| Research Article | ||
Open Vet. J.. 2025; 15(12): 6581-6601 Open Veterinary Journal, (2025), Vol. 15(12): 6581-6601 Research Article Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021)John Olusoji Abiola1, Oladipo Olufemi Omotosho1*, Emmanuel Ayopo Aderounmu2, Victor Ajibola Ogunlola1, Ridwan Olamilekan Adesola1, Abdulafees Hamzat1, Damilare Johnson Adebayo1, Mayowa Ayorinde1, Olalekan Taiwo Jeremiah1 and Temidayo Olutayo Omobowale11Department of Veterinary Medicine, Faculty of Veterinary Medicine, University of Ibadan, Ibadan, Nigeria 2Department of Theriogenology, Faculty of Veterinary Medicine, University of Ibadan, Ibadan, Nigeria *Corresponding Author: Oladipo Olufemi Omotosho. Department of Veterinary Medicine, Faculty of Veterinary Medicine, University of Ibadan, Ibadan, Nigeria. Email: oo.omotosho [at] gmail.com Submitted: 05/07/2025 Revised: 25/10/2025 Accepted: 07/11/2025 Published: 31/12/2025 © 2025 Open Veterinary Journal
AbstractBackground: African swine fever (ASF), a highly contagious, hemorrhagic viral disease of pigs, continues to decimate the global pig population. Aim: This study was designed to investigate key field events, morbidity patterns, mortality rate, clinical findings in pigs, and biosecurity breaches on farms during the ASF outbreak in Southwest Nigeria (2019–2021). Methods: Farms (n=62) accommodating 6,208 pigs in Oyo, Ogun, and Lagos states were investigated. Information on the demographic characteristics of farmers, their knowledge of and experience with ASF, the sequence of morbidity onset across age groups, and the rate of farm size reduction was obtained using structured questionnaires. Clinical findings in diseased pigs and biosecurity breaches related to farm infrastructures and management practices were observed and assessed using a modified standard template. Data were analyzed using descriptive statistics and chi-square at p < 0.05. Results: Most of the farmers were between 40 and 60 years old and were predominantly men. A significant proportion had over 5 years of experience, and most had a pre-outbreak herd of 200 pigs or less. Many farmers had experienced previous ASF outbreaks leading to losses. Pig age was associated with the sequence of morbidity onset, with the most frequent sequence being adults → piglets → weaners → growers. Mortality, farm size reduction, and financial loss were 60.6%, 92.1%, and ₦166,264,000, respectively. Clinical findings were consistent with hemorrhagic fever in pigs. Biosecurity breaches include a lack of quarantine facilities, limited use of fencing, gating, foot dips, and netting, pig farm aggregation, continuous pig flow on farms, poor vehicle disinfection, uncertified replacement stock, open waste dumping, uncontrolled visitor access, and unrestricted pig movement. Conclusion: Biosecurity breaches across farm infrastructure, management practices, and human behavior aided the spread of ASF in Nigeria, leading to significant economic losses. Strengthening biosecurity, regulating pig movement and market activities, educating stakeholders, and ensuring government led surveillance and compensation are critical to controlling ASF and safeguarding Nigeria’s pig industry. Keywords: African swine fever, Mass mortality; Panic sales, Pig disease, Biosecurity breaches. IntroductionPig production continues to serve as a crucial source of animal protein and income generation for many sub-Saharan African countries (Janse van Rensburg et al., 2022), with Nigeria ranking among the continent’s foremost pig producers (Robinson et al., 2014, Adesehinwa et al., 2024). Between 1984 and 1997, Nigeria experienced rapid growth in its pig population, expanding from approximately 2 million to over 7 million pigs. However, this upward trend was disrupted by the first officially confirmed outbreak of African swine fever (ASF) in 1997 (Odemuyiwa et al., 2000; Fasina et al., 2010). Subsequent outbreaks caused extensive losses in pig populations with lasting social and economic consequences (Otesile et al., 2005; Babalobi et al., 2007; Igbokwe et al., 2018; Omotosho, 2020). The disease has since emerged as a major constraint to pig farming in Nigeria, significantly impeding industry growth and profitability (Ebwanga et al., 2021; Ogundijo et al., 2023). Recurring outbreaks have been documented in vital pig producing and marketing hubs, particularly in the southwestern, central, and eastern regions of the country (Fasina et al., 2010; Owolodun et al., 2010; Igbokwe et al., 2018). The disease also remains endemic across many sub-Saharan African nations, with recent incidents reported in West African countries, such as Togo, Benin, Senegal, Côte d Ivoire, The Gambia, Burkina Faso, Ghana, Guinea Bissau, Mali, Guinea, Sierra Leone, and Nigeria (Wadoum et al., 2020). ASF poses a significant threat to food security across the African continent and is associated with considerable economic damage (Fadiga et al., 2013; Gebreyohannes, 2015). The disease continues to expand its geographic reach, affecting pig populations in multiple regions globally, thereby endangering the sustainability of pig farming worldwide (Blome et al., 2020; De Lorenzi et al., 2020). This acute, hemorrhagic viral disease targets the Suidae family, impacting both domestic and wild pig populations. It was first identified in Kenya in 1909 following the introduction of European domestic pigs (Montgomery, 1921; Mulumba-Mfumu et al., 2019). Since then, the virus has spread extensively across domestic pig populations in sub–Saharan Africa and beyond (Sánchez-Cordón et al., 2018). ASF persisted in Africa until it was likely introduced from Angola to Portugal in 1957 (Boinas, 2011; Danzetta et al., 2020; Ito et al., 2023). The disease is caused by the African swine fever virus (ASFV), a large, enveloped double stranded DNA virus (Rock, 2021). ASFV belongs to the genus Asfivirus within the family Asfarviridae and is the only known DNA arbovirus. The soft tick Ornithodoros spp. serves as its natural reservoir and biological vector (Njau et al., 2021; Jori et al., 2023). Domestic pigs (Sus scrofa) are highly susceptible to ASFV infection; however, subclinical infections have been observed in certain African wild pig species, including bush pigs (Potamochoerus spp.), warthogs (Phacochoerus spp.), and giant forest hogs (Hylochoerus meinertzhageni), which act as natural reservoirs (Stahl et al., 2019; Fischer et al., 2020). Under field conditions, ASF has an incubation period ranging from 4 to 19 days (Mebus et al., 1983, USDA, 2023), and infected pigs begin shedding the virus before clinical signs appear. High viral titers are typically present in all bodily fluids, including saliva, nasal secretions, feces, urine, conjunctival discharge, genital secretions, and blood from wounds, making transmission highly efficient (Sánchez-Vizcaíno et al., 2019). Surviving pigs may become chronic carriers and should be carefully considered in disease eradication strategies (Sánchez-Vizcaíno et al., 2019). Clinical manifestations vary according to viral virulence, dose, and route of infection (Li et al., 2022). Because ASF presents several clinical signs, its diagnosis can be challenging. The signs and pathological lesions in pigs often resemble those of other hemorrhagic diseases, such as classical swine fever, erysipelas, and septicemia. Consequently, laboratory confirmation is essential for accurate identification. Polymerase chain reaction (PCR), particularly targeting regions such as VP72 in the ASFV genome, remains the gold standard due to its high sensitivity and specificity (Fernández-Pinero et al., 2013). Considering the virus’s high morbidity and mortality rates, the absence of an effective vaccine, and its complex genome and replication cycle (Gaudreault et al., 2020), stringent biosecurity measures are essential for preventing the spread of the virus. These encompass both external protocols to prevent the introduction of the virus and internal strategies to limit its spread within pig farms (ASF-STOP, 2021; Rodrigues et al., 2019). Epidemiological investigations revealed that all 24 known ASFV genotypes circulate in Africa (Achenbach et al., 2017; Quembo et al., 2017), although only genotypes I and II have been implicated in outbreaks outside the continent (Njau et al., 2021). In Nigeria, ASF outbreaks have historically been associated predominantly with Genotype I strains (Odemuyiwa et al., 2000; Otesile et al., 2005; Awosanya et al., 2021). However, recent molecular investigations have confirmed the emergence of Genotype II in pig carcasses during recent outbreaks (Adedeji et al., 2021; Onoja et al., 2022). Regional investigations have confirmed the co circulation of ASFV genotypes I and II in West Africa. Meki et al. (2025), reported the detection of both genotypes in domestic pig populations in Nigeria, Ghana, Benin, Burkina Faso, Côte d Ivoire, and Mali, underscoring the growing complexity of ASF epidemiology in the region. The spread of ASF in Nigeria has been linked to weak on farm biosecurity infrastructure (Olugasa and Ijagbone, 2007; Fasina et al., 2010) and lapses in human oversight throughout the pig value chain. The unregulated movement of pigs, buyers, workers, and equipment has been frequently identified as a critical risk factor (Olugasa and Ijagbone, 2007; Fasina et al., 2010). This study was conducted to investigate key field events, morbidity patterns, clinical findings in pigs, and biosecurity breaches on farms that facilitated the spread of ASF during the 2019–2021 outbreak in Southwest Nigeria. Materials and MethodsStudy design, sampling technique, and study locationThe study was designed as a cross-sectional investigation, conducted on purposively selected pig farms located across 14 local governments in Oyo, Ogun, and Lagos states, Southwest Nigeria (Fig. 1), during an outbreak of a disease characterized by hemorrhagic fever and increased pig mortality. These states were selected because they are important pig-producing areas in Nigeria, and early disease outbreak reports were made by farmers in these locations.
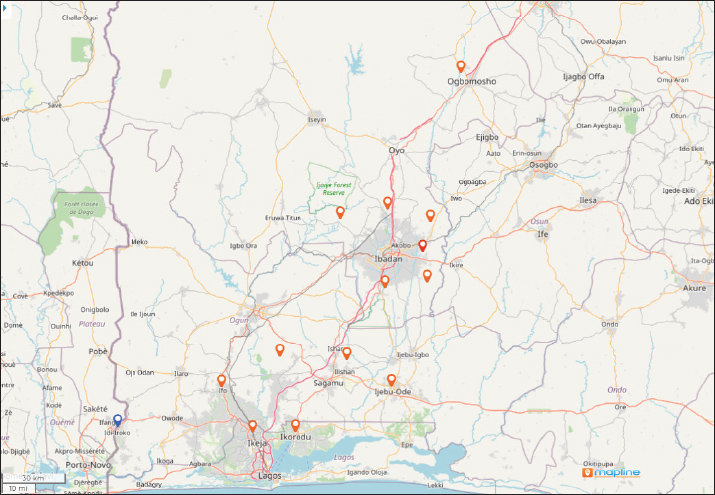
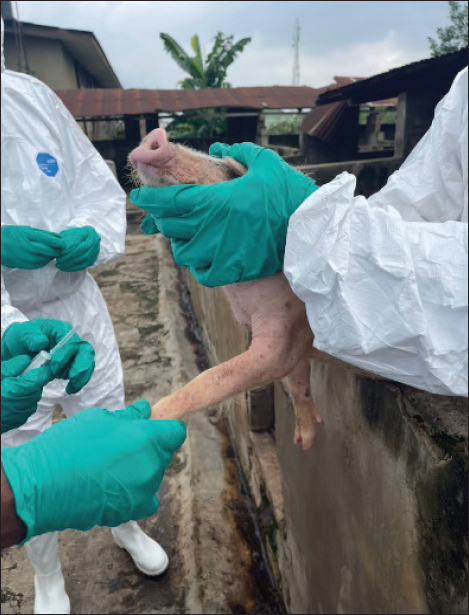
Fig. 1. A regional map of Southwest Nigeria displaying pin markers across 14 LGAs within Oyo, Ogun, and Lagos States. These pins represent the locations of 62 pig farms investigated for ASF between 2019 and 2021. The initial outbreak alert, designated by a blue pin, originated from the Ipokia LGA in Ogun State. To the west of this blue pin lies the international border with the Republic of Benin, along with its neighboring cities, beyond a clearly marked transecting line. Map created using Mapline (mapline.com). Disease outbreaks on pig farms (n=62) accommodating 6,208 pigs were investigated between December 2019 and March 2021 in the states of Oyo (n=14), Ogun (n=28), and Lagos (n=20). The farms were selected based on farmers’ reports of outbreaks to the Veterinary Teaching Hospital, University of Ibadan, directly or through receipt of outbreak information by two swine veterinarians affiliated to the hospital and other field veterinarians. Other farms were enlisted through the Veterinary Officers in the respective state ministry of agriculture of the three surveyed states. Only farms with acute morbidity and mortality were selected, visited, and investigated. Questionnaire and field observation guide on breaches of biosecurityA closed-ended questionnaire and field observation guide were designed to obtain information from farmers and guide field investigators, respectively. The questionnaires (n=62) that were administered to the farmers were used to obtain information on their demographic characteristics, farming practices, previous experience with ASF and on farm biosecurity infrastructures, and management practices. Information retrieved included farmers’ age, gender, religion, years of pig farming experience, and previous outbreak experience. The disease history, field observation, and continued interaction with enlisted farmers were used to document the disease spread pattern within herd, mortality level, rate of farm size reduction, and farmers’ responses to the outbreaks. During farm visits, investigators used the field observation guide to document key farm events and clinical signs in pigs affected by the outbreak. The date of disease onset and its geographic location were recorded for each farm, while the disease progression was monitored over a 30-day period from the initial onset. The investigation included cases reported between December 2019 and March 2021. The guide also enabled systematic documentation of biosecurity breaches, including infrastructure conditions and management practices, including pig movement patterns, and the handling or disposal of dead, sick, and exposed animals during the outbreak’s active phase. The questionnaires and observational templates were developed on the basis of a list of known risk factors for ASF transmission in West Africa (Fasina et al., 2012b) and adapted from the BioCheck risk-based weighted biosecurity scoring system available at www.biocheckgent.com. Emphasis was placed on subcategories related to external biosecurity (Backhans et al., 2015) to identify factors facilitating the entry of the ASF virus into farms and its subsequent spread within and to other farms and pig populations in Nigeria. Biosecurity measures taken by investigatorsTo prevent inter farm transmission of the disease, investigators adhered to strict biosecurity protocols during fieldwork. Disposable Personal Protective Equipment (PPE) was used throughout all farm visits (Fig. 2). Four investigator teams were deployed, with each team visiting one farm per day and conducting disinfection protocols both before and after each visit. Farm access was granted solely with the consent of the farmer and was limited to premises experiencing active disease problems. No team member made any visits to farms without reported outbreaks during the investigation period.
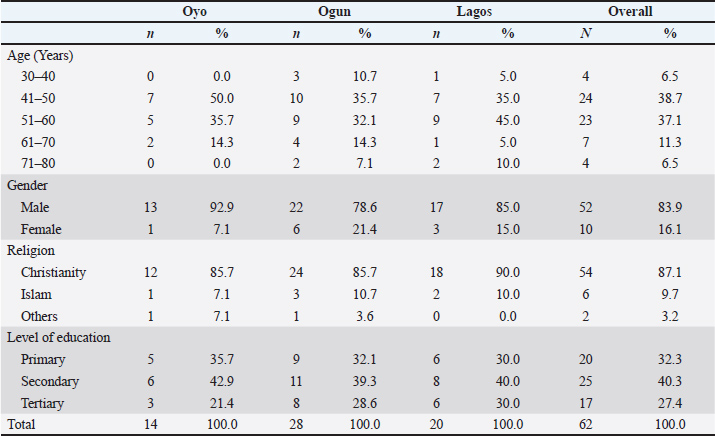
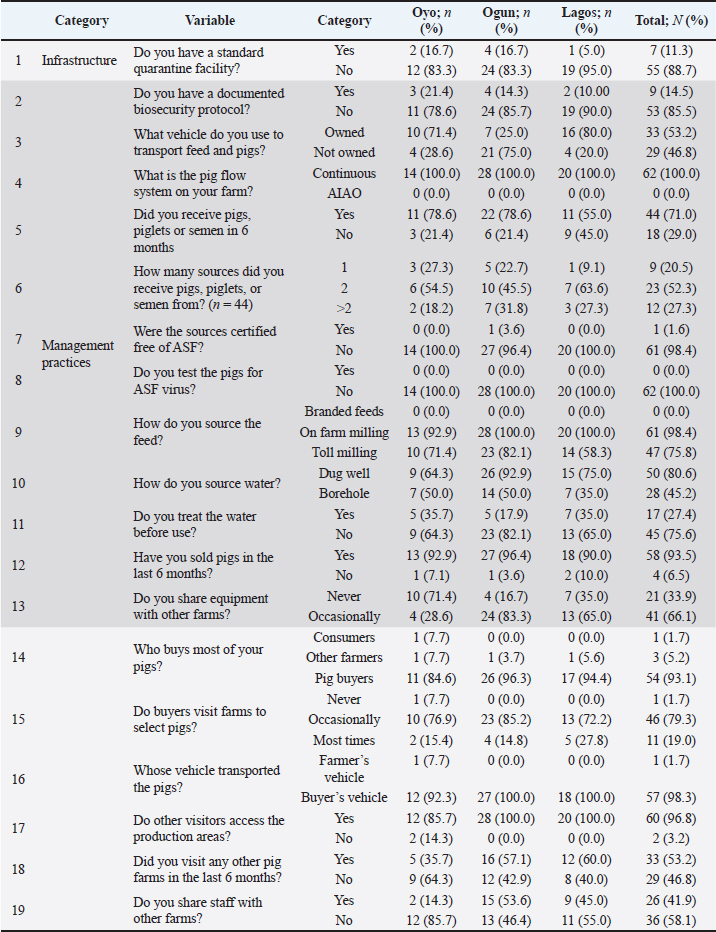
Fig. 2. Clinical evaluation and collection of blood samples from pigs by venipuncture during the ASF outbreak in Oyo state. Diagnosis of ASFOn each farm, we collected outbreak history data, including the timing of morbidity and mortality across various age groups, through interviews with farmers and farm records. Pigs were categorized into four age groups using farm records: piglets (pre-weaning), weaners (post-weaning to 12 weeks), growers (12 to 52 weeks), and adults (over 12 months). Clinical evaluations of affected pigs included assessments of rectal temperature, appetite, behavior, gait, and orifices. The skin was examined for lesions such as discoloration and hemorrhage. Group-housed pigs were further observed for huddling, shivering, respiratory distress, and other clinical indicators. Representative blood samples were collected via venipuncture for confirmatory diagnosis, and spleen tissue were obtained at postmortem. Labeled samples were submitted to the Molecular Virology Unit, Department of Virology, College of Medicine, University of Ibadan for the detection of ASFV using a p72 gene-targeted PCR assay (Onoja et al., 2022). Statistical analysisDescriptive statistics were used to analyze data to summarize outbreak patterns. The association between age categories and morbidity onset was assessed using the chi-square (χ²) test, with statistical significance set at p < 0.05. Ethical approvalInstitutional ethical approval for this study was obtained from the Animal Care and Use Research Ethics Committee of the University of Ibadan (Approval No. UI-ACUREC/099-0624/27). Informed consent was obtained from all participating farmers before the start of field investigations. ResultsDemographics and previous experience with ASFMost farmers (75.8%) were aged between 40 and 60 years, with a predominantly male representation (83.9%), and 87.1% identified as Christians (Table 1). Most respondents (87.1%) had over 5 years of experience in pig farming, and a substantial proportion (90.3%) reported pre-outbreak herd sizes of 200 pigs or less. All pig houses were open-sided. Notably, 72.6% of farmers had previously encountered ASF outbreaks, which were frequently associated with significant pig losses (Table 2). Table 1. Demographic characteristics of farmers affected by the ASF outbreak.
Table 2. Field experience of farmers and production practices on ASF-affected farms.
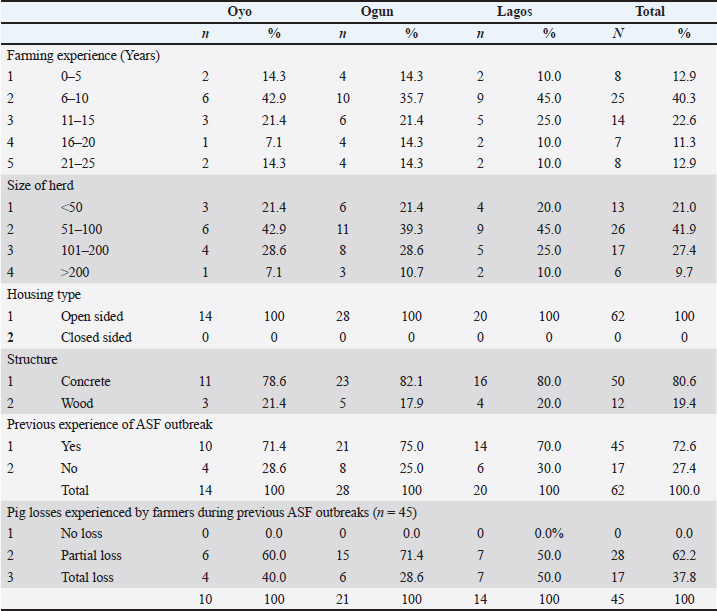
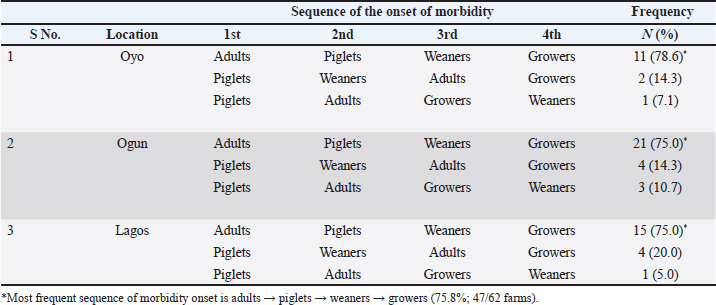
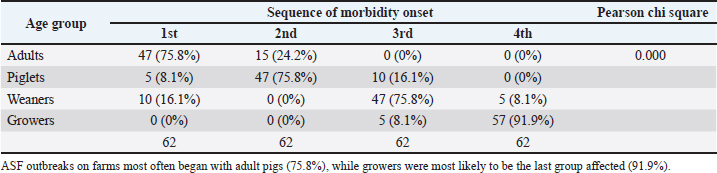
Morbidity patterns, mortality rate, and reduction in farm sizeRegarding morbidity patterns, the most reported sequence of onset across pig age groups was adults → piglets → weaners → growers (75.8%) (Table 3). A statistically significant association was observed between age and morbidity onset during ASF outbreaks (p=0.000). The likelihood of initial morbidity in adult pigs was 75.8% while growers were the most frequently the last group to exhibit clinical signs, with a probability of 91.9% (Table 4). Table 3. Sequence of onset of morbidity across age groups of pigs during ASF outbreak
Table 4. Association between pig age group and the onset of morbidity during the ASF outbreak in Southwest Nigeria (2019–2021).
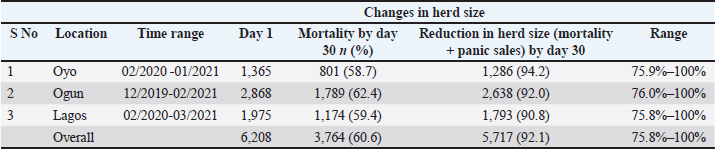
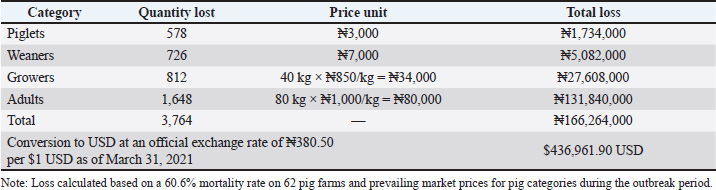
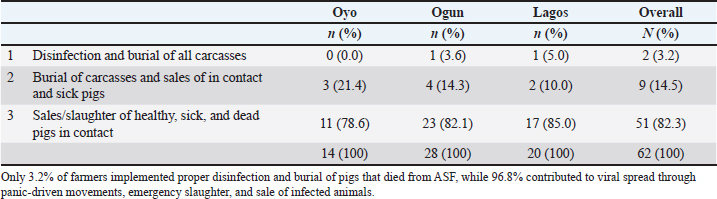
The overall mortality rate was 60.6%. Farm size reduction was driven by mortality, panic sales, emergency slaughter, and relocation of affected pigs, resulting in herd size declines ranging from 75.8% to 100% within the first 30 days of the outbreak (Table 5). The estimated financial loss was ₦166,264,000 ($436,961.90 USD) (Table 6). Most farmers (96.8%) contributed to viral spread by moving, slaughtering, and selling infected pigs to mitigate losses (Table 7 and Fig. 3).
Fig. 3. A: Mass mortality of pigs and their disposal by burial during the outbreak of ASF in Ogun state. B: Panic sales and transportation of apparently healthy pigs in Ogun state. Table 5. Mortality rate and reduction in herd size in the ASF outbreak in Oyo, Ogun, and Lagos, Southwest Nigeria (December 2019–March 2021).
Table 6. Financial loss due to ASF outbreak in Oyo, Ogun, and Lagos states, Southwest Nigeria (December 2019–March 2021).
Table 7. Farmers’ reaction and pattern of disposal of healthy, sick, and dead pigs during the ASF outbreak.
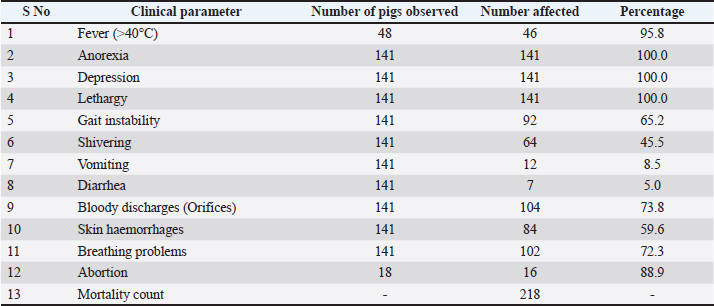
Clinical and epidemiological findingsClinical signs were consistently observed across the affected farms. Anorexia, depression, and lethargy were reported in 100% of cases, followed by fever in 95.8% of pigs. Other frequently noted signs included abortion in pregnant sows (88.9%), bloody discharges (73.8%), and gait instability (65.2%) (Table 8, Fig. 4). PCR targeting the VP72 gene of the ASF virus confirmed the presence of ASF on the affected farms.
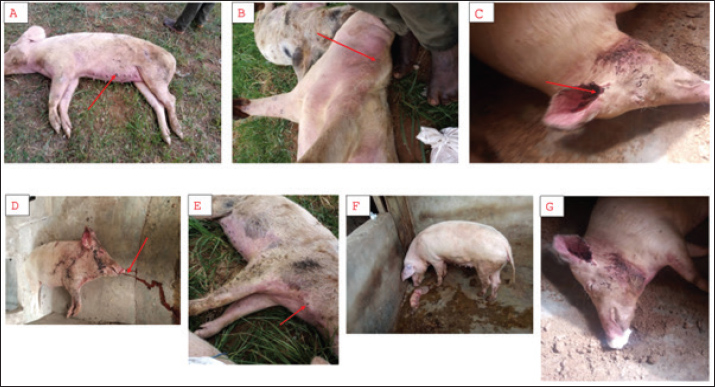
Fig. 4. Clinical signs of ASF in pigs in Southwest Nigeria. A: Cyanosis and purplish skin discoloration B: Hyperemia of the dermis. C and D: Hemorrhagic discharge from the nostrils and external ear canal. E: Petechial hemorrhages on the skin surface. F: Abortion in a pregnant sow, indicative of acute ASF. G: Acute respiratory distress with foamy oral exudate, culminating in death. Table 8. Clinical findings on some pigs affected by ASF in southwest Nigeria (2019–2021).
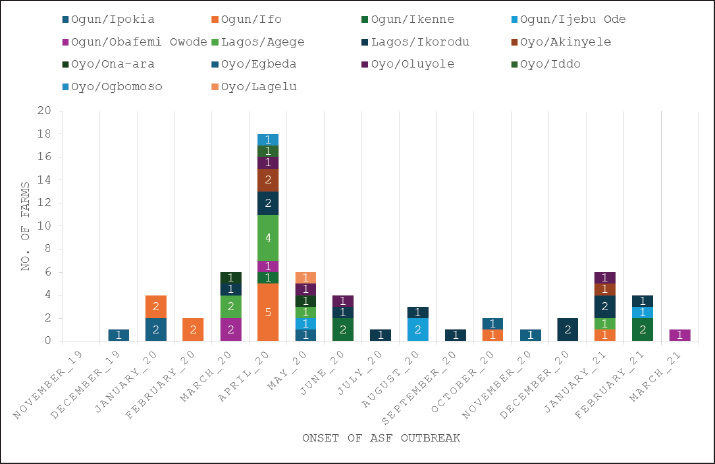
Epidemiological investigations across 62 pig farms (Figs. 1, 5, and 6) indicated that the index case emerged in December 2019 at a farm in Ipokia Local Government Area (LGA), Ogun State. This was followed by two confirmed cases within the same locality in January 2020. Later that same month, an additional outbreak was reported in Ifo LGA, located approximately 75 km from Ipokia. By March 2020, confirmed cases had extended to Agege LGA in Lagos State (approximately 74.5 km from Ipokia and 30 km from Ifo), as well as Ona-Ara LGA in Oyo State (approximately 211.5 km from Ipokia and 152 km from Ifo).
Fig. 5. Epidemiological curve illustrating the temporal spread of ASF across 62 pig farms located in 14 LGAs within Southwest Nigeria, spanning December 2019 to March 2021.
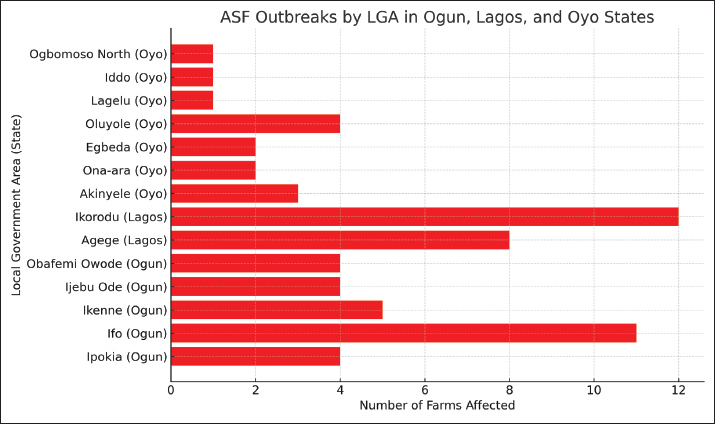
Fig. 6. Distribution of ASF outbreaks across LGAs in 62 pig farms in Ogun, Lagos, and Oyo states, Nigeria, between 2019 and 2021. The rapid geographic spread of ASF closely followed established pig trade routes and was intensified during the outbreaks by the ongoing movement and sale of pigs. Farmer panic led to increased marketing activity, which inadvertently accelerated the dissemination of the virus and infected pigs through regional market channels (Table 7, Fig. 3). Biosecurity breachesBiosecurity breaches were detected in both farm infrastructure and management practices, drawing on farmers survey responses (Table 9), and investigators on site observations during farm visits (Table 10). Table 9. Survey of biosecurity infrastructure conditions and management practices on pig farms affected by ASF in Oyo, Ogun, and Lagos states, Nigeria (2019–2021).
Table 10. Observation of biosecurity breaches on pig farms affected by ASF, (2019–2021).
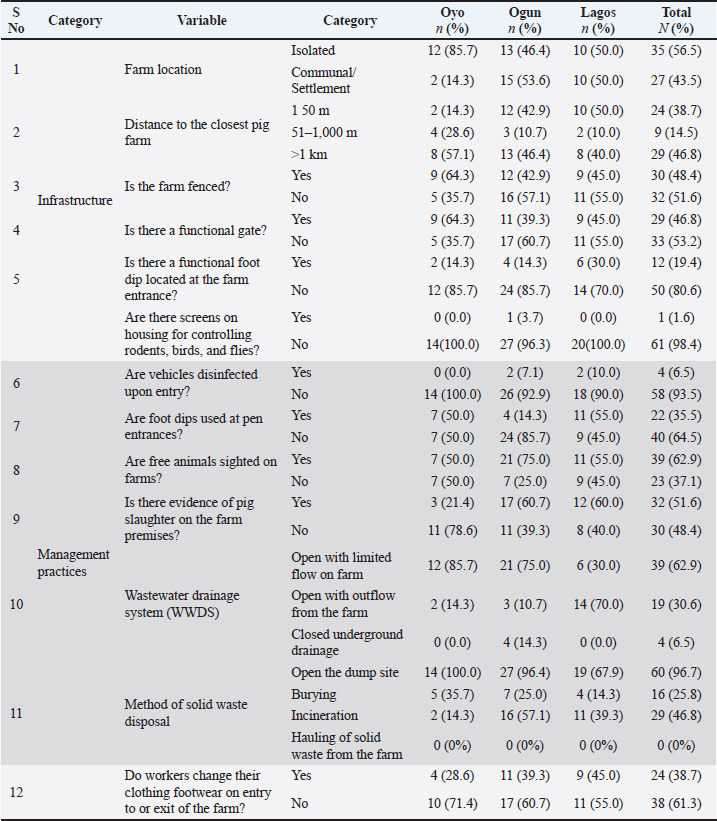
Farm infrastructureOnly 11.3% of the farms had quarantine facilities in place. Approximately 43.5% of the farms operated within communal farm arrangements, while the remaining 56.5% were isolated units. Perimeter fencing and gates were present on 48.4% and 46.8% of the farms, respectively. Functional foot dips were observed at the main entrance on just 19.4% of farms, and only 1.6% had netting installed on housing to restrict the entry of rodents, birds, and flies. Notably, all pig housing structures were open sided, lacking full enclosure (Tables 9 and 10). Farm management practices (based on surveys)Only 14.5% of the farmers had a documented biosecurity protocol for their farms. Over half (53.2%) used farm-owned vehicles to transport inputs and pigs, while the remainder relied on commercial transport. All farms (100%) operated a continuous pig flow system, indicating that the facilities constantly held pigs. A substantial proportion (79.6%) of breeding stock or semen were obtained from multiple suppliers; however, only 1.6% of these sources held certification as disease-free. None of the farmers assessed incoming stock for ASFV. Feed was usually not sourced from standard feed producers; instead, 98.4% relied on on-farm milling or toll milling (75.8%). Most water sources were dug wells (80.6%). Within a 6-month period, 93.5% of farms sold pigs to other farms or markets, primarily through pig buyers (93.1%) who accessed the production areas directly. These buyers provided their own vehicles for transportation in 98.3% of the cases. Additionally, 96.8% of farms allowed other visitors into production areas for varied reasons, and 41.9% shared staff with other farms (Table 9). Farm management practices (observation-based)Vehicle disinfection at farm entrances was observed in only 6.5% of farms, whereas foot dips at pen entrances were present on 35.5%. Free-range animals were on 62.9% of farms, and on-site pig slaughtering occurred at 51.6%. Wastewater was discharged on the farms through open drainage systems on 93.5% of farms, with 30.6% extending beyond farm boundaries. Solid waste disposal was primarily through open dumping (96.7%), with occasional burying or incineration. Furthermore, 61.3% of farms did not require workers to change clothing or footwear upon entering or exiting the production area (Table 10). DiscussionThis study presents a comprehensive analysis of the ASF outbreak that occurred between 2019 and 2021 in Southwest Nigeria. The study captures critical field events, profiles the demographics of affected pig farmers, morbidity and mortality patterns, and clinical manifestations in pigs. Furthermore, it highlights structural and operational deficiencies in biosecurity, encompassing both farm infrastructure and management practices, as well as human behaviors that collectively contributed to the persistence and spread of ASF in the region. Demographics and previous experience with ASFIn the study area, demographic data show that pig farming is a male-driven enterprise (83.9%). Previous research has similarly highlighted male dominance in the industry within the same region (Abiola et al., 2015; Obayelu et al., 2017; Omotosho et al., 2019). Comparable trends were observed in a Belgian study, which found that 76.8% of pig farmers were men (Laanen et al., 2014). This gender disparity in pig farming may be attributed to the industry’s physical demands and rigorous management practices. In addition to demographic patterns, socio-cultural and religious factors further shape pig farming practices across the region, with 87.1% of respondents identifying as Christians. Pork consumption is religiously prohibited in many northern regions of Nigeria, thereby constraining both the production and marketability of pigs. In contrast, pig farming is more culturally and religiously acceptable in the southern parts of the country, where it is more widely practiced. This socio religious divide positioned Southwest Nigeria as a major pig production hub, offering comparatively favorable conditions for enterprise expansion and market integration (Fasina et al., 2010; Abiola et al., 2015; Adesehinwa et al., 2024). Given the relatively high educational attainment of the farmers, with 67.7% having completed at least secondary school, they possess the capacity to adopt innovations in pig production and disease management. Previous reports have also highlighted the high educational attainment among pig farmers in Nigeria (Obayelu et al., 2017; Omowon et al., 2019). Additionally, many (75.8%) were between 40 and 60 years old, and 87.1% had over 5 years of experience in pig farming, reflecting a stable, experienced, and potentially adaptable workforce. This committed cohort represents a critical entry point for the successful rollout of improved ASF control strategies, provided targeted training and appropriate incentives are provided. Smallholder farmers continue to play a vital role in pig production and food security across Nigeria (Nwachukwu and Udegbunam, 2020; Chiaka et al., 2022). Most respondents (90.3%) reported pre-outbreak herd sizes of 200 pigs or less, reflecting the predominance of small-scale operations. This pattern aligns with earlier findings from the region (Omotosho et al., 2019; Ogundijo et al., 2023) and echoes broader trends observed in other African countries, where smallholder systems remain the backbone of the pork industry (Dixon et al., 2020). However, among the various challenges hindering the expansion of pig farming in Nigeria, disease outbreaks, particularly ASF, have emerged as a major constraint (Adesehinwa et al., 2024). Since the first confirmed ASF outbreak in Nigeria in 1997 (Babalobi et al., 2007; Awosanya et al., 2015) and an earlier unverified report in 1973 (Owolodun et al., 2010; Awosanya et al., 2021), the pig sector has faced repeated disruptions. In this study, 72.6% of the farmers surveyed reported prior ASF outbreaks, which were frequently accompanied by substantial pig losses (Table 2). These outbreaks have imposed significant financial strain on farmers, often without compensation or insurance. Consequently, the farmers may deliberately opt to maintain smaller herd sizes as a risk mitigation strategy, given the sporadic and unpredictable nature of ASF occurrences. Morbidity patterns, mortality rate, and reduction in farm sizeTo better understand transmission dynamics and inform control strategies, we examined the ASF spread sequence among age groups on affected farms. The most frequently observed pattern of morbidity onset followed the order: adults → piglets → weaners → growers, which was reported on 75.8% of farms (Table 3). A statistically significant association was found between age groups and the onset of morbidity with adults being the first affected group and growers being the last affected group (Table 4). This distribution reveals that ASF may initially affect older pigs, possibly due to higher exposure risk or a shorter incubation period, facilitating subsequent transmission to younger cohorts. Farmers commonly reported early signs of anorexia and lethargy in sows, boars, and finishers, preceding the appearance of clinical signs in younger age groups. Nursing piglets typically became sick after the adults, followed by weaned piglets and finally, growers. The disease onset rate and interval across these groups varied widely, from a few hours to several days. Although the precise factors driving this age specific progression remain unclear, it reflects the natural course of intra herd viral transmission or age-related differences in incubation periods and susceptibility to infection. Weaned pigs and growers are housed in separate units on many farms, often at varying distances from adult pigs and breeding stock, which can slow intra herd transmission of ASF across age groups. Adult pigs are typically more exposed to infectious agents because they are central to breeding activities and trade interactions. Their increased contact with farm visitors, other pigs, and potentially contaminated fomites increases their vulnerability. Furthermore, breeding practices, such as the introduction of semen, new boars, or replacement sows without proper health screening, pose a significant risk for the introduction of ASF into breeding units. The observed pattern of ASF spread in this study is consistent with findings from other outbreaks. For example, a 2019 investigation in Vietnam reported a high initial infection rate among adult pigs, with subsequent transmission to piglets and weaners (Nga et al., 2020). Similarly, research in Latvia found that virus transmission within large-scale farms progressed slowly, driven primarily by direct pig-to-pig contact and deficiencies in internal biosecurity (Lamberga et al., 2020). Further research is warranted to elucidate the drivers of these transmission patterns and compare outbreak characteristics across regions, thereby informing more targeted and effective ASF control measures. Beyond intra herd transmission dynamics, factors such as age, immune competence, and viral strain virulence play critical roles in shaping the clinical manifestations of ASF, which can range from acute hemorrhagic forms to chronic or subclinical presentations (Blome et al., 2020). The cumulative mortality rate was 60.6%, with herd depopulation ranging from 758.8% to 100% (Table 5). This level of mortality indicates infection with a moderately virulent ASFV strain, resulting in rapid disease progression and significant production losses. Compared with previous outbreaks in Nigeria, this mortality rate is somewhat lower than the historically reported extreme levels. For example, Babalobi et al. (2001) documented a 91% mortality rate during the 2001 Ibadan outbreak, while more recent outbreaks in Imo and Abia States in 2017 recorded mortality rates of 73% and 80%, respectively (Masembe et al., 2022). The variability in ASF outcomes is evident not only within Nigeria, where mortality rates range from 1.6% to 91.7% but also across West Africa. The reported mortality rates are 13.3%–83.5% in Burkina Faso, 39.7%–100% in Côte d’Ivoire, and 50%–75% in Mali (Meki et al., 2025). Variations in ASF mortality across outbreaks and regions reflect a complex interplay of factors, including differences in virus virulence, farm management practices, and response strategies. Several determinants may account for the observed disparities. Viral characteristics, such as the presence of distinct genotypes, e.g., genotype I versus genotype II and intra genotype variability, are known to influence disease severity and transmission dynamics (Dixon et al., 2020; Meki et al., 2025). Farm-level biosecurity measures and management practices also play a critical role in shaping outbreak outcomes (Dione et al., 2015). The timeliness of outbreak detection and the effectiveness of response efforts further affect mortality rates (Penrith and Vosloo, 2009). Herd demographics, particularly age structure, can influence susceptibility and disease progression (Babalobi et al., 2001). Additionally, regional differences in ASF impact are influenced by environmental and socioeconomic factors, such as pig population density, live animal movement, and access to veterinary services (Fasina et al., 2012b). Co infections and the baseline health status of herds may further exacerbate ASF outcomes, adding to the wide range of mortality figures reported in Nigeria and other West African countries (Arias et al., 2018). The outbreak had substantial financial implications, with an estimated direct loss of ₦166,264,000 (US$436,961.90 USD) across 62 affected farms (Table 6). This figure reveals the profound economic impact of ASF outbreaks on Nigeria’s pig sector, reflecting only mortality-related losses and excluding indirect costs from panic sales, emergency slaughter, relocation, and prolonged production disruptions. These findings are consistent with those of Fasina et al. (2012b), who modeled the economic consequences of ASF in smallholder farrow to finish units and projected annual losses of up to US$910,836.70 in a 122-sow piggery, emphasizing the cost effectiveness of biosecurity interventions. Additional studies have highlighted pig farmers’ economic vulnerability in Nigeria in the face of ASF. For instance, Adekola et al. (2022) reported that 78.3% of surveyed pig farmers in Ogun State experienced significant financial strain due to ASF, while Oyegbami et al. (2024) identified ASF as one of the leading barriers to profitability among pig farmers in Lagos State. Controlling the spread of ASF within a country or region relies heavily on strict movement restrictions for pigs. This is especially important in Africa and other areas where the virus mainly spreads between domestic pigs rather than through wild animals or ticks (Dixon et al., 2020). Despite this fact, field observations during the outbreak revealed heightened movement of pigs from infected farms through market systems, surpassing the typical trade activity. Such behavior runs counter to established ASF control measures and likely played a substantial role in the virus’s continued spread and persistence. Remarkably, 96.8% of farmers contributed to the dissemination of the virus through panic sales, on farm slaughter, and movement of infected pigs, whereas only 3.2% adhered to appropriate disposal protocols, such as thorough disinfection and burial of all ASF-related carcasses (Table 7 and Fig. 3). Similar behaviors have been documented in Kenya and Uganda, where the sale of diseased pigs through distant market channels facilitated long distance transmission of ASF (Lichoti et al., 2016). The unrestricted movement of pigs during outbreaks plays a critical role in the spread of ASFV, enabling the introduction of carrier pigs into new areas and contaminating the environment with viremic blood, feces, and other bodily secretions. Importantly, ASFV shedding begins before the onset of clinical signs, and once symptoms appear, the virus is present in high concentrations across all excretions and secretions (Sánchez-Vizcaíno et al., 2012), further amplifying the risk of transmission. Clinical and epidemiological findingsClinical assessment of affected pigs across farms revealed varying disease presentations (Table 8 and Fig. 4). Anorexia, depression, lethargy (100%), and fever (95.8%) were the most frequently observed clinical signs. These findings are consistent with the known clinical manifestations of ASF (Dixon et al., 2019). Although these clinical signs are not specific to ASF, their consistency with hemorrhagic fever diseases emphasizes the importance of differential diagnosis in outbreak investigations. The overlap of clinical presentations with other swine diseases, such as classical swine fever, erysipelas, and septicemia, necessitates laboratory confirmation to accurately identify ASF as the causative agent (WOAH, 2019). This is particularly critical in regions such as southwest Nigeria, where multiple swine diseases may co circulate and complicate field diagnoses (Fasina et al., 2012a). Epidemiological investigation revealed the rapid and severe spread of ASF along both local and transboundary trade routes (Figs. 1 and 5). The index case, which was reported in December 2019 on a farm in Ipokia LGA, Ogun State, illustrates the heightened vulnerability of farms situated near international borders. Ipokia LGA lies just 46 km by road from Porto Novo, the capital of the Republic of Benin, and is a hub for intense cross-border trade, including pigs and other livestock. Such movement of animals has been consistently identified as a key driver of transboundary animal disease spread, particularly where biosecurity at informal markets and border checkpoints is limited or poorly enforced (Penrith et al., 2013; Sánchez-Vizcaíno et al., 2019). The Ipokia outbreak subsequently propagated along established local trade networks, demonstrating how cross-border introductions can quickly evolve into regional outbreaks. In January 2020, the outbreak extended to Oke-Aro pig farm settlement in Ifo LGA, situated 75 km by road from Ipokia. Robust commercial interactions exist between these two locations, with input suppliers, pig buyers, and transporters actively involved in the trade of feed ingredients, breeding stock, finishing pigs, and other supplies. The Oke-Aro pig farm accommodates approximately 5,000 smallholder farms and emerged as a critical hot spot for ASF transmission during the outbreak cycle. This reaffirms the established correlation between high stocking density and inadequate biosecurity practices in amplifying disease risk (Costard et al., 2013). The swift spread to Lagos State, Agege LGA and Oyo State, Ona Ara LGA between January and March 2020 further illustrates the deep interconnected pig trade routes across Nigeria. Farmers trying to mitigate financial losses moved pigs into trade as a panic response during the outbreak. Although farmers initiated this process, it was often driven by pig buyers and transporters who sought profit and frequently circumvented regulatory oversight, thereby intensifying disease spread (Table 7, Fig. 3). Biosecurity breachesTo deepen our understanding of the risk factors for the spread of ASF across pig farms in Nigeria, this study undertook a comprehensive assessment of farm infrastructure and management practices. By systematically investigating affected farms, some critical breaches in biosecurity protocols that contributed to ASF transmission and persistence were identified. This investigation relied on information provided by farmers alongside observations made by the investigators, as detailed in Tables 9 and 10. Farm infrastructureThere was a lack of standard quarantine facilities for incoming stock (88.7%) on many farms, which is a significant breach. Quarantine facilities are vital for monitoring the health status of new or returning animals before they are integrated into the herd. Without these infrastructures, farms are at risk of introducing infected animals, which can act as reservoirs for ASF and other diseases (Penrith and Vosloo, 2009). This finding aligns with observations in other regions, where inadequate quarantine measures have been closely linked to the rapid spread of infectious diseases (Dione et al., 2016). The proximity of farms has a profound impact on disease dynamics, as spatial clustering plays a critical role in disease control. Short distances between farms can facilitate transmission through several pathways, such as airborne particles, rodents, and free-range birds or animals. (Alarcón et al., 2021). While 56.5% of the farms were isolated and independent, 43.5% were situated in communal or settlement farming systems. As noted earlier, communal systems, characterized by proximity and shared resources, can facilitate the transmission of ASF (Dixon et al., 2020). The purpose of establishing pig farm clusters in many parts of Africa for smallholder farmers was to make pig production more sustainable, competitive, and profitable (Adesehinwa et al., 2024). Ideally, the cluster arrangement would provide many benefits to smallholder farmers. First, these initiatives provide access to shared resources, such as feed mills, water systems, and veterinary services, which are often out of reach for smallholder farmers. Additionally, cost efficiency emerges as a significant benefit; this is achieved through bulk purchasing of essential items such as feed, medicine, and equipment. Collective marketing also enhances market access, allowing farmers to maintain a consistent supply and negotiate more effectively with buyers. The value of knowledge sharing within these networks is also beneficial. These platforms enable farmers to exchange best practices, receive training, and benefit from cooperative extension services, thereby fostering professional growth. Finally, the shared farming initiatives should ideally contribute to improved biosecurity control, promoting better health and safety measures across farming operations. Ultimately, smallholder farmers in cluster farms can leverage these advantages to reduce production costs, thereby enhancing their profitability. The presence of fencing (48.4%) and gates (46.8%) for traffic control reflects progress toward establishing perimeter biosecurity. However, enhancing perimeter defenses remains necessary within the study area. These measures help limit unauthorized access and reduce the pathogen introduction risk. Alarcón et al. (2021) emphasized that effective fencing and gating systems are essential for creating a secure perimeter and forming the first line of defense against external threats. Another area of concern is the rare use of foot dips at farm main entrances (19.4%). Foot dips are simple yet effective biosecurity measures for preventing the introduction of pathogens via footwear. The low adoption rate of this measure may reflect limited awareness or resource availability among farmers (Fasina et al., 2012a). Enhancing farmer education and providing access to affordable disinfection solutions could substantially improve compliance and aid in the control of ASF spread. Rodents, flies, and free-range birds or animals are established pathways of ASF and other disease spread across and within farms (Costard et al., 2013). Although all pig houses were open-sided buildings, as practiced in many tropical environments (Godyń et al., 2020, Omotosho et al. 2024), only 1.6% of the housing units had netting or screens to control access. The absence of such protective barriers significantly increases the risk of pathogen transmission. Therefore, as recommended by Dixon et al. (2020), implementing pest screens and rodent control measures could greatly enhance farm biosecurity. Farm management practices (based on surveys)Only a few farms had documented biosecurity protocols (14.5%), revealing a critical weakness in the existing disease prevention strategies. Biosecurity protocols are fundamental for mitigating the risk of disease introduction and spread, as they provide structured guidelines for farm management, visitor control, and sanitation practices (Alarcón et al., 2021). The absence of such protocols leaves farms highly vulnerable to outbreaks, as seen in other regions affected by ASF (Costard et al., 2013). Farm inputs, such as pigs and feed, were transported using personal vehicles (53.2%) or commercial transporters (46.8%). The observed practice of using the same vehicle to transport feed, pigs for replacement, and pigs for slaughter with limited emphasis on cleaning and disinfection raised significant biosecurity concerns. Notably, all farms employed a continuous pig flow system, which poses a substantial risk of disease transmission. Unlike All-In All-Out (AIAO) systems, continuous flow operations maintain a constant population of pigs on site, leading to higher pathogen loads and complicating disease management efforts (Alarcón et al., 2021). The reliance on continuous flow in the study area may stem from resource constraints or limited awareness of its implications, posing a critical barrier to effective ASF control. Many farmers sourced breeder pigs, piglets, or semen from multiple sources (79.6%), with only 1.6% of these acquisitions accompanied by health certification. This is a major lapse in biosecurity as health certification and quarantine protocols represent gold standard measures for preventing the introduction of ASF into swine populations (Bellini et al., 2016). The lack of diagnostic testing for incoming animals or semen further elevates the risk of ASF introduction, particularly in Nigeria, where the virus predominantly circulates within the domestic pig population (Dixon et al., 2020). Feed and water sourcing practices among farmers in the study area present notable biosecurity concerns. Feed ingredients are vulnerable to contamination by various pathogens during field exposure, processing, transport, and storage. These pathogens can persist in feed and introduce disease onto farms (Dee et al., 2016). An overwhelming 98.4% of farms rely on on-site feed milling, while 75.8% use toll milling services. Notably, none of the farms used branded feed produced under optimal feed biosecurity protocols. Farms frequently source ingredients from multiple suppliers, often with limited quality control measures in place. Contaminated feed is a well-established route for ASF transmission (Stoian et al., 2019), highlighting the critical importance of feed quality assessments as a key intervention point for disease prevention and control (Guinat et al., 2016; Niederwerder, 2021). Similarly, water sourcing practices posed additional risks. Most farms rely on dug wells (80.6%) and boreholes (45.2%), with minimal evaluation of water quality. Because poor water quality can compromise pig health and facilitate disease spread, routine testing and treatment of water sources are strongly recommended (Lozinski et al., 2020). Human activities pose significant challenges to ASF control, as it has been identified as the primary driver of long-distance transmission events and the introduction of ASFV into domestic pig farms (Fasina et al., 2012a; Bellini et al., 2021; Alotaibi et al., 2024). In the study area, pigs were frequently sold on farms, with buyers visiting farms to select, bargain, and collect pigs, occurring 79.3% occasionally and 19.0% always, thereby substantially elevating the risk of ASF transmission. The widespread practice of buyers transporting pigs (98.3%) worsens this risk. Buyers often rely on commercial transporters who indiscriminately use their vehicles and prioritize time efficiency and financial gains over biosecurity. This aligns with the findings of Penrith and Vosloo (2009), who observed that unrestricted farm access during pig sales significantly facilitates the transmission of ASF. Visitor access to production areas in 96.8% of farms, including technicians, input suppliers, and other service providers, revealed a significant breach of biosecurity. The unregulated movement of people, as noted by Alarcón et al. (2021), can inadvertently introduce pathogens into previously unaffected farms. Furthermore, the common practices of farmers visiting other pig farms (53.2%) and sharing staff between farms (41.9%) compound the risk of indirect disease transmission. Strengthening biosecurity measures, particularly by regulating the access and activities of people on farms, is a crucial step in mitigating these anthropogenic risks. Farm management practices (observation-based)At farm entrances, only 6.5% implemented the active vehicle disinfection protocols. This gap in farm biosecurity measures represents a significant weakness that requires immediate attention because livestock farms are known to face the risk of disease introduction through vehicles, which often serve as critical transmission pathways. (Dee et al., 2004; Lowe et al., 2014). Functional foot dips were observed at pen entrances on only 35.5% of farms, revealing a significant gap in farm biosecurity measures. These dips play a crucial role in disinfecting footwear and preventing pathogen introduction. This finding is consistent with previous reports that foot disinfection practices are frequently overlooked in resource-constrained environments (Alarcón et al., 2021). Ensuring proper maintenance and regular use of foot dips can significantly lower the risk of ASF virus transmission in Nigeria. Free-ranging pigs and other livestock were observed around pens on 62.9% of farms, revealing a significant risk to biosecurity. Such animals can serve as reservoirs or vectors for the ASF virus and other pathogens (Costard et al., 2013). Implementing measures, such as improved fencing and secure housing, to prevent contact between domestic pigs and free-ranging animals is essential to mitigate this transmission risk (Alarcón et al., 2021). The common practice of slaughtering pigs on farms (51.6%) raises significant biosecurity concerns. Without proper management, this on-site practice can become a source of environmental contamination and disease transmission. This aligns with the findings of Fasina et al. (2012a), who reported that inadequate slaughter waste disposal contributes to the spread of infectious diseases through environmental contamination. Waste management practices pose another significant risk of environmental contamination and disease transmission. On the farms, wastewater was disposed of via open drainage systems (62.9%), with 30.6% of these channels extending to nearby plots or water bodies, an approach that heightens the risk of ASF virus dissemination (Dixon et al., 2020), while solid waste disposal methods were also suboptimal. Open dumping was observed on 96.7% of farms, followed by incineration (46.8%) and burial (25.8%). These practices indicate the urgent need for improved waste management. Transitioning to closed drainage systems and adopting more controlled and sanitary waste disposal strategies could substantially reduce these biosecurity threats. Another notable lapse is that workers on many farms (61.3%) failed to change clothing or footwear when entering or leaving farms, which severely compromises biosecurity protocols. Adhering to stringent hygiene measures is essential for curbing pathogen transmission, as emphasized by Dione et al. (2015). Study limitationsAlthough this study provides valuable insights into the dynamics of ASF spread, clinical patterns, and associated biosecurity breaches on farms in the study area, certain limitations must be acknowledged. First, the reliance on farmer-reported data may have introduced recall bias, potentially affecting the accuracy of the reported sequence of disease spread and mortality patterns. Second, the study was geographically restricted to select states in southwest Nigeria, which may limit the generalizability of the findings to other regions or countries with varying farming practices, pig trade dynamics, and ecological conditions. Third, the epidemiological curve of the disease was based on a limited number of farms due to insufficient resources and personnel for comprehensive outbreak tracing. Finally, comprehensive laboratory testing of all samples from affected farms was not feasible, limiting the ability to confirm the presence of ASFV in every reported case. Future research that incorporates broader geographic coverage, improved resources, and laboratory diagnostics would enhance the robustness of findings and contribute to the development of more effective control measures. ConclusionThis study provides a comprehensive analysis of the ASF outbreak in Southwest Nigeria from 2019 to 2021, capturing critical field events, profiling affected pig farmers, and detailing the morbidity, mortality, and clinical manifestations observed in pigs. This study identifies biosecurity lapses spanning infrastructure, farm management, and human behavior as key drivers of the persistence and transmission of ASF in the region. Targeted interventions are imperative to mitigate future outbreaks and ensure the resilience of the pig industry in Nigeria. These include reinforcing farm level biosecurity, enhancing farmer education, regulating waste disposal and visitor access, and overhauling pig marketing and transport systems. Sustained government engagement in surveillance, policy enforcement, and emergency preparedness will be essential to building a robust ASF prevention and control framework. AcknowledgmentsWe gratefully acknowledge the invaluable support of the Pig Farmers Association of Nigeria (PFAN), particularly the Oyo, Ogun, and Lagos State chapters, for their cooperation during the investigation. We also extend our sincere appreciation to all participating pig farmers, farm staff, and field officers whose contributions made this study possible. Special thanks go to the Veterinary Teaching Hospital, University of Ibadan, and the Department of Virology, Molecular Virology Unit, College of Medicine, University of Ibadan, for their technical assistance and laboratory support. Conflicts of interestThe authors declare no conflicts of interest. FundingThe authors funded all aspects of the research. Authors’ contributionsAJO: Conceptualization, Methodology, Investigation, Data Curation, Writing Original Draft Writing Review Editing. OOO: Conceptualization, Methodology, Supervision, Project Administration, Funding Acquisition, Writing Review Editing. AEA: Investigation and data collection. OVA: Investigation, data collection, and data curation. ARO: Formal Analysis, Data Curation Writing Original Draft. HA: Formal Analysis, Data Curation, and Original Draft Writing. ADJ: Investigation and data collection; AM: Investigation and data collection; JOT: Methodology, supervision, writing, review, and editing; OTO: Conceptualization, methodology, supervision, project administration, funding acquisition, writing, review, and editing. Data availabilityAll data supporting this study’s findings are available within the manuscript. Further information may be retrieved through the corresponding author upon reasonable request. ReferencesAbiola, J., Omotosho, O., Adeniyi, O. and Ayoade, G. 2015. Sociodemographic characteristics of swine producers and swine management practices in Ibadan, Oyo State, Nigeria. Alex. J. Vet. Sci. 47(7), 7–17. Achenbach, J.E., Gallardo, C., Nieto-Pelegrín, E., Rivera-Arroyo, B., Degefa-Negi, T., Arias, M., Jenberie, S., Mulisa, D.D., Gizaw, D., Gelaye, E., Chibssa, T.R., Belaye, A., Loitsch, A., Forsa, M., Yami, M., Diallo, A., Soler, A., Lamien, C.E. and Sánchez-Vizcaíno, J.M. 2017. Identification of a new genotype of African swine fever virus in domestic pigs from Ethiopia. Transbound. Emerg. Dis. 64, 1393–1404. Adedeji, A.J., Luka, P.D., Atai, R.B., Olubade, T.A., Hambolu, D.A., Ogunleye, M.A., Muwanika, V.B. and Masembe, C. 2021. First-time presence of African swine fever virus genotype II in Nigeria. Microbiol. Resour. Announc. 10(26), 26. Adekola, O.A., Komolafe, S.E. and Raji, M.A. 2022. Economic impacts of African swine fever epidemic among pig cooperative farmers in Ogun State, Nigeria. Federal University of Agriculture, Abeokuta. Adesehinwa, A.O.K., Boladuro, B.A., Dunmade, A.S., Idowu, A.B., Moreki, J.C. and Wachira, A.M. 2024. Pig production in Africa: current status, challenges, prospects, and opportunities. Anim. Biosci. 37(4), 730–741. Alarcón, L.V., Allepuz, A. and Mateu, E. 2021. Biosecurity in pig farms: a review. Porcine Health Manag. 7(1), 5. Alotaibi, B.S., Wu, C.H., Khan, M., Nawaz, M., Chen, C.C. and Ali, A. 2024. African swine fever: insights into genomic aspects, reservoirs and transmission patterns of virus. Front. Vet. Sci. 11, 1–12 Arias, M., Jurado, C., Gallardo, C., Fernández-Pinero, J. and Sánchez-Vizcaíno, J.M. 2018. Gaps in African swine fever: analysis and priorities. Transbound. Emerg. Dis. 65(Suppl. 1), 235–247. ASF-STOP. 2021. Understanding and combatting African swine fever: A European perspective. Iacolina, L., Penrith, M.L., Bellini, S., Chenais, E., Jori, F., Montoya, M. and others, editors. Wageningen: Wageningen Academic Publishers. Atuhaire, D.K., Afayoa, M., Ochwo, S., Mwesigwa, S., Okuni, J.B., Olaho-Mukani, W. and Ojok, L. 2013. Molecular characterization and phylogenetic study of African swine fever virus isolates from recent outbreaks in Uganda. Virol. J. 10, 247. Awosanya, E.J., Olugasa, B., Ogundipe, G. and Grohn, Y.T. 2015. Sero-prevalence and risk factors associated with African swine fever on pig farms in Southwest Nigeria. BMC. Vet. Res. 11, 133. Awosanya, E.J., Olugasa, B.O., Gimba, F.I., Sabri, M.Y. and Ogundipe, G.A. 2021. Detection of African swine fever virus in pigs in Southwest Nigeria. Vet. World 14(7), 1840–1845. Backhans, A., Sjölund, M., Lindberg, A. and Emanuelson, U. 2015. Biosecurity level and health management practices in 60 Swedish farrow-to-finish herds. Acta. Vet. Scand. 57(1), 1–11. Bellini, S., Casadei, G., De Lorenzi, G. and Tamba, M. 2021. A review of risk factors of African swine fever incursion in pig farming within the European Union scenario. Pathogens 10, 84. Bellini, S., Rutili, D. and Guberti, V. 2016. Preventive measures aimed at minimizing the risk of African swine fever virus spread in pig farming systems. Acta. Vet. Scand. 58, 82. Blome, S., Franzke, K. and Beer, M. 2020. African swine fever – A review of current knowledge. Virus. Res. 287, 198099. Boinas, F.S., Wilson, A.J., Hutchings, G.H., Martins, C. and Dixon, L.J. 2011. The persistence of African swine fever virus in field-infected Ornithodoros erraticus during the ASF endemic period in Portugal. PLos One. 6(5), e20383. Chiaka, J.C., Zhen, L., Yunfeng, H., Xiao, Y., Muhirwa, F. and Lang, T. 2022. Smallholder farmers' contribution to food production in Nigeria. Front. Nutr. 9, 916678. Costard, S., Mur, L., Lubroth, J., Sanchez-Vizcaino, J.M. and Pfeiffer, D.U. 2013. Epidemiology of African swine fever virus. Virus. Res. 173(1), 191–197. Danzetta, M.L., Marenzoni, M.L., Iannetti, S., Tizzani, P., Calistri, P. and Feliziani, F. 2020. African swine fever: lessons to learn from past eradication experiences. Front. Vet. Sci. 7, 296. De Lorenzi, G., Borella, L., Alborali, G.L., Prodanov-Radulović, J., Štukelj, M. and Bellini, S. 2020. African swine fever: a review of cleaning and disinfection procedures in commercial pig holdings. Res. Vet. Sci. 132, 262–267. Dee, S., Neill, C., Singrey, A., Clement, T., Cochrane, R., Jones, C., Patterson, G., Spronk, G., Christopher-Hennings, J. and Nelson, E. 2016. Modeling the transboundary risk of feed ingredients contaminated with porcine epidemic diarrhea virus. BMC. Vet. Res. 12, 51. Dee, S.A., Deen, J., Otake, S. and Pijoan, C. 2004. An experimental model to evaluate the role of transport vehicles as a source of transmission of porcine reproductive and respiratory syndrome virus to susceptible pigs. Can. J. Vet. Res. 68(2), 128–133. Dione, M.M., Akol, J., Roesel, K., Kungu, J., Ouma, E.A., Wieland, B. and Pezo, D. 2015. Risk factors for African swine fever in smallholder pig production systems in Uganda. Transbound. Emerg. Dis. 62(2), 219–228. Dixon, L.K., Stahl, K., Jori, F., Vial, L. and Pfeiffer, D.U. 2020. African swine fever epidemiology and control. Annu. Rev. Anim. Biosci. 8, 221–246. Dixon, L.K., Sun, H. and Roberts, H. 2019. African swine fever. Antiviral Res. 165, 34–41. Ebwanga, E.J., Ghogomu, S.M. and Paeshuyse, J. 2021. African swine fever in Cameroon: a review. Pathogens 10(4), 421. Fadiga, M., Jost, C. and Ihedioha, J. 2013. Financial costs of disease burden, morbidity and mortality from priority livestock diseases in Nigeria: Disease burden and cost-benefit analysis of targeted interventions. ILRI Res. Rep. 33. Nairobi: ILRI. Fasina, F.O., Agbaje, M., Ajani, F.L., Talabi, O.A., Lazarus, D.D., Gallardo, C., Thompson, P.N. and Bastos, A.D.S. 2012a. Risk factors for farm-level African swine fever infection in major pig-producing areas in Nigeria, 1997–2011. Prev. Vet. Med. 107(1–2), 65–75. Fasina, F.O., Lazarus, D.D., Spencer, B.T., Makinde, A.A. and Bastos, A.D.S. 2012. Cost implications of African swine fever in smallholder farrow-to-finish units: economic benefits of disease prevention through biosecurity. Transbound. Emerg. Dis. 59(3), 244–255. Fasina, F.O., Shamaki, D., Makinde, A.A., Lombin, L.H., Lazarus, D.D., Rufai, S.A., Adamu, S.S., Agom, D., Pelayo, V., Soler, A., Simón, A., Adedeji, A.J., Yakubu, M.B., Mantip, S., Benshak, A.J., Okeke, I., Anagor, P., Mandeng, D.C., Akanbi, B.O., Ajibade, A.A., Faramade, I., Kazeem, M.M., Enurah, L.U., Bishop, R., Anchuelo, R., Martin, J.H. and Gallardo, C. 2010. Surveillance for African swine fever in Nigeria, 2006–2009. Transbound. Emerg. Dis. 57(4), 244–253. Fernández-Pinero, J., Gallardo, C., Elizalde, M., Robles, A., Gómez, C., Bishop, R., Heath, L., Couacy-Hymann, E., Fasina, F.O., Pelayo, V., Soler, A. and Arias, M. 2013. Molecular diagnosis of African swine fever by a new real-time PCR using universal probe library. Transbound. Emerg. Dis. 60(1), 48–58. Fischer, M., Hühr, J., Blome, S., Conraths, F.J. and Probst, C. 2020. Stability of African swine fever virus in carcasses of domestic pigs and wild boar experimentally infected with the ASFV "Estonia 2014" isolate. Viruses 12(10), 1118. Gallardo, M.C., Reoyo, A.D.L.T., Fernández-Pinero, J., Iglesias, I., Muñoz, M.J. and Arias, M.L. 2015. African swine fever: a global view of the current challenge. Porcine Health Manag. 1(1), 21. Gaudreault, N.N., Madden, D.W., Wilson, W.C., Trujillo, J.D. and Richt, J.A. 2020. African swine fever virus: an emerging DNA arbovirus. Front. Vet. Sci. 7, 215. Gebreyohannes, T.K. 2015. Potential methods of eliminating swine pathogens in livestock transport trailers. Saskatoon: University of Saskatchewan. Available via https://innovationporc.ca/media/1557-literature-review-2015-methods-to-eliminate-phatogens-in-trailers.pdf Godyń, D., Herbut, P., Angrecka, S. and Corrêa Vieira, F.M. 2020. Use of different cooling methods in pig facilities to alleviate the effects of heat stress—A review. Animals 10(9), 1459. Guetiya Wadoum, R.E., Kasiiti Lichoti, D.J., Noelina, D.N., Austine, D.B., Amara, D.L., Sesay, A.K., Jojo, D.H., Conteh, A.M., Leigh, M.H., Marah, J., Sesay, H.M., Caulker, G., Kanu, S.T., Sesay, B.H., Turay, J.A. and Bobo, D.G. 2020. Quantitative outcomes of a One Health approach to investigate the first outbreak of African swine fever in the Republic of Sierra Leone. Glob. J. Med. Res. 20(1), 27–33. Guinat, C., Wall, B., Dixon, L. and Pfeiffer, D.U. 2016. English pig farmers' knowledge and behaviour towards African swine fever suspicion and reporting. PLos One. 11(9), 161431. Igbokwe, I.O. and Maduka, C.V. 2018. Disease burden affecting pig production in Nigeria: review of current issues and challenges. Rev. Elev. Med. Vet. Pays Trop. 71(1–2), 87–95. Ito, S., Kawaguchi, N., Bosch, J., Aguilar-Vega, C. and Sánchez-Vizcaíno, J.M. 2023. What can we learn from the five-year African swine fever epidemic in Asia?. Front. Vet. Sci. 10, 1273417. Janse Van Rensburg, L., Penrith, M.L. and Etter, E.M.C. 2022. Prioritisation of provinces for African swine fever intervention in South Africa through decision matrix analysis. Pathogens 11(2), 135. Jori, F., Bastos, A., Boinas, F., Van Heerden, J.V., Heath, L., Jourdan-Pineau, H., Martinez-Lopez, B., Pereira De Oliveira, R.P.D., Pollet, T., Quembo, C., Rea, K., Simulundu, E., Taraveau, F. and Penrith, M.L. 2023. An updated review of Ornithodoros ticks as reservoirs of African swine fever in sub-Saharan Africa and Madagascar. Pathogens 12(3), 469. Laanen, M., Maes, D., Hendriksen, C., Gelaude, P., De Vliegher, S., Rosseel, Y. and Dewulf, J. 2014. Pig, cattle and poultry farmers with a known interest in research have comparable perspectives on disease prevention and on-farm biosecurity. Prev. Vet. Med. 115(1–2), 1–9. Lamberga, K., Oļševskis, E., Seržants, M., Bērziņš, A., Viltrop, A. and Depner, K. 2020. African swine fever in two large commercial pig farms in Latvia—Estimation of the high-risk period and virus spread within the farm. Vet. Sci. 7(3), 105. Li, Z., Chen, W., Qiu, Z., Li, Y., Fan, J., Wu, K. and others. 2022. African swine fever virus: a review. Life (Basel). 12(8), 1255. Lichoti, J.K., Davies, J., Kitala, P.M., Githigia, S.M., Okoth, E., Maru, Y., Bukachi, S.A. and Bishop, R.P. 2016. Social network analysis provides insights into African swine fever epidemiology. Prev. Vet. Med. 126, 1–10. Lowe, J., Gauger, P., Harmon, K., Zhang, J., Connor, J., Yeske, P., Loula, T., Levis, I., Dufresne, L. and Main, R. 2014. Role of transportation in spread of porcine epidemic diarrhea virus infection, United States. Emerg. Infect. Dis. 20(5), 872–874. Lozinski, B., Frederick, B., Hilbrands, A., Li, Y., Saqui-Salces, M., Shurson, G.C. and others. 2020. Effects of water quality on nursery pig performance and health. J. Anim. Sci. 98(Suppl. 3), 89. Masembe, C., Adedeji, A.J., Jambol, A.R., Weka, R., Muwanika, V. and Luka, P.D. 2023. Diversity and emergence of new variants of African swine fever virus Genotype I circulating in domestic pigs in Nigeria (2016–2018). Vet. Med. Sci. 9(2), 819–828. Mebus, C.A., McVicar, J.W. and Dardiri, A.H. 1983. Comparison of the pathology of high and low virulence African swine fever infections. In: Wilkinson, P.J., editor. African Swine Fever. Proceedings CEC/FAO Research Seminar, Sardinia, September 1981. EUR 8466 EN. Luxembourg: Commission of the European Communities, pp. 183–194. Meki, I.K., Adedeji, A.J., Ouoba, L.B., Koffi, Y.M., Diakité, A., Settypalli, T.B.K. and others. 2025. Detection of African swine fever virus genotype II in West Africa (2020) and its co-circulation with endemic genotype I: implications for pig production. Transbound. Emerg. Dis. 5396227, 1–14. Montgomery, R.E. 1921. On a form of swine fever occurring in British East Africa (Kenya Colony). J. Comp. Pathol. 34, 159–191. Mulumba-Mfumu, L.K., Saegerman, C., Dixon, L.K., Madimba, K.C., Kazadi, E., Mukalakata, N.T., Oura, C.A.L., Chenais, E., Masembe, C., Ståhl, K., Thiry, E. and Penrith, M.L. 2019. African swine fever: update on Eastern, Central and Southern Africa. Transbound. Emerg. Dis. 66(4), 1462–1480. Nga, B.T.T., Tran Anh Dao, B., Nguyen Thi, L., Osaki, M., Kawashima, K., Song, D., Salguero, F.J. and Le, V.P. 2020. Clinical and pathological study of the first outbreak cases of African swine fever in Vietnam, 2019. Front. Vet. Sci. 7, 392. Niederwerder, M. 2021. Risk and mitigation of African swine fever virus in feed. Animals 11(3), 792. Njau, E.P., Machuka, E.M., Cleaveland, S., Shirima, G.M., Kusiluka, L.J., Okoth, E.A. and Pelle, R. 2021. African swine fever virus (ASFV): biology, genomics and genotypes circulating in sub-Saharan Africa. Viruses 13(11), 2285. Nwachukwu, C.U. and Udegbunam, C. 2020. Rural pig production and pork consumption in Imo State, Nigeria. Niger. J. Anim. Sci. 22(1), 165–185. O Babalobi, O., O Olugasa, B., O Oluwayelu, D., F Ijagbone, I., O Ayoade, G. and A Agbede, S. 2007. Analysis and evaluation of mortality losses of the 2001 African swine fever outbreak, Ibadan, Nigeria. Trop. Anim. Health Prod. 39(7), 533–542. Obayelu, A.E., Ogunmola, O.O. and Sowande, O.K. 2017. Economic analysis and the determinants of pig production in Ogun State, Nigeria. Acta Sci. 50(2), 61–70. Odemuyiwa, S.O., Adebayo, I.A., Ammerlaan, W., Ajuwape, A.T.P., Alaka, O.O., Oyedele, O.I., Soyelu, K.O., Olaleye, D.O., Otesile, E.B. and Muller, C.P. 2000. An outbreak of African swine fever in Nigeria: virus isolation and molecular characterization of the VP72 gene of a first isolate from West Africa. Virus. Genes. 20(2), 139–142. Ogundijo, O.A., Omotosho, O.O., Al-Mustapha, A.I., Abiola, J.O., Awosanya, E.J., Odukoya, A., Owoicho, S., Oyewo, M., Ibrahim, A., Orum, T.G., Nanven, M.B., Bolajoko, M.B., Luka, P. and Adeyemo, O.K. 2023. A multi-state survey of farm-level preparedness towards African swine fever outbreak in Nigeria. Acta Trop. 246, 106989. Olugasa, B.O. and Ijagbone, I.F. 2007. Pattern of spread of African swine fever in south-western Nigeria, 1997–2005. Vet. Ital. 43(3), 621–628. Omotosho, O., Fowowe, O., Abiola, J., Oyagbemi, A. and Omobowale, T. 2024. High environmental temperature induces oxidative stress, reduced sow productivity and increased piglet mortality. J. Appl. Vet. Sci. 9(2), 42–54. Omotosho, O.O. 2020. Why Nigeria’s African swine fever outbreak will hit farmers hard. The Conversation. Available via https://theconversation.com/why-nigerias-african-swine-fever-outbreak-will-hit-farmers-hard-140764 Omotosho, O.O., Morakinyo, O.A., Adedayo, O.O., Odukaye, A.O. and Emikpe, B.O. 2019. Evaluation of the awareness and practices of farmers on gastric ulceration in pigs in the Oyo State, Nigeria. Bangladesh J. Vet. Med. 17(2), 147–153. Omowon, A.A., Daodu, O.B., Omowon, A.M. and Bello, I.I. 2019. Knowledge, attitude and practices of pig farmers post African swine fever outbreaks in Ogun and Oyo states of Nigeria. Sokoto J. Vet. Sci. 17(4), 14–24. Onoja, A.B., Ifeorah, I.M., Jolaoso, M. and Onoja, I.A. 2022. Detection of African swine fever virus genotype II in domestic pigs during a hemorrhagic fever outbreak in Ogun State, Nigeria. Niger. Vet. J. 43(3), 33–41. Otesile, E.B., Ajuwape, A.T.P., Odemuyiwa, S.O., Akpavie, S.O., Olaifa, A.K., Odaibo, G.N. and others. 2005. Field and experimental investigations of an outbreak of African swine fever in Nigeria. Rev. Elev. Med. Vet. Pays. Trop. 58(1-2), 21–26. Owolodun, O.A., Bastos, A.D.S., Antiabong, J.F., Ogedengbe, M.E., Ekong, P.S. and Yakubu, B. 2010. Molecular characterisation of African swine fever viruses from Nigeria (2003–2006) recovers multiple virus variants and reaffirms CVR epidemiological utility. Virus. Genes. 41(3), 361–368. Oyegbami, A., Idowu, A.B., Oluwole, O.O. and Omodewu, I.A. 2024. Knowledge and economic loss of pig farmers to African swine fever in Lagos State, Nigeria. J. Appl. Sci. Environ. Manage. 28(10), 3245–3252. Penrith, M.L. and Vosloo, W. 2009. Review of African swine fever: transmission, spread, and control. J. S. Afr. Vet. Assoc. 80(2), 58–62. Penrith, M.L., Vosloo, W., Jori, F. and Bastos, A. 2013. African swine fever virus in domestic pigs: transmission factors and control options. Vet. Microbiol. 165(1–2), 1–13. Quembo, C.J., Jori, F., Vosloo, W. and Heath, L. 2017. Genetic characterization of African swine fever virus isolates from soft ticks at the wildlife/domestic interface in Mozambique and identification of a novel genotype. Transbound. Emerg. Dis. 420, 431. Robinson, T.P., Wint, G.R.W., Conchedda, G., Van Boeckel, T.P., Ercoli, V., Palamara, E., Cinardi, G., D'Aietti, L., Hay, S.I. and Gilbert, M. 2014. Mapping the global distribution of livestock. PLos One. 9(5), e96084. Rock, D.L. 2021. Thoughts on African swine fever vaccines. Viruses 13(5), 943. Rodrigues Da Costa, M., Gasa, J., Calderón Díaz, J.A., Postma, M., Dewulf, J., Mccutcheon, G. and Manzanilla, E.G. 2019. Using the Biocheck. UGent™ scoring tool in Irish farrow-to-finish pig farms: assessing biosecurity and its relation to productive performance. Porcine Health Manage. 5(4), 4. Sánchez-Cordón, P.I., Montoya, M., Reis, A.L. and Dixon, L.K. 2018. African swine fever: a re-emerging viral disease threatening the global pig industry. Vet. J. 233, 41–48. Sánchez-Vizcaíno, J.M., Mur, L. and Martínez-López, B. 2012. African swine fever: an epidemiological update. Transbound. Emerg. Dis. 59(Suppl. 1), 27–35. Stoian, A.M.M., Zimmerman, J., Ji, J., Hefley, T.J., Dee, S., Diel, D.G., Rowland, R.R.R. and Niederwerder, M.C. 2019. Half-life of African swine fever virus in shipped feed. Emerg. Infect. Dis. 25(12), 2261–2263. United States Department of Agriculture (USDA). 2023. African Swine Fever Case Definition. Available via https://www.aphis.usda.gov/sites/default/files/asf-case-definition.pdf World Organisation for Animal Health (WOAH). 2019. African swine fever (ASF) report No. 27: September 13–26, 2019. Paris: WOAH. | ||
| How to Cite this Article |
| Pubmed Style Abiola JO, Omotosho OO, Aderounmu EA, Ogunlola VA, Adesola RO, Hamzat A, Adebayo DJ, Ayorinde M, Jeremiah OT, Omobowale TO. Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Vet. J.. 2025; 15(12): 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 Web Style Abiola JO, Omotosho OO, Aderounmu EA, Ogunlola VA, Adesola RO, Hamzat A, Adebayo DJ, Ayorinde M, Jeremiah OT, Omobowale TO. Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). https://www.openveterinaryjournal.com/?mno=208504 [Access: January 25, 2026]. doi:10.5455/OVJ.2025.v15.i12.42 AMA (American Medical Association) Style Abiola JO, Omotosho OO, Aderounmu EA, Ogunlola VA, Adesola RO, Hamzat A, Adebayo DJ, Ayorinde M, Jeremiah OT, Omobowale TO. Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Vet. J.. 2025; 15(12): 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 Vancouver/ICMJE Style Abiola JO, Omotosho OO, Aderounmu EA, Ogunlola VA, Adesola RO, Hamzat A, Adebayo DJ, Ayorinde M, Jeremiah OT, Omobowale TO. Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Vet. J.. (2025), [cited January 25, 2026]; 15(12): 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 Harvard Style Abiola, J. O., Omotosho, . O. O., Aderounmu, . E. A., Ogunlola, . V. A., Adesola, . R. O., Hamzat, . A., Adebayo, . D. J., Ayorinde, . M., Jeremiah, . O. T. & Omobowale, . T. O. (2025) Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Vet. J., 15 (12), 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 Turabian Style Abiola, John Olusoji, Oladipo Olufemi Omotosho, Emmanuel Ayopo Aderounmu, Victor Ajibola Ogunlola, Ridwan Olamilekan Adesola, Abdulafees Hamzat, Damilare Johnson Adebayo, Mayowa Ayorinde, Olalekan Taiwo Jeremiah, and Temidayo Olutayo Omobowale. 2025. Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Veterinary Journal, 15 (12), 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 Chicago Style Abiola, John Olusoji, Oladipo Olufemi Omotosho, Emmanuel Ayopo Aderounmu, Victor Ajibola Ogunlola, Ridwan Olamilekan Adesola, Abdulafees Hamzat, Damilare Johnson Adebayo, Mayowa Ayorinde, Olalekan Taiwo Jeremiah, and Temidayo Olutayo Omobowale. "Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021)." Open Veterinary Journal 15 (2025), 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 MLA (The Modern Language Association) Style Abiola, John Olusoji, Oladipo Olufemi Omotosho, Emmanuel Ayopo Aderounmu, Victor Ajibola Ogunlola, Ridwan Olamilekan Adesola, Abdulafees Hamzat, Damilare Johnson Adebayo, Mayowa Ayorinde, Olalekan Taiwo Jeremiah, and Temidayo Olutayo Omobowale. "Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021)." Open Veterinary Journal 15.12 (2025), 6581-6601. Print. doi:10.5455/OVJ.2025.v15.i12.42 APA (American Psychological Association) Style Abiola, J. O., Omotosho, . O. O., Aderounmu, . E. A., Ogunlola, . V. A., Adesola, . R. O., Hamzat, . A., Adebayo, . D. J., Ayorinde, . M., Jeremiah, . O. T. & Omobowale, . T. O. (2025) Field events, clinical findings, and biosecurity breaches in the African swine fever outbreak in Southwest Nigeria (2019–2021). Open Veterinary Journal, 15 (12), 6581-6601. doi:10.5455/OVJ.2025.v15.i12.42 |