| Review Article | ||
Open Vet. J.. 2026; 16(1): 1-14 Open Veterinary Journal, (2026), Vol. 16(1): 1-14 Review Article Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluationSaifur Rehman1, Shakeeb Ullah1,2, Mustofa Helmi Effendi3*, Budiastuti Budiastuti4, Iwan Sahrial Hamid5, Atta Ur Rahman2, Ali Zaman2, Muhammad Inam Ullah Malik2, Saqib Ali Rustam2, Sana Ullah2 and Zulqarnain Saleem21Faculty of Veterinary Sciences, University of Veterinary and Animal Sciences Swat, KPK, Pakistan 2Faculty of Veterinary and Animal Sciences, Gomal University Dera Ismail Khan, Dera Ismail Khan, Indonesia 3Division of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Study Program of Pharmacy Science, Faculty of Health Science, Universitas Muhammadiyah Surabaya, Indonesia 5Department of Basic Veterinary Science, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia *Corresponding Author: Mustofa Helmi Effendi. Division of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: jrcastro [at] ull.edu.es; mhelmieffendi [at] gmail.com Submitted: 15/04/2025 Revised: 11/11/2025 Accepted: 03/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
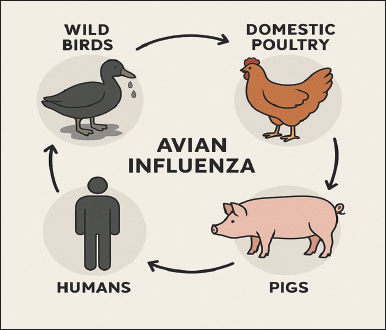
AbstractThe H5N1 and H9N2 subtypes of the avian influenza virus (AIV), part of the Orthomyxoviridae family, induce one of the most severe zoonotic diseases. The harmful impacts of H5 and H9 avian influenza viruses can affect avian and mammalian species, including human health, and result in economic loss and public health hazards. It has been isolated from several avian species and humans throughout all continents, except Antarctica. The incidence of avian influenza fluctuates based on regional and temporal variables of various avian species and humans. The AIV subtypes are categorized into low-pathogenic and high-pathogenic according to their virulence. H9N2 was classified as low pathogenic, whereas H5N1 was deemed highly pathogenic. Molecular diagnostic tests, such as real-time polymerase chain reaction and conventional polymerase chain reaction, are used to identify avian influenza viruses. The most common serological tests, such as enzyme-linked immunosorbent assay and hemagglutinin inhibition, are used to detect antibodies against influenza A and B viruses. AIV subtypes and host species affect the severity, clinical manifestations, and fatality rates of AIVs. Avian and swine infections are the primary vectors for the transmission of AIV to humans, presenting a considerable public health risk. An adequate surveillance system, a robust vaccination approach, and a segregation and culling protocol for avian influenza-infected birds are urgently required to eliminate avian influenza viruses. This comprehensive study provides a framework for understanding the risks associated with AIV subtypes H5 and H9. The goal is to avoid and control the disease, as it can spread into a major pandemic. Keywords: Epidemiology, Influenza virus, PCR, Public health, Zoonotic disease. IntroductionAvian influenza (AI) is a highly contagious viral illness that can be detected in several wild waterfowl populations (Alexander, 2007). It is also occasionally transmitted to the backyard poultry sector. AI viruses have been a significant veterinary and public health issue in the chicken industry for the past 30 years because of their potential to spread from chickens to other animals (Alexander, 2007). Since 1990, the highly pathogenic avian influenza (HPAI) virus subtype H5N1 has caused a large number of outbreaks in poultry around the world (Alexander, 2007). Additionally, it has been responsible for hundreds of human cases, the majority of which have been fatal (Yuan et al., 2017; Rehman FA Rantam et al., 2022;). From 2003 to 2008, Southeast Asia experienced significant outbreaks of highly pathogenic avian influenza virus (HPAIV) across seven of its eleven countries from 2003 to 2008, highlighting the region’s vulnerability to this zoonotic disease (Zhao et al., 2022). These outbreaks resulted in severe repercussions for both the poultry industry and public health, as HPAIV has the potential to transmit to humans (Peiris et al., 2007). To contain the spread, the affected countries, which included Thailand, Vietnam, Indonesia, Malaysia, Laos, Cambodia, and Brunei, implemented various control measures, such as culling infected flocks and imposing movement restrictions (Sims, 2005). The concept of free movement refers to the natural, uninhibited migration and dispersal behaviors exhibited by avian species across national and continental boundaries. This mobility not only plays a critical role in the ecology of birds but also has significant implications for the transmission of zoonotic pathogens, including avian influenza viruses. Migratory birds, especially waterfowl, serve as reservoirs and vectors for various avian influenza subtypes, facilitating their spread over vast geographic areas regardless of political borders (Olsen et al., 2006; Gilbert et al., 2008a,b). Consequently, international surveillance and coordinated biosecurity efforts are essential to monitor and mitigate disease risks associated with their movements. In contrast, the remaining four countries, namely, Singapore, the Philippines, Myanmar, and Timor-Leste, either reported fewer cases or managed to prevent outbreaks, showcasing diverse responses and preparedness levels within the region (FAO, 2011). These differing outcomes underscore the complex interplay of factors influencing disease transmission, including farming practices, biosecurity measures, and government readiness to handle public health emergencies. Following the H5N1 highly virulent avian influenza virus (AIV) infection that occurred in December 2003, Vietnam became the first country to record the number of fatalities in birds due to the virus. The AIV impacts multiple species, including birds, pigs, and mammals, and possesses zoonotic potential (Kibenge, 2023). AI virus subtypes, such as H5N1 and H9N2, have inflicted considerable economic damage on the poultry industry, particularly in backyard and commercial farms globally. Women have significantly contributed to the advancement of backyard poultry production systems in various geographical areas, enabling them to consistently fulfill their economic needs through the keeping of domestic birds. Low pathogenic avian influenza virus subtype H9N2 is frequently observed in Asian chicken populations. This low pathogenic (LP) subtype mutates to the high pathogenic (HP) subtype H5N1 and, having been isolated from both domestic and wild avian species, could present a significant zoonotic threat in the future. The identification of various bird species across Eurasia has led to considerable economic losses in the poultry sector (Olsen et al., 2006). The H9N2 virus has garnered significant interest owing to its fast propagation across indigenous avian species of Eurasia (e.g., China, Middle East, South Asia) and parts of Africa. The H9N2 viruses endemic to Asia are categorized into three genotypes based on their genetic and antigenic characteristics. This source may have been the origin of the internal genes of the HPAI subtype that struck Hong Kong in September 1997. H9N2 viruses belonging to the G1 lineage have been identified as the cause of influenza outbreaks among poultry in the Middle East and Indian subcontinent between 1997 and the present. A large amount of genetic recombination exists between the genomes of novelavian influenza (H9N2) viruses and the genomes of HPAI viruses. Initially, avian influenza began to spread only in Southeast Asia; however, this trend has now expanded to other regions, including Europe, the Middle East, and countries that were formerly part of the Soviet Union. Wild birds that live in aquatic environments are natural hosts of the AI virus (Olsen et al., 2006). In most cases, clinical indications of infection generally do not appear until the AI virus and its host have reached a state of ideal cohabitation. Additionally, the annual migration of wild avifauna increases the virus’s ability to spread globally (Machalaba Raufman et al., 2021). Despite the inability to inhibit the acquisition of HPAI viruses, the effectiveness of vaccination and concurrent infection with low pathogenic diseases in preventing symptomatic illness or mortality in infected birds are critical factors that are frequently overlooked in the process of assessing the risks associated with avian influenza disease (Dey et al., 2023; Kamal et al., 2023). Initially, the outbreaks of LP and HP avian influenza viruses were associated with poultry, specifically chickens and ducks, and were linked to human transmission only later. The regular interaction of humans with many bird species and other poultry in common locations, such as farms, marketplaces, and abattoirs, makes avian illness management a challenging and complicated task (Peiris et al., 2007; Ventana et al., 2023). As of now, there is no medication that is effective against influenza virus infections in various bird species and mammals, including humans. Consequently, the only therapeutic options available for human infections are supportive care and antiviral drugs to address complications and reduce viral replication. The emergence of resistance to antivirals is becoming an increasingly significant cause of concern. This review aims to help us learn more about the H5N1 and H9N2 types of the avian flu virus, including what caused them, how they spread, how the disease worsens, how to diagnose it, what its signs and symptoms are, how it affects people’s health, how much it costs, how to vaccinate against it, and how to keep it from spreading. This large investigation provides data that may help detect the hazards associated with AIV subtypes H5 and H9. The goal of this study is to prevent and treat this disease, as it can develop into a significant pandemic. EtiologyAI is a viral disease that causes mild or even asymptomatic acute infection, leading to fatal disease in turkeys, guinea fowls, chickens, migratory waterfowl, and other avian species. The causative agent of the disease is a virus that belongs to the AI virus type A and is classified under the family “Orthomyxoviridae.” Some influenza virus strains cause death or severe illness in guinea fowl, turkeys, or chickens. A total of 18 hemagglutinin (HA) and 11 neuraminidase (NA) antigens are present in influenza viruses. Among these, H5N1 is more pathogenic, causing 100% morbidity and mortality in poultry, whereas H9N2 is less pathogenic and causes mild respiratory signs and low mortality (Desouky et al., 2023). Proteins and carbohydrates are components of the envelope used by the influenza virus to encase the virus. The virus uses its spike proteins (NA and HA) to latch onto specific receptors present on host cells. There are two different kinds of spike proteins: those that are placed outside the virion consist of NA and HA. The influenza virus has four different types of antigenic proteins: NA, HA, matrix protein (MP), and nucleocapsid protein (NP). The NP and MP antigen features contribute to the categorization of influenza viruses into types A, B, and C. Influenza A virus causes an illness that is extremely harmful to both humans and animals (Carter et al., 2021) and is responsible for increased rates of morbidity and mortality worldwide (Peiris et al., 2007). The virus is very adaptable and can develop new strains that are more dangerous through antigenic drift or shift, which facilitates the virus’s global spread. According to epidemiological seroprevalence research, there are several influenza A virus subtypes linked to pandemic outbreaks. The influenza C virus is extremely uncommon, despite its potential to infect both humans and animals (Taubenberger and Morens, 2010). Conversely, the influenza B virus is capable of causing disease solely in humans. It is highly unlikely, if not impossible, for influenza virus types B to trigger pandemic outbreaks because they primarily infect humans and lack animal reservoirs, limiting their ability to reassort and cause widespread outbreaks (Javanian et al., 2021). Historical background and current situation of avian influenza in the WorldThe AIV was initially recognized in 1878 in Italy. Due to its virulence and elevated death rate relative to other avian diseases (Alexander and Brown, 2009), it was designated as fowl plague, closely associated with fowl cholera. In 1880, it was referred to as “Typhus Exudatious Gallinarum.” In 1918, it was referred to as the “Spanish influenza” and resulted in the deaths of approximately 30–50 million individuals globally. In 1955, the existence of the “Classical Fowl Plague” was verified in 1955. In 1957, it was designated as “Asian flu,” and in 1968, it was referred to as “Hong Kong influenza”. The initial classification of influenza viruses occurred in 1971, predicated on the antigenic characteristics of the structural and surface proteins of the viruses. Furthermore, the attributes encompassed the influenza species from which the virus derived, along with the NP, HA, and NA proteins (Alexander, 2003). By 1980, researchers had used this technique to classify influenza viruses regardless of their species of origin. This custom endures to this day. During the First International Symposium on Avian Influenza in 1981, the term “fowl plague” was replaced by the more accurate term “avian influenza” (Alexander, 2003), which will be used throughout this review when referring to any previously recognized fowl plague virus. Avian influenza viruses are categorized into two types: HPAI and Low pathogenic avian influenza (LPAI). In experimentally infected hens, a virus is categorized according to specific criteria encompassing viral characteristics and mortality rates. The primary virus causing avian influenza is LP, which causes negligible or absent symptoms in infected wild birds. From 1959 to 1995, 15 outbreaks of the AIV occurred in hens; however, the losses were negligible (Alexander and Brown, 2009). Before the 1990s, avian influenza in poultry resulted in a significant mortality rate; nevertheless, infections were infrequent and manageable. The A/goose/Guangdong/1/1996 (H5N1) virus, first isolated from a domestic goose in Guangdong, China, in 1996, is the ancestral strain that gave rise to all subsequent highly pathogenic H5N1 lineages responsible for major outbreaks across Asia, Europe, and Africa from 1996 onwards (Li South et al., 2004). Between 1996 and 2008, at least 11 outbreaks of avian influenza occurred in poultry, four of which affected millions of birds (Monne and Capua, 2014). The first documented case of human infection in Hong Kong occurred in 1997. Since 2003, nearly 700 cases of Asian H5N1 avian influenza in humans have been recorded, affecting more than 60 countries, primarily in 15 designated nations across the Middle East, Europe, Africa, and Asia (Chan, 2002). As of October 2023, avian influenza, particularly caused by the H5N1 strain, continues to pose significant threats to global poultry populations and, in some instances, to public health. Throughout the year, outbreaks among domesticated birds and wildlife have been reported across different continents. In Europe, countries such as France and the Netherlands have experienced significant outbreaks, resulting in large-scale culling of infected flocks to control the spread of the virus. In Asia, countries such as Vietnam and China have seen a resurgence of H5N1, raising concerns among agricultural authorities and prompting enhanced biosecurity measures (CDC, 2024). Meanwhile, sporadic cases have been reported in North America, with the United States and Canada confirming infections in both wild birds and commercial flocks, leading to strict surveillance and vaccination efforts (CDC, 2024). Public health experts remain vigilant about monitoring human cases and ensuring robust biosecurity practices in poultry farming (controlled access points, regular disinfection of equipment, use of protective gear, separation of flocks by age, and preventing contact between domestic and wild birds), given the highly pathogenic nature of these strains and their potential zoonotic capacity. Recent research has demonstrated sustained mammal-to-mammal transmission of HPAI H5N1 (clade 2.3.4.4b) in both terrestrial and marine species. Caserta et al. (2024) provided one of the first documented instances in dairy cattle, showing cow-to-cow spread and transmission to cats and raccoons via contaminated milking equipment (Caserta et al., 2024). Similarly, phylogenetic analysis of infections among pinnipeds in South America revealed distinct mammalian lineages with PB2 D701N and Q591K mutations, indicating efficient spread across long distances among marine mammals (Rimondi et al., 2024; Pardo et al., 2025). Experimental studies in ferrets using the A/Texas/37/2024 strain confirmed 100% direct-contact transmission and partial airborne spread, while mink farm outbreaks in Europe also showed evidence of mammal-to-mammal spread (Kareinen et al., 2024). Collectively, this mounting evidence underscores a critical shift in H5N1 ecology, with the virus now capable of sustaining transmission chains within mammalian populations. The HPAI H5N1 virus of clade 2.3.4.4b has emerged as the dominant lineage driving an unprecedented global panzootic. This strain evolved from the original A/goose/Guangdong/1/1996 (H5N1) isolate and has circulated extensively across bird populations worldwide since 2020, reaching Antarctica in late 2023, and spreading to every continent except Australia. Between January 2022 and December 2023, this virus caused over 11,400 outbreaks in more than 80 countries, affecting both wild birds and domestic poultry, making it the largest bird influenza epidemic in history. The ability of this panzootic to infect a growing range of mammals is a hallmark. The virus has been detected in dairy cattle, foxes, dogs, sheep, and marine mammals such as sea lions and seals across the United States (CDC, 2024). A new genotypic variant (D1.1) was identified in cattle from Nevada and Arizona, suggesting continued evolution within mammalian hosts (CDC, 2024). The first sheep infection in the United Kingdom was confirmed in March 2025, further highlighting the expanding host range. Although human infections are rare, they have been increasing. Between 2003 and April 2025, the World Health Organization (WHO) confirmed approximately 972 human cases across 24 countries, with approximately 470 deaths (~48% case-fatality rate). Between March and June 2025, 20 human infections were detected across Bangladesh, Cambodia, China, India, Mexico, and Vietnam, including four confirmed deaths (Lieber et al., 2023). Over 70 human infections have been identified in the United States since early 2024, primarily in farm workers exposed to infected poultry or cattle, and a first confirmed human fatality has been reported (CDC, 2024). In Mexico, the first confirmed pediatric case of the D1.1 variant emerged in Durango. Although no evidence of sustained human-to-human transmission has been observed, global health authorities consider the risk of those with occupational exposure to be moderate to high. The CDC and WHO have both emphasized that monitoring and pandemic preparedness remain critical due to the virus’s unpredictable nature and its deepening reservoir in both avian and mammalian species (WHO, 2022; CDC, 2024). International efforts have focused on updating candidate vaccines for humans to mitigate the threat, with several nations, including the UK, Canada, and EU states, expanding their vaccine stockpiles targeted against the 2.3.4.4b lineage. Epidemiology of H5N1 and H9N2 avian influenza virusEpidemiology refers to the examination and evaluation of the distribution, patterns, and determinants of health and disease conditions within specified populations. It is fundamental to public health by identifying disease risk factors and influencing policy decisions and evidence-based practices, and establishing preventive health care targets. The emergence of the HPAI H5N1 virus in southern China during the mid-1990s, its subsequent dissemination throughout East and Southeast Asia, and the extraordinary outbreaks documented in 2003–2004 exemplify this impact most prominently. Between 2014 and 2017, various subtypes of HPAI, such as H5N1, H5N6, and H5N8, emerged in East Asia and disseminated to other continents, underscoring the significance of this region as a source of novel HPAI subtypes (Gilbert et al., 2008a,b). The rapid and continuous advancement of AI viruses renders their monitoring and management challenging. Subsequently, regional and phylogeographical analyses were used to elucidate the mechanisms underlying the formation of highly pathogenic pathogens. The discovery of elevated viral reassortment rates and antigenic diversity in China and Vietnam shows the importance of more closely studying these areas, as they have a higher risk of starting HPAI outbreaks (Chowdhury et al., 2019). Between 2006 and September 2013, 173 human cases of AIV subtype H5N1 were documented in Egypt, resulting in 63 fatalities. These viruses were more widespread in commercial avians than through vaccinations. Mutations in subtype H5N1 may become transmissible among ferrets (Herfst et al., 2012; Imai et al., 2012). In February 2013, the H7N9 AIV was identified in China, resulting in human infections. In October 2013, the AIV subtype H7N9 mutated and infected 137 individuals, resulting in 45 fatalities. All viral genes in China were of avian origin, with six internal genes from the AIV subtype H9N2. A cross-sectional study was conducted from July 2011 to August 2011, using a convenience sample method across three regions of Maryland for domestic poultry (Vaziri et al., 2022). A total of 240 swab samples were obtained from the trachea and cloaca of 39 registered premises. Blood samples were obtained from these birds. Analysis revealed that the frequency of AIV subtype H5N1 was 4.2% in poultry birds (Vaziri et al., 2022). The real-time polymerase chain reaction (RT-qPCR) analysis indicated that the samples were negative for avian influenza Ribonucleic Acid, confirming the absence of H5, H7, or H9 avian influenza hemagglutinin strains. In 2011, AIV subtype H9N2 infection was detected in people living in Bangladesh. Between 2008 and 2011 in Bangladesh, a surveillance study revealed continuous isolation of AI subtype H9N2 viruses from live bird markets year-round between 2008 and 2011. In addition to subtype H9N2, subtype H5N1 viruses classified as HPAI were isolated in the chicken market during the winter months. Between 1995 and 2003, five significant AIV epidemics, including H5, H7, and H9, were associated with circulating subtypes of avian influenza (Naeem and Siddique, 2006). In these years, the highly HPAI subtype H7N3 caused the mortality of 3.2 million birds (Khan et al., 2021). In 1998, the initial outbreak of AIV H9N2 in poultry was reported in 1998. This outbreak had similarities with the AIV subtypes that were prevalent in Hong Kong during that period. The disease remains frequently occurring in the country. In India, approximately 70 outbreaks of H5N1 have occurred in poultry in the northern and eastern regions (Marchenko et al., 2023). Active monitoring of wild birds and poultry is crucial, as conducted in 2009, 2010, and 2011 in Bangladesh, which revealed the circulation and persistence of H5N1 across live bird markets and migratory bird habitats (Biswas et al., 2011). A total of 5722 samples, comprising fecal, blood, environmental samples, and tracheal swabs, were used to detect and isolate the virus via HI and PCR methods. Of the total collected samples, 57 tested positive for H9N2, 15 for H4N6, and one human case was also positive for H4N6. The ducks also tested positive for H4N6, which has minimal pathogenicity. Similarly, antibodies confirmed the presence of H5 and H7 viruses, whereas chickens tested positive for H9N2. This alerts us to implement all essential precautions to control the propagation of the virus. As of January 31, 2024, the World Health Organization has documented 94 instances of human infection with avian influenza A (H9N2) in the Western Pacific region. Pathogenesis of H5 and H9 avian influenza virusAvian influenza viruses can infect and lethally affect numerous bird species. Their virulence categorizes them into two groups. The HPAI has been recognized as a deadly virus responsible for avian epidemics (Chen et al., 2009). H5 and H7 were classified as highly pathogenic avian influenza viruses associated with approximately 100% elevated fatality rates. The H9N2 subtype of AIV, classified as LPAI, induces moderate respiratory illness (Alexander, 2007). There is ambiguity over the determinants that facilitate the transition of the virus from LPAI to HPAI (Swayne East and Suarez, 2000). Mutations develop rapidly under specific conditions following the introduction of wild birds. In other cases, the LPAI virus persisted in hens for several months before mutation (Xing et al., 2008). A specific set of genes primarily governs immune evasion, replication efficacy, and host and tissue tropism in AIVs. These viruses are polygenic and predominantly rely on this gene group. Moreover, characteristics unique to the host and those particular to the species influence the progression of infection following interspecies transfer. The LPAI virus can infect avian populations through several pathways, highlighting its transmission dynamics’ complexity. Direct contact with infected birds, where domesticated poultry and wild birds share habitats or interact in markets or farms, is a significant pathway (Alexander, 2007). Additionally, environmental contamination plays a crucial role; the virus can survive in the droppings of infected birds, contaminating water bodies, feed, or surfaces that other birds access. Indirect transmission may occur through contaminated equipment or personnel who handle infected birds and then come into contact with healthy flocks (Alexander, 2007). Furthermore, the migratory behavior of wild birds facilitates the spread of LPAI over long distances, as these birds can carry the virus asymptomatically and introduce it into new regions. Understanding these pathways is essential for implementing effective biosecurity measures and vaccination strategies to curb the spread of LPAI among avian populations (Xing et al., 2008). These viruses may develop into strains that significantly threaten sensitive bird populations (Monne and Capua, 2014). Upon the inhalation or ingestion of infected LPAI or HPAI virions, the degradation of surface HA by trypsin-like enzymes in intestinal and respiratory epithelial cells may lead to illness manifestation (Abd et al., 2020; Suardana et al., 2023). Consequently, many replication cycles occur in the intestines and respiratory tract, resulting in the release of infectious virions into the environment through feces and respiratory secretions, which facilitates further transmission to susceptible hosts. HPAI viruses may infiltrate the capillaries upon concluding their reproduction in the respiratory epithelium (Taubenberger and Morens, 2010). This event transpires after the viruses penetrate the submucosa. The virus initially replicates in endothelial cells before disseminating throughout the lymphatic and circulatory systems to infect more hosts that can infect and proliferate in various cell types found in the skin, brain, and internal organs. It is capable of spreading throughout the host before extensive replication occurs within Vascular Endothelial Cells (Short et al., 2014). The virus can be detected in erythrocytes, leukocytes, and plasma, indicating its ability to target and affect a range of blood cell types (Peiris et al., 2007). Evidence indicates that macrophages contribute to the dissemination of viruses within the structure of the organism (Short et al., 2014). The proteolytic cleavage sites of HA are catalyzed by the enzyme furin, which is essential for enabling pantropic replication (Short et al., 2014). Simultaneous failure of many organs can lead to clinical manifestations and elevated patient fatality rates. Typically, LPAI viruses primarily reproduce within the gastrointestinal or respiratory systems. Injuries to the respiratory system frequently lead to sickness or mortality, especially when they are linked to bacterial infection. Several organs are vulnerable to LPAI virus replication and damage. The organs include the renal tubules, pancreatic acinar epithelium, fallopian tubes, and other organs containing epithelial cells that sporadically display trypsin-like enzymes. The pathogenesis of AIV infection in birds that do not ingest gallinaceous diets is unknown. Laboratory diagnosisLaboratory diagnosis is essential for determining an individual’s influenza virus infection status during pandemics or seasonal outbreaks (Mahony, 2008). Laboratory testing must confirm probable cases and distinguish them from flu-like illnesses caused by other respiratory viruses, such as adenoviruses, picornaviruses, parainfluenza viruses, respiratory syncytial viruses, and rhinoviruses (Mahony, 2008). According to Lum et al. (2018), various diagnostic methods have been developed for identifying influenza virus infection, which may be classified as either direct or indirect. There are several direct approaches, including the direct identification of viral particles, rapid antigen detection techniques such as immunofluorescence, and enzyme-linked immunosorbent assay. Indirect methods utilize standard and accelerated cell culture, egg injection, or animal inoculation for viral propagation and classification. Nucleic acid testing, also known as RT–PCR, as well as serological diagnostic methods, such as complement fixation, hemagglutination inhibition, and neutralization assessments, have been utilized in the study of influenza virus The sensitivity of each diagnostic test varies, with unique advantages and limitations. Moreover, quantitative RT–PCR is the most sensitive and accurate technique for detecting and quantifying the influenza A virus (WHO, 2012). This technique is commonly used to detect the viral M gene, which is the most conserved gene across all influenza A virus subtypes. Hemagglutination inhibition and complement fixation are two serological assays employed to identify infections and evaluate the host’s response to influenza vaccination. Cell culture techniques can be used to detect influenza viruses (World Health Organization, 2011). Cell culture techniques play a pivotal role in the detection and study of influenza viruses, as they provide an environment for the virus to replicate and facilitate further analysis. The selection of suitable cell lines is critical for accurate detection, as different cell types exhibit varying susceptibilities to different influenza strains. Among the most commonly employed cell lines are Madin-Darby Canine Kidney (MDCK) cells, which are widely recognized for their high susceptibility to a broad range of influenza A viruses. MDCK cells are particularly advantageous for isolating circulating strains because they allow for efficient viral replication and yield high titers, making them a preferred choice in diagnostic laboratories and for vaccine production. The A549 lung carcinoma cell line, which originates from human alveolar epithelial cells, is another notable cell line. A549 cells are frequently used for studying human respiratory viruses, including influenza, due to their ability to mimic the respiratory tract environment and support the replication of both influenza A and B viruses (Leyrat et al., 2014). In addition, primary human airway epithelial cells have gained prominence in research because they provide a more physiologically relevant model for influenza replication and host-virus interactions, although their use is limited due to the complexity and cost of culturing primary cells. For strains that exhibit a preference for avian hosts, such as H5N1, chicken embryonic fibroblasts are also used effectively because they are derived from chicken embryos and facilitate the growth of avian-adapted influenza strains. Each cell line offers unique advantages, depending on the specific requirements of the study, including the type of influenza virus being isolated or studied, the need for vaccine development, or the exploration of viral pathogenesis (Pinto et al., 2022). Ultimately, the appropriate selection of cell lines in influenza virus research is crucial for accurate viral detection, characterization, and effective management strategies against influenza outbreaks. A specimen is introduced into a live culture system, and the virus is detected after a certain period of time. Furthermore, embryonated chicken eggs aged 9–11 days can be used to culture and isolate the influenza virus by inoculating the egg’s amniotic cavity with the sample. The viruses require a minimum of 3 days to replicate within the eggs before harvesting them (Pedersen et al., 2008; Pulit-Penaloza et al., 2018). Clinical signs and symptoms of H5 and H9The incubation period for influenza in hens ranges from 1 to 7 days. Low pathogenic avian influenza strains often present asymptomatically or with mild respiratory signs and decreased egg production. In contrast, HPAI causes severe clinical signs, including sudden death (with mortality rates approaching 100%), dark green diarrhea, anorexia, neurological symptoms, and systemic manifestations, such as facial edema, cyanosis, and disheveled plumage (Akanbi et al., 2015). Turkey exhibits particularly severe symptoms, including snood swelling, subcutaneous extremity hemorrhages, and internal petechial hemorrhages. Neurological involvement may present as paralysis, torticollis, or incoordination, whereas systemic infection can cause microvascular lesions in multiple organs, leading to edema, hemorrhage, and necrosis (Taubenberger and Morens, 2010). Reproductive impacts are common, with mucosal inflammation often causing decreased egg production or shell abnormalities. Notably, some fatal cases may show minimal premortem clinical signs (Korteweg and Gu, 2008). In humans, AI presents with symptoms resembling seasonal flu (fever, cough, and myalgia), although H5N1 and H7N9 subtypes can cause severe respiratory failure (Olsen et al., 2006; Li et al., 2004). Disease severity depends on viral characteristics and host factors, including age, comorbidities, and immune status (Rouse and Sehrawat, 2010), with immunocompromised individuals and those with cardiopulmonary disease having the highest risk (Kalil and Thomas, 2019). Avian influenza viruses are primarily transmitted from animals to humans through direct contact, and the cycle of transmission typically involves wild aquatic birds, domestic fowl, pigs, and humans (Montalvo-Corral Reyes et al., 2010). Wild aquatic birds act as natural reservoirs for all influenza A virus subtypes and are responsible for spreading the virus into the environment, especially through water sources via oral fecal transmission. This contamination can lead to infections in terrestrial (domestic) fowls, such as chickens and turkeys, which are more likely to have direct or indirect contact with humans and livestock (Stallknecht and Shane, 1988; Montalvo-Corral Reyes et al., 2010; Pulit-Penaloza et al., 2018). The first human infection with H5N1 was reported in Hong Kong in 1997, with 18 confirmed cases and 6 fatalities (Chan, 2002). Figure 1 illustrates the cyclical and interconnected transmission pathway among wild birds, domestic poultry, pigs, and humans, emphasizing the importance of a One Health approach in monitoring and controlling AIV transmission (Fig. 1).
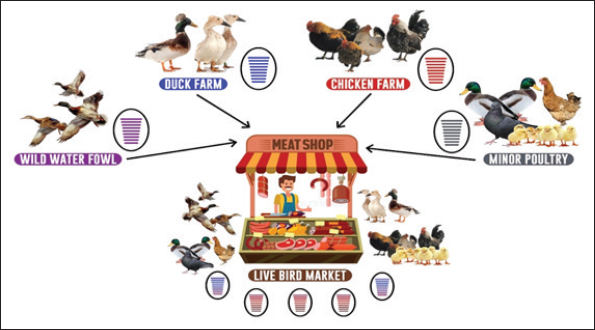
Fig. 1. Influenza A viruses and pigs serving as reservoirs for AIV transmission (Rehman Rantam et al., 2022). The virus can then be transmitted from domestic fowl to pigs, which serve as important “mixing vessels” due to their susceptibility to avian, swine, and human influenza A virus strains. This allows for genetic reassortment, which may lead to the emergence of novel influenza strains with zoonotic potential (Webby et al., 2004). Consequently, pigs can act as a bridge for the virus to infect humans, further facilitating interspecies transmission. AIV subtypes such as H5N1 and H9N2 are capable of replicating in human cells (Ma et al., 2008). Live bird markets (LBMs) and meat shops represent critical hubs in the transmission dynamics and ecological maintenance of AIVs, especially the H5N1 and H9N2 subtypes (Rehman Rantam et al., 2022). Figure 1 shows that wild waterfowl serve as the initial reservoir for AIV, spreading the virus to various types of poultry, including ducks, chickens, and minor poultry species via direct or indirect contact. These infected birds are then brought into duck farms, chicken farms, and small-scale poultry farms. The distribution of these birds is largely directed toward meat shops and live bird markets, especially through traditional and unregulated farming systems (Rehman et al., 2023). LBMs play a significant role in maintaining the endemicity of AIVs due to the dense cohabitation of multiple avian species in close proximity to humans. Ducks, chickens, and various minor poultry converge at these markets, facilitating cross-species transmission and viral reassortment (Fig. 2). Such environments are conducive to viral mutations, leading to the emergence of pandemic-potential novel influenza strains. Studies have highlighted that poultry density, cage stacking methods, hygiene practices (e.g., killing location and display surfaces), road and human density, nearby water bodies, and environmental variables are risk factors contributing to the spread of HPAI H5 and other AIVs in these markets (Rehman Rantam et al., 2022; Rehman et al., 2023). These conditions, when coupled with the tropical climate and traditional farming practices—such as allowing ducks to forage in harvested rice fields—create ideal conditions for virus survival and evolution. The presence of H5N1 in both household birds and commercial poultry at these market hubs, along with the increasing detection of H9N2, suggests an increasing likelihood of reassortment events. H9N2, in particular, has been identified as a key intermediate that facilitates the emergence of new human-infecting AIV strains, including H7N9 and H10N8. The reassortment of low-pathogenic H9N2 with highly pathogenic strains may further enhance their zoonotic potential. Given these transmission pathways and risk factors, ongoing surveillance in live bird markets, farms, and surrounding environments is crucial for the early detection and prevention of novel influenza virus outbreaks (Rehman et al., 2023). The figure underscores the central role of live bird trade networks in the spread and evolution of AIVs, highlighting the urgent need for biosecurity, monitoring, and policy interventions in these areas (Fig. 3).
Fig. 2. Contribution of live bird markets to the evolution of avian influenza viruses (Rehman Rantam et al., 2022).
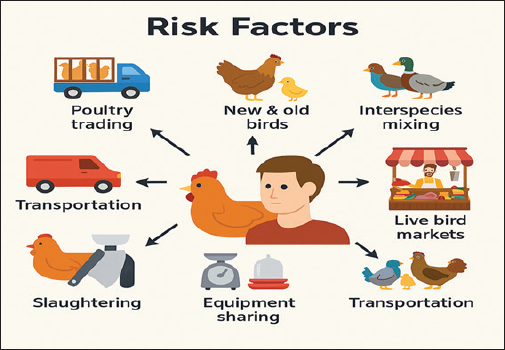
Fig. 3. Risk factors for influenza A virus infection. Several temporal and spatial risk factors are associated with the onset of sickness, the virulence of different avian influenza virus strains, and frequent mutations that impede the effectiveness of preventative and therapeutic efforts for controlling avian influenza. The primary risk factors identified include poultry trading patterns, the amalgamation of newly introduced birds with residual birds from prior batches interspecies mixing within enclosures, the source of chicken procurement, the communal use of slaughtering, feeding, watering, and weighing equipment, the daily volume of birds sold (Vaziri et al., 2022), and the transportation of poultry via LBMs. Slaughtering, butchering, handling contaminated chicken carcasses, and preparing virus subtypes H5, H7, or any other subtype for human exposure are all potential risk factors for disease transmission (Vaziri et al., 2022). Importance of public healthAvian influenza remains a significant public health concern, particularly given the recent resurgence of H5N1 virus outbreaks in both wild and domestic bird populations around the world. The ongoing circulation of H5N1 poses serious risks to public health, veterinarians, and medical professionals. Earlier studies have reported that H5N1 causes infections in humans, particularly in regions where the virus has been prevalent in poultry, underscoring the potential for severe illness and mortality among affected individuals (CDC, 2023). The transmission of H5N1 to humans, although still relatively rare, raises substantial concerns regarding its mutational ability and the potential for reassortment with circulating human influenza viruses, which could lead to a pandemic (Herfst et al., 2012). Emergence of avian influenza strains capable of efficient human-to-human transmission remains a critical issue. Such transmission would significantly increase the number of affected individuals, resulting in increased rates of illness and mortality (Imai et al., 2012). Recent data indicate that novel H5N1 strains can cause severe respiratory disease in humans, emphasizing the need for continuous surveillance and research to monitor changes in the pathogenicity and transmissibility of the virus (WHO, 2022). In addition to H5N1, other subtypes, such as H7N9 and H9N2, continue to pose threats to human health due to sporadic infections reported in various countries. These subtypes have been associated with severe respiratory illness and fatalities (Qi et al., 2018). The complex interplay of these viruses within both domestic and wild avian populations necessitates a vigilant public health response to prevent possible outbreaks among the human population. The One Health approach, which emphasizes the interconnectedness of human, animal, and environmental health, remains crucial in managing influenza (Pulit-Penaloza et al., 2018). Vaccination strategies for susceptible animals, including domestic birds and some farm animals, are vital in controlling the spread of the virus in animal populations and reducing the risk of zoonotic transmission to humans (Swayne East and Suarez, 2000). Enhanced biosecurity measures in poultry farms, continued research, and public health preparedness are essential components in mitigating the potential impact of influenza on global health. Economic impacts of AIV (H5N1 and H9N2)Because of the AI H5N1 and H9N2 outbreaks, several countries have reported losses in poultry that amount to approximately 1% of their gross domestic product. Vietnam has experienced losses that are as high as 1% (Otte et al., 2009). Consequently, the virus gradually spreads to new birds. During the month of June 2005, over 250 million birds died due to the disease, which had an estimated economic impact of 12 billion dollars across 62 countries. Information systems established by academic, commercial, and scientific institutions have been suggested to mitigate the impact of this condition (Perry and Sones, 2007). Between 1983 and 2005, 356.64 million chickens died due to efforts to avoid several AI virus epidemics (Capua and Alexander, 2009). Due to the introduction of the H5N1 strain of avian influenza in Hong Kong in 1997, the global pandemic risk has remained a persistent source of concern (Webster and Govorkova, 2006). Additionally, the H5N1 avian influenza subtype and its mutations have been responsible for the deaths of over 200 million birds, resulting in losses exceeding 10 billion dollars for the poultry industry. These losses have also impacted research and human life. Agricultural losses in China neared 1 billion dollars in 2004, and businesses’ income in China fell by two and a half billion dollars. To improve disease control, it is possible to construct a chronological chronology of events, beginning with the start of the epidemic and ending with the beginning of production. Losses in an impacted area may also bring benefits to unaffected areas (Peiris et al., 2007). The probability of an AI virus infection in Thailand was estimated in 2004 during the pandemic. This decision was informed by the virus’s genus and strain (Tiensin Chaitaweesub et al., 2005; Gilbert et al., 2006). The quail has the highest risk at 1.3%, followed by laying hens and broilers, each with a risk of 0.25%, then ducks and geese, each with a risk of 0.075%, and finally free-range chickens, which have the lowest risk of 0.05%. China saw a reduction in exports of 63%, Hong Kong experienced a decline of 55%, and the United States experienced a decline of 27% from January to April 2004 (Baffes et al., 2005). Thailand suffered the most significant impact of the outbreak, with a 75% decline in exports. Brazil was the only country reporting a 6% gain in exports. Between 2003 and 2005, Brazil benefited from restrictions imposed on Asian countries, namely Thailand and Turkey, regarding their chicken export capabilities (FAO, 2011). Although non-processed meat sales in Brazil quadrupled from US$ 1.5 billion to US$ 2.9 billion, processed meat sales in Brazil grew from US$ 220 million to US$ 398 million, while processed meat sales doubled (FAO, 2011). In 2006, the demand for meat and eggs fell across most Asian nations, even in 15% of countries unaffected by the disease, such as Argentina and Brazil, and 30% of countries severely impacted by the epidemic (FAO, 2011). During that particular year, China, Brazil, and the European Union all reduced their exports of chicken meat by 13%, 7%, and 2%, respectively. In the same year, the United States held a 36% share of the global market, Brazil held a 37% stake, the European Union held a 12% share, and China held a 1.9% share. Brazil now accounts for 17% of the total cooked beef exports worldwide, an increase from 13% in 2005. TreatmentAntiviral drugs are crucial in the treatment, reduction of disease severity, and management and prevention of disease. Antiviral medicines for influenza are classified into two primary categories: adamantanes and neuraminidase inhibitors (Chen et al., 2017). Rimantadine (flumadine) and amantadine (symmetrel) were the initial effective pharmaceuticals approved for the treatment of influenza. Amantadine was approved for prophylaxis by the Food and Drug Administration in 1966, and in 1976, it was used as an antiviral agent for children aged >1 year and adults. In 1993, the Food and Drug Administration sanctioned ribantadine for prophylactic use in preventing the disease in individuals aged 1 year and older, including adults (Chen et al., 2017). Among these two antiviral medications, amantadine is used less frequently because of its unfavorable effects on the central nervous system and a higher rate of drug resistance. Mutations in the viral M gene occur due to the modification of a single amino acid in the M2 transmembrane region. Mutations in the viral M gene confer resistance to amantadine, allowing the isolation and transmission of the resistant virus to a susceptible host. Consequently, the use of this medication was restricted to treating influenza (Kode et al., 2014). All influenza viruses are suppressed by neuraminidase inhibitors because they obstruct the release of virions from the infected cell. Zanamivir (Relenza) and oseltamivir (Tamiflu) are primary neuraminidase inhibitors that mitigate influenza infection transmission and severity. In 1999, the Food and Drug Administration initially introduced these drugs for treating uncomplicated acute influenza. Both medications are also effective in inhibiting disease transmission (chemoprophylaxis). These medications outperform M2 blockers due to their extensive efficacy against influenza A and B. Recently released pharmaceuticals in Japan, such as laninamivir and peramivir, are superior due to the central nervous system adverse effects associated with alternative medications. Vaccination of AIVsMany countries have established regular immunization programs. The following nations were responsible for administering 99% of the vaccination: (a) Indonesia (2.3%), (b) Egypt (4.6%), and (c) China (10.9%). The Hong Kong Special Administrative Region and Vietnam constitute 1.4% of the total. The AIV vaccine was used by less than 1% of individuals in Pakistan, as well as in other countries such as Mongolia, France, Israel, the Netherlands, Russia, Kazakhstan, Côte d’Ivoire, North Korea, and Sudan (Swayne East and Suarez, 2000). The H9N2 virus strain vaccine administered in Korea showed a substantial level of efficacy, leading to a reduction in the frequency, severity, and antigenic diversity of the disease in chickens. The interaction between humans and diseased poultry harboring H5N1 virus strains poses a significant risk and has the potential to result in rapid human infection (Richard et al., 2010). To enhance safety, individuals who have been exposed should be supplied with a readily available vaccine. The effectiveness of a formalin-inactivated vaccination against the circulating virus was evaluated for postexposure protection. Immunity and safety in deadly infections improved in a mouse model using the prototype HPAI H5N1 strain, designated A/Vietnam/1203/2004. Furthermore, this vaccine demonstrated a significant level of effectiveness when administered 24 hours after the initial exposure. Distributing vaccines is an effective means of stopping the spread of AI viruses in Indonesia. It is crucial to regularly update the vaccine master seed to keep up with the evolving viral landscape and ensure that field-used vaccines remain compatible. The LPAI H9N2 virus was selected as the vaccine strain due to its pathogenic, antigenic, and genetic properties of the BLi25Ut/18 virus. This strain, A/chicken/West Java/BBLitvet-RI/2017, was identified in 2017 and assigned Patent IDP000056903 (Rehman Rantam et al., 2022). Inactivated bivalent and monovalent H9N2 influenza vaccinations may induce an antibody response (Dharmayanti et al., 2025). It has the capacity to diminish the mortality rate and viral shedding associated with reassortant H9N2 virus infection. With support from the FAO and OFFLU agencies, the Indonesian government has established an effective immunization strategy against the H5N1 and H9N2 viruses. In Vitro Maturation online, an acronym for influenza virus monitoring, is a digital technology used for the administration of influenza virus in animal health laboratories. All structural proteins, including nucleoproteins and matrix proteins, can be quantified using readily available oil-based inactivated bivalent and monovalent vaccines. The use of either of these proteins or both as antigens in antibody testing is possible (Rehman et al., 2023). Using this method, it becomes very difficult, if not impossible, to differentiate vaccinated birds from those that have contracted the disease naturally. The absence of monitoring activities has been a major setback to avian flu vaccination campaigns. According to Rehman et al. (2023), genetic reassortment between H9N2 and H5N1 viruses is more likely to occur in Indonesia’s agricultural and avian markets if both viruses are present simultaneously. In addition to thoroughly and continuously monitoring chicken farms and live bird markets for the appearance of LP H9N2 variations, it is necessary to investigate vaccine seed viruses. Ensuring this will guarantee the effective implementation of future preventive actions while creating vaccines that are both cost-effective and highly immunogenic (Rehman Rantam et al., 2022). Indonesia is exploring inactivated bivalent and monovalent immunization and has developed some innovative vaccines based on technology. Combining new vaccines with effective adjuvants will increase their efficacy against the LPAI subtype H9N2. ConclusionThe avian influenza virus subtypes H5 and H9 are acknowledged as significant zoonotic illnesses due to their severe public health and economic repercussions, particularly with chicken mortality. Avian influenza is not confined to particular places, as it has been documented globally on nearly all continents. The influenza virus is very dangerous and can cause severe illnesses in humans through direct transmission from sick birds. A number of significant risk factors for zoonotic transmission to humans have been identified. These risk factors include contact with diseased birds, whether they are living or deceased, exposure to contaminated poultry markets, slaughtering, butchering, and handling tainted chicken carcasses. There have been reports of the influenza virus being found in domestic pets, wild birds, and a wide variety of other types of wildlife. Respiratory symptoms are a common manifestation of influenza virus-induced illness. Sore throat, nasal congestion, fever, cough, chills, myalgia, headache, weariness, and conjunctivitis are some of the other symptoms that are commonly experienced by people who are infected with another virus. Molecular or serological techniques frequently uncover the AIV in a laboratory setting. Therefore, there is currently no treatment that is effective for avian influenza virus infections in commercial poultry; however, supportive care and antiviral drugs are often the only treatments that are used for human infections. The fact that the influenza virus frequently develops resistance to regularly used antivirals makes the situation more complicated. Numerous studies and publications have indicated that implementing efficient vaccination strategies within the One Health framework, including the expedited development of effective vaccines and stringent hygiene protocols, will significantly reduce the incidence of avian influenza outbreaks and pandemics. This is the case, although a multitude of facts and reports AcknowledgmentsThe authors would like to express their profound gratitude to the Faculty of Veterinary Medicine. The authors would like to thank Airlangga University, the University of Veterinary and Animal Sciences Swat, and Gomal University for their cooperation in the data collection process. Conflict of interestAll authors declare no conflicts of interest. FundingThe Faculty of Veterinary Medicine, Universitas Airlangga, Indonesia, supported this research. The sponsors played no role in the study conception, data collection and analysis, publication decision, or paper writing. Author’s contributionsAll authors participated in data analysis, manuscript drafting, and revisions. Consent to assume responsibility for all aspects of this study. Data availabilityAll data were provided in the manuscript. ReferencesAkanbi CA Meseko., Odita I Shittu., Rimfa D Ugbe. and Shamaki, D. 2015. Epidemiology and clinicopathological manifestation of resurgent highly pathogenic avian influenza (H5N1) virus in Nigeria, 2015. Nigerian. Vet. J. 37, 175–186; doi:10.1016/j.nigevj.2015.07.010 Alexander, D.J. 2003. Report on avian influenza in the Eastern Hemisphere during 1997–2002. Avian. Dis. 47(s3), 792–797. Alexander, D.J. 2007. Summary of avian influenza activity in Europe, Asia, Africa, and Australasia, 2002–2006. Avian. Dis. 51, 161–166. Alexander, D.J. and Brown, I.H. 2009. History of highly pathogenic avian influenza. Revue. Scientifique. Et. Tech. (Int. Office. Epizootics). 28(1), 19–38; doi:10.20506/rst.28.1.1856 Baffes, J., Lewin, B., & Varangis, P. 2005. Global Agricultural Trade and Developing Countries, 297. World Bank. Washington, DC. Biswas, P.K., Christensen, J.P., Ahmed, S.S., Barua, H., Das, A., Rahman, M.H., Giasuddin, M., Habib, M.A., Hannan, A.S. and Debnath, N.C. 2011. Mortality rate and clinical features of highly pathogenic avian influenza in naturally infected chickens in Bangladesh. Revue. Scientifique. Et. Tech. (Int. Office. Epizootics). 30(3), 871–878; doi:10.20506/rst.30.3.2080 Capua, I. and Alexander, D.J. 2009. Avian influenza infection in birds: a challenge and opportunity for the poultry veterinarian. Poultry. Sci. 88(4), 842–846; doi:10.3382/ps.2008-00289 Carter, P., Megnin-Viggars, O. and Rubin, G.J. 2021. What factors influence symptom reporting and healthcare access during an emerging infectious disease outbreak? A rapid review of the evidence. Health. Secur. 19(4), 353–363. Caserta, L.C., Frye, E.A., Butt, S.L., Laverack, M., Nooruzzaman, M., Covaleda, L.M., Thompson, A.C., Koscielny, M.P., Cronk, B., Johnson, A., Kleinhenz, K., Edwards, E.E., Gomez, G., Hitchener, G., Martins, M., Kapczynski, D.R., Suarez, D.L., Alexander Morris, E.R., Hensley, T., Beeby, J.S., Lejeune, M., Swinford, A.K., Elvinger, F., Dimitrov, K.M. and Diel, D.G. 2024. Spillover of highly pathogenic avian influenza H5N1 virus to dairy cattle. Nature 634(8034), 669–676; doi:10.1038/s41586-024-07849-4 CDC. 2024–2025 influenza season: surveillance or novel influenza A and seasonal influenza viruses. Atlanta, GA: US Department of Health and Human Services, Centers for Disease Control and Prevention; 2024. Chan, P.K. 2002. Outbreak of avian influenza A(H5N1) virus infection in Hong Kong in 1997. Clin. Infect. Dis. 34 Suppl 2(Suppl 2), S58–S64; doi:10.1086/338820 Chen, J., Fang, F., Yang, Z., Liu, X., Zhang, H., Zhang, Z., Zhang, X. and Chen, Z. 2009. Characterization of highly pathogenic H5N1 avian influenza viruses isolated from poultry markets in central China. Virus. Res. 2019(1-2), 19–28. Chen, L.J., Lin, X.D., Tian, J.H., Liao, Y., Ying, X.H., Shao, J.W., Yu, B., Guo, J.J., Wang, M.R., Peng, Y., Shi, M., Holmes, E.C., Yang, Z.Q. and Zhang, Y.Z. 2017. Diversity, evolution and population dynamics of avian influenza viruses circulating in the live poultry markets in China. Virology 505, 33–41; doi:10.1016/j.virol.2017.02.009 Chowdhury, S., Hossain, M.E., Ghosh, P.K., Ghosh, S., Hossain, M.B., Beard, C., Rahman, M. and Rahman, M.Z. 2019. The Pattern of Highly Pathogenic Avian Influenza H5N1 Outbreaks in South Asia. Trop. Med. Infect. Dis. 4, 138; doi:10.3390/tropicalmed4040138 Desouky, A.I.M., Abdelnaser, A.H.T., Moustafa, M.M.A., Elboraay, I.M. and Abodalal, S.E.S.A. 2023. Epidemiology of rabbit hemorrhagic disease virus infection in different Egyptian governorates during 2021–2022 years. Int. J. Vet. Sci. 12(6), 768–775. Dey, P., Ahuja, A., Panwar, J., Choudhary, P., Rani, S., Kaur, M., Sharma, A., Kaur, J., Yadav, A.K., Sood, V., Suresh Babu, A.R., Bhadada, S.K., Singh, G. and Barnwal, R.P. 2023. Immune Control of Avian Influenza Virus Infection and Its Vaccine Development. Vaccines 11(3), 593; doi:10.3390/vaccines11030593 Dharmayanti, N.L., Indriani, R., Nurjanah, D., Nuradji, H., Suyatno, T. and Djufry, F. 2025. Antiviral activity of Eucalyptus globulus and Eucalyptus citriodora essential oils against H5N1 avian influenza virus and infectious bronchitis virus. Trop. J. Pharm. Res. 24(2), 185–193. East, E., Post, D.J., East, S., Roberts, P.C., Augustine, A.D., Ferguson, S. and Morens, D.M. 2018. A universal influenza vaccine: the strategic plan for the National Institute of Allergy and Infectious Diseases (NIAID). J. Infect. Dis. 218(3), 347–354; doi:10.1093/infdis/jiy103 Food and Agriculture Organization. 2011. Approaches to controlling, preventing and eliminating H5N1 highly pathogenic avian influenza in endemic countries. Paris: FAO, 2011. Gilbert, M., Chaitaweesub, P., Parakamawongsa, T., Premashthira, S., Tiensin, T. and Kalpravidh W Slingenbergh. 2006. Free-grazing ducks and highly pathogenic avian influenza in Thailand. Emerg. Infect. Dis. 12(2), 227–234; doi:10.3201/eid1202.050640 Gilbert, M., Xiao J Domenech., Lubroth V Martin. and Slingenbergh. 2008. Migration of Anatidae in the western Palearctic and spread of the highly pathogenic avian influenza H5N1 virus. Emerg. Infect. Dis. 12(11), 1650–1656; doi:10.3201/eid1211.060223 Gilbert, M., Xiao, X., Pfeiffer, D.U., Epprecht, M., Boles, S., Czarnecki, C., Chaitaweesub, P., Kalpravidh, W., Minh, P.Q., Otte, M.J., Martin, V. and Slingenbergh, J. 2008. Mapping H5N1 highly pathogenic avian influenza risk in Southeast Asia. Proc. Natl. Acad. Sci. U. S. A. 105(12), 4769–4774. Herfst, S., Schrauwen, E.J.A., Linster, M., Chutinimitkul, S., De Wit, E., Munster, V.J., Sorrell, E.M., Bestebroer, T.M., Burke, D.F., Smith, D.J., Rimmelzwaan, G.F., Osterhaus, A.D.M.E. and Fouchier, R.A.M. 2012. Airborne transmission of influenza A/H5N1 virus between ferrets. Science 336(6088), 1534–1541; doi:10.1126/science.1213362 Imai, M., Watanabe, T., Hatta, M., Das, S.C., Ozawa, M., Shinya, K., Zhong, G., Hanson, A., Katsura, H., Watanabe, S., Li, C., Kawakami, E., Yamada, S., Kiso, M., Suzuki, Y., Maher, E.A., Neumann, G. and Kawaoka, Y. 2012. Experimental adaptation of an influenza H5 HA confers respiratory droplet transmission to a reassortant H5 HA/H1N1 virus in ferrets. Nature 486(7403), 420–428; doi:10.1038/nature10831 Javanian, M., Barary, M., Ghebrehewet, S., Koppolu, V., Vasigala, V. and Ebrahimpour, S. 2021. A brief review of influenza virus infection. J. Med. Virol. 93(8), 4638–4646. Kalil, A.C. and Thomas, P.G. 2019. Influenza virus-related critical illness: pathophysiology and epidemiology. Crit. Care. 23(1), 258. Kamal, M.A., Khalaf, M.A., Zakia, A.M.A., Fathy, M., Hala, M.F.E.M., Mahmoud, A.M., Osman, A. and Zaki Ewiss, M.A. 2023. Effect of water organic load and total ammonia nitrogen on the humoral immune response of broilers against Newcastle disease virus vaccination in Egypt. Int. J. Vet. Sci. 12(1), 107–113; doi:10.47278/journal.ijvs/2022.164 Kareinen, L., Tammiranta, N., Kauppinen, A., Zecchin, B., Pastori, A., Monne, I., Terregino, C., Giussani, E., Kaarto, R., Karkamo, V., Lähteinen, T., Lounela, H., Kantala, T., Laamanen, I., Nokireki, T., London, L., Helve, O., Kääriäinen, S., Ikonen, N., Jalava, J., Kalin-Mänttäri, L., Katz, A., Savolainen-Kopra, C., Lindh, E., Sironen, T., Korhonen, E.M., Aaltonen, K., Galiano, M., Fusaro, A. and Gadd, T. 2024. Highly pathogenic avian influenza A (H5N1) virus infections on fur farms connected to mass mortalities of black-headed gulls, Finland, July to October 2023. Eurosurveillance 29(25), 2400063. Khan, M., Chaudhry, M., Fatima, Z., Khan, R.U., Ahmad, B., Ullah, R. and Khan, A. 2021. Effect of avian influenza H9N2 subtype virus infection on backyard poultry production. Sci. Lett. 9, 19–23. Kibenge F. S. B. 2023. A One Health approach to mitigate the impact of influenza A virus (IAV) reverse zoonosis is by vaccinating humans and susceptible farmed and pet animals. Am. J. Vet. Res. 84(6), ajvr.23.03.0053. doi: 10.2460/ajvr.23.03.0053 Kode, A., Manavalan, J.S., Mosialou, I., Bhagat, G., Rathinam, C.V., Luo, N., Khiabanian, H., Lee, A., Murty, V.V., Friedman, R., Brum, A., Park, D., Galili, N., Mukherjee, S., Teruya-Feldstein, J., Raza, A., Rabadan, R., Berman, E. and Kousteni, S. 2014. Leukaemogenesis induced by an activating β-catenin mutation in osteoblasts. Nature 506(7487), 240–244. Korteweg, C. and Gu, J. 2008. Pathology, molecular biology, and pathogenesis of avian influenza A (H5N1) infection in humans. Am. J. Pathol. 172(5), 1155–1170. Leyrat, C., Renner, M., Harlos, K., Huiskonen, J. T., and Grimes, J. M. 2014. Drastic changes in conformational dynamics of the antiterminator M2-1 regulate transcription efficiency in Pneumovirinae. Elife, 3, e02674. doi: 10.7554/eLife.02674 Li K South., Guan, Y., Wang, J., Smith, G.J.D., Xu, K.M., Duan, L. and Peiris J South. 2004. Genesis of a highly pathogenic and potentially pandemic H5N1 influenza virus in eastern Asia. Nature 430(6996), 209–213. Lieber, C.M., Aggarwal, M., Yoon, J.J., Cox, R.M., Kang, H.J., Sourimant, J., Toots, M., Johnson, S.K., Jones, C.A., Sticher, Z.M., Kolykhalov, A.A., Saindane, M.T., Tompkins, S.M., Planz, O., Painter, G.R., Natchus, M.G., Sakamoto, K. and Plemper, R.K. 2023. 4’-Fluorouridine mitigates lethal infection with pandemic human and highly pathogenic avian influenza viruses. PLos Pathogens 19(4), e1011342; doi:10.1371/journal.pone.00101342 Lum D Lee., Chua JJ Tan., Lee X Liu. and Ng, L. 2018. Zika virus infection preferentially counterbalances human peripheral monocyte and/or NK cell activity. MSphere 3(2), 10–128. Ma, W., Kahn R East. and Richt, J.A. 2008. The pig as a mixing vessel for influenza viruses: human and veterinary implications. J. Mol. Genet. Med. 3, 158. Machalaba J Raufman., Anyamba AM Berrian., Berthe GC Gray. and Weiss, L. 2021. Applying a one health approach in global health and medicine: enhancing involvement of medical schools and global health centers. Ann. Global Health 87(1), 30; doi:10.1016/j.agh.2016.09.010 Marchenko, V.Y., Svyatchenko, S.V., Onkhonova, G.S., Goncharova, N.I., Ryzhikov, A.B., Maksyutov, R.A. and Gavrilova, E.V. 2023. Review on the epizootiological situation on highly pathogenic avian influenza around the world and in Russia in 2022. Md-T, I., Roy, S., Talukdar, H., Shammi, S.A. and Ahmed, S. 2024. Sero-prevalence and associated risk factors of avian influenza virus infection in backyard chickens in the Sylhet region of Bangladesh. Int. J. Vet. Sci. 13(3), 362–368; doi:10.47278/journal.ijvs/2023.106 Monne, I. and Capua, I. 2014. AVI outbreaks in poultry and wild birds. Vet. Microbiol. 174(3–4), 163–174; doi:10.1016/j.vetmic.2014.09.021 Montalvo-Corral M Reyes., -Leyva, J. and Hernandez, J. 2010. Avian influenza: eco-epidemiological aspects of the virus in its natural hosts, the migratory waterfowls. Revista Chilena De Historia Natural 83(4), 543–556. Naeem, K. and Siddique, N. 2006. Strategic vaccination for avian influenza control in Pakistan. Develop. Biologicals 124, 145–150. Olsen, B., Munster, V.J., Wallensten, A., Waldenstro., m, J., Osterhaus, A.D. and Fouchier, R.A. 2006. Global patterns of influenza A virus in wild birds. Science 312(5772), 384–388. Otte, J., Hinrichs, J., Rushton, J., Roland-Holst, D. and Zilberman, D. 2009. Impacts of avian influenza virus on animal production in developing countries. CABI. Rev. 2008, 1–18. Pedersen, J.T., Ostergaard, J., Houen, G. and Heegaard, N.H. 2008. Affinity capillary electrophoresis for identification and investigation of human Gc-globulin (vitamin D-binding protein) and its isoforms interacting with G-actin. Electrophoresis 29(8), 1723–1733. Peiris, J.S., De Jong, M.D. and Guan, Y. 2007. Avian influenza virus (H5N1): a threat to human health. Clin. Microbiol. Rev. 20, 243–267. Perry, B.D. and Sones, K.R. 2007. Global roadmap for improving the tools to control foot and mouth disease in endangered settings. Pinto, R.M., Silva, M.D., Pastrana, L.M., Bañobre-López, M. and Sillankorva, S. 2022. This chapter was based on the following scientific articles. Maria Daniela Ferreira da Silva, 45, 1. Pulit-Penaloza, J.A., Jones, J., Sun, X., Jang, Y., Thor, S., Belser, J. and Davis, C.T. 2018. Antigenically diverse swine origin H1N1 variant influenza viruses exhibit differential ferret pathogenesis and transmission phenotypes. J. Virol. 92(11), 10–1128. Qi, X., An, X., Jiao, Y., Yu, H., Xu, K., Cui, L., Wang, S., Deng, F., Huo, X., Huang, H., Dai, Q. and Bao, C. 2018. Co-circulation of multiple genotypes of influenza A (H7N9) viruses in eastern China, 2016-2017. Arch. Virol. 163(7), 1779–1793; doi:10.1016/j.arv.2018.09.010 Rehman FA Rantam., Batool A Shehzad., Effendi AM Witaningrum. and Purnama, M.T. 2022. Emerging threats and vaccination strategies of H9N2 viruses in poultry in Indonesia: a review. F1000Research 11, 548; doi:10.1016/j.f1000research.2011.11.548 Richard, M., Herfst, S., Van Den Brand, J.M., Lexmond, P., Bestebroer, T.M., Rimmelzwaan, G.F., Koopmans, M., Kuiken, T. and Fouchier, R.A. 2010. Low virulence and lack of airborne transmission of the Dutch highly pathogenic avian influenza virus H5N8 in ferrets. PLos One. 10(6), e0129827. Rimondi, A., Vanstreels, R.E., Olivera, V., Donini, A., Lauriente, M.M. and Uhart, M.M. 2024. Highly pathogenic avian influenza A (H5N1) viruses from multispecies outbreak. Emerg. Infect. Dis. 30(4), 812–814; doi:10.3201/eid3004.231723 Rouse, B.T. and South, S. 2010. Immunity and immunopathology to viruses: lessons from infectious diseases. Nature. Rev. Immunol. 10(8), 754–765; doi:10.1038/nri2844 Rehman, S., Rantam, F. A., Batool, K., Shehzad, A., Effendi, M. H., Witaningrum, A. M. and Purnama, M. T. E. 2022. Emerging threats and vaccination strategies of H9N2 viruses in poultry in Indonesia: a review. F1000Research, 11, 548. Rehman, S., Effendi, M.H., Witaningruma, A.M., Nnabuikeb, U.E., Bilal, M., Abbas, A. and Hussain, K. 2023. Avian influenza (H5N1) virus, epidemiology and its effects on backyard poultry in Indonesia: a review. F1000Research 11, 1321. Doi: 10.1016/j.f1000research.2013.11.1321 Short, K.R., Kedzierska, K. and Van De Sandt, C. 2018. Back to the future: lessons learned from the 1918 influenza pandemic. Front. Cell. Infect. Microbiol. 8, 343. Sims, L., Domenech, J., Benigno, C., Kahn, S., Kamata, A., Lubroth, J. and Roeder, P. 2005. Origin and evolution of highly pathogenic H5N1 avian influenza in Asia. Vet. Rec. 157(6), 159–164. Suardana, I.B.K., Widyastuti, S.K., Pradnyadana, I.B.K. and Agustina, K.K. 2023. Effect of age and presence of maternal antibodies on the success of avian influenza and Newcastle disease vaccinations in broiler chickens. Int. J. Vet. Sci. 12(1), 101–106; doi:10.47278/journal.ijvs/2022.165 Swayne D East. and Suarez, D.L. 2000. Highly pathogenic avian influenza. Revue Scientifique Et Technique-office. Int. Des Epizooties 19(2), 463–475. Taubenberger, J.K. and Morens, D.M. 2010. Pandemic influenza: 2009 H1N1 and beyond. Nature Rev. Microbiol. 8(4), 407–415; doi:10.1038/nrmicro2400 The Yuan, R., Liang, L., Wu, J., Kang, Y., Song, Y. and Zou L Ke. 2017. Human infection with an avian influenza A/H9N2 virus in Guangdong in 2016. J. Infect. 74, 422–425. Tiensin P Chaitaweesub., Songserm A Chaisingh., Hoonsuwan C Buranathai. and Thanawongnuwech, R. 2005. Highly pathogenic avian influenza H5N1 (H5N1), 2004. Emerg. Infect. Dis. 11(11), 1664–1672; doi:10.3201/eid1111.050608 Vaziri, E., Maghsoudi, A., Feizabadi, M., Faraji-Arough, H. and Rokouei, M. 2022. Scientometric evaluation of 100-year history of Poultry Science (1921–2020). Poultry. Sci. 101, 102134. Ventana, G.A.Y., Suartha, I.N., Juliantari, P.T., Tama, K.T., Kendran, A.A.S., Sari, T.K., Agustina, K.K. and Tenaya, I.W.M. 2023. The safety and potential of avian influenza H5N1 isolated from Bali as a new vaccine candidate. Int. J. Vet. Sci. 12(6), 786–792; doi:10.1016/j.jwm.2023 Webby, R.J., Rossow, K., Erickson, G., Sims, Y. and Webster, R. 2004. Multiple lineages of antigenically and genetically diverse influenza A virus co-circulate in the United States swine population. Virus. Res. 103(1-2), 67–73. Webster, R.G. and Govorkova, E.A. 2006. H5N1 influenza—continuing evolution and spread. New England J. Med. 355(21), 2174–2177; doi:10.1056/NEJMp068205 World Health Organization (WHO). 2012. Cumulative number of confirmed human cases of avian influenza A (H5N1) reported to the World Health Organization. Geneva: WHO. World Health Organization. 2022. Integrated sentinel surveillance of influenza and SARS-CoV-2 and the development of the Global Influenza Surveillance and Response System Plus: virtual meeting, 12–14 October 2021. Geneva: WHO. Xing, Z., Cardona, C.J., Li, J., Dao, N., Tran, T. and Andrada, J. 2008. Modulation of the immune responses in chickens by low-pathogenicity avian influenza virus H9N2. J. Gen. Virol. 89(Pt 5), 1288–1299; doi:10.1099/vir.0.83362-0 Ye Tian., Kai Xu. and Ansari, N. 2005. TCP in wireless environments: problems and solutions. IEEE. Commun. Mag. 43(3), S27–S32. Zhao, G.Z., Li, B., Wang, Y.F., South, G., Du, Y., Ma, Q.X. and Liu. 2022. Detuning injection versus neuraminidase inhibitors in the treatment of influenza: a systematic review and meta-analysis. Chin. J. Integr. Med. 28(11), 1023–1031. | ||
| How to Cite this Article |
| Pubmed Style Rehman S, Ullah S, Effendi MH, Budiastuti B, Hamid IS, Rahman AU, Zaman A, Malik MIU, Rustam SA, Ullah S, Saleem Z. Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Vet. J.. 2026; 16(1): 1-14. doi:10.5455/OVJ.2026.v16.i1.1 Web Style Rehman S, Ullah S, Effendi MH, Budiastuti B, Hamid IS, Rahman AU, Zaman A, Malik MIU, Rustam SA, Ullah S, Saleem Z. Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. https://www.openveterinaryjournal.com/?mno=252720 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.1 AMA (American Medical Association) Style Rehman S, Ullah S, Effendi MH, Budiastuti B, Hamid IS, Rahman AU, Zaman A, Malik MIU, Rustam SA, Ullah S, Saleem Z. Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Vet. J.. 2026; 16(1): 1-14. doi:10.5455/OVJ.2026.v16.i1.1 Vancouver/ICMJE Style Rehman S, Ullah S, Effendi MH, Budiastuti B, Hamid IS, Rahman AU, Zaman A, Malik MIU, Rustam SA, Ullah S, Saleem Z. Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 1-14. doi:10.5455/OVJ.2026.v16.i1.1 Harvard Style Rehman, S., Ullah, . S., Effendi, . M. H., Budiastuti, . B., Hamid, . I. S., Rahman, . A. U., Zaman, . A., Malik, . M. I. U., Rustam, . S. A., Ullah, . S. & Saleem, . Z. (2026) Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Vet. J., 16 (1), 1-14. doi:10.5455/OVJ.2026.v16.i1.1 Turabian Style Rehman, Saifur, Shakeeb Ullah, Mustofa Helmi Effendi, Budiastuti Budiastuti, Iwan Sahrial Hamid, Atta Ur Rahman, Ali Zaman, Muhammad Inam Ullah Malik, Saqib Ali Rustam, Sana Ullah, and Zulqarnain Saleem. 2026. Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Veterinary Journal, 16 (1), 1-14. doi:10.5455/OVJ.2026.v16.i1.1 Chicago Style Rehman, Saifur, Shakeeb Ullah, Mustofa Helmi Effendi, Budiastuti Budiastuti, Iwan Sahrial Hamid, Atta Ur Rahman, Ali Zaman, Muhammad Inam Ullah Malik, Saqib Ali Rustam, Sana Ullah, and Zulqarnain Saleem. "Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation." Open Veterinary Journal 16 (2026), 1-14. doi:10.5455/OVJ.2026.v16.i1.1 MLA (The Modern Language Association) Style Rehman, Saifur, Shakeeb Ullah, Mustofa Helmi Effendi, Budiastuti Budiastuti, Iwan Sahrial Hamid, Atta Ur Rahman, Ali Zaman, Muhammad Inam Ullah Malik, Saqib Ali Rustam, Sana Ullah, and Zulqarnain Saleem. "Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation." Open Veterinary Journal 16.1 (2026), 1-14. Print. doi:10.5455/OVJ.2026.v16.i1.1 APA (American Psychological Association) Style Rehman, S., Ullah, . S., Effendi, . M. H., Budiastuti, . B., Hamid, . I. S., Rahman, . A. U., Zaman, . A., Malik, . M. I. U., Rustam, . S. A., Ullah, . S. & Saleem, . Z. (2026) Avian influenza virus H5N1 and H9N2 subtypes in different birds and humans: Findings from an extensive evaluation. Open Veterinary Journal, 16 (1), 1-14. doi:10.5455/OVJ.2026.v16.i1.1 |