| Research Article | ||
Open Vet. J.. 2026; 16(1): 686-698 Open Veterinary Journal, (2026), Vol. 16(1): 686-698 Research Article Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024Zanuba Ainena1,2, Okti Nadia Poetri3, Ekowati Handharyani2*1Veterinary Biomedical Sciences Study Programme, School of Veterinary Medicine and Biomedical Sciences, IPB University, Dramaga, Indonesia 2Division of Pathology, School of Veterinary Medicine and Biomedical Sciences, 3Division of Medical Microbiology, School of Veterinary Medicine and Biomedical Sciences, *Corresponding Author: Ekowati Handharyani. Division Pathology, School of Veterinary Medicine and Biomedical Sciences, IPB University, Dramaga, Indonesia. Email: ekowatieko [at] apps.ipb.ac.id Submitted: 01/07/2025 Revised: 07/12/2025 Accepted: 18/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
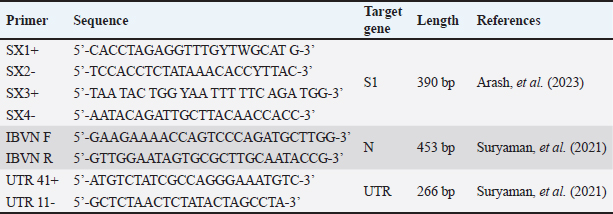
AbstractBackground: Infectious bronchitis virus (IBV) is an endemic poultry disease in Bogor Regency, West Java Province, Indonesia. This highly contagious disease spreads through direct contact with infected chickens, contaminated feces, virus-laden air, or asymptomatic carriers, such as birds. Among these carriers, pigeons (Columba livia) and spotted doves (Spilopelia chinensis) are commonly kept as pet birds and frequently interact with domesticated chickens, thereby increasing the risk of transmission. The high mutation rate of IBV, which generates numerous variants, further amplifies the risk. IBV can mutate within these asymptomatic carriers when pigeons and spotted doves act as reservoirs. Mutations may facilitate viral evolution and recombination, potentially leading to the emergence of new variants with varying pathogenicity. Aim:This study aimed to identify the potential of pigeons and spotted doves as reservoirs of IBV and perform molecular characterization of isolated IBV strains. Methods: A total of 7 pigeons and 8 spotted doves, acquired from Bogor Regency bird shops, were divided into four groups by species and observed for 12 weeks at the university animal facility. During this period, cloacal and oropharyngeal swabs were collected every 2 weeks and pooled to streamline analysis. Then, 24 pooled samples were inoculated into three 9-day-old embryonated chicken eggs to facilitate virus isolation. Following the inoculation process, IBV detection was conducted using agar gel precipitation assay, reverse transcription polymerase chain reaction (RT-PCR), and morphological observation of the embryos. In addition, agar gel precipitation (AGP) and RT-PCR were directly performed on the swabs without prior virus isolation to ensure comprehensive detection. RT-PCR targeted the nucleocapsid (N) and untranslated region genes, while the following nested RT-PCR targeted the spike gene (S1). To gain deeper insights into the viral characteristics, PCR products that tested positive for IBV were further analyzed through sequencing. Results: No birds showed clinical signs of IBV infection during the observation period. The AGP results showed IBV in direct swab samples collected between weeks 2 and 8. Virus isolation using embryonated chicken eggs extended the detection period, identifying IBV in samples collected from weeks 0 to 12. Similar results were obtained using RT-PCR, but only the S1 gene was successfully detected. Isolated IBV strains caused dwarfing and hemorrhage in embryos. The detected IBV isolates shared a high similarity with IBV 1/96, a 793B-like variant commonly used in disease vaccines, according to sequencing analysis. Conclusion: Pigeons and spotted doves may act as reservoirs for IBV, as the disease is detected in their swabs and through isolation in embryonated eggs. IBV from these isolates not only causes embryonic damage but is also closely related to the IBV 1/96 (793B-like) vaccine strain. The results show the importance of monitoring pigeons and spotted doves as part of comprehensive IBV control strategies. Keywords: Infectious bronchitis virus (IBV), Pigeons, Spotted doves, Reservoir, Molecular characterization. IntroductionInfectious bronchitis (IB) is caused by avian coronaviruses (ACoVs). According to the latest classification provided by the International Committee on Taxonomy of Viruses, ACoVs belong to the family Coronaviridae, subfamily Orthocoronavirinae, genus Gammacoronavirus, and species Avian coronavirus. IB frequently infects chickens and has significant economic implications (Bande et al., 2016). It can lead to morbidity and mortality rates of up to 80% and 20%, respectively (Hussain et al., 2005; Schalk and Hawn, 1931). In 2024, Medion, an Indonesian vaccine manufacturer, reported that there is a consistent increase in IBV cases in Indonesia, with 302 cases recorded during the year. This represents a 19.8% increase from the previous year, which recorded approximately 252 cases. The incidence of IBV primarily affects layer chickens, resulting in both mortality and reduced egg productivity. In layers, the disease not only reduces egg quality but also reduces production by 10%–50% (Tabbu, 2000; Dhama et al., 2014). IBV is a global concern, and Indonesia is no exception, as it is classified as an endemic disease country. Similar to other coronaviruses, IBV is highly prone to mutation, resulting in considerable genetic diversity and the emergence of numerous strains. This diversity presents a major challenge to vaccination aimed at controlling the disease (Woo et al., 2009; Thor et al., 2011; Setiawaty et al., 2019; Eid et al., 2024). In response to the high variability of IBV strains, Indonesian vaccination programs commonly include multiple vaccines containing different strains (Ababneh et al., 2020) to provide broader protection against circulating variants. IB clinical signs include lethargy, ruffled feathers, sneezing, dyspnea, hoarse vocalization, nasal discharge, and weight loss (Wickramasinghe et al., 2014). In cases of viremia, the virus may disseminate to the kidneys and reproductive system, resulting in reduced egg production and quality. IB transmission occurs both horizontally and vertically. The virus can be spread from infected chickens through respiratory droplets, tracheal secretions, and feces. Indirect transmission may occur via contaminated feeding and drinking equipment, housing facilities, egg packaging materials, and poultry manure (Cavanagh and Gelb, 2008). IBV is predominantly detected in the trachea during the first 3–5 days post-infection in the acute phase, after which it is excreted through feces and urine. During the chronic phase, the virus is more readily detected in the intestinal tract and cloaca (Alexander and Gough, 1977). Infected birds can transmit the virus to other flocks even in the absence of clinical signs (Ayala et al., 2020). Moreover, following apparent recovery from infection, re-excretion of the virus may occur when birds are exposed to stress or other environmental factors that trigger latent virus reactivation within the host (Naqi et al., 2003; Han et al., 2024). The presence of coronaviruses in wild birds has been reported across various continents, including Europe, Asia, Africa, North America, South America, and Australia (Wille et al., 2017; Gupta et al., 2022). In 2005, IBV strains homologous to the IBV H120 vaccine and wild-type IBV were detected in healthy peacocks (Pavo cristatus) and wild teals (Anas) (Liu et al., 2005). In Indonesia, IBV has also been identified in healthy parrots (Eclectus roratus) without any clinical symptoms (Suryaman et al., 2021). These results confirm that the disease can replicate in certain bird species without causing clinical signs. Moreover, IBV isolated from birds and inoculated into specific-pathogen-free embryonated chicken eggs (ECEs) caused nephritis and even embryo mortality (Sun et al., 2007). The results show that wild birds may serve as asymptomatic carriers of IBV, potentially transmitting the disease to susceptible chicken populations. Indonesia has a large poultry industry, with Bogor Regency in West Java Province serving as a key production region (Efendi et al., 2022). In this area, residents commonly keep birds as pets. Pigeons (Columba livia) and spotted doves (Spilopelia chinensis) are among the most popular bird species kept as pets. Considering that wild birds are known to act as reservoirs or asymptomatic carriers of IBV (Ujvári et al., 2003), there is growing interest in exploring the potential of pigeons and spotted doves. This exploration is important because the two birds frequently come into contact with chickens in the backyard. Therefore, this study aims to investigate the possibility of pigeons and spotted doves in Bogor Regency as reservoirs for IBV and to conduct molecular characterization of IBV strains isolated from the two bird species. Materials and MethodsBirdsThis study involved 15 birds, consisting of 7 pigeons and 8 spotted doves, which were purchased from four bird shops located in Bogor Regency. The birds were grouped based on their source and species as follows: three pigeons from Mang Ebel’s shop in Ciomas (Group A), four pigeons from Yoshe’s shop in Dramaga (Group B), four spotted doves from Judin’s shop in Cibanteng (Group C), and four spotted doves from Rizky’s shop in Ciampea (Group D). Therefore, each group contained three to four birds of the same species originating from the same vendor. All birds were clinically healthy and unvaccinated at the time of purchase. Each group was housed separately in a cage measuring 75 cm × 45 cm × 90 cm. Pigeons were fed a diet consisting of corn and brown rice, while spotted doves were fed brown rice supplemented with red and white millet. Feed and water were provided twice daily. The birds were maintained at the university’s animal research facility and monitored for clinical signs associated with IBV for 12 weeks. No medical treatment was administered during the study period. SamplingCloacal and oropharyngeal swab samples were collected from each bird every 2 weeks. The samples from each collection were then pooled according to their respective groups. The swab samples were placed into tubes containing Viral Transport Media (VTM) liquid consisting of phosphate buffer saline solution added with antibiotics (10,000 IU/ml penicillin and 10 mg/ml streptomycin) based on the World Organisation for Animal Health guideline for IBV. After pooling, the swab samples were stored at −20°C until further use. Virus isolationThis method was employed to propagate the virus in its natural host, i.e., the chicken. In cases where the viral titer in swab samples was low, this step was considered appropriate to increase the viral yield. Fertilized chicken eggs (clean eggs) were obtained from Perseroan Terbatas (PT) Inti Prima Satwa Sejahtera (IPSS), Sukabumi, Indonesia, and were free from major avian pathogens according to the supplier’s certificate. Egg inoculation was conducted immediately after swab samples were collected using the allantoic sac inoculation method. Pooling samples were first inoculated into three clean eggs (passage 1). Pooled samples were prepared as an inoculum by adding 10,000 IU/ml of penicillin–-streptomycin. After a 30-minute incubation at room temperature, 0.1 ml of this inoculum was injected into each of the three clean eggs. Eggs were incubated for 7 days and then refrigerated at 4°C–8°C for 24 hours. The allantoic fluid from passage 1 eggs was harvested and used as the inoculum for the next group of clean eggs (passage 2). The same procedure was repeated, in which the allantoic fluid from passage 2 was subsequently inoculated into clean eggs (passage 3). The allantoic fluid was clarified by low-speed centrifugation after harvesting to remove cellular debris. The clarified supernatant was aliquoted and stored at 20°C until further use. The allantoic fluid was tested for the presence of IBV using agar gel precipitation (AGP) and reverse transcription polymerase chain reaction (RT-PCR). Dwarf morphology was visually determined by comparing the size of inoculated embryos with that of negative control embryos (without inoculation) on the same day of incubation. Embryos were categorized as dwarfs when they appeared to be smaller than the controls, with characteristic features such as a relatively larger head and a shorter body (Tsai et al., 2016). Detection of IBV from inoculated samples using the AGP testVirus isolation was performed to increase the virus yield, especially when the viral titer in swab samples was low. Embryonated chicken eggs were obtained from IPSS, Sukabumi, Indonesia—confirmed free from major avian pathogens according to the supplier’s certificate—and were used for virus propagation. Egg inoculation was performed immediately after swab samples were collected using the allantoic sac inoculation method. Each pooled swab sample was inoculated into three 9-day-old clean eggs (passage 1). The procedure involved mixing the pooled samples with penicillin–streptomycin to reach a final concentration of 10,000 IU/ml. After incubation at room temperature for 30 minutes, each egg was injected with 0.1 ml of the inoculum. The eggs were incubated for 7 days and then chilled at 4°C–8°C for 24 hours. The allantoic fluid from eggs from passage 1 was collected and used as the inoculum for the next batch of eggs (passage 2). The same procedure was repeated for passage 3. The allantoic fluid was clarified by low-speed centrifugation after harvesting to remove cellular debris. The clear supernatant was aliquoted and stored at 20°C until further use. The harvested allantoic fluid was then tested for the presence of IBV using AGP and RT-PCR. Dwarfing in embryos was visually determined by comparing the size of inoculated embryos with that of negative control embryos (uninoculated) on the same day of incubation. Embryos were classified as dwarfs when they appeared to be smaller than the controls, typically showing a proportionally larger head and a shorter body. IBV detection using RT-PCRRT-PCR was conducted on allantoic fluid (from virus isolation) and pooled swab samples. Viral Ribonucleic Acid was extracted using the Viral Nucleic Acid Extraction Kit II (Geneaid Biotech Ltd., Taipei, Taiwan) according to the manufacturer’s instructions. CEVAC IBIRD® vaccine was used as a positive control. Primers for the S1 gene (SX1+/SX2) and (SX3+/SX4) were applied using nested PCR. The One-Step RT PCR kit (Bioline, UK) was used for the first amplification reaction, followed by the second amplification (nested-PCR) with MyTaq™ HS Red Mix (Bioline, UK), both following the manufacturer’s guidelines. Nested PCR offers a significant advantage over conventional PCR in terms of sensitivity and specificity. The two-step amplification process ensures that even low-copy Deoxyribonucleic Acid targets are amplified accurately, reducing background noise and nonspecific products. In addition, primers for the N gene (IBVN+/IBVN) and the untranslated region (UTR) gene (UTR41+/11) were applied using RT-PCR. The N and UTR genes were tested using conventional PCR to confirm the presence or absence of IBV in the samples (Promkuntod et al., 2014). This RT-PCR amplification was executed with the Bioline One-Step RT-PCR kit (Bioline, UK) according to the manufacturer’s instructions. Finally, the procedure visualized the amplicons using electrophoresis on a 1.5% agarose gel run at 100 V for 35 min. The specific acid–base sequence of the primers used is presented in Table 1. Table 1. Primers for detection of S1, N, and UTR genes in IBV.
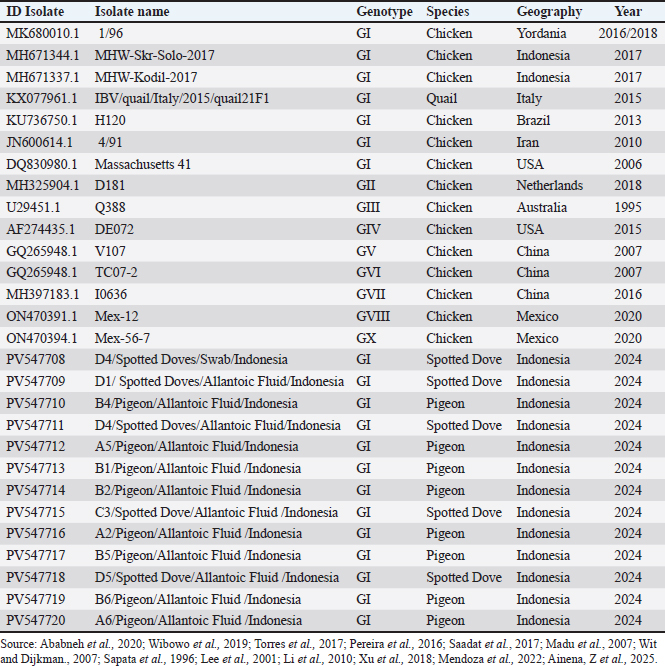
Sequencing and phylogenetic analysis of the genesThe isolates in this study were compared for their relatedness to vaccine strains, IBV isolates previously reported in other studies in GenBank, and IBV isolates obtained in Indonesia, as summarized in Table 6. Fifteen positive Polymerase Chain Reaction (PCR) products were subjected to Sanger sequencing at First BASE Laboratories Sdn Bhd (Malaysia), specifically targeting the S1 gene. The resulting sequences were then edited using the BLAST tool. Subsequently, a phylogenetic tree was reconstructed using the neighbor-joining method and 1,000 bootstrap replicates. All data analyses were performed using MEGA 7 software. Table 6. Summary of bird species, geographic origin, and detection year of IBV isolates.
Ethical approvalThis study adhered to Article 80 of Indonesian Law No. 18/2009 on Livestock and Animal Health. The Veterinary Ethics Commission of the School of Veterinary Medicine and Biomedical Sciences, IPB University, granted ethical approval under number 229 KEH/SKE/VII/2024. Date: 15 July 2024. ResultsClinical observation in pigeons and spotted dovesFor 3 months following inoculation, the treatment group showed no typical signs of IBV infection. The birds remained active, maintained a normal appetite, and continued to drink regularly. Furthermore, respiratory symptoms, such as coughing, sneezing, abnormal breathing sounds (rales), or gasping, were absent. There were also no indications of ruffled feathers or depression. Clinical observations confirmed that the virus isolates did not induce any obvious clinical manifestations in the test birds throughout this period. Detailed clinical observations are available in the Supplementary Material. The observed clinical signs are summarized in Table 2. Table 2. Clinical symptomps of observation table.
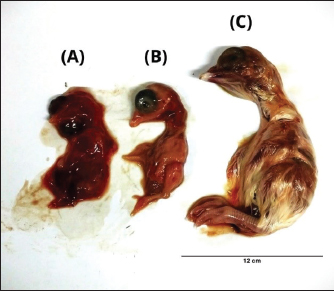
Morphological changes in chicken embryosEmbryos that were not inoculated (negative controls) showed normal morphology, whereas those inoculated with the positive control exhibited stunted growth and hemorrhaging. Some PCR-positive samples also caused stunted growth in the embryos; however, these effects were not consistently observed in subsequent inoculation passages. In contrast, only samples A5 (from pigeon) and D2 (from spotted dove) produced reproducible embryonic changes that could be passaged and were therefore selected for further analysis. As shown in Figure 1, embryos inoculated with the spotted dove isolate (D2/Spotted Dove/Allantoic Fluid/Indonesia) displayed clear signs of hemorrhage and dwarfing.
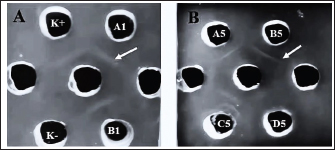
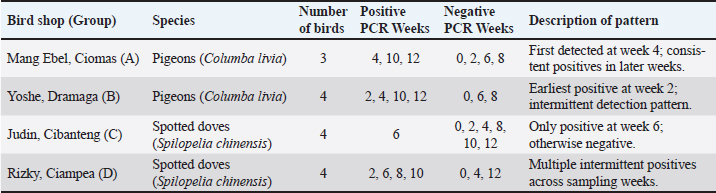
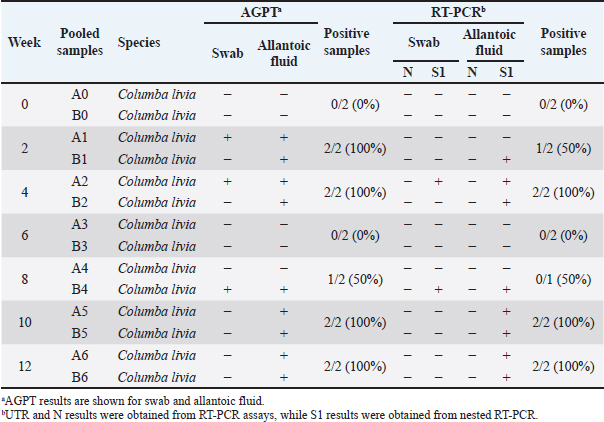
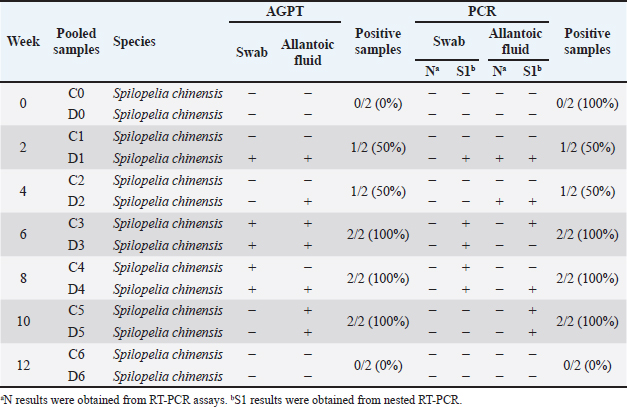
Fig. 1. The morphology of chicken embryos after inoculation from isolate D2/Spotted Doves/Allantoic Fluid/Indonesia. (A) Hemorrhage, (B) Dwarfing, (C) Normal embryo. AGPT and PCR resultsThe AGPT and PCR results indicated IBV in the swab and allantoic fluid samples from week 2, whereas all samples were negative at week 0. Positive AGPT samples are shown in Figure 2. PCR confirmed the presence of IBV, particularly after inoculation into ECEs, which facilitated viral replication. Positive PCR samples are shown in Figure 3. The IBV in swab and allantoic fluid samples from pigeons and spotted doves isolates are shown in Tables 4 and 5. The PCR results from each bird group obtained from different bird shops showed variable patterns of IBV detection throughout the study period. The detailed results for each shop are presented in Table 3.
Fig. 2. AGPT results from allantoic fluid samples. The arrow indicates a precipitation line showing a positive result for the IB virus. (A) A1, B1: Pigeon isolates (B) A5, B5, C5, D5: Spotted doves isolates.
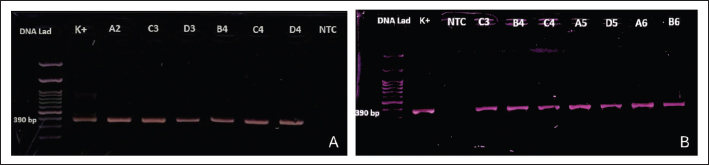
Fig. 3. (A). PCR results of the S1 gene targets from swab samples (A and B: pigeons). C and D: spotted doves). K+=positive control; NTC=no-template control. (B). PCR results of S1 gene targets from allantoic fluid samples (A and B: pigeons). C and D: spotted doves. K+=positive control; NTC=no-template control. Table 3. PCR detection of IBV in pigeons and spotted doves from different bird shops.
Table 4. Results for AGPT and PCR for Pigeon (Columba livia) samples.
Table 5. Results for AGPT and PCR for samples from spotted doves (Spilopelia chinensis).
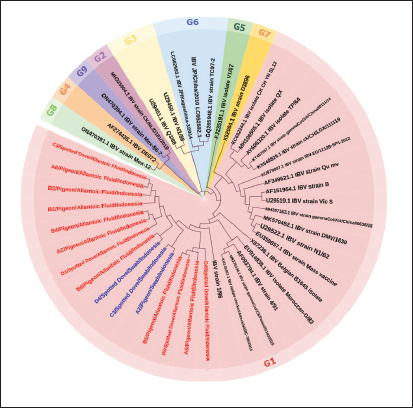
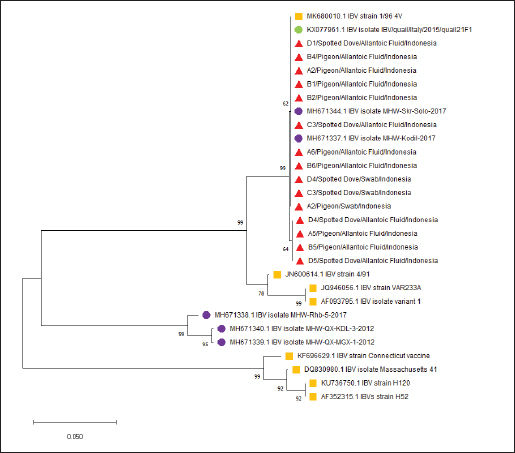
PCR targeting the UTR gene was negative for all samples, while PCR targeting the N gene was also negative except for two samples (D1 and D2), which tested positive. Table 5 shows the PCR result documentation. Phylogenetic analysis of partial S1 sequencesTwelve allantoic samples and 3 swab samples that showed positive PCR results for the S1 target gene (390 bp) were sequenced. Sequencing analysis showed that the isolates from this research were similar to vaccine isolate 1/96 (Fig. 6), a 793B-like variant vaccine (genotype 1) (Fig. 5).
Fig. 5. IBV phylogenetic tree based on partial S gene sequences including isolates from genotypes 1–9. Research isolates are highlighted in red (allantois fluid samples) and blue (swab samples).
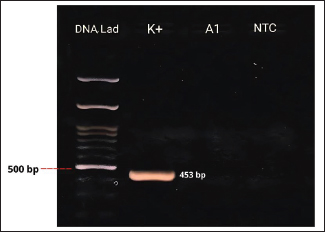
Fig. 6. IBV phylogenetic tree based on partial S gene sequences including research isolates (▲), Vaccine Isolates (■), IBV Isolates in Indonesia (●), and IBV Isolates from GenBank (●). DiscussionIBV, a member of the genus Gammacoronavirus within the family Coronaviridae, primarily infects chickens but has also been reported in other avian species (Schalk and Hawn, 1931; Cavanagh and Gelb, 2008). The increasing number of reports of IBV detection in non-galliform birds suggests that certain avian species may act as alternative hosts or reservoirs, facilitating viral maintenance and evolution in the environment (Liu et al., 2005; Suryaman et al., 2021 ; Tegegne et al., 2020). This study demonstrates that pigeons and spotted doves may harbor IBV without showing overt clinical signs, indicating their potential role as asymptomatic carriers. All samples collected at week 0 tested negative by both PCR and AGPT, indicating that no detectable viral load was present at the beginning of the study. This result suggests that the virus was either absent or present at levels below the detection limit at the time of sampling. Nevertheless, the possibility of low-level viral presence cannot be completely ruled out. The source of IBV detected in the subsequent weeks remains uncertain. Possible origins include prior exposure in the natural habitats of the birds, transmission during their stay at the bird shops, or exposure after transfer to the university facility. Since no poultry vaccinated with the IBV 1/96 strain were kept at the facility during the study period, the most likely explanation is infection from the bird shops or the birds’ original environments. Several factors, including environmental temperature and humidity, influence IBV transmission (Blanco-González et al., 2024). Gelb et al. (1991) indicated that the disease could survive at 28°C but became inactive at temperatures exceeding 41°C. Furthermore, IBV can spread through contaminated feeding or drinking utensils and cages (Bhuiyan et al., 2021). The incubation period in the host typically ranges from 24 to 48 hours. The disease is transmitted through the respiratory secretions, mucus, and feces of infected birds. Infected birds can intermittently shed IBV for up to 20 weeks post-infection without clinical symptoms (Cavanagh and Gelb, 2008). AGPT and PCR tests conducted on swab and allantoic fluid samples showed discrepancies in some cases. Virus inoculation of SPF ECEs is a standard practice for promoting viral replication. Although swab samples may initially have low viral titers, viral replication in ECEs can yield higher positive results in AGPT and PCR tests. Virus isolation using ECEs is a widely used method for propagating poultry viruses (Williams, 2016). AGPT serves as a preliminary test to detect viral antigens in samples before conducting molecular analyses. In this study, AGPT-positive samples were further confirmed using PCR to detect the genetic material of IBV. Figure 4 illustrates the PCR assay conducted to evaluate primer sensitivity. Amplification observed in the positive control confirms the specificity and sensitivity of the primers, while the absence of bands in the NTC indicates no contamination. However, discrepancies arose with two samples (A1 and D3), which were AGPT positive and PCR negative. This inconsistency likely resulted from viral degradation. As an RNA virus, IBV is more susceptible to damage than DNA viruses (Goebel et al., 2004). RNA molecules are particularly prone to hydrolysis, a chemical reaction that breaks down RNA in the presence of water under unstable conditions. Additionally, ribonuclease (RNase) enzymes, including endoribonucleases and exoribonucleases present in host cells, actively degrade RNA (Mathy et al., 2007; Jung et al., 2010; Sola et al., 2011).
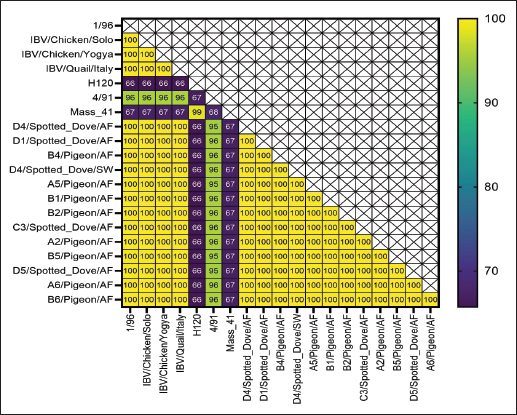
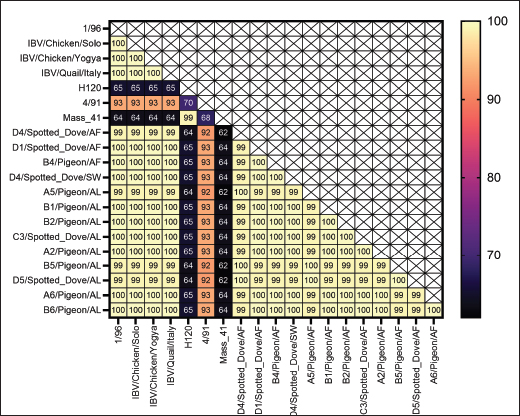
Fig. 4. PCR Results of N gene targets from swab samples. K+=positive control; A1=swab samples from pigeons; NTC=no-template control. Pigeons and spotted doves often act as asymptomatic carriers of IBV, harboring the virus without showing clinical signs. Periodic swab sampling was performed every 2 weeks to monitor the dynamics of IBV infection in both species. Because viral and antibody titers were not quantified, infection dynamics were interpreted solely based on the presence or absence of positive AGPT and PCR results. The intermittent IBV detection in swab and allantoic fluid samples shows that pigeons and spotted doves may indeed serve as asymptomatic carriers. The variation observed between sampling periods likely reflects fluctuations in viral load or intermittent viral shedding, a pattern that has also been reported in other avian species (Rasheed et al., 2023). However, stressful conditions or environmental triggers can reactivate latent viruses, resulting in increased titers and shedding. Domanska-Blicharz et al. (2014) highlighted that healthy birds from Anseriformes, Charadriiformes, and Galliformes orders could also serve as asymptomatic carriers of IBV. Partial sequencing of the S1 gene revealed that the isolates were closely related to the 1/96 vaccine strain, a 793B-like variant belonging to genotype 1 (Pellattiero et al., 2018). This genotype is widely distributed in Indonesia and is frequently used in vaccination programs (Darminto, 1992; Indriani and Darminto, 2000; Indi Dharmayanti and Indriani, 2017; Ababneh et al., 2020). The genetic similarity between field isolates from nonchicken species and vaccine strains shows that vaccine-derived viruses may have spilled over from vaccinated poultry populations (Tegegne et al., 2020; Han et al., 2024). Similar findings have been reported in wild birds carrying vaccine-related IBV genotypes (Bande et al., 2017). Continuous genetic monitoring is essential to differentiate between vaccine spillover and naturally circulating variants, as recombination or mutation events may alter virulence or antigenicity, complicating control efforts (Felippe et al., 2010; Zhao et al., 2011; Santos Fernando et al., 2017). Figures 7 and 8 shows the nucleotide and amino acid sequence similarity among the IBV isolates analyzed in this study. The comparison highlights conserved and variable regions, providing insight into the genetic relatedness and potential functional differences between isolates. Overall, this study provides preliminary evidence that pigeons and spotted doves can harbor IBV without clinical illness. These species may play a role in maintaining IBV in the environment and facilitating its evolution. Further research is needed to evaluate their potential as reservoirs through quantitative viral load studies, experimental transmission trials, and broader molecular surveillance across avian taxa.
Fig. 7. Nucleotide identity values of the obtained samples compared with vaccine isolates and research isolates in Indonesia.
Fig. 8. Amino acid identity values of the obtained samples compared with those of the vaccine isolates and research isolates in Indonesia. This study has several limitations that should be acknowledged. First, the sample size and geographic coverage were limited to birds obtained from a few shops, which may not represent the broader pigeon and spotted dove populations in Indonesia. Second, viral load quantification and serological testing were not performed, preventing the assessment of infection intensity or immune response dynamics. Third, the absence of experimental transmission trials limits the ability to confirm whether pigeons and spotted doves can transmit IBV to other avian species. In addition, only partial sequencing of the S1 gene was conducted; therefore, complete genomic characterization and potential recombination events could not be determined. Finally, environmental factors, such as temperature, humidity, and stress, which may have influenced viral replication or detection patterns, were not experimentally controlled. These limitations highlight the need for further studies using larger sample sizes, full-genome sequencing, and experimental infection models to better understand the epidemiological role of pigeons and spotted doves in IBV ecology. ConclusionThis study showed that pigeons and spotted doves can harbor IBV without developing clinical symptoms, indicating their potential role as asymptomatic carriers. Although all birds remained healthy during the 12-week observation period, IBV was intermittently detected in the swab and allantoic fluid samples. Embryo inoculation confirmed the presence of a viable virus capable of replication, particularly in the A5 (pigeon) and D2 (spotted dove) isolates. Phylogenetic analysis of the S1 gene revealed that the isolates were closely related to the 1/96 vaccine strain (793B group, genotype 1). These findings show the possible environmental persistence or spillover of vaccine-derived IBV in non-galliform birds. Further studies with larger sample sizes, full-genome sequencing, and transmission experiments are needed to confirm the reservoir potential of pigeons and spotted doves and to better understand their role in the epidemiology of IBV. AcknowledgmentsThe authors sincerely appreciate the financial support provided by the Ministry of Research, Technology, and Higher Education of the Republic of Indonesia through the Pendidikan Magister menuju Doktor untuk Sarjana Unggul (PMDSU) grant. Conflict of interestThe authors declare no conflict of interest. FundingFunding: The PMDSU grant funded this work. Authors’ contributionsZA, ONP, and EH conceptualized the research, designed the methodology, and conducted the data analysis. ZA contributed to the laboratory work, data collection, and analysis, and wrote the original manuscript. All authors contributed to the writing, review, and approval of the final version of the manuscript. Data availabilityAll data supporting the findings of this research are available within the manuscript. ReferencesAbabneh, M., Ababneh, O. and Al-Zghoul, M.B. 2020. High-resolution melting curve analysis for infectious bronchitis virus strain differentiation. Vet. World. 13(3), 400–406. Alexander, D.J. and Gough, R.E. 1977. Isolation of avian infectious bronchitis virus from experimentally infected chickens. Res. Vet. Sci. 23, 344–347. Ayala, A.J., Yabsley, M.J. and Hernandez, S.M. 2020. A review of pathogen transmission at the backyard chicken–wild bird interface. Front. Vet. Sci. 7, 1–15. Bande, F., Arshad, S., Omar, A., Hair-Bejo, M., Abubakar, M.S. and Abba, Y. 2016. Pathogenesis and diagnostic approaches of avian infectious bronchitis. Adv. Virol. 2016, 1–11. Bande, F., Arshad, S.S., Omar, A.R., Hair-Bejo, M., Mahmuda, A. and Nair, V. 2017. Distributions and strain diversity of avian infectious bronchitis virus: a review. Anim. Health Res. Rev. 18, 70–83. Bhuiyan, M.S.A., Amin, Z., Bakar, A.M.S.A., Saallah, S., Yusuf, N.H.M., Shaarani, S.M. and Siddiquee, S. 2021. Factor influences for diagnosis and vaccination of avian infectious bronchitis virus (gammacoronavirus) in chickens. Vet. Sci. 8, 1–25. Blanco-González, J., López-Rull, I., Cayuela, L., Majó, N., Busquets, N., Montalvo, T. and Senar, J.C. 2024. Native and invasive bird interactions increase the spread of Newcastle disease in urban environments. Biol. Invasions 26, 845–855. Cavanagh, J. and Gelb. 2008. Infectious bronchitis. In Diseases of Poultry. Swayne, D.E. (ed.), Hoboken, NY: Wiley-Blackwell, pp: 117–35. Darminto. and D. 1992. Serotype of IB viral isolates. Penyakit Hewan 24, 76–81. Das Gupta, S., Barua, B., Fournié, G., Hoque, M.A. and Henning, J. 2022. Village and farm-level risk factors for avian influenza infection on backyard chicken farms in Bangladesh. Sci. Rep. 12, 1–9. Dhama, K., Singh, S.D., Barathidas, R., Desingu, P.A., Chakrabort, S., Tiwari, R. and Kumar, M.A. 2014. Emergence of Avian Infectious Bronchitis Virus and its variants need better diagnosis, prevention and control strategies: a global perspective. Pak. J. Biol. Sci. 17(6), 751–767. Domanska-Blicharz, K., Jacukowicz, A., Lisowska, A., Wyrostek, K. and Minta, Z. 2014. Detection and molecular characterization of infectious bronchitis-like viruses in wild bird populations. Avian Pathol. 43(5), 406–413. Efendi, R., Sudarnika, E., Wibawan, I.W.T. and Purnawarman, T. 2022. An assessment of knowledge and attitude toward antibiotic misuse by small-scale broiler farmers in Bogor, West Java, Indonesia. Vet. World. 15, 707–713. Eid, A., Mahmoud, A., Hamouda, E., Metwally, M., Eldin, R. and Elbakrey, R. 2024. The efficacy of simultaneous successive classic and variant infectious bronchitis virus vaccines versus circulating variant II Egyptian field virus. Open. Vet. J. 14, 90–107. Felippe, P.A.N., Da Silva, L.H.A., Santos, M.M.A.B., Spilki, F.R. and Arns, C.W. 2010. Genetic diversity of avian infectious bronchitis virus isolated from domestic chicken flocks and coronaviruses from feral pigeons in Brazil between 2003 and 2009. Avian. Dis. 54, 1191–1196. Gelb, J., Lunt, R.L., Metz, A.L. and Fries, P.A. 1991. Attenuation of avian infectious bronchitis virus by cold-adaptation. Avian. Dis. 35(4), 847–853. Goebel, S.J., Hsue, B., Dombrowski, T.F. and Masters, P.S. 2004. Characterization of the RNA components of a putative molecular switch in the 30 untranslated region of the murine coronavirus genome. J. Virol. 78, 669–682. Han, Z., Xu, X., Li, H. and Liu, S. 2024. Replication and adaptation of avian infectious bronchitis viruses in pheasants (Phasianus colchicus). Virus Res. 350, 1–11. Hussain, A., Khan, A.S., Khalid, M. and Hamid, T. 2005. Seroprevalence polypeptide analysis of infectious bronchitis virus in broilers. Pak. Vet. J. 25(4), 194–196. Indi Dharmayanti, N.L.P. and Indriani, R. 2017. Identification and characterization of infectious bronchitis virus (IBV) in Indonesia. J. Biol. Ind. 13, 53–59. Indriani, R. and Darminto, D. 2000. Serotype variation infectious bronchitis virus isolates from several regions in Java. J. Int. Tech. Vet. 5(1), 1–7. Jung, M., Schaefer, A., Steiner, I., Kempkensteffen, C., Stephan, C., Erbersdobler, A. and Jung, K. 2010. Robust microRNA stability in degraded RNA preparations from human tissue and cell samples. Clin. Chem. 56, 998–1006. Liu, S., Chen, J., Chen, J., Kong, X., Shao, Y., Han, Z., Feng, L., Cai, X., Gu, S. and Liu, M. 2005. Isolation of avian infectious bronchitis coronavirus from domestic peafowl (Pavo cristatus) and teal (Anas). J. Gen. Virol. 86, 719–725. Mahmood M. Siddique I, Hussain. 2004. Carrier State Studies of Infectious Bronchitis Virus in Asymptomatic Layer in Pakistan. Int J Poult Sci. 3: 547-549. Mathy, N., Bénard, L., Pellegrini, O., Daou, R., Wen, T. and Condon, C. 2007. 5-to-3 exoribonuclease activity in bacteria: role of RNase J1 in rRNA maturation and 5’ stability of mRNA. Cell 129, 681–692. Naqi, S., Gay, K., Patalla, P., Mondal, S. and Liu, R. 2003. Establishment of persistent avian infectious bronchitis virus infection in antibody-free and antibody-positive chickens. Avian Dis. 47, 594–601. Pellattiero, E., Tucciarone, C.M., Franzo, G., Berto, G., Koutoulis, K., Meini, A., Zangrandi, C., Ramon, G., Drigo, M. and Cecchinato, M. 2018. Evaluation of unintended 1/96 infectious bronchitis vaccine transmission in broilers after direct contact with vaccinated ones. Vet. Med-Czech. 63, 287–291. Promkuntod, N., Van Eijndhoven, R.E.W., De Vrieze, G., Gröne, A. and Verheije, M.H. 2014. Mapping of the receptor-binding domain and amino acids critical for attachment in the spike protein of avian coronavirus infectious bronchitis virus. Virology 448, 26–32. Santos Fernando, F., Coelho Kasmanas, T., Diniz Lopes, P., Da Silva Montassier, M.D.F., Zanella Mores, M.A., Casagrande Mariguela, V., Pavani, C., Moreira Dos Santos, R., Assayag Jr, M.S. and Montassier, H.J. 2017. Assessment of molecular and genetic evolution, antigenicity and virulence properties during the persistence of the infectious bronchitis virus in broiler breeders. J. Gen. Virol. 98(10), 2470–2481. Schalk., A., Hawn. and M. 1931. An apparently new respiratory disease of baby chicks. J. Am. Vet. Med. Assoc. 78(4), 3–6. Setiawaty, R., Soejoedono, R.D. and Poetri, O.N. 2019. Genetic characterization of S1 gene of infectious bronchitis virus isolated from commercial poultry flocks in West Java, Indonesia. Vet. World. 12, 231–235. Sola, I., Mateos-Gomez, P.A., Almazan, F., Zuñiga, S. and Enjuanes, L. 2011. RNA-RNA and RNA-protein interactions in coronavirus replication and transcription. RNA Biol. 8, 237–248. Sun, L., Zhang, G.H., Jiang, J.W., Fu, J.D., Ren, T., Cao, W.S., Xin, C.A., Liao, M. and Liu, W.J. 2007. A Massachusetts prototype like coronavirus isolated from wild peafowls is pathogenic to chickens. Virus. Res. 130, 121–128. Suryaman, G.K., Soejoedono, R.D., Poetri, O.N., Setiyono, A. and Handharyani, E. 2021. Detection of infectious bronchitis-like virus isolated from psittacines at breeding facilities. Biodiversitas. J. Biol. Diversity. 22, 4261–4265. Tabbu, C. R. 2000. Penyakit Ayam dan Penangulangannya. Penyakit Bakterial, Mikal dan Viral. Kanesius. Tegegne, D., Deneke, Y., Sori, T., Abdurahaman, M., Kebede, N., Cecchinato, M. and Franzo, G. 2020. Molecular epidemiology and genotyping of infectious bronchitis virus and avian metapneumovirus in backyard and commercial chickens in Jimma Zone, Southwestern Ethiopia. Vet. Sci. 7, 1–11. Thor, S.W., Hilt, D.A., Kissinger, J.C., Paterson, A.H. and Jackwood, M.W. 2011. Recombination in avian gamma-coronavirus infectious bronchitis virus. Viruses 3, 1777–1999. Tsai, C., Chiou, H. and Wang, C. 2016. The infectious bronchitis vaccine strain virus is more pathogenic in chicken embryos than the wild virus strain 2575/98. Veterinarski. Arhiv. 86, 699–709. Ujvári, D., Wehmann, E., Kaleta, E.F., Werner, O., Savić, V., Nagy, E., Czifra, G. and Lomniczi, B. 2003. Phylogenetic analysis reveals extensive evolution of avian paramyxovirus type 1 strains of pigeons (Columba livia) and suggests multiple species transmission. Virus. Res. 96, 63–73. Wickramasinghe, I.N.A., Van Beurden, S.J., Weerts, E.A.W.S. and Verheije, M.H. 2014. The avian coronavirus spike protein. Virus. Res. 194, 37–48. Wille, M., Lindqvist, K., Muradrasoli, S., Olsen, B. and Järhult, J.D. 2017. Urbanization and the dynamics of RNA viruses in Mallards (Anas platyrhynchos). Infect. Genet. Evol. 51, 89–97. Williams, SM. 2016. A laboratory manual, the isolation, identification and characterization of avian pathogens, 6th edn. American Association of Avian Pathologists. Jacksonville, FL. Woo, P.C., Lau, S.K., Huang, Y. and Yuen, K.Y. 2009. Coronavirus diversity, phylogeny and interspecies jumping. Exp. Biol. Med. (Maywood). 234, 1117–1127. Zhao, W., Zhu, A.L., Yuan, C.L., Yu, Y., Zhu, C.X., Lan, D.L., Yang, Z.B., Cui, L. and Hua, X.G. 2011. Detection of astrovirus infection in pigeons (Columbia livia) during an outbreak of diarrhea. Avian Pathol. 40, 361–365. | ||
| How to Cite this Article |
| Pubmed Style Ainena Z, Poetri ON, Handharyani E. Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Vet. J.. 2026; 16(1): 686-698. doi:10.5455/OVJ.2026.v16.i1.64 Web Style Ainena Z, Poetri ON, Handharyani E. Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. https://www.openveterinaryjournal.com/?mno=268017 [Access: February 04, 2026]. doi:10.5455/OVJ.2026.v16.i1.64 AMA (American Medical Association) Style Ainena Z, Poetri ON, Handharyani E. Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Vet. J.. 2026; 16(1): 686-698. doi:10.5455/OVJ.2026.v16.i1.64 Vancouver/ICMJE Style Ainena Z, Poetri ON, Handharyani E. Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Vet. J.. (2026), [cited February 04, 2026]; 16(1): 686-698. doi:10.5455/OVJ.2026.v16.i1.64 Harvard Style Ainena, Z., Poetri, . O. N. & Handharyani, . E. (2026) Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Vet. J., 16 (1), 686-698. doi:10.5455/OVJ.2026.v16.i1.64 Turabian Style Ainena, Zanuba, Okti Nadia Poetri, and Ekowati Handharyani. 2026. Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Veterinary Journal, 16 (1), 686-698. doi:10.5455/OVJ.2026.v16.i1.64 Chicago Style Ainena, Zanuba, Okti Nadia Poetri, and Ekowati Handharyani. "Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024." Open Veterinary Journal 16 (2026), 686-698. doi:10.5455/OVJ.2026.v16.i1.64 MLA (The Modern Language Association) Style Ainena, Zanuba, Okti Nadia Poetri, and Ekowati Handharyani. "Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024." Open Veterinary Journal 16.1 (2026), 686-698. Print. doi:10.5455/OVJ.2026.v16.i1.64 APA (American Psychological Association) Style Ainena, Z., Poetri, . O. N. & Handharyani, . E. (2026) Detection and molecular characterization of infectious bronchitis virus in pigeons and spotted doves from Bogor Regency, West Java Province, Indonesia in 2024. Open Veterinary Journal, 16 (1), 686-698. doi:10.5455/OVJ.2026.v16.i1.64 |