| Research Article | ||
Open Vet. J.. 2025; 15(9): 4664-4670 Open Veterinary Journal, (2025), Vol. 15(9): 4664-4670 Research Article Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancyViski Fitri Hendrawan1*, Yefiandini Maya Wibawanti2, Dodik Prasetyo3, Citra Sari4, Ertika Fitri Lisnanti5, Dwi Rahmawati6 and Intan Firdha Olien Noor Al Ichsan21Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 2Undergraduate Student, Faculty of Veterinary Medicine, Universitas Brawijaya, Puncak Dieng Eksklusif, Malang, Indonesia 3Department of Internal Medicine, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 4Department of Veterinary Public Health, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia 5Program of Animal Husbandry, Faculty of Agriculture, Universitas Islam Kadiri, Kediri, Indonesia 6Departement of Orthodontic, Faculty of Dentistry, Universitas Brawijaya, Malang, Indonesia *Corresponding Author: Viski Fitri Hendrawan. Department of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia. Email: viski [at] ub.ac.id Submitted: 03/04/2025 Revised: 25/07/2025 Accepted: 19/08/2025 Published: 30/09/2025 © 2025 Open Veterinary Journal
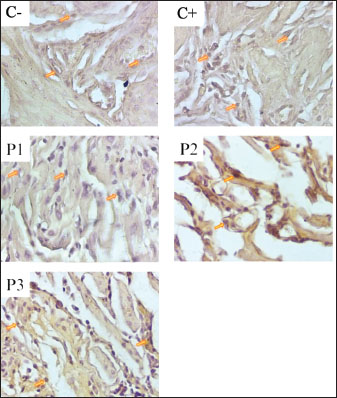
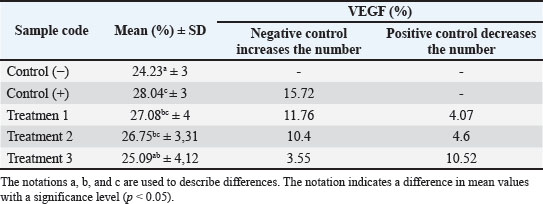
ABSTRACTBackground: Stress is a disorder that causes an imbalance in an organism’s internal environment, which can result in negative impacts such as discomfort and even death. Stressor induction with noise exposure of more than 110 dB can result in decreased organ function and angiogenesis. Aim: This study aimed to determine the effect of nanocurcumin administration in pregnant mice induced by 135 dB noise for 40 minutes on the expression of vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β) in the uterus. Methods: A post-test control group design was used with a sample of 25 female mice divided into five treatment groups. The division of the groups was based on (C−) not given treatment; (C+) induced by 135 dB noise for 40 minutes without being given nanocurcumin; (P1) induced by 135 dB noise for 40 minutes and given 14 mg/Kg body weight (BW) nanocurcumin; (P2) induced by noise of 135 dB for 40 minutes and nanocurcumin 21 mg/Kg BW; and (P3) induced by noise of 135 dB for 40 minutes and nanocurcumin 24.5 mg/Kg BW. VEGF and TGF-β expression were measured using immunohistochemistry. Results: Treatment 3 (P3), with exposure to noise of 135 dB for 40 minutes and administration of nanocurcumin 24.5 mg/Kg BW, increased VEGF and TGF expression by 3.55% and TGF-β expression by 1.79% compared to (C−), but P3 experienced a decrease in VEGF and TGF expression by 10.52% and TGF-β expression by 21.1% when compared to (C+). Conclusion: Nanocurcumin demonstrated strong anti-inflammatory activity by modulating VEGF and TGF-β expression in the uterus of pregnant mice exposed to high-intensity noise stress, particularly at a dose of 24.5 mg/kg BW. Keywords: Nanocurcumin, Noise, Stress, TGF-β, VEGF. IntroductionNoise is an unwanted sound. Its impacts on animals include alterations in behavior and productivity, depending on various factors, such as intensity (dB), frequency (Hz), duration, and pattern—including actinal vibrations. Additionally, the animal’s hearing ability, species, breed, age, and physiological state influence their response to noise. Prolonged or intense noise exposure contributes to several stress-induced diseases and disorders, such as hypertension and psychosomatic illnesses (Peng et al., 2023). Noise exposure above 90 dB has been identified as a significant stressor (Taban et al., 2017). Exposure to 110 dB noise can significantly impair ovarian and uterine function in female mice, reducing uterine blood flow, oxygenation, and the exchange of nutrients and waste products between mother and fetus. Long-term exposure to high-intensity noise elevates levels of adrenocorticotropic hormone and cortisol (Broucek, 2014). Increased cortisol levels promote inflammation, reduce uterine blood flow, and impair placental function (Squires, 2003). Stress is a reflexive reaction that occurs when animals are unable to adapt to environmental stimuli, potentially resulting in discomfort or death. It disrupts homeostasis—the organism’s internal balance—thereby interfering with normal biological functions and possibly causing more serious health issues (Seid and Ahmed, 2021). Stress-induced inflammation also decreases blood oxygenation, potentially leading to hypoxia—a state of reduced oxygen concentration in tissues that can cause organ dysfunction and cellular injury (Younis, 2023). Oxidative stress-induced inflammation can inhibit oxygen transport to tissues, leading to hypoxia. Vascular endothelial growth factor (VEGF) is one proangiogenic factor released during inflammation (Johnson and Wilgus, 2014). Long-term stress also increases the levels of pro-inflammatory cytokines, such as VEGF and transforming growth factor-β (TGF-β), which are key proangiogenic mediators that stimulate the formation of new blood vessels (Johnson and Wilgus, 2014). TGF-β) not only regulates inflammation by inhibiting inflammatory cells but also enhances collagen production for tissue repair and influences fibrosis and regeneration (Deng et al., 2024). The administration of curcumin, a compound known for its anti-inflammatory, antioxidant, antibacterial, antiviral, antifungal, anticancer, and antimalarial properties, is a potential therapeutic strategy. Curcumin inhibits molecules involved in inflammation, such as phospholipase, prostaglandin, elastase, and hyaluronidase. Curcumin inhibits phospholipase and prevents the release of arachidonic acid, a key player in inflammatory pathways (Razavi et al., 2021). However, the clinical application of curcumin is limited due to its low water solubility, instability, and poor bioavailability. These limitations can be overcome through nanoparticle-based formulations, which improve the stability and absorption of nanoparticles (Monika and Zayyanu, 2021). Nanocurcumin at a dose of 20 mg/kg body weight (BW) has been shown to significantly reduce inflammation-induced edema, reflecting its enhanced anti-inflammatory effect (Zeng et al., 2021). Exposure to noise during pregnancy poses specific risks to fetal development. High-intensity noise can impair placental function, disrupt hormonal balance, and reduce blood flow to the uterus, leading to hypoxia and impaired nutrient delivery to the fetus. These conditions may result in IUG, developmental delays, or even fetal loss. Prolonged maternal stress due to noise also elevates glucocorticoid levels, altering fetal programming and increasing the risk of metabolic and behavioral disorders in offspring. Therefore, this study aimed to evaluate the protective effects of nanocurcumin administration on uterine tissue and angiogenesis markers in mice exposed to high-intensity noise during pregnancy. Materials and MethodsResearch duration and participantsThis study was conducted over 3 months, from January to March 2025. The samples used were uterine tissues from 25 pregnant female mice (Mus musculus), each weighing approximately 36.5 g and aged 3 months as many as 25 pregnant mice. Laboratory proceduresThe procedure begins by inducing female mice using PMSG or Folligon hormones at a dose of 5 IU. After 48 hours, hCG or Chorulon hormones are injected, and the female mice are immediately mated with male mice by polymatting (five female mice and one male mouse in one cage). Within 17–24 hours after mating, the vagina of female mice is examined to determine the presence of a copulatory or vaginal plug from the cervix to the vulva. Female mice are considered pregnant if a vaginal plug is found. According to Roy et al. (2019), a copulatory plug or vaginal plug is a mixture of male ejaculate and female secretions that harden with a yellowish color. The procedure for providing noise stress treatment during pregnancy is carried out by dividing 25 pregnant mice into five treatment groups. The division of the groups is based on (C−) not given treatment; (C+) induced by 135 dB noise for 40 minutes without nanocurcumin; (P1) induced by 135 dB noise for 40 minutes and given 14 mg/Kg BW nanocurcumin; (P2) induced by 135 dB noise for 40 minutes and given 21 mg/Kg BW nanocurcumin; and (P3) induced by 135 dB noise for 40 minutes and given 24.5 mg/Kg BW nanocurcumin. Noise stress exposure was carried out by inserting mice into a soundproof box measuring 30 × 20 × 15 cm (p × l × t), and then sound was channeled into the box through a hose connected to an amplifier and speaker with an intensity of 135 dB for 40 minutes. According to Garber et al. (2011), mice weighing ≥25 g require a cage with a floor area of ≥96.7 cm2 and a height of 5 cm. Mice were euthanized using a combination of ketamine and xylazine anesthetic agents intraperitoneally, followed by taking as much intracardial blood as possible using a 3 cc syringe, which aims to minimize pain and stress (Feeney and Papakostas, 2023). Then, the uterus samples obtained were made into paraffin blocks. The tissue was exposed using a microtome until the tissue surface was visible, and sectioning was performed with a thickness of 5 μm. The sectioning results were transferred into a water bath and attached to a glass object smeared with albumin (Oktanella et al., 2023). The immunohistochemical staining procedure can be carried out by soaking the placental histopathology slide in xylol solution twice, then dehydrating the slide using sequentially graded alcohol (96%, 90%, 80%, 70%). Next, the slide was washed three times for 5 minutes each using PBS solution with pH 7.4. The slide was soaked in 3% hydrogen peroxide (in distilled water) for 20 minutes, washed again using PBS solution with pH 7.4 three times for 5 minutes each, then soaked in BSA solution for approximately 10–30 minutes at room temperature, and washed again using PBS solution with pH 7.4 three times for 5 minutes each. VEGF and TGF-β antibodies were added for 1 hour at room temperature and incubated overnight. After one night, the slide was washed three times for 5 minutes each using PBS solution with pH 7.4. Staining was performed using hematoxylin and eosin for 5 minutes, followed by washing three times with distilled water for 5 minutes each time. Mounting was performed using Entellan and then observed using a microscope at 400× and 1,000× magnification (Bintari and Yuliani, 2020). Positive results are indicated by brown spots in the area (%) (Vander et al., 2018). Data analysisThe percentage of the area showing positive VEGF and TGF-β expression was quantified using ImageJ software. Results were statistically analyzed using a one-way analysis of variance followed by Duncan’s multiple range test, with significance set at p < 0.05 using SPSS version 26 for Windows. Ethical approvalThis study was approved by the research ethics committee for the use of laboratory animals, Universitas Muhammadiyah Malang, Indonesia [No. E.5.a/038/KEPKUMM/III/2025]. ResultsEffect of nanocurcumin administration on VEGF expression in the uterus of pregnant mice under stress conditionsThe results of the study on the effect of nanocurcumin administration in mice (M. musculus) models on VEGF expression showed an increase in the positive control group compared to the negative control group, while there was a decrease in the therapy group compared to the positive control group. This indicates that VEGF expression will increase under stressful conditions during pregnancy and will decrease if nanocurcumin therapy is administered. In the picture, positive results are indicated by the brown color in the cell nuclei (endothelial cells). The more concentrated the color, the stronger the interaction between VEGF in the mice uterus and the antibody used, the fainter the bond, the weaker. Negative results are indicated by the purple or bluish color in the cell nuclei. Figure 1 shows the VEGF expression in the uterus of pregnant mice. The results of the VEGF expression examination showed that the positive control (C+) experienced a 15.72% increase from the negative control (C) with an average of 28.04±3., the treatment group (P1) with 135 dB noise induction for 40 minutes and 14 mg/kg nanocurcumin therapy showed an 11.76% increase from (C) with an average of 27.08±4., the treatment group (P2) with 135 dB noise induction for 40 minutes and 21 mg/kg nanocurcumin showed an increase of 10.4% from (C) with an average of 26.75±3.31., the treatment group (P3) given 135 dB noise induction for 40 minutes and 24.5 mg/kg nanocurcumin showed an increase of 3.55% from (C) with an average of 25.09±4.12. Compared with the positive control (C+), each treatment experienced a decrease of 4.07% in P1, 4.6% in P2, and 10.52% in P3. Therefore, it can be concluded that the highest increase in (C) and decrease in (C+) were observed in group (P3). The results of the examination of VEGF expression in the uterus of pregnant mice are shown in Table 1.
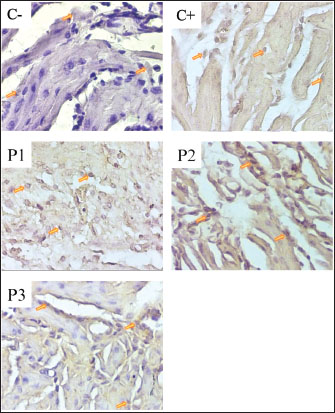
Fig. 1. Immunohistochemical staining of VEGF expression in the uterine tissue of pregnant mice. Positive VEGF expression is indicated by brown-stained nuclei, highlighted by orange arrows. Tissue sections were stained using immunohistochemistry and observed under a light microscope at 400× magnification. The image was captured using a Nikon Eclipse Ei microscope. Effect of Nanocurcumin Administration on TGF-β Expression in the Uterus of Pregnant Mice Under Stress ConditionsThe results of the study on the effects of nanocurcumin administration in mice (M. musculus) models on TGF-β expression showed an increase in the positive control group compared to the negative control group, while there was a decrease in the therapy group compared to the positive control group. This indicates that TGF-β expression will increase under stress conditions during pregnancy and will decrease if nanocurcumin therapy is administered. In Figure 2, the positive results are indicated by the brown color in the cell nuclei (endothelial cells). The more concentrated the color, the stronger the interaction between TGF-β in the mice uterus and the antibody used; the weaker the bond, the weaker the interaction. Negative results are indicated by the purple or bluish color in the cell nuclei. TGF-β expression in the uterus of pregnant mice can be seen in the following: Table 1. Results of the VEGF expression examination.
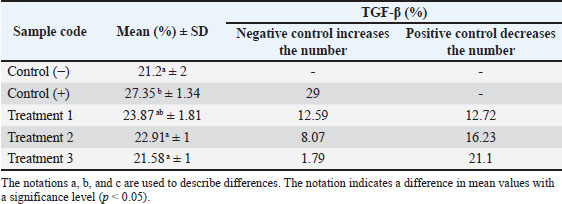
Fig. 2. Immunohistochemical staining of TGF- β expression in uterine tissue of pregnant mice. Positive TGF-β expression is indicated by brown-stained nuclei, highlighted by orange arrows. Tissue sections were stained using immunohistochemistry and observed under a light microscope at 400× magnification. The image was captured using a Nikon Eclipse Ei microscope. The results of the TGF-β expression examination showed that the positive control (C+) experienced a 29% increase from the negative control (C) with an average of 27.35 ± 1.34; the treatment group (P1) with 135 dB noise induction for 40 minutes and 14 mg/kg nanocurcumin therapy showed a 12.59% increase from (C) with an average of 23.87 ± 1.81; the treatment group (P2) with 135 dB noise induction for 40 minutes and 21 mg/kg nanocurcumin showed an 8.07% increase from (C) with an average of 22.91 ± 1; and the treatment group (P3) given 135 dB noise induction for 40 minutes and 24.5 mg/kg nanocurcumin showed a 1.79% increase from (C) with an average of 21.58 ± 1. Compared with the positive control (C+), each treatment experienced a decrease of 12.72%, 16.23%, and 21.1% in P1, P2, and P3, respectively. Therefore, it can be concluded that the highest increase in (C) and decrease in (C+) were observed in group (P3). The results of the TGF-β expression examination in the uterus of pregnant mice are shown in Table 2. DiscussionThe results of this study showed that there was no significant difference between groups (C−) and (P3) with an increase in VEGF of 3.55% and an increase in TGF-β of 1.79%. This shows that nanocurcumin administration has anti-inflammatory activity. In accordance with previous studies, curcumin has strong anti-inflammatory, antioxidant, anti-angiogenic, and anti-carcinogenic activities, and the nanoemulsion method has been proven to be effective. Thus, the use of nanoparticles can increase bioavailability and absorption in the body to achieve therapeutic effects, even when used in low doses (Mohammed et al., 2020). Inflammation can occur in various organs, including the uterus and placenta. Inflammation may reduce oxygen supply to the fetus, as seen in Hendrawan et al. (2017), because of an increase in inflammatory cytokines, which then enter the organs through the blood vessels. Cytokines, particulates, and hydroxyl radicals carried by blood to the placenta can affect cell membrane permeability. As a result, blood viscosity increases, which decreases the oxygen supply to the fetus. Hypoxia in the fetus can occur if the oxygen flow between the mother and fetus is disrupted through the placenta. Hypoxia is a condition of decreased blood oxygen levels that can disrupt the normal function of cells and organs. Placental hypoxia can cause abnormal placental villi development, a decrease in the number of villous and extravillous cytotrophoblasts, and suboptimal placental implantation, leading to trophoblast invasion. Trophoblasts produce VEGF and other angiogenic factors that support blood vessel formation in the placenta (Ozgokce et al., 2023). Oxygen supply to fetal tissues is reduced under hypoxic conditions, which can trigger physiological reactions, including activation of the inflammatory response. Fetal hypoxia can cause inflammation, potentially leading to premature birth or impaired fetal development (Burton and Jauniaux, 2018). Inflammation triggers the release of cytokines to prevent further damage. These cytokines activate macrophages to repair damaged tissue (Litchman et al., 2016). One of the pro-inflammatory cytokines involved in hypoxia is VEGF, the increase of which stimulates the formation of new blood vessels (angiogenesis) (Johnson and Wilgus, 2014). Unbalanced VEGF expression due to inflammatory factors in blood vessels can cause endothelial dysfunction, a condition in which the endothelial cells lining the walls of blood vessels do not function properly. In response to damage, the hypothalamus responds to inflammation by producing cytokines or growth factors, which affect endothelial function. This causes increased endothelial permeability, allowing inflammatory cells and large molecules, such as plasma, to exit the blood vessels and enter the inflamed tissue (Scheiner et al., 2019). Table 2. Results of TGF-β expression examination.
TGF-β is a protein that regulates differentiation, proliferation, and cell death. In addition, TGF-β can also trigger the formation of new blood vessels (angiogenesis). TGF-β will induce angiogenesis by increasing VEGF secretion. However, in high amounts, TGF-β can cause unprogrammed cell death (necrosis), thereby inhibiting endothelial proliferation (Kumar et al., 2018). TGF-β plays an important role in cell differentiation by regulating the expression of genes involved in cell proliferation. Cell differentiation is the main factor that functions in the formation of tissue, so it greatly influences the embryogenesis process (Larson et al., 2020). Increased expression of TGF-β can be caused by abnormalities in the body in the form of inflammation (Dejas et al., 2023). The process of vasculogenesis and angiogenesis during embryogenesis involves the role of VEGF and TGF-β. If inflammation occurs during embryogenesis due to stress, the expression of VEGF and TGF-β will change (Hendrawan et al., 2017). Therefore, anti-inflammatory therapy is necessary to control the increase in VEGF and TGF-β expression so that it can reduce stress-induced inflammation. Curcumin can be used as a therapy because it has anti-inflammatory, antimutagenic, and antioxidant properties. Although useful, several factors, such as low solubility in water, low bioavailability and pharmacokinetics, rapid metabolism, and low target penetration and efficacy, limit its use. Curcumin is also sensitive to alkaline conditions, high temperatures, and light. However, these obstacles can be overcome by making nanoemulsions, which increase the biological activity of curcumin by increasing its solubility and bioavailability, prolonging circulation and retention in the body, and overcoming physiological obstacles. Karthikeyan et al. (2020) reported a 30% reduction in VEGF levels with noise exposure. ConclusionThe administration of nanocurcumin during pregnancy in mice exposed to high-intensity noise stress showed a trend toward modulation of VEGF and TGF-β expression, especially at the highest dose of 24.5 mg/kg BW. Treatment 3 (P3), with exposure to noise of 135 dB for 40 minutes and administration of nanocurcumin 24.5 mg/kg BW, increased VEGF expression by 3.55% and TGF-β expression by 1.79% compared to (C−), but P3 experienced a decrease in VEGF expression by 10.52% and TGF-β expression by 21.1% when compared to (C+). Although the changes were not statistically significant, the observed patterns support the potential of nanocurcumin as a strong anti-inflammatory agent capable of mitigating stress-induced uterine inflammation and angiogenic disruption. These effects are consistent with the known ability of nanocurcumin to inhibit inflammatory mediators and promote tissue protection under stress conditions. Further research is needed to optimize dosing, confirm efficacy through functional and protein-level analyses, and investigate long-term fetal outcomes and PK profiles. The promising anti-inflammatory action of nanocurcumin highlights its potential as a therapeutic candidate for managing environmental stress-induced pregnancy-related complications. AcknowledgmentsThe authors express their gratitude to the academic advisors from the Faculty of Veterinary Medicine, Brawijaya University, Malang, for their guidance, suggestions, and provision of facilities that supported this research. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis research was funded by the Internal DPPSPP Faculty of Veterinary Medicine, Universitas Brawijaya, Malang, Indonesia (2024). Author’s contributionVFH: conceptualization, methodology, validation, formal analysis, investigation, resources, and original draft writing. VFH, CS: visualization, supervision, project administration, and funding acquisition. DP: Methodology, validation, investigation, and resources. DP, CS: Methodology, validation, investigation, and resources. YMW: Original draft, visualization, review, and editing. Data availabilityAll data supporting this study’s findings are available in the manuscript. ReferencesBintari, I.G. and Yuliani, M.G.A. 2020. Deteksi Aeromonas hydrophila pada Ginjal Mencit Mus musculus dengan Teknik Imunohistokimia. J. Agriektensia 19(2), 1. Broucek J. 2014. Effect of noise on performance, stress, and behaviour of animals. Slovak J. Anim. Sci. 47(2), 111–123. Burton, G.J. and Jauniaux, E. 2018. Pathophysiology of placental-derived growth restriction. Am. J. Obstect. Gynecol. 218(2), 745–761. Dejas, L., Santoni, K., Meunier, E. and Lamkanfi, M. 2023. Regulated cell death in neutrophils: from apoptosis to NETosis and pyroptosis. Semin. Immunol. 70, 101849. Deng, Z., Fan, T., Xiao, C., Tian, H., Zheng, Y., Li, C. and He, J. 2024. TGF-β Signaling in Health, Disease, and therapeutics. Springer Nat. 9(61), 1–40. Feeney, A. and Papakostas, G.I. 2023. Pharmacotherapy: ketamine and esketamine. Psychiatr. Clin. Res. 46(2), 277–290. Garber, J.C., Barbee, R.W., Bielitzki, J.T. and et al. 2011. Guide for the care and use of laboratory animals. Hendrawan V.F., Widjiati, Susilowati S. and dan Srianto P. 2017. Peningkatan Ekspresi vascular endothelial growth factor dan mitogen activating protein Kinase Plasenta Tikus yang Dipapar Carbon Black. J. Vet. 18(1), 59–68. Johnson, K.E. and Wilgus, T.A. 2014. Vascular endothelial growth factor and angiogenesis in cutaneous wound repair regulation. Wound Repair Regen. 3(10), 647–661. Karthikeyan, A., Senthil, N. and Min, T. 2020. Nanocurcumin: a promising candidate for therapeutic applications. Front. Pharmacol. 11(487), 1–24. Kumar, V., Abbas, A.K., and Aster, J.C. 2018. Robbins basic pathology. Philadelphia, PA: Elsevier. Larson, C., Oronsky, B., Carter, C.A., Oronsky, A., Knox, S.J., Sher, D. and Reid, T.R. 2020. TGF-beta: a master immune regulator. Expert Opinion Therapeutic Targets 24(5), 427–438; doi:10.1016/j.eott.2020.10.007 Lichtman, M.K., Otero-Vinas, M. and Falanga, V. 2016. Transforming growth factor beta (TGF-β) isoforms in wound healing and fibrosis. Wound Rehabil. Regen. 24, 215–222. Mohammed, E., El-Beih, N., El-Hussieny, E., El-Ahwany, E., Hassan, M. and Zoheiry, M. 2021. Effects of free and nanoparticulate curcumin on chemically induced liver carcinoma in an animal model. Arch. Med. Sci. 17(1), 218–227. Monika, K. and Zayyanu, T. 2021. Nanocurcumin: a Review. Bayer J. Med. Lab. Sci. 6(1), 115–121. Oktanella, Y., Untari, H., Wuragil, D.K., Ismiawati, H., Hasanah, N.A., Agustina, G.C. and Pratama, D.A.O. 2023. Evaluation of renal disturbance in animal models of polycystic ovary syndrome. Open Vet. J. 13(8), 1003–1011. Ozgokce, C., Ocal, A., Ermis, I.S. and Deveci, E. 2023. Histopathological, ultrastructural, and immunohistochemical examination of changes in the placenta because of severe preclampsia. ACTA Cirurg. Brasil. 38, 1–9; doi:10.1016/j.actacr.2018.09.013 Peng, X., Mao, Y., Tai, Y., Luo, B., Dai, Q., Wang, X., Wang, H., Liang, Y., Guan, R., Liu, C., Guo, Y., Chen, L., Zhang, Z. and Wang, H. 2023. Characterization of anxiety-like behaviors and neural circuitry following chronic moderate noise exposure in mice. Environ. Health Perspect. 131(10), 107004. Razavi, B.M., Ghasemzadeh Rahbardar, M. and Hosseinzadeh, H. 2021. A review of therapeutic potentials of turmeric (Curcuma longa) and its active constituent, curcumin, on inflammatory disorders, pain, and their related patents. Phytotherapy Res. PTR. 35(12), 6489–6513. Roy, T.V., Nurcahyani, N., Sutyarso. and Busman, H. 2019. Spinal structure of the fetus of mice (Mus musculus L.) Treated with Acanthus ilicifolius L. leaves extract. Nat. Rev. Dis. Primers 6(1), 27–31. Scheiner, K.C., Maas-Bakker, R.F., Nguyen, T.T., Duarte, A.M., Hendriks, G., Sequeira, L., Duffy, G.P., Steendam, R., Hennink, W.E. and Kok, R.J. 2019. Sustained release of vascular endothelial growth factor from poly(ε-caprolactone-PEG-ε-caprolactone)-b-Poly(l-lactide) multiblock copolymer microspheres. ACS Omega 4, 11481–11492. Seid, M.E. and Ahmed, E.F. 2021. Effect of stress on animal productivity and response to stressors. J. Anim. Vet. Adv. 20(1), 1–14. Squires, E.J. 2003. Applied animal endocrinology. London: UK: CABI Publishing. Taban, E., Mortazavi, S.B., Vosoughi, S., Khavanin, A. and Mahabadi, H.A. 2017. Effects of noise exposure on blood glucose, cortisol, and weight changes in male mice. Health Scope 6(2), 1–5. Vander Ark, A., Cao, J. and Li, X. 2018. TGF-β receptors: in and beyond TGF-β signaling. Cell. Signal. 52, 112–120. Younis, N.S. 2023. Myrrh essential oil mitigates renal ischemia/reperfusion-induced injury. Curr. Issues Mol. Biol. 45(2), 1183–1196. Zeng, L., Yu, G., Hao, W., Yang, K. and Chen, H. 2021. The efficacy and safety of Curcuma longa extract and curcumin supplements on osteoarthritis: a systematic review and meta-analysis. Bioscience. Rep. 41(6), BSR20210817. | ||
| How to Cite this Article |
| Pubmed Style Hendrawan VF, Wibawanti YM, Prasetyo D, Sari C, Lisnanti EF, Rahmawati D, . Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Vet. J.. 2025; 15(9): 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 Web Style Hendrawan VF, Wibawanti YM, Prasetyo D, Sari C, Lisnanti EF, Rahmawati D, . Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. https://www.openveterinaryjournal.com/?mno=250573 [Access: January 26, 2026]. doi:10.5455/OVJ.2025.v15.i9.70 AMA (American Medical Association) Style Hendrawan VF, Wibawanti YM, Prasetyo D, Sari C, Lisnanti EF, Rahmawati D, . Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Vet. J.. 2025; 15(9): 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 Vancouver/ICMJE Style Hendrawan VF, Wibawanti YM, Prasetyo D, Sari C, Lisnanti EF, Rahmawati D, . Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Vet. J.. (2025), [cited January 26, 2026]; 15(9): 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 Harvard Style Hendrawan, V. F., Wibawanti, . Y. M., Prasetyo, . D., Sari, . C., Lisnanti, . E. F., Rahmawati, . D. & (2025) Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Vet. J., 15 (9), 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 Turabian Style Hendrawan, Viski Fitri, Yefiandini Maya Wibawanti, Dodik Prasetyo, Citra Sari, Ertika Fitri Lisnanti, Dwi Rahmawati, and Intan Firdha Olien Noor Al Ichsan. 2025. Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Veterinary Journal, 15 (9), 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 Chicago Style Hendrawan, Viski Fitri, Yefiandini Maya Wibawanti, Dodik Prasetyo, Citra Sari, Ertika Fitri Lisnanti, Dwi Rahmawati, and Intan Firdha Olien Noor Al Ichsan. "Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy." Open Veterinary Journal 15 (2025), 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 MLA (The Modern Language Association) Style Hendrawan, Viski Fitri, Yefiandini Maya Wibawanti, Dodik Prasetyo, Citra Sari, Ertika Fitri Lisnanti, Dwi Rahmawati, and Intan Firdha Olien Noor Al Ichsan. "Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy." Open Veterinary Journal 15.9 (2025), 4664-4670. Print. doi:10.5455/OVJ.2025.v15.i9.70 APA (American Psychological Association) Style Hendrawan, V. F., Wibawanti, . Y. M., Prasetyo, . D., Sari, . C., Lisnanti, . E. F., Rahmawati, . D. & (2025) Effect of nanocurcumin administration on VEGF and TGF-β Expression in the uterus of mice exposed to noise stress during pregnancy. Open Veterinary Journal, 15 (9), 4664-4670. doi:10.5455/OVJ.2025.v15.i9.70 |