| Research Article | ||
Open Vet. J.. 2025; 15(12): 6527-6540 Open Veterinary Journal, (2025), Vol. 15(12): 6527-6540 Research Article Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley ratsYos Adi Prakoso1, Krestel Joy Viernes Isla2 and Agustina Dwi Wijayanti3*1Department of Pharmacology, Faculty of Veterinary Medicine, University of Wijaya Kusuma Surabaya, Surabaya, Indonesia 2College of Veterinary Medicine, Tarlac Agricultural University, Tarlac, Philippines 3Department of Pharmacology, Faculty of Veterinary Medicine, University of Gadjah Mada, Yogyakarta, Indonesia *Corresponding Author: Agustina Dwi Wijayanti. Department of Pharmacology, Faculty of Veterinary Medicine, University of Gadjah Mada, Yogyakarta, Indonesia. Email: wagustinadwi [at] gmail.com Submitted: 17/01/2025 Revised: 15/10/2025 Accepted: 03/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
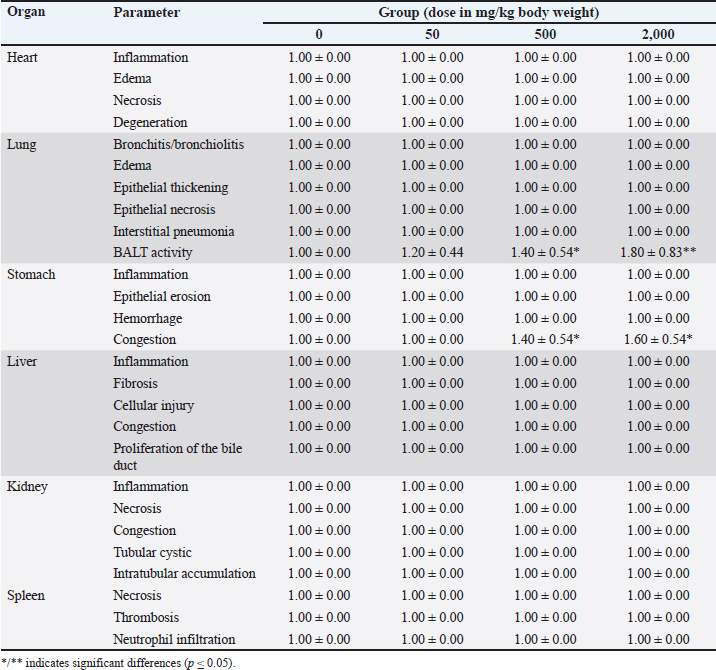
AbstractBackground: Fermented Crescentia cujete L supernatant of fermented Crescentia cujete (SFC) is a potential alternative therapy for ischemic stroke. However, the effects of SFC on various organs, the immune system, antioxidant levels, and malondialdehyde (MDA) still need to be better understood. Aim: This study aimed to analyze the effects of SFC in rat models through a 14-day repeated-dose oral toxicity study, focusing on histopathology, granulocyte-macrophage colony-stimulating factor (GM-CSF) levels, cyclooxygenase-2 (COX-2), antioxidants, and MDA. Methods: This study used 20 male Sprague Dawley rat models. Rats were divided into four groups: 0, 50, 500, and 2,000 mg/kg BW SFC. The therapy was administered orally and repeated once daily for 14 days. On day 15, the rats were euthanized, and samples were collected. The samples were tested against histopathology, enzyme-linked immunosorbent assay, antioxidant level, and MDA. The data were statistically analyzed. Results: The results indicated that SFC did not affect histopathology (p ≥ 0.05), except for an increase in bronchial associated lymphoid tissue activity in lung tissue and congestion in the stomach mucosa (p ≤ 0.05). SFC did not influence the rats’ serum protein levels (p ≥ 0.05), but it did increase the level of GM-CSF and decrease the level of COX-2 in the serum and various organs (p ≤ 0.05). Additionally, SFC increased the activity of catalase, glutathione peroxidase, glutathione, superoxide dismutase, and total antioxidants while decreasing the MDA level at a dosage of 2,000 mg/kg BW (p ≤ 0.05). Conclusion: The 14-day repeated-dose oral toxicity study of SFC did not show severe histopathological changes; however, it increased GM-CSF and antioxidant levels, which suppressed COX-2 and MDA levels. Keywords: Antioxidant, COX-2, Fermented Crescentia cujete L, GM-CSF, Histopathology. IntroductionA tropical country, Indonesia is renowned for its diverse flora and fauna. The calabash fruit (Crescentia cujete L.), which has unique therapeutic properties, is among its significant plant species. The calabash fruit possesses a wide range of benefits, including anti-inflammatory (Gonzales et al., 2023), neuroprotective, antibacterial, and antioxidant properties (Gonzales et al., 2022). These properties are attributed to specific compounds, such as saponin, tannin, glycoside, indomethacin, apigenin, and cinnamic acid (Das et al., 2014). Furthermore, SFC has demonstrated neuroprotective effects in rat models suffering from ischemic stroke, promoting neuronal survival and reducing pathological changes such as microgliosis, inflammation, and degeneration (Hidayah et al., 2023). These benefits are linked to the choline compound isolated after fermentation, where the fermentation process has been observed to stabilize the choline level at different preservation temperatures (Wilujeng et al., 2023). Notably, although the safety data, including toxicity and potential adverse effects, support the efficacy of fermented calabash as an ischemic stroke therapy, it is crucial to consider the extended duration of treatment for neurological defects, which may induce severe side effects and histopathological changes (Eikelboom et al., 2012). Furthermore, calabash is not only a potential therapy for ischemic stroke but also a rich source of natural antioxidants. Its antioxidant potential can be assessed through various biomarkers related to immunity and oxidative stress, including granulocyte-macrophage colony-stimulating factor (GM-CSF) (Lee et al., 2020), cyclooxygenase-2 (COX-2) (Zhuang et al., 2018), and levels of antioxidants (Li et al., 2022) and malondialdehyde (MDA) (Cordiano et al., 2023). Moreover, when combined with ciprofloxacin, SFC could be used as therapy against pneumonic pasteurellosis (Prakoso et al., 2024). These findings suggest that calabash could play a significant role in enhancing the quality of life and preventing oxidative stress-related diseases. Therefore, this study aimed to analyze the effects of SFC in rat models using a 14-day repeated-dose oral toxicity test, focusing on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels. Materials and MethodsFermented preparation of calabash (C. cujete L.)The calabash was obtained from the University of Wijaya Kusuma Surabaya. The calabash was prepared as a fermented product using the procedure described by Wilujeng et al. (2023) . The herbs were peeled before fermentation. The pulp weighed as much as 400 g. The pulp was mixed with water (1 L), sugar (40 g), and pectinase (40 ml) (Novozyme, UK). The mixture was then homogenized and stored at room temperature for 30 days. The mixture was filtered on the final day, and the fermented calabash (SFC) supernatant was stored at 4°C. Animal model and research designTwenty male Sprague Dawley rat models (6 months old, 259.00 ± 9.82 g) from the Laboratory of Pharmacology, Faculty of Veterinary Medicine, University of Wijaya Kusuma Surabaya were used in this study. The rats were acclimated for seven days in the laboratory under appropriate conditions (70% humidity, 25°C) before being used in the experiment. The rats were maintained in individual cages, and oven husk was used as bedding. They were also given ad libitum access to food (Ratbio®, Indonesia) and water. The rats were then divided into four groups as follows: 0, 50, 500, and 2,000 mg/kg BW SFC. The therapy was administered orally using a probe, and it was repeated once daily for 14 days. The doses used in this study were determined according to the preclinical toxicity test guidelines by Badan Pengawasan Obat dan Makanan, Indonesia Republic (2022) . Sample collectionChanges in animal behavior were recorded during the study. However, no behavioral changes were observed among the animal models during the observation times. The rats were terminated on day 15 after treatment. Before euthanasia, the rats were anesthetized using 50 mg/kg BW ketamine (Agrovet Market, Peru) for blood collection. Blood was collected using a microcapillary (Onemed, Indonesia) via the retro-orbital plexus and stored inside a plain tube (Onemed, Indonesia). The blood was centrifuged (DLab, Indonesia), and the serum was separated from the blood sediment. The rats were euthanized using cervical dislocation. The rats were then necropsied, and their abdominal walls were incised. Several organs (including the heart, lung, stomach, liver, kidney, and spleen) were collected and divided into two sections. For histopathology, the first section was fixed using 10% neutral-buffered formalin (Leica, UK). The second was stored inside a sterile plastic bag at −20°C for ELISA. Serum analysisSerum was measured for total protein and C-reactive protein (CRP) levels. It was tested using the procedure demonstrated by Henok et al. (2020) for total protein and by Kanaparthy et al. (2012) for CRP level. Hematoxylin and eosin stainingAfter 24 hours of fixation using 10% NBF, the organs were processed for histopathology against H&E staining using the standard procedure in the Laboratory of Pharmacology, UWKS, Indonesia. The organs were trimmed and stored inside a tissue cassette. They were then dehydrated for 2 hours using graded alcohol (70%, 80%, 90%, and absolute alcohol), each for 2 hours. The cassette was cleared using xylene (3×, each for 1 hour). The organs were then embedded using liquid paraffin for 4 hours. The organ was blocked in liquid paraffin using a mold until it cooled and hardened. The block was cut using a microtome at 3-5 µm of thickness and attached to the slide. The slide was then stained with H&E. The H&E staining procedure was as follows: the slide was cleared using xylene (3×, each 5’), absolute alcohol (2×, each 3’), 90% alcohol (3’), 80% alcohol (3’), 70% alcohol (3’), and distilled water (3’). The slides were then applied on the Mayer Hematoxylin (5’) gel and washed in running tap water (5’), 1% eosin (2’), and graded alcohol. The graded alcohol included 70% alcohol (3’), 80% alcohol (3’), 90% alcohol (3’), and absolute alcohol (2×, each 3’). Finally, the slide was cleared using xylene (3×, each 5’) and mounted using Entellan. All reagents used in the staining procedure in this study were manufactured by Leica. HistomorphometryHistopathological analysis of the tissue was performed by two pathologists under blindfold conditions. Analysis was performed against several parameters depending on the collected tissue (Table 1). The histomorphometry score was 1 (normal/no histopathological changes), 2 (mild changes), 3 (moderate changes), and 4 (severe changes). Table 1. Histopathological parameters of this study.
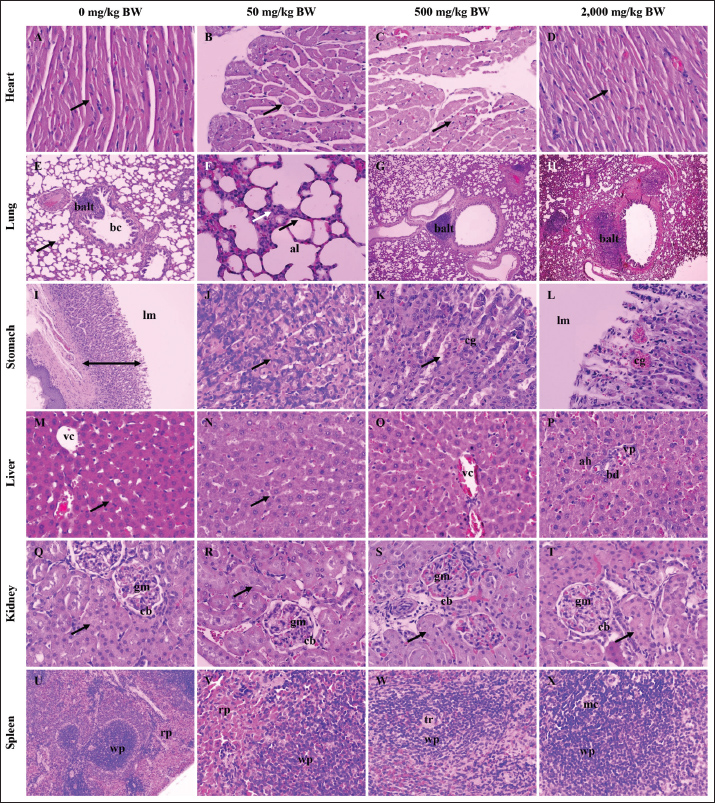
ELISAsELISA was used to measure fibrinogen, GM-CSF, and COX-2 levels. The fibrinogen level was measured using a serum specimen, whereas the GM-CSF and COX-2 levels were measured using serum and organs. Fibrinogen was measured following the procedure described by Zhang et al. (2023). However, GM-CSF (ERCSF2, Invitrogen) and COX-2 (E-EL-R0792, Elabscience, USA) levels were measured following the manufacturer’s standard procedure. Antioxidant measurementThe antioxidant level from the sample was measured against several parameters, including catalase activity, GPx, glutathione level, SOD, and total antioxidant capacity. Catalase activity was measured following the method of Farman and Hadwan (2021) . Furthermore, the GPx, glutathione, and SOD were tested using methods previously described by Bahrami et al. (2016) and the capacity of total antioxidants by Horváth-Szalai et al. (2023). MDA measurementThe serum MDA level was tested using colorimetric methods. MDA was measured using a standard manufacturing protocol (Thermo Fisher Scientific, EEA015, USA). Data analysisThis study included categorical and numeric data. Categorical data (e.g., histopathology score) were analyzed using a nonparametric test, especially the Kruskal–Wallis test, which was confirmed by the Mann-Whitney U test. However, the numeric data (e.g., total plasma protein, fibrinogen, CRP, GM-CSF, COX-2, antioxidant level, and MDA) were analyzed using parametric tests, especially ANOVA and Duncan test. Ethical approvalThis study was approved by the ethics committee of the Faculty of Veterinary Medicine, UWKS. The ethical approval was registered with voucher number 142-KKE-2023. The experiment was conducted in the Laboratory of Pharmacology, UWKS, Indonesia, from August 2023 to April 2024. ResultHistopathologyThe histopathological analysis revealed that the repeated 14-day oral administration of SFC did not induce any discernible effects on the heart, liver, kidney, or spleen (p ≥ 0.05), which is a reassuring finding. Conversely, there was a potential increase in BALT activity in the lung tissue in the 500 and 2,000 mg/kg BW groups following the 14-day application of SFC (p ≤ 0.05). Moreover, the same groups exhibited gastric mucosal congestion because of SFC administration (p ≤ 0.05) (refer to Table 2 for comprehensive details). Figure 1 shows the histopathological illustrations of the organs under investigation are available in Figure 1.
Fig. 1. Histopathology of the heart, lung, stomach, liver, kidney, and spleen of rat models after 14 days of repeated oral SFC toxicity. A typical myocardiocyte (black arrow) in the heart tissue among the treated group (A-D); interalveolar septum (black arrow) consisted of pneumocyte type 1 and 2 (white arrow) with clean bronchioles (bc) and lymphoid tissue (balt) (E-F); an increase of diameter of lymphoid tissue (balt) indicated a high activity of the immune system in group 500 (G) and 2,000 mg/kg BW (H); there is no histopathological changes from stomach’s mucosa (line) (I); with clear chief and parietal cells (black arrow) (J); however, group 500 and 2,000 mg/kg BW showed the congestion (cg) in the mucosal vein of stomach (K-L); the liver did not show any histopathological change marked by a typical hepatocyte (black arrow), central vein (vc), bile duct (bd), portal vein (vp), and hepatic artery (ah) (M-P); there is no histopathological change of kidneys in all treated group indicated by normal glomeruli (gm), capsule Bowman (cb), and tubules (black arrow) (Q-T); and it similar to the spleen histology after treatment, and the spleen showed dense white (wp) and red pulp (rp) (U-X). H&E, 100× (E, G-I, U); 400× (A-D, F, J-T, V-X). Table 2. Histopathology score after 14-day repeated-dose oral toxicity of fermented Crescentia cujete L in rat models.
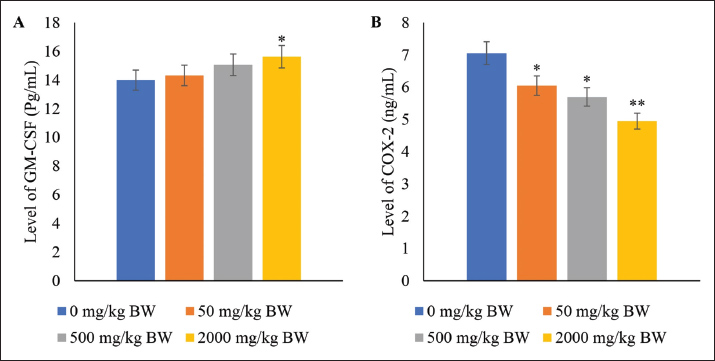
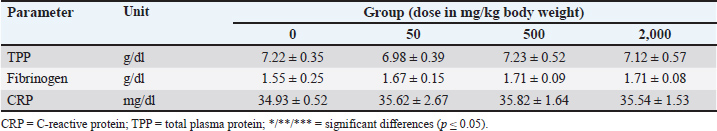
Serum protein, GM-CSF, and COX-2 levels in the plasmaOral administration of SFC exhibited no discernible impact on TPP, fibrinogen, and CRP levels in rat models (p ≥ 0.05) (Table 3). However, notable advantages were observed in elevating the serum levels of GM-CSF at the highest administered doses (p ≤ 0.05). Moreover, a declining trend was noted in the serum level of COX-2 corresponding to escalating doses of SFC (p ≤ 0.05). The most significant reduction in COX-2 levels was evident in the 2,000 mg/kg BW group (p ≤ 0.05). Conversely, no significant variance was observed between the 50 and 500 mg/kg BW groups (p ≥ 0.05) (Fig. 2).
Fig. 2. Levels of GM-CSF (A) and COX-2 (B) in the plasma of rat models after fourteen-day repeated dose oral toxicity of SFC. */** indicates significant differences. Table 3. Serum protein level after 14-day repeated-dose oral toxicity of fermented Crescentia cujete L.
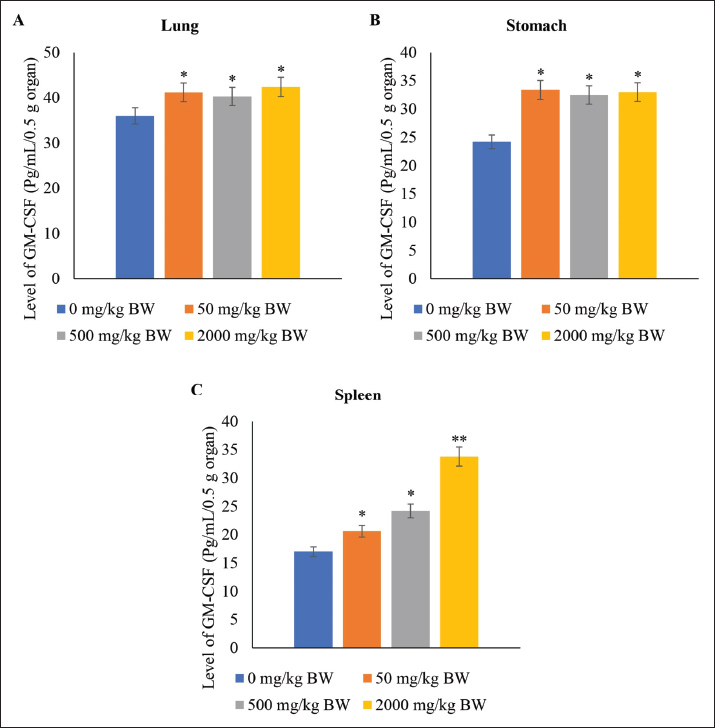
GM-CSF level in the organsThe presence of GM-CSF in heart, kidney, and liver tissue was not identified through ELISA analysis. This may be attributed to the GM-CSF levels in these organs falling below the detection threshold. Conversely, the lung, stomach, and spleen exhibited varying GM-CSF levels. Notably, the application of SFC led to elevated GM-CSF levels in the lungs and stomach compared with the control (p ≤ 0.05) (Fig. 3). Furthermore, no significant differences in GM-CSF levels were observed in the lung and stomach tissues among groups treated with 50, 500, and 2,000 mg/kg BW of SFC (p ≥ 0.05). Moreover, the administration of 2,000 mg/kg BW of SFC resulted in a substantial increase in GM-CSF levels within the spleen tissue compared to other doses, while the 50 mg/kg BW group did not present a significant difference compared to the 500 mg/kg BW group (p ≥ 0.05) (Fig. 3).
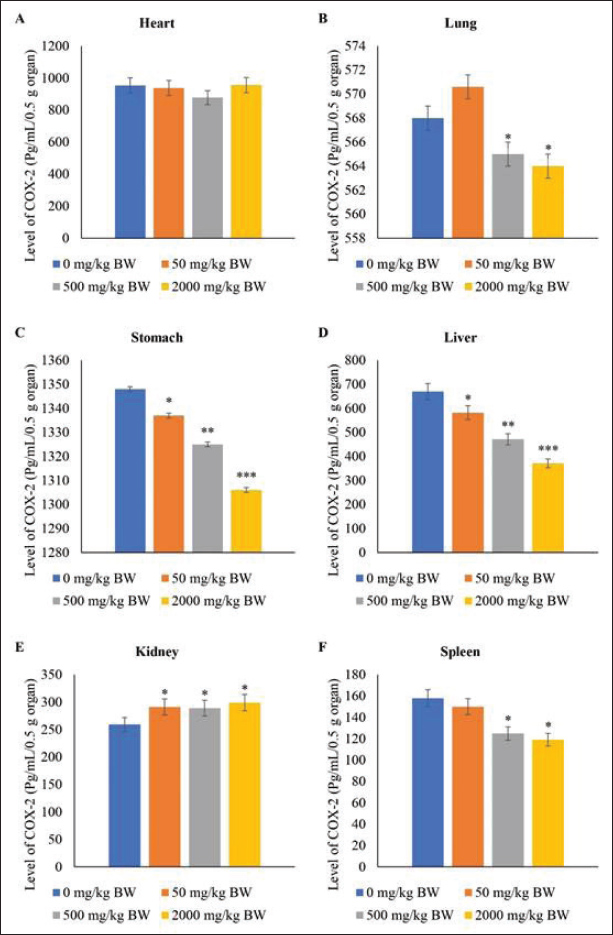
Fig. 3. Level of GM-CSF in the lungs, stomach, and spleen of rat models after fourteen-day repeated dose oral toxicity of SFC. */** indicates significant differences. Level of COX-2 in the organsThe study observed that administering SFC at various doses demonstrated no significant impact on COX-2 levels in the heart compared with the control group (p ≥ 0.05). However, it did affect COX-2 levels in the lung, stomach, liver, kidney, and spleen (p ≤ 0.05). Following 50 mg/kg BW SFC administration, the COX-2 levels in the lung and spleen did not differ significantly from those in the control group (p ≥ 0.05). However, repeated doses of 500 and 2,000 mg/kg BW SFC notably decreased COX-2 levels in the lung and spleen (p ≤ 0.05). After repeated doses of SFC, the stomach and liver exhibited a similar pattern in COX-2 levels, with higher doses resulting in a more significant decrease in COX-2 levels in these organs than in lower doses (p ≤ 0.05). Moreover, the level of COX-2 in the kidney increased following SFC application, showing higher levels of COX-2 in the treatment groups than in the control group (p ≤ 0.05). However, no differences in COX-2 levels were observed after applying different repeated doses of SFC (p ≥ 0.05) (Fig. 4).
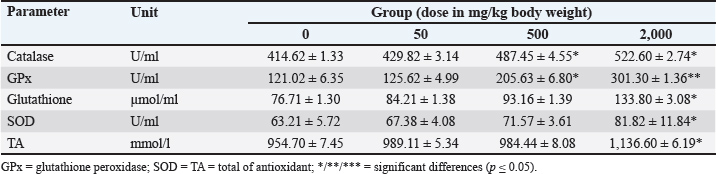
Fig. 4. Level of COX-2 in the heart, lung, stomach, liver, kidney, and spleen of rat models after 14 days of repeated oral SFC toxicity. */** indicates significant differences. Antioxidant and MDA levelsSignificant impacts on the antioxidant levels in rat models following treatment (p ≤ 0.05). The administration of 500 and 2,000 mg/kg BW SFC increased catalase and GPx levels after a 14-day treatment period (p ≤ 0.05). Specifically, the GPx level in the group receiving 2,000 mg/kg BW SFC was twice as high as that in the control group (p ≤ 0.05). The highest dose of SFC also effectively regulated the levels of glutathione, SOD, and total antioxidants compared with the other doses (p ≤ 0.05) (Refer to Table 4). Table 4. Antioxidant levels after 14-day repeated-dose oral toxicity of fermented Crescentia cujete L in rat models.
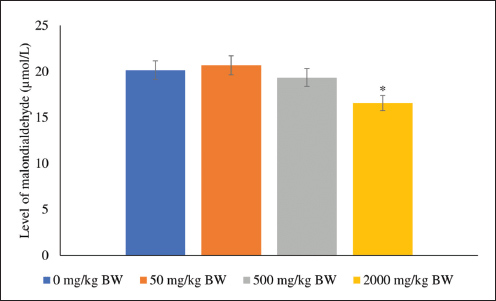
Furthermore, the samples were analyzed to determine the levels of MDA. The rat models treated with 2,000 mg/kg BW SFC exhibited the lowest MDA level compared with the other treatments (p ≤ 0.05); however, the levels observed in the 50 and 500 mg/kg BW groups did not differ significantly from those in the control group (p ≥ 0.05) (Fig. 5).
Fig. 5. Malondialdehyde activity in rat models after 14-day repeated oral SFC dose toxicity. */** indicates significant differences. DiscussionCalabash is a crucial fruit with significant therapeutic potential for various diseases. It contains essential biochemical compounds that play a crucial role in disease management and oxidative stress reduction. Das et al. (2014) highlighted the composition of various secondary metabolites in calabash, including phenolic, alkaloid, saponin, tannin, terpenoid, steroid, and flavonoid, which are instrumental in promoting cellular health. Additionally, it encompasses herbal-derived antioxidants such as ascorbic acid, phenol, and carotenoid. Another study reported that SFC contains choline (Wilujeng et al., 2023). Our unpublished data revealed α-tocopherol in SFC, demonstrating its efficacy as a scavenging agent against oxidative stress. As preventive or therapeutic agents, these compounds are vital for disease management. As indicated by Hidayah et al. (2023) SFC exerts beneficial effects on neuronal repair following ischemia. These therapeutic properties must be substantiated by toxicological data and thorough consideration of the factors involved during and after SFC use. Histopathological analysis demonstrated the safety of SFC over 14 days. After 14 days, SFC did not significantly affect the heart, liver, kidney, or spleen, except for the stomach, which exhibited mucosal congestion, known as gastric venous congestion (GVC). Changes in capillary mucosal pressure, gastrotomy, gastrectomy, and abdominal surgery are the primary factors contributing to GVC in the stomach (Nakao et al., 2018). In the context of drug utilization, drugs containing choline (including SFC) may induce abdominal pain and discomfort at standard doses (Arias et al., 2020). This effect arises from the decreased gastric blood pressure during choline metabolism and its conversion into trimethylamine, leading to blood entrapment within mucosal capillaries (Abbasi et al., 2023) and stimulating gastric antrum motility (Elfers et al., 2023). However, gastric mucosal congestion represents a reversible change. A prior study elucidated how congestion in organs caused by drug use can be remedied following drug cessation (Stoop et al., 2023). Furthermore, Sanchez (2012) contributed to the understanding that GVC is reversible in animal and human stomachs. Interestingly, the lungs exhibited an intriguing mechanism, manifesting increased BALT activity. Increased BALT activity results from the promotion of immunity by SFC via the phosphatidylcholine (PC) pathway (Garcia et al., 2018). The abundant choline and α-tocopherol content in SFC can serve as a source of membrane PC and antioxidant, aiding in the prevention of ceramide-induced inflammation, which is pivotal for enhancing lung function (Bernhard et al., 2019) and maintaining lung homeostasis (Jubinville et al., 2020). SFC administration did not affect serum protein levels, including TPP, fibrinogen, and CRP. This outcome suggests that SFC did not exert an influence on colloid osmotic pressure within the plasma (Liu et al., 2022) or organ histopathological changes (Całkosiński et al., 2016). The standard level of TPP indicates normal pressure within the circulatory system, reflecting the rat’s homeostasis condition following SFC administration. Acute phase proteins, such as fibrinogen and CRP, also play crucial roles in blood coagulation (Pieters and Wolberg, 2019) and inflammatory responses (Prakoso et al., 2021). Normal fibrinogen levels can serve as biomarkers in the absence of fibrinolysis (May et al., 2021), hematological disturbance (Vilar et al., 2020), and internal organ hemorrhage (Shafagh et al., 2022). Furthermore, CRP, an acute-phase protein, can significantly escalate following inflammation and is involved in the complement pathway, apoptosis, and phagocytosis in host responses to infection (Sproston and Ashworth, 2018). Consequently, there is no discernible impact on TPP, fibrinogen, and CRP levels in the rat model after SFC administration. These findings affirm the safety of using this herb for 14 days. These results align with the mild histopathological changes, including gastric mucosal congestion, observed in the organs during this study. The GM-CSF serves as a crucial immunomodulatory cytokine within the immune system. Various cells secrete GM-CSF, including lymphocytes, macrophages, fibroblasts, epithelial cells, and bronchoalveolar cells. This cytokine notably facilitates neutrophil activation, augmenting phagocytic and bactericidal functions (Kasahara et al., 2016). Furthermore, GM-CSF stimulates the activity of STAT-5 (Zhang et al., 2019), contributes to lipid homeostasis (Ataya et al., 2021), and induces cholesterol efflux from diverse cell types (Groenen et al., 2021). GM-CSF amplifies antigen-presenting cell functionality, microbicidal capacity (Mashima et al., 2020), adhesion, chemotaxis, adenosine triphosphate synthesis (Wessendarp et al., 2022), and intracellular elimination of leucocytes in macrophages (Rothchild et al., 2017). The current study underscores the regulatory effects of SFC comprising choline and α-tocopherol (unpublished data) on the immune system and its antioxidative properties. Choline and α-tocopherol have been observed to curtail LPS-induced proinflammatory cytokines (Baker et al., 2022), although the precise underlying mechanism remains unclear. Furthermore, α-tocopherol has been implicated in fostering lymphocyte proliferation (Lewis et al., 2019) and is associated with antibody synthesis processes (Iperi et al., 2021). After SFC administration, heightened GM-CSF levels were observed in the plasma, lung, stomach, and spleen. The raised plasma GM-CSF levels correlate with augmented antibody release into the circulatory system, whereas the increased GM-CSF levels within the lung, stomach, and spleen are associated with the innate presence of macrophages and lymphocytes in these tissues (Chen et al., 2023). Regrettably, GM-CSF levels in the heart, kidney, and liver were undetectable and were purportedly below the detection threshold. The administration of SFC significantly affects the level of COX-2, both in plasma and tissues, particularly in the lung, spleen, stomach, liver, and kidney. These findings suggest that SFC may alter immunity status by influencing COX-2 levels. COX-2 is known to be an inducible form during an inflammatory response and is linked to oxidative stress (Choi et al., 2010). Increased COX-2 levels in lung tissue are associated with PD and inflammation. Additionally, anti-inflammatory agents are crucial in supporting lung function during the pathogenesis of lung injury (Robertson et al., 2012). Therefore, COX-2 reduction in the lung is essential for maintaining lung homeostasis and ventilation. Conversely, COX-2 expression in gastrointestinal organs, such as the stomach, liver, and spleen, plays a significant role in the maintenance of these biological processes. For example, COX-2 in the stomach, COX-2 protects the mucosa from disruption (Wang et al., 2014) and the liver from injury (Martín-Sanz et al., 2017). In the spleen, COX-2 is essential for maintaining local splenic circulation (Kulesza et al., 2023). Decreased COX-2 levels in these organs can lead to functional disturbances, whereas COX-2 overexpression indicates severe inflammation and can be fatal (Yang et al., 2020). Nevertheless, the decrease in COX-2 levels in the stomach, liver, and spleen due to SFC is believed to have no impact on the biological function of these organs. Histopathological data supported this belief, which did not show significant impacts except for gastric mucosal congestion. With its choline and α-tocopherol compounds, SFC is suspected to act as antioxidants and protect cellular levels from oxidative stress (Niki, 2015) and injury (Karedath et al., 2022). Therefore, SFC selectively inhibits COX-2 rather than simultaneously inhibiting COX-1 and COX-2. It is essential to note that selective nonsteroidal anti-inflammatory drugs that inhibit COX-1 or COX-2 do not induce gastric ulceration in healthy animal models (Massó González et al., 2010). In this study, the administration of SFC with various doses resulted in an increase in the level of COX-2 in the kidney. COX-2 is beneficial in promoting the glomerular filtration rate (GFR) in the kidneys, which correlates with the excretion of toxicants in urine (Zeisel et al., 2018). Additionally, COX-2 is highly expressed in the kidney and regulates the response to changes in intravascular volume (Ferreira et al., 2023). The metabolite products of COX-2 mediate sodium excretion, renin release, and the local circulatory system in the kidney (Wang et al., 2019). The findings of this study suggest that choline and α-tocopherol from SFC may positively impact renal function. Hasson et al. (2022) noted that choline is essential for improving GFR and decreasing blood urea nitrogen and creatinine levels in mice with septic-associated acute kidney injury. Additionally, α-tocopherol, as a lipid-soluble antioxidant, is essential for controlling lipid peroxidation-related chronic kidney injury (Galli et al., 2022). The administration of SFC did not influence the COX-2 level in the hearts of the rat models in this study. This may be attributed to the maintenance and protection of the heart from oxidation and stress by choline and α-tocopherol derived from SFC (Wallert et al., 2019). Ilcol et al. (2005) indicated that choline supplementation can prevent cardiovascular defects during septic intervention. Furthermore, Parrish et al. (2008) reported that choline supplementation reduces proinflammatory cytokines during endotoxemia in mice. The study demonstrates a correlation between SFC administration and GM-CSF and COX-2 levels in various organs and antioxidant and MDA levels. SFC administration appears to be associated with an increase in antioxidant levels, particularly catalase, GPx, glutathione, SOD, and total antioxidants, alongside a decrease in MDA. This increase in antioxidants in the rat model suggests that the body can manage oxidative stress. Endogenous antioxidants, including catalase, GPx, glutathione, and SOD, have been shown to reduce oxidation processes (Chan and Chan, 2015) and associated damage (Zhu et al., 2023; Korczowska-Łącka et al., 2023). Furthermore, exogenous antioxidant supplementation, such as α-tocopherol from SFC, disrupts the redox balance by quenching oxygen-derived reactive species (Bouayed and Bohn, 2010). α-tocopherol and choline, both derived from SFC, have beneficial effects on cellular components (Zhu et al., 2014), protecting against oxidation, glycation, and nitrosylation (Bennett et al., 2020), and influencing cellular signaling (Edward et al., 2022). These effects are potentially correlated with increased immunity via the GM-CSF pathway, reduced COX-2 levels in various organs, and decreased MDA levels following SFC administration. MDA is a final product of lipid peroxidation (Hurşitoğlu et al., 2021). An increase in MDA promotes tissue destruction (Cherian et al., 2019). In contrast, the decrease in MDA levels suggests that SFC protects against tissue oxidative stress. Ultimately, the findings indicate that the repeated oral toxicity administration of SFC over 14 days did not induce severe histopathological changes but promoted increased immunity via the GM-CSF pathway and elevated endogenous antioxidants, thereby suppressing COX-2 and MDA levels. ConclusionIn conclusion, the 14-day repeated-dose oral toxicity of SFC did not potentially promote severe histopathological effects; however, it increased the levels of GM-CSF and antioxidants, which suppressed the levels of COX-2 and MDA. Hence, SFC can be assumed to be safe for use over 14 days. However, the potential toxicity effects must be observed using a longer observation time, such as a repeated oral toxicity study lasting 90 days, along with a reversibility test. AcknowledgmentThe authors would like to acknowledge Universitas Gadjah Mada, Indonesia, for the financial support of this study through the Program Post Doctoral 2024 with funding number: 4292/UN1.P1/PT.01.03/2024. Conflict of interestThe authors declare no conflict of interest. FundingThis study was financially supported by Universitas Gadjah Mada, Yogyakarta, Indonesia, through the Program Post Doctoral 2025 with funding number: 4454/UN1.P2/Dit-Lit/PT.01.03/2025. Authors’ contributionsYAP supervised this study. YAP, ADW, and KJVI contributed to the study design. YAP and ADW performed the experimental procedure. KJVI analyzed the data. YAP, ADW, and KJVI contributed to the drafting, revision, and approval of the final version of the manuscript. Data availabilityAll data supporting the findings are available within the manuscript. ReferencesAbbasi, M. S. P., Tousi, A. Z., Yazdani, Y., Vahdat, S., Gharebakhshi, F., Nikrad, N., Manzouri, A., Ardekani, A. M. and Jafarzadeh, F. 2023. Dietary choline and betaine intake, cardio-metabolic risk factors and prevalence of metabolic syndrome among overweight and obese adults. BMC Endocrine Dis. 23, 67. Arias, N., Arboleya, S., Allison, J., Kaliszewska, A., Higarza, S.G., Gueimonde, M. and Arias, J.L. 2020. The relationship between choline bioavailability from diet, intestinal microbiota composition, and its modulation of human diseases. Nutrients 12(8), 2340. Ataya, A., Knight, V., Carey, B.C., Lee, E., Tarling, E.J. and Wang, T. 2021. The role of GM-CSF autoantibodies in infection and autoimmune pulmonary alveolar proteinosis: a concise review. Front. Immunol. 12, 752856. Badan Pengawasan Obat dan Makanan Indonesia Republic. 2022. Peraturan Badan Pengawas Obat dan Makanan Nomor 10 Tahun 2022 tentang Pedoman Uji Toksisitas Praklinik secara In Vivo. BN.2022, 490. Baker, J.A., Breit, K.R., Bodnar, T.S., Weinberg, J. and Thomas, J.D. 2022. Choline supplementation modifies the effects of developmental alcohol exposure on immune responses in adult rats. Nutrients 14, 2868. Bahrami, S., Shahriari, A., Tavalla, M., Azadmanesh, S. and Hamidinejat, H. 2016. Blood levels of oxidant/antioxidant parameters in rats infected with Toxoplasma gondii. Oxid. Med. Cell Long. 2016, 8534604. Bennett, J.A., Mastrangelo, M.A., Ture, S.K., Smith, C.O., Loelius, S.G., Berg, R.A., Shi, X., Burke, R.M., Spinelli, S.L., Cameron, S.J., Carey, T.E., Brookes, P.S., Gerszten, R.E., Sabater-Lleal, M., De Vries, P.S., Huffman, J.E., Smith, N.L., Morrell, C.N. and Lowenstein, C.J. 2020. The choline transporter Slc44a2 controls platelet activation and thrombosis by regulating mitochondrial function. Nature Commun. 11, 3479. Bernhard, W., Lange, R., Graepler-Mainka, U., Engel, C., Machann, J., Hund, V., Shunova, A., Hector, A. and Riethmüller, J. 2019. Choline supplementation in cystic fibrosis-the metabolic and clinical impact. Nutrients 11(3), 656. Bouayed, J. and Bohn, T. 2010. Exogenous antioxidants--Double-edged swords in cellular redox state: Health beneficial effects at physiologic doses versus deleterious effects at high doses. Oxid. Med. Cell Longev. 3(4), 228–237. Całkosiński, I., Majda, J., Terlecki, G., Gostomska-Pampuch, K., Małolepsza-Jarmołowska, K., Sobolewska, S., Całkosińska, A., Kumala, A. and Gamian, A. 2016. Dynamic analysis of changes of protein levels and selected biochemical indices in rat serum in the course of experimental pleurisy. Inflammation 39(3), 1076–1089. Chan, J.Y. and Chan, S.H. 2015. Activation of endogenous antioxidants as a common therapeutic strategy against cancer, neurodegeneration and cardiovascular diseases: a lesson learnt from DJ-1. Pharmacol. Ther. 156, 69–74. Chen, Y., Li, F., Hua, M., Liang, M. and Song, C. 2023. Role of GM-CSF in lung balance and disease. Front. Immunol. 14, 1158859. Cherian, D. A., Peter, T., Narayanan, A., Madhavan, South S., Achammada, S. and Vynat, G. P. 2019. Malondialdehyde as a marker of oxidative stress in periodontitis patients. J. Pharm. Bioallied Sci. 11, S297–S300. Choi, S.H., Aid, S. and Bosetti, F. 2010. The distinct roles of cyclooxygenase-1 and -2 in neuroinflammation: implications for translational research. Trends Pharmacol. Sci. 30(4), 174–181. Cordiano, R., Di Gioacchino, M., Mangifesta, R., Panzera, C., Gangemi, S. and Minciullo, P.L. 2023. Malondialdehyde as a potential oxidative stress marker for allergy-oriented diseases: an update. Molecules 28(16), 5979; doi:10.1016/j.molecules.2016.09.016 Das, N., Islam, M.E., Jahan, N., Islam, M.S., Khan, A., Islam, M.R. and Parvin, M.S. 2014. Antioxidant activities of ethanol extracts and fractions of Crescentia cujete leaves and stem bark and the involvement of phenolic compounds. BMC. Complement. Altern. Med. 14, 45. Edwards, G., Olson, C.G., Euritt, C.P. and Koulen, P. 2022. Molecular mechanisms underlying the therapeutic role of vitamin E in age-related macular degeneration. Front. Neurosci. 16, 890021. Eikelboom JW, Hirsh, J., Spencer, F.A., Baglin, T.P. and Weitz, J.I. 2012. Antiplatelet drugs: antithrombotic therapy and prevention of thrombosis, 9th ed: Evidence-Based Clinical Practice Guidelines. Chest 141(2 Suppl), e89S–e119S. Elfers, K., Menne, L., Colnaghi, L., Hoppe, S. and Mazzuoli-Weber, G. 2023. Short- and long-term effects of cocaine on enteric neuronal functions. Cells 12(4), 577. Farman, A.A. and Hadwan, M.H. 2021. Simple kinetic method for assessing catalase activity in biological samples. MethodsX 8, 101434. Ferreira, P., Vaja, R., Lopes-Pires, M., Crescente, M., Yu, H., Nüsing, R., Liu, B., Zhou, Y., Yaqoob, M., Zhang, A., Rickman, M., Longhurst, H., White, W. E., Knowles, R. B., Chan, M. V., Warner, T. D., Want, E., Kirkby, N. S. and Mitchell, J. A. 2023. Renal function underpins the cyclooxygenase-2: asymmetric dimethylarginine axis in mouse and man. Kidney Intern Rep. 8(6), 1231–1238. Galli, F., Bonomini, M., Bartolini, D., Zatini, L., Reboldi, G., Marcantonini, G., Gentile, G., Sirolli, V. and Di Pietro, N. 2022. Antioxidants. Antioxidants 11(5), 899. Garcia, M., Mamedova, L.K., Barton, B. and Bradford, B.J. 2018. Choline regulates the function of bovine immune cells and alters the mRNA abundance of enzymes and receptors involved in its metabolism in vitro. Front. Immunol. 9, 2448. Gonzales, A.L., Huang, S.K.H., Sevilla, U.T.A., Hsieh, C.Y. and Tsai, P.W. 2023. In silico analysis of anti-inflammatory and antioxidant properties of bioactive compounds from Crescentia cujete L. Molecules 28, 3547. Gonzales A., Sevilla U., Tsai P.-W. and Huang S. 2022. Antioxidant and anti-inflammatory activities of bioactive compounds from Crescentia cujete L. leaves and fruit -A review. Int. J. Adv. Appl. Sci. 9, 64 -70. Groenen, A.G., Halmos, B., Tall, A.R. and Westerterp, M. 2021. Cholesterol efflux pathways, inflammation, and atherosclerosis. Crit. Rev. Biochem. Mol. Biol. 56(4), 426–439. Hasson, D.C., Watanabe-Chailland, M., Romick-Rosendale, L., Koterba, A., Miner, D.S., Lahni, P., Ma, Q., Goldstein, S.L., Devarajan, P. and Standage, S.W. 2022. Choline supplementation attenuates experimental sepsis-associated acute kidney injury. Am. J. Physiol. Renal. Physiol. 323(3), F255–F271. Henok, J.N., Okeleye, B.I., Omodanisi, E.I., Ntwampe, S.K.O. and Aboua, Y.G. 2020. Analysis of reference ranges of total serum protein in Namibia: clinical implications. Proteomes 8(2), 7. Hidayah, J.H., Prakoso, Y.A. and Widyarini, S. 2023. Histopathological changes after treatment with calabash fruit (Crescentia cujete L) in a rat model of artificially induced ischemic stroke. Adv. Anim. Vet. Sci. 11(12), 2003–2009. Hurşitoğlu, O., Orhan, F.O., Kurutaş, E.B., Doğaner, A., Durmuş, H.T. and Kopar, H. 2021. Diagnostic performance of increased malondialdehyde level and oxidative stress in patients with schizophrenia. Noro Psikiyatr. Ars. 58(3), 184–188. Horváth-Szalai, Z., Jakabfi-Csepregi, R., Szirmay, B., Ragán, D., Simon, G., Kovács-Ábrahám, Z., Szabó, P., Sipos, D., Péterfalvi, Á., Miseta, A., Csontos, C., Kőszegi, T. and Tóth, I. 2023. Serum total antioxidant capacity (TAC) and TAC/lymphocyte ratio as promising predictive markers in COVID-19. Int. J. Mol. Sci. 24(16), 12935. Ilcol, Y.O., Yilmaz, Z. and Ulus, I.H. 2005. Endotoxin alters serum-free choline and phospholipid-bound choline concentrations, and choline administration attenuates endotoxin-induced organ injury in dogs. Shock 24(3), 288–293. Iperi, C., Bordron, A., Dueymes, M., Pers, J.O. and Jamin, C. 2021. Metabolic program of regulatory B lymphocytes and influence in the control of malignant and autoimmune situations. Front. Immunol. 12, 735463. Jubinville, E., Milad, N., Maranda-Robitaille, M., Lafrance, M.A., Pineault, M., Lamothe, J., Routhier, J., Beaulieu, M.J., Aubin, S., Laplante, M. and Morissette, M.C. 2020. Critical importance of dietary methionine and choline in the maintenance of lung homeostasis during normal and cigarette smoke exposure conditions. Am. J. Physiol. Lung. Cell. Mol. Physiol. 319(2), L391–L402. Kanaparthy, A., Kanaparthy, R. and Niranjan, N. 2012. Evaluation of serum C-reactive protein levels in subjects with aggressive and chronic periodontitis and comparison with healthy controls. Dent. Res. J. 9(3), 261–265. Karedath, J., Javed, H., Ahsan Talpur, F., Lal, B., Kumari, A., Kivan, H., Anirudh Chunchu, V. and Hirani, S. 2022. Effect of vitamin E on clinical outcomes in patients with non-alcoholic fatty liver disease: a meta-analysis. Cureus 14, e32764. Kasahara, S., Jhingran, A., Dhingra, S., Salem, A., Cramer, R.A. and Hohl, T.M. 2016. Role of granulocyte-macrophage colony-stimulating factor signaling in regulating neutrophil antifungal activity and the oxidative burst during respiratory fungal challenge. J. Infect. Dis. 213(8), 1289–1298. Korczowska-Łącka, I., Słowikowski, B., Piekut, T., Hurła, M., Banaszek, N., Szymanowicz, O., Jagodziński, P.P., Kozubski, W., Permoda-Pachuta, A. and Dorszewska, J. 2023. Disorders of endogenous and exogenous antioxidants in neurological diseases. Antioxidants 12, 1811. Kulesza, A., Paczek, L. and Burdzinska, A. 2023. The role of COX-2 and PGE2 in the regulation of immunomodulation and other functions of mesenchymal stromal cells. Biomedicines 11, 445. Li, S., Fasipe, B. and Laher, I. 2022. Potential harms of supplementation with high doses of antioxidants in athletes. J. Exerc. Sci Fitness. 20(4), 269 -275. Lee, K.M.C., Achuthan. and Hamilton. 2020. GM-CSF: a promising target in inflammation and autoimmunity. Immun. Targets Ther. 9, 225–240. Lewis, E.D., Meydani, S.N. and Wu, D. 2019. Regulatory role of vitamin E in the immune system and inflammation. IUBMB. Life. 71(4), 487–494. Liu, B., Dong, D., Wang, Z., Gao, Y., Yu, D., Ye, S., Du, X., Ma, L., Cao, H., Liu, F., Zhang, R. and Li, C. 2022. Analysis of influencing factors of serum total protein and serum calcium content in plasma donors. PeerJ 10, e14474. Martín-Sanz, P., Casado, M. and Boscá, L. 2010. Cyclooxygenase 2 in liver dysfunction and carcinogenesis: facts and perspectives. World. J. Gastroenterol. 23(20), 3572–3580. Mashima, H., Zhang, R., Kobayashi, T., Hagiya, Y., Tsukamoto, H., Liu, T., Iwama, T., Yamamoto, M., Lin, C., Nakatsuka, R., Mishima, Y., Watanabe, N., Yamada, T., Senju, S., Kaneko, S., Idiris, A., Nakatsura, T., Ohdan, H. and Uemura, Y. 2020. Generation of GM-CSF-producing antigen-presenting cells that induce a cytotoxic T cell-mediated antitumor response. Oncoimmunol 9, 1814620. May J East., Wolberg A South. and Lim, M.Y. 2021. Disorders of fibrinogen and fibrinolysis. Hematol. Onco. Clin. North. Am. 35(6), 1197–1217. Massó González, E.L., Patrignani, P., Tacconelli, S. and García Rodríguez, L.A. 2010. Variability among nonsteroidal antiinflammatory drugs in risk of upper gastrointestinal bleeding. Arthrit. Rheum. 62(6), 1592 -1601. Nakao, A., Yamada, S., Fujii, T., Tanaka, H., Oshima, K., Oshima, Y., Iede, K., Kobayashi, H., Kimura, Y. and Kodera, Y. 2018. Gastric venous congestion and bleeding in association with total pancreatectomy. J. Hepato. Bil. Pancreat. Sci. 25(2), 150–154. Niki, E. 2015. Evidence for beneficial effects of vitamin E. Korean J. Inter. Med. 30(5), 571–579. Parrish, W.R., Rosas-Ballina, M., Gallowitsch-Puerta, M., Ochani, M., Ochani, K., Yang, L.H., Hudson, L., Lin, X., Patel, N., Johnson, S.M., Chavan, S., Goldstein, R.S., Czura, C.J., Miller, E.J., Al-Abed, Y., Tracey, K.J. and Pavlov, V.A. 2008. Modulation of TNF release by choline requires alpha7 subunit nicotinic acetylcholine receptor-mediated signaling. Mol. Med. 14(9-10), 567–574. Pieters, M. and Wolberg, A. 2019. Fibrinogen and fibrin: an illustrated review. Res. Pract. Throm. Haemos. 3, 161–172. Prakoso, Y.A., Hidayah, N., Rini, C.S. and Kurniasih, K. 2021. Dynamic change of blood profile in rat models with acute skin injury artificially infected with methicillin-resistant Staphylococcus aureus. Vet World. 2018(14), 2085–2090. Prakoso, Y.A., Susilo, A. and Widyarini, S. 2024. The standardization and efficacy of fermented Crescentia cujete (L.) in combination with enrofloxacin against artificially induced pneumonic pasteurellosis in rat models. Open. Vet. J. 14(12), 3404–3416. Robertson, J.A., Sauer, D., Gold, J.A. and Nonas, S.A. 2012. The role of cyclooxygenase-2 in mechanical ventilation-induced lung injury. Am. J. Regulatory. Cellular. Mol. Biol. 47(3), 387–394. Rothchild, A.C., Stowell, B., Goyal, G., Nunes-Alves, C., Yang, Q., Papavinasasundaram, K., Sassetti, C.M., Dranoff, G., Chen, X., Lee, J. and Behar, S.M. 2017. Role of granulocyte-macrophage colony-stimulating factor production by T cells during Mycobacterium tuberculosis infection. mBio 8(5), e01514-17. Sanchez, L.C. 2018. Disorders of the gastrointestinal system. Equine Intern. Med. 2, 709–842. Shafagh, S., Barooni, M., Davoodabadi, A., Gilasi, H. and Hajian, A. 2022. Evaluation of plasma level of fibrinogen as a diagnostic criterion in acute appendicitis: a cohort study. Ann. Med. Surg. 75, 103393. Sproston, N.R. and Ashworth, J.J. 2018. Role of C-reactive protein at sites of inflammation and infection. Front. Immunol. 9, 754. Stoop, T.F., Von Gohren, A., Engstrand, J., Sparrelid, E., Gilg, S., Del Chiaro, M. and Ghorbani, P. 2023. Risk factors, management, and outcome of gastric venous congestion after total pancreatectomy: an underestimated complication requiring standardized identification, grading, and management. Annal. Surg. Oncol. 30(12), 7700–7711. Vilar, R., Fish, R.J., Casini, A. and Neerman-Arbez, M. 2020. Fibrin(ogen) in human disease: both friend and foe. Haematologica 105, 284–296. Wallert, M., Ziegler, M., Wang, X., Maluenda, A., Xu, X., Yap, M.L., Witt, R., Giles, C., Kluge, S., Hortmann, M., Zhang, J., Meikle, P., Lorkowski, S. and Peter, K. 2019. Α-tocopherol preserves cardiac function by reducing oxidative stress and inflammation in ischemia/reperfusion injury. Redox Biol. 26, 101292. Wang, T., Fu, X., Chen, Q., Patra, J.K., Wang, D., Wang, Z. and Gai, Z. 2019. Arachidonic acid metabolism and kidney inflammation. Int. J. Mol. Sci. 20(15), 3683. Wang, Z., Chen, J.Q. and Liu, J.L. 2014. COX-2 inhibitors and gastric cancer. Gastroenterol. Res. Pract. 2014, 132320. Wessendarp, M., Watanabe-Chailland, M., Liu, S., Stankiewicz, T., Ma, Y., Kasam, R.K., Shima, K., Chalk, C., Carey, B., Rosendale, L.R., Dominique Filippi, M. and Arumugam, P. 2022. Role of GM-CSF in regulating metabolism and mitochondrial functions critical to macrophage proliferation. Mitochondrion 62, 85–101. Yang, H., Xuefeng, Y., Shandong, W. and Jianhua, X. 2020. COX-2 in liver fibrosis. Clin. Chim. Acta. 506, 196–203. Wilujeng, S., Prakoso, Y.A. and Wirjaatmadja, R. 2023. Effects of extraction, fermentation, and storage processes on the level of choline derived from calabash fruit (Crescentia cujete L.). J Res Pharm. 27(2), 620 -626. Zhang, F., Wang, Y., Liu, Y., Han, H., Zhang, D., Fan, X., Du, X., Gamper, N. and Zhang, H. 2019. Transcriptional regulation of voltage-gated sodium channels contributes to gm-csf-induced pain. J. Neurosci. 39(26), 5222–5233. Zhang, J., Zhao, Y., Jing, Y., Han, L., Ma, X., Yu, L. and Diao, T. 2023. The plasma fibrinogen levels in the nitroglycerin-induced chronic migraine rat model and its association between migraine-associated vestibular dysfunction. Front. Neurol. 14, 980543. Zeisel, S.H., Klatt, K.C. and Caudill, M.A. 2018. Choline. Adv. Nutr. 9(1), 58 -60. Zhu, J., Lian, J., Wang, X., Wang, R., Pang, X., Xu, B., Wang, X., Li, C., Ji, S. and Lu, H. 2023. Role of endogenous and exogenous antioxidants in risk of six cancers: evidence from the Mendelian randomization study. Front. Pharmacol. 14, 1185850. Zhu, J., Wu, Y., Tang, Q., Leng, Y. and Cai, W. 2014. The effects of choline on hepatic lipid metabolism, mitochondrial function and antioxidative status in human hepatic C3A cells exposed to excessive energy substrates. Nutrients 6, 2552–2571. Zhuang, Y., Wang, C., Wu, C., Ding, D., Zhao, F., Hu, C., Gong, W., Ding, G., Zhang, Y., Chen, L., Yang, G., Zhu, C., Zhang, A. and Jia, Z. and Huang, S. 2018. Mitochondrial oxidative stress activates COX-2/mPGES-1/PGE2 cascade induced by albumin in renal proximal tubular cells. Oncotarget 9(10), 9235–9245. | ||
| How to Cite this Article |
| Pubmed Style Prakoso YA, Isla KJV, Wijayanti AD. Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Vet. J.. 2025; 15(12): 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 Web Style Prakoso YA, Isla KJV, Wijayanti AD. Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. https://www.openveterinaryjournal.com/?mno=233334 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.37 AMA (American Medical Association) Style Prakoso YA, Isla KJV, Wijayanti AD. Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Vet. J.. 2025; 15(12): 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 Vancouver/ICMJE Style Prakoso YA, Isla KJV, Wijayanti AD. Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 Harvard Style Prakoso, Y. A., Isla, . K. J. V. & Wijayanti, . A. D. (2025) Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Vet. J., 15 (12), 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 Turabian Style Prakoso, Yos Adi, Krestel Joy Viernes Isla, and Agustina Dwi Wijayanti. 2025. Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Veterinary Journal, 15 (12), 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 Chicago Style Prakoso, Yos Adi, Krestel Joy Viernes Isla, and Agustina Dwi Wijayanti. "Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats." Open Veterinary Journal 15 (2025), 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 MLA (The Modern Language Association) Style Prakoso, Yos Adi, Krestel Joy Viernes Isla, and Agustina Dwi Wijayanti. "Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats." Open Veterinary Journal 15.12 (2025), 6527-6540. Print. doi:10.5455/OVJ.2025.v15.i12.37 APA (American Psychological Association) Style Prakoso, Y. A., Isla, . K. J. V. & Wijayanti, . A. D. (2025) Effects of repeated 14-day oral toxicity of fermented Crescentia cujete (L.) on histopathology, GM-CSF, COX-2, antioxidants, and MDA levels in male Sprague Dawley rats. Open Veterinary Journal, 15 (12), 6527-6540. doi:10.5455/OVJ.2025.v15.i12.37 |