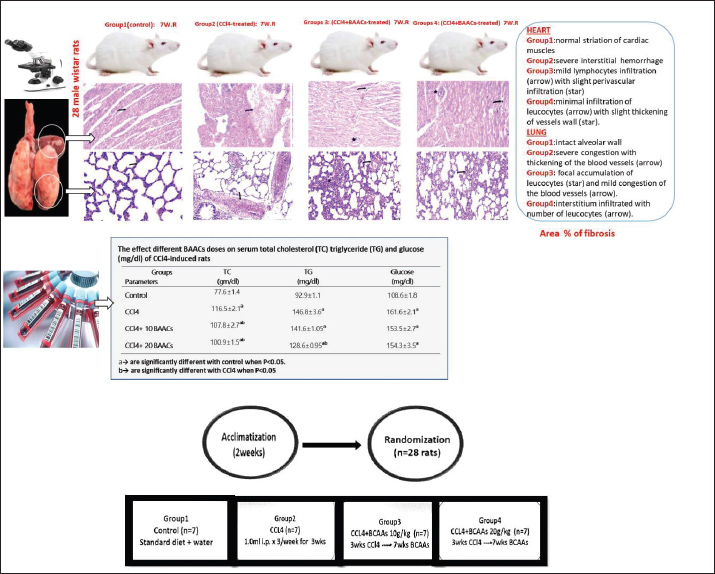
| Research Article | ||
Open Vet. J.. 2025; 15(12): 6644-6659 Open Veterinary Journal, (2025), Vol. 15(12): 6644-6659 Research Article Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar ratsMariam M. Jad1, Zeinab Al-Amgad2, Fatma A. Madkour3, Hend Al-Amgad4, Asmaa W. Basher5, Heba A. Mohammed6, Abeer M. Mahmoud7, Asmaa Elnagar8,9, Ibrahim F. Rehan8,9,10*, František Zigo11, Martina Zigová12 and Amany Ahmed Abd El-Aziz131Department of Zoology, Faculty of Science, Luxor University, Luxor, Egypt 2Medical Technical College, Al-Farahidi University, Baghdad, Iraq 3Department of Anatomy and Embryology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 4Human Internal Medicine and Rheumatism, Faculty of Medicine, Kasr Al Ainy, Cairo University Hospitals, Cairo, Egypt 5Department of Pharmacology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 6Doctor of Physiology, Post Graduated by Faculty of Science, Qena, Egypt 7Department of Human Anatomy and Embryology, Faculty of Medicine, South Valley University, Qena, Egypt 8Department of Pathobiochemistry, Faculty of Pharmacy, Meijo University Yagotoyama, Nagoya-shi, Japan 9Department of Animal Behavior and Husbandry, Faculty of Veterinary Medicine, Menoufia University, Shebin Alkom, Menoufia, Egypt 10Institute of Genetics and Animal Biotechnology, Polish Academy of Sciences, Jastrzebiec, Poland. 11Department of Nutrition and Animal Husbandry, University of Veterinary Medicine and Pharmacy, Košice, Slovakia 12Department of Pharmacology, Faculty of Medicine, Pavol Jozef Safarik University, Kosice, Slovakia 13Home Economic Department, Nutrition and Food Science, Faculty of Specific Education, South Valley University, Qena, Egypt *Corresponding Author: Ibrahim F. Rehan. Department of Pathobiochemistry, Faculty of Pharmacy, Meijo University Yagotoyama, Nagoya-shi, Japan. Email: ibrahim.rehan [at] vet.menofia.edu.eg; Submitted: 10/06/2025 Revised: 25/10/2025 Accepted: 08/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Carbon tetrachloride (CCl4) is a xenobiotic hepatotoxic agent that causes pathophysiological disorders in tissues and the liver. CCl4-induced cardiopulmonary toxicity is attributed to free radical-induced dyslipidemia and oxidative damage. Branched-chain amino acids (BCAAs) stimulate systemic defense against oxidative stress, which is probably caused by CCl4, through an improvement in the antioxidant system. Aim: This study aimed to explore the therapeutic benefits of BCAAs against CCl4-induced cardiopulmonary pathologies. Methods: A total of 28 inclusive Wistar rats stand under control was standard; group 2 comprised CCl4-received rats, whereas groups 3 and 4 comprised CCl4-rats supplemented with two varying levels of BCAAs. Results: Biochemical findings indicated that CCl4 provoked a rise in glucose and cholesterol levels compared with the control. The oxidative profile suggested increased malondialdehyde level and decreased superoxide dismutase activity in the CCl4-intoxicated heart. Supplemented BCAAs downregulated dyslipidemia and restored atrial changes in antioxidant components to be close to normal. Histopathological investigations using hematoxylin-eosin stain revealed damage to the inflamed heart-lung parenchyma of CCl4-intoxicated rats. An increase in fibrosis expression was detectable, simultaneously with a deprivation of protein content in cardiopulmonary sections, as shown by Masson trichrome and bromophenol blue techniques. Co-treated BCAAs intensified cardiopulmonary integrity, inducing scarce histological fibrosis with abundant protein. Accordingly, BCAA supplementation in CCl4-treated rats mitigated complications by reducing serum toxicity, enhancing antioxidant capacity, restoring protein expression, and improving organ health. Conclusion: The findings support the protective role of BCAAs with antioxidant benefits against CCl4-induced damage, suggesting their potential as a therapeutic agent for cardiopulmonary pathology. Keywords: CCl4, BCAAs, Dyslipidemia, Antioxidant profile, Cardiac injury. IntroductionCardiovascular ailments, including coronary diseases, stroke, and myocardial infarction, are the dominant cause of morbidity and mortality worldwide (Balanescu et al., 2010). Some clinical studies have suggested a distinct relationship between dyslipidemia and the incidence of cardiovascular hazards (Eisen et al., 2008). Certain dyslipidemia indices, involving a pronounced rise in blood total cholesterol (TC) and triglyceride (TG) levels, are identified as a pervasive factor for the development of cardiovascular risks in the world population (Jung et al., 2022). Possibility of persistent oxidative stress, a mediator of allostatic burden, probably causes prolonged alterations in lipid signal with subsequent disturbance of the antioxidant machinery, accompanied by severe sequelae involving atherosclerosis (Goldstein and McEwen, 2002). Carbon tetrachloride (CCl4) is involved in multisystem dysregulation and oxidant/antioxidant imbalance. The hypercholesterolemia ascribed to CCl4 is implicated in oxidative stress (Niemann et al., 2017), owing to its possibility to impair the β-oxidation of fatty acids (Mahmoodzadeh et al., 2017), stimulate the esterification lipid process (Mesalam et al., 2021), and block the action of the lysosomal lipase enzyme (Marimuthu et al., 2013). CCl4 is a deleterious hepatotoxin (Xiao et al., 2012), suggesting the progression of profound complications consequent to the heart by producing free radicals (Islam et al. 2017). At a high dose, CCl4 profusely induces oxidative stress, cell necrosis, and inflammation, accompanied by acute tissue injury and apoptosis. Toxicity by CCl4, which offers a critical role in degenerative tissue conditions mediated through over generation of free radicals (CCl3 and CCl3OO) via the metabolism of the NADPH-cytochrome P450 system, ultimately promoting lipid peroxidation (Shi et al., 2013). Generated free radicals are involved in many pathological events, including dyslipidemia, atheroma development, endothelial damage, low-density lipoprotein oxidation, plaque injury, myocardial ischemic dysfunction, and recurrent thrombosis (Gupta et al., 2015). Oxidative stress is also responsible for diabetic heart conditions and congestive cardiomyopathy (Bugger and Abel, 2010). Various chemically induced methods in vitro and in vivo are available for provoking cardiac diseases and heart failure (Ahsan et al., 2020). The mechanism of CCl4-induced toxicity in laboratory animals has often been demonstrated via inhalation, oral, and intraperitoneal administration (Taslidere et al. 2014). Earlier studies have suggested that the toxicity of CCl4 can accelerate the generation of free radicals in various tissues (Khan and Ahmed, 2009). Free radicals and several associated species prompt oxidative damage through profound alterations in cell metabolism, thereby raising the calcium ions within cells, which then destroys the membrane permeability and cells via lipid peroxidation. Earlier supported findings through CCl4 model is not only associated with the rising level of malondialdehyde (MDA) but also with alleviating the antioxidant catalase (CAT), glutathione peroxidase (GPx), and superoxide dismutase (SOD) enzymes levels in the heart cells, elucidating that the heart is certainly targeted by CCl4 (Eshaghi et al., 2012). The release of reactive oxygen species (ROS) into the myocardium leads to subsequent mitochondrial dysfunction and cell injury. In this context, elevated ROS levels have been clearly identified in several models of heart dysfunction. Formerly, a growing scale in oxidative injury owing to increased ROS production and a close shortage in the antioxidant system has been proposed as a possible biomarker of cardiotoxicity/cardiac injury (Abdel-Wahab and Metwally, 2015). Many reports have suggested that CCl4 is often involved in the occurrence and progression of significant tissue damage by aggravated induction of free radicals rather than experimental cardiotoxicity induction (Ahsan et al., 2020). CCl4 is a powerful toxicant to the lungs and constitutes a potent peroxidizing model that usually creates systemic toxicity (Unsal et al., 2021). Pulmonary diseases are also devastating complications in patients with liver disease (Ma et al., 2013). The mortality experienced by patients, in addition to pulmonary diseases, is clinically relevant (Krowka et al. 2012). Generally, CCl4 causes lung injury and pneumonia during inhaler exposure (García De Acilu et al., 2015). However, CCl4, when applied intraperitoneally at low doses, prompts liver cirrhosis, lung injury, and pulmonary circulation disruption (Das et al. 2014). Similarly, the intraperitoneal application of 1 ml/kg CCl4 for 10 consecutive days induced an appreciable alveolar deficit (Ahmad et al., 2015). To achieve equilibrium between the extremely produced radical species and antioxidant machinery, a variety of potentials have been detected to lower the toxicity of CCl4 (Wang et al. 2021). Growing attention has confirmed that dietary supplementation with specific nutrients ameliorates oxidative injury in mammals (Hao et al., 2021). This evidence indicates that antioxidant nutrients in diets are an essential approach to minimize the possible damage emitted by ROS (Liu et al., 2018). Supplementation with branched-chain amino acids (BCAAs), mainly comprising isoleucine, leucine, and valine, confers beneficial roles in the regulation of ROS (Hu et al. 2017). Furthermore, dietary BCAAs prevent cardiovascular issues associated with ameliorating hypercholesterolemia (Li et al., 2022). The anti-atherogenic potential of valine and leucine has been shown to conquer the cardiovascular risks by Cojocaru et al. (2014), as well as to help modulate serum hyperlipidemia. Valine and leucine exhibited a protective impact on antioxidant activities with a restoring effect in rats with hypercholesterolemia. Dietary BCAAs could improve the role of pulmonary rehabilitation in chronic lung diseases associated with low BCAAs (De Bisschop et al., 2021). To date, dearth concerns describe the consequences of CCl4 toxicity in the incidence of cardiopulmonary injury. However, this protocol was prepared to decode the prophylactic measures of BCAAs by evaluating their consequences on different signaling pathways, such as oxidative stress and dyslipidemia profile, in cardiopulmonary-induced CCl4 experimental toxicity. In this study, toxicity of the heart and lung was induced through the administration of CCl4 by adult Wistar rats, and the scavenging free radical properties of the BCAAs were measured by assessing the SOD activity level as a known antioxidant and the extent of MDA content as a lipid peroxidation index. Materials and MethodsEthical approvalAll research assays were conducted in accordance with the Ethics Guidelines for the Laboratory Animals Use of the Faculty of Science. Ethical approval and protocol were obtained on 3rd September 2024 from South Valley University, Qena, Egypt, with a protocol No. 003/09/24. Experimental ratsAnimals of this experiment were twenty-eight male Wistar rats (200 ± 20 g), aged 3–4 months, supplied by Animal House from the University of Sohag, Egypt. All Wistar rats were secured in safeguarded cages after arrival into the laboratory room. Animals were kept in housing conditions and randomly assigned in 2–3 rats per cage to avoid any overcrowding between group-housed albino rats. Cages were changed biweekly and established using a constant light/dark system of 12 hours for each, with an air conditioning system of 22°C temperature and 50% relative air humidity. Animals were fed standard pellets with free-to-access tap water ad libitum for 2 weeks before the experimentation for acclimatization. ChemicalsCarbon tetrachloride is a chemical compound with the chemical structure of CCl4. Al-Nasr Chemicals, Cairo, Egypt, provided it. Using corn oil, CCl4 was liquefied in a 1:1 ratio and injected intraperitoneally into rats three times a week for 3 weeks (Khedr and Khedr, 2017). BCAAs were produced by Sigma-Aldrich, Louis (MO, USA). High-quality reagents and analytical standard chemicals are used in laboratory research. Experimental design and dosageTwenty-eight Wistar rats were obtained from the Laboratory Animal House of Sohag University, Egypt. Rats were acclimatized for two successive weeks in controlled environmental conditions with access to water and a suitable pelleted diet. Rats were randomly bifurcated under the random selection based on the body weights, into four categories as shown in Figure 1; seven rats were categorized for each group as follows: Group 1 (Control) consisted of seven un-treated animals, which employed a negative control, where the rats supplied standard pellets with free access to freshwater ad-libitum through the experiment period. Group 2 (CCl4-treated group) consisted of seven animals injected with 1.0 ml CCl4 dissolved in corn oil i.p. for 3 weeks, where the rats supplied standard pellets with free access to freshwater ad-libitum along the experiment. Group 3 (CCl4+10 g BCAAs-treated group) consisted of seven prior animals injected with 1.0 ml CCl4 dissolved in corn oil i.p. for 3 weeks and then supplied 10 g BCAAs g/kg bwt for an additional seven weeks. Group 4 (CCl4+20 g BCAAs-treated group) consisted of seven animals prior injected with 1.0 ml CCl4 dissolved in corn oil i.p. for 3 weeks and then supplied 20 g BCAAs g/kg bwt for additional 7 weeks. The injection of CCl4 was trice i.p. per a week for 3 successive weeks in groups 2, 3 & 4 Khedr and Khedr (2017) till cardiopulmonary injury fulfilled. After the induction of CCl4 toxicity through cardiopulmonary injury using diagnostic biopsies, groups 3 and 4 were orally supplemented with 10 g and 20 BCAAs g/kg but, respectively, according to the protocol of Marrone et al. (2023). Iqbal et al. (2021) recommended a dose of 10 g/kg bwt BCAAs containing 5.026 g leucine, 2.835 g isoleucine, 2.734 g valine, and 189.405 g casein once a day. A dose of 20 g/kg but BCAAs containing 9.446 g leucine, 6.002 g isoleucine, 5.224 g valine, and 179.328 g casein was also administered once a day for an additional 7 weeks. Rats in groups 1 and 2 were fed standard pellets deprived of BCAAs. However, the same diet was provided by groups 3 and 4 and full in BCAAs, with free access to freshwater ad libitum throughout the experiment period.
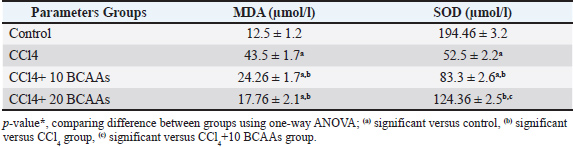
Fig. 1. Experimental design and dosage. Clinical signs and body weight (BW)Changes in behavioral patterns or clinical aberrations were observed in experimental animals, and the mean body weight was noted at 0 days, 3 weeks, and 10 weeks. Sample collectionAfter the feeding period, the rats of all groups were immediately euthanized through cervical dislocation. Blood and tissue samples were immediately extracted from each group for further biochemical and histopathological analysis after 12 hours of fattening. To segregate serum, blood samples were obtained through the tail vein in sterilized plain vacutainer tubes. Serum was centrifuged at 4,000 rpm for 10 minutes and collected for biochemical monitoring. Collected tissues from the heart and lungs were carefully immersed in a proper fixative for 24 hours until histological analysis (Bancroft et al., 2013). Other aseptic cardiac autopsies were further homogenized by washing in buffered saline, grinding, and centrifugation at 4°C. The heart tissues were assessed for oxidative and antioxidant activity profiles. Biochemical analysis of serumUsing standard diagnostic kits, spectrophotometric analyses of serum cholesterol and triglyceride levels (CAT. No. CH 12 20 & TR 20 30, Dokki, Egypt), respectively and were performed according to the methods of Richmond (1973) and Fossati and Prencipe (1982), respectively. Moreover, the enzymatic colorimetric method (Trinder, 1969) was described for assessing serum glucose levels (CAT. NO. GL 13 20, Dokki, Egypt). Oxidative stress analysisThe MDA level, a lipid peroxidation biomarker, was measured in the heart tissue following a spectrophotometric procedure (CAT. No. MD 25 28, Dokki, Egypt) illustrated by Ohkawa et al. (1979). Nishikimi et al. (1972) assessed SOD activity (CAT. No. SD 25 20, Dokki, Egypt). Kits for oxidative stress markers were procured by a bio-diagnostic company, Giza, Egypt. Histological evaluationFresh small-sized specimens of the heart and lungs with an average diameter of 2 cm2 were immediately dissected from each rat of the existing groups and rinsed in 10% neutral formalin for a day. Then, the specimens were processed in an automated tissue processor through the completion of dehydration in ascending scales of methanol (70%, 80%, 90%, and 95%), clearing in xylene to discard the methanol, and softening through embedding into a paraffin block built from paraffin waxes (melting point 55°C–60°C). Then, the sectioning process included mounting the tissue samples on a microtome and harvesting into desiccated sections of 4 μm thicknesses, blemished by hematoxylin and eosin (H&E), Masson’s trichrome, and bromophenol blue (BPB) stains according to standard protocols (Drury et al., 1980). The stained slides were inspected, photographed, and scored using a light microscope equipped with a digital camera. A total of 9 sections/3 slides were selected from each group for histopathological scoring. The ordinary semi-quantitative method was run using a scale sorted into none (0), minimal (+), moderate (++), and severe damage (+++) according to Gibson-Corley et al. (2013). Fibrosis scoringAnalysis of the photomicrographs of the heart and lung of the control, CCl4, CCl4 + 10 g BCAAs, and CCl4 + 20 g BCAAs was performed using Masson trichrome-stained tissues by the ImageJ software (Collins, 2007). Collagen fibers were separated greatly from the background, and their intensity was calculated using the “Threshold” index tool to evaluate the density and area of collagen fibers in Masson trichrome-stained sections. The threshold was manually adjusted until the entire green area of collagen fibers was highlighted in red. Then, the threshold area was measured. Statistical analysis of dataStatistical data analysis was performed using one-way analysis of variance using Windows Statistics, version 21.0 (SPSS Inc., Chicago, USA). The Scheffe post hoc test was used to evaluate all experimental data of the biochemical findings to evaluate significant variations between groups. Triplicates (n=3) were used for each treatment. Data are defined as means ± SD. The significance of the variations between means is based on p < 0.05 (Mustafy and Rahman, 2024). ResultsClinical signs and body weightThe CCl4 rats exhibited lowered appetite, decreased feeding intake, dullness, and inactivity. On day 0 of the experiment, the mean body weights of the rats in the CCl4, CCl4 + 10 BCAAs, and CCl4 + 20 BCAAs groups were 197.3 ± 3.0, 199.6 ± 4.7 and 198.3 ± 2.1 g, respectively, exhibiting no significant variations compared with the control weight of 203.0 ± 4.0 g. In contrast, in the second week after the injection, the mean body weights of the CCl4 group (217.0 ± 3.0 g), CCl4 + 10 BCAAs (220.3 ± 2.5 g), and CCl4 + 20 BCAAs (219.0 ± 3.0 g) were significantly decreased (p < 0.05) compared with the mean body weight of the control group (239.6 ± 4.0 g). In a comparison with CCl4 values (230.6 ± 2.1 g), the mean body weights at 10 weeks from experimentation were significantly increased (p < 0.05) to 256.3 ± 2.1 and 260.6 ± 2.5 g of the CCl4 + 10 BCAAs and CCl4 + 20 BCAAs groups, respectively, exhibiting significant improvement compared with the CCl4 group (Table 1). Table 1. Changes in body weight (mean ± standard deviation g) of rats treated with carbon tetrachloride and bovine serum albumin (BCAAs).
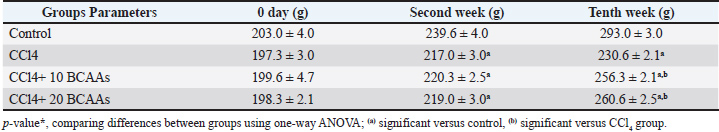
Biochemical findingTable 2 shows the obtained concentrations of some serum enzymatic lipograms (cholesterol and triglyceride) in the rats. Substantial increases (p < 0.05) were observed in the total cholesterol and triglyceride levels after CCl4 administration (116.5 ± 2.1 and 146.8 ± 3.6 mg/dl, respectively) compared with the control non-administrated rats (77.6 ± 1.4 and 92.9 ± 1.1 mg/dl, respectively). However, the concomitant administration of low and high doses of BCAAs with CCl4 significantly declined (p < 0.05) the serum cholesterol (107.8 and 100.9 ± 1.5 mg/dl, respectively) and triglyceride levels (128.6 ± 0.95 mg/dl) compared with CCl4, indicating that the BCAA-induced diet supports the improvement in lipid levels in the blood. Notably, the mean glucose level displayed an observable increase (p < 0.05) in the CCl4 group (161.6 ± 2.1 mg/dl). However, they increased significantly (p < 0.05) in both combined groups of CCl4 plus BCAAs (153.5 ± 2.7 and 154.3 ± 3.5 mg/dl, respectively) compared with the control (108.6 ± 1.8 mg/dl), as shown in Table 2. Table 2. Effect of different doses of branched-chain amino acids on serum total cholesterol (TC), triglyceride (TG), and glucose levels in carbon tetrachloride-induced rats (mean ± SD mg/dl each).
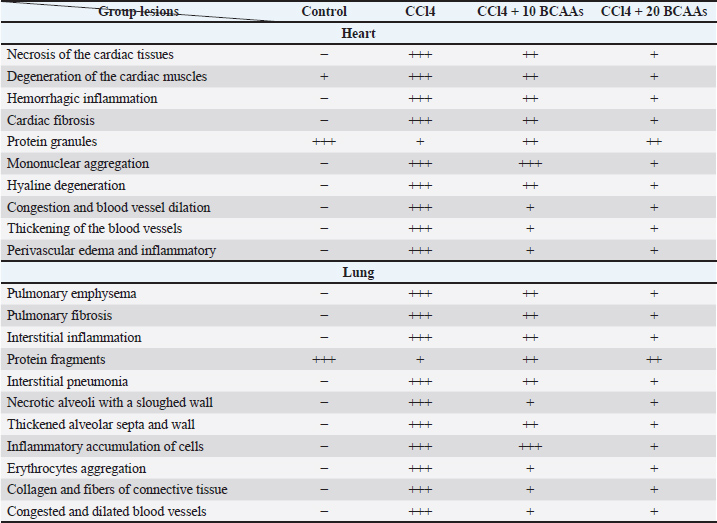
Results of oxidative stressCCl4 toxicity tends to induce consequential changes in the biomarker of lipid peroxidative stress (LPO), characterized by a significantly higher (p < 0.05) in the MDA (43.5 ± 1.7 µmol/l) compared with the control values (12.5 ± 1.2 µmol/l), opposing SOD activity, which manifested a significant reduction (p < 0.05) among CCl4-treated hearts (52.5 ± 2.2 µmol/l) compared with the activity of control (194.46 ± 3.2 µmol/l). In comparison with the CCl4 values, the combined treatment of CCl4 with both 10 and 20 BCAAs resulted in a considerable decrease (p < 0.05) in MDA level was 24.26 ± 1.7 and 17.76 ± 2.1 µmol/l, respectively. A significant rise (p < 0.05) in the SOD activity, however, was 83.3 ± 2.6 and 124.36 ± 2.5 µmol/l, respectively. Accordingly, the administration of BCAAs decreased the unusual increase in LPO level and greatly enhanced the enzymatic antioxidant activity relative to the control findings (Table 3). Table 3. The effect of different branched-chain amino acids doses on carbon tetrachloride-induced rats on lipid peroxidative biomarker (MDA), and antioxidant activity (SOD) (mean ± SD µmol/l each).
Histological and histochemical changesThe histological and histochemical alterations (Table 4) illustrated that the CCl4-received group had more extensive and intense alterations than the CCl4 + BCAA-treated groups. Table 4. Nominal semi-quantitative histological scores of the heart and lung of the control, carbon tetrachloride, and branched-chain amino acid-treated groups.
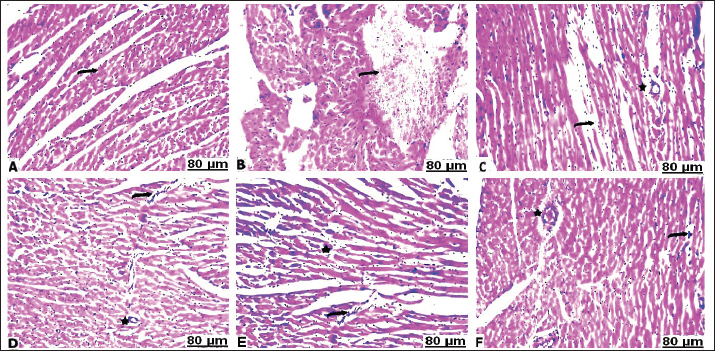
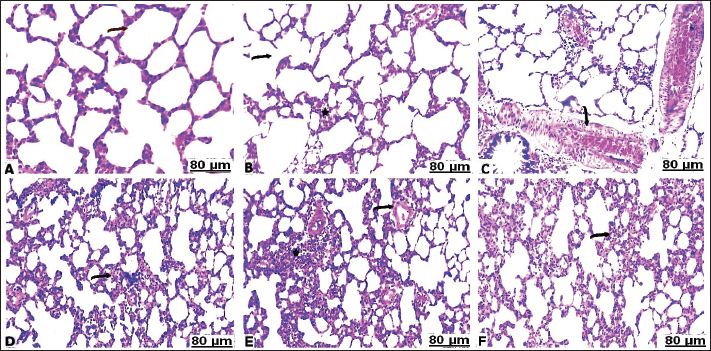
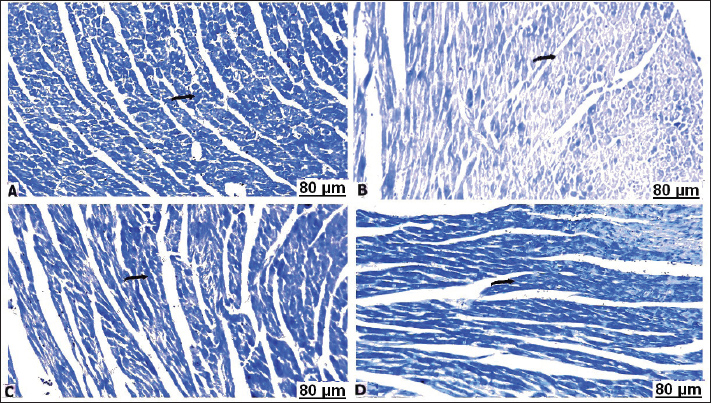
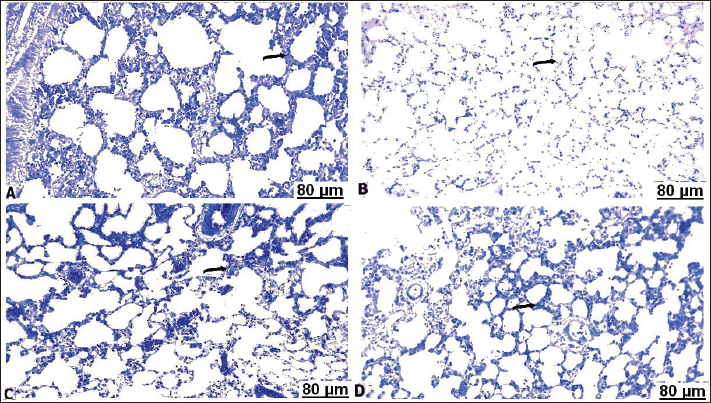
H&E stainHistopathological examination of the heart in control group showed normalization of cardiac tissue, indicating good striation of the cardiac muscles and an intact vein (Fig. 2A). In contrast, CCl4-intoxicated rats showed severe hemorrhage with massive infiltration of red blood cells, perivascular inflammatory edema, hyaline deposition, fibrotic collagen precipitates on the cardiac surface, ballooning degenerative changes of the myocardium, myolysis, and intense muscle necrosis replaced by focal mononuclear aggregation (Fig. 2B and C). BCAA treatment at varying doses resulted in an improvement in the heart architecture characterized by improved cardiac muscle cells, minimal mononuclear infiltrates, weak hyaline deposition, and minimally congested blood vessels (Fig. 2D–F). The histopathological examination of the lung alveoli and bronchi of the control rats was histologically normal (Fig. 3A). Conversely, in the lung parenchyma of CCl4-treated rats, there were distinct areas of emphysema and interstitial pneumonia associated with abundant leukocyte infiltration. Many alveoli had thickened walls containing inflammatory cells, mostly mononuclear cells, eosinophils, and neutrophils. Some alveoli also revealed necrotic alveolar walls, indicating diffuse damage. Numerous erythrocytes were observed in and around the alveoli. The interstitial cell population infiltrated and thickened with mononuclear cells. The inflamed areas exhibited focal collagen and connective tissue fiber accumulation. Several alveolar macrophages were also invaded. The blood vessels showed extensive congestion, dilatation, and thickening of their walls with numerous inflammatory cells and fibrous tissues (Fig. 3B and C). CCl4+BCAA-treated rats showed a restored normal histological structure of the lung. Moderate degree of alveolar thickening with moderate accumulation of leucocytes Blood vessels were also slightly congested and dilated (Fig. 3D and E). Furthermore, the CCl4+BCAA-treated rats showed mild alveolar thickening with a mild leukocyte accumulation (Fig. 3F).
Fig. 2. Heart sections of control (A), carbon tetrachloride (B, C), carbon tetrachloride + 10 g branched-chain amino acids (D, E), and carbon tetrachloride + 20 g branched-chain amino acids (F): A) normal striation of cardiac muscles. B) severe interstitial hemorrhage. C) necrosis of muscle fiber with a loss of striation (arrow), perivascular edema, and cellular infiltrates (star). D) Mild lymphocyte infiltration (arrow) with slight perivascular infiltration (star). E) Mild cardiac myolysis (star) and interstitial inflammation (arrow). F) Minimal leucocyte infiltration (arrow) with slight vessel wall thickening (star). [Hematoxylin and eosin staining].
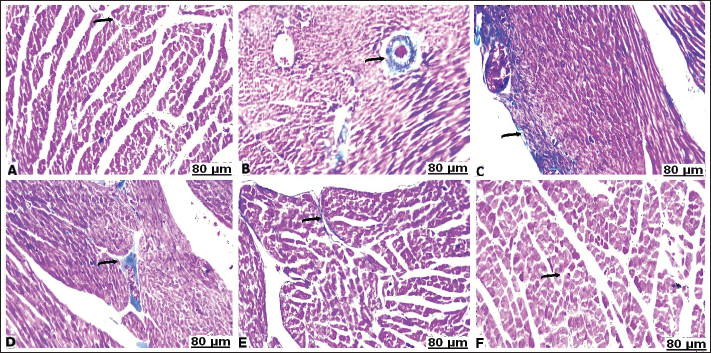
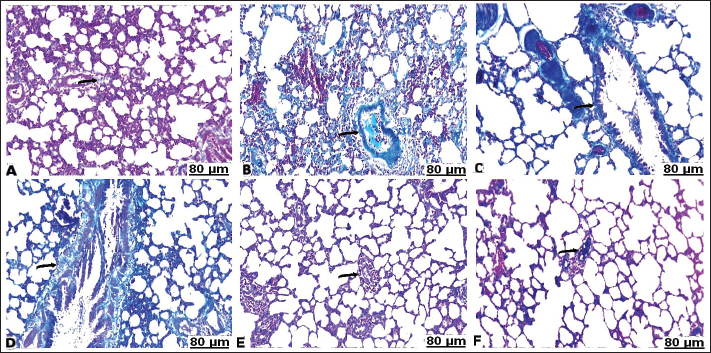
Fig. 3. Lung sections of control (A), carbon tetrachloride (B, C), carbon tetrachloride +10 g branched-chain amino acids (D, E), carbon tetrachloride +20 g branched-chain amino acids (F): A) showing intact alveolar wall. B) showing scattered areas of alveolar emphysema (arrow) and thickening of the interalveolar septa with mononuclear cells (star). C) showing severe congestion with thickening of the blood vessels (arrow). D) showing interstitial infiltration of alveolar septa with lymphocytes (arrow). E) showing focal accumulation of leucocytes (star) and mild congestion of the blood vessels (arrow). F) showing interstitium infiltrated with several leucocytes (arrow). [Hematoxylin and eosin staining]. Masson’s trichrome staining methodThe area of fibrosis (%) of the heart and lung sections depends on the average proportion and collagen distribution determined by morphometric analysis (Table 5). The area percentage of heart fibrosis was significantly increased (p < 0.05) in all treated groups compared with the control, as evidenced by increased areas of the collagen depositions. The heart sections of the CCl4 group recorded a higher percentage than those of the BCAA group. Co-treated CCl4 with 20 BCAAs exhibited the fewest percentage of heart fibrosis, comparable with 10 BCAAs. Although the mean proportion (%) of lung fibrosis was significantly increased (p < 0.05) CCl4 and CCl4 + 10 BCAA-treated groups compared with the control group, the CCl4 + 20 BCAA-treated group recorded a smaller proportion (%) of lung fibrosis; however, BCAAs verified dose-dependent alterations. Accordingly, the Masson staining method exhibited almost hard collagen detection in the control heart (Fig. 4A). Significant fibrosis characterized by the deposition of large amounts of collagen was observed after CCl4 treatment (Fig. 4B–D). Treatment with 10 and 20 mg/kg BCAAs significantly diminished collagen deposition (Fig. 4E and F). The same context of the control lung showed scarce collagen infiltrates in the pulmonary parenchyma (Fig. 5A). CCl4 treatment was accompanied by the pronounced activation of fibrosis with high collagen and fibrous tissue infiltration (Fig. 5B–D). Lung fibrosis was alleviated after using varying doses of BCAAs, as evidenced by faintly stained fibrous tissues (Fig. 5E and F).
Fig. 4. Heart sections of control (A), carbon tetrachloride (B, C, D), carbon tetrachloride + 10 g branched-chain amino acids (E), and carbon tetrachloride + 20 g branched-chain amino acids (F): A) weak intensity of the collagen fibers staining (arrow). (B, C, D) increased fibrosis evident by a great rise in the stained collagen fibers (arrow). (E) Low percentages of the infiltrated collagen fibers (arrow). F) Scarcely distributed collagen fibers (arrow). [Masson’s trichrome stain].
Fig. 5. Lung sections of control (A), carbon tetrachloride (B, C, D), carbon tetrachloride + 10 g branched-chain amino acids (E), and carbon tetrachloride + 20 g branched-chain amino acids (F): (A) faintly stained nuclei of collagen fibers (arrow). (B, C, D) marked incidence of perivascular and interstitial fibrosis (arrow). (E) significant depletion of collagen fibers (arrow). (F) weakly precipitated collagen fibers (arrow). [Masson’s trichrome stain]. Table 5. Fibrosis scoring area (%) of the heart and lung of the experimental groups compared with control tissues.
BPB techniqueThe control heart was specified by high total protein content (Fig. 6A). A highly positive bromophenol blue reaction was observed in the cytoplasm, characterized by equally distributed bluish granules of definite sizes. Total protein revealed a striking decrease in the cytoplasm of the heart cells of the CCl4 group, displaying high percentages of protein-devoid cells and cytoplasmic vacuolation, particularly around the blood vessels (Fig. 6B). However, oral BCAA administration concomitant with CCl4 could reduce alterations in protein content in rat hearts compared with the CCl4 group. The BPB reaction was increased, exhibiting dark-blue spotted granules of varying protein contents, peripheral in position or perinuclear, particularly adjacent to the vasculature (Fig. 6C and D).
Fig. 6. Heart sections of control (A), carbon tetrachloride (B), carbon tetrachloride + 10 g branched-chain amino acids (C), and carbon tetrachloride + 20 g branched-chain amino acids (D): (A) a considerable distribution of protein elements within the cytoplasm (arrow). (B) a weak response of the cell's cytoplasm toward the BPB reaction (arrow). (C) a low decrease in protein elements (arrow). (D) a noticeable increase in protein content (arrow). [BPB stain]. The lungs of the control vehicle revealed marked protein content in the cytoplasm of the lung tissues, as indicated by the large number of bluish-colored fine materials of different sizes (Fig. 7A). Conversely, CCl4 injection into rats for three consecutive weeks resulted in a sustainable reduction in protein content, as evidenced by less BPB-stained cells. In these lungs, the protein content displayed minimal intensity of the reaction and became barely detectable (Fig. 7B). However, the combined administration of BCAAs and CCl4 strengthened the BPB reaction and resulted in a more detectable number of bluish-colored granules of varying sizes, indicating increased protein contents similar in intensity to those in the control lung (Fig. 7C and D).
Fig. 7. Lung sections of control (A), carbon tetrachloride (B), carbon tetrachloride +10 g branched-chain amino acids (C), and carbon tetrachloride +20 g branched-chain amino acids (D): (A) Homogenous dense, blue-stained granules indicating positive affinity to BPB. B) Very low protein particles (arrow). (C) A low decrease in protein elements (arrow). (D) A noticeable increase in protein content (arrow). [BPB stain]. DiscussionOxidative stress is an imbalance between the radical species and the physiological antioxidant enzymatic machinery that neutralizes ROS or scavenges the resulting damage The emitted ROS can destroy all cellular components, yielding fatal consequences (Shaban et al., 2017), and excites a crucial part in the pathogenesis of multiple tissues, including the kidney, brain, spleen, liver, and lung (Ragab et al., 2013). Reducing ROS expulsion is essential for cell survival and the prevention of further oxidative damage (Morgan and Liu, 2011). Several toxic chemicals, including carbon tetrachloride, methanol, bromobenzene, and aromatic hydrocarbons, can cause systemic injury by producing reactive oxygen species (Rahmouni et al., 2017). Carbon tetrachloride is a copious environmental toxicant that can cause oxidative stress with over-created free radicals in various organs, notably the lungs and kidneys, motivating generalized toxicity (Habashy et al., 2021). Carbon tetrachloride (CCl4), a vigorous carcinogenic agent, is an organic industrial solvent that may cause liver, kidney, nervous system, and lung dysfunction. CCl4 is taken up by the respiratory tract, gastrointestinal system, and skin, and then, through cytochrome P-450 metabolism, it exerts toxic impacts via the free radicals of trichloromethyl and trichloromethyl peroxyl metabolites (Khan, 2012). These radicals combine fatty acids in the lung cell membrane to enhance DNA fragmentation and lipid peroxidation. Besides this, they disturb the antioxidant enzyme system, encompassing superoxide dismutase, catalase, and glutathione enzymes. Because of aggregations of exaggerated fibroblasts, monocytes, macrophages, and neutrophils in blood vessels, CCl4 caused lung toxicity by interstitial cell degeneration, fibrosis, and ruptures of intra-alveolar septa. Increased ROS is involved in pulmonary fibrosis, lung carcinoma, emphysema, chronic bronchitis, and pleural diseases (Ahmad et al., 2015). CCl4 may cause excessive transforming growth factor beta1, leading to systemic fibrosis in multiple tissues, primarily the lung and liver (Fan et al., 2013). CCl4 promotes lung injury by accelerating lipid peroxidation and increasing MDA levels (Ganie et al., 2010). CCl4 induces cellular apoptosis by activating the caspase pathway, thereby promoting tissue injury (Xie et al., 2015). Carbon tetrachloride induced heart dysfunction in rats Intoxicated rats exhibited cardiac histopathology in the form of fibrotic strands in the heart wall, massive deposition of collagen on the cardiac layer, and ballooning degeneration of endocardial muscle fibers (El-Baz et al., 2015). In this study, the H&E-stained sections of CCl4-treated heart revealed necrosis and degenerative changes of the cardiac muscles, inflammation, hemorrhage, and congestion of the blood vessels. Toxic response of CCl4-enhanced heart infarction A single CCl4 injection at l.2 ml/kg induced myocardial damage identified by focal masses of huge degeneration with scattered, blue-stained fibrosis zones of fibrous tissues. CCl4-intoxicated rats had increased levels of serum pro-inflammatory biomarkers and C-reactive protein in heart cells (Al-Rasheed et al., 2014). CCl4-mediated cardiotoxicity and cardiac injury interfere with antioxidant mechanisms and may involve oxidative stress. The elevated rate of lipid oxidation was more detectable in CCl4-induced cardiac injury. CCl4 is linked to scattered necrosis, widespread inflammation, cytoplasmic vacuolization, interstitial edema, and disintegrated myofibrils (Alsaffar et al., 2022). H&E-lung sections of the CCl4 group exhibited interstitial pneumonia, abundant leukocyte infiltration, thickening of the interalveolar wall, and extensive congestion. Intense damage was detected in lung morphology after CCl4 exposure, which was recognized by disorganized alveolar architecture, widespread thickening of the interalveolar space, and collapse of alveolar septa similar to those findings by Habashy et al. (2021). Acute and chronic lung injury in rats by i.p. CC14 injection for 2 and 5 weeks, respectively, exhibited features of discrete alveolar damage. Acute lung injury is distinguished by acute hemorrhagic interstitial pneumonia, whereas chronic interstitial pneumonia is caused by long-term injury. Connective tissue revealed intra- and interstitial alveolar fibrosis in patients with short-term injury. The mechanism of CC14 lung injury is suggested by lipid peroxidation-correlated radicals (Pääkkö et al., 1996). Light microscopy of the CCl4-exposed lung showed an abnormal histological appearance in the form of alveolar epithelial injury, edema, sloughed epithelial cells, and mononuclear inflammatory infiltrates in the lung’s interstitial tissues. Intensive congestion of the large blood vessels, swelling of epithelial cells, shedding of cellular epithelium, and scattered visualizations, with intensive fibrosis of interstitial tissues with fibrous cells using Masson’s trichrome. An examination of the lungs of cirrhotic mice treated with CCl4 revealed the presence of strong fibrotic lesions in the lungs of CCl4-associated cirrhotic animals. H&E staining revealed that the most evident changes in lung sections were a lack of normal alveolar organization, significant pulmonary fibrosis, notable thickening of the alveolar interstitium, and prominent disorganization of the alveolar space (Das et al., 2014). The current design of Masson’s trichrome-stained sections recorded an enormous increase in the collagen and fibrous tissue areas that induce fibrosis. Similar findings were consistent with those of Ding and Choi (2014), who detected the overexpression of collagen and fibrous tissues by mesangial cells of the kidney. Trichrome staining illustrated blue staining, intense extracellular matrix accumulation, and peribronchial and perivascular lung fibrosis in CCl4-cirrhotic mice. Picrosirius Red staining showed substantial collagen deposition around the vasculature and in the lung parenchyma (Das et al., 2014). The incidence of protein elements by BPB-stained microscopic sections displayed a marked decrease in cardiac and lung cells following CCl4 intoxication, while significantly reversed close to normal protein level in BCAA-treated groups. This alteration is attributed to oxidative damage to cell proteins and deterioration in cellular function and contents (Ohta et al., 2000). In this study, the total weight of rats dropped remarkably after intraperitoneal exposure to CCl4 because of hyperlipidemia activity. CCl4 supplementation tended to increase lipid and glucose levels in the serum, contrary to the BCAA-induced groups. Significant decreases in serum insulin and increases in blood glucose levels were observed after CCl4 supplementation (Khedr and Khedr, 2017). The subsequent administration of CCl4 is associated with hyperlipidemia activity characterized by a significant elevation in glucose and cholesterol levels. Changes in the lipid profile are caused by subsequent liver injury due to CCl4 toxicity (Dutta et al., 2018). Ultimately, Hussein and Khan (2022) showed that acute exposure to CCl4 induced a marked increase in triglyceride, cholesterol, low-density lipoproteins, and glucose levels. CCl4 intoxication triggered lipid metabolism parameters, leading to a significant increase in triglyceride and cholesterol levels. However, orally administered BCAAs lowered the serum levels of cholesterol and triglyceride in patients with liver cirrhosis (Takeshita et al., 2009). In addition, Arakawa et al. (2011) reported that BCAA-rich meals improved and reversed triglyceride levels in a fat-rich mouse model. Generally, leucine supplementation improved body weight, attributed to marked improvements in glucose and lipid metabolism (Jiao et al., 2016). Macotela et al. (2011) explored that leucine causes motivation in glucose tolerance and insulin index without changing the weight gain of mice with obesity. The improved influence of leucine on body weight and lipid markers is correlated with the regulation of lipid and carbohydrate metabolism (Chen et al., 2012). Leucine supplementation induces the beneficial role of BCAAs in improving the lipid profile in the blood serum, provoking a marked decrease in microvesicular steatosis illustrated by decreased liver oxidation and improved lipid processing with concomitant inhibition of de novo lipid synthesis associated with obesity (Fu et al., 2015). Improvement in liver lipid catabolism is associated with a prominent decline in insulin level, lipid peroxidation, liver diacylglycerol, and inflammation, further confirmed by profound repression of crucial inflammatory proteins and pathways. The valine and isoleucine constituents found in BCAAs serve as nutritional treatments to restore metabolic or inflammatory homeostasis in mice with obesity-associated steatotic livers (Gart et al., 2022). Noguchi et al. (2006) detected the beneficial effect of BCAAs to regulate lipid metabolism and heart function, who confirmed that leucine and valine neutralized the deleterious effect of lipid levels immediately in atherosclerotic episodes. However, valine had a quicker response than isoleucine and leucine on the improvement of biochemical indices, according to the histopathological evaluation (Ifrim et al., 2018). In this experimental work, the oxidative component of MDA in CCl4-treated hearts was significantly higher than that in control animals, contrary to a significant decline in SOD antioxidant enzyme. The increased MDA level suggests the continual stimulation of the peroxidation process, inducing tissue injury and insufficiency of the antioxidant procedures to neutralize the excessively generated radical species (Ganie et al., 2011). CCl4-induced lipid peroxidation was significantly attenuated by branched-chain amino acids, which was reversed by an improvement in antioxidant enzyme activity. The potential influence of BCAAs for the treatment of various disorders is shown through the enhancement of metabolic processes and the strengthening of mitochondrial bioenergetics for maintaining mitochondrial health (Ruocco et al., 2021). BCAAs can reduce oxidative stress and modulate the antioxidant defense system in medium-aged mice (D’Antona et al., 2010). Valine plays a favorable role in lipid metabolism (Bishop et al., 2020). While studies on BCAAs, particularly leucine and isoleucine, have shown an advantageous impact on mitochondrial function, cellular metabolism, and oxidative stress (Tamanna and Mahmood, 2014), valine confers vital function in protecting mitochondrial viability by neutralizing cellular/mitochondrial harms activated by oxidative stress (Sharma et al., 2024). Dietary BCAAs had a protective effect on heart and lung architectures. The beneficial contribution of leucine, isoleucine, and valine supplementation results in significant mitigation of histopathological lesions due to a rich cholesterol diet. Similar histopathological examinations of leucine, isoleucine, and valine revealed mild lesions in the vascular endothelium (Ifrim et al., 2018). The reduced microscopic lesions are a consequence of the addition of BCAAs to the diet of mice, which ultimately strengthens the ROS defense mechanism in the tissues and improves mitochondrial health status (Valerio et al., 2011). ConclusionTaken together, the current experimental results explicated the systemic toxicity of CCl4 in the heart and lungs of rats and the significant therapeutic potential of BCAAs. The biochemical findings and histopathological results elicited the potency of BCAAs in alleviating CCl4-induced oxidative toxicity induced by CCl4 through suppressing biochemical disturbance and fibrosis. Accordingly, this investigation suggests that BCAAs, notably valine, leucine, and isoleucine, have an influential protector with strong radical scavenger function against CCl4-induced heart and lung damage. Thus, in dose-dependent amelioration, BCAAs are being proposed as a promising and efficient anti-toxic agent that targets different pathways in various organs, particularly the heart and lung tissues. However, specific mechanisms through BCAAs remain unclear and require further investigation. AcknowledgmentsThe authors would like to thank the technicians at the Faculty of Medicine, South Valley University, for their assistance in completing this study. Conflict of interestThe authors declare no conflict of interest. FundingThis work was supported by the Slovak grants VEGA no. 1/0162/23 and KEGA no. 011UVLF-4/2024. Authors’ contributionsAll authors contributed to the study conception and design. Conceptualization: ZA-A, FAM, and HA-A; methodology: ZA-A, FAM, HA-A, AWB, and AMM; software: ZA-A, FAM, HA-A, AWB, AMM, and MMJ; validation: ZA-A, FAM, HA-A, AWB, and AMM; formal analysis: ZA-A, FAM, HA-A, AWB, AMM, MMJ, AE, IFR, FZ, and MZ; resources: ZA-A, FAM, HA-A, AWB, AMM, and MMJ.; data curation: ZA-A, FAM, HA-A, AWB, AMM, MMJ, AE, IFR, FZ, and MZ; writing—original draft preparation: ZA-A, FAM, HA-A, AWB, AMM, MMJ, AE, IFR, FZ, and MZ; writing—review and editing: ZA-A, FAM, HA-A, AWB, AMM, MMJ, AE, and IFR; visualization: HA-A, AWB, AMM, MMJ, AE, IFR, FZ, and MZ; supervision: ZA-A, FAM, and MMJ; funding acquisition: MZ and FZ. All authors have read and approved the published version of the manuscript. Statement of the institutional review boardAll research assays were conducted in accordance with the Ethics Guidelines for the Laboratory Animals Use of the Faculty of Science. Ethical approval and protocol were obtained on 3rd September 2024 from South Valley University, Qena, Egypt (Protocol No. 003/09/24). Data availability statementThe datasets generated for this study are available upon request from the corresponding author. ReferencesAbdel-Wahab, B.A. and Metwally, M.E. 2015. Clozapine-Induced Cardiotoxicity: role of Oxidative Stress, Tumour Necrosis Factor Alpha and NF-κβ. Cardiovasc. Toxicol. 15, 355–365; doi:10.1007/s12012-014-9304-9 Ahmad, B., Khan, M.R. and Shah, N.A. 2015. Amelioration of carbon tetrachloride-induced pulmonary toxicity with Oxalis corniculata. Toxicol. Ind. Health. 31, 1243–1251; doi:10.1177/074823371348724 Ahsan, F., Mahmood, T., Usmani, S., Bagga, P., Shamim, A., Tiwari, R., Verma, N. and Siddiqui, M.H. 2020. A conglomeration of preclinical models related to myocardial infarction. Braz. J. Pharm. Sci. 56, 18365; e18365 Al-Rasheed, N.M., Al-Rasheed, N.M., Faddah, L.M., Mohamed, A.M., Mohammad, R.A. and Al-Amin, M. 2014. Potential impact of silymarin in combination with chlorogenic acid and/or melatonin in combating cardiomyopathy induced by carbon tetrachloride. Saudi J. Biol. Sci. 21, 265–274; doi:10.1016/j.sjbs.2013.09.006 Alsaffar, R.M., Rashid, S., Ahmad, S.B., Rehman, M.U., Hussain, I., Parvaiz Ahmad, S. and Ganaie, M.A. 2022. D-limonene (5-(one-methyl-four-1-methylethenyl) cyclohexane) diminishes CCl4-induced cardiac toxicity by alleviating oxidative stress, inflammatory and cardiac markers. Redox Rep. 27, 92–99. Arakawa, M., Masaki, T., Nishimura, J., Seike, M. and Yoshimatsu, H. 2011. The effects of branched-chain amino acid granules on the accumulation of tissue triglycerides and uncoupling proteins in diet-induced obese mice. Endocrine. J. 58, 161–170. Balanescu, S., Calmac, L., Constantinescu, D., Marinescu, M., Onut, R. and Dorobantu, M. 2010. Systemic inflammation and early atheroma formation: are they related?. Maedica 5, 292. Bancroft, J.D., Layton, C. and Suvarna, S.K. 2013. Bancroft’s theory and practice of histological. Elsevier, pp. 15. Bishop, C.A., Schulze, M.B., Klaus, S. and Weitkunat, K. 2020. The branched-chain amino acids valine and leucine have differential effects on hepatic lipid metabolism. FASEB. J. 34, 9727–9739; doi:10.1096/fj.202000195R Bugger, H. and Abel, E.D. 2010. Mitochondria in the diabetic heart. Cardiovasc. Res. 88, 229–240; doi:10.1093/cvr/cvq239 Chen, H., Simar, D., Ting, J.H., Erkelens, J.R. and Morris, M.J. 2012. Leucine improves glucose and lipid status in offspring from obese dams, dependent on diet type, but not caloric intake. J. Neuroendocrinol. 24, 1356–1364; doi:10.1111/j.1365-2826.2012.02339.x Cojocaru, E., Filip, N., Ungureanu, C., Filip, C. and Danciu, M. 2014. Effects of valine and leucine on some antioxidant enzymes in hypercholesterolemic rats. Health 6, 2313. Collins, T.J. 2007. ImageJ for microscopy. Biotechniques 43(sup1), S25–S30. D'Antona, G., Ragni, M., Cardile, A., Tedesco, L., Dossena, M., Bruttini, F., Caliaro, F., Corsetti, G., Bottinelli, R., Carruba, M.O., Valerio, A. and Nisoli, E. 2010. Branched-chain amino acid supplementation promotes survival and supports cardiac and skeletal muscle mitochondrial biogenesis in middle-aged mice. Cell. Metab. 12(2010), 362–372; doi: 10.1016/j.cmet.2010.08.016 Das, M., Boerma, M., Goree, J.R., Lavoie, E.G., Fausther, M., Gubrij, I.B., Pangle, A.K., Johnson, L.G. and Dranoff, J.A. 2014. Pathological changes in pulmonary circulation in carbon tetrachloride (CCl4)-induced cirrhotic mice. PLos One. 9, e96043; doi:10.1371/journal.pone.0096043 De Bisschop, C., Caron, F., Ingrand, P., Bretonneau, Q., Dupuy, O. and Meurice, J.C. 2021. Does branched-chain amino acid supplementation improve pulmonary rehabilitation effect in COPD?. Respiratory. Med. 189, 106642; doi:10.1016/j.rmed.2021.106642 Ding, Y. and Choi, M.E. 2014. Regulation of autophagy by TGF-β: emerging role in kidney fibrosis. Seminars Nephrology 34, 62–71; doi:10.1016/j.semnephrol.2013.11.009 Drury, R., Wallington, A. and Cameron, R. 1980. Carleton’s Histological Techniques. New York, NY: Oxford University Press. Dutta, S., Chakraborty, A.K., Dey, P., Kar, P., Guha, P., Sen, S., Kumar, A., Sen, A. and Chaudhuri, T.K. 2013. Amelioration of CCl4 induced liver injury in swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill. PLos One. 13, e0196411; doi:10.1371/journal.pone.0196411 Eisen, A., Tenenbaum, A., Koren-Morag, N., Tanne, D., Shemesh, J., Imazio, M., Fisman, E.Z., Motro, M., Schwammenthal, E. and Adler, Y. 2008. Calcification of the thoracic aorta as detected by spiral computed tomography among stable angina pectoris patients: association with cardiovascular events and death. Circulation 118, 1328–1334; doi:10.1161/circulationaha.107.712141 El-Baz, F.K., Khalil, W.K., Aly, H.F., Shoman, T.M. and Saad, S.A. 2015. The ameliorating effect of J. curcas extract against CCl4 induced cardiac toxicity and genotoxicity in albino rats. Int. J. Pharm. Sci. Rev. Res. 34, 223–233. Eshaghi, S.Z., Banihabib, V.N. and Farokhi, P.M. 2012. Cardioprotective effect of Cornus mas fruit extract against carbon tetrachloride induced-cardiotoxicity in albino rats. J. Basic Appl. Sci. Res. 2, 11106–11114. Fan, X., Zhang, Q., Li, S., Lv, Y., Su, H., Jiang, H. and Hao, Z. 2013. Attenuation of CCl4-induced hepatic fibrosis in mice by vaccinating against TGF-β1. PLos One. 8, e82190; doi:10.1371/journal.pone.0082190 Fossati, P. and Prencipe, L. 1982. Serum triglycerides determined colorimetrically with an enzyme that produces hydrogen peroxide. Clin. Chem. 28(10), 2077–2080; doi:10.1093/clinchem/28.10.2077 Fu, L., Bruckbauer, A., Li, F., Cao, Q., Cui, X., Wu, R., Shi, H., Zemel, M.B. and Xue, B. 2015. Leucine amplifies the effects of metformin on insulin sensitivity and glycemic control in diet-induced obese mice. Metab. Clin. Exp. 64, 845–856; doi:10.1016/j.metabol.2015.03.007 Ganie, S.A., Haq, E., Hamid, A., Qurishi, Y., Mahmood, Z., Zargar, B.A., Masood, A. and Zargar, M.A. 2011. Carbon tetrachloride induced kidney and lung tissue damages and antioxidant activities of the aqueous rhizome extract of Podophyllum hexandrum. BMC Complement. Altern. Med. 28, 17; doi:10.1186/1472-6882-11-17 Ganie, S.A., Haq, E., Masood, A. and Zargar, M.A. 2010. Amelioration of carbon tetrachloride induced oxidative stress in kidney and lung tissues by ethanolic rhizome extract of Podophyllum hexandrum in Wistar rats. J. MedPlants. Res. 4, 1673–1677; doi:10.5897/JMPR10.232 García De Acilu, M., Leal, S., Caralt, B., Roca, O., Sabater, J. and Masclans, J.R. 2015. The Role of Omega-3 Polyunsaturated Fatty Acids in the Treatment of Patients with Acute Respiratory Distress Syndrome: a Clinical Review. BioMed Res. Int. 2015, 653750; doi:10.1155/2015/653750 Gart, E., Van Duyvenvoorde, W., Caspers, M.P.M., Van Trigt, N., Snabel, J., Menke, A., Keijer, J., Salic, K., Morrison, M.C. and Kleemann, R. 2022. Intervention with isoleucine or valine corrects hyperinsulinemia and reduces intrahepatic diacylglycerols, liver steatosis, and inflammation in Ldlr-/-.Leiden mice with manifest obesity-associated NASH. FASEB. J. Off. Publication. Fed. Am. Societies. For. Exp. Biol. 36, e22435; doi:10.1096/fj.202200111R Gibson-Corley, K.N., Olivier, A.K. and Meyerholz, D.K. 2013. Principles for valid histopathologic scoring in research. Vet. Pathol. 50, 1007–1015; doi:10.1177/0300985813485099 Goldstein, D.S. and McEwen, B. 2002. Allostasis, homeostats, and the natu. Stress 5, 55–58. Gupta, A.K., Irchhaiya, R. and Misra, C. 2015. Free radical scavenging activity of Rauwolfia serpentina rhizome against CCl4 induced liver injury. Int. J. Pharm. 2, 123–126. Habashy, N.H., Kodous, A.S. and Abu-Serie, M.M. 2021. Targeting ROS/NF-κB signaling pathway by the seedless black Vitis vinifera polyphenols in CCl4-intoxicated kidney, lung, brain, and spleen in rats. Scientific Rep. 11, 16575. Hao, Y., Xing, M. and Gu, X. 2021. Research Progress on Oxidative Stress and Its Nutritional Regulation Strategies in Pigs. Animals 11, 1051384. Doi: 10.3390/ani11051384 Hu, J., Nie, Y., Chen, S., Xie, C., Fan, Q., Wang, Z., Long, B., Yan, G., Zhong, Q. and Yan, X. 2017. Leucine reduces reactive oxygen species levels via an energy metabolism switch by activation of the mTOR-HIF-1α pathway in porcine intestinal epithelial cells. Int. J. Biochem. &. Cell Biol. 89, 42–56; doi:10.1016/j.biocel.2017.05.026 Hussein, M. and Khan, R. 2022. CCL4-induced hepatotoxicity: study in rats intoxicated with carbon tetrachloride and treated with camel milk and urine. J. Chem. Stud. 1, 07–11; doi:10.32996/jcs.2022.1.1.2 Ifrim, S., Amalinei, C., Cojocaru, E. and Matei, M.C. 2018. Administration of valine, leucine, and isoleucine improved plasma cholesterol and mitigated the preatherosclerotic lesions in rats fed with hypercholesterolemic diet. Revista Romana De Medicina De Laborator 26, 65–75. Islam, M.A., Al Mamun, M.A., Faruk, M., Ul Islam, M.T., Rahman, M.M., Alam, M.N., Rahman, A., Reza, H.M., Alam, M.A., 2017. Astaxanthin ameliorates hepatic damage and oxidative stress in carbon tetrachloride-administered rats. Pharmacogn. Res. 9:S84–S91. doi: 10.4103/pr.pr_26_17 Iqbal, U., Jadeja, R.N., Khara, H.S. and Khurana, S. 2021. A Comprehensive Review Evaluating the Impact of Protein Source (Vegetarian vs. Meat). Nutrients 13, doi:10.3390/nu13020370 Jiao, J., Han, S.F., Zhang, W., Xu, J.Y., Tong, X., Yin, X.B., Yuan, L.X. and Qin, L.Q. 2016. Chronic leucine supplementation improves lipid metabolism in C57BL/6J mice fed with a high-fat/cholesterol diet. Food. Nutr. Res. 60, 31304; doi:10.3402/fnr.v60.31304 Jung, E., Kong, S.Y., Ro, Y.S., Ryu, H.H. and Shin, S.D. 2022. Serum Cholesterol Levels and Risk of Cardiovascular Death: a Systematic Review and a Dose-Response Meta-Analysis of Prospective Cohort Studies. Int. J. Environ. Res. Public Health 19, doi:10.3390/ijerph19148272 Khan, M.R. and Ahmed, D. 2009. Protective effects of Digera muricata (L.) Mart. on testis against oxidative stress of carbon tetrachloride in rat. Food. Chem. Toxicol. Int. J. Published. For. Br. Ind. Biol. Res. Assoc. 47, 1393–1399; doi:10.1016/j.fct.2009.03.020 Khan, R.A. 2012. Protective effect of Launaea procumbens (L.) on lungs against CCl4-induced pulmonary damages in rat. BMC. Complementary. Alternative. Med. 12, 133; doi:10.1186/1472-6882-12-133 Khedr, N.F. and Khedr, E.G. 2017. Branched chain amino acids supplementation modulates TGF-β1/Smad signaling pathway and interleukins in CC l4-induced liver fibrosis. Fundam. Clin. Pharmacol. 31, 534–545; doi:10.1111/fcp.12297 Krowka, M.J., Miller, D.P., Barst, R.J., Taichman, D., Dweik, R.A., Badesch, D.B. and Mcgoon, M.D. 2019. Portopulmonary hypertension: a report from the US-based REVEAL Registry. Chest 141, 906–915; doi:10.1378/chest.11-0160 Li, Z., Zhang, R., Mu, H., Zhang, W., Zeng, J., Li, H., Wang, S., Zhao, X., Chen, W., Dong, J. and Yang, R. 2022. Oral Administration of Branched-Chain Amino Acids Attenuates Atherosclerosis by Inhibiting the Inflammatory Response and Regulating the Gut Microbiota in ApoE-Deficient Mice. Nutrients 14, doi:10.3390/nu14235065 Liu, Z., Ren, Z., Zhang, J., Chuang, C.C., Kandaswamy, E., Zhou, T. and Zuo, L. 2018. Role of reactive oxygen species and nutritional antioxidants in human diseases. Front. Physiol. 9, 477; doi:10.3389/fphys.2018.00477 Ma, C., Crippin, J.S., Chapman, W.C., Korenblat, K., Vachharajani, N., Gunter, K.L. and Brunt, E.M. 2013. Parenchymal alterations in cirrhotic livers in patients with hepatopulmonary syndrome or portopulmonary hypertension. Liver Transplantation Off. Publication Am. Assoc. For Study Liver Dis. Int. Liver Transplantation Soc. 19, 741–750; doi:10.1002/lt.23632 Macotela, Y., Emanuelli, B., Bång, A.M., Espinoza, D.O., Boucher, J., Beebe, K., Gall, W. and Kahn, C.R. 2011. Dietary leucine--an environmental modifier of insulin resistance acting on multiple levels of metabolism. PLos One. 6, doi:10.1371/journal.pone.0021187 Mahmoodzadeh, Y., Mazani, M. and Rezagholizadeh, L. 2017. Hepatoprotective effect of methanolic Tanacetum parthenium extract on CCl4-induced liver damage in rats. Toxicol. Rep. 4, 455–462; doi:10.1016/j.toxrep.2017.08.003 Marimuthu, S., Adluri, R.S., Rajagopalan, R. and Menon, V.P. 2013. Protective role of ferulic acid in carbon tetrachloride-induced hyperlipidemia and histological alterations in experimental rats. J. Basic. Clin. Physiol. Pharmacol. 24, 59–66; doi:10.1515/jbcpp-2012-0053 Marrone, G., Serra, A., Miele, L., Biolato, M., Liguori, A., Grieco, A. and Gasbarrini, A. 2023. Branched chain amino acids in hepatic encephalopathy and sarcopenia in liver cirrhosis: evidence and uncertainties. World. J. Gastroenterol. 29, 2905–2915; doi:10.3748/wjg.v29.i19.2905 Mesalam, N.M., Aldhumri, S.A., Gabr, S.A., Ibrahim, M.A., Al-Mokaddem, A.K. and Abdel-Moneim, A.M.E. 2021. Putative abrogation impacts of Ajwa seeds on oxidative damage, liver dysfunction and associated complications in rats exposed to carbon tetrachloride. Mol. Biol. Rep. 48, 5305–5318; doi:10.1007/s11033-021-06544-1 Morgan, M.J. and Liu, Z.G. 2011. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 21, 103–115; doi:10.1038/cr.2010.178 Mustafy, T. and Rahman, M.T.U. 2024. SPSS.In Statistics and data analysis for engineers and scientists. Berlin/Heidelberg, Germany: Springer, pp: 135–80. Niemann, B., Rohrbach, S., Miller, M.R., Newby, D.E., Fuster, V. and Kovacic, J.C. 2017. Oxidative Stress and Cardiovascular Risk: obesity, Diabetes, Smoking, and Pollution: Part 3. J. Am. Coll. Cardiol. 70, 230–251; doi:10.1016/j.jacc.2017.05.043 Nishikimi, M., Appaji, N., Yagi, K., 1972. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. 46, 849–854. doi: 10.1016/s0006-291x(72)80218-3 Noguchi, Y., Zhang, Q.W., Sugimoto, T., Furuhata, Y., Sakai, R., Mori, M., Takahashi, M. and Kimura, T. 2006. Network analysis of plasma and tissue amino acids and the generation of an amino index for potential diagnostic use. Am. J. Clin. Nutr. 83, 513s–519s; doi:10.1093/ajcn/83.2.513S Ohkawa, H., Ohishi, W. and Yagi, K. 1979. Colorimetric method for determination of MDA activity. Biochemistry 95, 90733–90738. Ohta, Y., Kongo, M., Sasaki, E., Nishida, K. and Ishiguro, I. 2000. Therapeutic effect of melatonin on carbon tetrachloride-induced acute liver injury in rats. J. Pineal. Res. 28, 119–126; doi:10.1034/j.1600-079x.2001.280208.x Pääkkö, P., Anttila, S., Sormunen, R., Ala-Kokko, L., Peura, R., Ferrans, V.J. and Ryhänen, L. 1996. Biochemical and morphological characterization of carbon tetrachloride-induced lung fibrosis in rats. Arch. Toxicol. 70, 540–552; doi:10.1007/s002040050311 Ragab, G.M., El-Denshary, E.S., Hassan, A.M., Abdel-Azeim, S.H., Hassan, N.S., Mannaa, F.A. and Abdel-Wahhab, M.A. 2013. Grape (Vitis vinifera) seed extract inhibits the cytotoxicity and oxidative stress in liver of rats treated with carbon tetrachloride. Global J. Pharmacol. 7, 258–269; doi:10.5829/idosi.gjp.2013.7.3.7637 Rahmouni, F., Hamdaoui, L., Badraoui, R. and Rebai, T. 2017. Protective effects of Teucrium polium aqueous extract and ascorbic acid on hematological and some biochemical parameters against carbon tetrachloride (CCl4) induced toxicity in rats. Biomed. &. Pharmacotherapy 91, 43–48; doi:10.1016/j.biopha.2017.04.071 Richmond. and W. 1973. Colorimetrically method used for cholesterol measurement. Clin. Chem. 19, 1350. Ruocco, C., Segala, A., Valerio, A. and Nisoli, E. 2021. Essential amino acid formulations to prevent mitochondrial dysfunction and oxidative stress. Curr. Opinion. Field. Clin. Nutr. Metabolic. Care. 24, 88–95; doi:10.1097/mco.0000000000000704 Shaban, N.Z., Ahmed Zahran, A.M., El-Rashidy, F.H. and Abdo Kodous, A.S. 2017. Protective role of hesperidin against γ-radiation-induced oxidative stress and apoptosis in rat testis. J. Biol. Res. 24, 5; doi:10.1186/s40709-017-0059-x Sharma, S., Zhang, X., Azhar, G., Patyal, P., Verma, A., Kc, G. and Wei, J.Y. 2024. Valine improves mitochondrial function and protects against oxidative stress. Journal 88, 168–176; doi:10.1093/bbb/zbad169 Shi, H., Dong, L., Jiang, J., Zhao, J., Zhao, G., Dang, X., Lu, X. and Jia, M. 2013. Chlorogenic acid reduces liver inflammation and fibrosis through inhibition of toll-like receptor 4 signaling pathway. Toxicology 303, 107–114; doi:10.1016/j.tox.2012.10.025 Takeshita, S., Ichikawa, T., Nakao, K., Miyaaki, H., Shibata, H., Matsuzaki, T., Muraoka, T., Honda, T., Otani, M., Akiyama, M. and et al. 2009. A snack enriched with oral branched-chain amino acids prevents a fall in albumin in patients with liver cirrhosis undergoing chemoembolization for hepatocellular carcinoma. Nutr. Res. (New. York. NY). 29, 89–93; doi:10.1016/j.nutres.2008.12.005 Tamanna N, Mahmood N. 2014. Emerging Roles of Branched-Chain Amino Acid Supplementation in Human Diseases. International scholarly research notices. 2014:235619. Epub 2014/01/01. https://doi.org/10.1155/2014/235619 Taslidere, E., Esrefoglu, M., Elbe, H., Cetin, A. and Ates, B. 2014. Protective effects of melatonin and quercetin on experimental lung injury induced by carbon tetrachloride in rats. Exp. Lung Res. 40, 59–65; doi:10.3109/01902148.2013.866181 Trinder, P. 1969. Determination of blood glucose using 4-amino phenazone as oxygen acceptor. J. Clin. Pathol. 22(2), 246; doi:10.1136/jcp.22.2.246-b Unsal V, Cicek M, Sabancilar İ. 2021. Toxicity of carbon tetrachloride, free radicals and role of antioxidants. Reviews on the environmental health Jun 25;36:279-295. Epub 2020/09/25. https://doi.org/10.1515/reveh-2020-0048 Valerio, A., D'Antona, G. and Nisoli, E. 2011. Branched-chain amino acids, mitochondrial biogenesis, and healthspan: an evolutionary perspective. Aging 3, 464–478; doi:10.18632/aging.100322 Wang, Y., Wang, S., Wang, R., Li, S. and Yuan, Y. 2021. Neferine Exerts Antioxidant and Anti-Inflammatory Effects on Carbon Tetrachloride-Induced Liver Fibrosis by Inhibiting the MAPK and NF-κB/IκBα Pathways. Evidence-based Complementary Alternative Med. ECAM. 2021, doi:10.1155/2021/4136019 Xiao, J., Liong, E.C., Ching, Y.P., Chang, R.C., So, K.F., Fung, M.L. and Tipoe, G.L. 2012. Lycium barbarum polysaccharides protect the liver of mice from carbon tetrachloride-induced oxidative stress and necroinflammation. J. Ethnopharmacology 139, 462–470; doi:10.1016/j.jep.2011.11.033 Xie, J., Liu, J., Chen, T.M., Lan, Q., Zhang, Q.Y., Liu, B., Dai, D., Zhang, W.D., Hu, L.P. and Zhu, R.Z. 2015. Dihydromyricetin alleviates carbon tetrachloride-induced acute liver injury in mice via a JNK-dependent mechanism in mice. World. J. Gastroenterol. 21, 5473–5481; doi:10.3748/wjg.v21.i18.5473/ | ||
| How to Cite this Article |
| Pubmed Style Jad MM, Al-amgad Z, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Mahmoud AM, Elnagar A, Rehan IF, Zigo F, Zigová M, El-aziz AAA. Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Vet. J.. 2025; 15(12): 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 Web Style Jad MM, Al-amgad Z, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Mahmoud AM, Elnagar A, Rehan IF, Zigo F, Zigová M, El-aziz AAA. Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. https://www.openveterinaryjournal.com/?mno=263834 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.46 AMA (American Medical Association) Style Jad MM, Al-amgad Z, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Mahmoud AM, Elnagar A, Rehan IF, Zigo F, Zigová M, El-aziz AAA. Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Vet. J.. 2025; 15(12): 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 Vancouver/ICMJE Style Jad MM, Al-amgad Z, Madkour FA, Al-amgad H, Basher AW, Mohammed HA, Mahmoud AM, Elnagar A, Rehan IF, Zigo F, Zigová M, El-aziz AAA. Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 Harvard Style Jad, M. M., Al-amgad, . Z., Madkour, . F. A., Al-amgad, . H., Basher, . A. W., Mohammed, . H. A., Mahmoud, . A. M., Elnagar, . A., Rehan, . I. F., Zigo, . F., Zigová, . M. & El-aziz, . A. A. A. (2025) Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Vet. J., 15 (12), 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 Turabian Style Jad, Mariam M., Zeinab Al-amgad, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Abeer M. Mahmoud, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Amany Ahmed Abd El-aziz. 2025. Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Veterinary Journal, 15 (12), 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 Chicago Style Jad, Mariam M., Zeinab Al-amgad, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Abeer M. Mahmoud, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Amany Ahmed Abd El-aziz. "Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats." Open Veterinary Journal 15 (2025), 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 MLA (The Modern Language Association) Style Jad, Mariam M., Zeinab Al-amgad, Fatma A. Madkour, Hend Al-amgad, Asmaa W. Basher, Heba A. Mohammed, Abeer M. Mahmoud, Asmaa Elnagar, Ibrahim F. Rehan, František Zigo, Martina Zigová, and Amany Ahmed Abd El-aziz. "Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats." Open Veterinary Journal 15.12 (2025), 6644-6659. Print. doi:10.5455/OVJ.2025.v15.i12.46 APA (American Psychological Association) Style Jad, M. M., Al-amgad, . Z., Madkour, . F. A., Al-amgad, . H., Basher, . A. W., Mohammed, . H. A., Mahmoud, . A. M., Elnagar, . A., Rehan, . I. F., Zigo, . F., Zigová, . M. & El-aziz, . A. A. A. (2025) Therapeutic potential of branched-chain amino acids against carbon tetrachloride-induced cardiopulmonary injuries in Wistar rats. Open Veterinary Journal, 15 (12), 6644-6659. doi:10.5455/OVJ.2025.v15.i12.46 |