| Research Article | ||
Open Vet. J.. 2025; 15(12): 6635-6643 Open Veterinary Journal, (2025), Vol. 15(12): 6635-6643 Research Article Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosteroneJunianto W. A. Pratama1, Pudji Astuti2*, Claude M. Airin2 and Yos A. Prakoso31Doctoral Student, Veterinary Science Doctoral Program, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 2Department of Physiology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia 3Department of Pharmacology and Therapeutics, Faculty of Veterinary Medicine, Universitas Wijaya Kusuma Surabaya, Surabaya, Indonesia *Corresponding Author: Pudji Astuti. Department of Physiology, Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. Email: pastuti2 [at] ugm.ac.id Submitted: 01/06/2025 Revised: 22/10/2025 Accepted: 07/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: Brassica is one of the herbs with potential in the field of medicine, containing active compounds such as glucosinolates. These compounds can be hydrolyzed into indole-3-carbinol (I3C) and 3,3′-diindolylmethane, which have been shown to have a positive impact on health. The stem part of cauliflower is often not utilized; therefore, it was used in this study. Aim: This study aimed to detect the presence of I3C in cauliflower stem powder (BOVB) and evaluate its potential as an aromatase inhibitor in vivo. Methods: This study used cauliflower stems that were freeze-dried and milled into powder. The experimental animals used were Sprague–Dawley rats with five treatments: P1=distilled water (negative control); P2=pure I3C 10.34 mg/day (positive control); P3=171.5 g/day; P4=343 g/day; and P5=686 g/day. Treatment was conducted for 56 days with blood serum collection on day 7 (week 1), day 28 (week 4), day 42 (week 6), and day 56 (week 8). Blood serum was tested using an enzyme-linked immunosorbent assay to measure serum testosterone and aromatase levels. Data were statistically analyzed using two-way analysis of variance and the Duncan test. Results: Rats administered BOVB suspension after 56 days showed a significant decrease in aromatase levels in the blood serum (P4 and P5) compared with those not administered (P1) in the eighth week. In contrast, serum testosterone levels tended to increase but showed no significant change in all groups and all weeks. In the eighth week, rats also showed a significant increase in body weight in the groups given BOVB suspension (P3, P4, and P5) compared to those not given the BOVB suspension (P1). Conclusion: BOVB supplementation significantly decreased serum aromatase levels, increased body weight, and showed a non-significant upward trend in testosterone levels. Further research is needed to elucidate the metabolic pathways, determine optimal safe dosages, and evaluate long-term clinical relevance. Keywords: Aromatase blocker, Cauliflower, Herbal therapy, Indole-3-carbinol, Testosterone. IntroductionBrassica is one of the herbs that has potential in the field of medicine, and it contains active compounds such as glucosinolates (Kjær 1976; Fahey et al., 2001). One variant of this plant is cauliflower [Brassica oleracea var. botrytis (L.)] (BOVB). A study showed that the stem part of green broccoli has glucosinolate content equivalent to that of the flower part and is often not used (Casajús et al., 2023). Cauliflower is a variety of Brassica and belongs to the same species as green broccoli. Therefore, cauliflower stems are expected to have similar properties to broccoli stems and contain high concentrations of important compounds such as glucosinolates. Indol-3-ylmethyl glucosinolate (glucobrassicin), a major glucosinolate, is a precursor to indole-3-carbinol (I3C), a compound with reported health benefits (Bennett et al., 2004; Cartea et al., 2008). The administration of I3C may affect estrogen metabolism by inhibiting the aromatization of testosterone in vitro (Sundar et al., 2006; Santi et al., 2015). Indole-3-carbinol inhibits estrogen-induced cell proliferation through transcriptional downregulation and ubiquitination/degradation of estrogen receptor alpha proteasomes (Marconett et al., 2010). A study also showed that the I3C content in red cabbage extract can increase testosterone levels in a rat model of hyperthyroidism, which suppresses ERa expression so that testosterone can perform its function. Similar results were also confirmed in a cadmium-induced infertility model, where red cabbage supplementation increased testosterone and reproductive function. (Bakr and El-Yamany, 2020). The decrease in testosterone is associated with aromatization, i.e., the synthesis of estradiol from testosterone by aromatase or 17β-hydroxysteroid dehydrogenase. Several studies have demonstrated the aromatization mechanism of testosterone. A study reported a decrease in testosterone and up to 3 times increase in estradiol levels in the testes of rats during aromatase hyperactivity (Boulanger et al., 2015). In the vertebrate brain, aromatase is expressed in the neurons of the hypothalamus and amygdala (Brooks et al., 2020). Studies have shown that the weak expression of aromatase CYP19 results from the inhibition of aromatase using natural substances, resulting in low estrogen and high testosterone levels in rats (Astuti et al., 2019). In addition, natural aromatase inhibitors (AI) improve the quality of secondary changes, such as voice, by increasing testosterone in male canaries and male chickens (Astuti et al., 2020; Yuneldi et al., 2021). Furthermore, natural AI can increase testosterone levels, reducing the expression of aromatase CYP19 receptors in the brain, syrinx, and testes (Astuti et al., 2022). Based on this background, the objectives of this study were to detect the I3C content in BOVB stems and evaluate the potential of BOVB stems as a raw material for AI, as well as to utilize the waste from BOVB stems to optimize natural resources and provide sustainable treatment and nutrition alternatives. Materials and MethodsTime and location of researchThis study was conducted from March to June 2025 at the Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia. Determination of the herbal speciesThe herbs used were stems from harvested cauliflower. Herbal species were determined at the Center for Research and Development of Medicinal Plants and Traditional Medicines (B2P2TO-OT), Tawangmangu, Central Java, with registration number: KM.04.02/H.IX/489/2023. The results of the determination are Brassica oleracea var. botrytis (L.) Lyophilization preparationFresh BOVB stems were weighed and finely cut. The cuttings were placed in a freeze-dryer for 2-3 days. The cut yield was removed from the freeze-dryer and pulverized in a chopper. The remaining unrefined yield was pulverized with a mortar and sieved. The dry weight of the refined yield was weighed and then stored in a dry place at room temperature and not exposed to direct sunlight (Biegańska-Marecik et al., 2017). Indole-3-carbinol detection using thin-layer chromatographyThe levels of indole-3-carbinol were determined using Thin Layer Chromatography (TLC). A total of 100 mg of freeze-dried BOVB sample was weighed and placed in a 2 ml microtube. Absolute ethanol (1 ml) was added to the sample. The suspension was vortexed and sonicated for 60 minutes and then macerated for 24 hours. The suspension was vortexed and centrifuged to obtain samples (1 µl) were spotted on a silica gel plate and equalized with the comparison standard using pure I3C. Samples and comparison standards were placed in a chamber containing saturated toluene-chloroform mobile phase in a ratio of 2:3. The samples were eluted to the limit, removed, and dried by aeration. The samples were densitized at a wavelength of 278 nm using a densitometer. The chromatography details are as follows: volume of 1 µl; stationary phase on silica gel 60 F254; mobile phase on toluene-chloroform (2 ml:3 ml, v/v); creepage distance 8.8 cm; and wavelength 278 nm. The Rf was calculated using the following equation:
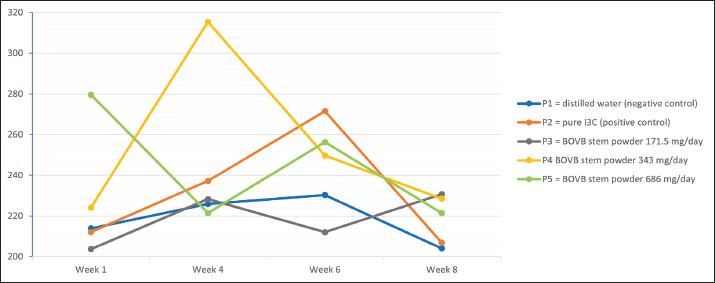
The Rf value obtained for I3C was 0.37. The I3C content of the sample was quantified by comparing the densitometric response of the sample spots with that of the reference standard, and the result was expressed as mg/kg dry weight of freeze-dried BOVB. The TLC analysis of I3C levels in BOVB stems showed that a sample weight of 0.1044 g with a spotting volume of 1.0 µl resulted in an area measurement of 10,222.52 AU. This corresponds to a detection result of 506.65 ng of I3C. Based on these calculations, the I3C content in the stem of freeze-dried BOVB is 4,853 mg/kg of dry weight. Detection of indole-3-carbinol using Gas Chromatography–Mass Spectrometry (GC-MS)Identification of the I3C compound was performed using GC-MS (Thermo ScientificTM, ISQD1702517_1). The sample was dissolved in a 1.5 ml microtube with EtOH as the solvent, vortexed until homogeneous, and, if necessary, centrifuged at 9,500 rpm for 5 minutes. The supernatant was placed in a GC vial. The sample solution was ready to be injected into the GC-MS machine. The capillary column used was HP-5MS UI (30 m × 0.25 mm; film thickness, 0.25 µm), with the following equipment conditioning: carrier gas: UHP helium (He); injector temperature: 230°C; split flow: 50 ml/minute; split ratio: 50:1; front inlet flow: 1.00 ml/minute; MS transfer line temperature: 250°C; ion source temperature: 200°C; mass list range (amu): 40–500; purge flow : 3 ml/minute; gas saver flow: 5 ml/minute; gas saver time: 5 minute; column temperature: retention time: 2 minute, rate: 0°C/minute, target value: 60°C, hold time: 2 minutes; retention time: 32 minutes, rate: 10°C/minutes, target value: 280°C, and hold time: 8 minutes. Experimental animals and treatment dose calculationThe experimental animals used in this study were 25 Sprague–Dawley rats, which were randomly divided into five treatment groups with five rats in each treatment group. Each rat was then marked for identification. During the measurement of results, a single-blind method was applied such that the researchers conducting the measurements did not know the group allocation to minimize bias. Based on the results of the TLC test on the sample, the dose to be administered was calculated based on a review article (Nair and Jacob, 2016). The dose given to rats is 343 mg/day, so that the treatment is given to 5 treatment groups as follows: P1=distilled water (negative control); P2=pure I3C (positive control); P3=BOVB stem powder 171.5 mg/day; P4=BOVB stem powder 343 mg/day; and P5=BOVB stem powder 686 mg/day. Administration of Simplicia from BOVB stemsThe BOVB stem powder was dissolved in distilled water to a total volume of 3 ml. The dissolution process was conducted at room temperature. Treatment administration is accomplished through oral ingestion. The BOVB stem powder solution is administered through a feeding tube. The maximum volume of solution administered to rats is ± 3 ml/250 g of the rat’s body weight in one administration. The solution volume was adjusted to the dose to be given per day for 56 days. Serum samplingBlood samples were collected from each rat that had been treated on day 7 (week 1), day 28 (week 4), day 42 (week 6), and day 56 (week 8) using a microcapillary (Onemed, Indonesia). Before blood collection, the animals were anesthetized using ketamine (50 mg/kg BW, IP, Ket-A-100, Agrovet Market, Lima, Peru) and xylazine 2% (4 mg/kg BW, IP, Xyla, Interchemie, Venray, Holland). After the animals were anesthetized, blood samples were collected via the plexus retro-orbital. Furthermore, blood samples were separated by centrifugation at 1,372 × g for 10 minutes, and serum was obtained. Blood serum was placed in an Eppendorf tube and stored at 4°C until the next test. Serum analysis was performed after the last blood collection (Wijayanti et al., 2024). Examination of aromatase levels using enzyme-linked immunosorbent assay (ELISA)Serum samples were homogenized using a vortex before being subjected to ELISA. Standards and reagents were prepared according to the ELISA Kit guidelines (ABclonal Inc., Catalog No. RK04428, Woburn, United States). Each sample and standard was assayed in duplicate to ensure the precision, reliability, and reproducibility of the measurements. Examination of testosterone levels using ELISASerum samples were homogenized using a vortex before ELISA measurement. Standards and reagents were prepared according to the ELISA Kit guidelines (Calbiotech Inc., Catalog No. TE373S, El Cajon, United States). Data analysisData analysis in this study was performed using the SPSS software. Data on body weight and aromatase concentration were analyzed using a two-way analysis of variance. The difference is significant if p < 0.05; if the effect is significant, the Duncan test will be continued. Ethical approvalThis study was conducted in strict compliance with ethical guidelines. This study was approved by the Research Ethics Commission for Experimental Animals, Faculty of Dentistry, Universitas Airlangga, Surabaya, with registration number: 0018/HRECC.FODM/I/2024. Date: March 2, 2023. ResultsIndole-3-carbinol detectionThe TLC method was used to detect I3C content, yielding a result of 4,853 mg/kg for BOVB stem. GC-MS analysis of BOVB stems detected 4-Piperidineacetic acid, 1-acetyl-5-ethyl-2-[3-(2-hydroxyethyl)-1H-indol-2-yl]-a-methyl-, methyl ester with retention time 28.461 on Hit#3, consistent with a derivative related to I3C. Aromatase levelsStatistical analysis showed a significant difference in serum aromatase levels between the groups at week 8 (p=0.01, p < 0.05). Groups P4 (228.47) and P5 (221.41) had significantly lower levels of aromatase than group P1 (204.07), whereas groups P1 and P3 showed a decrease in aromatase compared to P2, although the difference was not statistically significant (p > 0.05) (Fig. 2).
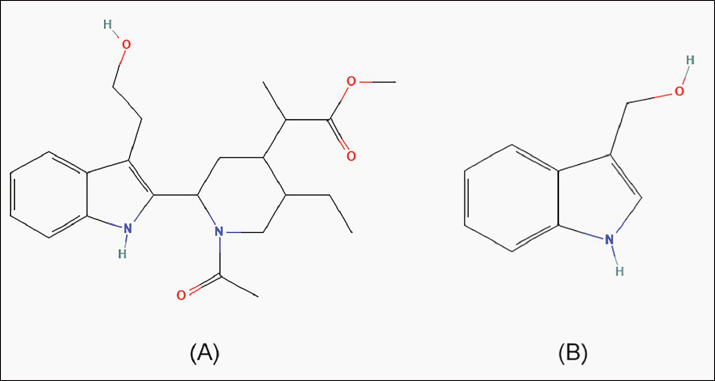
Fig. 1. Chemical Structures. A) 4-Piperidineacetic acid, 1-acetyl-5-ethyl-2-[3-(2-hydroxyethyl)-1H-indol-2-yl]-a-methyl-, methyl ester (National Center for Biotechnology Information, 2025a); B) indole-3-carbinol (National Center for Biotechnology Information, 2025b).
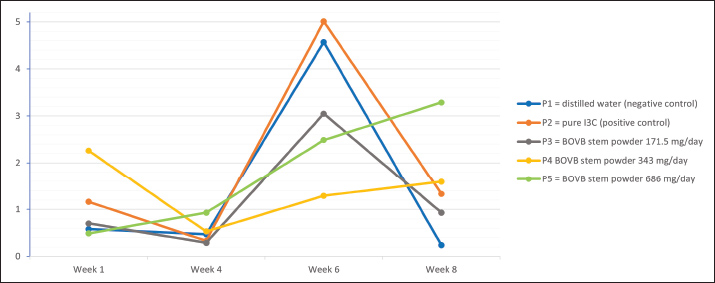
Fig. 2. Serum aromatase of rats after treatment. Testosterone levelThis contrasts with the levels of serum testosterone in rats after treatment. Serum testosterone levels increased across all treatment groups. However, these changes did not reach statistical significance (p > 0.05) 8 weeks after treatment (Fig. 3).
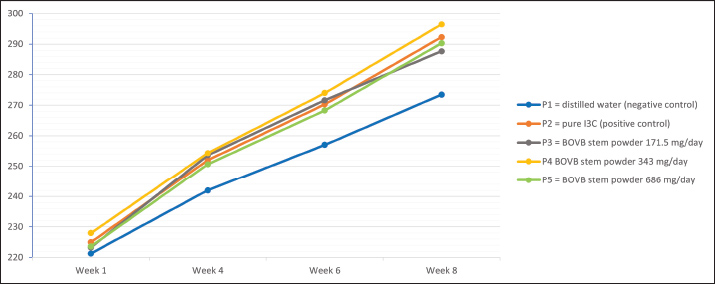
Fig. 3. Serum testosterone levels in rats after treatment. Body weightThe results of the statistical analysis show that treatment has an effect on the body weight of rats (p < 0.05). There was a pattern of increasing the mean body weight of rats with increasing therapy time. Group P1 had the lowest body weight compared with the other groups (p < 0.05). The difference between P2 and P3 and P4 and P5 was not statistically significant (p > 0.05). Group P4 showed the highest body weight, which was significantly different from groups P1 and P5 (p < 0.05) at week 8 (Fig. 4).
Fig. 4. Body weight of mice treated. DiscussionThe TLC test results showed I3C in the BOVB stem powder. Indol-3-carbinol is an indole compound derived from the hydrolysis of glucosinolates by the enzyme myrosinase found in cruciferous vegetables (Singh et al., 2021). GC-MS test results did not find I3C but did find the compound 4-Piperidineacetic acid, 1-acetyl-5-ethyl-2-[3-(2-hydroxyethyl)-1H-indol-2-yl]-a-methyl-, methyl ester. The non-identification of I3C on GC-MS can be due to the lack of stability of the compound, so that other compounds that have almost the same structure, such as 4-Piperidineacetic acid, 1-acetyl-5-ethyl-2-[3-(2-hydroxyethyl)-1H-indol-2-yl]-a-methyl-, methyl ester, are detected. The compound has an ethyl hydroxy group at position 2 of the indole ring, whereas I3C has a methyl hydroxy group at position 3. The ethyl hydroxy group in I3C and the methyl hydroxy group in I3C are similar but are located at different positions on the indole ring. Indole-3-carbinol and 4-Piperidineacetic acid, 1-acetyl-5-ethyl-2-[3-(2-hydroxyethyl)-1H-indol-2-yl]-a-methyl-, methyl ester have almost the same structure, which contains an indole group, which is a benzopyrrole structure in which the benzene and pyrrole rings are fused through positions 2 and 3 of the pyrrole core (Fig. 1) (Srivastava et al., 2011). Indole compounds have a benzene ring structure fused to a pyrrole ring. Both have similarities in the ability to participate in hydrogen bonding and their important role in biological activity (Smith and Wermuth, 2011; Delgado et al., 2015; Van Quan et al., 2019; Amarakoon et al., 2023a; Centofanti et al., 2023). After 56 days, rats given a suspension of BOVB showed a significant decrease in aromatase levels in the blood serum (P4 and P5) compared with those not given BOVB suspension (P1) (Fig. 2). This occurred due to the I3C content of the BOVB stems. Indole-3-carbinol is a compound found in cruciferous vegetables such as cauliflower and has been studied for its various biological activities, including its potential as an AI (Choi et al., 2012; Singh et al., 2021). Aromatase converts testosterone to estradiol. Inhibiting aromatase reduces this conversion, resulting in higher testosterone levels and lower estradiol levels (Korani, 2023). When metabolized in the stomach, I3C can form 3,3′-diindolylmethane (DIM), which is its by-product (Amarakoon et al., 2023b; Tian et al., 2024). Studies have shown the potential of I3C as an AI that can affect the expression of the CYP19 gene encoding aromatase (Amalia et al., 2025). I3C and its condensation product, DIM, successfully reduced aromatase expression in estrogen-dependent MCF7 breast cancer cells. This suggests that I3C may affect aromatase at the transcriptional level as an AI, thereby decreasing estrogen production (Licznerska et al., 2013). Furthermore, the testosterone levels (Fig. 3) in the blood serum did not show statistically significant changes after treatment despite an increase over time. This insignificant increase may be due to the conversion of testosterone to dihydrotestosterone (DHT). Testosterone is converted to DHT catalyzed by the enzyme 5α-reductase, which exists in three isoenzyme forms encoded by different genes (SRD5A1, SRD5A2, and SRD5A3) (Li et al., 2011; Jifei et al., 2023). DHT synthesis involves the conversion of dehydroepiandrosterone and androstenedione to testosterone and then to DHT (Gauthier-Landry et al., 2015; Lee and Kim, 2022). This conversion is important because DHT has a 2-5 times higher binding affinity to androgen receptors and a 10 times higher potency in inducing androgen receptor signaling than testosterone (Azzouni and Mohler, 2012). Therefore, androgenic effects can persist even if serum testosterone levels remain stable (Li et al., 2018; Bhasin and Jasuja, 2019). The rats that received BOVB suspension showed a significant increase in body weight (P3, P4, and P5) compared with those who did not receive BOVB suspension (P1) (Fig. 4). This indicates that I3C content as an AI affects weight gain. Aromatase inhibitors indirectly increase body weight by reducing estrogen levels and increasing testosterone levels, although the increase in testosterone levels was not significant in this study. Testosterone plays an important role in body weight and muscle building. Higher testosterone levels are associated with increased muscle mass and strength (Linden-Hirschberg, 2019; Hirschberg, 2020). Supraphysiological testosterone levels can lead to significant muscle hypertrophy and fat mass reduction (Mouser et al., 2016). Testosterone promotes muscle fiber hypertrophy by increasing protein synthesis through satellite cell activation and increased myonuclear accretion, which supports further protein synthesis when existing myonuclei are insufficient. Furthermore, testosterone promotes satellite cell proliferation and differentiation into myotubes, which are essential for muscle repair and growth (Stárka, 2006; Kadi, 2008). Testosterone mediates anabolic effects through androgen receptors in muscle cells. Binding to these receptors triggers a series of anabolic signaling pathways, including the PI3K/Akt/mTOR pathway, which is critical for muscle protein synthesis and hypertrophy (Wu et al., 2010; Basualto-Alarcón et al., 2013). Testosterone interacts with other anabolic hormones, such as growth hormone and insulin-like growth factor, which further modulate muscle protein turnover and hypertrophy. These interactions are critical for the overall anabolic environment in muscle (Kraemer et al., 2020; Gharahdaghi et al., 2021). Long-term testosterone replacement therapy in older adult men with low testosterone levels increased muscle mass and improved muscle performance. This effect was partly due to a reduction in protein breakdown via the ubiquitin–proteasome pathway (Kruse et al., 2020). ConclusionBOVB contains measurable levels of I3C. Oral administration to rats for 56 days significantly reduced serum aromatase levels, suggesting potential aromatase inhibitory activity. Testosterone levels showed a non-significant upward trend, and body weight increased significantly in the treated groups. These findings warrant further research to determine the optimal safe dose, clarify metabolic effects, and evaluate long-term safety before clinical applications are considered. AcknowledgmentsThe authors are grateful to the Faculty of Veterinary Medicine, Universitas Gadjah Mada, for providing the necessary facilities for the study. Conflict of interestThe authors declare no conflict of interest. FundingThis article was funded by the Directorate of Research (Direktorat Penelitian), Universitas Gadjah Mada, through the 2025 Final Project Recognition Program (Program Rekognisi Tugas Akhir Tahun 2025) with the number 4193/UN1.P1/Dit-Lit/PT.01.03/2025. Authors contributionsConceptualization and design: PA; acquisition of data: JWAP; formal analysis and interpretation of data: CMA and YAP; writing, original draft preparation: PA and JWAP; writing, review, and editing: PA, CMA, and YAP. All authors have read and approved the published version of the manuscript. Data availabilityAll data supporting the results of this study are available within the manuscript, and no additional sources are required. ReferencesAmalia, R., Ohama, T., Parhar, I.S., Airin, C.M., Sato, H., Budiyanto, A. and Astuti, P. 2025. Potential of Indole-3-Carbinol compounds from broccoli (Brassica oleracea var. italica) as natural aromatase blockers: &In silico prediction and in vivo studies. Open. Vet. J. 15(4), 18; doi:10.5455/OVJ.2025.v15.i4.18 Amarakoon, D., Lee, W., -J., Tamia, G., Lee, S. and -H. 2023b. The Annual Review of Food Science and Technology. Annu. Rev. Food. Sci. Technol. 14, 347–366; doi:10.1146/annurev-food-060721 Amarakoon, D., Lee, W.J., Tamia, G. and Lee, S.H. 2023a. Indole-3-Carbinol: occurrence, Health-Beneficial Properties, and Cellular/Molecular Mechanisms. Annu. Rev. Food Sci. Technol. 14, 347–366; doi:10.1146/annurev-food-060721-025531 Astuti, P., Airin, C.M., Nurrurozi, A., Aidi, R., Hana, A., Hadi, S. and Harimurti, H. 2020. Potential Natural Aromatase Blockers on Enhance the Frequency and Sound Quality of Male Canaries. In E3S Web of Conferences, EDP Sciences, Banda Aceh. Astuti, P., Airin, C.M., Sarmin, S., Nururrozi, A. and Harimurti, S. 2019. Effect of shell as natural testosterone boosters in Sprague Dawley rats. Vet. World 12, 1677–1681; doi:10.14202/vetworld.2019.1677-1681 Astuti, P., Putra, M.N.P., Shiddiq, M.F.A., Yuneldi, R.F., Airin, C.M. and Sarmin. 2022. The Potency of Anadara nodifera Shell as Natural Testosterone Booster for Male Canary (Serinus canaria). Hayati 29, 107–113; doi:10.4308/HJB.29.1.107-113 Azzouni, F. and Mohler, J. 2012. Role of 5α-reductase inhibitors in benign prostatic diseases. Prostate. Cancer. Prostatic. Dis. 15, 222–230; doi:10.1038/pcan.2012.1 Basualto-Alarcón, C., Jorquera, G., Altamirano, F., Jaimovich, E. and Estrada, M. 2013. Testosterone Signals through mTOR and Androgen Receptor to Induce Muscle Hypertrophy. Med. Sci. Sports Exerc. 45, 1712–1720; doi:10.1249/MSS.0b013e31828cf5f3 Bennett, R.N., Mellon, F.A. and Kroon, P.A. 2004. Screening Crucifer Seeds as Sources of Specific Intact Glucosinolates Using Ion-Pair High-Performance Liquid Chromatography Negative Ion Electrospray Mass Spectrometry. J. Agric. Food Chem. 52, 428–438; doi:10.1021/jf030530p Bhasin, S. and Jasuja, R. 2019. Reproductive and Nonreproductive Actions of Testosterone.In Encyclopedia of Endocrine Diseases. Huhtaniemi, I. and Martini, L. (eds). Oxford: Elsevier, pp: 721–34. https://doi.org/ 10.1016/B978-0-12-801238-3.65412-0 Biegańska-Marecik, R., Radziejewska-Kubzdela, E. and Marecik, R. 2017. Characterization of phenolics, glucosinolates and antioxidant activity of beverages based on apple juice with addition of frozen and freeze-dried curly kale leaves (Brassica oleracea L. var. acephala L.). Food. Chem. 230, 271–280; doi:10.1016/j.foodchem.2017.03.047 Boulanger, G., Cibois, M., Viet, J., Fostier, A., Deschamps, S., Pastezeur, S., Massart, C., Gschloessl, B., Gautier-Courteille, C. and Paillard, L. 2015. Hypogonadism Associated with Cyp19a1 (Aromatase) Posttranscriptional Upregulation in Celf1 Knockout Mice. Mol. Cell. Biol. 35, 3244–3253; doi:10.1128/mcb.00074-15 Brooks, D.C., Coon V, J.S., Ercan, C.M., Xu, X., Dong, H., Levine, J.E., Bulun, S.E. and Zhao, H. 2020. Brain Aromatase and the Regulation of Sexual Activity in Male Mice. Endocrinology 161, 1–15. doi: 10.1210/endocr/bqaa137 Cartea, M.E., Velasco, P., Obregón, S., Padilla, G. and De Haro, A. 2008. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern Spain. Phytochemistry 69, 403–410; doi:10.1016/j.phytochem.2007.08.014 Casajús, V., Howe, K., Fish, T., Civello, P., Thannhauser, T., Li, L., Gómez Lobato, M. and Martínez, G. 2023. Evidence of glucosinolates translocation from inflorescences to stems during postharvest storage of broccoli. Plant Physiol. Biochem. 195, 322–329; doi:10.1016/j.plaphy.2023.01.012 Centofanti, F., Buono, A., Verboni, M., Tomino, C., Lucarini, S., Duranti, A., Pandolfi, P.P. and Novelli, G. 2023. Synthetic Methodologies and Therapeutic Potential of Indole-3-Carbinol (I3C) and Its Derivatives. Pharmaceuticals 16, 240; doi:10.3390/ph16020240 Choi, Y., Kim, Y., Park, S., Lee, K.W. and Park, T. 2012. Indole-3-carbinol prevents diet-induced obesity through modulation of multiple genes related to adipogenesis, thermogenesis or inflammation in the visceral adipose tissue of mice. J. Nutr. Biochem. 23, 1732–1739; doi:10.1016/j.jnutbio.2011.12.005 Delgado, G.E., Mora, A.J., Ávila, E.E., González, T., Briceño, A. and Chacón, C. 2015. N-Carbamoyl-Piperidine-4-Carboxylic Acid: an X-ray and Density Functional Tight-Binding Studies. Mol. Crystals. Liquid. Crystals. 623, 358–364; doi:10.1080/15421406.2015.1036498 Fahey, J.W., Zalcmann, A.T. and Talalay, P. 2001. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 56(00), 5–51. Gauthier-Landry, L., Bélanger, A. and Barbier, O. 2015. Multiple roles for udp-glucuronosyltransferase (UGT)2B15 and UGT2B17 enzymes in androgen metabolism and prostate cancer evolution. J. Steroid Biochem. Mol. Biol. 145, 187–192; doi:10.1016/j.jsbmb.2014.05.009 Gharahdaghi, N., Phillips, B.E., Szewczyk, N.J., Smith, K., Wilkinson, D.J. and Atherton, P.J. 2021. Links Between Testosterone, Oestrogen, and the Growth Hormone/Insulin-Like Growth Factor Axis and Resistance Exercise Muscle Adaptations. Front. Physiol. 11, 1–12. doi:10.3389/fphys.2020.621226 H. Bakr, E.S. and El-Yamany, M.A. 2020. Chemical and nutraceutical studies on infertility of albino rats induced by cadmium chloride. Pak. J. Biol. Sci. 23, 1245–1252; doi:10.3923/pjbs.2020.1245.1252 Hirschberg, A.L. 2020. Female hyperandrogenism and elite sport. Endocr. Connect. 9, R81–R92; doi:10.1530/EC-19-0537 Jifei, P., Xiaona, W., Haijiao, G., Suzhen, Y. and Jianying, C. 2023. The establishment and application of an in vitro evaluation system for 5α-reductase inhibitors. China Surfactant Detergent Cosmetics 53, 1280–1284; doi:10.3969/j.issn.2097-2806.2023.11.006 Kadi, F. 2008. Cellular and molecular mechanisms responsible for the action of testosterone on human skeletal muscle. A basis for illegal performance enhancement. Br. J. Pharmacol. 154, 522–528; doi:10.1038/bjp.2008.118 Kjær, A. 1976. Glucosinolates in the cruciferae. In: The Biology and Chemistry of the Cruciferae. Academic Press, London. Korani, M. 2023. Aromatase inhibitors in male: a literature review. Medicina Clínica Práctica 6, 100356; doi:10.1016/j.mcpsp.2022.100356 Kraemer, W.J., Ratamess, N.A., Hymer, W.C., Nindl, B.C. and Fragala, M.S. 2020. Growth Hormone(s), Testosterone, Insulin-Like Growth Factors, and Cortisol: roles and Integration for Cellular Development and Growth With Exercise. Front. Endocrinol. (Lausanne) 11:33; doi: 10.3389/fendo.2020.00033 Kruse, R., Petersson, S.J., Christensen, L.L., Kristensen, J.M., Sabaratnam, R., Ørtenblad, N., Andersen, M. and Højlund, K. 2020. Effect of long-term testosterone therapy on molecular regulators of skeletal muscle mass and fibre-type distribution in aging men with subnormal testosterone. Metabolism 112, 154347; doi:10.1016/j.metabol.2020.154347 Lee, H.G. and Kim, C.J. 2022. Classic and backdoor pathways of androgen biosynthesis in human sexual development. Ann. Pediatr. Endocrinol. Metab. 27, 83–89; doi:10.6065/apem.2244124.062 Li, J., Ding, Z., Wang, Z., Lu, J.F., Maity, S.N., Navone, N.M., Logothetis, C.J., Mills, G.B. and Kim, J. 2011. Androgen Regulation of 5α-Reductase Isoenzymes in Prostate Cancer: implications for Prostate Cancer Prevention. PLos One 6, e28840; doi:10.1371/journal.pone.0028840 Li, L., Zirkin, B.R. and Papadopoulos, V. 2018. Leydig Cell Androgen Synthesis.In Encyclopedia of Reproduction. Skinner, M.K. and Jégou, B. (eds). Oxford: Elsevier, pp: 215–21. https://doi.org/10.1016/B978-0-12-801238-3.64583-X Licznerska, B.E., Szaefer, H., Murias, M., Bartoszek, A. and Baer-Dubowska, W. 2013. Modulation of CYP19 expression by cabbage juices and their active components: indole-3-carbinol and 3,3′-diindolylmethene in human breast epithelial cell lines. Eur. J. Nutr. 52, 1483–1492; doi:10.1007/s00394-012-0455-9 Linden-Hirschberg, A. 2019. The Role of Androgens for Body Composition and Physical Performance in Women.In Menstrual Cycle Related Disorders. Berga, S.L., Genazzani, A.R., Naftolin, F. and Petraglia, F Springer Cham, Switzerland, pp: 43–54. https://doi.org/ 10.1007/978-3-030-14358-9_4 Marconett, C.N., Sundar, S.N., Poindexter, K.M., Stueve, T.R., Bjeldanes, L.F. and Firestone, G.L. 2010. Indole-3-Carbinol Triggers Aryl Hydrocarbon Receptor-dependent Estrogen Receptor (ER)α Protein Degradation in Breast Cancer Cells Disrupting an ERα-GATA3 Transcriptional Cross-Regulatory Loop. Mol. Biol. Cell 21, 1166–1177; doi:10.1091/mbc.e09-08-0689 Mouser, J.G., Loprinzi, P.D. and Loenneke, J.P. 2016. The association between physiologic testosterone levels, lean mass, and fat mass in a nationally representative sample of men in the United States. Steroids 115, 62–66; doi:10.1016/j.steroids.2016.08.009 Nair, A. and Jacob, S. 2016. A simple practice guide for dose conversion between animals and human. J. Basic Clin. Pharm. 7, 27; doi:10.4103/0976-0105.177703 National Center for Biotechnology Information, 2025a. PubChem Substance Record for SID 319233018, Source: ToxPlanet [WWW Document]. URL https://pubchem.ncbi.nlm.nih.gov/substance/319233018 (accessed 2.26.25). National Center for Biotechnology Information, 2025b. PubChem Compound Summary for CID 3712, Indole-3-Carbinol [WWW Document]. URL https://pubchem.ncbi.nlm.nih.gov/compound/Indole-3-Carbinol (accessed 2.26.25). Santi, M., Carloni, E., Galluzzi, L., Diotallevi, A., Lucarini, S., Magnani, M. and Brandi, G. 2015. Inhibition of Testosterone Aromatization by the Indole-3-carbinol Derivative CTet in CYP19A1-overexpressing MCF-7 Breast Cancer Cells. Anticancer Agents Med. Chem. 15, 896–904; doi:10.2174/1871520615666150121123053 Singh, A.A., Patil, M.P., Kang, M.J., Niyonizigiye, I. and Kim, G.D. 2021. Biomedical application of Indole-3-carbinol: a mini-review. Phytochem. Lett. 41, 49–54; doi:10.1016/j.phytol.2020.09.024 Smith, G. and Wermuth, U.D. 2011. Hydrogen-Bonding in the Structures of the Hydrated Proton-Transfer Compounds of Isonipecotamide with the Isomeric Indole-2- and Indole-3-Carboxylic Acids. J. Chem. Crystallogr. 41, 1850–1854; doi:10.1007/s10870-011-0186-4 Srivastava, A., Pandeya, S.N. and Khan, A.A. 2011. Anticonvulsant and convulsant effects of indole derivatives against chemical models of epilepsy. Int. J. Pharmtech. Res. 3, 2029–2037. Stárka, L. 2006. Testosterone treatment of sarcopenia. Vnitr. Lek. 52, 909–911. Sundar, S.N., Kerekatte, V., Equinozio, C.N., Doan, V.B., Bjeldanes, L.F. and Firestone, G.L. 2006. Indole-3-carbinol selectively uncouples expression and activity of estrogen receptor subtypes in human breast cancer cells. Mol. Endocrinol. 20, 3070–3082; doi:10.1210/me.2005-0263 Tian, C., Deng, S., Yang, M., Bai, B., Pan, Y., Xie, G., Zhao, D. and Wei, L. 2024. Indole-3-carbinol and its main derivative 3,3′-diindolylmethane: regulatory roles and therapeutic potential in liver diseases. Biomed. &. Pharmacotherapy 180, 117525; doi:10.1016/j.biopha.2024.117525 Van Quan, V., Phu, H.T., Thao, P.T.T. and Nam, P.C. 2019. Substituent effects on antioxidant activity of monosubstituted indole-3-carbinols: a DFT study. Vietnam. J. Chem. 57, 728–734; doi:10.1002/vjch.2019000110 Wijayanti, A., Prakoso, Y. and Isla, K. 2024. Effects of fermented Crescentia cujete L. on the profile of haematology, clinical chemistry and circulatory CD4+/CD8+ in Sprague Dawley rats. Open. Vet. J. 14(9), 2475; doi:10.5455/OVJ.2024.v14.i9.36 Wu, Y., Bauman, W.A., Blitzer, R.D. and Cardozo, C. 2010. Testosterone-induced hypertrophy of L6 myoblasts is dependent upon Erk and mTOR. Biochem. Biophysical. Res. Commun. 400, 679–683; doi:10.1016/j.bbrc.2010.08.127 Yuneldi, R.F., Astuti, P., Saragih, H.T.S. and Airin, C.M. 2021. Anadara granosa shell powder improves the metabolism, testosterone level, and sound frequency of Pelung chickens. Vet. World. 14, 1564–1571; doi:10.14202/vetworld.2021.1564-1571 | ||
| How to Cite this Article |
| Pubmed Style Pratama JWA, Astuti P, Airin CM, Prakoso YA. Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Vet. J.. 2025; 15(12): 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 Web Style Pratama JWA, Astuti P, Airin CM, Prakoso YA. Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. https://www.openveterinaryjournal.com/?mno=262033 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.45 AMA (American Medical Association) Style Pratama JWA, Astuti P, Airin CM, Prakoso YA. Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Vet. J.. 2025; 15(12): 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 Vancouver/ICMJE Style Pratama JWA, Astuti P, Airin CM, Prakoso YA. Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 Harvard Style Pratama, J. W. A., Astuti, . P., Airin, . C. M. & Prakoso, . Y. A. (2025) Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Vet. J., 15 (12), 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 Turabian Style Pratama, Junianto W. A., Pudji Astuti, Claude M. Airin, and Yos A. Prakoso. 2025. Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Veterinary Journal, 15 (12), 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 Chicago Style Pratama, Junianto W. A., Pudji Astuti, Claude M. Airin, and Yos A. Prakoso. "Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone." Open Veterinary Journal 15 (2025), 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 MLA (The Modern Language Association) Style Pratama, Junianto W. A., Pudji Astuti, Claude M. Airin, and Yos A. Prakoso. "Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone." Open Veterinary Journal 15.12 (2025), 6635-6643. Print. doi:10.5455/OVJ.2025.v15.i12.45 APA (American Psychological Association) Style Pratama, J. W. A., Astuti, . P., Airin, . C. M. & Prakoso, . Y. A. (2025) Role of Brassica oleracea var. botrytis (L.) as an aromatase inhibitor in Sprague–Dawley rats: Analysis of serum aromatase and testosterone. Open Veterinary Journal, 15 (12), 6635-6643. doi:10.5455/OVJ.2025.v15.i12.45 |