| Research Article | ||
Open Vet. J.. 2025; 15(12): 6814-6822 Open Veterinary Journal, (2025), Vol. 15(11): 6814-6822 Review Article Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virusEqbal Salman Najem1, Ehssan N. Jasim AL-Obaidy2, Ali Majhool Kane3, Ali Ibrahim Ali Al-Ezzy4*1Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Diyala, Baqubah, Iraq 2Department of Physiology Biochemistry and Pharmacology, College of Veterinary Medicine, University of Diyala, Baqubah, Iraq 3Department of Pathology, College of Veterinary Medicine, University of Kufa, Kufa, Iraq 4Department of Pathology, College of Veterinary Medicine, University of Diyala, Baqubah, Iraq *Corresponding Author: Ali Ibrahim Ali Al-Ezzy. Department of Pathology, College of Veterinary Medicine, University of Diyala, Baqubah, Iraq. Email: ali.ib [at] uodiyala.edu.iq Submitted: 21/04/2025 Revised: 30/09/2025 Accepted: 14/10/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
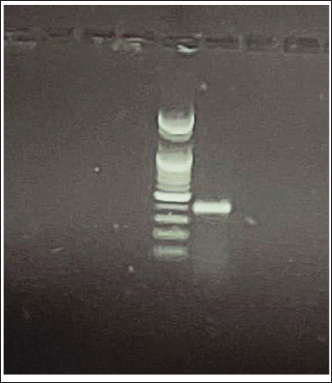
AbstractBackground: A serious mortality epizootic affected the populations of common carp (Cyprinus carpio L.) in several fish farms that are located at Al–jadeda–Diyala province, Iraq, during the autumn 2022. The diseased fish were showing clinical signs associated with a viral cause. Aim: To conduct molecular detection using polymerase chain reaction (PCR) to examine the carp interstitial nephritis and gill necrosis virus (CNGV) outbreak, and characterize histopathological changes in major organs, as well as serum liver enzyme profile and kidney function profile. Methods: A total of 50 live common carp (Cyprinus carpio L.) with weight of 128 to 2,300 g that showed clinical signs such as necrosis on the gills, overproduction of mucus, or pallor in the skin were randomly sampled from five positive farms. Blood samples were obtained from the caudal vein for measurement of the level of biochemical liver enzymes (alanine transaminase and aspartate amino transferase) markers for kidney function (creatinine). At 15 d postimmunization, fish (n=3) from each group were euthanized, and tissues (gill, liver, spleen, kidney, and intestine) were aseptically removed. Hematoxylin/eosin-stained histopathology tissues were processed by routine methods after fixation with 10% neutral buffered formaldehyde. Gill tissue was maintained in phosphate-buffered saline for DNA extraction and PCR amplification to a region of the conserved genome between CNGVs for molecular analysis. Results: The positive presence of CNGV in the fish samples was confirmed by PCR. Histopathological analysis showed extensive lesions in all the analyzed organs, such as severe gill necrosis, hepatitis, splenitis, extensive interstitial nephritis, and enteritis. They were associated with marked increases in serum liver enzyme activities and alterations of renal end-points. Conclusion: This is the first molecular confirmation of CNGV infection as the cause of an outbreak of disease in common carp in Iraq. The virus caused severe systemic lesions with characteristic histopathological alterations in major organs and remarkable hepatic and renal function impairment. These results bring CNGV as a serious new threat to carp farming in the region. Keywords: Carp interstitial nephritis and gill necrosis virus, Histopathology, Blood biochemistry, Common carp. IntroductionCarp interstitial nephritis and gill necrosis virus (CNGV) is an important pathogen in both common carp (Cyprinus carpio L.) and koi fish (Dishon et al., 2005). CNGV is a large DNA virus with physical properties similar to members of the Herpesviridae family and carries an equally large linear double-stranded DNA genome (approximately 280 kbp) (Uma and A, 2025). The virus has been recorded at multiple farms in Europe, Asia, and Africa since the early reports, making it a serious threat to worldwide carp farming (Bavarsad et al., 2024). CNGV infections are often associated with lethal diseases, which cause catastrophic mortalities that can reach higher than 80% in affected populations (Gilad et al., 2003). Outbreaks usually take place in spring and autumn at water temperatures of 16°C–25°C (Razzaq et al., 2025). Infected fish show unspecific signs such as depression, anorexia, and erratic swimming that evolve to more pathognomonic including severe gill necrosis, higher secretion of dermal/branchial mucus; skin hemorrhage, paleness of the skin (dermal pallor), and Enophtalmia (sunken eye) (Uma and A, 2025). An important aspect of CNGV epidemiology is the infection’s capacity to establish a latent infection in carriers without clinical signs (Boutier et al., 2019). Similarly, to other viral infections, survivors are considered as potential virus carriers and constitute an essential mode for the persistence and spreading of the virus among fish populations (Eide et al., 2011). The economic impact of CNGV is substantial, affecting a cornerstone of global aquaculture. According to the FAO Yearbook (2004), the common carp is the third most cultivated freshwater fish species worldwide, with major production centers in South America, Russia, Europe, Asia, and Africa (Taal et al., 2004). Furthermore, the vibrant koi carp, a colored variety of C. carpio, holds significant value in the international ornamental fish trade, making CNGV outbreaks a threat to both food security and a lucrative industry (Eide et al., 2011). Hematological and biochemical analyses can be used to effectively assess fish health, along with their responses against pathogens like CNGV. Changes in blood parameters act as sensitive markers for the diagnosis of disease, measurement of physiological stress, or the extent of an animal’s exposure to environmental contaminants (Thrall et al., 2012; Bojarski et al., 2025). For detecting infectious diseases and their systemic cascades, clinical biochemistry is a useful option (Najem et al., 2020). Autumn 2022, outbreaks of mortalities were observed in common carp at a number of fish-farm sites in Al–Jadeda District, Diyala Province, Iraq. This aside, the field observation and pathological examination strongly suggested that CNGV was an aetiological agent for high mortalities. Hence, the current study was carried out to (1) confirm molecularly CNGV infection in diseased carp population using polymerase chain reaction (PCR), (2) follow paired changes of liver enzyme and kidney function parameters in response to virus, and (3) take down these histopathological alterations in different organs, i.e., the gill, liver spleen kidney and intestine controlled by this virus. Materials and MethodsFish samplingFifty clinically affected common carp (Cyprinus carpio) producing gill necrosis, excessive dermal and branchial mucus secretion, in addition to dermal pallor, were randomly obtained during November 2022 from five fish farms located at Al-Jadeda of Diyala Province, Iraq. The weight of the individuals ranged between 128 and 2,300 g; all were bagged in oxygen, carried to the laboratory, and euthanized after approved ethical proceedings. Blood collection and serum preparationBlood samples were aseptically collected from the caudal vein with sterile syringes and placed in evacuated serum gel tubes. Blood was clotted at room temperature, and then centrifuged at 3,000 × g for 15 minutes to collect the serum. The serum aliquots obtained were kept at −20°C until biochemical assay could be carried out. Biochemical analysisThe level of key liver enzymes (serum alanine transaminase, ALT, and aspartate amino transferase AST) activity was determined calorimetrically using a commercial method (Najem et al., 2020). Moreover, according to the manufacturers’ instructions, renal function markers (urea and creatinine) were measured using commercial diagnostic kits (Najem et al., 2020). Given the high prevalence of virus within the study site at the time of investigation, with most of the fish population showing signs of clinical infection, it was not possible to establish a reliable contemporaneous control group. Asymptomatic fish regardless that could not be negative controls, since they can be the conduit of the virus. As such, measurements from a previously compiled control cohort (Najem et al., 2020) were utilized for comparison in the present study. Histopathological examinationAfter dissection, five tissues were harvested from the major organs, including gill, liver, spleen, kidney, and intestine. Tissues were fixed in 10% neutral buffered formalin and allowed to fix for at least 48 hours; tissues were processed (dehydrated, cleared, and embedded in paraffin wax) based on routine histological techniques. The thickness at 4–5 µm was sliced using a microtome mounted on glass slides, stained with hematoxylin and eosin (H&E) for microscopic examination. Histological evaluation, histopathology was conducted based on previous reports (Gunasegaran, 2016; Najem et al., 2024). Molecular detection of CNGV by PCRGill tissue samples from each fish were preserved in phosphate-buffered saline (pH 7.0) for molecular diagnosis. Genomic DNA was extracted from these tissues using the QIAamp DNA Mini Kit (QIAGEN, USA), following the manufacturer’s instructions. The extracted DNA was subsequently used as a template in a conventional PCR assay targeting the koi herpes virus major capsid protein as a specific genomic region of the CNGV for infection confirmation. The primer for the major capsid protein of the virus used in the PCR product was designed using the Primer-Blast tool: (F: TCTCACCCAGTACACCACCA, R: GTTCATGGCGCCAAAGT AGT), annealing temperature 59, product size 382 bp) for koi herpes virus major capsid protein Statistical analysisData are expressed as means ± standard error. Data were analyzed using the “Statistical Package for the Social Sciences (version 18.0)”. The t-test was used for comparison between groups. Significant difference at p < 0.05 (Hameed et al., 2020; Hameed et al., 2024). Ethical approvalAll specimens were immediately transported in oxygenated bags to the laboratory and subsequently euthanized following the approved ethical guidelines by the College of Veterinary Medicine, University of Diyala, Iraq. No. DP, 2023-01, in 1-Sep-2023. ResultsMolecular detection of CNGVCNGV nucleic acid was successfully detected via PCR in gill tissues from 4 of the 50 sampled fish (8% prevalence), while the remaining 46 cases were negative (Fig. 1).
Fig. 1. Positive sample for CNGV. Biochemical analysisLiver enzymes: Table 1 presents the serum levels of liver enzymes. No significant difference (p > 0.05) was observed in ALT activity among the control, PCR-positive, and PCR-negative groups. In contrast, a statistically significant difference (p ≤ 0.05) was found in AST activity between these groups. Table 1. Liver enzyme (ALT and AST) in blood of infected common carp.
Kidney function markers: Table 2 summarizes the results for renal function parameters. Urea levels did not differ significantly (p > 0.05) between the control, PCR-positive, and PCR-negative groups. However, a significant difference (p < 0.05) was detected in creatinine levels across the groups. Table 2. Urea and creatinine in blood of infected common carp.
Histopathological findingsLiver:
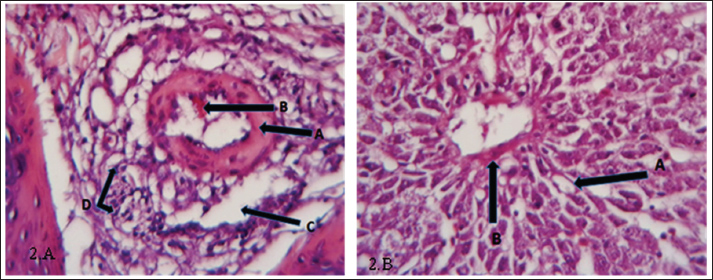
Fig. 2. (a) Histopathological section of liver of common carp showing fibrosis (A), hemorrhage(B), edema (C) inflammatory cells infiltration (D). H&E ×40.; (b) Histopathological section of the liver of common carp appear degeneration (A) and fibrosis (B). H&E ×20.
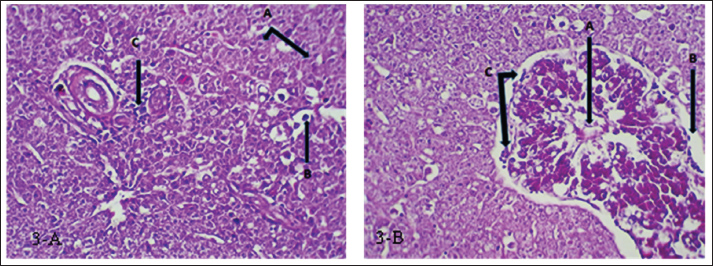
Fig. 3. (a) Histopathological section of liver of common carp showing severe necrosis (A) with fatty degeneration (B) and infiltration of inflammatory cells (C). H&E ×20.; (b) Histopathological section of liver of common carp showing necrosis in the bile duct (A) and edema surrounded the bile duct (B) with mononuclear cells infiltration (C). H&E ×20. Kidney:
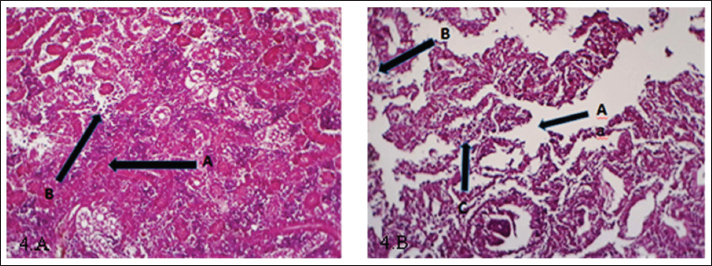
Fig. 4. (a) Histopathological section of kidney of common carp showing damage of glomeruli and tubules (A), inflammatory cells infiltration (B). H&E ×20.; (b) Histopathological section of kidney of common carp showing edema (A) and sloughing (B) inflammatory cells infiltration (C). H&E ×10.
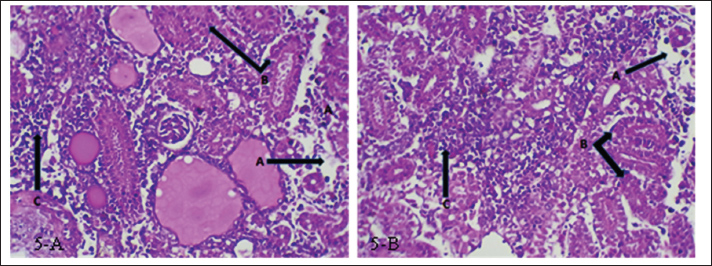
Fig. 5. (a and b) Histopathological section of kidney of common carp showing necrosis (A), cellular swelling (B), inflammatory cells infiltration (C). H&E ×20. Spleen:
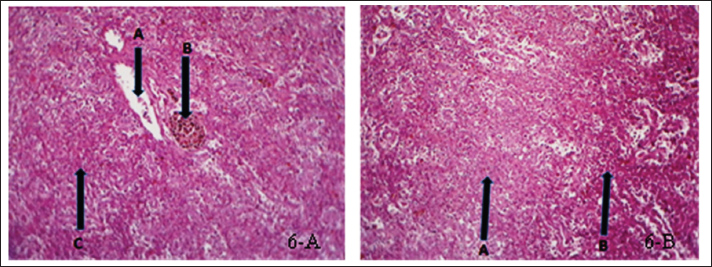
Fig. 6. (a) Histopathological section of spleen of common carp showing edema (A), hemosidrein (B) and damage in red pulp (C). H&E ×10.; (b) Histopathological section of spleen of common carp showing damage in red pulp (A) and inflammatory cells infiltration (B). H &E ×10.
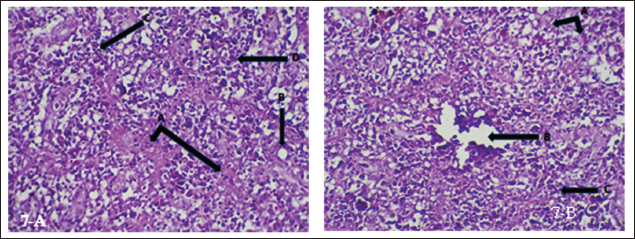
Fig. 7. (a) Histopathological section of spleen of common carp showing necrosis (A), vacuolation (B) apoptosis (C) and hemosiderin precipitation (D). H&E ×20.; (b) Histopathological section of spleen of common carp showing necrosis (A), edema (B) and inflammatory cells infiltration (C). H&E ×20. Intestine:
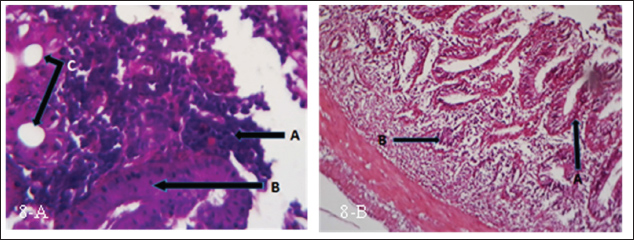
Fig. 8. (a) Histopathological section of intestine of common carp showing inflammatory cells infiltration (A), thickening in lamina properia (B) and vacuolation (C). H&E ×10.; (b) Histopathological section of intestine of common carp showing shortening in lamina properia (A) and inflammatory cells infiltration (B). H&E ×10.
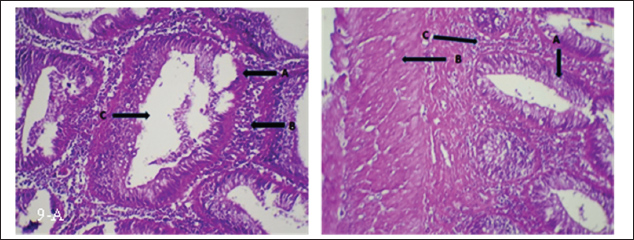
Fig. 9. (a) Histopathological section of intestine of common carp showing hyperplasia of intestinal gland (A), inflammatory cells infiltration (B) and complete vacuolation (C). H&E ×20.; (b) Histopathological section of intestine of common carp showing enlargement of intestinal gland (A) and thicked of muscular layer (B) and inflammatory cells infiltration (C). H&E ×20. Gill:
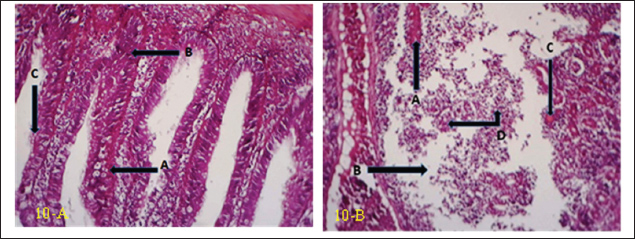
Fig. 10. (a) Histopathological section of gill of common carp showing hemorrhage (A), edema (B) and blood congestion (C) with infiltration of mononuclear cells (D). H&E ×20.; (b) Histopathological section of gill of common carp showing increase number of mucous cells (A), blood congestion (B) and elongated of secondary lamellae (C). H&E ×10.
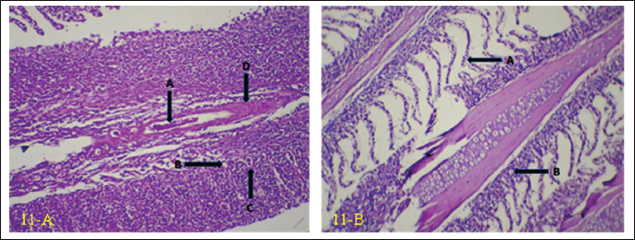
Fig. 11. (a) Histopathological section of gill of common carp showing hemorrhage (A), vacuolation (B) apoptosis (C) with hyperplasia of primary lamellae (D). H&E ×20.; (b) Histopathological section of gill of common carp showing epithelial lifting in secondary lamellae (A), with epithelial vacuolation (B). H&E ×10. DiscussionCNGV is a significant pathogen that affects common carp, with its prevalence peaking during autumn and spring. Outbreaks are highly dependent on water temperature, typically occurring within a range of 16°C–25°C. The virus has a global distribution and is responsible for substantial economic losses in common carp aquaculture (Mancheva et al., 2021). This study provides crucial confirmation of CNGV infection and characterizes the associated alterations in liver enzymes, kidney function, and histopathological changes in key organs, including the gill, liver, spleen, kidney, and intestine. Due to the widespread prevalence of the virus within the study area during the investigation period, whereby a majority of the fish population exhibited clinical signs of infection, establishing a valid contemporaneous control group in the current study was not feasible. Fish that appeared asymptomatic could not serve as true negative controls, as they are potential carriers of the virus. CNGV is known to persist in a latent state without manifesting overt clinical disease (Boutier et al., 2019). Consequently, fish that survive an outbreak are considered subclinical viral carriers (Eide et al., 2011). Therefore, data from a control group established in a previous study (Najem et al., 2020) were utilized for comparative analysis in the present investigation. CNGV transmission occurs through water, sediment, fecal material, and direct contact with infected fish (Uma, 2025). The gills, gut, and kidneys are believed to be the primary portals of entry (Haenen and Hedrick, 2006) pathogenicity of CNGV is primarily characterized by gill necrosis and interstitial nephritis (Ronen et al., 2003; Pikarsky et al., 2004). These investigations have documented necrotic changes and hemorrhage in the gills, often accompanied by renal enlargement (Miyazaki et al., 2008; Mouchira, 2011). Additional clinical signs, including swollen and necrotic gill tissue, excessive mucus production, skin discoloration, and splenomegaly, have been described (Walster, 1999; Bavarsad et al., 2024). The results of this study align with these established findings, confirming the presence of CNGV via PCR and correlating the infection with elevated liver enzyme activities, impaired kidney function, and significant histopathological lesions in the aforementioned organs. The high environmental transmission potential of CNGV in pond systems probably results, at least in part, from its stability and the fact that the virus remains infective for at least 4 hours in water (Dorfman et al., 2024). Although the precise route of entry into gilthead seabream—intestinal or branchial—has not been unequivocally established (Perelberg et al., 2005), histopathological findings obtained here and elsewhere lend support to the postulate that gills act as a principal site where viral entry/infections are initiated. This primary replication results in mucosal sloughing and necrosis, and severe gill pathology is probably a significant cause of the overall sick feeling of fish. A complete description of host-virus interactions in gill tissue is required to understand the mechanisms leading up to such damage. The gill virus replication and subsequent release into the water column provide a novel mechanism that can account for the efficient, rapid spread of viral hemorrhagic septicemia within populations. Following initial infection, the virus is thought to be rapidly transported to the kidneys, potentially via infected white blood cells (Wolf, 2019), where it replicates within leukocytes and renal epithelial cells, culminating in severe Interstitial Nephritis Syndrome (INS) (Gilad et al., 2004; Negenborn et al., 2015; Hadfield, 2021). This leukocyte-associated viremia may also explain the relatively low PCR positivity rate observed in the current study, as the virus might be sequestered within cellular components rather than freely circulating in all tissues at the time of sampling. Gill tissue samples obtained from clinically affected fish were subjected to PCR analysis to detect CNGV. Among these, viral nucleic acid was successfully amplified in four samples, confirming the presence of active infection. However, the negative PCR results in the remaining 46 samples should not be interpreted as definitive evidence of the absence of infection. A negative result from gill tissue does not preclude a systemic infection, as the virus may have already disseminated to and be primarily replicating in other internal organs, such as the kidney or intestine. This viral kinetics profile is supported by what is known about CNGV pathogenesis. After initial reception, the virus is quickly transferred to the kidneys, frequently via the gills. Here, it invades leukocytes, inducing severe interstitial nephritis (Gilad et al., 2004; Negenborn et al., 2015; Hadfield, 2021). This extremely fast change of viral tropism and load to a non-primary infection site offers a mechanism to explain the low occurrence rate of positive PCR gill results observed in this study. Virus sequestration within leukocytes raises the likelihood of leukocyte-associated viremia, a route by which pathogens can be effectively delivered to visceral organs (Wolf, 2019). In conclusion, this is the first molecular evidence of CNGV being the aetiological agent of the outbreak in cultured common carp in Iraq. The virus was accompanied by severe systemic pathology, such as obvious histopathological alterations in important organs and marked hepatic and renal functional disorders. These results reveal CNGV as an acute emerging threat to carp aquaculture in the region. AcknowledgmentsSpecial thanks to the Department of Pathology, College of Veterinary Medicine, University of Diyala, for providing all the necessary facilities for this work. Conflict of interestThe authors declare no conflict of interest. FundingNo funding agencies are available. Authors’ contributionsAll authors equally contributed to the planning, laboratory work, manuscript preparation, and statistical analysis. Data availabilityAll related data are presented in the text. ReferencesBavarsad, M., Abed-Elmdoust, A., Tabandeh, M., Farahmand, H., Alishahi, M., Mirvaghefi, A., Avazeh, A., Adel, M., Jafari, A. and Zorriehzahra, M. 2024. Cyprinid herpesvirus 3 (CyHV-3), koi herpes virus disease (KHVD) and their current status in Iran: a review. Iranian J. Fisheries Sci. 23(5), 783–802. Bojarski, B., Witeska, M. and Kondera, E. 2025. Blood biochemical biomarkers in fish toxicology—a review. Animals 15(7), 965. Boutier, M., Gao, Y., Donohoe, O. and Vanderplasschen, A. 2019. Current knowledge and future prospects of vaccines against cyprinid herpesvirus 3 (CyHV-3). Fish. Shellfish. Immunol. 93, 531–541. Dishon, A., Perelberg, A., Bishara-Shieban, J., Ilouze, M., Davidovich, M., Werker, S. and Kotler, M. 2005. Detection of carp interstitial nephritis and gill necrosis virus in fish droppings. Appl. Environ. Microbiol. 71(11), 7285–7291. Dorfman, B., Marcos-Hadad, E., Tadmor-Levi, R. and David, L. 2024. Disease resistance and infectivity of virus susceptible and resistant common carp strains. Scientific Rep. 14(1), 4677. Eide, K., Miller-Morgan, T., Heidel, J., Bildfell, R. and Jin, L. 2011. Results of total DNA measurement in koi tissue by koi herpes virus real-time PCR. J. Virol. Methods 172(1-2), 81–84. Gilad, O., Yun, S., Adkison, M.A., Way, K., Willits, N.H., Bercovier, H. and Hedrick, R.P. 2003. Molecular comparison of isolates of an emerging fish pathogen, koi herpesvirus, and the effect of water temperature on mortality of experimentally infected koi. J. Gen. Virol. 84(10), 2661–2667. Gilad, O., Yun, S., Zagmutt-Vergara, F., Leutenegger, C., Bercovier, H. and Hedrick, R. 2004. Concentrations of a Koi herpesvirus (KHV) in tissues of experimentally-infected Cyprinus carpio koi as assessed by real-time TaqMan PCR. Dis. Aquatic Organisms 60(3), 179–187. Gunasegaran, J. 2016. Textbook of histology and A practical guide-E-Book, 3rd ed. New Delhi, India: Elsevier Health Sciences. Hadfield, C.A. 2021. Viral diseases. In Clinical guide to fish medicine. Eds., Hadfield, M.A. and Clayton, L.A. Hoboken, NJ: John Wiley & Sons, vol. 1, pp: 407–30. Haenen, O. and Hedrick, R. 2006. Koi herpesvirus workshop. Bulletin-European. Assoc. Fish Pathologists 26(1), 26. Hameed, M.S., Al-Ezzy, A.I.A., Jalil, W.I. and Al-Khalidi, A.A.H. 2020. Physiological protective effects of ascorbic acid versus Dl-A-tocopheryl acetate–sodium selenite combination in mice under experimental sodium nitrate intoxication. Biochem. Cell. Arch. 20(1), 2593–2601. Hameed, M.S., Hassoon, S.J., Mahmood, M.A. and Al-Ezzy, A.I.A. 2024. Physiological effect of multivitamins supplementation on hematological parameters, lipid profile, hepato-renal function of ross 308 broilers. Assiut. Vet. Med. J. 70(183), 585–595. Mancheva, K., Danova, S., Vilhelmova-Ilieva, N., Dobreva, L., Kostova, K., Simeonova, L. and Atanasov, G. 2021. Viral pathogens with economic impact in aquaculture. Acta Microbiol. Bulgarica 37, 111–121. Miyazaki, T., Kuzuya, Y., Yasumoto, S., Yasuda, M. and Kobayashi, T. 2008. Histopathological and ultrastructural features of Koi herpesvirus (KHV)-infected carp Cyprinus carpio, and the morphology and morphogenesis of KHV. Dis. Aquatic Organisms 80(1), 1–11. Mouchira, M. 2011. Histopathological studies in experimentally infected koi carp (Cyprinus carpio koi) with koi herpesvirus in Japan. World J. Fish. Mar. Sci. 3, 252–259. Najem, E.S., Al-Shammari, S.M.H., Ismail, M.M., Al-Ezzy, A.I.A., Ibrahim, S.N. and Hassen, D.H. 2020. Assessment the effect of Saccharomyces cerevisiae on body biochemical composition of common carp (Cyprinus carpio L). Biochem. Cell.. Arch. 20(1), 2629–2634. Najem, E.S., Hussein, S.A., Kane, A.M. and Al-Ezzy, A.I.A. 2024. Bioaccumulation of lead, arsenic, and mercury in vital organs of common carp (Cyprinus carpio L.): assessment of pathological effects and possible hazards associated with human consumption. Open. Vet. J. 14(11), 2780. Negenborn, J., Van Der Marel, M.C., Ganter, M. and Steinhagen, D. 2015. Cyprinid herpesvirus-3 (CyHV-3) disturbs osmotic balance in carp (Cyprinus carpio L.)—a potential cause of mortality. Vet. Microbiol. 177(3-4), 280–288. Perelberg, A., Ronen, A., Hutoran, M., Smith, Y. and Kotler, M. 2005. Protection of cultured Cyprinus carpio against a lethal viral disease by an attenuated virus vaccine. Vaccine 23(26), 3396–3403. Pikarsky, E., Ronen, A., Abramowitz, J., Levavi-Sivan, B., Hutoran, M., Shapira, Y., Steinitz, M., Perelberg, A., Soffer, D. and Kotler, M. 2004. Pathogenesis of acute viral disease induced in fish by carp interstitial nephritis and gill necrosis virus. J. Virol. 78(17), 9544–9551. Razzaq, H., Haroon, W., Fatima, S.A., Ullah, S., Waheed, S.F., Ali, M., Javaid, T. and Ashfaq, K. 2025. Introduction to koi herpes virus (KHV) disease. In Diseases across life: from humans to land and sea. Eds., Ismael, S.S., Nisa, Z.U. and Aziz, S. Faisalabad, Pakistan: Unique Scientific Publishers, pp: 277–83. Ronen, A., Perelberg, A., Abramowitz, J., Hutoran, M., Tinman, S., Bejerano, I., Steinitz, M. and Kotler, M. 2003. Efficient vaccine against the virus causing a lethal disease in cultured Cyprinus carpio. Vaccine 21(32), 4677–4684. Taal, C., Klok, A., Oostenbrugge J v., Smit, M., Wijk M v. and Wilde J d. 2004. Fishery statistics 2003 (Vol. 95Den Haag, Netherlands: Landbouw-Economisch Instituut (LEI), 95. Thrall, M.A., Weiser, G., Allison, R.W. and Campbell, T.W. 2012. Veterinary hematology and clinical chemistry, 2nd ed. Ames, IA: John Wiley & Sons. Uma, A. 2025. Pathogen and disease transmission in aquatic animals. In Management of Fish Diseases. Eds., Mallik, S.N. and Pandey, S.K. Singapore: Springer, 1, pp: 19–55. Walster. 1999. Clinical observations of severe mortalities in koi carp, Cyprinus carpio, with gill disease. Fish. Vet. J. 3, 54–58. Wolf, K. 2019. Fish viruses and fish viral diseases. Ithaca, NY: Cornell University Press. | ||
| How to Cite this Article |
| Pubmed Style Najem ES, Al-obaidy ENJ, Kane AM, Al-ezzy AIA. Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Vet. J.. 2025; 15(12): 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 Web Style Najem ES, Al-obaidy ENJ, Kane AM, Al-ezzy AIA. Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. https://www.openveterinaryjournal.com/?mno=242927 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.61 AMA (American Medical Association) Style Najem ES, Al-obaidy ENJ, Kane AM, Al-ezzy AIA. Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Vet. J.. 2025; 15(12): 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 Vancouver/ICMJE Style Najem ES, Al-obaidy ENJ, Kane AM, Al-ezzy AIA. Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 Harvard Style Najem, E. S., Al-obaidy, . E. N. J., Kane, . A. M. & Al-ezzy, . A. I. A. (2025) Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Vet. J., 15 (12), 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 Turabian Style Najem, Eqbal Salman, Ehssan N. Jasim Al-obaidy, Ali Majhool Kane, and Ali Ibrahim Ali Al-ezzy. 2025. Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Veterinary Journal, 15 (12), 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 Chicago Style Najem, Eqbal Salman, Ehssan N. Jasim Al-obaidy, Ali Majhool Kane, and Ali Ibrahim Ali Al-ezzy. "Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus." Open Veterinary Journal 15 (2025), 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 MLA (The Modern Language Association) Style Najem, Eqbal Salman, Ehssan N. Jasim Al-obaidy, Ali Majhool Kane, and Ali Ibrahim Ali Al-ezzy. "Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus." Open Veterinary Journal 15.12 (2025), 6814-6822. Print. doi:10.5455/OVJ.2025.v15.i12.61 APA (American Psychological Association) Style Najem, E. S., Al-obaidy, . E. N. J., Kane, . A. M. & Al-ezzy, . A. I. A. (2025) Histopathological, liver enzymes, and kidney function anomalies in fish infected with carp interstitial nephritis and gill necrosis virus. Open Veterinary Journal, 15 (12), 6814-6822. doi:10.5455/OVJ.2025.v15.i12.61 |