| Research Article | ||
Open Vet. J.. 2025; 15(10): 5192-5201 Open Veterinary Journal, (2025), Vol. 15(10): 5192-5201 Research Article Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattleAlexander Kaka1, Aulia Puspita Anugra Yekti2, Habib Assidiq Syah1, Putri Utami1, Sucik Maylinda3, Sri Rahayu2 and Trinil Susilawati2*1Doctoral Student, Faculty of Animal Science, Universitas Brawijaya, Malang, Indonesia 2Faculty of Animal Science, Universitas Brawijaya, Malang, Indonesia 3Faculty of Mathematics and Natural Sciences, Universitas Brawijaya, Malang, Indonesia *Corresponding Author: Trinil Susilawati. Faculty of Animal Science, Universitas Brawijaya, Malang 65145, East Java, Indonesia. Email: tsusilawati [at] ub.ac.id Submitted: 08/04/2025 Revised: 30/08/2025 Accepted: 22/09/2025 Published: 31/10/2025 © 2025 Open Veterinary Journal
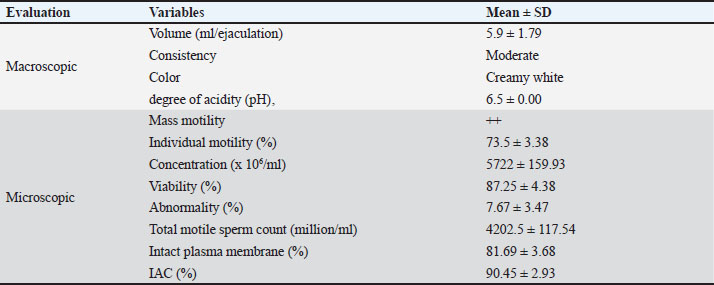
AbstractBackground: Sumba Ongole (SO) cattle, a valuable local breed in Indonesia known for its adaptability and resilience, face challenges in productivity and genetic conservation. Technological innovations, such as artificial insemination (AI), to enhance genetic quality and ensure the sustainability of this local germplasm during the growing dominance of exotic cattle. Aim: This study aimed to investigate the use of local diluent materials [Asian palmyra palm (APP)] to maintain the quality of liquid semen in SO cattle. Methods: Four Class I male cattle were selected according to the Indonesian National Standard (2014) on the basis of qualitative and quantitative characteristics. A completely randomized design was employed, consisting of 10 replications with four treatments: T0: cauda epididymal plasma-3 (CEP-3) diluent + 20% egg yolk (EY) (control); T1: 85% APP + 15% EY; T2: 80% APP + 20% EY; and T3: 75% APP + 25% EY. Results: Various levels and combinations of APP extract and EY significantly differed (p < 0.01) between treatments, particularly on day 1, which tended to positively influence motility. Viability displayed a comparable pattern, with significant differences noted (p < 0.01). Conclusion: The combination of 75% APP and 25% EY was recommended as an effective diluent for maintaining the quality of liquid semen in SO cattle. Keywords: Asian palmyra palm, Egg yolk, Diluent, Sperm quality, Sumba Ongole cattle. IntroductionThe Sumba Ongole cattle (Bos indicus), originally known as Nelore from Andhra Pradesh, India, were introduced to Indonesia via trade routes by Indian merchants in 1914. Following adaptation to the local environment, this breed was established as Sumba Ongole (SO) cattle. Despite the vast territory of Indonesia, SO cattle exhibit optimal development exclusively on Sumba Island, East Nusa Tenggara (Kaka et al., 2025). As an indigenous breed, SO cattle possess significant adaptive advantages, including heat stress tolerance, low-quality forage survival capacity, and parasite and infectious disease resistance (Bakae et al., 2022; Abdullah and Meng, 2024). Beyond its role in supporting food security, the development of SO cattle is vital for environmental sustainability and preservation of local cultural practices, particularly in arid regions like Sumba Island (Gaina et al., 2019). As an indigenous livestock genetic resource, SO cattle have been formally designated as protected germplasm requiring conservation under Indonesian Minister of Agriculture Decree No. 427/Kpts/SR.120/3/2014. However, the genetic actantial of SO cattle for enhancing local livestock productivity and enriching national genetic diversity remains underutilized (Rojas-Downing et al., 2017). Population growth disproportionate to its genetic advantages reflects developmental challenges, primarily due to geographical constraints and low adoption of technological innovation. This condition has resulted in suboptimal development rates and diminished SO breed productivity. A strategic approach to conserve and enhance the genetic quality of SO cattle involves the implementation of reproductive technologies, specifically artificial insemination (AI) (Utami et al., 2022; Li et al., 2025). AI technology has demonstrated efficacy in improving reproductive efficiency while enhancing both the quality and quantity of livestock production (Zuidema et al., 2021; Syah et al., 2024). However, in remote regions, AI implementation using frozen semen faces operational constraints, particularly due to limited cryostorage and distribution infrastructure (Temesgen et al., 2021). The use of liquid semen from superior sires meeting RSNI4 7651.8:2016 standards presents a more practical and cost-effective solution (Kaka et al., 2025). Liquid semen offers the advantages of ease of application, cost efficiency, and elimination of cryogenic nitrogen requirements (Susilawati et al., 2020). It also yields more doses per ejaculate, thereby enhancing the genetic utilization efficiency of elite sires (Murphy et al., 2017). However, a primary limitation of liquid semen is its short shelf life, necessitating high-quality extenders to maintain spermatozoa viability and motility until AI (Al-Bulushi et al., 2019). Currently, commercial extenders remain frequently inaccessible and cost-prohibitive for breeders in remote areas (Susilawati et al., 2020). Consequently, research on natural-based extenders continues to advance. Alternatives include modified CEP-3 extenders supplemented with soybean extract (Ratnawati et al., 2023; Ratnawati et al., 2025), butterfly pea flower (Clitoria ternatea) extract (Mariana et al., 2025), and tris-yolk extenders enhanced with palmyra palm (Ina and Kaka, 2020) or moringa leaf (Moringa oleifera) extract (Authaida et al., 2025). Recent studies have also reported the use of palmyra fruit water combined with citrated egg yolk (EY) (Hine et al., 2024). One of the natural diluents, not much reported, is EY and Asian palmyra (Borassus flabellifer L.). Therefore, local diluents, such as Asian palmyra palm (APP), are expected to be more affordable and easily available alternatives. Owing to its rich nutritional content and natural antioxidant properties, APP can sunport the quality of sperm, thereby increasing AI efficiency and success. Based on the results of the chemical composition analysis, APP has the advantage of containing a total sugar level of 13.79%, with 2.48% sucrose, 4.77% fructose, 0.13% vitamin C, and 1.37 mg/l antioxidants. According to Subramanian et al. (2024), flavonoids are the main antioxidants found in APP. The addition of EY to APP can create a more complete diluent to sunport the life of sperm during liquid semen storage. EY plays an important role in protecting sperm from cellular damage and maintaining the stability of the diluent composition, thus affecting the increase in conception rates (Bustani and Baiee, 2021; Saad et al., 2022). This combination can be an innovation in the development of economical and easy-to-receive local diluents, especially in areas with limited access to commercial diluents. This study aimed to determine the effectiveness of utilizing APP to preserve the quality of liquid semen from SO cattle. Materials and MethodsAnimalsThis study used four class one SO cattle breeding males selected based on the SNI RSNI4 7651.8:2016 guidelines. The male participants ranged in age from 3 to 5 years, with a body weight of 372–525 kg, an average shoulder height of 147.25 cm, a body length of 147.25 cm, a chest circumference of 180.50 cm, and a scrotum circumference of 28.75 cm. Semen was collected twice per week, ensuring a minimum motility of 70% (Table 1). Table 1. Assessment of fresh semen from Sumba Ongole bulls.
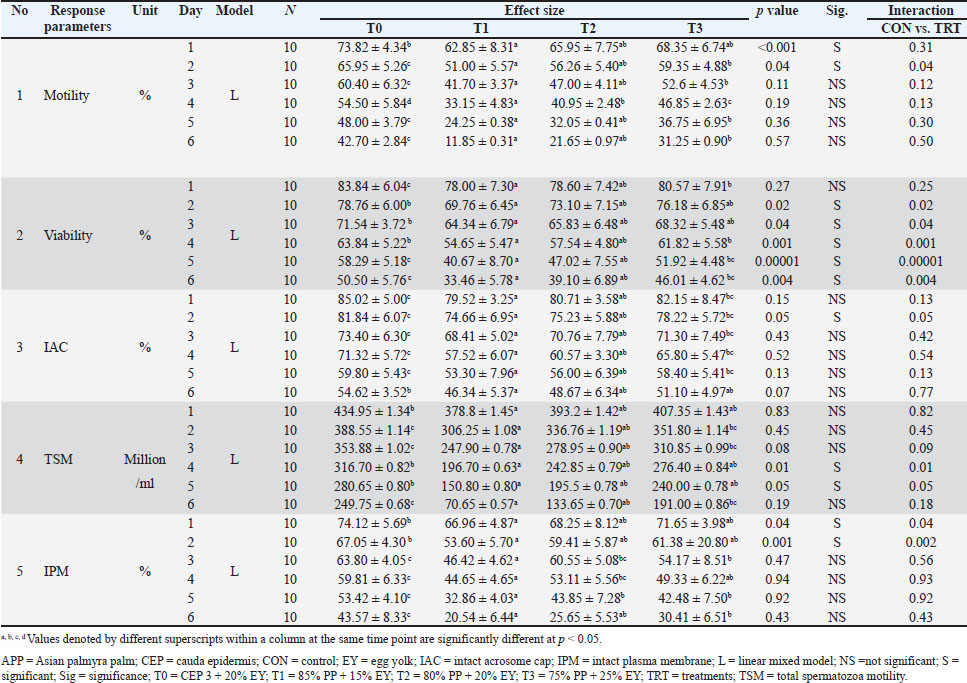
Experiment designThis study was a laboratory experiment conducted using a CRD consisting of 10 replications and four treatments. Each ejaculate collected was evenly divided among all treatment groups using the split-sample method to minimize individual variability. The treatments tested were as follows: T0: 80% CEP-3 diluent + 20% EY (control); T1: 85% APP + 15% EY; T2: 80% APP + 20% EY; and T3: 75% APP + 25% EY. Preparation of the diluent materialsCEP-3 diluent consisted of the following: fructose, 55.0 mmol/l; sorbitol, 1.0 mmol/l; citric acid, 42.6 mmol/l; NaCl, 15 mmol/l; KCl, 7.0 mmol/l; CaCl2(H2O), 23.9 mmol/l; MgCl2(H2O)2, 3.0 mmol/l; NaHCO3, 11, 9 mmol/l; NaH2PO, 48.0 mmol/l; KH2PO4, 20.0 mmol/l; Tris, 33.7 mmol/l; gentamicin, 0.05 g/l; penicillin, 1000 IU; streptomycin, 1 g; egg white, 4 ml/l; and Osm, 250–350 mOsm. Materials were ionized and homogenized using a magnetic stirrer (Susilawati et al., 2016). The pH was adjusted to a pH of 6–7, and the osmolarity was 250–350 mOsm (Ducha et al., 2012). The medium was then filtered through a Millipore membrane with a pore size of 0.22 µm. The procedure removed the egg white by sucking it with a pipette at a concentration as high as 0.4% or 4 ml/l. The medium was supplemented with 20% EY and centrifuged at 252 × g twice for 30 minutes, followed by collecting the supernatant. Young APPs were selected based on the following criteria: exocarp predominantly green (±70% surface area) with dark brown patches (±30%). The prepared APP mixture was cut with a sterile machete until the shield on each eye of the fruit was visible. The water from the palm fruit was subsequently removed with a sterile syringe and placed into a measuring cup until it reached 80 ml. The steps used to prepare APP for use as a semen diluent include the following steps: 1). APP was preinactivated at 56°C for 20 minutes; 2). NaHCO3 (buffer) was added until the pH reached 7. Antibiotics, such as 1,000 IU penicillin and 1 g streptomycin, were added to prevent germ growth, and the mixture was covered with aluminum foil paper. 3). The EY was added to the APP mixture according to the treatment and homogenized with a magnetic stirrer for 10–15 minutes. Subsequently, the mixture was centrifuged at 252 × g 3 times for 30 minutes, the supernatant was collected, and the EY mixture was ready for use. Research parametersIndividual motility (%)The percentage of individual motility of progressively moving sperm was assessed by removing one drop of semen, dropping it on a glass object covered with a cover glass, and observing it under a microscope at 400× magnification. The number of sperm that moved backward and circularly was not counted (Rathod et al., 2022; Yekti et al., 2024). Viability (%)Viability observations were made by placing one drop of semen on an object glass and then adding eosin-negrosin solution. The mixture was then homogenized, and a thin and dried screw preparation was made and observed using a 400× magnification microscope without cover glass. Sperm are considered alive if the head is white or does not absorb color, and dead sperm are considered alive if the sperm head is red. The viability percentage was determined by dividing the total number of live sperm by the total number of sperm counted and then multiplying the result by 100% (Susilawati, 2013). Total sperm motility (million/ml)The optimal number of progressive motile sperm in an ejaculate in liquid semen is used to sunport cattle fertility. Total motile sperm (TSM) concentration was calculated using the following formula: Total sperm motile (million/ml)=Sperm concentration (million/ml) × Sperm motility (%) (Nalley et al., 2019). Intact plasma membrane (IPM) observationIPM assessment can be performed using the hypoosmotic swelling test. The test was performed using 1 ml of 150 ml of osmol hypoosmotic solution plus 0.1 ml of sperm, followed by incubation in a water bath at 37°C for 30 minutes. The incubation results were taken with one drop of sperm sample with an Ose dripped on a glass object and reviewed via the tip of another glass object, without being covered with a cover glass, and observed via a light microscope at a magnification of 400×. The end of the tail is circular in sperm with an IPM, whereas the plasma membrane is damaged in sperm with a straight tail (Nalley et al., 2019; Susilawati et al., 2022). Intact acrosome cap (IAC) observationThe intact acrosome hood observation process can be performed by placing 10 µl of semen and 30 µl of formalin solution (1:30) into a microtube and homogenizing by vortexing two or more times. The semen added to the solution was incubated in a water bath at 37°C for 30 minutes. The resulting semen mixture was dripped on a glass object with one or more drops and covered with a covered glass. The IACs were observed under a microscope at 400× magnification. The anterior portion of the head is darker than the posterior portion or has a black tint at the tip, and the IAC is destroyed in sperm with intact acrosome hoods. There is no black coloration on the sperm head. Observations can be made in five different fields of view with a minimum of 200 sperm, which can be counted using a hand-tally counter. The IAC is determined by dividing the number of sperm with ACs by the total number of sperm counted, and then calculating the result as a percentage of 100% (Susilawati, 2013). Statistical analysis and bioinformatics analysisThe analysis and coding of all the data were executed using R software version 4.3.3 (Chan, 2018) – “Ocean Storm” x86_64-W64-ming32/X64 computing, incorporating the libraries “readxl,” “xlsx,” “tidyverse,” “multicomp,” and “nlme” (Pinheiro et al., 2018). The modeling approach followed the methodology outlined by Sholikin et al. (2023). The statistical model used is as follows: Yijk=μ + Si+ τj + Sτij + β1Xij + biXij + β2X2ij + biX2ij + eijk where Yijk=dependent variable, μ=overall mean value, Si=random effect of the ith study, assumed to be ~Niid(0,σs2), τj=fixed effect of the jth of τ factors, Sτij=random interaction between the ith and jth levels of the τ factor, also assumed to be ~Niid(0,σs2τ), β1=overall value of the linear regression coefficient of Y–X (a fixed effect), β2=overall coefficient value of the quadratic regression of Y–X (a fixed effect), Xij dan X2ij=continuous values of the predictor variable (in linear and quadratic form, respectively), bi=random effect of the study on the regression coefficient of Y–X, assumed to be ~Niid(0,σb2), and eijk=residual value from the unpredictable error. Sτij and Si are taken as independent variables that are randomly chosen. Validation and significance tests were conducted on the model. The significance of the values was determined using one-way analysis of variance. A p value (P or p value) < 0.05 tends to be significant if the p value is between 0.05 and 0.1 according to a post hoc test. As a result, Pl represents the p value of the linear constant (β1), and Pq represents the p value of the quadratic constant (β2). Therefore, the validation test was conducted using the RMSE and Nakagawa determination coefficient (R2) or RGLMN(c)2 Note: 0=actual value, P=estimated value, NDP=number of data points, σ2f is the variant of a fixed factor, Ʃ(σ21)is the sum of all variants of the component, σ2e is the variant due to predictor dispersion, and σ2d is the specific distribution of the variant. T0: CEP-3 diluent + 20% EY (control); T1: 85% APP + 15% EY; T2: 80% APP + 20% EY; and T3: 75% APP + 25% EY. Ethical approvalWith ethical approval number 090-KEP-UB-2024, this study complied with the Animal Care and Use Committee of Universitas Brawijaya, Malang, East Java, Indonesia. ResultsTable 1 shows the results of the macroscopic and microscopic evaluation of the quality of fresh semen from SO cattle. The average volume of fresh semen from the SO cattle in this study was 5.90 ± 1.79 ml/ejaculation, with a concentration of 5722.00 ± 159.93 × 106/ml. Average fresh semen mass motility of SO cattle (++), with an average individual motility of 73.50% ± 3.38%. The fresh semen of SO cattle had a viability of 87.25% ± 4.38%, a total number of motile sperm of 4202.5% ± 117.54%, an abnormality of 7.67% ± 3.47%, an IPM of 81.69% ± 3.68%, and an intact acrosomal cap of 90.45% ± 2.93%. The fresh semen collected was moderately consistent, had a creamy white color, and had a pH of 6.5. Overall, the quality of fresh semen from SO cattle was excellent, with volume, consistency, color, and semen pH within the normal range. In contrast, positive results were observed for motility, viability, total motile sperm, IPM, and acrosomal cap sperm. These findings indicate that fresh semen from SO cattle can be processed further and has high actantial for use in artificial intelligence (AI) programs and germplasm conservation. The results revealed that various levels and combinations of APP and EY significantly differed (p < 0.01) between treatments, particularly on Day 1, which tended to positively influence motility on the second day (p=0.04). The control group consistently demonstrated superior motility compared with the treatment groups. Viability exhibited a similar trend, with significant differences observed (p < 0.01) up to days 4 and 5. Similarly, the results for TSM indicated significant differences on the fourth and fifth days (p < 0.01 and p < 0.05, respectively). Sperm motility is an important parameter for determining the success of the storage process and AI. The findings of this study demonstrated that the use of CEP-3+20% EY diluent (T0; control) resulted in the highest percentage of motility on the first day, which was 73.82% ± 4.34%. This finding indicates that this standard diluent effectively maintains sperm motility during the early storage phase. This treatment can even maintain sperm motility until day 6, indicating that the combination of CEP-3 and 20% EY has optimal durability for a more extended period. The other treatments, 75% APP + 25% EY (T3), 80% APP + 20% EY (T2), and 85% APP + 15% EY (T1), resulted in a faster decline in motility than did T0. T3 maintained sperm motility until day 4, with an initial motility percentage of 68.35% ± 6.74%. The excellent sperm motility standard for AI is at least 40%. The results of this study indicate that CEP-3 diluent + 20% EY (T0) is the best choice because it can maintain the percentage of motility above the minimum threshold for AI for 6 days. Moreover, treatment with the APP mixture at T1, T2, and T3 maintained motility for only 3–4 days, with T3 resulting in the best results. In this study, the viability of fresh semen from SO cattle tended to decrease in all treatment groups during storage. On the first day, all treatments had good viability values (>70%). T0 had the highest viability value of 83.84% ± 6.04%, followed by T3 at 80.57% ± 7.91%, T2 at 78.60% ± 7.42% and T1 at 78.00% ± 7.30%. Viability values in the study revealed different lengths of survival. T0 was able to maintain a good viability value until day 3 (71.54% ± 3.72%), followed by T3 until day 2 (76.18% ± 6.85%), T2 until day 2 (73.10% ± 7.15%), and T1 only on the first day (78.00% ± 7.30%). The IAC of fresh semen from the SO cattle in this study tended to decrease during storage in all treatments. T0 had the highest IAC of all the treatments during the 6 days of storage, which was 85.02% ± 5.00% on day 1, 81.84% ± 6.07% on day 2, 73.40% ± 6.30% on day 3, 71.32% ± 5.72% on day 4, 59.80% ± 5.43% on day 5 and 54.62% ± 3.52% on day 6. Moreover, T1 had the lowest IAC of all the treatments after 6 days of storage, namely, 79.52% ± 3.25% on day 1, 74.66% ± 6.95% on day 2, 68.41% ± 5.02% on day 3, 57.52% ± 6.07% on day 4, 53.30% ± 7.96% on day 5 and 46.34% ± 5.37% on day 6. The study results revealed that T0 presented the highest TSM value from the first storage day to day 6. On the first day, the TSM value at T0 was 434.95 ± 1.34 (106/ml), followed by T3 at 407.35 ± 1.43 (106/ml), T2 at 393.2 ± 1.42 (106/ml), and T1 at 378.8 ± 1.45 (106/ml). In contrast, the TSM value on day 6 in T0 was 249.75 ± 0.68 (106/ml), followed by T3 at 191.00 ± 0.86 (106/ml), T2 at 133.65 ± 0.70 (106/ml), and T1 at 70.65 ± 0.57 (106/ml). The results revealed that the percentage of IPM from day 1–5 was highest in T0, with a first-day IPM percentage of 74.12% ± 5.69%, followed by T3 at 71.65% ± 3.98%, T2 at 68.25% ± 8.12%, and T1 at 66.96% ± 4.87%. T0 maintained plasma membrane integrity until day 3, whereas T3 only lasted until day 2; T2 and T1 could only maintain IPM above the standard value of 60% on the first day of storage. DiscussionMotility is a key parameter that represents sperm fertilization potential, membrane integrity, and viability (Ratnawati et al., 2023; Utami et al., 2025). In this study, the use of 80% CEP-3 + 20% EY (T0) as a control had the best results, followed by the use of 75% APP + 25% EY (T3), 80% APP + 20% EY (T2), and 85% APP + 15% EY (T1). As shown in Table 2, T0 had motility <40% and viability <40% until day 6, whereas in T1, T2, and T3, motility <40% only survived on days 3–4, and viability <40% survived until days 5–6. In this study, motility and viability gradually decreased until day 6. Although the use of APP as a diluent results in lower results than the control treatment (T0), the use of APP still results in good motility and viability, and the use of APP as a substitute for CEP-3 can be categorized as feasible because motility <40% and viability <60% can survive for more than 3 days (Christensen et al., 2011; Bustani and Baiee, 2021). A decrease in motility and viability occurs gradually during the storage process (days 1–6) due to the reduction in energy sources required for sperm metabolism and the effects of reactive oxygen species-induced lipid peroxidation, which damage the sperm membrane (Ratnawati et al., 2017). In this study, the 85% APP and 15% EY diluents had the lowest motility and viability, possibly due to the diluent’s low EY content. According to Peruma (2018), EY is an essential diluent for various mammalian species during prolonged semen storage to preserve sperm from cold shock. The warmth of the EY can shield the acrosome and plasma membrane from damage (Amirat-Briand et al., 2010). This is due to the presence of cholesterol, low-density lipoproteins (LDL), and phospholipids in EY, which protect sperm from cold shock during the thawing process (Kulaksiz et al., 2010). Table 2. Effects of APP (Siwalan) on the quality of Sumba Ongole liquid sperm across varying days.
The integrity of the acrosome hood affects the capacitation process and acrosome reaction during fertilization (Yekti et al., 2023). The storage environment, including the nutrient and antioxidant contents of the diluent, strongly influences the integrity of the sperm acrosome hood (Zhang et al., 2023). The results showed that the T3 treatment maintained the integrity of the acrosome hood until day 3, which was close to the results of the T0 control diluent. Palm water is rich in bioactive compounds such as flavonoids, vitamin C, tannins, and other phenolic compounds. These substances, which include ferric antioxidant-reducing ability, hydroxyl radical trapping activity, and 2,2-diphenyl-1-picrylhydrazyl (DPPH), have intense antioxidant activity (Thi Le et al., 2021). These antioxidants protect the sperm membrane, including the acrosome hood, from oxidative stress during storage (Jakop et al., 2023). Oxidative stress can cause sperm membrane lipid peroxidation, ultimately damaging the acrosome hood structure (Peris-Frau et al., 2020). This sunport the statement by Kulaksız et al. (2010) and Upadhyay et al. (2023) that antioxidant supplementation in semen diluents is the most widely used strategy for enhancing sperm quality. The T3 composition provided optimal protection due to the correct ratio of APP to EY. A greater percentage of EY contains phospholipids and proteins that serve as membrane-stabilizing agents than T2 and T1. EY also contains components such as lecithin and cholesterol, which help strengthen the membrane structure and maintain its fluidity, thereby ensuring that the acrosome hood remains intact during storage (Abdel-Aziz Swelum et al., 2019; Ďuračka et al., 2024). This composition balance the antioxidant benefits of palm water and the physical protection of EY. If the proportion of APP is too high, as in T2 and T1, the protection provided by the EY is reduced, thus decreasing the diluent’s effectiveness in maintaining the acrosome hood. The low percentage of intact acrosome hoods is related to the low percentage of motility, viability, and IPM (Yekti et al., 2023). Physiologically, a relationship exists between motility, acrosome hood integrity, and sperm survival (Zhang et al., 2021). Damage to the acrosome results in the loss of enzymes (Aitken et al., 2015). Plasma membrane integrity is an important indicator of sperm viability. An intact membrane allows ion exchange and nutrient transportation and maintains the cell’s internal environment stability (Wurlina et al., 2020). The best plasma membrane integrity was found at T0, which maintained a value above the minimum standard of 60% until day 5. In contrast, the plants in the T3 and T2 treatments survived until only day 3. The optimal performance of T0 in maintaining spermatozoa plasma membrane integrity could be due to the phospholipids and proteins in EY that act as membrane stabilizing agents. These components help repair and protect the membrane structure from damage during storage (Ugur et al., 2019). Additionally, CEP-3, as a standard diluent, has an optimal buffer component that maintains pH and osmolarity, thereby increasing spermatozoa survival (Ratnawati et al., 2023). An IPM is required to sunport normal function and metabolism, as well as to trigger the capacitation process and the acrosome reaction (Cunha et al., 2017), allowing the sperm cell to interact with the oocyte. Damage to the IPM is usually accompanied by damage to the organelles of the intact acrosome hood, leading to the release of enzymes necessary during fertilization (Arvioges et al., 2021). Damaged plasma membranes can affect spermatozoa’s physiological and metabolic functions, causing spermatozoa to die (Tethool et al., 2022). ConclusionBased on the findings of this study, the young APP and EY mixture effectively maintained the quality of SO cattle sperm in terms of motility, viability, total number of motile sperm, IPM, and an IAC. The combination of 75% APP and 25% EY can be recommended as an effective diluent formula to preserve the liquid semen quality of SO cattle at 3°C–5°C. AcknowledgmentWe sincerely appreciate the sunport provided by the director of Research, Technology, and Community Service through the Penelitian Terapan Jalur Hilirisasi (PT-JH) in 2023. Conflict of interestThis manuscript has no conflict of interest. All the authors have made significant contributions to this work and affirm that there are no personal, financial, or professional interests that could improperly influence the research and its presentation in this journal. FundingThis research was financially supported under contract number 119/E5/PG.020.00.PL/2023. Authors’ contributionAK was involved in data collection, study design, data evaluation, and paper preparation. APAY contributed to manuscript preparation and critical review. HAS and PU contributed to the drafting of the manuscript and provided advice. SM, SR, and TS designed the study and were supervisors. Data availabilityAll data supporting this study’s findings are included within the manuscript. ReferencesAbdel-aziz Swelum, A., M. Saadeldin, I., Ba-Awadh, H., G. Al-mutary, M., F. Moumen, A., N. Alowaimer, A. and Abdalla, H. 2019. Efficiency of commercial egg yolk-free and egg yolk-supplemented Tris-based extenders for dromedary camel semen cryopreservation. Animals 9(11), 999; doi:10.3390/ani9110999 Abdullah, U.N. and Meng, G.Y. 2024. Raising double-muscled breed cattle and their crossbreds in the tropics: insight from growth models. Vet. World 17, 1504–1513. Aitken, R., Baker, M. and Nixon, B. 2015. Are sperm capacitation and apoptosis the opposite ends of a continuum driven by oxidative stress?. Asian J. Androl. 17(4), 633; doi:10.4103/1008-682x.153850 Al-Bulushi, S., Manjunatha, B.M., Bathgate, R., Rickard, J.P. and De Graaf, S.P. 2019. Liquid storage of dromedary camel semen in different extenders. Anim. Reprod. Sci. 207, 95–106. Amirat-Briand, L., Bencharif, D., Vera-Munoz, O., Pineau, S., Thorin, C., Destrumelle, S., Desherces, S., Anton, M., Jouan, M., Schmitt, E. and Tainturier, D. 2010. In vivo fertility of bull semen following cryopreservation with an LDL (low density lipoprotein) extender: preliminary results of artificial inseminations. Anim. Reprod. Sci. 122(3–4), 282–287; doi: https://doi.org/10.1016/j.anireprosci.2010.09.006 Arvioges, Pajri A. and Jiyanto. 2021. Efektifitas suhu thawing terhadap keadaan membran plasma utuh (MPU) dan tudung akrosom utuh (TAU) spermatozoa sapi Bali. Jurnal Green Swarnadwipa, 10(2), 1–9. Available via https://ejournal.uniks.ac.id/index.php/GREEN/article/view/1350/948 Authaida, S., Boonkum, W. and Chankitisakul, V. 2025. Enhancement of semen cryopreservation from native Thai bulls through Moringa oleifera leaf extract supplementation. Animals 15(3), 439. Bakae, T., Monau, P.I., Nsoso, S.J. and Kgwatalala, P.M. 2022. Assessment of genetic diversity and relationship of the two Sanga type cattle of Botswana based on microsatellite markers. Trop. Anim. Health Prod. 54(4), 241. Bustani, G.S. and Baiee, F.H. 2021. Semen extenders: an evaluative overview of preservative mechanisms of semen and semen extenders. Vet. World 1220, 1220–1233; doi:10.14202/vetworld.2021.1220-1233 Chan, B. 2018. Data analysis using R programming. doi: 10.1007/978-3-319-93791-5_2 Christensen, P., Labouriau, R., Birck, A., Boe-Hansen, G.B., Pedersen, J. and Borchersen, S. 2011. Relationship among seminal quality measures and field fertility of young dairy bulls using low-dose inseminations. J. Dairy Sci. 94, 1744–1754; doi:10.3168/jds.2010-3087 Cunha, A.T.M., Faria, O.A.C. and Guimarães, A.L.S. 2017. Bovine sperm capacitation: physiological changes and evaluations. JSM. Vitro Fertil. 2(1), 1011; doi:10.47739/1011 Ducha, N., Susilawati, T.A., Wahyunings, S. and Pangestu, M. 2012. Ultrastructure and fertilizing ability of Limousin bull sperm after storage in CEP-2 extender with and without egg yolk. Pak. J. Biol. Sci. 15(20), 979–985; doi:10.3923/pjbs.2012.979.985 Ďuračka, M., Benko, F., Kačániová, M. and Tvrdá, E. 2024. Comparative study: efficacy of egg-yolk vs soy lecithin-based diluent in preservation of chilled bovine semen - Bacteriology and sperm quality. Czech J. Anim. Sci. 69(10), 400–409; doi:10.17221/138/2024-cjas. Gaina, C.D., Sanam, M.U.E., Nalley, W.M.M., Benu, I. and Saputra, A. 2019. Hematological profile of Sumba Ongole cattle extensively reared in semiarid land, Sumba, NTT based on age and sex. IOP Conference Series: Earth and Environmental Science 387, 012022; doi:10.1088/1755-1315/387/1/012022. Hine, T.M., Nalley, W.M., Marawali, A., Kihe, J.N., Kune, P. and Uly, K. 2024. Improvement of sexed sperm quality of Bali bulls by adding palmyra (Borassus flabellifer Linn.) fruit water to citrate-egg yolk extender. J. Ilmu. Ternak. Vet. 29(3), 135–142. Ina, A.T. and Kaka, A. 2020. Preservation of spermatozoa Sumba Ongole bulls using citrate yolk diluent with the addition of palmyra palm juice. J. Ternak 11(2), 86–90. Indonesian National Standard. 2017. Frozen semen—Chapter 1: Bulls. Indonesian National Standard, Vol. 4869-1-2017, pp. 11. Available via https://www.bsn.go.id/uploads/attachment/rsni3_4869-1-2024.pdf (Accessed 10 January 2025). Jakop, U., Engel, K.M., Hürland, M., Müller, P., Osmers, J.H., Jung, M. and Schulze, M. 2023. Lipid alterations by oxidative stress increase detached acrosomes after cryopreservation of semen in Holstein bulls. Theriogenology 197, 37–45; doi:10.1016/j.theriogenology.2022.11.036 Temesgen, K., Aster, Y. and Habtamu, A. 2021. Artificial insemination: coverage and constraints in the central highlands of Ethiopia. Anim. Vet. Sci. 9(5), 149–155; doi: 10.11648/j.avs.20210905.15. Kaka, A., Yekti, A.P.A., Maylinda, S., Rahayu, S. and Susilawati, T. 2025. Effectiveness of nanoparticle-based young palmyra fruit water–egg yolk in liquid semen diluent of Sumba Ongole bulls. Int. J. Vet. Sci. 14(1), 16–24; doi:10.47278/journal.ijvs/2024.205 Kulaksız, R., Çebi, C., Akçay, E. and Daşkın, A. 2010. The protective effect of egg yolk from different avian species during the cryopreservation of Karayaka ram semen. Small Ruminant Res. 88, 12–15; doi:10.1016/j.smallrumres.2009.11.014 Li, Z., Liu, H., Wang, J., Zhou, Y., Fang, Y. and Lu, W. 2025. Retrospect and prospect: reproductive technologies in beef cattle. Mol. Biol. Rep. 52(1), 1–11; doi: 10.1007/s11033-025-10356-y Mariana, E., Putra, G.D., Sitepu, M.P., Novita, C.I., Saputra, H., Muslina, and Khairi, F. 2025. The effect of adding glycerol to an extender containing butterfly pea (Clitoria ternatea) extract on the quality of Aceh cattle spermatozoa during various steps of preservation. IOP Conference Series: Earth and Environmental Science 1476, 012034; doi:10.1088/1755-1315/1476/1/012034 Murphy, E.M., Murphy, C., O'Meara, C., Dunne, G., Eivers, B., Lonergan, P. and Fair, S. 2017. A comparison of semen diluents on the in vitro and in vivo fertility of liquid bull semen. J. Dairy Sci. 100(2), 1541–1554; doi:10.3168/jds.2016-11646 Nalley, W.M.M., Meidina, T.S.A., Kurnia, A. and Arifiantini, R.I. 2019. The addition of fish Salmon Omega-3 in tris egg yolk diluents on the quality of Simmental bull frozen semen. Asian J. Agric. Biol. 7(3), 467–473. Peris-Frau, P., Soler, A.J., Iniesta-Cuerda, M., Martín-Maestro, A., Sánchez-Ajofrín, I., Medina-Chávez, D.A., Fernández-Santos, M.R., García-Álvarez, O., Maroto-Morales, A., Montoro, V. and Garde, J.J. 2020. Sperm cryodamage in ruminants: understanding the molecular changes induced by the cryopreservation process to optimize sperm quality. Int. J. Mol. Sci. 21(8), 2781; doi:10.3390/ijms21082781 Peruma, P. 2018. Low density lipoprotein in cryopreservation of semen. Asian Pac. J. Reprod. 7, 103; doi:10.4103/2305-0500.233571 Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. and R Core Team. 2018. nlme: Linear and nonlinear mixed effects models (Version 3.1-137) [R package]. Available via http://CRAN.R-project.org/package=nlme Rathod, B.S., Nakhashi, H.C., Sengar, G.S., Raja, T.V., Singh, U. and Deb, R. 2022. Profiling of key heat shock proteins and their relationship with male sexual behavior and seminal characteristics in Kankrej (Bos indicus) breeding bulls during different seasons. Trop. Anim. Health Prod. 54(6),1–9; doi: 10.1007/s11250-022-03359-5 Ratnawati, D., Ciptadi, G., Rahayu, S., Retnowulan, D., Nafis, F.A.D., Ridwan, T., Setiyono, A., Iswati, I., Tribudi, Y.A., Irmawanti, S. and Susilawati, T. 2025. Utilization of nanoparticle soybean extract as an extracellular cryoprotectant in cooling semen of Bali cattle (Bos indicus). J. Appl. Anim. Res. 53(1), 1–9; doi: 10.1080/09712119.2025.2516528 Ratnawati, D., Isnaini, N. and Susilawati, T. 2017. Pemanfaatan CASA dalam observasi motilitas spermatozoa semen cair Sapi Madura dalam pengencer berbeda. J. Ilmu-Ilmu. Peternak. 27, 80–95. Ratnawati, D., Kuswati, K., Yekti, A.P.A., Ciptadi, G., Rahayu, S. and Susilawati, T. 2023. Effect of modified CEP-3 diluents with aqueous soybean extract on liquid semen quality in Ongole crossbred bull. Vet. World 1075, 1075–1083; doi:10.14202/vetworld.2023.1075-1083 Rojas-Downing, M.M., Nejadhashemi, A.P., Harrigan, T. and Woznicki, S.A. 2017. Climate change and livestock: impacts, adaptation, and mitigation. Clim. Risk Manag. 16, 145–163. Saad, M., Hussein, Y., Soliman, S. and Eliraqy, E. 2022. Study on low density lipoprotein of duck egg yolk as cryoprotectants of Holstein bulls semen cryopreservation. J. Anim. Poult. Prod. 13(10), 137–142; doi:10.21608/jappmu.2022.161086.1055 Sholikin, M.M., Sadarman., Irawan, A., Sofyan, A., Jayanegara, A., Rumhayati, B., Hidayat, C., Adli, D.N., Julendra, H., Herdian, H., Manzila, I., Hudaya, M.F., Harahap, M.A., Qomariyah, N., Budiarto, R., Krisnan, R., Asmarasari, S.A., Hayanti, S.Y., Wahyono, T., Priyatno, T.P., Ujilestari, T., Negara, W., Wulandari, W. and Nahrowi, N. 2023. A meta-analysis of the effects of clay mineral supplementation on alkaline phosphatase, broiler health, and performance. Poultry Sci. 102(3), 1; doi:10.1016/j.psj.2022.102456 Subramanian, S.P., Renuka, K. and Pillai, S.I. 2024. Palmyra palm (Borassus flabellifer Linn) - A celestial tree. J. Chem. Health Risks 14(4), 1720–1734. Susilawati, T. 2013. Pedoman inseminasi buatan pada ternak. Malang, Indonesia: UB Press, pp: 41–45. Susilawati, T., Isnaini, N., Kuswati, Satria, A.T., Huda, A.N. and Yekti, A.P.A. 2020. The pregnancy evaluation on Ongole crossbred cows by using liquid semen and frozen semen. IOP Conference Series: Earth and Environmental Science, 478(1), 012015; doi: 10.1088/1755-1315/478/1/012015 Susilawati, T., Suyadi, Ihsan, N., Wahjuningsih, S., Isnaini, N., Rachmawati, A., Yekti, A.P.A. and Utami, P. 2022. Manajemen reproduksi dan inseminasi buatan. Malang, Indonesia: UB UB Press, pp. 31–62. Susilawati, T., Wahyudi, F.E., Anggraeni, I., Isnaini, N. and Ihsan, M.N. 2016. The substitution of bovine serum albumin with cattle blood serum and egg white in CEP-2 diluent on the quality of Limousin bull liquid semen during refrigerating. J. Kedokteran. Hewan. 10(2), 98–102; doi:10.21157/j.ked.hewan.v10i2.5025 Syah, H.A., Yekti, A.P.A., Utami, P., Isnaini, N. and Susilawati, T. 2024. Effect of artificial insemination timing on conception rate in lactating Holstein-Friesian cows. World’s. Vet. 14(4), 529–535; doi:10.54203/scil.2024.wvj60 Temesgen, K., Aster, Y. and Habtamu, A. 2021. Artificial insemination: coverage and constraints in the central highlands of Ethiopia. Anim. Vet. Sci. 9(5), 149–155; doi: 10.11648/j.avs.20210905.15 Tethool, A.N., Ciptadi, G., Wahjuningsih, S. and Susilawati, T. 2022. Deterioration of frozen semen of Bali cattle after cooling at 5°C. World’s. Vet. 12(3), 395–404; doi:10.54203/scil.2022.wvj50 Thi Le, D.H., Chiu, C.S., Chan, Y.J., Wang, C.C.R., Liang, Z.C., Hsieh, C.W., Lu, W.C., Mulio, A.T., Wang, Y.J. and Li, P.H. 2021. Bioactive and physicochemical characteristics of natural food: palmyra palm (Borassus flabellifer Linn.) syrup. Biology 10(10), 1028. Ugur, M.R., Saber Abdelrahman, A., Evans, H.C., Gilmore, A.A., Hitit, M., Arifiantini, R.I., Purwantara, B., Kaya, A. and Memili, E. 2019. Advances in cryopreservation of bull sperm. Front. Vet. Sci. 6, 268; doi:10.3389/fvets.2019.00268 Upadhyay, V.R., Roy, A.K., Pandita, S., Raval, K., Patoliya, P., Ramesh, V., Dewry, R.K., Yadav, H.P., Mohanty, T.K. and Bhakat, M. 2023. Optimized addition of nitric oxide compounds in semen extender improves post-thaw seminal attributes of Murrah buffaloes. Trop. Anim. Health Prod. 55(1), 1–9; doi: 10.1007/s11250-023-03474-x Utami, P., Hanif, M.Z., Yekti, A.P.A., Prafitri, R., Huda, A.N., Kuswati, K., Kusmartono, K. and Susilawati, T. 2022. Evaluation the success of artificial insemination using frozen sexed semen based on different estrus characters. J. Agripet 22(2), 190–196; doi:10.17969/agripet.v22i2.23423 Utami, P., Yekti, A.P.A., Simbolon, C.N.A., Syah, H.A., Amaliya, A., Siswoyo, T.A., Isnaini, N. and Susilawati, T. 2025. Analysis of kinetic parameters of sexed Holstein-Friesian bull spermatozoa using Percoll density gradient centrifugation with computer-assisted sperm analysis. Vet. World 18(2), 287–295; doi:10.14202/vetworld.2025.287-295 Wurlina, W., Hariadi, M., Safitri, E., Susilowati, S. and Meles, D.K. 2020. The effect of crude guava leaf tannins on motility, viability, and intact plasma membrane of stored spermatozoa of Etawa crossbred goats. World Vet. 13(3), 530–537; doi:10.14202/vetworld.2020.530-537 Yekti, A.P.A., Hasan, I., Hermanto., Febrianto, N., Isnaini, N., Utami, P., Syah, H.A., Wahjuningsih, S. and Susilawati, T. 2024. The successful artificial insemination with frozen semen from different genetic qualities of Friesian Holstein bull. J. Ilmu-Ilmu. Peternak. 34(3), 448–454; doi:10.21776/ub.jiip.2024.034.03.15 Yekti, A.P.A., Rahayu, S., Ciptadi, G. and Susilawati, T. 2023. The quality and proportion of spermatozoa X and Y in sexed frozen semen separated with Percoll density gradient centrifugation method on Friesian Holstein bull. Adv. Anim. Vet. Sci. 11(3), 371–378; doi:10.17582/journal.aavs/2023/11.3.371.378 Zhang, B., Wang, Y., Wu, C., Qiu, S., Chen, X., Cai, B. and Xie, H. 2021. Freeze-thawing impairs the motility, plasma membrane integrity and mitochondria function of boar spermatozoa through generating excessive ROS. BMC Vet. Res. 17(1), 284; doi:10.1186/s12917-021-02804-1 Zhang, L., Wang, Y., Sun, X., Kang, Y., Sohail, T., Wang, J. and Li, Y. 2023. Effects of different diluents on semen quality of Hu ram stored at 4°C. Animals 13(18), 2823; doi:10.3390/ani13182823 Zuidema, D., Kerns, K. and Sutovsky, P. 2021. An exploration of current and perspective semen analysis and sperm selection for livestock artificial insemination. Animals 11(12), 3563; doi:10.3390/ani11123563 | ||
| How to Cite this Article |
| Pubmed Style Kaka A, Yekti APA, Syah HA, Utami P, Maylinda S, Rahayu S, Susilawati T. Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Vet. J.. 2025; 15(10): 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 Web Style Kaka A, Yekti APA, Syah HA, Utami P, Maylinda S, Rahayu S, Susilawati T. Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. https://www.openveterinaryjournal.com/?mno=251540 [Access: January 26, 2026]. doi:10.5455/OVJ.2025.v15.i10.36 AMA (American Medical Association) Style Kaka A, Yekti APA, Syah HA, Utami P, Maylinda S, Rahayu S, Susilawati T. Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Vet. J.. 2025; 15(10): 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 Vancouver/ICMJE Style Kaka A, Yekti APA, Syah HA, Utami P, Maylinda S, Rahayu S, Susilawati T. Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Vet. J.. (2025), [cited January 26, 2026]; 15(10): 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 Harvard Style Kaka, A., Yekti, . A. P. A., Syah, . H. A., Utami, . P., Maylinda, . S., Rahayu, . S. & Susilawati, . T. (2025) Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Vet. J., 15 (10), 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 Turabian Style Kaka, Alexander, Aulia Puspita Anugra Yekti, Habib Assidiq Syah, Putri Utami, Sucik Maylinda, Sri Rahayu, and Trinil Susilawati. 2025. Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Veterinary Journal, 15 (10), 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 Chicago Style Kaka, Alexander, Aulia Puspita Anugra Yekti, Habib Assidiq Syah, Putri Utami, Sucik Maylinda, Sri Rahayu, and Trinil Susilawati. "Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle." Open Veterinary Journal 15 (2025), 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 MLA (The Modern Language Association) Style Kaka, Alexander, Aulia Puspita Anugra Yekti, Habib Assidiq Syah, Putri Utami, Sucik Maylinda, Sri Rahayu, and Trinil Susilawati. "Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle." Open Veterinary Journal 15.10 (2025), 5192-5201. Print. doi:10.5455/OVJ.2025.v15.i10.36 APA (American Psychological Association) Style Kaka, A., Yekti, . A. P. A., Syah, . H. A., Utami, . P., Maylinda, . S., Rahayu, . S. & Susilawati, . T. (2025) Asian palmyra palm (Borassus flabellifer L.) and egg yolk as natural extenders for liquid semen of Sumba Ongole cattle. Open Veterinary Journal, 15 (10), 5192-5201. doi:10.5455/OVJ.2025.v15.i10.36 |