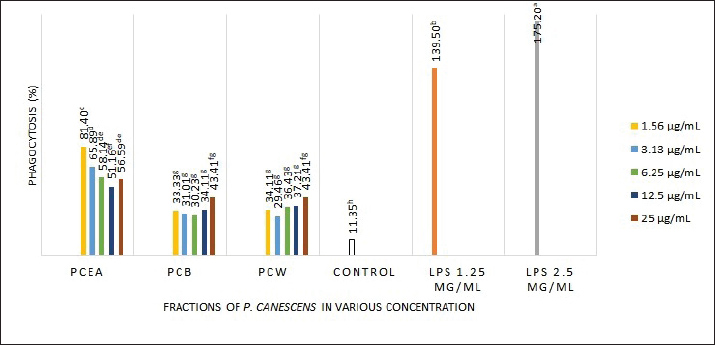
Fig. 1. Effect of three P. canescens leaf fractions at concentrations of 3.1, 6.25, 12.5, 25, and 50 µg/ml on RAW 264.7 cell viability. A distinct superscript in each bar (p < 0.05) indicates a significant difference between the means. Effect of P. canescens fractions on the phagocytic capacity of RAW 264.7 cellsThe RAW 264.7 cells treated with LPS at both concentrations, 1.25 and 2.5 µg/ml, showed significantly higher phagocytosis percentages (139.50% ± 24.43% and 175.20% ± 16.28%) compared to the control group (11.35% ± 0.46%, p < 0.05). RAW 264.7 cells treated with PcEA at concentrations from 1.56 to 25 µg/ml showed phagocytosis capacities ranging from 50.39% ± 17.61% to 81.40% ± 4.84%. RAW 264.7 cells treated with PcB showed phagocytosis rates ranging from 30.23% ± 1.34% to 43.41% ± 4.84%, while PcW treatment resulted in rates ranging from 29.46% ± 6.71% to 43.41% ± 1.93%. Figure 2 demonstrates the effect of P. canescens leaf fractions on the phagocytic activity of RAW 264.7 cells.
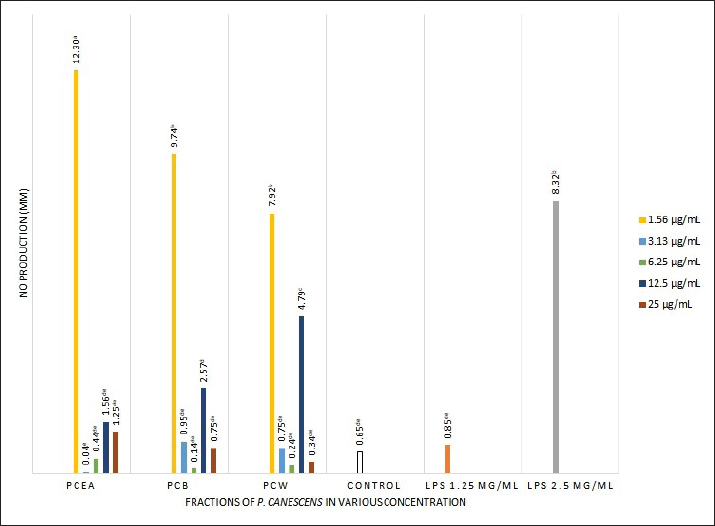
Fig. 2. Effect of three P. canescens leaf fractions at concentrations of 3.1, 6.25, 12.5, 25, and 50 µg/ml on RAW264.7 cell phagocytic capacity. A distinct superscript in each bar (p < 0.05) indicates a significant difference between the means. Effect of P. canescens fractions on RAW 264.7 cell NO productionIn this study, LPS induction at a concentration of 2.5 µg/ml in RAW 264.7 cells significantly increased NO production by almost 13 times compared with the untreated cells (p < 0.05). NO production was significantly higher when RAW 264.7 cells were incubated with the three P. canescens fractions at 1.56 µg/ml compared with the control group (p < 0.05). Notably, following treatment with 1.56 µg/ml of PcEA fraction, NO production was higher than that in the group exposed to LPS of 2.5 µg/ml. Conversely, a contrary outcome was observed in RAW 264.7 cells, which were incubated with PcEA at a dosage of 3.13 µg/ml, where NO production was considerably lower than that of the control group. However, this difference was not significant (p > 0.05) (Fig. 3). These findings demonstrate that the concentration of the samples significantly affects the NO production from RAW 264.7 cells treated with the three P. canescens fractions.
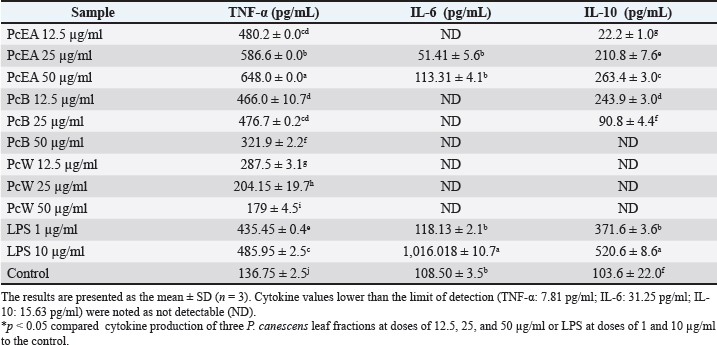
Fig. 3. Effect of three P. canescens leaf fractions at concentrations of 3.1, 6.25, 12.5, 25, and 50 µg/ml on NO production in RAW 264.7 cells. A distinct superscript in each bar (p < 0.05) indicates a significant difference between the means. Effect of P. canescens fractions on RAW 264.7 cell cytokine productionThe impacts of RAW 264.7 cells exposed to PcEA, PcB, and PcW at the concentrations of 12.5, 25, and 50 µg/ml on cytokine production, including TNF-α, IL-6, and IL-10, are shown in Table 2. Table 2 shows that the cultures of RAW 264.7 cells treated with LPS at dosages of 1.25 and 2.5 µg/ml, as well as those incubated with all three P. canescens fractions at concentrations ranging from 12.5, 25, and 50 µg/ml, markedly enhanced the release of the cytokine TNF-α in comparison to the control group (p < 0.05). Meanwhile, the release of IL-6 cytokines by RAW 264.7 cells treated with PcEA at 25 and 50 µg/ml dosages, along with the group stimulated by LPS at 1 µg/ml, showed no significant difference compared with the control cells. Elevated production of the cytokine IL-10 was noted in RAW 264.7 cells treated with PcEA at doses ranging from 12.5 to 50 µg/ml and PcB at concentrations from 12.5 to 25 µg/ml, in addition to RAW 264.7 cells stimulated by 1 and 10 µg/ml LPS. Table 2. Effect of three P. canescens leaf fractions at doses of 12.5, 25, and 50 µg/ml on the production of cytokines in RAW 264.7 cells.
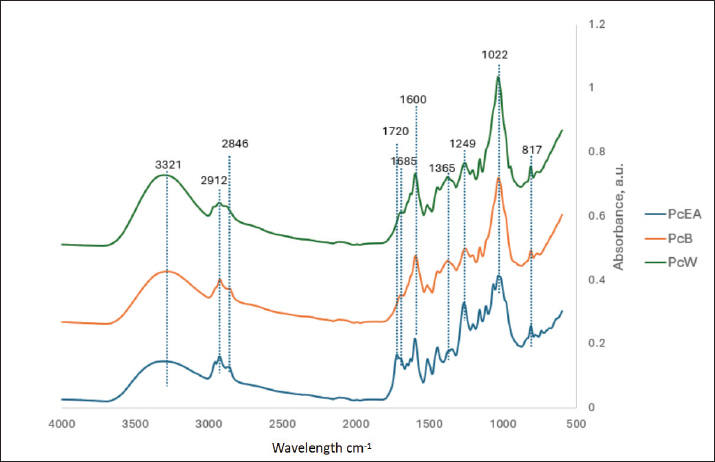
FT-IR spectroscopy analysisThe FT-IR spectra are shown in Figure 4. The three fractions of P. canescens leaf extract exhibited several similar absorption peak patterns, indicating the existence of common functional groups. A broad absorption peak near 3,250–3,475 cm−1 indicates absorption from the hydroxyl group, whereas the absorption peak around 3,321 cm−1 denotes stretch from the secondary amine (NH) group. The appearance of spectral peaks at 2,912 and 2,846 cm−1 indicates the stretching vibrations of the CH group. The carbonyl group (C=O) appears at an absorption peak of 1,720 cm−1. This C=O group is more abundant in the water fraction (PcW), which is characterized by its greater intensity. The peak at 1,685 cm−1 signifies the C=C stretching group, whereas the peak at 1,600 cm−1 denotes the NH (amide) group. Peak 1,022 cm−1 shows that the C-O group is stretching, whereas peak 1,249 cm−1 confirms that the C-O group could be from phenol. The absorbance peak at 1,365 cm−1 indicates CH bending groups from alkanes, while the peak at 817 cm−1 indicates wagging from CH2.
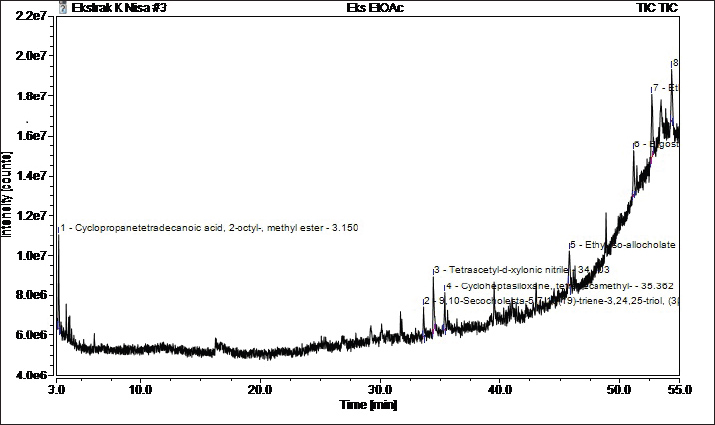
Fig. 4. FT-IR spectra of the three P. canescens leaf fractions. GC-MS analysisIn the present work, the PcEA fraction showed the highest immunomodulatory activity based on the pro-inflammatory and anti-inflammatory cytokine production balance from RAW 264.7 cells incubated in PcEA. Furthermore, the compounds that contributed to the immunomodulatory activity of PcEA were investigated by metabolite analysis using a GC-MS instrument. GC-MS analysis revealed eight compounds in PcEA (Fig. 5 and Table 3).
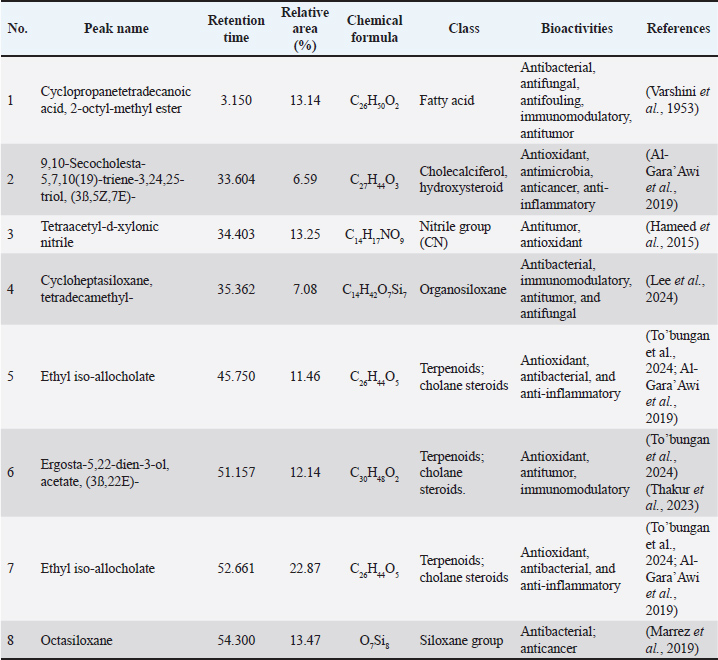
Fig. 5. Chromatogram of the PcEA fraction of P. canescens leaves based on GC-MS. Table 3. Compounds identified from the PcEA fraction of P. canescens leaves based on GC–MS analysis.
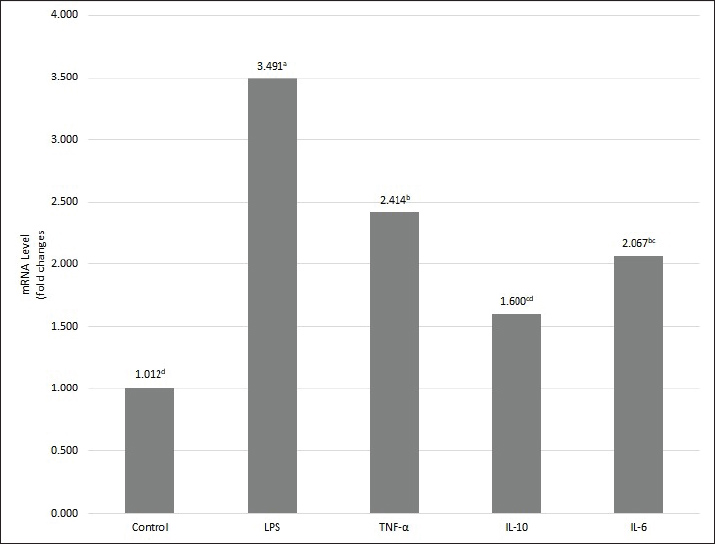
Gene expression analysisCytokine secretion from the cells was measured using a kit for cytokine production. RT-qPCR was employed to assess changes in cytokine mRNA gene expression in RAW 264.7 cells after PcEA treatment. The 6-hour PcEA treatment resulted in a notable increase in the mRNA expression levels of both pro-inflammatory and anti-inflammatory cytokines, particularly IL-6, IL-10, and TNF-α, when contrasted with the control group (Fig. 6). These changes in mRNA expression were consistent with the cytokine production results shown in Table 2. Collectively, these findings further support the immunomodulatory activity of PcEA. Based on previous references, Table 3 shows that several substances found in PcEA have potential immunomodulatory properties.
Fig. 6. The alteration of TNF-α, IL-6, and IL-10 mRNA expression in RAW 264.7 cells after PcEA treatment. The gene expression of TNF-α, IL-6, and IL-10 in RAW 264.7 cells treated with PcEA for 6 hours using Reverse Transcription quantitative Polymerase Chain Reaction analysis. Glyceraldehyde 3-phosphate dehydrogenase was used as an internal control for Reverse Transcription Polymerase Chain Reaction.. Fold changes were calculated from 2^-DELTA DELTA Cq. A distinct superscript in each bar (p < 0.05) indicates a significant difference between the means. DiscussionThis study used RAW 264.7 cell viability assays using the MTT assay method to determine nontoxic concentrations of PcEA, PcB, and PcW in RAW 264.7 cells. The MTT assay is based on the principle that mitochondrial dehydrogenase enzymes convert MTT into purple formazan crystals through the action of metabolically active and viable cells (Stockert et al., 2012). Based on the guidelines for cytotoxicity assays using the MTT method, a decrease in cell viability to below 70% of the control indicates that the cells exhibit cytotoxic potential (ISO, 2009). Pratama et al. (2024) reported that the evaluation of the extract concentration, which produces a percentage of live cells >80%, indicates that the extract is not toxic to RAW 264.7 cells, so further testing is warranted. This study showed 100% cell viability in the untreated control group, indicating ideal cell growth conditions and a safe medium for cellular development. For RAW 264.7 cells treated with PcEA, PcB, and PcW at concentrations of 3.1, 6.25, 12.5, 25, and 50 µg/ml, cell viability ranged from 97.88% to 82.39% (Fig. 1). These results indicate that all three fractions are non-toxic to RAW 264.7 cells and can be used for further evaluation within the tested concentration range. This finding is supported by Hikariastri et al. (2019), who reported that treatment doses resulting in cell viability above 80% are deemed safe for subsequent assays using RAW 264.7 cells. Ryu et al. (2019) investigated the anti-inflammatory properties of various herbal plant extracts using the MTT method and determined that extract concentrations with a RAW 264.7 cell viability percentage exceeding 80% are suitable for use in subsequent assays. Innate immune system cells are the first line of defense against foreign pathogens. The rapid response to antigens is a key factor in strengthening innate immunity. Several phagocytic cells, including macrophages, monocytes, and neutrophils, play crucial roles in the innate immune response. Macrophages are an early and essential step in the immune response (Qian et al., 2023). In this study, the neutral red phagocytosis assay was used to evaluate the activation of the immune response induced by P. canescens fractions. The neutral red assay assesses phagocytic activity by quantifying the uptake of neutral red dye by phagocytic cells. Cells engulf antigens during the initial phase of phagocytosis, a process facilitated by increased membrane fluidity and the activation of various membrane-associated proteins. Membrane fluidity is crucial for phagocytic cells to internalize foreign particles. Upon activation, macrophages also initiate the production of several immunomodulatory molecules, including NO, inducible NO synthase (iNOS), cyclooxygenase-2, and pro-inflammatory cytokines (Lee et al., 2022). In vitro studies using the RAW 264.7 cell line to evaluate the phagocytic activity of P. canescens extract have not been conducted. RAW 264.7 cells are frequently used as macrophage models for evaluating in vitro immunomodulatory activity. RAW 264.7 cells closely resemble primary murine macrophages, which are crucial for macrophage maturation (Riastri et al., 2023). Activated macrophages can eliminate pathogens through direct phagocytosis in the innate response. The neutral red absorption test can be used to evaluate the phagocytic mechanism of RAW 264.7 cells (Teng et al., 2015). Dillasamola et al. (2021) determined the phagocytic activity of macrophages in vivo and demonstrated that white mice treated with P. canescens extract at concentrations of 800, 400, and 200 mg/kg BW exhibited significantly higher phagocytic capacity than the control group, which received 0.5% Na CMC (p < 0.05). In another in vivo study, Latief et al. (2023) demonstrated that the administration of methanol and ethyl acetate extracts of P. canescens significantly increased phagocytic activity compared with the negative control group. The results of both phagocytic in vivo assays corroborate those of the in vitro phagocytic assays in this study. The findings obtained in this study corroborate the information regarding the potential bioactivity of sugai leaves in modulating the immune system. NO production was measured using the Griess reagent assay. This method involves the reaction of nitrite, a stable end-product of NO metabolism, with a diazotization reagent to form a transient diazonium salt. This intermediate then reacts with N-naphthyl-ethylenediamine, a coupling agent, to produce a stable, purple azo compound. The intensity of the resulting color allows for the sensitive and accurate quantification of nitrite levels, thereby serving as an indirect measure of NO production (Paul et al., 2021). NO is a lipophilic gas generated from the arginine substrate by the activity of the enzyme NO synthase (Priyanga et al., 2024). NO is crucial for mediating cellular immune signals and exhibits various biological functions, such as immunomodulation, inflammation, and the eradication of microbes and tumors (Cinelli et al., 2020). Macrophages activated by cytokines or microbial antigens, such as LPS, release several effector molecules that can eradicate microbial antigens, one of which is NO (Shreshtha et al., 2018). However, this multifunctional effector molecule has a dual role. It is advantageous and modulates physiological processes within the body at lower concentrations. Conversely, at elevated doses, it adversely affects microorganisms or tumor cells and may induce harmful effects on host cells (Ahmad et al., 2021). The induction of LPS at a concentration of 2.5 µg/ml in RAW 264.7 cells led to a significant increase in NO production compared with the control group (p < 0.05). The incubation of RAW 264.7 cells with P. canescens leaf fraction samples at a concentration of 1.56 µg/ml resulted in a significant increase in NO production compared with the control group (p < 0.05). Conversely, treatment with fractions at dosages ranging from 3.13 to 6.25 µg/ml resulted in the suppression of NO production, leading to levels that were lower or comparable to those of the control group (p > 0.05) (Fig. 9). The findings of this study indicate that dosage and concentration significantly affect NO production in RAW 264.7 cells treated with all P. canescens leaf fractions. Overall, the NO production analysis showed that Sungkai leaf fractions at a 1.56 µg/ml concentration have immunostimulatory activity. Several previous studies have reported the in vivo immunostimulatory activity of sungkai leaves. In their study, Dillasamola et al. (2021) stated that ethanol extract of sungkai leaves at doses of 800, 400, and 200 mg/kg BW has potential as an immunostimulant by increasing the phagocytic activity of peritoneal macrophage cells, the number of leukocytes, and the percentage of lymphocytes, and reducing segmental neutrophil cells in vivo in white mice, as well as increasing the levels of pro-inflammatory cytokines (TNF-α and IL-6), and is non-toxic to RAW 264.7 cells. Dillasamola et al. (2022) also demonstrated that white mice treated with a butanol fraction at a dose of 1 mg/kg BW experienced an increase in the concentration of NK cells, whereas those treated with a water fraction at a dose of 100 mg/kg BW experienced an increase in the concentration of CD8+ T cells. NO production is closely related to macrophages’ phagocytic function against various pathogens. Therefore, increased NO production can induce immunostimulatory activity in macrophages (Abbas et al., 2022). Cytokines are a substantial category of secreted proteins characterized by varied structures and functions. They regulate and coordinate numerous immune cell functions, encompassing both innate and adaptive immune systems. All immune cells secrete at least some cytokines and express specific receptors for certain cytokines. The initial reaction of the innate immune system to infection and damaged tissues is the release of cytokines by cells in the damaged area, which are essential for the acute inflammatory response. TNF-α, IL-1, and IL-6 are important proinflammatory mediators in the innate immune system (Abbas et al., 2022). Although various inflammatory mediators exist, evidence from various sources indicates that IL-10 functions as an anti-inflammatory cytokine, whereas IL-6 and TNF-α serve as pro-inflammatory cytokines. All of them play an important role in producing a balanced immune response (Lim et al., 2021). TNF-α is a cytokine that stimulates macrophages and other phagocytic cells to produce additional cytokines, including IL-1 and IL-6, that trigger inflammation (Abbas et al., 2022). Besides, TNF-α also has the ability to attenuate the immune response by inhibiting T lymphocyte receptor signaling, enhancing the death of lymphoid T cells, suppressing dendritic cell co-stimulation, and promoting other cytokines that can decrease immunological mediation, including IL-10 and TGF-β (Smith, 2018). The measurement results of the cytokine release of TNF-α in this work correspond with the results obtained by Dillasamola et al. (2021), demonstrating that P. canescens leaf extract significantly boosts TNF-α release in RAW 264.7 cells relative to the control group of cells. The findings indicate that the metabolites in PcEA enhance immune response activity by stimulating cytokine production that promotes inflammatory activity (Ashaolu et al., 2024). This study reveals a synergistic pathway for IL-6 and IL-10 cytokine production, demonstrating the suppression of inflammation in RAW 264.7 cells that receive PcEA at dosages of 25 and 50 µg/ml. Cytokine production indicates that the compounds in PcEA inhibit the production of proinflammatory IL-6 cytokines. Inhibition of IL-6 production is an effect linked to high IL-10 cytokine production. All stimuli that trigger pro-inflammatory activation in immunocompetent cells concurrently enhance the production of IL-10, thereby inhibiting the synthesis of cytokines that stimulate inflammation and leading to the cessation of inflammatory activity (Stenvinkel et al., 2005). The two principal functions of IL-10 are to suppress the release of various cytokines, specifically TNF-α, IL-1, and IL-6, and to promote an immunosuppressive or anti-inflammatory state by inhibiting macrophage function in activating T cells. IL-10 suppresses Major Histocompatibility Complex class II expression and reduces the expression of costimulatory molecules on macrophages (Tizard, 2018). IL-10 (18 kD) is synthesized by immunologically active cells and is essential for the control of anti-inflammatory immune responses. IL-10 release is typically delayed, occurring after the secretion of proinflammatory mediators such as TNF and IL-6. This sequential pattern ensures that the initial inflammatory response is strong, while the later release of IL-10 contributes to its gradual resolution over time. In local inflammatory responses, IL-10 may operate in an autocrine or paracrine manner and even act systemically at the endocrine level (Smith, 2018). Many types of studies have investigated the anti-inflammatory potential of P. canescens through in vivo, in vitro, and in silico approaches. Tarigan et al. (2022) reported that the ethanol-soluble fraction of P. canescens contains various flavonoids and steroid compounds with anti-inflammatory properties, exhibiting inhibition rates between 56.59% and 58.12%. In addition, Latief et al. (2021) reported that topical preparations containing 15% ethanol extract of P. canescens leaves exhibited up to 50% anti-inflammatory activity, as evidenced by a reduction in exudate volume, lymphocytes, band neutrophils, and segmented neutrophils. Rahardhian et al. (2022) conducted an insilico analysis showing that Peronemin C1 has better molecular docking performance compared to six other compounds against IL-6 and TNF-α, as reflected by its binding affinity, inhibition constant, and interactions with protein amino acids, indicating its potential as an effective inhibitor. FT-IR spectroscopy is commonly used to identify functional groups in plant extracts and analyze their composition without damaging the sample (Sravan Kumar et al., 2015). This study conducted FT-IR analysis of P. canescens leaf fractions obtained from three types of organic solvents to determine the functional groups of chemical compounds in each fraction, as per previous research (Noviana et al., 2022). Although there was no significant difference in the FT-IR spectra obtained from the three fractions of P. canescens, the FT-IR spectra could confirm the presence of major functional groups because they serve as a fundamental fingerprint analysis of the extract’s chemical composition. The FT-IR spectra obtained in this study agree with previous findings on ethanol extracts of P. canescens leaves (Maigoda et al., 2022). The absorption peaks observed in the FT-IR spectra of the three P. canescens leaf extract fractions indicate the presence of phenolic compounds. Previous studies have reported that P. canescens leaves contain a variety of bioactive constituents, including phenolics, flavonoids, terpenoids, alkaloids, tannins, and saponins (Shalihin et al., 2024). According to Suryani et al. (2025), the PcEA, n-butanol, and water fractions of P. canescens leaf extracts exhibited the highest total phenolic and flavonoid contents compared with ethanol extracts and fractions obtained with other organic solvents. These findings are consistent with the FT-IR-based functional group analysis conducted in this study. Metabolite compound analysis by GC-MS showed eight compounds were successfully identified from PcEA, including: Ethyl iso-allocholate and ergosta-5,22-dien-3-ol acetate (3ß,22E), which were identified as terpenoid compounds (To’bungan et al., 2024; Al-Gara’Awi et al., 2019); cycloheptasiloxane tetradecamethyl- and octasiloxane are included in the siloxane group (Rasyid et al., 2023; Lee et al., 2024). Cyclopropanetetradecanoic acid 2-octyl-methyl ester is included in the fatty acid group; 9,10-Secocholesta-5,7,10(19)-triene-3,24,25-triol, (3ß,5Z,7E)- is classified as a steroid, and Tetraacetyl-d-xylonate nitrile is categorized as a nitrile (Hameed et al., 2015). The active compounds derived from the P. canescens plant were estimated to be ethyl iso-alchocolate, ergosta-5,22-dien-3-ol acetate (3ß,22E)-, cyclopropanetetradecanoic acid 2-octyl-methyl ester, 9,10-secocholesta-5,7,10(19)-triene-3,24,25-triol, (3ß,5Z,7E)-, and tetraacetyl-d-xylonate nitrile. Meanwhile, cyclopentasiloxane tetradecamethyl- and octasiloxane may contaminate the GC-MS results (Chainet et al., 2011). The appearance of contaminant peaks from the siloxanes group in the GC system can be attributed to contaminants in dirty injectors, carrier gases, extractable contaminants, autosampler vials and caps, or pollutants carried over from previous injections (Kumar and Sharma, 2022). Juswardi et al. (2023) reported that ethyl iso-allocholate is a compound in young and mature leaf extracts of P. canescens based on GC-MS analysis. Ergosta-5,22-dien-3-ol acetate (3ß,22E)- is a natural compound from the Hyptis capitata plant (To’bungan et al., 2024), which belongs to the Lamiaceae family and is a plant in the same family as P. canescens. Based on the metabolite profiling in PcEA, several compounds that interact synergistically might influence immunomodulatory activity, thereby strengthening the immunomodulatory effect. In previous preclinical studies, Renda et al. (2022) reported that terpenoid compounds possess a wide range of pharmacological properties, including antioxidant, anti-inflammatory, anti-atherosclerotic, antiviral, anticancer, hepatoprotective, and immunomodulatory effects. Similarly, Patel and Savjani (2015) highlighted that plant-derived steroids exhibit diverse biological activities, such as anticancer, antitumor, antibacterial, immunosuppressive, hepatoprotective, cytotoxic, anthelmintic, and cardiotonic effects. PcEA may exert its immunomodulatory effects by modulating the levels of inflammation-promoting cytokines and those that inhibit inflammation, which are crucial for defending against intracellular pathogens (Geum et al., 2021). Proinflammatory cytokines encourage immune cell recruitment and activation, whereas anti-inflammatory cytokines suppress inflammation and support tissue repair (Al-Qahtani et al., 2024). The production of these cytokines is modulated through various signaling pathways, such as phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT), NF-κB, and mitogen-activated protein kinases. For instance, polysaccharides derived from Ophiocordyceps sinensis mycelia modulate cytokine gene expression by activating the MAPK and PI3K/AKT pathways (Liu et al., 2021). Similarly, sprouts of bok choy (Brassica campestris var. chinensis) induce the production of NO, iNOS, IL-1β, IL-6, IL-12, TNF-α, and MCP-1 via the TLR2-mediated MAPK, NF-κB, and PI3K/AKT pathways (Geum et al., 2021). Bok choy is a vegetable rich in phenols, flavonoids, and terpenoids (Dong et al., 2024). These compounds are also present in P. canescens. Ergosterol, a derivative also detected in the PcEA fraction (Table 2), showed an immune response by MAPK phosphorylation (Saake et al., 2025). PcEA may influence pro- and anti-inflammatory cytokine regulation through similar signaling mechanisms. However, further studies are required to clarify the specific pathways responsible for PcEA immunomodulatory activities. ConclusionOur findings indicate that the fraction of PcEA from P. canescens leaves exhibits immunomodulatory activity in vitro by modulating macrophage phagocytosis, NO, and cytokine production. Therefore, future research should focus on the isolation and purification of the identified compound and further in vivo and mechanistic studies to confirm its therapeutic potential. AcknowledgmentThe authors express their gratitude to the Research Center for Food Technology and Processing-National Research and Innovation Agency, for the provision of facilities and collaborative efforts among the research group division. Conflict of interestWe officially confirm that there are no conflicts of interest arising from any financial, personal, or other affiliations with individuals or organizations that relate to the topics presented in the journal. FundingThe National Research and Innovation Agency (BRIN) financially supported this research through the RP-ORPP Project. Authors' contributionsThe study was designed by AES, KN, YN, and HW. Laboratory work was conducted by AES, KN, SaH, SH, and WNMN. The data were analyzed using AES, SH, KN, and AWI. AES, HW, KN, SH, and AWI wrote the manuscript. All authors have reviewed and approved the final version of the manuscript. Data availabilityThe data supporting the results and conclusions of this study are available from the corresponding author upon request. ReferencesAbbas, A.K., Lichtman, A.H., and Pillai, S. 2022. Cellular And Molecular Immunology, 10th Edition. Antimicrob. Agents Chemother. 58. Ahmad, T., Eapen, M.S., Ishaq, M., Park, A.Y., Karpiniec, S.S., Stringer, D.N., Sohal, S.S., Fitton, J.H., Guven, N., Caruso, V. and Eri, R. 2021. Anti-inflammatory activity of fucoidan extracts in vitro. Mar. Drugs 19, 702. doi:10.3390/md19120702 Al-Gara, awi, N.I., Abu-Serag, N.A., Alee Shaheed, K.A. and Bahadly, Z.K.A. 2019, Analysis of bioactive phytochemical compound of (Cyperus alternifolius L.) by using gas chromatography–mass spectrometry. In IOP conference series: Materials science and engineering, 571, 012047. IOP Publishing Ltd. England and Wales, United Kingdom. Al-Qahtani, A.A., Alhamlan, F.S. and Al-Qahtani, A.A. 2024. Pro-Inflammatory and Anti-Inflammatory Interleukins in Infectious Diseases: a Comprehensive Review. Trop. Med. Infect. Dis. , 9(1), 13. doi: 10.3390/tropicalmed9010013 Ashaolu, T.J., Zarei, M., Agrawal, H., Kharazmi, M.S. and Jafari, S.M. 2024. A critical review on immunomodulatory peptides from plant sources; action mechanisms and recent advances. Crit. Rev. Food Sci. Nutr., 64(20), 7220–7236 doi:10.1080/10408398.2023.2183380 Behl, T., Kumar, K., Brisc, C., Rus, M., Nistor-Cseppento, D.C., Bustea, C., Aron, R.A.C., Pantis, C., Zengin, G., Sehgal, A., Kaur, R., Kumar, A., Arora, S., Setia, D., Chandel, D. and Bungau, S. 2021. Exploring the multifocal role of phytochemicals as immunomodulators. Biomed. Pharmacother. , 133, 110959. doi:10.1016/j.biopha.2020.110959 Bewick, V., Cheek, L. and Ball, J. 2004. Statistics review 9: one-way analysis of variance. Crit. Care 9, 130–136. doi:10.1186/cc2836 Bunga, C.D., Haresmita, P.P., Rianawati, L., Nurkhayati, N., Azizah, B.R., Latifah, E. and Hermawansyah, A. 2024. Aktivitas Fagositosis Ekstrak Etil Asetat Daun Binahong dari Borobudur untuk Imunomodulator Pandemi COVID-19. J. Mandala Pharmacon Indones. 10, 707–714. Chainet, F., Courtiade, M., Lienemann, C.P., Ponthus, J. and Donard, O.F.X. 2011. Silicon speciation by gas chromatography coupled to mass spectrometry in gasolines. J. Chromatogr. A. 1218, 9269–9278. doi:10.1016/j.chroma.2011.10.047 Cinelli, M.A., Do, H.T., Miley, G.P. and Silverman, R.B. 2020. Inducible nitric oxide synthase: regulation, structure, and inhibition. Med. Res. Rev., 40(1), 158–189. doi:10.1002/med.21599 Dillasamola, D., Aldi, Y., Wahyuni, F.S., Rita, R.S., Dachriyanus D, D., Umar, S. and Rivai, H. 2021. Study of Sungkai (Peronema canescens, Jack) leaf extract activity as an immunostimulators with in vivo and in vitro methods. Pharmacogn. J. 13, 1397–1407; doi:10.5530/PJ.2021.13.177 Dillasamola, D., Wahyuni, F.S., Rita, R.S. and Aldi, Y. 2023. Immunostimulating Activity of Sungkai Leaf Stigmasterol Isolate against Cluster Differentiated 8+ T (CD8+T) from Covid 19 Disease Exposure. Pharmacogn. J. 15, 145; doi:10.5530/pj.2023.15.145 Dillasamola, D., Wahyuni, F.S., Rita, R.S., Dachriyanus, D., Alen, Y., Umar, S. and Aldi, Y. 2022. Immunostimulating Study of Active Agent Fraction from Sungkai (Peronema canescens Jack.) Leaf from SARS-COV-2 Virus Antigen Exposure to NK and CD8+T Cells. Pharmacogn. J. 14, 344–351. doi:10.5530/pj.2022.14.105 Dong, S., Fang, S., Li, J., Zheng, W., Wang, Z., Hu, J., Zhao, X., Liu, Z., Feng, H. and Zhang, Y. 2024. Comparative metabolic profiling of different pakchoi cultivars reveals nutritional diversity via widely targeted metabolomics. Food Chem. X 22, 101379 doi: 10.1016/j.fochx.2024.101379 Geum, N.G., Yeo, J.H., Yu, J.H., Choi, M.Y., Lee, J.W., Baek, J.K. and Jeong, J.B. 2021. In vitro Immunostimulatory Activity of Bok Choy (Brassica campestris var. chinensis) Sprouts in RAW264.7 Macrophage Cells. Korean. J. Plant. Resour. 34, 203–215. Hameed, I.H., Hussein, H.J., Kareem, M.A. and Hamad, N.S. 2015. Identification of five newly described bioactive chemical compounds in Methanolic extract of Mentha viridis by using gas chromatography – mass spectrometry (GC-MS). J. Pharmacogn. Phyther. 7, 107–125. doi:10.5897/JPP2015.0349 Hikariastri, P., Winarno, H., Kusmardi, K., Laksmitawati, D.R. and Abdillah, S. 2019. Aktivitas Antiinflamasi Crude Extract Fukoidan dari Sargassum crassifolium pada Sel RAW 264.7 yang Diinduksi LPS. J. Kefarmasian. Indones. , 9(2), 97–105. doi:10.22435/jki.v9i2.1547 Hossen, I., Kaiqi, Z., Hua, W., Junsong, X., Mingquan, H. and Yanping, C. 2023. Epigallocatechin gallate (EGCG) inhibits lipopolysaccharide-induced inflammation in RAW 264.7 macrophage cells via modulating nuclear factor kappa-light-chain enhancer of activated B cells (NF-κB) signaling pathway. Food Sci. Nutr. 11, 4634–4650. doi:10.1002/fsn3.3427 Indrianingsih, A.W., Apriyana, W., Nisa, K., Rosyida, V.T., Hayati, S.N., Darsih, C. and Kusumaningrum, A. 2019. Antiradical activity and physico-chemical analysis of crackers from Cucurbita moschata and modified cassava flour. Food Res. 3, 484–490. doi:10.26656/fr.2017.3(5).093 ISO, 2009. International Standard ISO 10993-5 Biological evaluation of medical devices. Iso 40, 984–989. Juswardi, J., Delsy Amalia, I. and Sriwijaya, U. 2023. Metabolite profile of false elder leaves (Peronema canescens Jack.) based on development levels. Int. J. Life. Sci. 11, 143–150. Kumar, A. and Sharma, C. 2022. Recent update of the various sources originating ghost peaks in gas chromatography: a review. J. Chromatogr. A , 1685, 463625. doi:10.1016/j.chroma.2022.463625 Latief, M., Fisesa, A.T., Sari, P.M. and Tarigan, I.L. 2021. Anti-Inflammatory Activity of Sungkai Leaves (Peronema canescens Jack) Ethanol Extract in Carrageenan-Induced Mice. Jfsp 7, 2579–4558. Latief, M., Dasrinal, E., Safitri, W. and Tarigan, I.L. 2023. Immunomodulator activity of 5, 7-dihydroxy isoflavones and β-sitosterol from Peronema canescens Jack leaves methanol and ethyl acetate extract. In 4th Green Development International Conference (GDIC 2022), Atlantis Press, 558–572. Lee, J., Kim, M., Son, H., Kim, S., Jo, S., Janchiv, A., Kim, S.Y., Lee, T. and Yoo, H.Y. 2024. Phytochemical Characterization and Bioactivity Evaluation of Extracts Obtained via Ultrasound-Assisted Extraction of Medicinal Plant Phedimus aizoon. Plants 13, 1915. doi:10.3390/plants13141915 Lee, J., Kim, S. and Kang, C.H. 2022. Immunostimulatory Activity of Lactic Acid Bacteria Cell-Free Supernatants through the Activation of NF-κB and MAPK Signaling Pathways in RAW 264.7 Cells. Microorganisms 10, 2247; doi:10.3390/microorganisms10112247 Lim, Y.J., Sidor, N.A., Tonial, N.C., Che, A. and Urquhart, B.L. 2021. Uremic Toxins in the Progression of Chronic Kidney Disease and Cardiovascular Disease: mechanisms and Therapeutic Targets. Toxins. (Basel). 13 13(2), 142; doi: 10.3390/TOXINS13020142 Liu, Y., Li, Q.Z., Li, L.D.J. and Zhou, X.W. 2021. Immunostimulatory effects of the intracellular polysaccharides isolated from liquid culture of Ophiocordyceps sinensis (Ascomycetes) on RAW264.7 cells via the MAPK and PI3K/Akt signaling pathways. J. Ethnopharmacol. 275, 114130. doi:10.1016/j.jep.2021.114130 Maigoda, T., Judiono, J., Purkon, D.B., Haerussana, A.N.E.M. and Mulyo, G.P.E. 2022. Evaluation of Peronema canescens Leaves Extract: fourier Transform Infrared Analysis, Total Phenolic and Flavonoid Content, Antioxidant Capacity, and Radical Scavenger Activity. Open. Access. Maced. J. Med. Sci. 10, 117–124; doi:10.3889/oamjms.2022.8221 Marrez, D.A., Naguib, M.M., Sultan, Y.Y. and Higazy, A.M. 2019. Antimicrobial and anticancer activities of Scenedesmus obliquus metabolites. Heliyon 5, e01404. doi:10.1016/j.heliyon.2019.e01404 Noviana, E., Indrayanto, G. and Rohman, A. 2022. Advances in Fingerprint Analysis for Standardization and Quality Control of Herbal Medicines. Front. Pharmacol. 13, 853023.doi:10.3389/fphar.2022.853023 Noviarni I. 2023. Review Artikel: Potensi Ekstrak Daun Sungkai (Peronema canescens Jack.) sebagai Antioksidan. JSSIT: Jurnal Sains dan Sains Terapan, 1(1), 1–6. Park, H.Y., Oh, M.J., Kim, Y. and Choi, I. 2018. Immunomodulatory activities of Corchorus olitorius leaf extract: beneficial effects in macrophage and NK cell activation immunosuppressed mice. J. Funct. Foods 46, 220–226. doi:10.1016/j.jff.2018.05.005 Patel, S.S. and Savjani, J.K. 2015. Systematic review of plant steroids as potential antiinflammatory agents: current status and future perspectives. J. Phytopharm. 4, 121–125. doi:10.31254/phyto.2015.4212 Paul, S., Pan, S., Mukherjee, A. and De, P. 2021. Nitric Oxide Releasing Delivery Platforms: design, Detection, Biomedical Applications, and Future Possibilities. Mol. Pharm. 18(9), 3181–3205.doi:10.1021/acs.molpharmaceut.1c00486 Pratama, R.R., Sari, R.A., Sholikhah, I., Mansor, H., Chang, H.I., Sukardiman. and Widyowati, R. 2024. Inhibition of nitric oxide production in RAW 264.7 cells and cytokines IL-1β in osteoarthritis rat models of 70 % ethanol extract of Arcangelisia flava (L.) merr stems. Heliyon 10, e35730; doi:10.1016/j.heliyon.2024.e35730 Priyanga, S., Velusamy, M., Ramasubramanian, R. and Mayilmurugan, R. 2024. Fe(III) Complexes as the Optical Imaging Probe for l-Arginine via the Redox Mechanism. Cryst. Growth. Des. 25(2), 182–190.doi:10.1021/acs.cgd.4c00822 Qian, L., Du, M., Yang, X., Wang, Q., Huang, S., Ma, Y. and Sun, Y. 2023. Microanalysis Characterization and Immunomodulatory Effect for Selenium-Enriched Polysaccharide from Morchella esculenta (L.) Pers. Molecules 28, 7285; doi:10.3390/molecules28072885 Rahardhian, M.R.R., Susilawati, Y., Musfiroh, I., Febriyanti, R.M., Muchtaridi. and Sumiwi, S.A. 2022. In Silico Study of Bioactive Compounds From Sungkai (Peronema canescens) As Immunomodulator. Int. J. Appl. Pharm. 14, 135–141. doi:10.22159/ijap.2022.v14s4.PP33 Rasyid, A., Putra, M.Y. and Yasman. 2023. Antibacterial and antioxidant activity of Sea cucumber extracts collected from Lampung waters, Indonesia. Kuwait. J. Sci. 50, 615–621; doi:10.1016/j.kjs.2023.03.012 Renda, G., Gökkaya, I. and Şöhretoğlu, D. 2022. Immunomodulatory properties of triterpenes. Phytochem. Rev. 21, 537–563; doi:10.1007/s11101-021-09785-x Riastri, A., Putri, D.D.P., Sa’adah, M., Gani, A.P. and Murwanti, R. 2023. RAW 264.7 Macrophage Cell Line: in Vitro Model for the Evaluation of the Immunomodulatory Activity of Zingiberaceae. Trop. J. Nat. Prod. Res. 7(2), 2316.doi:10.26538/tjnpr/v7i2.3 Ryu, H.Y., Lee, H., Kong, H.J. and Kang, J.H. 2019. Anti-inflammatory Effects of Complex Extract including Eucommia ulmoides in LPS-induced RAW 264.7 Cells. J. Acupunct. Res. 36, 256–263. doi:10.13045/jar.2019.00276 Saake, P., Brands, M., Endeshaw, A.B., Stolze, S.C., Westhoff, P., Balcke, G.U., Hensel, G., Holton, N., Zipfel, C., Tissier, A., Nakagami, H. and Zuccaro, A. 2025. Ergosterol-induced immune response in barley involves phosphorylation of phosphatidylinositol phosphate metabolic enzymes and activation of diterpene biosynthesis. New Phytol. 246, 1236–1255; doi:10.1111/nph.70022 Sari, R.M., Wahono, S.K., Anwar, M., Rizal, W.A., Suryani, R. and Suwanto, A. 2023. Pyrolysis of coconut shells for liquid smoke production: effect of integrated water scrubber on reduction of tar. Biomass Convers. Biorefinery 14(20), 26105–26119. doi:10.1007/s13399-023-04675-y Shreshtha, S., Sharma, P., Kumar, P., Sharma, R. and Singh, S.P. 2018. Nitric oxide: it’s role in immunity. J. Clin. Diagnostic. Res. 12(7), 1.doi:10.7860/JCDR/2018/31817.11764 Smith, J.A. 2018. Regulation of cytokine production by the unfolded protein response; Implications for infection and autoimmunity. Front. Immunol. 9, 422. doi:10.3389/fimmu.2018.00422 Spagnuolo, C., Moccia, S. and Russo, G.L. 2018. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur. J. Med. Chem. 153, 105–115. doi:10.1016/j.ejmech.2017.09.001 Sravan Kumar, S., Manoj, P. and Giridhar, P. 2015. Fourier transform infrared spectroscopy (FTIR) analysis, chlorophyll content, and antioxidant properties of native and defatted foliage of green leafy vegetables. J. Food Sci. Technol. 52, 8131–8139. doi:10.1007/s13197-015-1959-0 Stenvinkel, P., Ketteler, M., Johnson, R.J., Lindholm, B., Pecoits-Filho, R., Riella, M., Heimbürger, O., Cederholm, T. and Girndt, M. 2005. IL-10, IL-6, and TNF-α: central factors in the altered cytokine network of uremia - The good, the bad, and the ugly. Kidney. Int. 67, 1216–1233. doi:10.1111/j.1523-1755.2005.00200.x Stockert, J.C., Blázquez-Castro, A., Cañete, M., Horobin, R.W. and Villanueva, A. 2012. MTT assay for cell viability: intracellular localization of the formazan product is in lipid droplets. Acta Histochem. 114, 785–796. doi:10.1016/j.acthis.2012.01.006 Sujono, T.A., Nurrochmad, A., Lukitaningsih, E. and Nugroho, A.E. 2021. Immunomodulatory effect of petroleum ether extract and ethyl acetate fraction of bengkoang (Pachyrhizus erosus (L.) Urban) in vitro. Indonesian J. Pharm. 32(4), 454–463. Suryani, A.E., Nisa, K., Handayani, S., Darsih, C., Wuryastuty, H. and Yanuartono, Y. 2025. The UHPLC-HRMS profiling, in vitro-antioxidant and pancreatic lipase inhibitory activities of Peronema canescens leaves extract and fractions from Indonesia. J. Appl. Pharm. Sci. 15, 75–89; doi:10.7324/JAPS.2025.214965 Tarigan, I.L., Sutrisno, S., Rumaida, R., Aini, I.P.S. and Latief, M. 2022. Isolation of a Flavone Apigenin and a Steroids Squalene from Peronema canescens Jack Leaves with Anti-Inflammatory Activities. Pharmacogn. J. 14, 744–752. doi:10.5530/pj.2022.14.162 Teng, L., Fu, H., Wang, M., Deng, C. and Chen, J. 2015. Stimulation of RAW264.7 macrophages by sulfated Escherichia coli K5 capsular polysaccharide in vitro. Mol. Med. Rep. 12, 5545–5553. doi:10.3892/mmr.2015.4082 Thakur, S., Singla, A., Chhabra, R., Kaur, M., Gupta, N., Bhardwaj, U. and Sharma, J.P. 2023. GC-MS Analysis for Phytochemical Profiling of Different Salix Clones. Russ. J. Plant Physiol. 70, 122. doi:10.1134/S1021443723600708 Tizard, I., 2018. Veterinary Immunology, 10th edition, Elsevier. To’bungan, N., Widhiastuti, S.S., Hida, F.N.L. and Mahardhika, I.W.S. 2024. Phytochemical Properties, Antioxidant, and Cytotoxicity Activity of Knobweed (Hyptis capitata) from South Sulawesi, Indonesia. J. Trop. Biodivers. Biotechnol. 9, 90976. doi:10.22146/jtbb.90976 Varshini, R., Manokiruthika, V., Hariprasath, L., Muthusami, S., and Suganyadevi, P. 1953. Cycloheptasiloxane, tetradecamethyl identified from petroleum ether fraction of Sargassum wightii by Gas Chromatography-Mass Spectrometry Analysis. Versita, R. and Setiani, A.D. 2023. Effectiveness Anti-Inflammatory of Sungkai Leaf (Peronema canescens Jack.) Ethanol Extract on Male Rabbit (Oryctolagus cuniculus). Med. Sains. J. Ilm. Kefarmasian. 8, 1135–1140. doi:10.37874/ms.v8i3.822 Zhou, Y., Wang, S., Feng, W., Zhang, Z. and Li, H. 2021. Structural characterization and immunomodulatory activities of two polysaccharides from Rehmanniae Radix Praeparata. Int. J. Biol. Macromol. 186, 385–395. doi:10.1016/j.ijbiomac.2021.06.100 | ||
| How to Cite this Article |
| Pubmed Style Suryani AE, Nisa K, Haryanti S, Handayani S, Indrianingsih AW, Nuzulia WNM, Wuryastuty H, Yanuartono Y. Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Vet. J.. 2025; 15(12): 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 Web Style Suryani AE, Nisa K, Haryanti S, Handayani S, Indrianingsih AW, Nuzulia WNM, Wuryastuty H, Yanuartono Y. Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. https://www.openveterinaryjournal.com/?mno=269415 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.66 AMA (American Medical Association) Style Suryani AE, Nisa K, Haryanti S, Handayani S, Indrianingsih AW, Nuzulia WNM, Wuryastuty H, Yanuartono Y. Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Vet. J.. 2025; 15(12): 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 Vancouver/ICMJE Style Suryani AE, Nisa K, Haryanti S, Handayani S, Indrianingsih AW, Nuzulia WNM, Wuryastuty H, Yanuartono Y. Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 Harvard Style Suryani, A. E., Nisa, . K., Haryanti, . S., Handayani, . S., Indrianingsih, . A. W., Nuzulia, . W. N. M., Wuryastuty, . H. & Yanuartono, . Y. (2025) Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Vet. J., 15 (12), 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 Turabian Style Suryani, Ade Erma, Khoirun Nisa, Sari Haryanti, Sri Handayani, Anastasia Wheni Indrianingsih, Wafda Naufi Marva Nuzulia, Hastari Wuryastuty, and Yanuartono Yanuartono. 2025. Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Veterinary Journal, 15 (12), 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 Chicago Style Suryani, Ade Erma, Khoirun Nisa, Sari Haryanti, Sri Handayani, Anastasia Wheni Indrianingsih, Wafda Naufi Marva Nuzulia, Hastari Wuryastuty, and Yanuartono Yanuartono. "Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis." Open Veterinary Journal 15 (2025), 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 MLA (The Modern Language Association) Style Suryani, Ade Erma, Khoirun Nisa, Sari Haryanti, Sri Handayani, Anastasia Wheni Indrianingsih, Wafda Naufi Marva Nuzulia, Hastari Wuryastuty, and Yanuartono Yanuartono. "Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis." Open Veterinary Journal 15.12 (2025), 6856-6871. Print. doi:10.5455/OVJ.2025.v15.i12.66 APA (American Psychological Association) Style Suryani, A. E., Nisa, . K., Haryanti, . S., Handayani, . S., Indrianingsih, . A. W., Nuzulia, . W. N. M., Wuryastuty, . H. & Yanuartono, . Y. (2025) Evaluation of the immunomodulatory properties of Peronema canescens leaf fractions: A molecular and chemical analysis. Open Veterinary Journal, 15 (12), 6856-6871. doi:10.5455/OVJ.2025.v15.i12.66 |