| Research Article | ||
Open Vet. J.. 2026; 16(1): 401-418 Open Veterinary Journal, (2026), Vol. 16(1): 401-418 Research Article Effect of metformin treatment on ovarian function in hypothyroid ratsNabil Abbas Soliman1*, Amr A. Shalaby1, Khaled A. Abulfadle2, Heba Mohammed3, Samia Hussien1, Mohamed Ahmed Ammar4, Aya Sh. Metwally5 and Eman E. L. Bahaie11Zoology Department, Faculty of Science, Zagazig University, Sharkya, Egypt 2Physiology Department, Faculty of Medicine, Zagazig University, Sharkya, Egypt 3Human Anatomy and Embryology Department, Faculty of Medicine, Zagazig University, Sharkya, Egypt 44Doctor of Physiological Zoology, Faculty of Science, Abu Surra University of Medical Sciences, Zawia City, Libya 5Assistant Professor of Pharmacology, Faculty of Veterinary Medicine, Aswan University, Egypt *Corresponding Author: Nabil A. Soliman. Zoology Department, Faculty of Science, Zagazig University, Zagazig, Egypt.Email: nabilSoliman54 [at] yahoo.com Submitted: 30/10/2025 Revised: 02/12/2025 Accepted: 06/12/2025 Published: 31/01/2026 © 2025 Open Veterinary Journal
AbstractBackground: Among women in their reproductive years, hypothyroidism is a prevalent endocrine condition linked to insulin resistance and abnormalities in ovarian function. Aim: The current study sought to determine how metformin treatment affected thyroid-stimulating hormone (TSH) levels, thyroid morphology in hypothyroidism, and ovarian alterations in polycystic ovary syndrome. It also sought to determine whether metformin was beneficial as an insulin sensitizer. Methods: Four equal groups of forty adult, healthy female albino rats of the local strains weighed an average of 150–170 g apiece. Group III and Group IV received propylthiouracil (Thyrocil 50 mg tablets, Amoun, Egypt) dissolved in distilled water (15 mg/kg body weight) orally for 45 days by dose calculation equation is Dosage each Rats=15 mg × weight of Rat (gm)/1,000 gm, while Group I received distilled water orally for 90 days. Group II received distilled water orally daily for the first 45 days of the study, and then received metformin Hydrochloride (1 g tablets, Chemical Industries Development, Giza) (50 mg/100 g body weight), daily from the 46th day of the study until the end (90th day). Group III received 1 ml of distilled water orally every day from the 46th day of the study until the study's 90th day. Group IV was administered metformin orally at a dose of (50 mg/100 g bwt) every day starting on the 46th day of the research and continuing until the 90th day. Biochemical measurements, thyroid, and ovarian histology were examined. Results: These findings supported the hypothesis that thyroid hypofunction is linked to disrupted metabolic and ovarian function. They also showed that metformin treatment improved thyroid and ovarian changes in a rat model of hypothyroidism. Conclusion: The anti-inflammatory and antioxidant characteristics of metformin were confirmed, as they enhanced ovarian function alterations while having no influence on ovarian modifications in the rat model of hypothyroidism. These results showed that metformin had no effect on ovarian architecture in euthyroid rats. Keywords: Metformin, Propylthiouracil, Hypothyroidism, Ovary. IntroductionWhen the thyroid gland does not create enough thyroid hormones, the condition is known as hypothyroidism (Krysiak et al., 2016). Triiodothyronine (T3) and thyroxin (T4), which are vital for cell growth and development, are produced, stored, and released by the thyroid gland (Vargas-Uricoechea and Bonelo-Perdomo, 2017). Globally, thyroid disorders are extremely common (Müller et al., 2001; Atasayar and Guler Demir, 2019). Among the most prevalent endocrinopathies in women of reproductive age, thyroid problems impact approximately 14% of adult women (Strikić Đula et al., 2022). Thyroid hormones have a significant impact on sexual and reproductive processes in both humans and animals, according to Nelson et al. (2011). Additionally, Brown et al. (2023)observed a decline in ovarian follicle quantity in hypothyroid rats correlated with a drop in Follicle-Stimulating Hormone (FSH) levels. Furthermore, Silva et al. (2018) observed elevated serum levels of Luteinizing hormone (LH) in rats with hypothyroidism. Regrettably, a number of thyroid conditions have negative implications on metabolic and reproductive health (Shekhar et al., 2020). Metabolic effects of thyroid hormones. The thyroid hormones increase the basal metabolic rate and have effects on almost all body tissues. Appetite, absorption of substances, and gut motility are all influenced by thyroid hormones (Villar et al., 2007 ). Thyroid hormones act to increase protein turnover. This might serve an adaptive function in regard to long-term calorie restriction with adequate protein (Fontana et al., 2006). They increase the absorption in the gut, generation, uptake by cells, and breakdown of glucose. They stimulate the breakdown of fats and increase the number of free fatty acids. Despite increasing free fatty acids, thyroid hormones decrease cholesterol levels, perhaps by increasing the rate of secretion of cholesterol in bile (Hall et al., 2011). For example, polycystic ovary syndrome (PCOS), a reproductive condition marked by hyperandrogenism, ovulatory dysfunction, and polycystic ovarian morphology, is linked to subclinical hypothyroidism (SCH) (Fatima et al., 2020). Comorbidities as insulin resistance and dyslipidaemia are linked to SCH in women with PCOS (de Medeiros et al., 2018). The primary first-line treatment for type 2 diabetes is metformin, which is marketed under the brand names Glucophage and other names (Maruthur et al., 2016; Cosentino et al., 2020; Draznin et al., 2022), especially in overweight individuals. Furthermore, Jensterle et al. (2020) discovered that long-term metformin treatment led to body mass index (BMI) reduction and stabilisation, improvements in the androgen profile, and a low conversion rate to diabetes in overweight-obese women with PCOS and normal baseline glycaemmeostasis. One of the safest anti-hyperglycemic medications is metformin. Gastrointestinal (GI) intolerance, which includes nausea, diarrhoea, dyspepsia, and stomach discomfort, is the primary adverse effect. Less than 2% of patients discontinue therapy due to GI symptoms, despite the fact that they may be present in up to 28% of patients (Blonde et al., 2004). TSH levels were significantly lowered in obese diabetic women with primary hypothyroidism as a result of metformin treatment; however, TH levels remained unchanged and were restored upon stopping the medication. It's interesting to see that the baseline TSH concentration and the extent of TSH reduction were related. In 2007 (Isidro et al., 2007). TSH levels in patients with T2DM or PCOS have been shown to drop after taking metformin, according to a number of studies (Isidro et al., 2007; Ittermann et al., 2013). Furthermore, a number of studies have connected the usage of metformin with a lower incidence and mortality rate from cancer (Col et al., 2012; Noto et al., 2012). Currently ranked among the most used oral hypoglycemic medications worldwide, metformin is a cornerstone medication for both diabetes prevention and management (Ezzamouri et al., 2023). Numerous studies have demonstrated that, as metformin use increases, it may also be crucial for preserving cardiovascular health, delaying the ageing process, and halting the growth of tumors (Lascu et al., 2022; Wang et al., 2022; Nojima and Wada, 2023). Its beneficial effects on liver, lung, gastric, esophageal, and thyroid cancer have also been documented by numerous clinical trials (Jin et al., 2022; Lan et al., 2022; Sekino et al., 2022; Morale et al., 2022; Han et al., 2023). This study aimed to investigate the effects of metformin medication on ovarian changes in PCOS, thyroid morphology in hypothyroidism, and TSH levels. Whether metformin was advantageous as an insulin sensitizer was another goal it attempted to ascertain. Materials and MethodsBiochemical agents
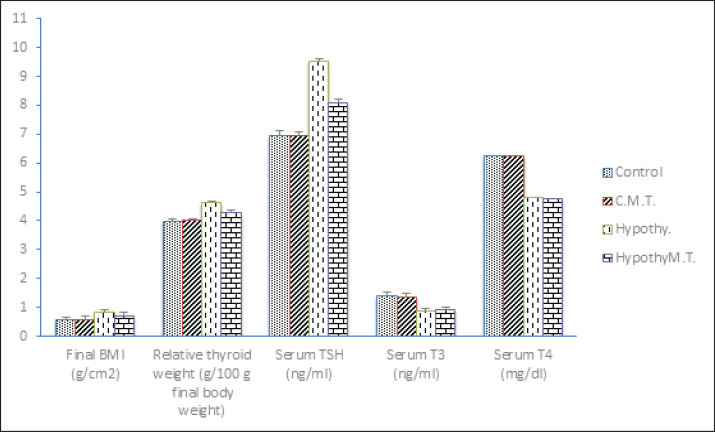
Material preparationVerma et al. (1994) reported that a suspension of metformin diluted in distilled water was given orally at a dose of (50 mg/100 g body weight). In order to cause hypothyroidism, a different solution containing PTU diluted in distilled water (15 mg/kg body weight) was given orally for 45 days (Cooper, 2005). Investigational animalsForty adult, healthy female albino rats were provided for the experiment by Zagazig University's Veterinary Laboratory Animal Farm. Ten rats per metallic cage were housed in a laboratory-style environment with temperature control and air exposure. The estimated animals had free reign over an ordinary meal (a viable rodent food) and an endless supply of water. All of the animals were kept under observation and acclimatisation for a duration of 2 weeks prior to the perfect conditions already starting the trial phase. Experimental designRats weighing between 150 and 170 g at baseline were randomly assigned to four equal groups. The control group, designated as Group I, received oral distilled water. Rats in Group II were given distilled water orally for the first 45 days of the study, and from the 46th day until the 90th day, they were given metformin (50 mg/100 g body weight) orally. Groups III were given PTU dissolved in distilled water (15 mg/kg body weight) orally for 45 days, then they given distilled water from the 46th day of the trial until the 90th day, Group IV were given PTU dissolved in distilled water (15 mg/kg body weight) orally for 45 days and from the 46th day they took metformin daily, until the 90th day. For the histology studies, the thyroid and ovaries of every rat were cleansed with regular cold saline, dried with filter paper, and then preserved in 10% formalin-Saline. Inspection or examinationRats that were fasting had their blood drawn to create serum samples. Blood samples were brought in non-heparinized tubes under light ether anesthesia on the last day of the dosage. At the end of the experiment, all animals were fasted overnight and subjected to light diethy ether anesthesia (through inhalation) before sacrifice. Blood specimens (3–7 ml) were withdrown from the retro-orbital plexus using sterile hepariniged glass capillary (singl drow vascutainer neddle) into collecting tubes and left for 10 minutes to coagulate, then centrifuged at 3,000 rpm for 10 minutes using a cooling centrifuge (Andreas Hettich GmbH Co.12D-78532 Tuttlingen Germany). Finally, the serum was separated at once divided into aliguote and stored at −20ºC until carrying out the biochemical measurement as soon as possible. Thyroid function tests: serum T3, T4, and TSH analyses from serum were measured using the enzyme-linked immunosorbent test in an ELISA device with a commercial test kit (competitive ELISA kit) according to Burger and Patel 1977. Serum FSH, LH (Tietz, 1995), Cata FSH: BC-1031, BioCheck, Inc 323 Vintage Park Dr. Foster City, CA 94404 estradiol, progesterone and testosterone (Tietz, 1995), serum levels were quantitatively measured by BC-1111 from BioCheck, Inc. at 323 Vintage Park Dr. Foster City, CA 94404 Serum glucose and insulin levels, Calculation of Homeostatic Model Assessment - Insulin Resistance (HOMA-IR)=insulin (µU/ml) × glucose (mg/dl) /405 and Homeostatic Model Assessment - Beta-cell function (HOMA-B)=360 × fasting insulin (μU/ml) / (fasting glucose (mg/dl) - 63) . Lipid profile: serum triglyceride, cholesterol (Tietz, 1995), and High-Density Lipoprotein (HDL) Calculation of low-density lipoprotein-cholesterol (LDL) level: LDL. Serum inflammatory markers: tumor necrosis factor alpha (TNF-α) and interleukin 6 (IL-6) levels and Serum antioxidant markers: superoxide dismutase and catalase levels were measured. Kits were provided by Biodiagnos-tics (Giza, Egypt) Following the animals' sacrifice, each rat's entire thyroid and ovary were taken, cleaned in regular saline, dried with filter paper, and preserved in 10% formalin-Saline at room temperature for the histology analyses. The international federation of clinical chemistry with T3 Enzyme-Linked Immunosorbent Assay (ELISA) kit (Catalogue No: 201-11-0535, T4 ELISA kit (Catalogue No: 201–11–0338, TSH ELISA kit (Catalogue No: 201–11–0181, FSH ELISA kit (Catalog Number: BC-1029), LH ELISA kit (Catalog Number: BC-1031), Estradiol (E2) Enzyme-Linked Immunosorbent Assay Kit: (Catalog Number: 2011) used for the in vitro quantitative measurement of estradiol in serum by the immunoassay analyzers. Progesterone Enzyme-Linked Immunosorbent Assay Kit: (Catalog Number: 2011) used for the in vitro quantitative measurement of progesterone in serum by the immunoassay analyzers. Testosterone ELISA kit (Catalog Number: BC–1,115). Glucose (GOD-PAP)-Liquizyme Kit: Enzymatic colorimetric method for quantitative measurement of glucose in the serum. Insulin Enzyme-Linked Immunosorbent Assay Kit: (Product Number: RAB0904) for the quantitative measurement of Insulin in serum by the immunoassay analyzers. Cholesterol Enzyme-Linked Immunosorbent Assay Kit: (Catalog Number: 2,011). Triglycerides Enzyme-Linked Immunosorbent Assay Kit: (Catalog Number: 2,011), HDL-Cholesterol Enzyme-Linked Immunosorbent Assay Kit: (Catalog Number: 2,011), TNF-α Immunoassay Kits: (Catalog Number KRC3011 ), IL-6 ELISA kit (Catalogue No: 201–11–0136), Superoxide dismutase (SOD), and Catalase ELISA kit (Egyptian Company for Biotechnology, Obour city, Cairo, Egypt). Statistical analysisThe information was displayed in triplicate as mean ± standard deviation (SD). When comparing two-group variance, the data were statistically examined utilizing the unpaired Student's t-test, which was followed by a one-way ANOVA. The Statistical Package for Social Sciences (SPSS), 16 ver. for Windows, Chicago, (SPSS Inc) was utilized throughout this analysis. Ethical approvalThe research was conducted at Zagazig University's Faculty of Medicine's Scientific & Medical Research Centre, approved by the International Animals and Use Committee and the ZU-IACUC Committee. The approval number is (ZU-IACUC/1/F/44/2023). ResultsFinal BMI and relative thyroid weight changes among different studied groupsThere was no significant change (p > 0.05) in both final BMI and relative thyroid weight in the control metformin-treated group in comparison to that in the control group. On the other hand, there was a significant increase (p < 0.05) in both final BMI and relative thyroid weight in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in both final BMI and relative thyroid weight in comparison to that in hypothyroid rats (Table 1 and Fig. 1).
Fig. 1. Final BMI and relative thyroid weight changes besides Thyroid function changes among different studied groups. Table 1. Comparison between the studied groups.
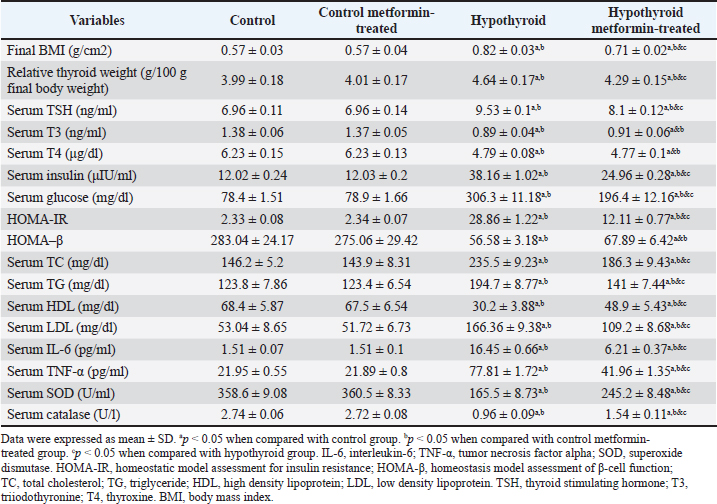
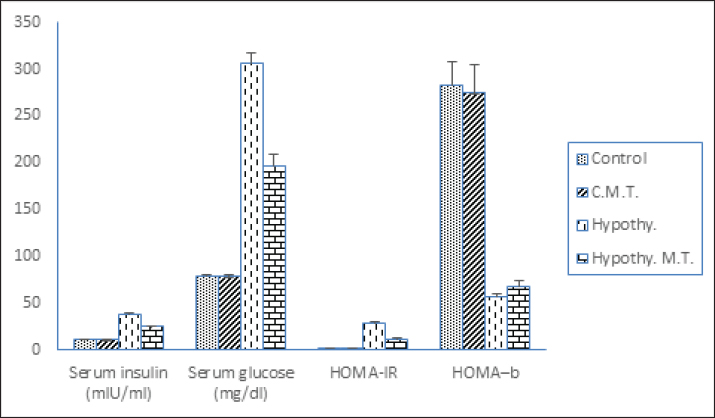
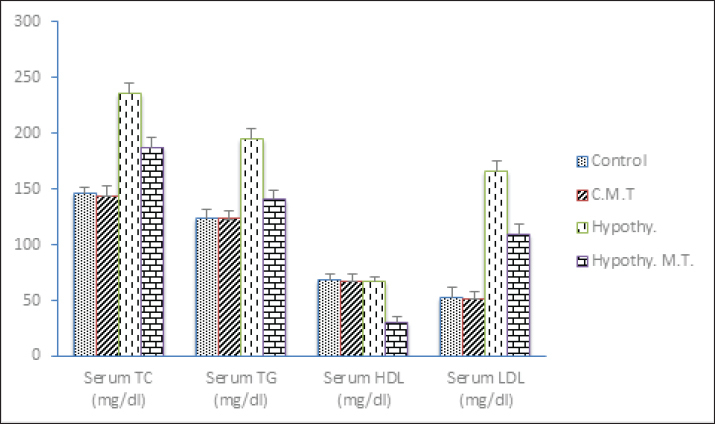
Thyroid function changes among different studied groupsThere were no significant changes (p > 0.05) in serum levels of TSH, T3, and T4 in the control metformin-treated group in comparison to that in control group. On the other hand, there was a significant increase (p < 0.05) in serum level of TSH, with a significant decrease (p < 0.05) in serum levels of T3 and T4 in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in serum level of TSH in comparison to that in hypothyroid rats. There were no significant changes (p > 0.05) in serum levels of T3 and T4 in the hypothyroid metformin-treated group in comparison to that in hypothyroid group (Table 1 and Fig. 1). Metabolic changes among different studied groupsThere were no significant changes (p > 0.05) in serum levels of (insulin, glucose, TC, TG, HDL, and LDL), HOMA-IR, and HOMA-B in the control metformin-treated group in comparison to that in control group. On the other hand, there was a significant increase (p < 0.05) in serum levels of (insulin, glucose, TC, TG, and LDL), and HOMA-IR, with a significant decrease (p < 0.05) in HOMA-IR and serum levels of HDL in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in serum levels of (insulin, glucose, TC, TG, and LDL), and HOMA-IR, with a significant increase (p < 0.05) in serum levels of HDL in comparison to that in hypothyroid rats. There were no significant changes (p > 0.05) in HOMA-· in the hypothyroid metformin-treated group in comparison to that in the hypothyroid group (Table 1 and Figs. 2 and 3).
Fig. 2. Serum levels of (insulin, glucose,), HOMA-IR and HOMA- B, changes among different studied groups.
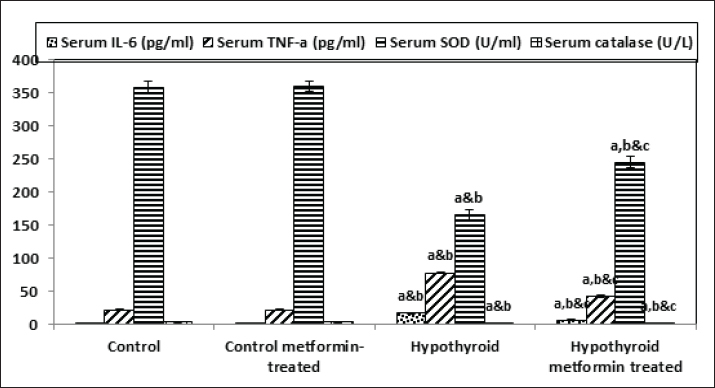
Fig. 3. Serum levels of TC, TG, HDL, and LDL among different studied groups. Inflammatory and oxidative stress biomarkers changes among different studied groupsThere were no significant changes (p > 0.05) in serum levels of IL-6, TNF-·, SOD, and catalase in control metformin-treated group in comparison to that in control group. On the other hand, there was a significant increase (p < 0.05) in serum levels of IL-6 and TNF-α, with a significant decrease (p < 0.05) in serum levels of SOD and catalase in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in serum levels of IL-6 and TNF-·, with a significant increase (p < 0.05) in serum levels of SOD and catalase in comparison to that in hypothyroid rats (Table 1 and Fig. 4).
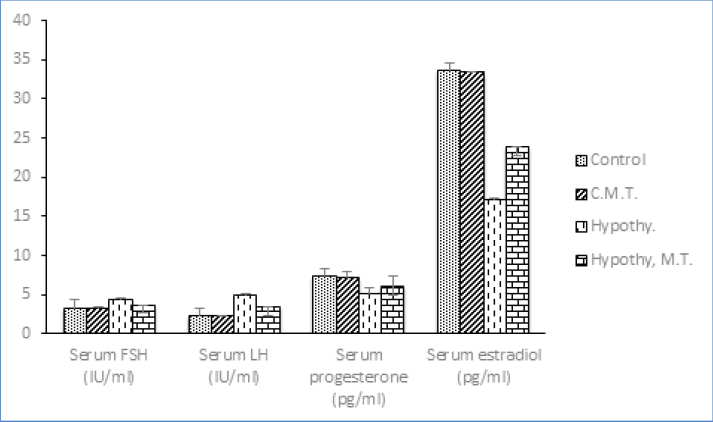
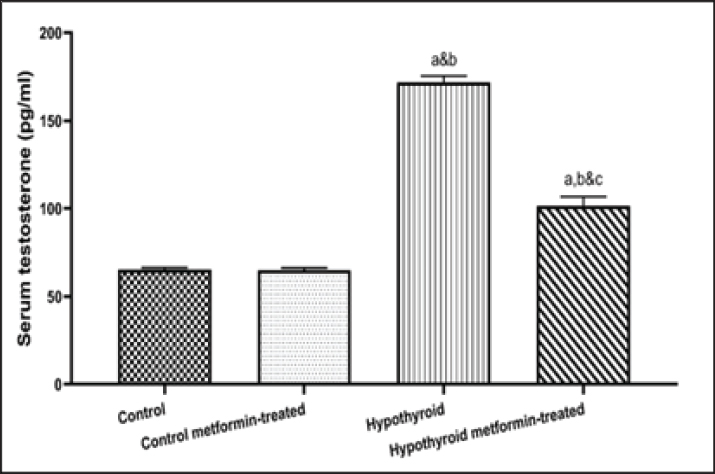
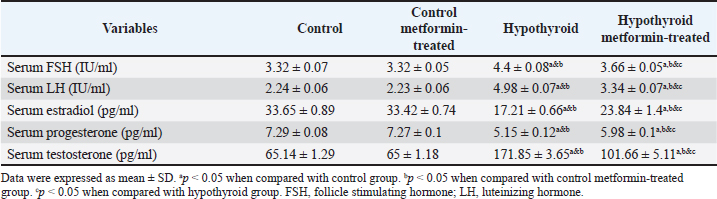
Fig. 4. Inflammatory and oxidative stress biomarkers changes among different studied groups. Ovarian function changes among different studied groupsThere were no significant changes (p > 0.05) in serum levels of FSH, LH, estradiol, progesterone, and testosterone in control metformin-treated group in comparison to that in control group. On the other hand, there was a significant increase (p < 0.05) in serum levels of FSH, LH and testosterone, with a significant decrease (p < 0.05) in serum levels of estradiol and progesterone in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in serum levels of FSH, LH, and testosterone, with a significant increase (p < 0.05) in serum levels of estradiol and progesterone in comparison to that in hypothyroid rats (Table 2 and Figs. 5 and 6).
Fig. 5. Ovarian function changes among different studied groups.
Fig. 6. Serum testosterone (pg/ml) changes among different studied groups. Table 2. Ovarian function changes among different studied groups.
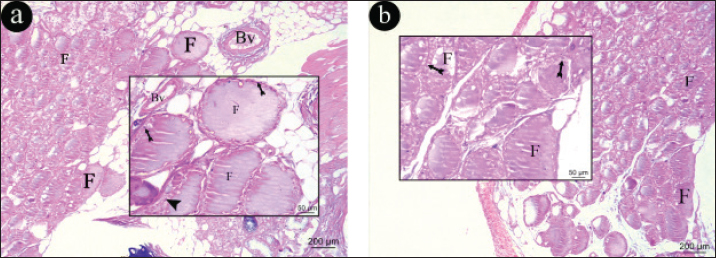
Histopathological analysis results H&EThyroid glandThyroid section of the control and control-metformin treated groups revealed a normal structure of thyroid gland as it appeared formed of different size follicles filled with acidophilic colloid, lined with low cuboidal epithelium lining with rounded nucleus, with inter follicular cells and few blood vessels in between (Fig. 7a and b).
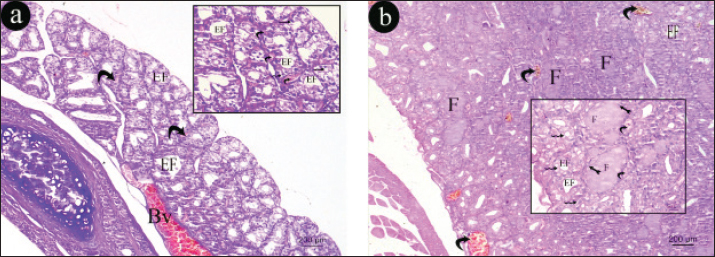
Fig. 7. Representative photomicrographs of H&E-stained sections of thyroid gland. a) thyroid section of the control group, b) thyroid section of the control-metformin treated group. Thyroid gland is formed of different size follicles filled with acidophilic colloid (F), lined with low cuboidal epithelium lining with rounded nucleus (tailed arrow), with inter follicular cells (arrow) and few blood vessels (Bv) in between (H&E × 100 & inset magnification × 400). Thyroid gland of hypothyroid group appeared formed mostly of small size empty follicles, lined with vacuolated epithelium lining, with congested blood vessel in the periphery and multiple congested blood vessels in between the follicles (Fig. 8a) while, thyroid section of the hypothyroid-metformin treated group appeared having different size follicles some were filled with acidophilic colloid while others were empty, lined with low cuboidal epithelium lining with rounded nucleus, some epithelial lining were vacuolated with few congested blood vessels in between the follicles (Fig. 8b).
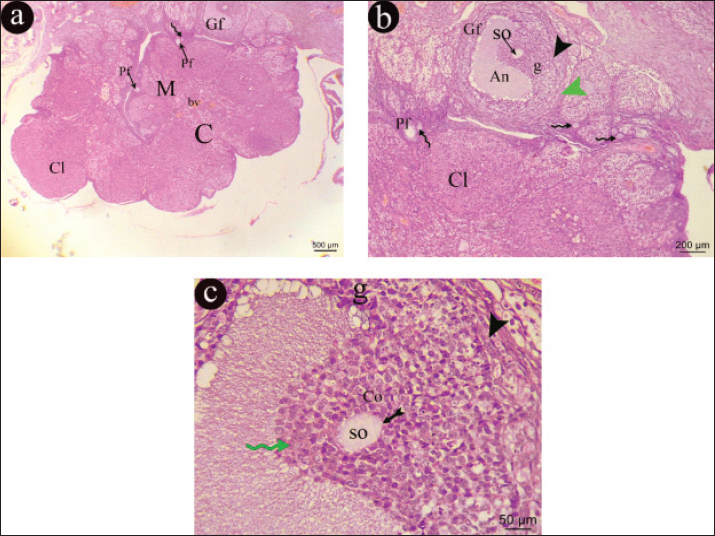
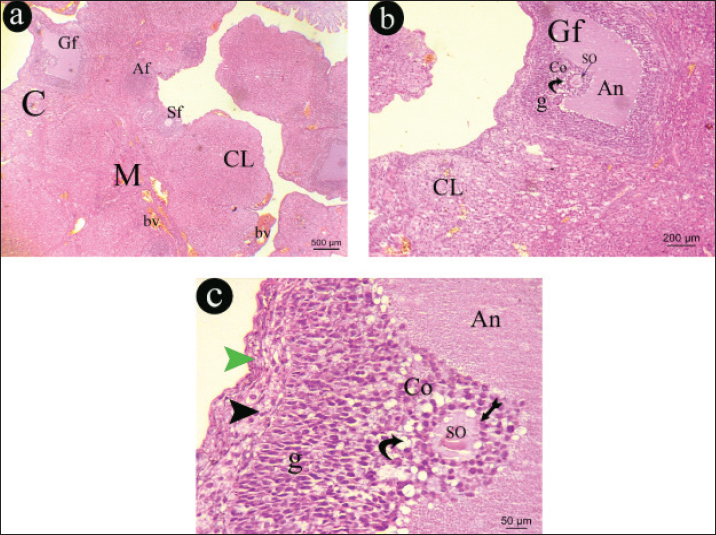
Fig. 8. Representative photomicrographs of H&E-stained sections of thyroid gland. a) thyroid section of the hypothyroid group, Thyroid gland is formed of mostly small size empty follicles (EF), lined with vacuolated epithelium lining (zigzag arrow ), with congested blood vessel (Bv)in the periphery and multiple congested blood vessels in between the follicles(curved arrow). b) thyroid section of the hypothyroid-metformin treated group. Thyroid gland is formed of different size follicles some are filled with acidophilic colloid (F) while others are empty (EF), lined with low cuboidal epithelium lining with rounded nucleus (tailed arrow),some epithelial lining are vacuolated (zigzag arrow) with few congested blood vessels in between the follicles (curved arrow) (H&E × 100 & inset magnification × 400). OvaryThe ovarian sections of control and control-metformin groups appeared formed of cortex and medulla. Cortex had multiple stages of ovarian follicles, primordial follicle, primary follicle, corpus luteum. Graafian follicle that had secondary oocyte surrounded by zona pellucida, corona radiate, and attached by cumulus oophorus to the wall of follicle. The antrum of Graafian follicle was surrounded with granulosa cells, theca interna cells, and theca externa cells (Figs. 9 and 10).
Fig. 9. Representative photomicrographs of H&E-stained sections of thyroid gland. a) thyroid section of the hypothyroid group, Thyroid gland is formed of mostly small size empty follicles (EF), lined with vacuolated epithelium lining (zigzag arrow ), with congested blood vessel (Bv)in the periphery and multiple congested blood vessels in between the follicles(curved arrow). b) thyroid section of the hypothyroid-metformin treated group. Thyroid gland is formed of different size follicles some are filled with acidophilic colloid (F) while others are empty (EF), lined with low cuboidal epithelium lining with rounded nucleus (tailed arrow),some epithelial lining are vacuolated (zigzag arrow) with few congested blood vessels in between the follicles (curved arrow) (H&E × 100 & inset magnification × 400).
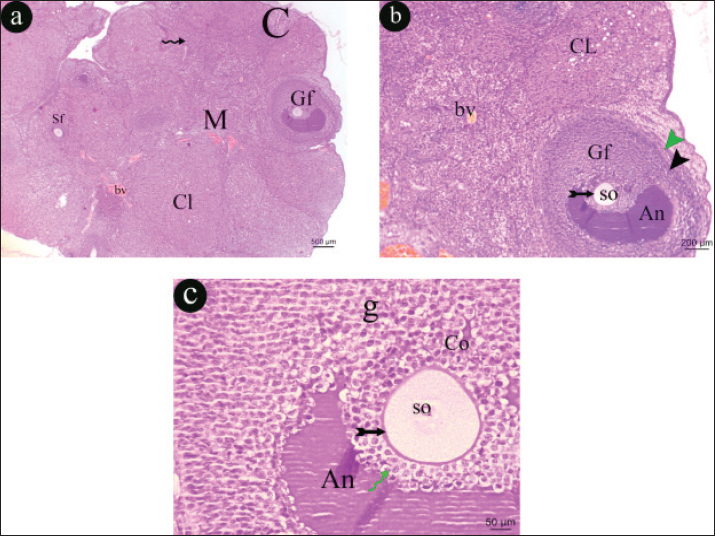
Fig. 10. A representative photomicrographs of H&E section in an ovary of control rat. a) The ovarian section appears formed of cortex (c) and medulla (M), with corpus luteum (Cl), Graafian follicle (Gf) in cortex and small blood vessels(bv)within medulla (H&E, × 40). b) The cortex appears with multiple stages of ovarian follicles, primordial follicle (zigzag arrow), primary follicle (Pf) and corpus luteum (Cl). The Graafian follicle (Gf) is containing secondary oocyte (SO) and appeared with antrum (An) is surrounded with granulosa cells (g), theca interna cells (black arrow head) and theca externa cells (green arrow head) (H&E, × 100). c) The Graafian follicle (Gf) appears with antrum (An) surrounded with granulosa cells (g), theca interna cells (black arrow head). secondary oocyte (SO), surrounded by zona pellucida (tailed arrow), corona radiata (green zigzag arrow) and attached by cumulus oophorus (Co) (H&E, × 400). The ovarian cortex of hypothyroid group revealed multiple follicular stages with secondary follicles, primary follicles, multiple corpus luteum, multiple cystic follicles, and congested blood vessels in medulla. The cystic follicles were surrounded with vacuolated cells, and corpus luteum cells showed marked vacuolation, while the medulla was filled with congested blood vessels. The secondary follicle was lined with multiple-layered follicular cells with vacuolated cytoplasm was containing a primary oocyte was surrounded with zona pellucida. A small primary follicle appeared with containing a primary oocyte (Fig. 11).
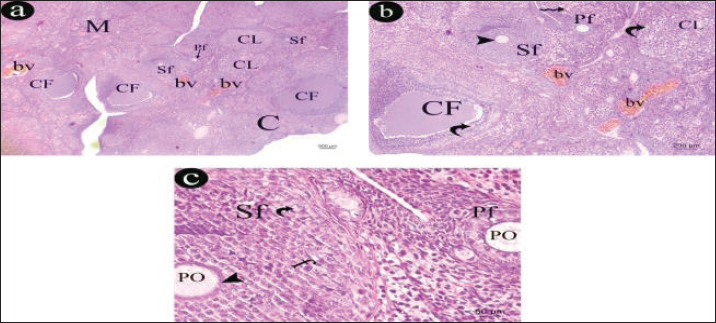
Fig. 11. A representative photomicrographs of H&E section in an ovary of control rat. a) The ovarian section appears formed of cortex (c) and medulla (M), with corpus luteum (Cl), Graafian follicle (Gf) in cortex and small blood vessels(bv)within medulla (H&E, × 40). b) The cortex appears with multiple stages of ovarian follicles, primordial follicle (zigzag arrow), primary follicle (Pf) and corpus luteum (Cl). The Graafian follicle (Gf) is containing secondary oocyte (SO) and appeared with antrum (An) is surrounded with granulosa cells (g), theca interna cells (black arrow head) and theca externa cells (green arrow head) (H&E, × 100). c)The Graafian follicle (Gf) appears with antrum (An) surrounded with granulosa cells (g), theca interna cells (black arrow head). secondary oocyte (SO), surrounded by zona pellucida (tailed arrow), corona radiata (green zigzag arrow) and attached by cumulus oophorus (Co) (H&E, × 400). The ovarian sections of hypothyroid-metformin groups appeared formed of cortex and medulla. cortex had multiple stages of ovarian follicles, primordial follicle, primary follicle, and corpus luteum. Graafian follicle that had a secondary oocyte surrounded by zona pellucida, corona radiate, and attached by vacuolated cumulus oophorus to the wall of follicle. The antrum of Graafian follicle was surrounded with vacuolated granulosa cells, theca interna cells, and theca externa cells. The medulla showed some congested blood vessels (Fig. 12).
Fig. 12. A representative photomicrographs of H&E sections in an ovary of hypothyroid- metformin treated rat. a) The ovarian section appears formed of cortex (c) and medulla (M), the cortex appears with multiple stages of ovarian follicles, secondary follicle (Sf), corpus luteum (Cl) and Graafian follicle (Gf) and atretic follicle (Af) with congested blood vessels (bv) within medulla (H&E, × 40) b) The cortex appears with corpus luteum (Cl) and Graafian follicle (Gf) is containing secondary oocyte (SO) and appeared with antrum (An) surrounded with vacuolated (curved arrow) granulosa cells (g) (H&E, × 100). c) The Graafian follicle (Gf) appears with antrum (An) surrounded with granulosa cells (g), theca interna cells (black arrowhead). secondary oocyte (SO), surrounded by zona pellucida (tailed arrow), and attached by vacuolated (curved arrow) cumulus oophorus (Co) (H&E, × 400). DiscussionTriiodothyronine and T4, which are vital for cell growth and development, are produced, stored, and released by the thyroid gland, an endocrine gland (Vargas-Uricoechea and Bonelo-Perdomo, 2017). One of the disorders of the thyroid gland that causes disruptions in the synthesis and secretion of hormones is hypothyroidism, which is characterised as a lack of thyroid hormone production (Priya et al., 2015). The current investigation sought to determine how hypothyroidism affected ovarian structure and function, as well as whether metformin could have any mitigating effects on hypothyroidism. Propylthiouracil is one of the essential agents that can cause hypothyroidism by decreasing circulating levels of T3 and T4, increasing levels of TSH by inhibiting peroxidase, and decreasing peripheral deiodinase. For this reason, we used it in this study to induce hypothyroidism (EL-Tantawi and Abozeid, 2019). According to the study's findings, a rat model of hypothyroidism showed improved ovarian and thyroid alterations when treated with metformin. The results of the present investigation indicate that metformin treatment has no effect on the final BMI or relative thyroid weight in euthyroid rats, as there was no statistically significant difference observed between the metformin-treated group and the control group. However, compared to the control and control metformin-treated groups, there was a significant increase in both the final BMI and the relative thyroid weight in the hypothyroid and hypothyroid metformin-treated groups. This finding is consistent with the findings of Nyrnes et al. (2006); Hayat et al. (2016); and Brown et al. (2023) that hypothyroidism is linked to an increase in body weight. Furthermore, BMI was found to be negatively connected with serum free T4 (FT4), positively correlated with serum TSH, and uncorrelated with serum free T3 (FT3), according to Knudsen et al. (2005). Furthermore, subclinical hypothyroidism has been demonstrated to be associated with a notable rise in body weight and to be a risk factor for overweight and obesity by Sanyal and Raychaudhuri (2016). They attributed this to the reduction in metabolic rate and thermogenesis that is seen in hypothyroidism. However, compared to the control group, morbidly obese individuals exhibited greater levels of total T3, FT3, total T4, and TSH (Michalaki et al., 2006). This was likely due to the reset of their central thyrostat at a higher level. Conversely, Figueroa et al. (2008) found no correlation between BMI and thyroid function in patients with subclinical hypothyroidism or euthyroid adults. The present investigation revealed that metformin administration significantly reduced the end BMI and relative thyroid weight of hypothyroid rats compared to hypothyroid rats. This finding may support the idea that metformin treatment can mitigate the alterations linked to hypothyroidism. This was consistent with the findings of Robert et al. (2006), who reported that using metformin for 2–8 months caused reversible reduction of TSH without affecting levels of free T4 or free T3. Conversely, Oleandri et al. (1999) observed no variation in thyroid function tests among 28 patients on metformin for obesity. Yerevanian and Soukas (2019) linked the weight reduction related with metformin to changes in the gut microbiome and hypothalamus appetite-regulating centres. The study's findings demonstrated that there were no appreciable variations in the TSH, T3, and T4 serum levels between the metformin-treated control group and the control group, indicating that metformin treatment had no influence on euthyroid function. Conversely, compared to the control and control metformin-treated groups, there was a substantial decrease in serum levels of T3 and T4 and a significant increase in serum TSH in the hypothyroid and hypothyroid metformin-treated groups. These results validated the success of the paradigm (primary hypothyroidism) and were consistent with Treesh and Khair (2014). As also stated by Choksi et al. (2003), the hypothyroid group's elevated serum TSH level is the consequence of decreased T3 and T4, which is caused by the pituitary gland's release of TSH via the negative feedback role. When hypothyroid rats are given metformin, their blood TSH levels significantly drop when compared to the hypothyroid rats' levels. This finding is consistent with the findings of Ittermann et al. (2013), who found that diabetic patients receiving metformin had smaller thyroid volumes and were less likely to develop thyroid nodules than controls. This finding suggests that metformin has anti-proliferative properties, which justifies its use in the treatment of thyroid proliferative diseases. Furthermore, blood levels of T3 and T4 in the hypothyroid metformin-treated group did not significantly differ from those in the hypothyroid group, which is consistent with Amirabadizadeh et al. (2023) and Krysiak et al. (2024). The hypothalamo-pituitary-thyroid axis is the target of a complex and multifaceted mode of action for metformin. Metformin may increase central dopaminergic tone, change the affinity and/or quantity of thyroid hormone receptors, or directly affect TSH regulation, all of which would enhance the impact of thyroid hormones on the pituitary gland (Krysiak and Okopien, 2011). Adenosine monophosphate-activated protein kinase (AMPK) pathway may be the mechanism by which metformin's central effects on thyrotropin-releasing hormone (TRH)/TSH regulation are mediated (Lopez et al., 2010). In contrast to its peripheral effects, metformin increases the inhibitory effect of thyroid hormones on TSH secretion and inhibits AMPK activity in the hypothalamus (Amirabadizadeh et al., 2023). According to our findings, the serum levels of insulin, glucose, TC, TG, HDL, and LDL, as well as HOMA-IR and HOMA-·, did not significantly differ between the metformin-treated control group and the control group, indicating that metformin had no effect on euthyroid rats' metabolism. In contrast to the control and control metformin-treated groups, the hypothyroid and hypothyroid metformin-treated groups showed a significant decrease in serum levels of HDL and a significant increase in serum levels of insulin, glucose, TC, TG, and LDL. Abulfadle and Ahmed (2019) provided support for these findings. Furthermore, according to ROOS et al. (2007), serum FT4 was only positively correlated with HDL and negatively correlated with total cholesterol and triglycerides, whereas serum TSH levels were only positively correlated with triglycerides. However, compared to hypothyroid rats, metformin-treated hypothyroid rats exhibited a large rise in HDL serum levels and a significant decrease in insulin, glucose, TC, TG, and LDL serum levels, as well as HOMA-IR. Its insulin sensitising properties may account for the improvement in metabolic alterations (Zhao et al., 2021). According to Dehkordi et al. (2018), metformin also effectively lowers insulin resistance, fasting plasma insulin level, the peripheral blood glucose by AMPK, and the synthesis of glucose from liver cells (gluconeogenesis). When comparing the hypothyroid metformin-treated group to the hypothyroid group, there were no appreciable differences in HOMA-β. According to the current findings, there were no appreciable differences between the metformin-treated control group's serum levels of IL-6, TNF-α, SOD, and catalase and those of the control group. However, compared to the control and control metformin-treated groups, there was a substantial drop in blood levels of SOD and catalase and a significant increase in serum levels of IL-6 and TNF-α in the hypothyroid and hypothyroid metformin-treated groups. These findings were consistent with those of Khodabandeh et al. (2022), who discovered that hypothyroidism is linked to lower blood SOD levels while MDA levels are elevated. They also validated the existence of inflammation and oxidative stress, which are connected with hypothyroidism and may contribute to its pathogenesis. According to Agarwal et al. (2012), oxidative stress disrupts the balance between pro-oxidant molecules like reactive oxygen species (ROS) and defensive antioxidants, which appears to be a major factor in the pathophysiology of infertility. Furthermore, according to Jha et al. (2018), an increase in inflammatory markers is associated with an increase in ROS formation, which in turn contributes to tissue damage. Agarwal et al. (2008) provided an explanation for this, stating that the highly reactive unstable radicals injure cells by obtaining their electrons from intracellular biological components like proteins, lipids, nucleic acids, and carbohydrates. According to Shokoohi et al. (2019), ovarian tissue alterations brought on by oxidative stress are detrimental. However, compared to hypothyroid rats, metformin-treated hypothyroid rats exhibited a large rise in serum levels of catalase and SOD and a significant drop in serum levels of TNF-α and IL-6. According to these findings, metformin has anti-oxidant and anti-inflammatory properties in rats that have hypothyroidism. When ovarian function changes were examined between the several groups under investigation, there was a significant increase (p < 0.05) in serum levels of FSH, LH, and testosterone, with a significant decrease (p < 0.05) in serum levels of estradiol and progesterone in hypothyroid and hypothyroid metformin-treated groups in comparison to that in control and control metformin-treated groups. But hypothyroid rats treated with metformin showed a significant decrease (p < 0.05) in serum levels of FSH, LH, and testosterone, with a significant increase (p < 0.05) in serum levels of estradiol and progesterone in comparison to that in hypothyroid rats. These findings suggested that hypothyroidism may have an impact on ovarian function. These findings supported the findings of Nelson et al. (2011), who found that thyroid hormones significantly impacted the reproductive and sexual systems of both humans and animals. Additionally, Rashied et al. (2024) observed a decline in ovarian follicle quantity in hypothyroid rats correlated with a drop in FSH levels. Furthermore, Silva et al. (2018) observed elevated serum levels of LH in rats with hypothyroidism. According to Panciera et al. (2012), hypothyroidism is also linked to anovulation, ovarian cyst development, irregular oestrus cycles, and infertility, among other compromised reproductive processes. Furthermore, in hypothyroid rats, AL-Saaidi and AL-Bedary (2022) observed a reduction in ovarian folliculogenesis and serum levels of FSH and oestradiol. Additionally, hypothyroidism has been shown to negatively impact reproductive physiology and certain metabolic processes, which are compromised by problems related to sex hormones that follow thyroid hormone suppression (Seifi et al., 2022). Furthermore, hypothyroidism has been linked to menstrual irregularities, anovulation, and hyperprolactinemia, which result in ovulation disorders and decreased fertility in women of childbearing age. These factors also increase the risk of miscarriage, early delivery, placental abruption, and poor perinatal outcomes with low birth weight, according to Hapon et al. (2010). However, compared to hypothyroid rats, metformin-treated hypothyroid rats exhibited significantly higher serum levels of progesterone, progesterone, and oestradiol and significantly reduced serum levels of FSH, LH, and testosterone. These results confirmed that the changes in ovarian function observed in the rat model of hypothyroidism were improved by metformin. Serum TSH showed an inverse relationship with serum levels of HDL, oestradiol, T4, T3, and SOD, while demonstrating a notable positive correlation with BMI, HOMA-IR, and serum levels of TC, LDL, FSH, LH, testosterone, glucose, insulin, and TNF-α. These results bolstered the hypothesis that abnormal thyroid function is associated with changes in ovarian and metabolic functions. These findings concurred with those of Kabodmehri et al. (2021), who established the negative correlation between blood TSH levels and ovarian function. In the thyroid section of the control and control metformin-treated groups, the thyroid gland was shown to have a normal structure consisting of follicles of varying sizes filled with acidophilic colloid, surrounded by low cuboidal epithelium lining a rounded nucleus, interfollicular cells, and a few blood vessels. These findings demonstrated that in euthyroid rats, metformin had no effect on ovarian anatomy. According to histopathological findings, the thyroid gland in the hypothyroid group is primarily composed of small, empty follicles that are surrounded with vacuolated epithelium. The follicles have several congested blood vessels in between them and a periphery that is coated with a congested blood vessel. These outcomes highlighted how well hypothyroidism was induced. The findings were corroborated by Kabodmehri et al. (2021), who reported that thyroid hormone receptors are expressed by a variety of ovarian cells, including granulosa cells, oocytes, and epithelium. Follicle-stimulating hormone and TSH work in concert to encourage the growth of granulosa cells (Vissenberg et al., 2015). However, Zhang et al. (2013) have demonstrated that thyroid hormones control the activation of FSH in follicles and stop them from apoptosing. Furthermore, Kabodmehri et al. (2021) discovered that follicular fluid contains thyroid hormone. It plays a key function in the process of follicle growth, and its dysregulation may hamper follicular development. Thyroid section of the hypothyroid metformin-treated group appeared having different size follicles some were filled with acidophilic colloid while others were empty, lined with low cuboidal epithelium with rounded nucleus, some epithelial linings were vacuolated with few congested blood vessels in between the follicles. These findings demonstrated that metformin medication had a positive effect on thyroid structure in hypothyroidism. The cortical and medulla-shaped ovarian sections were observed in the control and control metformin-treated groups. Multiple phases of ovarian follicles were present in the cortex, including the primordial, primary, corpus luteum, and Graafian follicles, which contained secondary oocytes surrounded by zona pellucida and corona radiata and linked to the follicle wall by cumulus oophorus. Granulosa, theca interna, and theca externa cells encircled the antrum of the Graafian follicle. These findings corroborated those of Brent (2012), who discovered that the regulation of growth, metabolism, development, and ovarian differentiation depends on proper thyroid hormone levels. The ovarian cortex of hypothyroid group revealed multiple follicular stages with secondary follicles, primary follicles, multiple corpus luteum, multiple cystic follicles, and congested blood vessels in medulla. The cystic follicles were surrounded with vacuolated cells, and corpus luteum cells showed marked vacuolation. The primary oocyte within the secondary follicle was encircled by zona pellucida and bordered by multiple-layered follicular cells with vacuolated cytoplasm. These findings corroborated Mcdermott (2012) assertion that hypothyroidism in females reduced fertility via altering endometrial and ovarian structure, which hypothyroidism deteriorates ovarian structure. Our study's hypothyroid rats' numerous corpora lutea could be attributed to elevated serum levels of LH, which is consistent with Silva et al. (2018). Moreover, Kumar et al. (2015) linked the large corpora lutea found in the ovaries of hypothyroid rats to a decrease in thyroid hormone levels, which are necessary for the transformation of follicular granulosa cells into granulosa lutein cells. Furthermore, as oestrogen is essential to the viability of follicular cells. Younis et al. (2024) found that hypothyroid rats had lower oestrogen levels, which was thought to be the cause of the rise in atretic follicles and the decrease in developing follicles. Moreover, Ajayi et al. (2013) found that hypothyroidism interfered with the pituitary-ovarian axis's normal operation, affecting follicular maturation and leading to the production of cysts. Brown et al. (2024) observed that rats in the hypothyroid group had significant congestion of medullary blood vessels, which was explained by ovarian hyperactivity in response to hormonal changes that occur in hypothyroidism. These findings are consistent with our findings. According to van den Van Den Boogaard et al. (2011), hypothyroidism affects the reproductive axis and is linked to decreased ovarian function and female fertility. Furthermore, Jiang (2001) discovered that anovulation and follicular growth and development defects are brought on by thyroid hypofunction. Furthermore, Priya et al. (2015) discovered that ovulatory dysfunction brought on by thyroid abnormalities is linked to endocrine reproductive problems. Furthermore, according to Seifi et al. (2022), thyroid hormones function by attaching to an ovarian receptor and stimulating follicles as a result. As a result, hypothyroidism may have a direct impact on ovarian function. Additionally, Hasegawa et al. (2022) found that thyroid hormone-expressing cell surface receptors on oocytes influence follicle-stimulating hormone and luteinizing hormone functions through steroid production. The ovarian portions in the hypothyroid metformin groups showed a cortical and medulla formation. Multiple phases of ovarian follicles were seen in the cortex, including the primordial, primary, corpus luteum, and Graafian follicles, which contained secondary oocytes surrounded by zona pellucida and corona radiata and linked to the follicle wall by vacuolated cumulus oophorus. Vacuolated granulosa cells, theca externa cells, and theca interna cells encircled the antrum of the Graafian follicle. There were some clogged blood arteries in the medulla. These results corroborate the findings of Priya et al. (2015), who found that treating hypothyroidism resolves ovulatory dysfunctions associated with hypothyroidism. Metformin treatment was found to mitigate ovarian abnormalities associated with hypothyroidism. The anti-inflammatory and antioxidant properties of metformin, along with improvements in body metabolism and hormonal balance, help to improve ovarian structure and function, which are negatively impacted by hypothyroidism. ConclusionThe anti-inflammatory and antioxidant properties of metformin, along with improvements in body metabolism and hormonal balance, help to improve ovarian structure and function, which are negatively impacted by hypothyroidism. AcknowledgmentsNot applicable. Conflict of interestThe authors have no competing interests to declare that are relevant to the content of this article. FundingNo funds, grants, or other support was received. Authors' contributionsThe study was conceptualized and designed by all authors, with the experiment being supervised by N.A.S., A.A.S., and the rest of the authors carried out the study's practical components, conducted data analysis, wrote and revised the manuscript. All authors read and appropriate the manuscript. Data availabilityAll datasets generated during this study are included in the article. ReferencesAbulfadle, K.A. and Ahmed, N.A. 2019. Effect of exercise training and adropin treatment on thyroid and testicular function changes in obese type 2 diabetic rats. Am. J. Biomed. Sci. 11, 209–225. Agarwal, A., Aponte-mellado, A., Premkumar, B.J., Shaman, A. and Gupta, S. 2012. The effects of oxidative stress on female reproduction: a review. Reprod. biol. Endocrinol. 10, 1–31. Agarwal, A., Gupta, S., Sekhon, L. and Shah, R. 2008. Redox considerations in female reproductive function and assisted reproduction: from molecular mechanisms to health implications. A. R. S. 10, 1375–1404. Ajayi, A., Akhigbe, R. and Ajayi, L. 2013. Hypothalamic-pituitary-ovarian axis in thyroid dysfunction. West Indian Med. J. 62, 835–838. AL-Saaidi, J.A. and AL-Bedary, J.K. 2022. Gonadotropin profile in experimentally induced hypothyroid and hyperthyroid cyclic female rats. Iraqi J. Vet. 36, 745–751. American Diabetes Association Professional Practice Committee (ADAPPC). 2022. 9. pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2022. Diabetes Care. 45 (Suppl 1): S125–S143. Amirabadizadeh, A., Amouzegar, A., Mehran, L. and Azizi, F. 2023. The effect of metformin therapy on serum thyrotropin and free thyroxine concentrations in patients with type 2 diabetes: a meta-analysis. Sci. Rep. 13, 18757. Atasayar, S. and Guler Demir, S. 2019. Determination of the Problems Experienced by Patients Post-Thyroidectomy. Clin. Nurs. Res. 28(5), 615–635. Blonde, L., Dailey, G.E., Jabbour, S.A., Reasner, C.A. and Mills, D.J. 2004. Gastrointestinal tolerability of extended-release metformin tablets compared to immediate-release metformin tablets: results of a retrospective cohort study. Curr. Med. Res. Opin. 20, 565–572. Brent. 2012. Mechanisms of thyroid hormone action. J. Clin. Invest. 122, 3035–3043. Brown, E.D.L., Obeng-Gyasi, B., Hall, J.E. and Shekhar, S. 2023. The thyroid hormone axis and female reproduction. Int. J. Mol. Sci. 24(12), 9815. Burger, H.G. and Patel, Y.C. 1977. Thyrotropin releasing hormone - TSH Clinic. Endocrinol. Metab. 6, 831–800. Choksi, N.Y., Jahnke, G.D., St. Hilaire, C. and Shelby, M. 2003. Role of thyroid hormones in human and laboratory animal reproductive health. Birth. Defects. Res. Part. B. Develop. Reproductive. Toxicol. 68, 479–491. Col, N.F., Ochs, L., Springmann, V., Aragaki, A.K. and Chlebowski, R.T. 2012. Metformin and breast cancer risk: a meta-analysis and critical literature review. Breast. Cancer. Res. Treatment. 135, 639–646. Cooper. 2005. Antithyroid Drugs. N. Engl. J. Med. 352, 905–917. Cosentino, F., Grant, P.J., Aboyans, V., Bailey, C.J., Ceriello, A., Delgado, V., Federici, M., Filippatos, G., Grobbee, D.E., Hansen, T.B., Huikuri, H.V., Johansson, I., Jüni, P., Lettino, M., Marx, N., Mellbin, L.G., Östgren, C.J., Rocca, B., Roffi, M., Sattar, N., Seferović, P.M., Sousa-Uva, M., Valensi, P. and Wheeler, D.C. 2020. "2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD". Eur .Heart J . 41 (2): 255–323. De Medeiros, S.F., De Medeiros, M.A.S., Ormond, C.M., Barbosa, J.S. and Yamamoto, M.M.W. 2018. Subclinical Hypothyroidism Impact on the Characteristics of Patients with Polycystic Ovary Syndrome. A Meta-Analysis of Observational Studies. Gynecol. Obstet. Investig. 83, 105–115. Dehkordi, A.H., Abbaszadeh, A., Mir, S. and Hasanvand, A. 2018. Metformin and its anti-inflammatory and anti-oxidative effects; new concepts. J. R. I. P. 8, 54–61. El-Tantawi, H. and Abozeid, F. 2019. Impact of Spirulina on Propylthiouracil-Induced Hypothyroidism in Albino Rats, a histological, immunohistochemical and biochemical approach. E. J. H. 42, 849–860. Ezzamouri, B., Rosario, D., Bidkhori, G., Lee, S., Uhlen, M. and Shoaie, S. 2023. Metabolic modelling of the human gut microbiome in type 2 diabetes patients in response to metformin treatment. NPJ. Syst. Biol. Applications. 9(1), 2. Fatima, M., Amjad, S., Sharaf Ali, H., Ahmed, T., Khan, S., Raza, M. and Inam, M. 2020. Correlation of Subclinical Hypothyroidism With Polycystic Ovary Syndrome (PCOS). Cureus 12, 8142. Fernando, B., Marley, R., Holt, S., Anand, R., Harry, D., Sanderson, P., Smith, R., Hamilton, G. and Moore, K., 1998. Figueroa, B., Velez, H. and Irizarry-Ramirez, M. 2008. Association of thyroid-stimulating hormone levels and body mass index in overweight Hispanics in Puerto Rico. Ethn. Dis. 18, 151–154. Fontana, L., Klein, S., Holloszy, J.O. and Premachandra, B.N. 2006. Effect of long-term calorie restriction with adequate protein and micronutrients on thyroid hormones. J Clin Endocrinol Metab. 91, 3232–3235. Han, P., Zhou, J., Xiang, J., Liu, Q. and Sun, K. 2023. Research progress on the therapeutic effect and mechanism of metformin for lung cancer (Review). Oncol. Rep. 49(1), 3. Hapon, M. B., Gamarra-Luques, C. and Jahn G. A. 2010. Short term hypothyroidism affects ovarian function in the cycling rat. R B &E, 8, 14. Hall, A., Rog, T. and Vattulainen, I. 2011. Effect of galactosylceramide on the dynamics of cholesterol-rich lipid membranes. J Phys Chem B. 115, 14424–14434. Hasegawa, Y., Kitahara, Y., Osuka, S., Tsukui, Y., Kobayashi, M. and Iwase, A. 2022. Effect of hypothyroidism and thyroid autoimmunity on the ovarian reserve: a systematic review and meta-analysis. Reprod. Med. Biol. 21, e12427. Hayat, N.Q., Nadir, S. and MuneeraUNEERA, M.J. 2016. The effect of hypothyroidism on the body weight of adult albino Wistar rats. J. R. M. C. 20, 20. Isidro, M.L., Penín, M.A., Nemiña, R. and Cordido, F. 2007. Metformin reduces thyrotropin levels in obese, diabetic women with primary hypothyroidism on thyroxine replacement therapy. Endocr. J. 32, 79–82. Ittermann, T., Markus, M.R., Schipf, S., Derwahl, M., Meisinger, C. and Volzke, H. 2013. Metformin inhibits goitrogenous effects of type 2 diabetes. Eur. J. Endocrinol. 169, 9–15. Jensterle, M., Kravos, N.A., Ferjan, S., Goricar, K., Dolzan, V. and Janez, A. 2020. Long-term efficacy of metformin in overweight-obese PCOS: longitudinal follow-up of retrospective cohort. Endocr. Connect. 9(1), 44–54. Jha, J.C., HO, F., Dan, C. and Jandeleit-Dahm, K. 2018. A causal link between oxidative stress and inflammation in cardiovascular and renal complications of diabetes. J. Clin. Sci. 132, 1811–1836. Jiang, J. 2001. Characteristics of infertility in female hypothyroid (hyt) mice. Rev. Reprod. 122, 695–700. Jin, P., Jiang, J., Zhou, L., Huang, Z., Qin, S., Chen, H.N., Peng, L., Zhang, Z., Li, B., Luo, M., Zhang, T., Ming, H., Ding, N., Li, L., Xie, N., Gao, W., Zhang, W., Nice, E.C., Wei, Y. and Huang, C. 2022. Disrupting metformin adaptation of liver cancer cells by targeting the TOMM34/ATP5B axis. EMBO. Mol. Med. 14(12), e16082. Kabodmehri, R., Sharami, S.H., Sorouri, Z.Z., Gashti, N.G., Milani, F., Chaypaz, Z. and Ghalandari, M. 2021. The relationship between thyroid function and ovarian reserve: a prospective cross-sectional study. Thyroid. Res. 14, 22. Khair Ns, T.S. 2014. Effect of thyroid disorders on the adult female albino rats (Histological and Histochemical Study). J. Cytology. &. Histology. 5(1), 1. Khodabandeh, S., Hosseini, A., Khazali, H. and Azizi, V. 2022. Interplay between polycystic ovary syndrome and hypothyroidism on serum testosterone, oxidative stress and StAR gene expression in female rats. Endocrinol. Diabetes Metab. J. 5, e359. Knudsen, N., Laurberg, P., Rasmussen, L.B., Bulow, I., Perrild, H., Ovesen, L. and Jørgensen, T. 2005. Small differences in thyroid function may be important for body mass index and the occurrence of obesity in the population. J. Clin. Endocrinol. Metab. 90, 4019–4024. Krysiak, R. and Okopien, B. 2011. Thyrotropin-lowering effect of metformin in a patient with resistance to thyroid hormone. J. Clin. Endocr. 75, 404–406. Krysiak, R., Gilowska, M., Szkróbka, W. and Okopień, B. 2016. The effect of metformin on the hypothalamic-pituitary-thyroid axis in patients with type 2 diabetes and amiodarone-induced hypothyroidism. Pharmacol. Rep. 68(2), 490–494. Krysiak, R., Kowalcze, K. and Okopień, B. 2024. Impact of metformin on hypothalamic–pituitary–thyroid axis activity in women with autoimmune and non autoimmune subclinical hypothyroidism: a pilot study. Pharmacol. Rep. 76, 195–206. Kumar, V., Kumar, V., Abbas, A.K. and Aster, J.C. 2015. Robbins and cotran pathologic basis of disease. 9th Ed. Philadelphia: Elsevier. Lan, W.H., Lin, T.Y., Yeh, J.A., Feng, C.L., Hsu, J.T., Lin, H.J., Kuo, C.J. and Lai, C.H. 2022. Mechanism underlying metformin action and its potential to reduce gastric cancer risk. Int. J. Mol. Sci. 23(22), 14163. Lascu, A., Ionică, L.N., Merce, A.P., Dănilă, M.D., Petrescu, L., Sturza, A., Muntean, D.M. and Streian, C.G. 2022. Metformin acutely mitigates oxidative stress in human atrial tissue: a pilot study in overweight non-diabetic cardiac patients. Life. (Basel). 12(12), 2058. Lopez, M., Varela, L., Vazquez, M.J., Rodriguez-Cuenca, S., Gonzalez, C.R., Velagapudi, V.R., Morgan, D.A., Schoenmakers, E., Agassandian, K. and Lage, R. 2010. Hypothalamic AMPK and fatty acid metabolism mediate thyroid regulation of energy balance. J. Nat. Med. 16, 1001–1008. Maruthur, N.M., Tseng, E., Hutfless, S., Wilson, L.M., Suarez-Cuervo, C., Berger, Z., Chu, Y., Iyoha, E. and Segal, J.B. 2016. Bolen S. Diabetes medications as monotherapy or metformin-based combination therapy for type 2 diabetes: a systematic review and meta-analysis. Ann. Intern. Med. 164, 740–751. Mcdermott, M.T. 2012. Does combination T4 and T3 therapy make sense?. Endocr. J. 18, 750–757. Michalaki, M.A., Vagenakis, A.G., Leonardou, A.S., Argentou, M.N., Habeos, I.G., Makri, M.G., Psyrogiannis, A.I., Kalfarentzos, F.E. and Kyriazopoulou, V.E. 2006. Thyroid function in humans with morbid obesity. J. Thyroid Res. 16, 73–78. Morale, M.G., Tamura, R.E. and Rubio, I.G.S. 2022. Metformin and cancer hallmarks: molecular mechanisms in thyroid, prostate and head and neck cancer models. Biomolecules 12(3), 100–105. Müller, P.E., Jakoby, R., Heinert, G. and Spelsberg, F. 2001. Surgery for recurrent goitre: its complications and their risk factors. Eur. J. Surg.=. Acta. Chirurgica. 167(11), 816–821. Nelson, E.R., Allan, E.R.O., Pang, F.Y. and Habibi, H.R. 2011. Auto-regulation of thyroid hormone receptors in the goldfish ovary and testis. Gen. Comp. Endocrinol. 172, 50–55. Nojima, I. and Wada, J. 2023. Metformin and its immune-mediated effects in various diseases. Int. J. Mol. Sci. 24(1), 755. Noto, H., Goto, A., Tsujimoto, T. and Noda, M. 2012. Cancer risk in diabetic patients treated with metformin: a systematic review and meta-analysis. PLos One 7(3), e33411. Nyrnes, A., Jorde , R. & Sundsfjord , J. 2006. Serum TSH is positively associated with BMI. 1. Oleandri, S.E., Maccario, M., Rossetto, R., Procopio, M., Grottoli, S., Avogadri, E., Gauna, C., Ganzaroli, C. and Ghigo, E. 1999. Three-month treatment with metformin or dexfenfluramine does not modify the effects of diet on anthropometric and endocrine-metabolic parameters in abdominal obesity. J. Endocrinol. Invest. 22, 134–140. Panciera, D., Purswell, B., Kolster, K., Werre, S. and Trout, S. 2012. Reproductive effects of prolonged experimentally induced hypothyroidism in bitches. Jvim 26, 326–333. Persani, L., Rodin, C., Moran, W.E., Visser, W.S., Groeneweg, S., Peeters, S., Refetoff, S. and Gurnell, M. 2024. European Thyroid Association Guidelines on diagnosis and management of genetic disorders of thyroid hormone transport, metabolism and action. ETJ 13(4), 1–62. Priya, D.M., Akhtar, N. and Ahmad, J. 2015. Prevalence of hypothyroidism in infertile women and evaluation of response of treatment for hypothyroidism on infertility. Indian J. Endocrinol. Metab. 19, 504. Robert, A., Vigersky, A. and FilmoreI, N. 2006. Thyrotropin suppression by metformin. J. Clin. Endocrinol. Metab. 91, 225–227. Roos, A., Bakker, S.J., Links, T.P., Gans, R.O. and Wolffenbuttel, B.H. 2007. Thyroid function is associated with components of the metabolic syndrome in euthyroid subjects. J. Clin. Endocrinol. Metab. 92, 491–496. Sanyal, D. and Raychaudhuri, M. 2016. Hypothyroidism and obesity: an intriguing link. Indian J. Endocrinol. Metab. 20, 554–567. Seifi, A., Taheri, N., Kia, H., Mansourian, H.R. and Mansourian, A.R. 2022. Adverse Effects of Hypothyroidism on Fertility and Pregnancy: a Mini Review. Med. Lab. J. 16, 1–9. Sekino, N., Kano, M., Kobayashi, S., Murakami, K., Sakata, H., Toyozumi, T., Endo, S., Matsumoto, Y., Suito, H., Takahashi, M., Otsuka, R., Yokoyama, M., Shiraishi, T., Okada, K., Kamata, T., Ryuzaki, T., Hirasawa, S., Kinoshita, K., Sasaki, T., Iida, K., Komatsu, A. and Matsubara, H. 2022. Metformin-induced heat shock protein family a member 6 is a promising biomarker of esophageal squamous cell carcinoma. Oncology 100(5), 267–277. Shekhar, S., Hall, J.E. and Klubo-Gwiezdzinska, J. 2020. The Hypothalamic Pituitary Thyroid Axis and Sleep. Curr. Opin. Endocr. Metab. Res. 17(8), 8–14. Shokoohi, M., Soltani, M., Abtahi -Eivary, Seyed-Hosein, Niazi, V., Rafeei, P.M., Mohammed, J., Ravaei, H., Salimnejad, R., Moghimian, M. and Shoorei, H. 2019. Effect of hydro--alcoholic extract of Olea europaea on apoptosis--related genes and oxidative stress in a rat model of torsion/detorsion--induced ovarian damage. Asian Pac. J. Reprod. 8, 148–156. Silva, J.F., Ocarino, N.M. and Serakides, R. 2018. Thyroid hormones and female reproduction. Biol. Reprod. 99, 907–921. Strikić Đula, I., Pleić, N., Babić Leko, M., Gunjača, I., Torlak, V., Brdar, D., Punda, A., Polašek, O., Hayward, C. and Zemunik, T. 2022. Epidemiology of Hypothyroidism, Hyperthyroidism and Positive Thyroid Antibodies in the Croatian Population. Biology 11, 394. Suvarna, K.S., Layton, C. and Bancroft, J.D. 2018. Bancroft’s Theory and Practice of Histological Techniques E-Book; Elsevier Health Sciences. Amsterdam, The Netherlands: Elsevier Health Sciences. Temple, R.C., Clark, P.M. and Hales, C.N. 1992. Measurement of insulin secretion in type 2 diabetes: problems and pitfalls. J. Diabetic. 9, 503–512. Tietz N.W. 1995. Clinical Guide to Laboratory Tests. 3rd Ed. W.B. Saunders Company, Phila, 509–580. Treesh, S. and Khair, N. 2014. Effect of thyroid disorders on the adult female albino rats (Histological and Histochemical Study). J. Cytol. Histol. 5, 1-9. Van Den Boogaard, E., Vissenberg, R., Land, J.A., Van Wely, M., Van Der Post, J.A.M., Goddijn, M. and Bisschop, P.H. 2011. Significance of (sub)clinical thyroid dysfunction and thyroid autoimmunity before conception and in early pregnancy: a systematic review. Hum. Reprod. Update. 17, 605–619. Vargas-Uricoechea, H. and Bonelo-Perdomo, A. 2017. Thyroid Dysfunction and Heart Failure: mechanisms and Associations. Curr. Heart Fail. Rep. 14, 48–58. Villar, H.C., Saconato, H., Valente, O. and Atallah A.N. 2007. Thyroid hormone replacement for subclinical hypothyroidism. Cochrane Database Syst. Rev. 2007, CD003419. Vissenberg, R., Manders, V.D., Mastenbroek, S., Fliers, E., Afink, G.B., Ris-Stalpers, C., Goddijn, M. and Bisschop, P.H. 2015. Pathophysiological aspects of hyroid hormone disorders/thyroid peroxidase autoantibodies and reproduction. Hum. Reprod. Update 21, 378–387. Verma, S., Bhanot, S. and Mcneill, J.H. 1994. Antihypertensive effects of metformin in fructose-fed hyperinsulinemic, hypertensive rats. J. Pharmacol. Exp. Ther. 271, 1334–1337. Vissenberg, R., Manders, V.D., Mastenbroek, S., Fliers, E., Afink, G.B., Ris-Stalpers, C., Goddijn, M. and Bisschop, P.H. 2015. Pathophysiological aspects of thyroid hormone disorders/thyroid peroxidase autoantibodies and reproduction. Hum. Reprod. Update 21, 378–387. Wang, N.F., Jue, T.R., Holst, J. and Gunter, J.H. 2022. Systematic review of antitumour efficacy and mechanism of metformin activity in prostate cancer models. BJUI. Compass. 4(1), 44–58. Yerevanian, A. and Soukas, A.A. 2019. Metformin: mechanisms in human obesity and weight loss. Curr. Obes. Rep. 8, 156–164. Younis A.H, Hilal A.R., Rajesh K., Muddasir B. and Mohd S.R.A. 2024. Review on critical appraisal and pathogenesis of polycystic ovarian syndrome. Endocrinol. Metab. (EnM). 14(31), M 100162e.–M 1001-4-A. Zhang, C., Wang, X., Wang, Z., Niu, W., Zhu, B. and Xia, G. 2013. Effect of different culture systems and 3, 5, 3'-triiodothyronine/follicle-stimulating hormone on preantral follicle development in mice. PLos One 8, e61947. Zhao, H., Xing, C., Zhang, J. and He, B. 2021. Comparative efficacy of oral insulin sensitizers metformin, thiazolidinediones, inositol, and berberine in improving endocrine and metabolic profiles in women with PCOS: a network meta-analysis. Reprod. Health. 18, 1–12. | ||
| How to Cite this Article |
| Pubmed Style Soliman NA, Shalaby AA, Abulfadle KA, Mohammed H, Hussien S, Ammar MA, Metwally AS, Bahaie EEL. Effect of metformin treatment on ovarian function in hypothyroid rats. Open Vet. J.. 2026; 16(1): 401-418. doi:10.5455/OVJ.2026.v16.i1.37 Web Style Soliman NA, Shalaby AA, Abulfadle KA, Mohammed H, Hussien S, Ammar MA, Metwally AS, Bahaie EEL. Effect of metformin treatment on ovarian function in hypothyroid rats. https://www.openveterinaryjournal.com/?mno=226759 [Access: January 31, 2026]. doi:10.5455/OVJ.2026.v16.i1.37 AMA (American Medical Association) Style Soliman NA, Shalaby AA, Abulfadle KA, Mohammed H, Hussien S, Ammar MA, Metwally AS, Bahaie EEL. Effect of metformin treatment on ovarian function in hypothyroid rats. Open Vet. J.. 2026; 16(1): 401-418. doi:10.5455/OVJ.2026.v16.i1.37 Vancouver/ICMJE Style Soliman NA, Shalaby AA, Abulfadle KA, Mohammed H, Hussien S, Ammar MA, Metwally AS, Bahaie EEL. Effect of metformin treatment on ovarian function in hypothyroid rats. Open Vet. J.. (2026), [cited January 31, 2026]; 16(1): 401-418. doi:10.5455/OVJ.2026.v16.i1.37 Harvard Style Soliman, N. A., Shalaby, . A. A., Abulfadle, . K. A., Mohammed, . H., Hussien, . S., Ammar, . M. A., Metwally, . A. S. & Bahaie, . E. E. L. (2026) Effect of metformin treatment on ovarian function in hypothyroid rats. Open Vet. J., 16 (1), 401-418. doi:10.5455/OVJ.2026.v16.i1.37 Turabian Style Soliman, Nabil Abbas, Amr A. Shalaby, Khaled A. Abulfadle, Heba Mohammed, Samia Hussien, Mohamed Ahmed Ammar, Aya Sh. Metwally, and Eman E. L. Bahaie. 2026. Effect of metformin treatment on ovarian function in hypothyroid rats. Open Veterinary Journal, 16 (1), 401-418. doi:10.5455/OVJ.2026.v16.i1.37 Chicago Style Soliman, Nabil Abbas, Amr A. Shalaby, Khaled A. Abulfadle, Heba Mohammed, Samia Hussien, Mohamed Ahmed Ammar, Aya Sh. Metwally, and Eman E. L. Bahaie. "Effect of metformin treatment on ovarian function in hypothyroid rats." Open Veterinary Journal 16 (2026), 401-418. doi:10.5455/OVJ.2026.v16.i1.37 MLA (The Modern Language Association) Style Soliman, Nabil Abbas, Amr A. Shalaby, Khaled A. Abulfadle, Heba Mohammed, Samia Hussien, Mohamed Ahmed Ammar, Aya Sh. Metwally, and Eman E. L. Bahaie. "Effect of metformin treatment on ovarian function in hypothyroid rats." Open Veterinary Journal 16.1 (2026), 401-418. Print. doi:10.5455/OVJ.2026.v16.i1.37 APA (American Psychological Association) Style Soliman, N. A., Shalaby, . A. A., Abulfadle, . K. A., Mohammed, . H., Hussien, . S., Ammar, . M. A., Metwally, . A. S. & Bahaie, . E. E. L. (2026) Effect of metformin treatment on ovarian function in hypothyroid rats. Open Veterinary Journal, 16 (1), 401-418. doi:10.5455/OVJ.2026.v16.i1.37 |