| Research Article | ||
Open Vet. J.. 2025; 15(12): 6541-6551 Open Veterinary Journal, (2025), Vol. 15(12): 6541-6551 Research Article Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposureWurlina Wurlina1, Imam Mustofa1, Sri Mulyati1*, Aswin Rafif Khairullah2, Dewa Ketut Meles3, Adeyinka Oye Akintunde4, Niluh Suwasanti5, Ulul Khoiriyah6, Gusti Hardyani6, Deny Rama Kusnadi6, Nove Hidajati3, Rahmi Sugihartuti3, Budi Utomo1, Erma Safitri1, Lilik Maslachah3, Rochmah Kurnijasanti3 and Suryo Kuncorojakti71Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 2Research Center for Veterinary Science, National Research and Innovation Agency (BRIN), Bogor, Indonesia 3Division of Basic Veterinary Medicine, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 4Department of Agriculture and Industrial Technology, Babcock University, Ilishan Remo, Nigeria 5Department of Clinical Pathology, Faculty of Medicine, Universitas Katolik Widya Mandala Surabaya, Surabaya, Indonesia 6Profession Program of Veterinary Medicine, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia 7Division of Veterinary Anatomy, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia *Corresponding Author: Sri Mulyati. Division of Veterinary Reproduction, Faculty of Veterinary Medicine, Universitas Airlangga, Surabaya, Indonesia. Email: sri-m [at] fkh.unair.ac.id Submitted: 01/04/2025 Revised: 02/11/2025 Accepted: 14/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
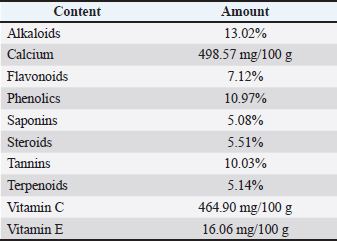
AbstractBackground: Moringa oleifera leaves are particularly high in quercetin compared with other flavonoids. Quercetin is known for its strong antioxidant properties. Aim: This study sought to determine the protective effects of ethanolic extract of M. oleifera leaves on lead acetate-induced testicular histopathological alterations, testosterone suppression, oxidative stress marker (MDA) elevation, and antioxidant enzyme (SOD) depletion in rats. Methods: Twenty-five rats were randomly assigned to five groups. The rats in the negative control group received 0.5 ml of distilled water and 0.5 ml of 0.5% carboxymethyl cellulose sodium (CMC Na) every 4 hours for 21 days. The positive control group (T0) received 0.5 ml of lead acetate solution containing 50 mg/kg BW/day, followed by 0.5 ml of 0.5% CMC Na solution 4 hours later. Groups T1, T2, and T3 were exposed to 0.5 ml of the lead acetate solution (50 mg/kg BW) and subsequently received 0.5 ml of M. oleifera leaf extracts at dosages of 200, 316, and 500 mg/kg BW/day, respectively, after 4 hours. Treatments were administered orally over 20 days. On day 21, all rats were sacrificed for evaluation of serum and testicular parameters. Results: Lead exposure led to significant decreases (p < 0.05) in the diameter and epithelial thickness of the number of spermatogonia, spermatids, Leydig, seminiferous tubules, Sertoli cells, and spermatocytes, as well as testosterone levels and SOD activity, while MDA levels increased (p < 0.05). The administration of Moringa leaf extract at 316–500 mg/kg BW/day helped alleviate these disorders. Conclusion: Moringa oleifera leaf extract has a beneficial effect on the histological and antioxidant parameters of testes in rats with lead acetate-induced Rattus norvegicus. Keywords: Environmental pollution, Moringa oleifera, Seminiferous tubule, Lead acetate, Testosterone. IntroductionHuman health is seriously at risk from Pb pollution. Lead mining, processing, manufacturing, use, and recycling are the main causes of environmental contamination (Raj and Das, 2023). The emissions of PbSO4, PbO, Pb, and PbO2 account for approximately 90% of these pollutants (Yang et al., 2020). For instance, from 2015 to 2018, the water quality index in Central Java was classified as mildly to moderately polluted (Suhardono et al., 2022). Research indicates a notable difference in urine lead levels between affected and healthy toddlers, particularly in children with stunted development (Wardoyo et al., 2022). Oxidative stress is the main cause of the negative effects of lead exposure (Obeng-Gyasi, 2018). One of the main causes of male infertility is oxidative stress, which disrupts the equilibrium between antioxidants and reactive oxygen species (ROS), resulting in an imbalance between the body’s defenses against free radicals and antioxidants (Monageng et al., 2023; Pavuluri et al., 2024). Theoretically, lead exposure can negatively impact libido and sperm quality by reducing sperm count, integrity, viability, and motility, as well as increasing morphological abnormalities and compromising sperm Deoxyribonucleic Acid (DNA) integrity. Collectively, these changes can decrease fertility potential (Kumar, 2018). Antioxidants may mitigate or lessen the harmful effects of free radicals in the body by donating single electrons or hydrogen atoms to stabilize these reactive species (Chaudhary et al., 2023). Antioxidants are considered a potential treatment for heavy metal toxicity in the testes (Koyama et al., 2024). Natural sources of antioxidants can be derived from plants, such as Moringa oleifera Lam, being one notable example. Reproductive functions in female and male rat models were impacted by the consumption of different Moringa plant components (Ogunsola et al., 2017). Moringa oleifera is rich in flavonoids, polyphenols, lycopene, and β-carotene, with quercetin being the primary flavonoid (Kou et al., 2018). Moringa oleifera leaves are particularly high in quercetin compared with other flavonoids (Kashyap et al., 2022). Quercetin is known for its strong antioxidant properties (Qi et al., 2022). Nevertheless, there is no documented use of moringa extract as an antioxidant to treat male infertility caused by lead exposure. This study sought to determine the protective effects of the ethanolic extract of M. oleifera leaves on lead acetate-induced testicular histopathological alterations, testosterone suppression, oxidative stress marker (MDA) elevation, and antioxidant enzyme (SOD) depletion in rats. Materials and MethodsPlant extractionLead acetate [Pb(CH3COO)2] was sourced from Sigma-Aldrich (USA; Cat. No. 6080-56-4). Lead acetate was diluted in distilled water to create the treatments, which were then administered as a daily dose of 50 mg/kg body weight (Revina Ayu Septiani et al., 2022). Moringa oleifera leaf extract was extracted by macerating it with 96% ethanol, then freeze-drying it and diluting it with 0.5% carboxymethyl cellulose sodium (CMC Na) (Mardatillah et al., 2022). Phytochemical analysis of M. oleifera leaf extract was conducted using the High-Performance Liquid Chromatography (HPLC) method (Shafiq et al., 2024). Experimental animalsRattus norvegicus male rats weighing 200–250 g and 2.5–3.0 months of age were kept in climate-controlled plastic cages. The 12-hour light-dark cycle was used to maintain the temperature at 26°C ± 2°C. The rats were given unlimited access to regular commercial meals and drinking water. Twenty-five rats were divided into five equal and random groups. Rats in the negative control group (NC) received 0.5% CMC Na and were exposed to distilled water, which is a solvent for Pb acetate. A 0.5% CMC Na solution and 50 mg/kg BW of lead acetate were administered daily to the T0 positive control group. In addition to receiving 200, 316, and 500 mg/kg BW/day of M. oleifera leaf extract, the T1, T2, and T3 groups were exposed to 50 mg/kg BW of lead acetate. Rats were orally administered a 0.5 ml solution of lead acetate at 7:00 a.m. before eating, and four hours later, they were administered a 0.5 ml solution of M. oleifera leaf extract. This routine was followed for 20 days. On day 21, 40 mg/kg BW of intraperitoneal ketamine was administered to all animals to induce sleep. Blood was extracted from the heart using a subcostal arch laparotomy. Serum was also collected to gauge testosterone levels. The testicles were removed for histological hematoxylin–eosin slide staining and MDA levels and SOD activity measurements. Measurement of the seminiferous tubule diameterA light microscope fitted with Image Raster Software Version 3.7 was used to measure five tubules at 100x magnification in order to determine their diameter (µm). The diameter was defined as the maximum distance from the central point between two opposing points. Four diameters were measured at 45-degree angular intervals from each other for each seminiferous tubule, and the values were averaged (Mardatillah et al., 2022). Epithelium thickness measurementFive tubules were examined at 100x magnification to determine the epithelial thickness (µm) of the seminiferous tubules using a Nikon Eclipse Ci light microscope equipped with Image Raster Software Version 3.7. The distance between the spermatogonia cell layer surface and the basement membrane was defined as the epithelial thickness. Four measurements of epithelial thickness were taken and averaged for each seminiferous tubule (Mardatillah et al., 2022). Examination of the spermatogenic cellsRat testicular microanatomical preparations were stained with hematoxylin-eosin and seen at 400x magnification using a Nikon Eclipse Ci microscope. Random selection of three round-cut seminiferous tubules was analyzed for averaging. The quantity of spermatogonia, primary spermatocytes, and spermatid cells was measured. The oval-round nuclei that are closer to the basal lamina are what distinguish spermatogonia. The seminiferous tubules contain primary spermatocytes, which are distinguished by their tiny chromatin grains and the largest nucleus at the equator. Spermatids, which are also found in seminiferous tubules, are distinguished by their close proximity to the tubule lumen, tiny size, and dense chromatin regions (Machmudia et al., 2021). Leydig cell countThe eosinophilic granular cytoplasm of round Leydig cells aggregates to create polyhedral interstitial tissue in the testes (Aladamat and Tadi, 2022). Five interstitial seminiferous tubules were randomly selected to calculate the number of Leydig cells, and the average count was recorded (Fitri et al., 2019; Ngizzah et al., 2023). At 400× magnification, the observations were made using a light microscope with Optilab Viewer Software Version 2.2 installed. Sertoli cell countThese Sertoli cells have a pyramidal shape with a polymorphic and pale nucleus attached to the basal lamina. Sertoli cells were counted at 400× magnification using a Nikon Eclipse Ci light microscope and Optilab Viewer Software Version 2.2. Five random average observation replications of seminiferous tubules were performed (Panggalih et al., 2021). Testosterone levelsThe Siemens testosterone test kit (Immulite® 1,000 total testosterone) was used to measure serum testosterone levels using the solid-phase competitive chemiluminescence enzyme immunoassay technique (Cinquanta et al., 2017). MDA levelsThe thiobarbituric acid-reactive substance assay was used to measure the amount of MDA in testicular tissue using a thiobarbituric acid kit (NWLSS, USA, Cat. No. NWK-MDA01). A UV-1601 spectrophotometer was used to measure the absorbance at a maximum wavelength of 535 nm (Janero, 1990). SOD levelsA Cayman Chemicals test kit (USA, Cat. No. 706,002) was used to measure the amount of SOD in the testicular tissue. The absorbance was measured at a wavelength of 550 nm (Weydert and Cullen, 2010). Data analysisData were analyzed using one-way analysis of variance, with results reported as F-values, degrees of freedom (df), exact p-values, effect sizes (η²), and 95% confidence intervals. Post-hoc comparisons were performed using Duncan’s multiple range test at a 95% confidence level. All statistical analyses were conducted using the Statistical Package for the Social Sciences version 26 for Windows (IBM Corporation). Ethical approvalThe ethical approval date for this study is 11 October 2022 (Certificate No. 158/HRECC.FODM/XI/2022, Universitas Airlangga Research Ethical Clearance Commission). ResultsPhytochemical analysisPhytochemical analysis using the thin-layer chromatography method showed that M. oleifera leaf extract contained alkaloids, tannins, terpenoids, steroids, flavonoids, saponins, phenolics, calcium, vitamin C, and vitamin E (Table 1). Table 1. Results of phytochemical analysis of M. oleifera leaf extract using high-performance liquid chromatography.
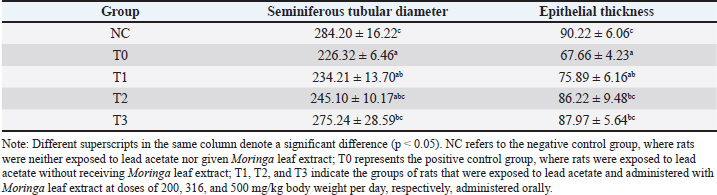
Seminiferous tubule width and epithelial thicknessSeminiferous tubule width and epithelial thickness were lower in rats exposed to lead acetate alone (group T0) than in the control group (p < 0.05). Conversely, the rats in groups T2 and T3, which were treated with lead acetate and then given moringa leaf extract, showed greater seminiferous tubule width and epithelial thickness (p < 0.05) than the group T0, which was treated with lead acetate alone. However, compared with the control rats, the maximum dose of Moringa leaf extract (500 mg/kg body weight per day) reduced the seminiferous tubule width and epithelial thickness (p < 0.05) (Table 2). Table 2. Diameter and epithelial thickness (µm) of seminiferous tubules in the testes of rats (Rattus norvegicus) treated with lead acetate following oral administration of Moringa leaf extract.
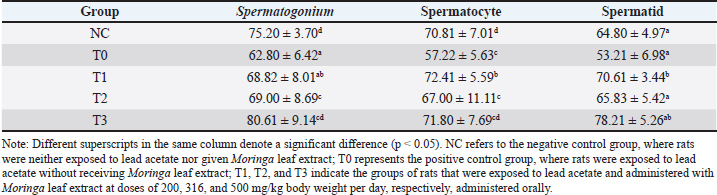
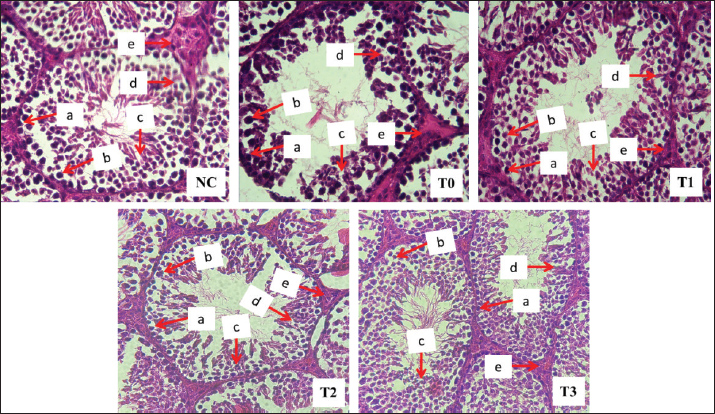
Spermatogenic cellsThe spermatids, spermatocytes, and spermatogonia of rats exposed to lead acetate alone (group T0) were smaller than those of control rats NC (p < 0.05). In contrast, the groups of rats that were exposed to Pb acetate and subsequently given Moringa leaf extract (groups T2 and T3) displayed larger spermatids, spermatocytes, and spermatogonia (p < 0.05) in comparison to the group exposed to Pb acetate alone (group T0). The highest dose of Moringa leaf extract (group T3) resulted in spermatogonia, spermatocytes, and spermatids that were not significantly different (p > 0.05) from those of the control rats NC (Table 3 and Fig. 1).
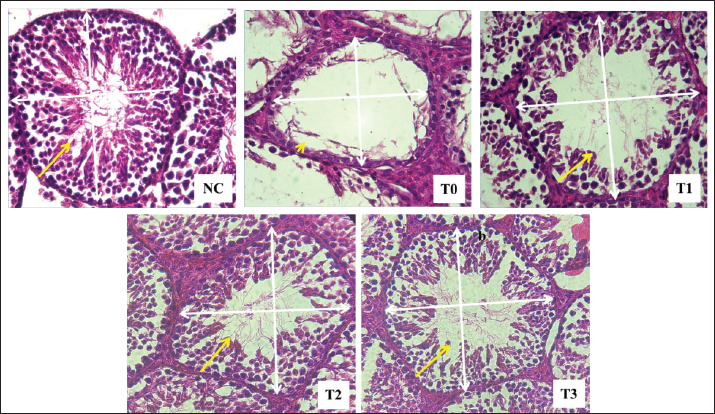
Fig. 1. Diameter (white arrow) and thickness (yellow arrow) of the seminiferous tubule epithelium of rats exposed to lead acetate with or without Moringa leaf extract. Hematoxylin-eosin staining, 400x magnification on a light microscope (Nikon Eclipse Ci). NC refers to the negative control group, where rats were neither exposed to lead acetate nor given Moringa leaf extract; T0 represents the positive control group, where rats were exposed to lead acetate without receiving Moringa leaf extract; T1, T2, and T3 indicate the groups of rats that were exposed to lead acetate and administered with Moringa leaf extract at doses of 200, 316, and 500 mg/kg body weight per day, respectively, administered orally. Table 3. Spermatogonium, spermatocytes, and spermatid counts in the testes of rats (Rattus norvegicus) treated with lead acetate following oral administration of Moringa leaf extract.
Leydig and Sertoli cells were usedThe rats exposed solely to lead acetate (group T0) displayed smaller Leydig and Sertoli cells (p < 0.05) compared to the control rats NC. In contrast, the groups of rats that were exposed to Pb acetate and subsequently supplemented with Moringa leaf extract (groups T2 and T3) exhibited larger Leydig and Sertoli cells (p < 0.05) compared with the lead acetate-only group (group T0). The Leydig and Sertoli cells produced by the maximum dosage of Moringa leaf extract (500 mg/kg body weight daily, group T3) did not differ substantially (p > 0.05) from those of the control rats (NC) (Table 4 and Fig. 2).
Fig. 2. Spermatogonial cells (a), spermatocytes (b), spermatids (c), Sertoli (d), and Leydig (e) in rats exposed to Pb acetate without or with Moringa leaf extract. Hematoxylin-eosin staining, 400x magnification on a light microscope (Nikon Eclipse Ci). NC refers to the negative control group, where rats were neither exposed to lead acetate nor given Moringa leaf extract; T0 represents the positive control group, where rats were exposed to lead acetate without receiving Moringa leaf extract; T1, T2, and T3 indicate the groups of rats that were exposed to lead acetate and administered with Moringa leaf extract at doses of 200, 316, and 500 mg/kg body weight per day, respectively, administered orally. Table 4. Leydig and Sertoli cell counts in the testes of rats (Rattus norvegicus) with lead acetate-induced infection following oral administration of Moringa leaf extract.
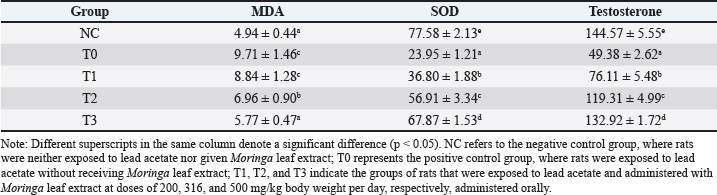
Oxidative stress markers and hormonal changes (MDA, SOD, and testosterone levels)Rats exposed to lead acetate alone (group T0) showed higher levels of MDA (p < 0.05) and lower levels of testicular tissue SOD and serum testosterone (p < 0.05) than control rats NC. Rats exposed to lead acetate and then administered Moringa leaf extract (groups T2 and T3) had lower MDA levels (p < 0.05) and higher serum testosterone levels and testicular tissue SOD (p < 0.05) than the lead acetate-only group (group T0). However, when compared to control rats NC, the maximum dose of Moringa leaf extract (500 mg/kg body weight per day, group T3) produced reduced serum testosterone levels and testicular tissue SOD, while MDA levels were not substantially different (p > 0.05) (Table 5). Table 5. Levels of malondialdehyde (MDA, nmol/ml), superoxide dismutase (SOD) activity (%), and testosterone (ng/dl) following oral administration of Moringa leaf extract in lead acetate-induced rats (Rattus norvegicus).
DiscussionCompared with normal rats, testicular tissue from rats exposed solely to lead acetate had reduced SOD activity and elevated MDA levels, indicating oxidative stress. Lead induces the generation of ROS while simultaneously impairing antioxidant defense systems, particularly enzymes such as SOD and catalase (Flora et al., 2012; Dobrakowski et al., 2017). The disruption of these enzymes reduces the clearance of free radicals, leading to lipid peroxidation and cellular damage (Awad et al., 2014; Ogunlade et al., 2022; Ragab et al., 2024). This imbalance between ROS production and antioxidant defenses is a key mechanism underlying lead-induced testicular toxicity (Singh et al., 2018; Sani and Musa, 2021). In this study, rats exposed to Pb acetate and subsequently treated with M. oleifera leaf extract showed increased testicular SOD activity and reduced MDA levels compared with the Pb-only group. These findings indicate the antioxidant potential of Moringa in mitigating oxidative stress, consistent with previous reports showing its ability to enhance enzymatic antioxidants and suppress lipid peroxidation in animal models (Alkafafy et al., 2021; Mustafa, 2023; Reda et al., 2023; Su et al., 2023; Ragab et al., 2024). Lead exposure can deplete endogenous antioxidant reserves due to increased radical production, resulting in oxidative stress. This oxidative stress can damage mitochondria, subsequently affecting apoptotic processes. Excessive apoptosis of testicular cells causes damage and seminiferous tubule degeneration (Pratama et al., 2021). This apoptotic process begins with the release of cytochrome c, which occurs when ROS damages the inner and outer mitochondrial membranes. Increased calcium ion (Ca2+) levels are associated with the release of cytochrome c, which is caused by ROS interactions with lipids and membrane proteins that impair membrane permeability (Kowalczyk et al., 2021). High ROS levels damage lipids, proteins, and DNA, ultimately impairing testicular function. Although ROS can also activate apoptotic pathways in testicular tissue (Zhu et al., 2015; Korshunova et al., 2021), apoptosis was not directly assessed in this study. Our findings focused on oxidative stress markers (MDA and SOD), which provide indirect evidence of ROS-related tissue injury (Yu et al., 2024). Seminiferous tubules are the major component of testicular tissue and are essential for spermatogenesis (Suede et al., 2023). In this study, lead exposure reduced tubule diameter and epithelial thickness, consistent with previous findings in rodents (Revina Ayu Septiani et al., 2022). These changes are attributed to the loss of spermatogenic and Sertoli cells (Jensen et al., 2022), reflecting the harmful effects of Pb on testicular structure. Lead may disrupt the hypothalamic–pituitary–gonadal axis by reducing GnRH release, leading to lower Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH) levels (Marques et al., 2022). Consequently, testosterone synthesis by Leydig cells and androgen-binding protein (ABP) production by Sertoli cells are impaired, which negatively affects spermatogenesis and seminiferous tubule structure (Shah et al., 2021). Moringa oleifera leaf extract improves seminiferous tubule diameter and epithelial thickness in lead-exposed rats. These effects are attributed to its bioactive compounds, including flavonoids and vitamins C and E, which act as antioxidants and support spermatogenesis (Nobossé et al., 2018; Kalwar et al., 2019; Li et al., 2020; Wurlina et al., 2021a; Wurlina et al., 2021b; Meles et al., 2022). The creation of viable sperm and the facilitation of spermiogenesis are dependent on Sertoli cells. They emit several important molecules, such as activin, inhibin B, and ABP, that either directly or indirectly stimulate spermatogenesis through negative hormonal feedback pathways. Sertoli cells also respond to pituitary hormones such as FSH, initiating spermatogenesis and assisting nearby spermatogonia (Shah et al., 2021). Moringa oleifera leaf extract partly ameliorated damage to the seminiferous tubules caused by lead-induced oxidative stress. Its flavonoids and polyphenols act as antioxidants, protecting testicular tissue and supporting spermatogenesis (Ye et al., 2020; Alkafafy et al., 2021; Kashyap et al., 2022). However, even at the highest dose, the treatment did not fully restore the seminiferous tubule structure to normal levels (Abarikwu et al., 2020; Mardatillah et al., 2022; Ragab et al., 2024). Lead acetate exposure significantly reduced the numbers of spermatids, spermatocytes, and spermatogonia, indicating impaired spermatogenesis (Asadi et al., 2021). This effect is linked to oxidative stress and hormonal disruption, particularly decreased LH and testosterone, which are essential for spermatogenic cell development (Asadi et al., 2017; El-Magd et al., 2017; Ajayi and Akhigbe, 2020; Shah et al., 2021). Rats treated with M. oleifera leaf extract after exposure to lead showed increased numbers of spermatogonia, spermatocytes, and spermatids compared with the lead-only group. This improvement is attributed to the antioxidant compounds in the extract, which reduce oxidative stress and help restore the hormonal balance required for spermatogenesis (Oduwole et al., 2021; Srivastava et al., 2023; Ragab et al., 2024). Exposure to lead decreased the number of Leydig and Sertoli cells, consistent with previous findings in rodents (Revina Ayu Septiani et al., 2022). This reduction is largely attributed to oxidative stress and lipid peroxidation, which damage cell membranes and impair testicular cell function. The lead-induced disruption of gonadotropins (ICSH and FSH) further contributes to the decline in Leydig cell function and testosterone production (Hentrich et al., 2011; Zirkin and Papadopoulos, 2018). Rats treated with M. oleifera leaf extract after exposure to lead showed larger Leydig and Sertoli cells compared with the lead-only group. This effect is likely due to Moringa’s antioxidant properties, which reduce oxidative stress and help maintain testosterone production, which is essential for spermatogenesis (Elmore, 2007; Redza-Dutordoir and Averill-Bates, 2016; Vergara-Jimenez et al., 2017; Oduwole et al., 2018; Shahbaz et al., 2024). Sertoli cells facilitate spermatogenesis through ABP, whereas Leydig cells regulate testosterone production (Griswold, 2018; Zirkin and Papadopoulos, 2018). Lead-induced oxidative stress disrupts these processes by damaging Leydig cell organelles and reducing testosterone synthesis, thereby impairing spermatogenesis (Gill-Sharma, 2018; Huang et al., 2021). The highest dose of M. oleifera leaf extract (500 mg/kg BW/day) restored Leydig and Sertoli cells to levels comparable to those in normal rats. Moringa treatment improved spermatogenic cell counts and increased serum testosterone in lead-exposed rats, likely due to its flavonoid and quercetin content with antioxidant properties (Dias et al., 2021; Monageng et al., 2023). Moringa oleifera leaves alleviate male infertility by reducing oxidative stress and improving sperm parameters and hormone levels (Ayala et al., 2014; Chaudhary et al., 2023). Their strong antioxidant activity, attributed to phenolic compounds and vitamin E, helps neutralize free radicals and prevent lipid peroxidation (Peñalver et al., 2022; Mohlala et al., 2023). Oxidative stress from lead exposure disrupts steroidogenesis in Leydig cells, leading to reduced testosterone and impaired spermatogenesis (Wurlina et al., 2022). Treatment with M. oleifera leaf extract helped restore testosterone production and supported sperm development (Miller and Bose, 2011; Manna et al., 2016). Oxidative stress from lead exposure disrupts Leydig cell function by impairing androgen receptor signaling and damaging organelles, ultimately reducing testosterone synthesis (Eacker et al., 2008; Barati et al., 2020). This disruption also affects the hypothalamic–pituitary–testicular axis, leading to hormonal imbalance and impaired spermatogenesis (Wang et al., 2021; Wurlina et al., 2025). The brain, pituitary gland, and testes all control spermatogenesis. Some neurons in the brain produce GnRH, which promotes the production of FSH and LH. While FSH encourages Sertoli cells to produce more ABP, which is essential for binding testosterone during spermatogenesis, LH stimulates testosterone synthesis in Leydig cells (Oduwole et al., 2021). Although the highest dose of M. oleifera leaf extract did not fully restore testosterone and SOD levels, it effectively reduced MDA to normal levels, indicating protection against oxidative membrane damage (Alkafafy et al., 2021; Ragab et al., 2024). ConclusionLead exposure causes a drop in the number of spermatogenic cells (including spermatogonia, spermatocytes, and spermatids), Leydig cells, and Sertoli cells in rats, as well as a reduction in the seminiferous tubule diameter and epithelial thickness. Additionally, this exposure leads to lower testosterone, malondialdehyde, and superoxide dismutase levels. The administration of Moringa leaf extract helps alleviate these disorders. AcknowledgmentThe authors express their sincere gratitude to the Directorate of Research and Community Service, Deputy for Strengthening Research and Technology, Ministry of Research and Technology/National Research and Innovation Agency. The authors would like to thank Ulul Khoiriyah, Baich R Tyarraushananda Defvyanto, and Sila Faredy Heriana for their technical support. Conflict of interestThe authors declare no conflict of interest. FundingThe authors would like to express their sincere gratitude to the Directorate of Research and Community Service, Deputy for Strengthening Research and Technology, Ministry of Research and Technology/National Research and Innovation Agency for the 2022 fiscal year, Chancellor’s Decree number: 770/UN3.14/PT/2022. Author’s contributionsLaboratory work and data collection: WW, IM, LM, and DKM. Field sampling: ARK, AOK, NS, and GH. Data analysis and manuscript writing: UK, DRK, RS, SM, and SK. Research concept: NH, ES, BU, and RK. All authors have read, revised, and approved the final version of the manuscript. Data availabilityAll data supporting the findings of this study are available within the manuscript, and no additional data sources are required. ReferencesAbarikwu, S.O., Simple, G. and Onuoha, C.S. 2020. Morphometric evaluation of the seminiferous tubules and the antioxidant protective effects of gallic acid and quercetin in the testis and liver of butyl phthalate treated rats. Indian. J. Clin. Biochem. 35(1), 20–31. Ajayi, A.F. and Akhigbe, R.E. 2020. The physiology of male reproduction: impact of drugs and their abuse on male fertility. Andrologia 52(9), e13672. Aladamat, N. and Tadi, P. 2022. Histology, Leydig Cells. StatPearls Publishing, Treasure Island, FL, USA Alkafafy, M.E., Sayed, S.M., El-Shehawi, A.M., El-Shazly, S., Farouk, S., Alotaibi, S.S., Madkour, D.A., Orabi, S.H., Elbaz, H.T. and Ahmed, M.M. 2021. Moringa oleifera ethanolic extract ameliorates the testicular dysfunction resulted from HFD-induced obesity rat model. Andrologia 53(8), e14126. Asadi, A., Arazi, H., Ramirez-Campillo, R., Moran, J. and Izquierdo, M. 2017. Influence of maturation stage on agility performance gains after plyometric training: a systematic review and meta-analysis. J. Strength Cond. Res. 31(9), 2609–2617. Asadi, A., Ghahremani, R., Abdolmaleki, A. and Rajaei, F. 2021. Role of sperm apoptosis and oxidative stress in male infertility: a narrative review. Int. J. Reprod. Biomed. 19(6), 493–504. Awad, N., Shanan, S.K. and Al-Fartosy, A.J.M. 2014. Effect of lead exposure on Malondialdehyde (MDA) level and some biochemical parameter in workers of gasoline stations. Res. J. Pharm. Biol. Chem. Sci. 5(6), 711–716. Ayala, A., Muñoz, M.F. and Argüelles, S. 2014. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell. Longev. 2014(1), 360438. Barati, E., Nikzad, H. and Karimian, M. 2020. Oxidative stress and male infertility: current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cellular Mol. Life Sci. 77(1), 93–113. Chaudhary, P., Janmeda, P., Docea, A.O., Yeskaliyeva, B., Razis, A.F.A., Modu, B., Calina, D. and Sharifi-Rad, J. 2023. Oxidative stress, free radicals and antioxidants: potential crosstalk in the pathophysiology of human diseases. Front. Chem. 11(1), 1158198. Cinquanta, L., Fontana, D.E. and Bizzaro, N. 2017. Chemiluminescent immunoassay technology: what does it change in autoantibody detection?. Auto. Immun. Highlights 8(1), 9. Dias, M.C., Pinto, D.C.G.A. and Silva, A.M.S. 2021. Plant Flavonoids: chemical Characteristics and Biological Activity. Molecules (Basel. Switzerland). 26(17), 5377. Dobrakowski, M., Pawlas, N., Kasperczyk, A., Kozłowska, A., Olewińska, E., Machoń-Grecka, A. and Kasperczyk, S. 2017. Oxidative DNA damage and oxidative stress in lead-exposed workers. Hum. Exp. Toxicol. 36(7), 744–754. Eacker, S.M., Agrawal, N., Qian, K., Dichek, H.L., Gong, E.Y., Lee, K. and Braun, R.E. 2008. Hormonal regulation of testicular steroid and cholesterol homeostasis. Mol. Endocrinol. 22(3), 623–635. El-Magd, M.A., Kahilo, K.A., Nasr, N.E., Kamal, T., Shukry, M. and Saleh, A.A. 2017. A potential mechanism associated with lead-induced testicular toxicity in rats. Andrologia 49(9), e12725. Elmore, S. 2007. Apoptosis: a review of programmed cell death. Toxicol. Pathol. 35(4), 495–516. Fitri, P.E., Wurlina, W., Chusniati, S., Suwanti, L.T., Plumeriastuti, H. and Mufasirin, M. 2019. Effect honey in spermatogenesis staging and leydig cells in mice (Mus musculus) infected by Toxoplasma gondii. Ovozoa. J. Anim. Reprod. 8(1), 10–16. Flora, G., Gupta, D. and Tiwari, A. 2012. Toxicity of lead: a review with recent updates. Interdiscip. Toxicol. 5(2), 47–58. Gill-Sharma, M.K. 2018. Testosterone Retention Mechanism in Sertoli Cells: a Biochemical Perspective. Open Biochem. J. 12(1), 103–112. Griswold, M.D. 2018. 50 years of spermatogenesis: sertoli cells and their interactions with germ cells. Biol. Reprod. 99(1), 87–100. Hentrich, A., Wolter, M., Szardening-Kirchner, C., Lüers, G.H., Bergmann, M., Kliesch, S. and Konrad, L. 2011. Reduced numbers of Sertoli, germ, and spermatogonial stem cells in impaired spermatogenesis. Mod. Pathol. 24(10), 1380–1389. Huang, H., Wang, M., Hou, L., Lin, X., Pan, S., Zheng, P. and Zhao, Q. 2021. A potential mechanism associated with lead-induced spermatogonia and Leydig cell toxicity and mitigative effect of selenium in chicken. Ecotoxicol. Environ. Saf. 209(1), 111671. Janero, D.R. 1990. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 9(6), 515–540. Jensen, C.F.S., Wang, D., Mamsen, L.S., Giwercman, A., Jørgensen, N., Fode, M., Ohl, D., Dong, L., Hildorf, S.E., Pors, S.E., Fedder, J., Ntemou, E., Andersen, C.Y. and Sønksen, J. 2022. Sertoli and Germ Cells Within Atrophic Seminiferous Tubules of Men With Non-Obstructive Azoospermia. Front. Endocrinol. 13(1), 825904. Kalwar, Q., Chu, M., Ahmad, A.A., Ding, X., Wu, X., Bao, P. and Yan, P. 2019. Morphometric Evaluation of Spermatogenic Cells and Seminiferous Tubules and Exploration of Luteinizing Hormone Beta Polypeptide in Testis of Datong Yak. Animals 10(1), 66. Kashyap, P., Kumar, S., Riar, C.S., Jindal, N., Baniwal, P., Guiné, R.P.F., Correia, P.M.R., Mehra, R. and Kumar, H. 2022. Recent Advances in Drumstick (Moringa oleifera) Leaves Bioactive Compounds: composition, Health Benefits, Bioaccessibility, and Dietary Applications. Antioxidants (Basel. Switzerland). 11(2), 402. Korshunova, A., Blagonravov, M., Neborak, E., Syatkin, S., Sklifasovskaya, A., Semyatov, S. and Agostinelli, E. 2021. BCL2-regulated apoptotic process in myocardial ischemia-reperfusion injury (Review). Int. J. Mol. Med. 47(1), 23–36. Kou, X., Li, B., Olayanju, J., Drake, J. and Chen, N. 2018. Nutraceutical or Pharmacological Potential of Moringa oleifera Lam. Nutrients 10(3), 343. Kowalczyk, P., Sulejczak, D., Kleczkowska, P., Bukowska-Ośko, I., Kucia, M., Popiel, M., Wietrak, E., Kramkowski, K., Wrzosek, K. and Kaczyńska, K. 2021. Mitochondrial Oxidative Stress-A Causative Factor and Therapeutic Target in Many Diseases. Int. J. Mol. Sci. 22(24), 13384. Koyama, H., Kamogashira, T. and Yamasoba, T. 2024. Heavy Metal Exposure: molecular Pathways, Clinical Implications, and Protective Strategies. Antioxidants (Basel. Switzerland). 13(1), 76. Kumar, S. 2018. Occupational and Environmental Exposure to Lead and Reproductive Health Impairment: an Overview. Indian. J. Occup. Environ. Med. 22(3), 128–137. Li, G., Ding, K., Qiao, Y., Zhang, L., Zheng, L., Pan, T. and Zhang, L. 2020. Flavonoids Regulate Inflammation and Oxidative Stress in Cancer. Molecules (Basel. Switzerland). 25(23), 5628. Machmudia, A., Eliyani, H., Widjiati, W. and Wurlina, W. 2021. Effect of α-tocopherolon spermatogenic and Leydig cell counts of white rats (Rattus norvegicus) exposed to 2,3,7,8-Tetrachlorodibenzo-p-dioxin. Ovozoa. J. Anim. Reprod. 10(3), 74–79. Manna, P.R., Stetson, C.L., Slominski, A.T. and Pruitt, K. 2016. Role of the steroidogenic acute regulatory protein in health and disease. Endocrine 51(1), 7–21. Mardatillah, M., Wurlina, W., Yudaniayanti, I.S., Plumeriastuti, H., Primarizky, H. and Hamid, I.S. 2022. Moringa oleifera leaf extract restored the diameter and thickness of the seminiferous tubules of rat (Rattus norvegicus) injected with Gentamicin. Ovozoa J. Anim. Reprod. 11(1), 15–21. Marques, P., Skorupskaite, K., Rozario, K.S., Anderson, R.A., and George, J.T. 2022. Physiology of GnRH and Gonadotropin Secretion. In Endotext, Feingold, K.R., Ed. MDText.com, Inc., South Dartmouth, MA.. Meles, D.K., Rachmawati, K., Hamid, I.S., Mustofa, I., Wurlina, W., Suwasanti, N., Putri, D.K.S.C. and Utama, S. 2022. Α-Tocopherol Prevents Sperm Apoptosis and Necrosis in Rats Exposed to 2,3,7,8-Tetrachlorodibenzo-p-dioxin. Vet. Med. Int. 2022(1), 3685686. Miller, W.L. and Bose, H.S. 2011. Early steps in steroidogenesis: intracellular cholesterol trafficking. J. Lipid Res. 52(12), 2111–2135. Mohlala, K., Offor, U., Monageng, E., Takalani, N.B. and Opuwari, C.S. 2023. Overview of the Effects of Moringa oleifera leaf extract on oxidative stress and male infertility: a review. Appl. Sci. 13(7), 4387. Monageng, E., Offor, U., Takalani, N.B., Mohlala, K. and Opuwari, C.S. 2023. a review on the impact of oxidative stress and medicinal plants on leydig cells. Antioxidants. (Basel. Switzerland). 12(8), 1559. Mustafa, H.N. 2023. Ameliorative potential of the quercetin on lead-induced testicular damage: morphohistometric and biochemical analysis. Afr. J. Urol. 29(1), 36. Ngizzah, N., Wurlina Wurlina., Poedji Hastutiek., Iwan Sahrial Hamid., Eka Pramyrtha Hestianah. and Lita Rakhma Yustinasari. 2023. Effect of ethanolic extract of Moringa oleifera leaves on the number of spermatogenic cells and Leydig cells of Gentamicin-induced rats. Ovozoa. J. Anim. Reprod. 12(2), 93–100. Nobossé, P., Fombang, E.N. and Mbofung, C.M.F. 2018. Effects of age and extraction solvent on phytochemical content and antioxidant activity of fresh Moringa oleifera L. leaves. Food Sci. Nutr. 6(8), 2188–2198. Obeng-Gyasi, E. 2018. Lead exposure and oxidative stress-a life course approach in U.S. Adults. Toxics 6(3), 42. Oduwole, O.O., Huhtaniemi, I.T. and Misrahi, M. 2021. The roles of luteinizing hormone, follicle-stimulating hormone and testosterone in spermatogenesis and folliculogenesis revisited. Int. J. Mol. Sci. 22(23), 12735. Oduwole, O.O., Peltoketo, H. and Huhtaniemi, I.T. 2018. Role of Follicle-Stimulating Hormone in Spermatogenesis. Front. Endocrinol. (Lausanne). 9(1), 763. Ogunlade, B., Jeje, S.O., Adelakun, S.A. and Akingbade, G.T. 2022. Moringa oleifera restored semen quality, hormonal profile, and testicular morphology against Highly Active Antiretroviral Therapy-induced toxicity in adult male Wistar rats. JBRA. Assist. Reprod. 26(1), 3–12. Ogunsola, O.A., Owalabi, J.O., Fabiyi, O.S., Nwobi, N.L., Faluyi, B. and Akinbola, A.S. 2017. Moringa plant parts consumption had effects on reproductive functions in male and female rat models. IOSR. J. Dent. Med. Sci. 16(10), 82–86. Panggalih, A., Susilowati, S., Maslachah, L., Ratnani, H. and Suprayogi, T.W. 2021. The effect of watermelon (Citrullus lanatus) rind ethanolic extract on the number of Leydig, Sertoli, and spermatogenic cells of rat (Rattus novergicus) exposed to heat. Ovozoa 10(1), 7–11. Pavuluri, H., Bakhtiary, Z., Panner Selvam, M.K. and Hellstrom, W.J.G. 2024. Oxidative stress-associated male infertility: current diagnostic and therapeutic approaches. Medicina (Kaunas. Lithuania). 60(6), 1008. Peñalver, R., Martínez-Zamora, L., Lorenzo, J.M., Ros, G. and Nieto, G. 2022. Nutritional and antioxidant properties of Moringa oleifera leaves in functional foods. Foods 11(8), 1107. Pratama, A.P.I., Susilowati, S., Maslachah, L., Ratnani, H. and Suprayogi, T.W. 2021. The effect of watermelon (Citrullus lanatus) rind ethanolic extract on the number of Leydig, Sertoli, and spermatogenic cells of rat (Rattus novergicus) exposed to heat. Ovozoa. J. Anim. Reprod. 10(1), 7–11. Qi, W., Qi, W., Xiong, D. and Long, M. 2022. Quercetin: its Antioxidant Mechanism, Antibacterial Properties and Potential Application in Prevention and Control of Toxipathy. Molecules (Basel. Switzerland). 27(19), 6545. Ragab, S.M.M., Almohaimeed, H.M., Alghriany, A.A.I., Khalil, N.S.A. and Abd-Allah, E.A. 2024. Protective effect of Moringa oleifera leaf ethanolic extract against uranyl acetate-induced testicular dysfunction in rats. Sci. Rep. 14(1), 932. Raj, K. and Das, A.P. 2023. Lead pollution: impact on environment and human health and approach for a sustainable solution. Environ. Chem. Ecotoxicol. 5(1), 79–85. Reda, R.M., Helmy, R.M.A., Osman, A., Ahmed, F.A.G., Kotb, G.A.M. and El-Fattah, A.H.A. 2023. The potential effect of Moringa oleifera ethanolic leaf extract against oxidative stress, immune response disruption induced by abamectin exposure in Oreochromis niloticus. Environ. Sci. Pollut. Res. 30(20), 58569–58587. Redza-Dutordoir, M. and Averill-Bates, D.A. 2016. Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica Et Biophysica Acta (BBA). Bioenergetics. 1863(12), 2977–2992. Revina, A.S., Iwan, S.H., Emy, K.S., Anwar, M.R., Eka, P.H. and Maslichah, M. 2022. Tomato (Lycopersicon esculentum Mill.) juice restored the number of Leydig cells, and the diameter of the seminiferous tubules of mice (Mus musculus) exposed to lead acetate. Ovozoa J. Anim. Reprod. 11(3), 123–129. Sani, A.H. and Amanabo, M. 2021. Lead: A concise review of its toxicity, mechanism and health effect. GSC Biol. Pharm. Sci. 15(1), 55–62. Shafiq, N.E., Mahdee, A.F. and Hasan, Z.Y.M. 2024. Leaf Extracts of Moringa oleifera cultivated in Baghdad: characterization and antimicrobial potential against endodontic pathogens. Sci. World J. 2024(1), 6658164. Shah, W., Khan, R., Shah, B., Khan, A., Dil, S., Liu, W., Wen, J. and Jiang, X. 2021. The Molecular Mechanism of Sex Hormones on Sertoli Cell Development and Proliferation. Front. Endocrinol. 12(1), 648141. Shahbaz, M., Naeem, H., Batool, M., Imran, M., Hussain, M., Mujtaba, A., Alsagaby, S.A., Al Abdulmonem, W., El-Ghorab, A.H., Ghoneim, M.M., Shaker, M.E., Abdelgawad, M.A. and Al Jbawi, E. 2024. Antioxidant, anticancer, and anti-inflammatory potential of Moringa seed and Moringa seed oil: a comprehensive approach. Food. Sci. Nutr. 12(9), 6157–6173. Singh, N., Kumar, A., Gupta, V.K. and Sharma, B. 2018. Biochemical and Molecular Bases of Lead-Induced Toxicity in Mammalian Systems and Possible Mitigations. Chem. Res. Toxicol. 31(10), 1009–1021. Srivastava, S., Pandey, V.K., Dash, K.K., Dayal, D., Wal, P., Debnath, B., Singh, R. and Dar, A.H. 2023. Dynamic bioactive properties of nutritional superfood Moringa oleifera: a comprehensive review. J. Agric. Food. Res. 14(1), 100860. Su, X., Lu, G., Ye, L., Shi, R., Zhu, M., Yu, X., Li, Z., Jia, X. and Feng, L. 2023. Moringa oleifera Lam.: a comprehensive review on active components, health benefits and application. RSC. Adv. 13(35), 24353–24384. Suede, S.H., Malik, A. and Sapra, A. 2023. Histology, Spermatogenesis. Treasure Island, FL: StatPearls Publishing; 2024. Available via https://www.ncbi.nlm.nih.gov/books/NBK553142/ Suhardono, S., Septiariva, I.Y., Sari, M.M. and Suryawan, I.W.K. 2022. Analysis of metal pollution index in waters in the central Java area, Indonesia. Aquat. Sci. Manag. 10(2), 35–41. Vergara-Jimenez, M., Almatrafi, M.M. and Fernandez, M.L. 2017. Bioactive Components in Moringa oleifera Leaves Protect against Chronic Disease. Antioxidants (Basel). 6(4), 91. Wang, H.Q., Zhang, W.D., Yuan, B. and Zhang, J.B. 2021. Advances in the Regulation of Mammalian Follicle-Stimulating Hormone Secretion. Animals 11(4), 1134. Wardoyo, S., Nurjazuli, N. and Darundiati, Y.H. 2022. Lead exposure and stunting incidents in children aged 3–5 years in Pontianak City, West Kalimantan, Indonesia. Toxicol. Anal. Clin. 34(2), 111–116. Weydert, C.J. and Cullen, J.J. 2010. Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat. Protoc. 5(1), 51–66. Wurlina, W., Mustofa, I., Meles, D.K., Khairullah, A.R., Akintunde, A.O., Rachmawati, K., Suwasanti, N., Putra, D.M.S., Mulyati, S., Utama, S., Khoiriyah, U., Tyarraushananda Defvyanto, B.R., Heriana, S.F., Riwu, K.H.P., Ahmad, R.Z. and Riwu, A.G. 2025. Restoration of sperm quality in lead acetate-induced rats via treatment with Moringa oleifera leaf extract. Open Vet. J. 15(1), 416–427. Wurlina, W., Mustofa, I., Meles, D.K., Mulyati, S., Putri, D.K.S.C. and Suwasanti, N. 2021a. Administration of the α-tocopherol for repairing testicle histological damage in rats exposed to dioxin. Thai J. Vet. Med. 51(2), 293–301. Wurlina, Racmawati, K., Utama, S., Mahasri, G., Suwasanti, N. and Putri, D.K.S.C. 2021b. Recovery and development of creative economy processed sea products in coastal bulak Surabaya in new normal era for supporting economic growth. Bull. Community Serv. 1(1), 12–20. Wurlina, W., Mustofa, I., Meles, D.K., Safitri, E., Susilowati, S., Mulyati, S., Utomo, B. and Utama, S. 2022. Α-Tocopherol restores semen quality in rats exposed to 2,3,7,8-tetrachlorodibenzo-p-dioxin. Vet. World. 15(2), 316–323. Yang, J., Li, X., Xiong, Z., Wang, M. and Liu, Q. 2020. Environmental Pollution Effect Analysis of Lead Compounds in China Based on Life Cycle. Int. J. Environ. Res. Public. Health. 17(7), 2184. Ye, R.J., Yang, J.M., Hai, D.M., Liu, N., Ma, L., Lan, X.B., Niu, J.G., Zheng, P. and Yu, J.Q. 2020. Interplay between male reproductive system dysfunction and the therapeutic effect of flavonoids. Fitoterapia 147(1), 104756. Yu, W., Zhu, H., Huang, R., Yan, B., Xu, B., Shi, Y., Mao, J., Liu, Z. and Wang, J. 2024. Roles of Cyt-c/Caspase-9/Caspase-3/Bax/Bcl-2 pathway in Cd-induced testicular injury in rats and the protective effect of quercetin. Toxicon 237(1), 107561. Zhu, L., Han, M.B., Gao, Y., Wang, H., Dai, L., Wen, Y. and Na, L.X. 2015. Curcumin triggers apoptosis via upregulation of Bax/Bcl-2 ratio and caspase activation in SW872 human adipocytes. Mol. Med. Rep. 12(1), 1151–1156. Zirkin, B.R. and Papadopoulos, V. 2018. Leydig cells: formation, function, and regulation. Biol. Reprod. 99(1), 101–111. | ||
| How to Cite this Article |
| Pubmed Style Wurlina W, Mustofa I, Mulyati S, Khairullah AR, Meles DK, Akintunde AO, Suwasanti N, Khoiriyah U, Hardyani G, Kusnadi DR, Hidajati N, Sugihartuti R, Utomo B, Safitri E, Maslachah L, Kurnijasanti R, Kuncorojakt S. Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Vet. J.. 2025; 15(12): 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 Web Style Wurlina W, Mustofa I, Mulyati S, Khairullah AR, Meles DK, Akintunde AO, Suwasanti N, Khoiriyah U, Hardyani G, Kusnadi DR, Hidajati N, Sugihartuti R, Utomo B, Safitri E, Maslachah L, Kurnijasanti R, Kuncorojakt S. Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. https://www.openveterinaryjournal.com/?mno=250279 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.38 AMA (American Medical Association) Style Wurlina W, Mustofa I, Mulyati S, Khairullah AR, Meles DK, Akintunde AO, Suwasanti N, Khoiriyah U, Hardyani G, Kusnadi DR, Hidajati N, Sugihartuti R, Utomo B, Safitri E, Maslachah L, Kurnijasanti R, Kuncorojakt S. Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Vet. J.. 2025; 15(12): 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 Vancouver/ICMJE Style Wurlina W, Mustofa I, Mulyati S, Khairullah AR, Meles DK, Akintunde AO, Suwasanti N, Khoiriyah U, Hardyani G, Kusnadi DR, Hidajati N, Sugihartuti R, Utomo B, Safitri E, Maslachah L, Kurnijasanti R, Kuncorojakt S. Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 Harvard Style Wurlina, W., Mustofa, . I., Mulyati, . S., Khairullah, . A. R., Meles, . D. K., Akintunde, . A. O., Suwasanti, . N., Khoiriyah, . U., Hardyani, . G., Kusnadi, . D. R., Hidajati, . N., Sugihartuti, . R., Utomo, . B., Safitri, . E., Maslachah, . L., Kurnijasanti, . R. & Kuncorojakt, . S. (2025) Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Vet. J., 15 (12), 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 Turabian Style Wurlina, Wurlina, Imam Mustofa, Sri Mulyati, Aswin Rafif Khairullah, Dewa Ketut Meles, Adeyinka Oye Akintunde, Niluh Suwasanti, Ulul Khoiriyah, Gusti Hardyani, Deny Rama Kusnadi, Nove Hidajati, Rahmi Sugihartuti, Budi Utomo, Erma Safitri, Lilik Maslachah, Rochmah Kurnijasanti, and Suryo Kuncorojakt. 2025. Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Veterinary Journal, 15 (12), 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 Chicago Style Wurlina, Wurlina, Imam Mustofa, Sri Mulyati, Aswin Rafif Khairullah, Dewa Ketut Meles, Adeyinka Oye Akintunde, Niluh Suwasanti, Ulul Khoiriyah, Gusti Hardyani, Deny Rama Kusnadi, Nove Hidajati, Rahmi Sugihartuti, Budi Utomo, Erma Safitri, Lilik Maslachah, Rochmah Kurnijasanti, and Suryo Kuncorojakt. "Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure." Open Veterinary Journal 15 (2025), 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 MLA (The Modern Language Association) Style Wurlina, Wurlina, Imam Mustofa, Sri Mulyati, Aswin Rafif Khairullah, Dewa Ketut Meles, Adeyinka Oye Akintunde, Niluh Suwasanti, Ulul Khoiriyah, Gusti Hardyani, Deny Rama Kusnadi, Nove Hidajati, Rahmi Sugihartuti, Budi Utomo, Erma Safitri, Lilik Maslachah, Rochmah Kurnijasanti, and Suryo Kuncorojakt. "Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure." Open Veterinary Journal 15.12 (2025), 6541-6551. Print. doi:10.5455/OVJ.2025.v15.i12.38 APA (American Psychological Association) Style Wurlina, W., Mustofa, . I., Mulyati, . S., Khairullah, . A. R., Meles, . D. K., Akintunde, . A. O., Suwasanti, . N., Khoiriyah, . U., Hardyani, . G., Kusnadi, . D. R., Hidajati, . N., Sugihartuti, . R., Utomo, . B., Safitri, . E., Maslachah, . L., Kurnijasanti, . R. & Kuncorojakt, . S. (2025) Protective potential of Moringa oleifera leaf extract on rat testicular histology following lead acetate exposure. Open Veterinary Journal, 15 (12), 6541-6551. doi:10.5455/OVJ.2025.v15.i12.38 |