| Research Article | ||
Open Vet. J.. 2025; 15(12): 6487-6496 Open Veterinary Journal, (2025), Vol. 15(12): 6487-6496 Research Article Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cellsJuma Al-Mutaani1,2, Tahani Zorgui3, Lazhar Zourgui4, Omar Alqawi5 and Nabiha Missaoui1*1Research Laboratory LR21ES03, Oncogenesis and Tumor Progression, Faculty of Medicine of Sousse, University of Sousse, Sousse, Tunisia 2Primary Health Care, Jaalan Bani Bu Hassan Hospital, Ministry of Health, Muscat, Sultanate of Oman 3Research Laboratory on Energy, Water, Environment and Processes, National School of Engineering, Gabes University, Gabes, Tunisia 4Research Laboratory BMA LR22ES02, Higher Institute of Applied Biology of Medenine, University of Gabes, Gabes, Tunisia 5National Cancer Institute, Misurata, Libya *Corresponding Author: Nabiha Missaoui. Research Laboratory LR21ES03, Oncogenesis and Tumor Progression, Faculty of Medicine of Sousse, University of Sousse, Sousse, Tunisia. Email: missaouinabiha [at] live.fr Submitted: 02/07/2025 Revised: 11/11/2025 Accepted: 17/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
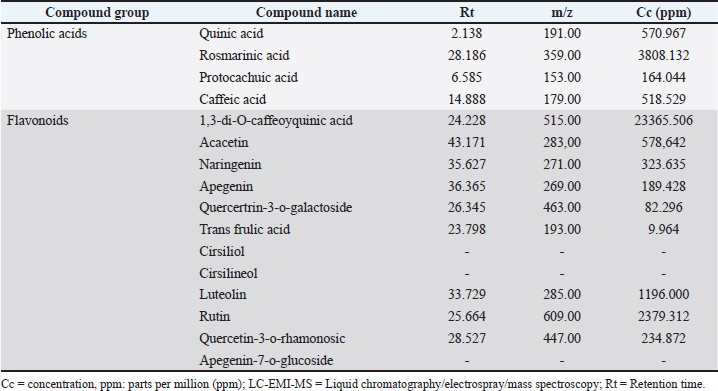
AbstractBackground: Natural plant-derived compounds are increasingly explored as safer alternatives to chemotherapy due to their multi-targeted anticancer properties. Despite Oman’s rich floral biodiversity, data on the anticancer potential of its native species remain limited. Ocimum basilicum has emerged as a plant with promising bioactivities. Aim: This study examined the cytotoxic, antimigratory, and antiadhesive effects of O. basilicum ethanolic extract on HCT116 colorectal cancer (CRC) cells. Methods: Ocimum basilicum collected from South Al Sharqiyah, Oman, was ethanol-extracted and tested against HCT116 cells at concentrations of 0–200 µg/ml. Cytotoxicity was evaluated using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, whereas cell adhesion and migration were assessed using fibrinogen/fibronectin-coated matrices and the wound-healing assay, respectively. Data were analyzed using one-way analysis of variance. Results: The ethanolic extract was rich in 1,3-di-O-caffeoylquinic acid (23,366 ppm), rosmarinic acid (3,808 ppm), rutin (2,379 ppm), and luteolin (1,196 ppm). The extract significantly reduced HCT116 cell viability in a dose-dependent manner, from 87.7% at 50 µg/ml to 32.4% at 200 µg/ml (p < 0.0001). Migration was moderately inhibited at 50 µg/ml and strongly suppressed at concentrations ≥ 100 µg/ml (p < 0.0001), while adhesion remained unaffected at 50 µg/ml but was significantly decreased at higher concentrations (p < 0.0001). A strong dose–response correlation was observed across all assays (R² > 0.89). Conclusion: Ocimum basilicum ethanolic extract exhibits pronounced cytotoxic and antimigratory activities against HCT116 CRC cells, highlighting its potential as a natural anticancer candidate. Keywords: Ocimum basilicum, Colorectal cancer, HCT116 cells, Natural anticancer agent; Oman. IntroductionColorectal cancer (CRC) is the most prevalent malignancy of the gastrointestinal tract, ranking third in global cancer incidence and remaining the second leading cause of cancer-related mortality worldwide, accounting for an estimated 904,019 deaths in 2022 (Bray et al., 2024). According to recent Global Burden of Disease estimates, the Middle East and North Africa region reported approximately 670,000 new cases and 418,000 deaths in 2020, reflecting a rapidly increasing cancer burden driven by population growth, increased life expectancy, and lifestyle as well as dietary transitions associated with socioeconomic development (Hofmarcher et al., 2023; Poor et al., 2025). CRC has become the most frequently diagnosed cancer among men in many Middle Eastern countries, with a notable increase in incidence among younger adults (Makhlouf et al., 2021). In Oman, the burden of CRC has risen sharply over the past two decades. The age-standardized incidence rate for colon cancer increased from 2.7 per 100,000 in 1996 to 11.2 in 2019, and that for rectal cancer increased from 1.5 to 5.3 during the same period, with annual percentage changes of 6.92% and 4.24%, respectively (Al-Sayegh, 2024). These alarming trends underscore the urgent need for more effective preventive and therapeutic strategies. Oxidative stress is a major contributor to CRC initiation and progression, promoting epithelial injury and compromising intestinal barrier integrity (Bardelčíková et al., 2023; Matsuda et al., 2025). Conventional CRC management relies on surgery, chemotherapy, and radiotherapy depending on the disease stage (Puzzo et al., 2025). Although surgical resection remains the mainstay for early-stage tumors and systemic chemotherapy regimens [Folinic acid, 5-Fluorouracil (5-FU), and Oxaliplatin (FOLFOX) and Folinic acid, 5-Fluorouracil (5-FU), and Irinotecan (FOLFIRI)] are standard for advanced disease, nearly half of non-metastatic patients experience relapse, and approximately 20% present with distant metastases at diagnosis (Tatsuta et al., 2025). Targeted therapies, including anti-epidermal growth factor receptor antibodies, have improved survival outcomes in rat sarcoma (RAS) wild-type tumors. However, drug resistance, treatment-related toxicity, and interpatient variability remain major clinical challenges (Puzzo et al., 2025). The current therapies’ limitations have intensified the search for safer and more effective alternatives, particularly from natural product sources with multitargeted mechanisms of action. Over the past few decades, numerous plant species have been recognized as valuable sources of anticancer agents (Boulaaba et al., 2013; Ashwini et al., 2017). Plant-derived compounds exhibit the remarkable ability to modulate multiple cellular signaling pathways and demonstrate diverse biological activities, including antiproliferative, proapoptotic, antioxidant, and antimetastatic effects against various types of cancer (Sheng et al., 2017; Khajeh et al., 2020; Xiang et al., 2020; Jovankić et al., 2022; Chammam et al., 2024). Ocimum basilicum (basil) is among the medicinal plants reported to possess significant anticancer potential in studies conducted across several countries (Aburigal et al., 2017). Nonetheless, factors such as environmental conditions, genetic variability, growth stage, and extraction methods can markedly influence the composition and concentration of bioactive constituents (Zhakipbekov et al., 2024). For example, Aburigal et al. (2017) reported marked variability in antioxidant activity and phenolic content among basil accessions from six countries, with the Maldivian strain exhibiting the highest levels (Aburigal et al., 2017). Medicinal plants from Oman share species with other regions but often display distinctive phytochemical profiles shaped by the country’s diverse climatic and ecological conditions. Notably, Omani O. basilicum is enriched in flavonoids and phenolic acids, conferring strong antioxidant and anticancer properties (Al-Mutaani et al., 2025). Therefore, the present study aimed to evaluate the anticancer effects of the ethanolic extract of Omani Ocimum basilicum using HCT-116 CRC cells as an in vitro model. Specifically, we investigated its effects on cell viability, migration, and adhesion. Materials and MethodsReagentsThe study employed ethanol, 70% ethanol spray, and distilled water (H2O), along with Roswell Park Memorial Institute (RPMI) medium (Sigma, Burlington, MA) supplemented with 10% fetal calf serum (FCS), 1% L-glutamine (200 mM), penicillin (100 U/ml), and streptomycin (100 µg/ml). Additional reagents included bovine serum albumin (BSA), fibrinogen (50 µg/ml), fibronectin (10 µg/ml), dimethyl sulfoxide (DMSO), phosphate-buffered saline (PBS), PBS containing calcium ions (PBS/Ca²+), PBS with Ethylenediaminetetraacetic Acid (EDTA) (0.53 mM), trypsin, sodium dodecyl sulfate (SDS), sodium hydroxide (NaOH), sodium phosphate (28 mM), sulfuric acid (0.6 M), ammonium molybdate (4 mM), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT). All chemicals and reagents were obtained from the Faculty of Sciences, University of Gabes, Tunisia. EquipmentThe major equipment included a biosafety cabinet, HCT116 human colorectal carcinoma cell line, 96-well tissue culture plates, CO2 incubator, personal protective equipment, water bath, centrifuge, hemocytometer, inverted phase-contrast microscope, microplate reader (Thermo Labsystems, Franklin, MA), cell culture flasks, pipettes with disposable tips, and tube racks. Collection of plant materialOcimum basilicum specimens were collected from the South Al Sharqiyah Governorate, Sultanate of Oman, between July and September 2023. Identification and authentication of plantsA taxonomist at the University of Gabes, Tunisia, taxonomically authenticated the plant, and a voucher specimen was deposited at the University Herbarium and assigned. The species’ taxonomic identity was further verified using the World Flora Online database (World Flora Online, 2024). Extraction processThe ethanolic extract was prepared according to established protocols (Zourgui et al., 2020; Al-Mutaani et al., 2025). Leaves and stems (excluding roots) were washed, shade-dried, chopped, and ground into a fine powder (Zourgui et al., 2020; Al-Mutaani et al., 2025). Briefly, 200 g of powdered plant material was mixed with 800 ml of ethanol and shaken at room temperature for 24 hours. The mixture was filtered, centrifuged, and the supernatant was evaporated under reduced pressure, followed by lyophilization at 4°C. The dry extract was stored at 4°C until use. Liquid chromatography/electrospray ionization/mass spectrometry analysisThe phytochemical composition of O. basilicum ethanolic extract was previously determined and reported in our recent publication (Al-Mutaani et al., 2025) using liquid chromatography/electrospray ionization/mass spectrometry (LC-ESI-MS). Briefly, flavonoids and phenolic acids were extracted following the method of Ayaz et al. (2005) with slight modifications. LC–ESI–MS analysis was performed on a Shimadzu LCMS-2020 quadrupole mass spectrometer (Kyoto, Japan) equipped with an electrospray ionization source operating in negative ionization mode. Phytochemical compounds were identified by comparing their retention times and mass spectra with those of authentic standards (≥99% purity; Sigma Chemical Co., St. Louis, MO) (Chahbani et al., 2018). Antioxidant activityThe antioxidant activity was previously evaluated using five complementary assays, as detailed in our recent publication (Al-Mutaani et al., 2025). The 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical-scavenging activity was determined according to Sanchez et al. (2014) by measuring the reduction of DPPH absorbance at 517 nm after incubation with the extracts (Sanchez et al., 2014). Nitric oxide radical-scavenging activity was assessed following Jagetia and Baliga (2004) by measuring nitrite formation at 546 nm after the reaction with Griess reagent. Ferric reducing antioxidant power was determined as described by Yildirim et al. (2001) based on the increase in absorbance at 700 nm reflecting Fe³+ reduction (Yildirim et al., 2001). The ABTS•+ radical-scavenging capacity was measured following Chang et al. (2008) by comparing the absorbance reduction at 734 nm to the Trolox standards. Finally, the total antioxidant capacity was evaluated using the phosphomolybdenum method (Phatak and Hendre, 2014) by measuring the absorbance at 695 nm and expressing the results as mg ascorbic acid equivalents per g extract. Anti-cancer activity assaysCell cultureHCT116 cells were cultured in RPMI medium supplemented with 10% FCS and 1% penicillin–streptomycin. The cells were maintained at 37°C in a humidified atmosphere with 5% CO2. Upon reaching 80%–90% confluence, the cells were trypsinized, centrifuged at 1,000 rpm for 5 minutes, and re-suspended. All steps were conducted under sterile laminar-flow conditions (Nelson et al., 2020; Padauleng et al., 2023). Cell countingCell viability was assessed using the trypan blue exclusion method by mixing cells 1:1 with 0.4% trypan blue and manually counting viable (unstained) and non-viable (blue-stained) cells with a hemocytometer (Daham, 2021). Cell density was calculated as follows: Cells/ml=(average cells/square) × 2 (dilution factor) × 104. MTT cytotoxicity assayCytotoxicity was evaluated using the MTT assay according to established protocols (Jovankić et al., 2022; Fayazi et al., 2024; Abdulla et al., 2025). The freeze-dried extract was first reconstituted to prepare a 200 µg/ml stock solution, from which 100 and 50 µg/ml working concentrations were obtained by serial dilution. HCT116 cells were seeded in 96-well plates (10,000 cells/well) and treated with 0, 50, 100, or 200 µg/ml of the extract for 24 hours. After treatment, cells were washed with PBS, and 50 µl of MTT solution (5 mg/ml) was added per well and incubated for 1 hour at 37°C for 1 hour. The formazan crystals were dissolved in 100 µl DMSO, and the absorbance was measured at 570 nm using a multidetection microplate reader (Thermo Labsystems, Franklin, MA). Negative controls contained RPMI medium alone. Based on these measurements, the cytotoxicity (%) was calculated as follows: Cytotoxicity (%)=(1−A570 treated/A570 control) × 100. Proliferation assayThe cells (5,000/well) were seeded and incubated for 48 hours before treatment with 0, 50, 100, or 200 µg/ml of extract for 72 hours. The fixed cells were stained with 50 µl crystal violet for 30 minutes at 37°C, washed, and solubilized in DMSO. The absorbance was measured at 560 nm (Shimizu et al., 2013). Cell adhesion assayThe 96-well plates were pre-coated overnight at 4°C with fibrinogen (50 µg/ml) or fibronectin (10 µg/ml) in PBS/Ca²+. The wells were blocked with PBS/BSA at 37°C for 30 minutes. The cells treated with 0–200 µg/ml extract were seeded and incubated for 3 hours at 37°C for 3 hours. Non-adherent cells were removed by washing with PBS. The adherent cells were fixed with 1% glutaraldehyde (10 minutes), stained with crystal violet (30 minutes), and solubilized in 1% SDS. The absorbance was recorded at 560 nm (Buttner et al., 2020). Wound healing migration assayA linear scratch was made in a confluent monolayer with a sterile 200 µl pipette tip, and detached cells were removed with PBS (Dahham et al., 2021). The remaining cells were treated with 0–200 µg/ml extract and incubated for 24 hours. Wound closure was monitored using an inverted microscope under 100× magnification (Leica Quin; Microsystems computerized imaging system, USA). The mean percentage of migration inhibition was calculated by comparing with the control using the following formula: % migration inhibition=[1 − (Ds/Dc)] × 100, where Ds=migration distance in treated cells and Dc=migration distance in control cells. Statistical analysisAll experiments were performed in triplicate. To ensure precision and reliability, the findings were analyzed using mean values and standard deviations and presented as mean ± SEM (Abdulla et al., 2025). Statistical evaluation was conducted using IBM SPSS version 25. The data were analyzed using analysis of variance followed by Tukey’s post hoc test to compare the mean values. A multiple comparison test was used to determine significant differences among groups. A p-value of 0.05 was considered statistically significant. Ethical approvalNot needed for this study. ResultsTable 1 presents the results of the LC-ESI-MS analysis (Al-Mutaani et al., 2025). The extract was rich in phenolic acids and flavonoids, with rosmarinic acid (3,808 ppm) and quinic acid (571 ppm) being the predominant phenolic acids. The flavonoid fraction was dominated by 1,3-di-O-caffeoylquinic acid (23,366 ppm), rutin (2,379 ppm), and luteolin (1,196 ppm). Table 1. LC-EMI-MS analysis of ethanolic extracts of Ocimum basilicum (Al-Mutaani et al., 2025).
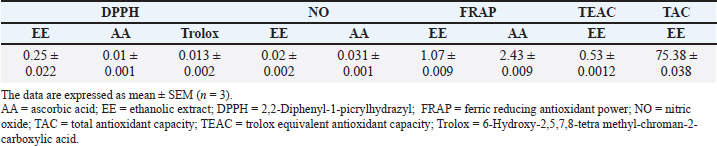
Table 2 summarizes the antioxidant activities of the Ocimum basilicum ethanolic extract (Al-Mutaani et al., 2025). The extract showed strong DPPH scavenging (IC50=0.25 ± 0.022 mg/ml), surpassing ascorbic acid (IC50=0.01 ± 0.001 mg/ml) and Trolox (IC50=0.013 ± 0.002 mg/ml), and notable nitric oxide scavenging (IC50=0.02 ± 0.002 mg/ml vs. ascorbic acid=0.031 ± 0.001 mg/ml). FRAP analysis indicated substantial ferric-reducing power (1.07 ± 0.009 mg/ml), while Trolox equivalent antioxidant capacity was moderate (0.53 ± 0.0012), and total antioxidant capacity was high (75.38 ± 0.038 mg AAE/g dw), collectively highlighting the extract’s potent and broad-spectrum antioxidant potential. Table 2. Anti-oxidant activities of Ocimum basilicum ethanolic extract (Al-Mutaani et al., 2025).
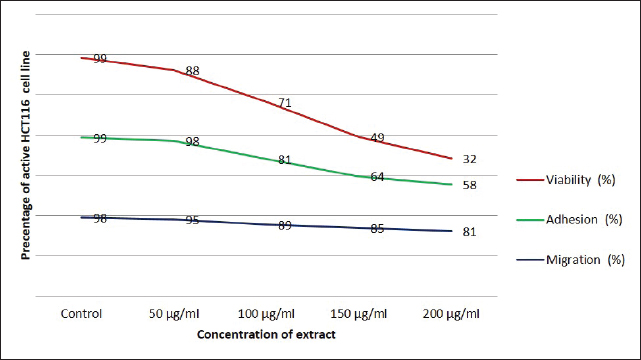
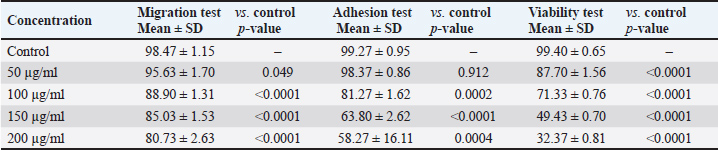
The anticancer activity of the O. basilicum ethanolic extract collected from Oman against HCT116 CRC cells is summarized in Table 3 and illustrated in Fig. 1. The effects of the extract were assessed using three complementary in vitro assays: cell viability (MTT), adhesion, and wound-healing migration. Cells were treated with increasing concentrations of the extract (50, 100, 150, and 200 µg/ml) and compared with untreated controls.
Fig. 1. Anticancer activities of Omani Ocimum Basilicum. Table 3. Effects of ethanolic Ocimum basilicum extract on HCT116 cells assessed by MTT viability, adhesion, and migration assays.
The extract exhibited a concentration-dependent inhibitory effect across all assays. Cell viability was significantly reduced at all tested concentrations, decreasing from 87.70% ± 1.56% at 50 µg/ml to 32.37% ± 0.81% at 200 µg/ml (p < 0.0001 for all vs. control) (Table 3, Fig. 1). Cell migration was moderately inhibited at the lowest concentration (95.63% ± 1.70%, p=0.049), with a progressive and significant reduction at higher concentrations: 88.90% ± 1.31% (100 µg/ml), 85.03% ± 1.53% (150 µg/ml), and 80.73% ± 2.63% (200 µg/ml) (p < 0.0001 for all, vs. control) (Table 3, Fig. 1). Cell adhesion was not significantly affected at 50 µg/ml (98.37% ± 0.86%, p=0.912). However, significant impairment was observed at 100 µg/ml (81.27% ± 1.62%, p=0.0002), 150 µg/ml (63.80% ± 2.62%, p < 0.0001), and 200 µg/ml (58.27% ± 16.11%, p=0.0004) relative to controls (Table 3, Fig. 1). Overall, the extract demonstrated potent anti-proliferative and anti-migratory effects, with anti-adhesive effects emerging at concentrations ≥100 µg/ml (Table 3, Fig. 1). Cell viability exhibited the highest sensitivity to treatment, followed by migration and adhesion. A robust dose–response relationship was observed across all assays, as reflected by high correlation coefficients (R² > 0.89), indicating a strong concentration-dependent mode of action (Fig. 1). DiscussionThis study demonstrated that the ethanolic extract of O. basilicum, collected in Oman, exerts potent anticancer effects on HCT116 cells, as evidenced by a significant, dose-dependent reduction in cell viability, migration, and adhesion. Among the tested parameters, cell viability was the most sensitive to treatment, followed by migration and adhesion, with a consistent dose–response relationship. The ethanolic extract’s strong bioactivity likely reflects its efficient solubilization of polyphenolic compounds, which possess well-documented anticancer properties (Rao et al., 2024). Owing to its intermediate polarity, ethanol is widely recognized for its ability to extract both hydrophilic and lipophilic bioactive compounds, including flavonoids, phenolic acids, and terpenoids (Ermi Hikmawanti et al., 2021). In contrast, solvents, such as petroleum ether, are less effective for polyphenol extraction and may co-extract undesirable non-polar substances (Zarlaha et al., 2014). The use of the HCT116 cell line is methodologically justified because of its aggressive phenotype, self-renewal capabilities, and gene expression profiles that closely resemble those of primary colorectal tumors (Yeung et al., 2010). These cells also exhibit high proliferative rates and secrete immune-modulatory chemokines (CXCL16, CCL27, and CCL28), enhancing their relevance in the evaluation of anticancer agents (Elemam et al., 2019). The use of RPMI-1640 medium, which supports HCT116 proliferation through its balanced nutrient composition, further ensured optimal assay conditions (Wu et al., 2009). Our MTT assay results demonstrate that O. basilicum extract markedly reduces the viability of HCT116 CRC cells, which is in agreement with previous studies investigating the anticancer activity of Ocimum species. Among the bioactive constituents of O. basilicum, quercetin—a plant-derived flavonol and one of the most extensively studied polyphenols in fruits, vegetables, and medicinal plants—has been widely reported to exert potent anticancer effects in various malignancies, including colon, gastric, cervical, breast, hepatocellular, and glioblastoma cancers. The anticancer mechanisms of quercetin are primarily mediated through the modulation of key signaling pathways such as Wnt/β-catenin, phosphoinositide 3-kinase/protein kinase B/mammalian target of rapamycin (PI3K/Akt/mTOR), Janus kinase/signal transducer and activator of transcription (JAK/STAT), mitogen-activated protein kinase (MAPK), and non-coding RNA regulatory networks (Hijazi et al., 2025). Additionally, quercetin has been shown to inhibit the JAK/STAT and RAS/RAF/MEK/ERK cascades, thereby suppressing cell proliferation and promoting apoptosis in cancer cells (Rao et al., 2024). Comparable findings have been reported for other Ocimum species in various cancer cell lines. For instance, ethanolic and aqueous extracts of O. sanctum exhibited cytotoxic effects in HT-15, HT-29, A549, and IMR-32 cells (Jovankić et al., 2022). Similarly, O. minimum extracts induced apoptosis in HCT116 cells through enhanced ROS generation, lipid peroxidation, mitochondrial membrane destabilization, and apoptotic signaling (Jovankić et al., 2022). Furthermore, aqueous O. basilicum extract was shown to upregulate pro-apoptotic markers Bcl-2 Associated X-protein (BAX) and Bcl-2-associated death (BAD) in CRC cell lines while sparing normal colon epithelial cells (Heydarnezhad Asl et al., 2022), indicating a degree of tumor selectivity. The effective dose range for plant-derived anticancer agents varies considerably. Panicker et al. (2020) reported 70% cytotoxicity in MCF-7 and MCF-10A cells at 125 µg/ml of Cleome droserifolia extract, whereas Chakroun et al. (2024) observed significant cytotoxicity at 30 µg/ml of Phoenix dactylifera methanolic extract in glioblastoma and breast cancer cell lines. These findings reinforce that the cytotoxicity of natural products depends on extract composition, cell line sensitivity, and experimental conditions (Dulay et al., 2022). Similarly, Panicker et al. (2020) highlighted the need for analytical validation techniques, such as nuclear magnetic resonance (NMR), to ensure specificity and minimize solvent-related artifacts (Panicker et al., 2020). The anti-migratory effects of O. basilicum extract were dose-dependent, with substantial inhibition at higher concentrations. Loss of epithelial characteristics and basement membrane integrity facilitates tumor metastasis (Martin et al., 2009; Son et al., 2017), and migration suppression is a crucial indicator of anti-metastatic potential. This is consistent with previous reports showing that flavonoids, such as rutin and apigenin, present in Ocimum, can suppress cancer cell migration by modulating the Wnt and JAK2/STAT3 pathways in CRC and lung cancer models (Jovankić et al., 2022; Rao et al., 2024). Thus, migration inhibition represents a key strategy to prevent metastasis, particularly in aggressive CRC phenotypes. Similarly, cell adhesion was significantly reduced at concentrations ≥100 µg/ml The broad-spectrum anti-cancer activity of the extract likely results from the synergistic effects of its constituent polyphenols and flavonoids, including quercetin, rutin, luteolin, rosmarinic acid, and caffeic acid. These compounds are known to induce apoptosis, arrest cell cycle progression, inhibit angiogenesis, and modulate oncogenic pathways such as c-MYC, TP53, caspase-3/8, and NOS2 (Jovankić et al., 2022; Rao et al., 2024). The observed reduction parallels the findings of Dolghi et al. (2021), who reported diminished Caco-2 cell adhesion following O. basilicum oil treatment. Loss of adhesion reflects interference with integrin-mediated attachment and extracellular matrix interactions, processes crucial for metastasis and tumor progression (Dolghi et al., 2021). These results also align with those of Nelson et al. (2020) who observed Eclipta alba migration inhibition at comparable concentrations. Some variability was noted in the adhesion assay at the highest extract concentration. Such variation can arise from multiple factors, including cell density, culture conditions, pH, and contamination, all of which must be strictly controlled to ensure reproducibility. Low seeding density may produce sparse or inconsistent data, whereas overseeding can lead to surface overcrowding, thereby reducing measurement accuracy. Contamination—whether microbial or chemical—can cause misleading results or interfere directly with cellular behavior. Moreover, environmental factors, such as pH and specific ions (e.g., calcium), affect cell viability and electrostatic interactions with the substrate. Overall, the ethanolic extract of O. basilicum exhibited robust, dose-dependent anticancer activity in HCT116 CRC cells by inhibiting proliferation, migration, and adhesion, supporting its potential as a promising source of natural anticancer agents. However, several limitations should be acknowledged. Experiments were conducted with a limited number of replicates (n=3) and involved only one CRC cell line, without a normal-cell comparison, which constrains the generalizability of the findings. Future studies should include both malignant and nonmalignant colon cell models (e.g., HT-29 and CCD-18Co) to better assess selectivity, safety, and translational relevance. The lack of mechanistic assays, such as apoptosis (e.g., annexin V, caspase activation), necrosis, oxidative stress markers, and cell cycle analyses, also limits the understanding of the mode of action of the extract. Further in vivo validation using xenograft models and molecular-level studies (protein, DNA, and RNA) are warranted to elucidate the underlying biological mechanisms. Finally, the isolation and characterization of individual bioactive compounds using advanced analytical techniques (e.g., NMR) will be essential to confirm their contribution to the observed anticancer effects. ConclusionIn summary, our study primarily constitutes a toxicity assessment of the ethanolic extract of O. basilicum from Oman, suggesting dose-dependent anticancer effects against HCT116 CRC cells through inhibition of proliferation, migration, and adhesion. These findings highlight its translational potential and support further preclinical evaluation. Although the results are promising, the underlying molecular mechanisms remain unclear. Future studies should aim to isolate the active constituents, delineate their molecular targets and signaling pathways, and validate efficacy and safety in preclinical CRC models, such as xenografts, to establish a mechanistic basis for therapeutic application. AcknowledgmentsThe authors gratefully acknowledge Professor Mustapha Gorai from the Higher Institute of Applied Biology of Medenine, University of Gabes (Tunisia), for his valuable assistance in authenticating the Ocimum basilicum plant specimen collected in Oman. Conflict of interestThe authors have no competing interests to declare. FundingNot applicable. Author contributionsJAM and TZ: conceptualization, data collection, data curation, and paper drafting. LZ: conceptualization, experimental design, resources, and supervision. OA: conceptualization, visualization, and data analysis. NM: conceptualization, visualization, supervision, and paper writing. All authors have reviewed and approved the final version of the manuscript. Data availabilityThe supplementary data will be made available by the corresponding author upon reasonable request. ReferencesAbdulla, S.A., Elshaari, F.A., Alshintari, M. and Zourgui, L. 2025. Phytochemical profiling and biological activity of the ethanolic extract of Phragmites australis. Open. Vet. J. 15(6), 2478–2491; doi:10.5455/OVJ.2025.v15.i6.20 Aburigal, Y.A.A., Mirghani, M.E.S., Elmogtaba, E.Y., Sirible, A.A.M., Hamza, N.B. and Hussein, I.H. 2017. Total phenolic content and antioxidant capacity of basil (Ocimum basilicum L.) leaves from different locations. Int. Food. Res. J. 24, S378–S381. Al-Mutaani, J., Zorgui, T., Mohamed, A.A., Smetanska, I., Zourgui, L. and Missaoui, N. 2025. Comparative antioxidant activity of four Omani medicinal plants: Ocimum basilicum, Teucrium polium, Caralluma arabica, and Cleome amblyocarpa. Open Vet. J. 15(9), 4671–4680; doi:10.5455/OVJ.2025.v15.i9.71 Al-Sayegh, H., Al-Zadjali, S. and Al-Moundhri, M. 2024. Analyzing cancer incidence trends in oman from 1996 to 2019: a comprehensive study of the National Cancer Annual Reports. JCO. Glob. Oncol. 10, e2300337; doi:10.1200/GO.23.00337 Ashwini, S., Ezhilarasan, D. and Anitha, R. 2017. Cytotoxic effect of Caralluma fimbriata against human colon cancer cells. Pharmacogn. J. 9(2), 204–207; doi:10.5530/pj.2017.2.34 Ayaz, F.A., Hayirlioglu-Ayaz, S., Gruz, J., Novak, O. and Strnad, M. 2005. Separation, characterization, and quantitation of phenolic acids in a Little-Known Blueberry (Vaccinium arctostaphylos L.) Fruit by HPLC-MS. J. Agric. Food Chem. 53(21), 8116–8122; doi:10.1021/jf058057y Bardelčíková, A., Šoltys, J. and Mojžiš, J. 2023. Oxidative stress, inflammation and colorectal cancer: an overview. Antioxidants 12(4), 901; doi:10.3390/antiox12040901 Boulaaba, M., Mkadmini, K., Tsolmon, S., Han, J., Smaoui, A., Kawada, K., Ksouri, R., Isoda, H., and Abdelly, C. 2013. In vitro antiproliferative effect of Arthrocnemum indicum extracts on Caco-2 cancer cells through cell cycle control and related phenol LC-TOF-MS identification. Evid. Based Complement. Alternat. Med. 2013(1), 529375. doi: 10.1155/2013/529375 Bray, F., Laversanne, M., Sung, H., Ferlay, J., Siegel, R.L., Soerjomataram, I. and Jemal, A. 2024. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 74(3), 229–263; doi:10.3322/caac.21834 Büttner, H., Perbandt, M., Kohler, T., Kikhney, A., Wolters, M., Christner, M., Heise, M., Wilde, J., Weißelberg, S., Both, A., Betzel, C., Hammerschmidt, S., Svergun, D., Aepfelbacher, M. and Rohde, H. 2020. A giant extracellular matrix binding protein of Staphylococcus epidermidis binds surface-immobilized fibronectin via a novel mechanism. mBio 11(5), e01612-20; doi:10.3204/PUBDB-2020-04302 Chahbani, A., Fakhfakh, N., Balti, M.A., Mabrouk, M., El-Hatmi, H., Zouari, N. and Kechaou, N. 2018. Microwave drying effects on drying kinetics, bioactive compounds and antioxidant activity of green peas (Pisum sativum L.). Food Bioscience 25, 32–38; doi:10.1016/j.fbio.2018.07.004 Chakroun, M., Morjen, M., Mabrouk, H.B., Mejdoub, H., Srairi-Abid, N., Marrakchi, N., Jebali, J. and Khemakhem, B. 2024. Anticancer properties of different varieties of date palm (Phoenix dactylifera L.) leaf extracts in human tumor cells: a comparative study. Plant. Foods. Hum. Nutr. 79(2), 518–525; doi:10.1007/s11130-024-01162-1 Chammam, A., Romdhane, M., Fillaudeau, L. and Bouajila, J. 2024. Phytochemical composition and characterization of in vitro bioactivities from pinus using green process. Molecules 29(22), 5295; doi:10.3390/molecules29225295 Chang, S.F., Hsieh, C.L. and Yen, G.C. 2008. The protective effect of Opuntia dillenii haw fruit against low-density lipoprotein peroxidation and its active compounds. Food Chem. 106(2), 569–575; doi:10.1016/j.foodchem.2007.06.017 Daham, H.R. 2021. Cell culture protocol: cell counting using a hemocytometer. Basrah: College of Veterinary Medicine University of Basrah. Dahham, S.S., Tabana, Y., Asif, M., Ahmed, M., Babu, D., Hassan, L.E., Ahamed, M.B.K., Sandai, D., Barakat, K., Siraki, A. and Majid, A.M.S.A. 2021. Β-Caryophyllene induces apoptosis and inhibits angiogenesis in colorectal cancer models. Int. J. Mol. Sci. 22(19), 10550; doi:10.3390/ijms221910550 Dolghi, A., Buzatu, R., Dobrescu, A., Olaru, F., Popescu, G.A., Marcovici, I., Pinzaru, I., Navolan, D., Cretu, O.M., Popescu, I. and Dehelean, C.A. 2021. Phytochemical analysis and in vitro cytotoxic activity against colorectal adenocarcinoma cells of Hippophae rhamnodies L., Cymbopogon citratus (D.C.) Stapf, and Ocimum basilicum L. essential oils. Plants 10(12), 2752; doi:10.3390/plants10122752 Dulay, R.M.R., Cabrera, E.C., Kalaw, S.P., Reyes, R.G. and Undan, J.R. 2022. Cytotoxicity of Lentinus isolates mycelial extracts on human colorectal carcinoma HCT-116 cells. J. Appl. Pharm. Sci. 12(6), 76–85; doi:10.7324/JAPS.2022.120608 Ermi Hikmawanti, N.P., Fatmawati, S. and Asri, A.W. 2021. The effect of ethanol concentrations as the extraction solvent on antioxidant activity of Katuk (Sauropus androgynus (L.) Merr.) leaves extracts. IOP Conf. Ser.: Earth Environ. Sci. 755(1), 12060. doi: 10.1088/1755-1315/755/1/012060 Fayazi, M., Sajjadi, M. and Mousavi, S.Y. 2024. Cytotoxicity evaluation of hydro-alcoholic extract of Prangos pabularia Lindl root on breast cancer MCF-7 cell line. J. Herbmed. Pharmacol. 14(1), 63–70; doi:10.34172/jhp.2025.52583 Heydarnezhad Asl, M., Ahmadi, A., Karari, K., Haghi, M., Tohidkia, M.R., Pasban, F. and Safaralizadeh, R. 2022. Anti-proliferative effects of Ocimum basilicum leaf aqueous extract on colon cancer cell lines and the expression of apoptotic genes. Jentashapir. J. Cell. Mol. Biol. 13(1), e123890; doi:10.5812/jjcmb-123890 Hjazi, A., Mohammed, S.N., Abosaoda, M.K., Ahmad, I., Rekha, M.M., Kundlas, M., Ullah, M.I., Abd, B., Ray, S. and Nathiya, D. 2025. Quercetin as a multi-targeted therapeutic agent in breast cancer: molecular targets and therapeutic potential. Med. Oncol. 42(8), 365; doi:10.1007/s12032-025-02907-x Hofmarcher, T., Manzano García, A., Wilking, N. and Lindgren, P. 2023. The Disease Burden and Economic Burden of Cancer in 9 Countries in the Middle East and Africa. Value Health Reg. Issues 37, 81–87; doi:10.1016/j.vhri.2023.05.005 Jagetia, G.C. and Baliga, M.S. 2004. The evaluation of nitric oxide scavenging activity of certain Indian medicinal plants in vitro: a preliminary study. J. Med. Food 7(3), 343–348; doi:10.1089/jmf.2004.7.343 Jovankić, J., Nikodijević, D., Blagojević, S., Radenković, N., Jakovljević, D., Grbović, F. and Cvetković, D. 2022. The biological activity of Ocimum minimum L. flowers on redox status parameters in HCT-116 colorectal carcinoma cells. Kragujevac J. Sci. 44, 155–168; doi:10.5937/KgJSci2244155J Juma, A.-M., Lazhar, Z. and Nabiha, M. 2025. Phytochemicals, bioactive compounds, and antimicrobial activities of Ocimum basilicum, Teucrium polium, Cleome amblyocarpa, and Caralluma arabica extracts: a comparative Omani study. Cell. Mol. Biol. 71(3), 134–145; doi:10.14715/cmb/2025.71.3.16 Makhlouf, N.A., Abdel-Gawad, M., Mahros, A.M., Lashen, S.A., Zaghloul, M., Eliwa, A., Elshemy, E.E., Ali-Eldin, Z., Abdeltawab, D., El-Raey, F., Omran, D., Khalaf, M., Fanous, N., Abdelmohsen, A.S., Abu-Elfatth, A., Abdelghani, M., Farouk, M., Abdelaziz, M. and Alboraie, M. 2021. Colorectal cancer in Arab world: a systematic review. World J. Gastrointestinal Oncol. 13(11), 1791–1798; doi:10.4251/wjgo.v13.i11.1791 Martin, T.A. and Jiang, W.G. 2009. Loss of tight junction barrier function and its role in cancer metastasis. Biochimica Et Biophysica Acta (BBA). -. Biomembranes. 1788(4), 872–891; doi:10.1016/j.bbamem.2008.11.005 Matsuda, T., Fujimoto, A. and Igarashi, Y. 2025. Colorectal cancer: epidemiology, risk factors, and public health strategies. Digestion 106(2), 91–99; doi:10.1159/000543921 Nelson, V.K., Sahoo, N.K., Sahu, M., Sudhan, H.H., Pullaiah, C.P. and Muralikrishna, K.S. 2020. In vitro anticancer activity of Eclipta alba whole plant extract on colon cancer cell HCT-116. BMC. Complement. Med. Ther. 20(1), 355; doi:10.1186/s12906-020-03118-9 Padauleng, N., Mustofa, M., Wahyuningsih, T. and Purnomosari, D. 2023. Chalcone-3 inhibits the proliferation of human breast cancer MDA-MB-231 cell line. Asian Pac. J. Cancer Prev. 24(2), 683–691; doi:10.31557/APJCP.2023.24.2.683 Panicker, N.G., Balhamar, S.O.M.S., Akhlaq, S., Qureshi, M.M., Rehman, N.U., Al-Harrasi, A., Hussain, J. and Mustafa, F. 2020. Organic extracts from Cleome droserifolia exhibit effective caspase-dependent anticancer activity. BMC. Complement. Med. Ther. 20(1), 74; doi:10.1186/s12906-020-2858-0 Phatak, R.S. and Hendre, A.S. 2014. Total antioxidant capacity (TAC) of fresh leaves of Kalanchoe pinnata. J. Pharmacogn. Phytochem. 2(5), 32–35. Poor, A.R.H., Mehdizadeh, P., Shokouh, S.M.H. and Amiri, M.M. 2025. Socioeconomic and political determinants of five common cancers in the MENA region. Acta Psychol. 258, 105137; doi:10.1016/j.actpsy.2025.105137 Puzzo, M., De Santo, M., Morelli, C., Leggio, A., Catalano, S. and Pasqua, L. 2025. Colorectal cancer: current and future therapeutic approaches and related technologies addressing multidrug strategies against multiple level resistance mechanisms. Int. J. Mol. Sci. 26(3), 313; doi:10.3390/ijms26031313 Rao, P.P., Shenoy Belle, V., Nayak, A.G., Kumar, N., Rao, V., Cheruku, S.P. and Prabhu, K. 2024. Evaluation of the merit of ethanolic extract of Annona reticulata as an anti-cancer agent in human colon cancer cell lines (HCT-116). F1000Research 12, 1571; doi:10.12688/f1000research.141542.2 Rao, Y., Chen, R., Ahmad, H. and Ahmad, U. 2024. Reverse zagreb indices and their application in the evaluation of physiochemical properties of anticancer/antibacterial drugs. ACS Omega 9(28), 31056–31080; doi:10.1021/acsomega.4c04409 Rasmi, Y., Khajeh, E., Kheradmand, F., Malekinejad, H., Aramwit, P., Saboory, E., Daeihassani, B. and Nasirzadeh, M. 2020. Crocetin suppresses the growth and migration in HCT-116 human colorectal cancer cells by activating the p-38 MAPK signaling pathway. Res. Pharm. Sci. 15(6), 592; doi:10.4103/1735-5362.301344 Sánchez, E., Dávila-Aviña, J., Castillo, S.L., Heredia, N., Vázquez-Alvarado, R. and García, S. 2014. Antibacterial and antioxidant activities in extracts of fully grown cladodes of 8 cultivars of Cactus pear. J. Food. Sci. 79(4), M659-M664; doi: 10.1111/1750-3841.12416 Sheng, X., Zhu, P., and Qin, J. 2017. Study of the biological effect of bufalin on anti- proliferation, adhesion and invasion in liver cancer cells. Remedy Publications LLC 2, 1363. Shimizu, T., Torres, M.P., Chakraborty, S., Souchek, J.J., Rachagani, S., Kaur, S., Macha, M., Ganti, A.K., Hauke, R.J. and Batra, S.K. 2013. Holy Basil leaf extract decreases tumorigenicity and metastasis of aggressive human pancreatic cancer cells in vitro and in vivo: potential role in therapy. Cancer. Lett. 336(2), 270–280; doi:10.1016/j.canlet.2013.03.017 Son, E.S., Kim, Y.O., Park, C.G., Park, K.H., Jeong, S.H., Park, J.W. and Kim, S.H. 2017. Coix lacryma-jobi var. ma-yuen Stapf sprout extract has anti-metastatic activity in colon cancer cells in vitro. BMC. Complement. Altern. Med. 17(1), 486; doi:10.1186/s12906-017-1990-y Tatsuta, K., Sakata, M., Kojima, T., Booka, E., Kurachi, K. and Takeuchi, H. 2025. Updated insights into the impact of adjuvant chemotherapy on recurrence and survival after curative resection of liver or lung metastases in colorectal cancer: a rapid review and meta-analysis. World J. Surg. Oncol. 23(1), 56; doi:10.1186/s12957-025-03714-4 World Flora Online. 2024. An online flora of all known plants. Available via http://www.worldfloraonline.org Wu, X., Lin, M., Li, Y., Zhao, X. and Yan, F. 2009. Effects of DMEM and RPMI 1640 on the biological behavior of dog periosteum-derived cells. Cytotechnology 59(2), 103–111; doi:10.1007/s10616-009-9200-5 Xiang, L., He, B., Liu, Q., Hu, D., Liao, W., Li, R., Peng, X., Wang, Q. and Zhao, G. 2020. Antitumor effects of curcumin on the proliferation, migration and apoptosis of human colorectal carcinoma HCT 116 cells. Oncol. Rep. 44(5), 1997–2008; doi:10.3892/or.2020.7765 Yeung, T.M., Gandhi, S.C., Wilding, J.L., Muschel, R. and Bodmer, W.F. 2010. Cancer stem cells from colorectal cancer-derived cell lines. Proc. Natl. Acad. Sci. U. S. A. 107(8), 3722–3727; doi:10.1073/pnas.0915135107 Yıldırım, A., Mavi, A. and Kara, A.A. 2001. Determination of antioxidant and antimicrobial activities of Rumex crispus L. extracts. J. Agric. Food. Chem. 49(8), 4083–4089; doi:10.1021/jf0103572 Zarlaha, A., Kourkoumelis, N., Stanojkovic, T.P. and Kovala-Demertzi, D. 2014. Cytotoxic activity of essential oil and extracts of Ocimum basilicum against human carcinoma cells. Molecular docking study of isoeugenol as a potent COX and LOX inhibitor. Dig. J. Nanomater. Biostruct. 9(3), 907–917. Zhakipbekov, K., Turgumbayeva, A., Akhelova, S., Bekmuratova, K., Blinova, O., Utegenova, G., Shertaeva, K., Sadykov, N., Tastambek, K., Saginbazarova, A., Urazgaliyev, K., Tulegenova, G., Zhalimova, Z. and Karasova, Z. 2024. Antimicrobial and other pharmacological properties of Ocimum basilicum, Lamiaceae. Molecules 29(2), 388; doi:10.3390/molecules29020388 Zourgui, M.N., Hfaiedh, M., Brahmi, D., Affi, W., Gharsallah, N., Zourgui, L. and Amri, M. 2020. Phytochemical screening, antioxidant, and antimicrobial activities of Opuntia streptacantha fruit skin. J. Food Meas. Charact. 14(5), 2721–2733; doi:10.1007/s11694-020-00518-w | ||
| How to Cite this Article |
| Pubmed Style Al-mutaani J, Zorgui T, Zourgui L, Alqawi O, Missaoui N. Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Vet. J.. 2025; 15(12): 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 Web Style Al-mutaani J, Zorgui T, Zourgui L, Alqawi O, Missaoui N. Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. https://www.openveterinaryjournal.com/?mno=268451 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.33 AMA (American Medical Association) Style Al-mutaani J, Zorgui T, Zourgui L, Alqawi O, Missaoui N. Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Vet. J.. 2025; 15(12): 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 Vancouver/ICMJE Style Al-mutaani J, Zorgui T, Zourgui L, Alqawi O, Missaoui N. Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 Harvard Style Al-mutaani, J., Zorgui, . T., Zourgui, . L., Alqawi, . O. & Missaoui, . N. (2025) Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Vet. J., 15 (12), 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 Turabian Style Al-mutaani, Juma, Tahani Zorgui, Lazhar Zourgui, Omar Alqawi, and Nabiha Missaoui. 2025. Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Veterinary Journal, 15 (12), 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 Chicago Style Al-mutaani, Juma, Tahani Zorgui, Lazhar Zourgui, Omar Alqawi, and Nabiha Missaoui. "Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells." Open Veterinary Journal 15 (2025), 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 MLA (The Modern Language Association) Style Al-mutaani, Juma, Tahani Zorgui, Lazhar Zourgui, Omar Alqawi, and Nabiha Missaoui. "Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells." Open Veterinary Journal 15.12 (2025), 6487-6496. Print. doi:10.5455/OVJ.2025.v15.i12.33 APA (American Psychological Association) Style Al-mutaani, J., Zorgui, . T., Zourgui, . L., Alqawi, . O. & Missaoui, . N. (2025) Anticancer properties of ethanolic extract of Omani Ocimum basilicum in HCT116 colorectal cancer cells. Open Veterinary Journal, 15 (12), 6487-6496. doi:10.5455/OVJ.2025.v15.i12.33 |