| Research Article | ||
Open Vet. J.. 2026; 16(2): 987-998 Open Veterinary Journal, (2026), Vol. 16(2):987-998 Research Article Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smokeBrian Christian Sarniem1, Tyagita Hartady1,2*, Mas Rizky A.A Syamsunarno2, Shafia Khairani1,2, Andi Hiroyuki1,2, Nur Atik2, Septiyani Septiyani1,2, Faisal Amri Satrio1,2 and Briliant Maharani11Veterinary Medicine Study Program, Faculty of Medicine, Padjadjaran University, Bandung, Indonesia 2Department of Biomedical Science, Faculty of Medicine, Padjadjaran University, Bandung, Indonesia *Corresponding Author: Tyagita Hartady. Veterinary Medicine Study Program, Faculty of Medicine, Padjadjaran University, Bandung, Indonesia. Email: tyagita [at] unpad.ac.id Submitted: 02/07/2025 Revised: 06/04/2025 Accepted: 30/12/2025 Published: 28/02/2026 © 2026 Open Veterinary Journal
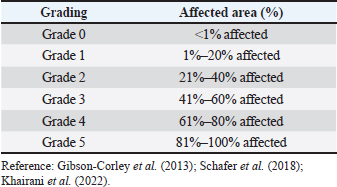
AbstractBackground: Exposure to cigarette smoke, whether from traditional or electronic sources, is a growing health concern affecting both humans and animals. Although electronic cigarettes (ECs) (vapes) are often marketed as a safer alternative, evidence supporting their reduced toxicity is still lacking. Aim: This study aimed to evaluate and compare the pathological effects of exposure to traditional cigarette smoke and EC vapor in rats, particularly focusing on respiratory and reproductive organs. Methods: A total of 32 male rats were randomly assigned to four groups (n=8 per group): control group (no exposure), traditional cigarette smoke exposure group, an EC vapor exposure group, and a combined exposure group. Smoke exposure was administered daily for a specific period. Macroscopic and microscopic evaluations of lung and testicular tissues were performed using a semiquantitative scoring system. Statistical analysis was performed using analysis of variance, with p < 0.05 considered significant. Results: Significant pathological differences (p < 0.05) were observed between all exposed groups and the control group. However, no significant differences (p > 0.05) were found among the three smoke-exposed groups. Common pathological findings included nodules, fibrosis, emphysema, testicular vacuolization, decreased spermatogenesis, and degenerative changes. Conclusion: Exposure to both traditional and ECs induced comparable pathological changes in lung and testicular tissues. These findings suggest that ECs may pose similar health risks as conventional cigarettes, emphasizing the need for caution in their use and further investigation into their safety profile. Keywords: Cigarette, Electronic cigarette, Pathology, Toxicology, Secondhand smoker, Experimental. IntroductionElectronic cigarette (EC) consumption has increased over the years, according to the Global Adult Tobacco Survey (2021 a,b), which states that 6.2 million people (3.0%) in Indonesia use EC products, representing a 2.7% increase from 2011. In 2021, 70.2 million people (34.5%) used cigarette tobacco products. In addition, there is a difference between the male and female smokers by 62% for tobacco cigarettes and 5.5% for tobacco cigarettes and ECs, respectively. These data show the risks posed to active smokers, passive smokers, and the surrounding environment. According to publications from the World Health Organization (2020), the death toll in Indonesia due to diseases that are correlated with smoking activities reaches 225.700 people each year. The effects of nicotine in these two products can be addictive (Indonesia Food and Drug Administration, 2017) and affect animals within the vicinity. Smoke from cigarettes can settle on animal hair and water, which is toxic to Carassius auratus (Swanson, 2013). Cigarette smoke can cause physiological disorders in gestational animals and potentially interfere with brain formation (Slotkin et al., 2016), inducing lymphoma, asthma, and squamous cell carcinoma in cats, and inducing nasal and lung cancer in dogs. In addition, residual products from cigarettes, such as cigarette butts and residual tobacco, can be lethal when consumed by animals (Swanson, 2013). Exposure to tobacco smoke and ECs is an important topic for further review, both in the field of animal and human health. The interest in knowing the negative impact of tobacco and ECs on health is the basis of this research. This study aims to examine the transition between tobacco cigarettes, ECs, and their comparisons. Specifically, it seeks to investigate differences between tobacco and ECs, the transition between the two, and their comparisons based on pathological findings. Materials and MethodsThe research design applied in this study is a true experiment, semiquantitative, and randomized. The main goal of this study is to prove whether vaping is safer than smoking based on the pathological findings of the respiratory and reproductive organs from 1-month exposure to tobacco and EC smoke. All rats in this experimental study were acclimated for 6 days in a controlled laboratory environment. The temperature was set at 25°C, humidity was around 70%, a 12-hour dark-light cycle was applied, a clean water supply and pellets were provided ad libitum (CitraFeed® RatBio), and bedding was changed every 2 days. Food was changed daily and replaced immediately if contamination, such as litter, wetness, or bugs, occurred. The activity is monitored twice daily to safeguard the safety and well-being of rats and to maintain optimal laboratory conditions. The inclusion criteria for this study are that the animals obtained must be healthy, Wistar male rats, and have never been used in a previous experiment. The exclusion criteria were rats weighing >300 and 200 g. This study used a male Wistar Rattus norvegicus strain, approximately 10 weeks old, with an average weight of approximately 268 g (SD ± 26.05). The rats were bred in the Laboratory of Pharmacology and Therapy, Faculty of Medicine, Universitas Padjadjaran, and met all the requirements as experimental animals for the study. In this research, rats were divided into four groups, namely Control (C), Tobacco (T), EC, and Shifting, the transition of exposure from tobacco cigarette smoke to EC vapor (S). The shifting category was selected based on human behavior patterns, specifically the transition from conventional tobacco smoking to EC use. Exposure to cigarette smoke was conducted in the morning and afternoon. Exposure was delivered using a whole-body method. The animals were placed in cages inside an exposure chamber, where they were exposed to smoke in their normal environment with free access to feed and water. This study used Federer’s formula and corrected sample size to calculate the sample size for each group and anticipate potential experiment errors/dropout criteria, such as rats becoming sick, abnormalities being observed, clinical signs being present, or high stress levels. The T group was exposed to six cigarettes (“DC” Kretek, 2.4 mg nicotine, and 38 mg tar/cigarette) per day, and the rats in the EC group were exposed to 1.2 ml EC liquid (“FL”, 0mg nicotine) per day for 30 consecutive days. Meanwhile, the S group received six cigarettes/day for 15 days and then switched to ECs 1.2 ml/day for 15 days. Our team consists of experienced pathologists and researchers. Anesthesia procedures, including cervical dislocation methods, were performed in accordance with established guidelines. The laboratory was prepared the day before euthanasia was conducted. The laboratory, including all tables, chairs, walls, and floors, was thoroughly cleaned with disinfectant. Subsequently, the wall and table surfaces were covered with disposable plastic to ensure personnel safety and minimize contamination. Rats were placed in a safe, non-transparent, and isolated container to ensure that they were not stressed. The rat is injected intraperitoneally using Ketamine and Xylazine overdose (approved method according to AVMA Guidelines, 2020) and waits until it takes effect. Necropsy was performed with a midline incision from the thorax. The rats were isolated and performed by professionals. The scoring method was used to determine the severity level of each sample. An ordinal (percentage) grading system ranging from 0 to 5 was applied for both macroscopic pathological (MP) and histopathological (HP) findings (Table 1). A binocular Olympus CX23 microscope was used for the HP examination. Six HP parameters were assessed in the respiratory system, including emphysema, thickening of the alveolar septa, obstruction, fibrosis, tissue destruction, and haemorrhage (Ziad et al., 2013; Reyna-Sepúlveda et al., 2016; Phillips et al., 2018; Butt et al., 2019; Jawad et al., 2019; Monica et al., 2019; Wawryk-Gawda et al., 2019; Kaous et al., 2020; Smith et al., 2021). Six HP parameters were evaluated in the reproductive system, including vacuolization, degeneration, decreased spermatogenesis, tissue destruction, necrosis, and congestion (Kolawole et al., 2015; He et al., 2016; Wawryk-Gawda et al., 2019; Szumilas et al., 2020). Data analysis was performed using statistical methods. The Shapiro–Wilk normality test was used in this study, and One-Way analysis of variance was used to analyze differences between the four groups. Then, Tukey’s honest significant difference (HSD) post hoc test was performed in SPSS version 25. Scattered plots and histograms were used to visualize the distribution of data. The Shapiro–Wilk normality test indicated whether the test was insignificant. Therefore, no abnormal data were found among the group or the whole sample. After confirming the Shapiro–Wilk test, one-way analysis of variance (ANOVA) was used to identify mean differences between and within groups. Tukey’s HSD post hoc tests were used only when the ANOVA showed a p-value < 0.05 to compare the means of paired groups. Table 1. Grading for pathological changes.
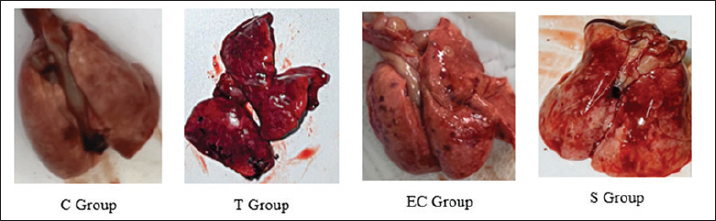
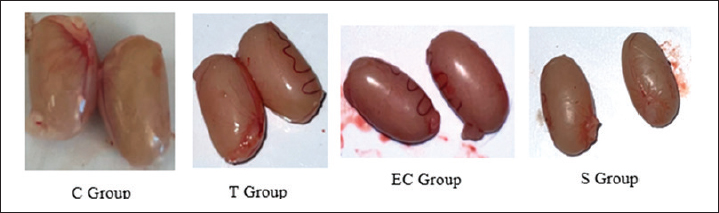
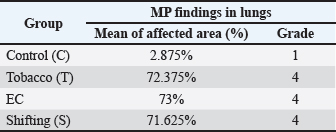
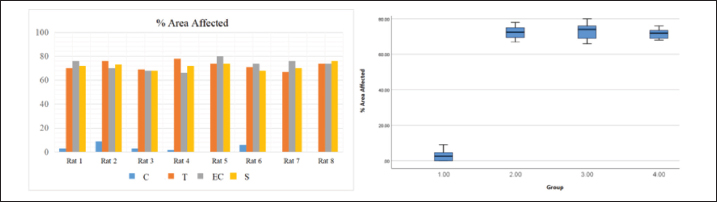
HypothesisThe study’s hypothesis serves as a provisional statement to establish relationships among variables, ensure the research’s replicability, and form logical conclusions. This study proposes that there is no significant difference in the pathological findings of the respiratory and reproductive organs of male rats exposed to tobacco cigarette smoke, EC vapor, and the transition between the two types of smoke (Ha). Alternatively, a significant difference in these pathological changes was observed among the exposure groups (Hₐ). The experimental groups consisted of a control group (C), a tobacco cigarette smoke exposure group (T), an EC vapor exposure group (EC), and a transitional cigarette smoke exposure group (S). Ethical approvalThis study was approved by the Ethics Committee of the Faculty of Medicine, Padjadjaran University, with ethical clearance number 589/UN6.KEP/EC/2023. All laboratory animals used in this experiment exhibited full adherence to the five animal welfare principles. Good laboratory practice is also conducted in this research. The study was conducted from April to May 2023 at the Laboratory of Padjadjaran Veterinary Hospital and in June 2023 at the Pathology Laboratory of Subang Veterinary Center, Subang Regency, Indonesia. ResultLung and testicular organ weightThe organ weight results from the lung and testicle provide new information that could indicate pathological changes, such as fluid accumulation (Graphs 1 and 2). Organ weight parameters were established at 1.0–1.2 g for each pulmonary lobe, which were considered within the normal range, and 2.9–3.2 g for both testicular lobes. The mean pulmonary organ weight was within the normal range (1.156 g) and showed no statistical difference compared to each group (p=0.687). The testicular organ weight was within the standard parameter (2.916 g), resulting in a non-significant difference (p=0.213). MP findingsSmoke-exposed rats show noticeable differences compared with the C group. The T, EC, and S groups showed the same result (Grade 4), affecting 72.38%, 73%, and 71.63% of pathological changes around the lungs, respectively. Compared with the C group (Grade 1), only about 3% of the lung was damaged (Graph 2). Statistically significant p-values (p=0.000) were obtained using Tukey’s honest significant difference test (Graph 3). Black spots and darkening are the main findings in the T group, whereas nodules were observed in the EC group, resulting in pale-colored lungs. MP findings in the S group showed intermediate characteristics between those of the T and EC groups (Fig. 1). Similar results were observed across all groups in the reproductive organ (testicle) and showed normal anatomical structure from both lobes and at all angles (Fig. 2). HP findingsRespiratory systemMicroscopic findings revealed significant differences between the smoke-exposed and control groups (p < 0.05). The respiratory semiquantitative scoring is shown (Table 3). Notable findings were identified in the T, EC, and S groups, including brown to black pigments distributed within the alveolar walls and macrophages. Previous research found the same findings from human autopsy cases with a history of heavy vaping for year (Butt et al., 2019). Microscopic findings show significant damage in the smoke-exposed groups, including emphysematous changes in the alveolar structure, thickening of the alveolar septa, obstruction of alveolar lumens, fibrosis around the alveolar structure, and destructive changes, as shown in the HP pictures below (Figs. 3–5). Mild to severely damaged alveolar walls, indicating emphysematous changesSignificant results were found (p=0.000) compared to the control group. The T and S groups achieved the same semiquantitative parameter result (Lesion score: 3 ± 0.189 and 3 ± 0.267). Moderate to heavy alveolar septal thickening and chronic inflamation were found around the septal wallA significant difference was observed (p=0.000) between the smoke-exposed and control groups. Mediocre obstructive changes in the alveolar lumen led to increased resistance of the airwayThe obstructive changes in the lumen ranged from 10% to 20%, and in some cases, they reached up to 30%, as illustrated in the representative images of Group 4. A significant difference was found (p=0.000) compared with the control group, whereas no significant difference was found (p=1.000) compared with the smoke-exposed group. Fibrosis around the parenchymal tissue was found; the highest score was is in the EC groupThe EC and S groups were significantly different from the control group (p=0.000 and p=0.047, respectively), but the T group was not (p=0.332). Statistically, the difference between the EC group and the S group was also non-significant (p=0.098), and the S group compared with the T group showed the same result (p=0.732). The fibrosis lesion in the semiquantitative parameter showed that the highest score was obtained in the EC group (1.875 ± 0.398), followed by the S group (1 ± 0.189) and the T group (0.625 ± 0.263). Destructive damage to the alveolar and parenchymal tissues was foundSignificant results were obtained (p=0.000) against the control group; thus, non-significant results (p=0.778 and p=0.964) were found against the smoke exposure groups.
Fig. 1. Gross pathological finding in lung.
Fig. 2. Gross pathological finding in testicle. Table 2. MP findings in lung.
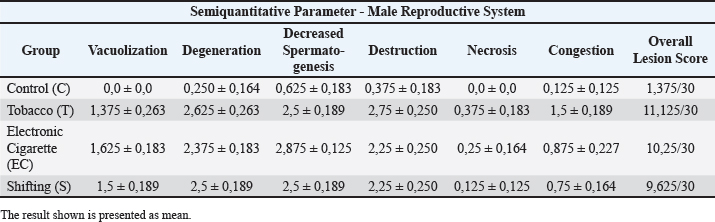
Hemorrhages in the smoke-exposed group indicates some capillary damagesThe T, EC, and S groups have similar results from the semiquantitative parameter (Lesion score: 3.25 ± 0.25, 3.125 ± 0.227, and 3.5 ± 0.189). Significant results were found (p=0.000 and p=0.001) compared with the control group, and non-significant results (p=0.568, p=0.820, and p=0.972) were observed in comparison with the smoke-exposed group. Pulmonary capillary inflammation was found in the T and EC groups. Pulmonary capillary inflammation was found in T and EC groupsVasculitis is a chronic inflammatory reaction that occurs in the body’s blood vessels of the body and was found in the T and EC groups. Reproductive systemFindings from microscopic observation reveal a massive difference between the smoke exposure group and the control group, which was statistically significant (p < 0.05). The male reproductive semiquantitative results are shown in Table 4. Microscopically, various abnormalities with varying degrees of severity were observed, including vacuolization, degeneration, decreased spermatogenesis, destruction, obstruction, and congestion, which could be visualized using a microscope with 40× and 100× magnification. HP findings are presented in Figures 6 and 7. Mild vacuolization of the seminiferous tubules is observed in the smoke-exposed groupsThe T, EC, and S groups have similar results from the semiquantitative parameter (Lesion score: 1.35 ± 0.25, 1.625 ± 0.183, and 1.5 ± 0.189). Significant results were found (p=0.000) compared with the control group, while non-significant results were found (p=0.778 and p=0.964) when compared with the smoke-exposed group. Destructive changes and degeneration of the seminiferous tubule indicate extensive damage caused by cigarette substancesThe degeneration parameter showed similar results for the T, EC, and S groups (Lesion score: 2.65 ± 0.263, 2.375 ± 0.183, and 2.5 ± 0.189). The destruction parameter was similar (Lesion score: 3.125 ± 0.227, 3 ± 0.189, and 2.875 ± 0.125). Tukey’s HSD showed significant results (p=0.000) compared with the control group, whereas non-significant results were found (degeneration, p=0.820 and p=0.972; destruction, p=0.449 and p=1.000) compared to the smoke-exposed group. Table 3. Semiquantitative parameter in respiratory system.
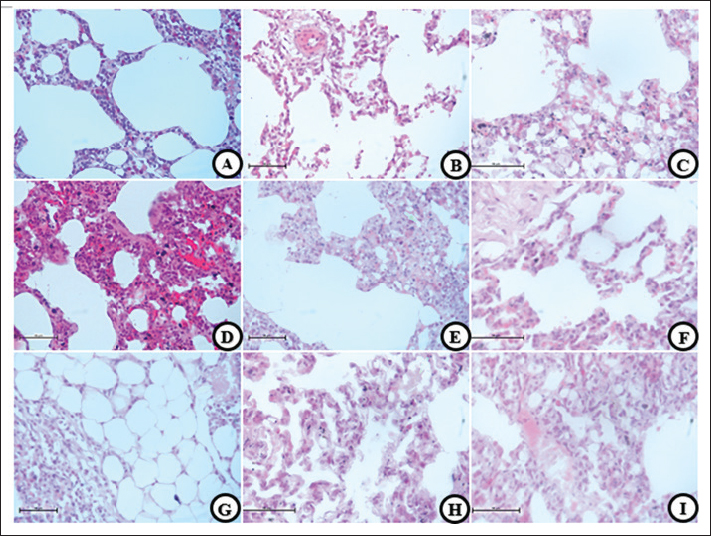
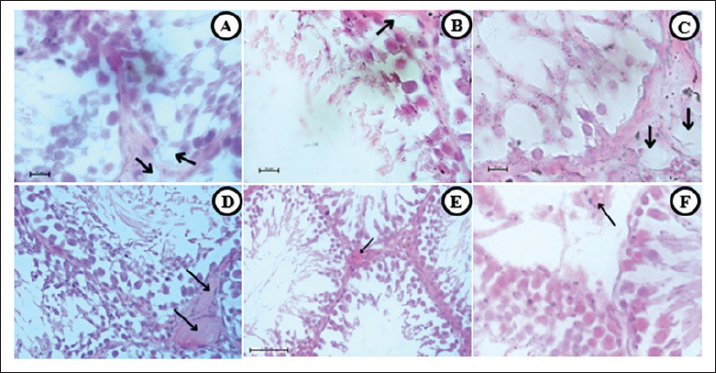
Fig. 3. HP changes. (A, D, and G) T group,(B, E, and H) EC group, and (C, F, and I) S group. Emphysematous changes are shown in all smoke groups (A, C, E, and F) characterized by alveolar lumen widening and decimation of the septal wall. Septal thickening (D, E, and F) characterized by septal wall width increases thus infiltration of chronic inflammatory cells. Obstruction (G, H, and I) shows some fluid accumulation in alveolar lumen. Fibrosis (E, G, H, and I) were characterized by hairy-looking connective tissue along the parenchymal wall. 40×, H&E.
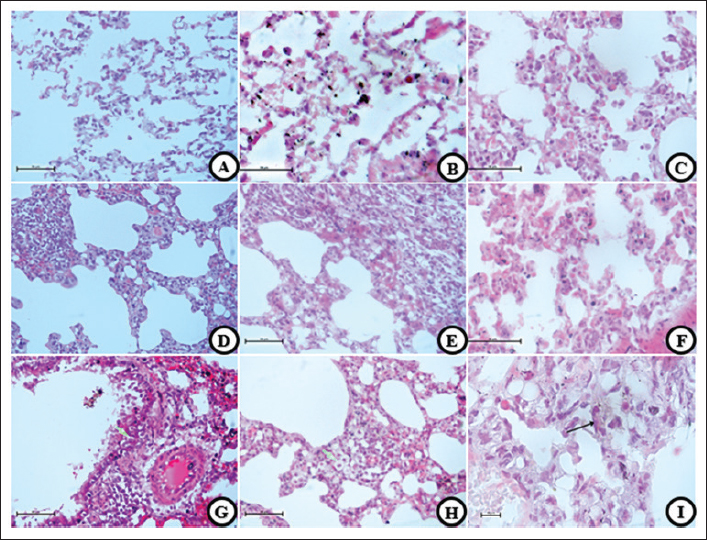
Fig. 4. HP changes. (A, D, and G) T group, (B, E, and H) EC group, and (C, F, and I) S group. Destructive damages were shown in all smoke exposed groups (A, B, and C) characterized by multiple pathological changes, such as damaged alveolar wall, inflammatory cells infiltration, and alveolar lumen widening. Hemorrhages (D–F) characterized by red blood cell that was found outside the vascular. Pulmonary Vacuolization (G–I) shows hydropic degeneration in pulmonary cells and balloon look-alike appearance. 40× and 100× (I), H&E. Decreased spermatogenesis activity and abnormal spermatozoa are the result of all the testicle damagesThe results from the semiquantitative parameter show that the EC group has the highest score, followed by the T, S, and C groups (EC: 2.875 ± 0.125, T and S: 2.5 ± 0.189, and C: 0.625 ± 0.183). Significant results were found (p=0.000) compared with the control group, whereas non-significant results were found (p=0.435 and p=1.000) when compared with the smoke-exposed group. Mild cellular death in Sertoli and Leydig cells are present in all smoke-exposed groupsIn this parameter, all groups show similar results (severity grade: 0) and non-significant results (p=0.241, p=0.581, and p=0.918) when compared within the smoke-exposed group and against the control group. Vascular congestion and capillaritis indicate decreased blood flow to the testicular tissueThe semiquantitative parameter shows that the T group has the highest lesion score, followed by the EC, S, and C groups. The one-way analysis of variance test yields a significant result (p=0.000) between groups. DiscussionThe reaction that causes this particular pathological finding begins in the immune system. Smoke-induced cellular signaling triggers an immunological response in the alveolar walls, helping to reduce damage from oxidative stress and cellular injury. The other mechanism is vasodilatation to compensate for the reduction in oxygen diffusion capability caused by oxidative stress. Vasodilatation helps increase the blood flow to the tissue. Alveolar septal thickening negatively affects the respiratory system, reducing the lumen space and increasing airway resistance. Obstruction can result from a reduction in lumen size, decreased parenchymal elasticity, tumors, or objects that block it, such as tumors, cysts, foreign bodies, and mucous exudates (Lechner et al., 2012). Fibrosis is characterized by the formation of connective tissue caused by the accumulation of excess extracellular matrix fluid. It is activated by repeated damage to alveolar epithelial cells induced by reactive oxygen species (ROS) (Wuyts et al., 2012).
Fig. 5. HP changes. (A and D) T group, (B and E) EC group, and (C) S group. Brown to black unknown substance were found in all smoke groups (A–C). This findings mostly found in the alveolar wall, alveolar lumen, and some go through the parenchymal wall. Vasculitis (D and E) was found only in T and EC group, characterized by inflammation around the vascular wall thus thickening of the vascular wall. 40×, 100× (D and E) H&E. Table 4. Semiquantitative parameter in male reproductive system.
Destruction is classified by the presence of two abnormalities in the alveolar wall, obvious damage, and an HP picture that can indicate an emphysema condition (Reyna-Sepúlveda et al., 2016). The pathogenesis of alveolar and parenchymal tissue destruction is similar to that described above, and is caused by smoke components containing ROS, toxins, chemicals, tar, and additional flavorings that interfere with cellular activity. Decreased tissue elasticity impacts cellular dysfunction and cell death. It leads to tissue destruction, which is accompanied by other pathological findings (Reyna-Sepúlveda et al., 2016). Haemorrhage findings are characterized by a reddish color in the cellular arrangement and not in the vascular or capillary arrangement. These findings indicate bleeding caused by vascular wall damage due to the toxic components of cigarettes. However, from another point of view, post-mortem bleeding, such as the incision made during organ resection from the body, can cause hemorrhages.
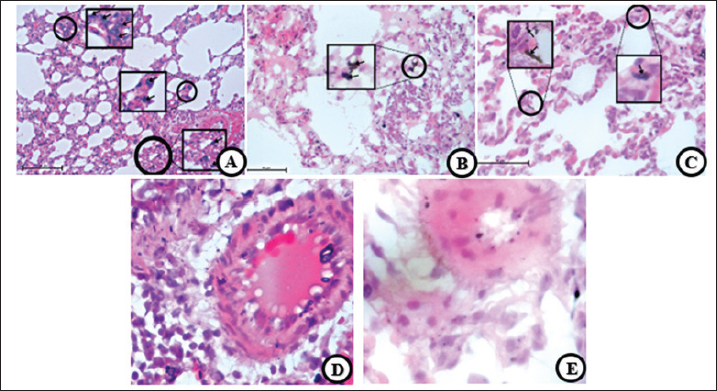
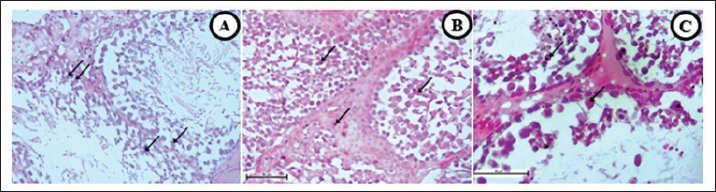
Fig. 6. H changes. (A and D) T group, (B and E) EC group, and (C and F) S gProup. Vacuolization (large arrow) along seminiferous tubules were found in all smoke groups (A–C). Decreased spematogenesis activity with abnormal sperm also present. Vascular congestion (D–F) are present (thin arrow) and characterized by clumps of red blood cell filling the vascular lumen. In addition, another pathological changes was also found, namely destruction, degeneration, and separation of seminiferous tubules. 40× (D–F), 100× (A–C) H&E.
Fig. 7. HP changes. (A) T group, (B) EC group, and (C) S group. Necrotic cells (arrow) were found in all smoke groups (A–C), followed by thickening of the seminiferous tubule wall (B), thus the declined activity of spematogenesis, destructive damages, and abnormal sperms. 40, H&E. Vacuolization, vacuolar degeneration, hydropic degeneration, or ballooning degeneration is characterized by an increase in cell size and a balloon-like cytoplasm. The characteristic of vacuolization is that the cytoplasm changes in color to bluish–gray and appears pale (Cesta et al., 2023). The negative effect of vacuolization in pneumocytes is decreased oxygen diffusion capability, impaired cellular activity, and cell death. Brown to black unknown substances with a size of approximately 10 microns were spread across the lumen of alveoli, parenchymal tissue, pulmonary capillaries, and alveolar macrophages. A previous study also found similar substances in a patient who vapes daily (Butt et al., 2019) and a biopsy from a heavy chronic smoker (NMPV, 2017). Although tobacco and ECs may have different residual products, they have the same effect on the lungs. The last finding in the discussion of lung organs is that some rats had vasculitis or pulmonary capillaritis. In addition, vasculitis can be used to describe the incidence of both primary and secondary diseases (Jatwani et al., 2023). Vacuolated structures in the epithelium of the seminiferous tubules characterize vacuolization. It can disrupt spermatogenesis, thereby reducing sperm production (Xie et al., 2014). Vacuolization can be caused by oxidative stress due to ROS (Acikel-Elmas et al., 2023), which is present in tobacco and ECs. These findings can be attributed to various factors, including trauma, changes in temperature, environment, nutrition, stress, and toxins (Koziol and Palmer, 2023). In addition, other studies have reported that degeneration can be accompanied by vacuolization, atrophy, apoptosis, germ cells’ multinucleation, and germinal layer structure (Creasy et al., 2023). The decline in spermatogenesis is caused by nicotine, ROS, and various toxic components that circulate in the blood, leading to antioxidant and hormonal imbalances and, over a long period, DNA damage (Cui et al., 2016). ROS disrupts the hypothalamic–pituitary–gonadal axis, leading to alterations in luteinizing hormone and testosterone (Acikel-Elmas et al., 2023). Necrosis, which typically occurs in the testes, is likely caused by ischemia. In contrast, the other factor includes hemodynamic changes, vascular endothelial damage, or decreased blood flow to the testicular region, which can cause ischemia (Creasy and Chapin, 2018). Some factors likely contribute to the minimal severity of testicular necrosis. First, rat bodies can manage cellular death through a clearance mechanism, and mild vascular congestion leads to good blood perfusion. The exposure time and nicotine dosage are also insufficient to cause cell necrosis. Vascular congestion refers to excessive fluid accumulation within the vascular lumen that negatively impacts testicular cells and tissues. Decent blood flow is required to perform normal activities and maintain homeostasis, such as spermatogenesis and sexual hormone secretion. Vascular congestion leads to reduced blood flow and decreased blood and oxygen supply. Cigarettes (tobacco or electronic) are products that satisfy the users (Food and Drug Administration, 2016), starting from inhaling the smoke and emitting residual smoke. The smoke that arises from smoking activities can produce residual substances that are likely to be inhaled by the surrounding humans and animals. This phenomenon is known as secondhand smoke. Interactions involve the diffusion of various components of cigarette smoke, which are spread throughout the body via the vascular system. The free radical ROS causes oxidative stress, which can damage cellular structures and interfere with cell function, leading to abnormalities in cell function, tissue damage, and potentially carcinogenic effects (American Lung Association, 2020). The previous study is in line with our pathological findings as described above, lungs that are exposed to tobacco cigarette smoke experienced hemorrhage, thickening, and deposition of collagen in the alveolar septa, found intrabronchial mucus, erythrocytes, macrophages (Zhu et al., 2012; Ziad et al., 2013; Phillips et al., 2018; Jawad et al., 2019; Wawryk-Gawda et al., 2019) peribronchial congestion, perivascular congestion (Jatwani et al., 2023), monocyte infiltration (Reyna-Sepúlveda et al., 2016), alveolar inflammation, obstruction, destruction, increased airway resistance, accompanied by decreased ability to diffuse with oxygen (Reyna-Sepúlveda et al., 2016), edema, alveolar collapse, until signs of atelectasis are found (Sousa et al., 2019). On the other hand, lungs that are exposed to ECs experience similar pathological symptoms such as damage to alveolar and hyaline membranes, deposition of fibrin, accumulation of macrophages, widening of the cytoplasm, fibrosis (Smith et al., 2021), infiltration of eosinophils, erythrocytes and mononuclear cells, thickening of septa alveolaris, hyperemia, and collagen deposition in alveolar septa and peribronchiolar areas (Wawryk-Gawda et al., 2019; Wawryk-Gawda et al., 2020) pneumocyte hyperplasia, interstitial lymphocytic infiltration, multifocal interstitial lymphocytic pneumonitis (Kaous et al., 2020), mucosal epithelial degeneration and inflammation, necrosis of the epithelium, endotheliosis, thrombosis (Monica et al., 2019), as well as macrophages vacuolization and pneumocytes (Wawryk-Gawda et al., 2020). These pathological changes can lead to many diseases, some of the disease, including chronic obstructive pulmonary disease, interstitial lung disease, acute eosinophilic pneumonia, bronchiolitis, desquamative interstitial pneumonia (Sousa et al., 2019), airspace enlargement with fibrosis (AEF), combined pulmonary fibrosis and emphysema (CPFE) (Iwasawa et al., 2018), pulmonary tumors/cancer, idiopathic pulmonary fibrosis (IPF), acute and chronic hypoxemia, endothelial dysfunction (Nevzorova et al., 2018), smoking-related interstitial fibrosis (SRIF) (Wick, 2018). ConclusionThis study provides HP evidence that both tobacco and EC smoke exposure induce significant structural alterations in the lungs and testes of male rats. Exposure to ECs produced the highest degree of pulmonary fibrosis (p < 0.05) compared with the control, tobacco, and transition groups, indicating pronounced alveolar parenchymal fibrotic remodeling. In contrast, tobacco smoke exposure resulted in significant vascular congestion within testicular tissue (p < 0.05), suggesting impaired testicular perfusion and potential disruption of spermatogenic activity. Collectively, these findings demonstrate that EC vapor is not a harmless alternative to tobacco smoke, as both exposures elicit comparable degenerative and inflammatory responses in vital organ systems. Continued investigation using extended exposure durations, diverse dosages, and comprehensive molecular analyses is recommended to further elucidate the mechanisms underlying these pathological effects and their long-term health implications. Research limitationsThe study’s findings are limited to the structural observations of the lung and testicular organs. No additional tests were conducted, such as X-ray imaging, immunohistochemistry, or other staining techniques, were conducted on the HP preparations. Future studies are recommended to explore different dosages, animal models, and exposure durations using the same or different substances. Furthermore, this study did not include an analysis of the chemical composition of the electronic and tobacco cigarettes used. AcknowledgmentsThe author would like to thank the Department of Basic Medical Sciences, Veterinary Medicine Study Program, Faculty of Medicine, Padjadjaran University, for facilitating the implementation of this research activity. Conflict of interestThe authors have no conflicts of interest to declare in this article. FundingThe authors would like to thank the Ministry of Education, Culture, Research, and Technology of the Republic of Indonesia and Research Fund No.1549/UN.6.3.1/PT.00/2023 Padjadjaran University for the funds and facilities provided by Dr.drh.Tyagita, M.VSc, during the research. Authors’ contributionsThe author conducted the research and writing of this article, with contributions from data collection and analysis. Writing the manuscript: Brian Christian Sarniem and Briliant Maharani: Collecting data and analysis: Brian Christian Sarniem, Mas Rizky A.A Syamsunarno, Tyagita Hartady: Data Analysis and Review Manuscript; Shafia Khairani, Nur Atik, Septiyani, Andi Hiroyuki, and Faisal Amri Satrio. Data availabilityAll data were provided in the manuscript. ReferefrncesAcikel-Elmas, M.,Algilani, S.A., Sahin, B., Bingol Ozakpinar, O., Gecim, M., Koroglu, K. and Arbak, S. 2023. Apocynin ameliorates monosodium glutamate-induced testis damage by impaired blood-testis barrier and oxidative stress parameters. Life 13, 822. American Lung Association. 2020. What’s in a cigarette? American Lung Association. Butt, Y.M., Smith, M.L., Tazelaar, H.D., Vaszar, L.T., Swanson, K.L., Cecchini, M.J., Boland, J.M., Bois, M.C., Boyum, J.H., Froemming, A.T., Khoor, A., Mira- Avendano, I., Patel, A. and Larsen, B.T. 2019. Pathology of vaping-associated lung injury. N. Engl. J. Med. 381, 1780–1781; doi: 10.10 56/NEJMc1913069 Cesta, M.F., Dixon, D., Herbert, R.A. and Staska, L.M. 2023. Lung – vacuolization, cytoplasmic. National Toxicology Program – Nonneoplastic Lesion Atlas [Online]. Available via https://ntp.niehs.nih.gov/sites/default/files/nnl/respiratory/lung/vaccyto/lung-vacuolization-cytoplasmic-508.pdf(Accessed 28 June 2023) Creasy, D.M. and Chapin, R.E. 2018. Male reproductive system. In Fundamentals of toxicologic pathology, 3rd ed. Oxford, UK: Elsevier. Creasy, D.M., Maronpot, R.R. and Giri, D.K. 2023. Testis – seminiferous tubule – necrosis. National Toxicology Program – Nonneoplastic Lesion Atlas [Online]. Available via https://ntp.niehs.nih.gov/sites/default/files/nnl/male_reproductive/testis/setubnecr/testis_seminiferous_tubule_-necrosis_508.pdf(Accessed 30 June 2023) Cui, X., Jing, X., Wu, X., Wang, Z. and Li, Q. 2016. Potential effect of smoking on semen quality through DNA damage and downregulation of Chk1 in sperm. Mol. Med. Rep. 14, 753–761. Food and Drug Administration. 2016. How a cigarette is engineered. U.S. Food and Drug Administration. Gibson-Corley, K.N., Olivier, A.K. and Meyerholz, D.K. 2013. Principles for valid histopathologic scoring in research. Vet. Pathol. 50, 1007–1015. Global Adult Tobacco Survey. 2021a. Comparison fact sheet Indonesia 2011 & 2021. World Health Organization. Global Adult Tobacco Survey. 2021b. Fact sheet Indonesia 2021. World Health Organization. He, L., Gong, H., Zhang, J., Zhong, C., Huang, Y., Zhang, C. and Ashraf, M.A. 2016. Interaction of exposure concentration and duration in determining apoptosis of testis in rats after cigarette smoke inhalation. Saudi J. Biol. Sci. 23, 579–586. Indonesia Food and Drug Administration. 2017. Kajian rokok elektronik di Indonesia, 2nd ed. BPOM. Iwasawa, T., Takemura, T. and Ogura, T. 2018. Smoking-related lung abnormalities on computed tomography images: comparison with pathological findings. Jpn. J. Radiol. 36, 165–180. Jatwani, S. and Goyal, A. 2023. Vasculitis. Treasure Island, FL: StatPearls Publishing [Online]. Available via: https://www.ncbi.nlm.nih.gov/books/NB K545186/(Accessed 29 June 2023) Jawad, M., Eissenberg, T., Salman, R., Soule, E., Alzoubi, K.H., Khabour, O.F., Karaoghlanian, N., Baalbaki, R., El Hage, R., Saliba, N.A. and Shihadeh, A. 2019. Toxicant inhalation among singleton waterpipe tobacco users in natural settings. Tobacco Control 28, 181–188. Kaous, M., Xian, J., Rongo, D., McDonald, M., Ocasionez, D., Mathew, R., Estrada- Y-Martin, R., Patel, B., Cherian, S.V. and Jani, P.P. 2020. Clinical, radiologic, pathologic patterns and outcomes of vaping-related pulmonary injury: a case series. Respir. Med. 173, 106153. Khairani, S., Fauziah, N., Wiraswati, H.L., Panigoro, R., Setyowati, E.Y. and Berbudi, A. 2022. Oral administration of piperine as curative and prophylaxis reduces parasitaemia in Plasmodium berghei ANKA-infected mice. J. Trop. Med. 2022, 1–11. Kolawole, T.A., Oyeyemi, W.A., Adigwe, C., Leko, B., Udeh, C. and Dapper, D.V. 2015. Honey attenuates detrimental effects of nicotine on testicular functions in Wistar rats. Niger. J. Physiol. Sci. 30(11), 11–16. Koziol, J. and Palmer, C. 2023. Pathophysiology, diagnosis, and management of testicular diseases in the bull. Clin. Theriogenol. 15, 9271. Lechner, A.J., Matuschak, G.M. and Brink, D.S. 2012. Pathology of obstructive pulmonary diseases. In Respiratory: an integrated approach to disease. Eds., Schwartz, M.I. and King, T.E. Jr. New York, NY: McGraw-Hill. Monica, M., Adi, A.A.A.M. and Winaya, I.B.O. 2019. Histopatologi bronkiolus dan pembuluh darah paru mencit jantan pasca terpapar asap rokok elektrik. Bul. Vet. Udayana. 11, 157–165. National Toxicology Program. 2023a. Lung – vacuolization, cytoplasmic. Nonneoplastic Lesion Atlas. National Toxicology Program. 2023b. Testis – germinal epithelium – degeneration. Nonneoplastic Lesion Atlas. Nevzorova, V., Brodskaya, T. and Zakharchuk, N. 2018. Smoking, respiratory diseases and endothelial dysfunction. NMPV and Valoti, F. 2017. Man. Lung of smoker. Vertical section. 250×. Nature Microscope Photo Video [Online]. Available via https://www.nature-microscope-photo-video.com/en/photos/animal-histology/comparative-histology-of-vertebrates/other-systems/respiratory-system/mammals/man/010505c0201050101z01-man-lung-of-smoker-vertical-section-250x.html(Accessed 21 June 2023) Phillips, B.W., Schlage, W.K., Titz, B., Kogel, U., Sciuscio, D., Martin, F., Leroy, P., Vuilaume, G., Krishnan, S., Lee, T., Veljkovic, E., Elamin, A., Merg, C., Ivanov, N.V., Peitsch, M.C., Hoeng, J. and Vanscheeuwijck, P. 2018. A 90-day OECD TG 413 rat inhalation study with systems toxicology endpoints. Food Chem. Toxicol. 115, 388–413. Reyna-Sepúlveda, F., Caballero-Mendoza, E., Guzmán-de-la-Garza, F., Fernández-Garza, N., Muñoz-Maldonado, G.E. and Ancer-Rodríguez, J. 2016. Emphysema model in rats exposed to tobacco smoke. Med. Univ. 18, 79–84. Schafer, K.A., Eighmy, J., Fikes, J.D., Halpern, W.G., Hukkanen, R.R., Long, G.G., Meseck, E.K., Patrick, D.J., Thibodeau, M.S., Wood, C.E. and Francke, S. 2018. Use of severity grades to characterize histopathologic changes. Toxicol. Pathol. 46, 256–265. Slotkin, T.A., Stadler, A., Skavicus, S., Card, J., Ruff, J., Levin, E.D. and Seidler, F.J. 2016. Developmental neurotoxicity of low-level tobacco smoke exposure. Toxicol. Sci. 155, 75–84. Smith, M.L., Gotway, M.B., Crotty Alexander, L.E. and Hariri, L.P. 2021. Vaping-related lung injury. Virchows. Arch. 478, 81–88. Sousa, C., Rodrigues, M., Carvalho, A., Viamonte, B., Cunha, R., Guimarães, S., de Moura, C.S., Morais, A. and Pereira, J.M. 2019. Diffuse smoking-related lung diseases: radiologic–pathologic correlation. Insights Imaging 10, 73. Swanson, A. 2013. Secondhand smoke harms pets and wildlife. Humane Society Veterinary Medical Association. Szumilas, K., Szumilas, P., Grzywacz, A. and Wilk, A. 2020. Effects of e-cigarette vapor components on reproductive systems. Int. J. Environ. Res. Public. Health. 17, 6152. Wawryk-Gawda, E., Chylińska-Wrzos, P., Zarobkiewicz, M.K., Chłapek, K. and Jodłowska-Jędrych, B. 2020. Lung histomorphological alterations in rats exposed to cigarette smoke and e-cigarette vapour. Exp. Ther. Med. 19, 2826–2833. Wawryk-Gawda, E., Zarobkiewicz, M.K., Chłapek, K., Chylińska-Wrzos, P. and Jodłowska-Jędrych, B. 2019. Histological changes in the reproductive system of male rats exposed to cigarette smoke or e-cigarette vapor. Toxicol. Environ. Chem. 101, 1–16. Wick, M.R. 2018. Pathologic features of smoking-related lung diseases. Semin. Diagn. Pathol. 35, 271–281. World Health Organization. 2020. Pernyataan: Hari Tanpa Tembakau Sedunia 2020. WHO Indonesia. Wuyts, W.A., Agostini, C., Antoniou, K.M., Bouros, D., Chambers, R.C., Cottin, V., Egan, J.J., Lambrecht, B.N., Lories, R., Prasse, A., Robalo-Cordeiro, C., Verbeken, E., Verschakelen, J.A., Wells, A.U. and Verleden, G.M. 2012. Pathogenesis of pulmonary fibrosis. Eur. Respir. J. 41, 1207–1218. Xie, B., Li, J. and Zhu, W. 2014. Pathological changes of testicular tissue in normal adult mice. Exp. Ther. Med. 7, 654–656. Zhu, F., Qiu, X., Wang, J., Jin, Y., Sun, Y., Lv, T. and Xia, Z. 2012. A rat model of smoke inhalation injury. Inhalation. Toxicol. 24, 356–364. Ziad, S., Wajdy, A.A. and Darwish, B. 2013. Effect of cigarette smoking on trachea and lung histology of albino rat. Res. Opin. Anim. Vet. Sci. 3, 256–265.
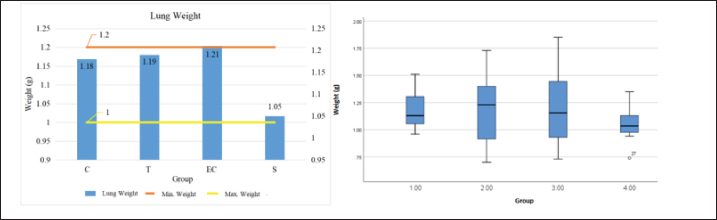
Graph 1. Histogram and Boxplot of rat lung weight data visualization. Bar and boxplot charts showing the mean lung weight (g) across four experimental groups: Control (C), Tobacco cigarette smoke exposure (T), EC vapor exposure, and Shifting exposure group (S). The mean lung weights were 1.18 g (C), 1.19 g (T), 1.21 g (EC), and 1.05 g (S). The reference range for normal lung weight was 1.0 -1.2 g. Statistical analysis revealed no significant difference among groups (p=0.687), indicating that cigarette smoke exposure did not significantly alter total lung mass after 30 days of exposure (Piao et al., 2013).
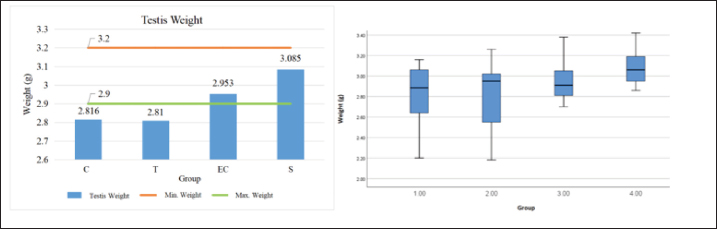
Graph 2. Histogram and Boxplot of testicle lung weight data visualization Bar and boxplot charts showing the mean testis weight (g) across four experimental groups: Control (C), Tobacco cigarette smoke exposure (T), EC vapor exposure, and Shifting exposure group (S). The mean testis weights were 2.816 g (C), 2.81 g (T), 2.953 g (EC), and 3.085 g (S). The reference range for normal testis weight was 2.9–3.2 g. Statistical analysis showed no significant difference among groups (p=0.213), suggesting that short-term exposure to either tobacco or electronic cigarette smoke did not significantly affect testicular weight.
Graph 3. Histogram and Boxplot of rat MP % area affected data visualization. | ||
| How to Cite this Article |
| Pubmed Style Sarniem BC, Hartady T, Syamsunarno MRA, Khairani S, Hiroyuki A, Atik N, Septiyani S, Satrio FA, Maharani B. Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Vet. J.. 2026; 16(2): 987-998. doi:10.5455/OVJ.2026.v16.i2.20 Web Style Sarniem BC, Hartady T, Syamsunarno MRA, Khairani S, Hiroyuki A, Atik N, Septiyani S, Satrio FA, Maharani B. Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. https://www.openveterinaryjournal.com/?mno=268411 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.20 AMA (American Medical Association) Style Sarniem BC, Hartady T, Syamsunarno MRA, Khairani S, Hiroyuki A, Atik N, Septiyani S, Satrio FA, Maharani B. Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Vet. J.. 2026; 16(2): 987-998. doi:10.5455/OVJ.2026.v16.i2.20 Vancouver/ICMJE Style Sarniem BC, Hartady T, Syamsunarno MRA, Khairani S, Hiroyuki A, Atik N, Septiyani S, Satrio FA, Maharani B. Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 987-998. doi:10.5455/OVJ.2026.v16.i2.20 Harvard Style Sarniem, B. C., Hartady, . T., Syamsunarno, . M. R. A., Khairani, . S., Hiroyuki, . A., Atik, . N., Septiyani, . S., Satrio, . F. A. & Maharani, . B. (2026) Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Vet. J., 16 (2), 987-998. doi:10.5455/OVJ.2026.v16.i2.20 Turabian Style Sarniem, Brian Christian, Tyagita Hartady, Mas Rizky A.a Syamsunarno, Shafia Khairani, Andi Hiroyuki, Nur Atik, Septiyani Septiyani, Faisal Amri Satrio, and Briliant Maharani. 2026. Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Veterinary Journal, 16 (2), 987-998. doi:10.5455/OVJ.2026.v16.i2.20 Chicago Style Sarniem, Brian Christian, Tyagita Hartady, Mas Rizky A.a Syamsunarno, Shafia Khairani, Andi Hiroyuki, Nur Atik, Septiyani Septiyani, Faisal Amri Satrio, and Briliant Maharani. "Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke." Open Veterinary Journal 16 (2026), 987-998. doi:10.5455/OVJ.2026.v16.i2.20 MLA (The Modern Language Association) Style Sarniem, Brian Christian, Tyagita Hartady, Mas Rizky A.a Syamsunarno, Shafia Khairani, Andi Hiroyuki, Nur Atik, Septiyani Septiyani, Faisal Amri Satrio, and Briliant Maharani. "Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke." Open Veterinary Journal 16.2 (2026), 987-998. Print. doi:10.5455/OVJ.2026.v16.i2.20 APA (American Psychological Association) Style Sarniem, B. C., Hartady, . T., Syamsunarno, . M. R. A., Khairani, . S., Hiroyuki, . A., Atik, . N., Septiyani, . S., Satrio, . F. A. & Maharani, . B. (2026) Pathological findings of respiratory and reproductive organs in male rats exposed to tobacco and electronic cigarette smoke. Open Veterinary Journal, 16 (2), 987-998. doi:10.5455/OVJ.2026.v16.i2.20 |