| Research Article | ||
Open Veterinary Journal, (2024), Vol. 14(1): 545-552 Original Research Application of some inorganic metal oxide nanoparticles to control E. coli in raw milkAshraf M. Nazem1, Eman K. Abo Shaala1* and Sameh A. Awad21Department of Food Hygiene, Faculty of Veterinary medicine, Alexandria University, Alexandria, Egypt 2Department of Dairy Science and Technology, Faculty of Agriculture, Alexandria University, Alexandria, Egypt *Corresponding Author: Eman K. Abo Shaala. Department of Food Hygiene, Faculty of Veterinary medicine, Alexandria University, Egypt. Email: keman718 [at] gmail.com Submitted: 01/10/2023 Accepted: 15/12/2023 Published: 31/01/2024 © 2024 Open Veterinary Journal
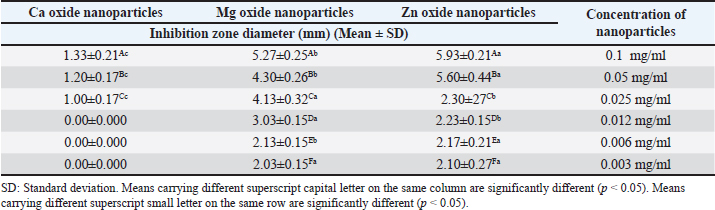
AbstractBackground: Nanoparticles are regarded as magical bullets because of their exclusive features. Recently, the usage of nanoparticles has progressed in almost all aspects of science and technology due to its ability to revolutionize certain fields. In the field of food science and technology, the application of nanoparticles is being researched in many various areas thus provides the dairy industry with a variety of new attitudes for developing the quality, prolong shelf life, ensure the safety and healthiness of foods. Aim: This study aimed to focus on the application of some inorganic metal oxide nanoparticles (zinc oxide (ZnO), magnesium oxide (MgO), and calcium oxide (CaO)) to control E. coli in raw milk and ensure its safety. Methods: The antibacterial action of certain nanoparticles (ZnO, MgO, and CaO) with multiple concentrations (0.1, 0.05, 0.025, 0.0125, 0.006, and 0.003 mg/ml) was evaluated against E. coli strains in ultra heat treated (UHT) milk samples. Also, storage temperature and storage period effects were studied. Results: The findings of the current research revealed that inorganic metal oxide nanoparticles had a significant antibacterial role against E. coli, in the following order; ZnO, MgO, and CaO, respectively. The antibacterial effect of inorganic metal oxide nanoparticles is more noticeable at lower temperatures. Conclusion: Inorganic metal nanoparticles can be used in the food industry for the purpose of the control of E. coli, and extension of the shelf life of dairy products. Keywords: Calcium oxide, E. coli, Magnesium oxide, Nanoparticles, Zinc oxide. IntroductionBecause it contains vital components including protein, carbohydrates, fat, vitamins, minerals, and water, milk is a perfect food source. However, bacteria may grow and multiply with the aid of these nutrients (Yohannes, 2018). Bacterial contamination found in milk has significant health issues that have raised concerns among the general public worldwide due to their potential to harm human health and cause social and economic difficulties. One member of the facultative anaerobic, oxidase-negative, Gram-negative Enterobacteriaceae family of bacteria that ferments glucose to produce acid is Escherichia coli (E. coli). The latter can induce meningitis, sepsis, diarrhea, and urinary tract infections. Based on their virulence agents, different E. coli strains were named and classified. One of the most important food-borne infections that can be spread by milk and dairy products is E. coli (Hasan et al., 2018). According to Zeinhom and Abdel-Latef (2014), E. coli is linked to potentially fatal conditions such as thrombotic thrombocytopenic purpura, hemolytic uremic syndrome, and hemorrhagic colitis (HC). In addition, E. coli is one of the most important organisms that might result in difficulties and potentially fatal issues like bloodstream infections (Ibrahim et al., 2022). More affordable methods for creating nanoparticles with unique physical and chemical properties and restricted resistance have emerged as a result of recent advancements in microbial resistance to numerous antibiotics (Nazoori and Kariminik, 2018). The use of materials smaller than 100 nm is the focus of nanotechnology. In reality, they are used in many fields, such as material science, farming, the dairy sector, cosmetics, medicine, and diagnostics (Siddiqi et al., 2018). Nanoparticles are referred to as "a wonder of modern medicine" these days. Because of their unique chemical and physical characteristics, nanostructures are now used more frequently (Abbas et al., 2020). The most recent advancements in the field of nanoparticle components have a significant impact on the food industry, and nanotechnology has completely changed our social structure and is used extensively in daily life (Siddiqui and Alrumman, 2021). The majority of the many types of nanoparticles found in food are made of inorganic substances including iron oxide, zinc oxide, calcium oxide (CaO), and magnesium oxide (MgO), all of which are safe for ingestion by humans (Pietroiusti et al., 2016). It is yet unknown how exactly nanoparticles work to inhibit bacteria. The antibacterial mechanism of nanoparticles has been explained by a number of mechanisms, including the generation of reactive oxygen species (ROS), which encourage lipid peroxidation in bacteria, the interaction of nanoparticles with bacteria that ultimately results in the destruction of the bacterial cell, and an alkaline impact. According to Tang and Lv (2014), the specific mechanism of action of metal oxide nanoparticles against bacteria is unclear and complex. Worldwide antibacterial agents, zinc oxide nanostructures are thought to be nontoxic and biosafe (Navale et al., 2015). It has been discovered that ZnO nanoparticles selectively poison bacteria while having no negative effects on human health. These findings support the potential uses of ZnO nanoparticles in the dairy industry (Stankic et al., 2016). The Food and Drug Administration (FDA) in the United States has approved zinc oxide as a food additive that extends shelf life and is "generally recognized as safe" (GRAS) (Siddiqui and Alrumman, 2021). Moreover, milk-borne pathogenic bacteria are inhibited by nanoscale MgO (Jin and He, 2011). An antibacterial agent based on light metals that the body can process and completely resorb is the nano-MgO particle. Because MgO nanoparticles can be broken down and metabolized effectively in the body and can actually be removed from the body as long as kidney function is normal, MgO nanoparticles are an attractive alternative to heavy metal-based nanomaterials like silver nanoparticles. This helps to prevent excessive metal accumulation in the body (Nguyen et al., 2018). The US FDA presently lists MgO nanoparticles as safe GRAS and as an addition to human feed. According to Anicˇić et al. (2018), MgO nanoparticles are important inorganic metal oxide nanoparticles with a typical broad band-gap. The most significant aspect of CaO nanoparticles is their environmentally friendly character, which makes them a unique agent for future industrial prospects. They are inexpensive, non-corrosive, and have an antibacterial role and potential heterogeneous catalytic impact (Boey et al., 2021). When used at allowed concentrations, CaO nanoparticles are generally not harmful and remain stable in harsh environments. Because CaO nanoparticles have the ability to generate ROS, which ultimately causes microbial mortality, they may offer a therapeutic approach for inhibiting microbial growth (Kumari et al., 2023). According to Bertu et al. (2010), milk that is meant for human consumption cannot include any harmful bacteria. In order to assess the antibacterial activity of several nanoparticles (ZnO, MgO, and CaO) against E. coli strains in vitro on Muller Hinton Agar media and in vivo in ultra heat treated (UHT) milk samples contaminated with E. coli, the current study was conducted. Furthermore, the impact of storage temperature and duration on the antimicrobial activity of nanoparticles was investigated. Materials and MethodsPreparation of the bacterial strainThe tested E. coli strain was isolated from examined raw milk samples obtained from small holders and biochemically identified by VITEK B Compact (BioMérieux, USA) at Mbarret El Asafra Labs, Alexandria. The molecular identification was carried out in the Reference Laboratory for Veterinary Quality Control in Poultry Production, Animal Health Research Institute, Giza, Egypt. Preparation of nanoparticles (Zn oxide, mg oxide, and ca oxide)For use in photo-electronic applications, ZnO nanoparticles with the NT-ZONP brand and an analytical certificate were purchased from NanoTech Egypt. The nanoparticles were 99.98% pure and had an average size of 30 nm. To achieve a homogenous solution of nanoparticles at different concentrations, distilled water was added to the nanoparticles in glass containers. The particles were then sonicated for 30 minutes to create a colloidal suspension and uniform dispersion. NanoTech Egypt purchased MgO nanoparticles for Photo-Electronics under the NT-MgONP brand, which has an average size of 10 nm and a purity of 99.98%, along with an analytical certificate. To achieve a homogenous solution of nanoparticles at different concentrations, distilled water was added to the nanoparticles in glass containers. The particles were then sonicated for 30 minutes to establish a colloidal suspension and uniform dispersion (Ali et al., 2018). NanoTech Egypt purchased CaO nanoparticles with an average size of 80 nm and a purity of 99.98% for use in photo-electronics, as shown by the NT-CaO brand and an analytical certificate. To achieve a homogenous solution of nanoparticles at different concentrations, distilled water was added to the nanoparticles in glass containers. The particles were then sonicated for 30 minutes to establish a colloidal suspension and uniform dispersion (Ali et al., 2018). Evaluation of antibacterial activity of various metal oxide nanoparticles against the E. coli isolatesMany of the antibacterial activities can be assessed using the agar-well diffusion method (Magaldi et al., 2004; Valgas et al., 2007; Balouiri et al., 2016). Inorganic metal oxide nanoparticle dilutions were made at concentrations of 0.1, 0.05, 0.025, 0.0125, 0.006, and 0.003 mg/ml. The tested isolate's colony suspension was calibrated to meet the 0.5 McFarland criteria. To put it succinctly, the bacterial isolate was cultivated on Plate Count Agar (HiMedia, India) and incubated for twenty-four hours at 37°C. A 0.5 McFarland standard solution was used to compare the four to five isolated colonies of the tested strain that were picked up and injected at a concentration of 1.5 × 108 CFU/mL in tubes containing sterile saline (5 ml in each) (Saeed and Tariq, 2005). To ensure that the isolated bacteria were evenly distributed throughout the surface of a Muller-Hinton agar plate, 100 µl of the bacterial solution was carefully transferred using a micropipette. Subsequently, a sterile cork borer or tip was used to punch an aseptic hole measuring 6 to 8 mm in diameter. A volume of 20–100 µl of the inorganic metal oxide nanoparticle suspensions at varying concentrations was then added to the wells. Agar plates were then incubated for 24 hours at 37°C ± 2°C. The studied microbial strain's growth is inhibited by the antimicrobial agent as it diffuses throughout the agar media. The zones of inhibition were measured and the results were interpreted. Evaluation of antibacterial activity of different metal oxide nanoparticles against the E. coli isolates in UHT milk modelThe study conducted by Mirhosseini and Afzali (2016) involved the application of varying concentrations of different inorganic metal oxide nanoparticle suspensions, such as zinc oxide, MgO, and calcium oxide nanoparticles, to UHT milk model samples that had already been inoculated with a known quantity of E. coli. The aim was to evaluate the effect of inorganic nanoparticles on E. coli. The inoculated milk was then stored at cold storage, while a similar group of inoculated milk was kept at room temperature to ascertain the impact of temperature and storage conditions on the effectiveness of nanoparticles for inhibiting E. coli over a three-day period. Statistical analysisThe data were was presented as means ± SD and subjected to analysis of variance and the Tukey's Kramer HSD test, where a significance level of p < 0.05 was applied. Ethical approvalNot needed for this study. ResultsResults in Table 1 exhibited that the highest concentration (0.1 mg/ml) of ZnO nanoparticles had the widest zone of inhibition (ZOI) against E. coli on Muller Hinton agar media with a ZOI (mm) of 5.93 ± 0.21. ZnO nanoparticles at a concentration of 0.012 mg/ml also exhibited a high inhibition zone 2.23 ± 0.15. A concentration (0.1 mg/ml) of MgO nanoparticles resulted in the widest ZOI against E. coli with 5.27 ± 0.25, additionally a concentration (0.1 mg/ml) of CaO nanoparticles had a ZOI of 1.33 ± 0.21. Table 1. Impact of different concentrations of various nanoparticles on viability of E. coli isolated from the examined milk sample using agar well diffusion method on Muller Hinton agar.
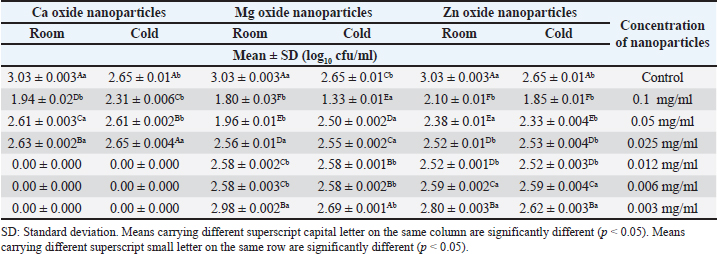
Data given in Table 2 summarize the effect of different concentrations of various nanoparticles on E. coli count inoculated into UHT milk stored at cold storage (4°C ± 1°C) at the 1st day. Data revealed that the most effective concentration of zinc oxide, MgO, and CaO was 0.1 mg/ml with a mean inhibition zone value of 1.85 ± 0.01, 1.33 ± 0.01, 2.31 ± 0.01 (log10 cfu/ml), respectively. Summarized data pointed out the impact of different concentrations of nanoparticles on E. coli inoculated in UHT milk stored at ambient temperature (25°C ± 1°C) on the 1st day. Results showed that ZnO, MgO, and CaO nanoparticles with a concentration of 0.1 mg/ml significantly reduced E. coli counts to 2.10 ± 0.01, 1.80 ± 0.03, and 1.94 ± 0.02. Results mentioned in Table 3 revealed the impact of different concentrations of various nanoparticles on E. coli count inoculated in UHT milk stored at cold storage (4°C + 1°C) on the 2nd day. ZnO, MgO, and CaO nanoparticles at 0.1 mg/ml significantly reduced E. coli counts to 2.75 ± 0.001, 2.52 ± 0.001, 2.76 ± 0.004 (log10 cfu/ml), respectively. The impact of different concentrations of various nanoparticles on E. coli inoculated into UHT milk stored at ambient temperature (25°C ± 1°C) on the 2nd day revealed that 0.1 mg/ml was the most effective concentration that achieved a significant reduction in E. coli counts to 2.78 ± 0.001, 2.69 ± 0.002, 2.60 ± 0.001 log10 cfu/ml, respectively. Also, data in Table 3 illustrated a comparison among different concentrations of various nanoparticles on E. coli inoculated into UHT milk at both cold storage (4°C ± 1°C) and ambient temperature (25°C ± 1°C) on the 2nd day. Table 2. Impact of different concentrations of various nanoparticles on viability of E. coli inoculated in UHT milk stored at (4˚C ± 1˚C) and (25˚C ± 1˚C) for 1st day.
Table 3. Impact of different concentrations of various nanoparticles on viability of E. coli inoculated in UHT milk stored at (4˚C ± 1˚C) and (25˚C ± 1˚C) on the 2nd day.
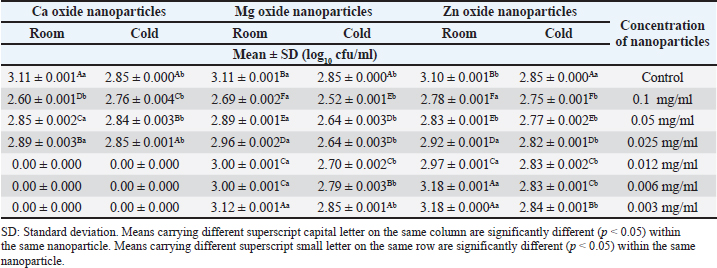
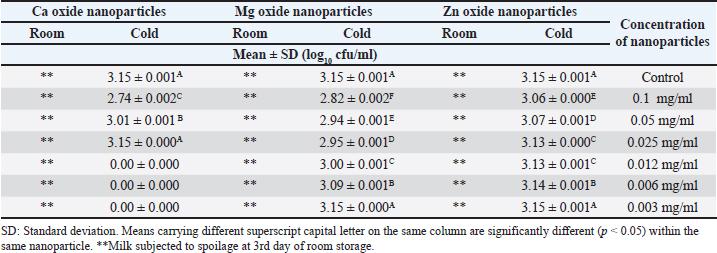
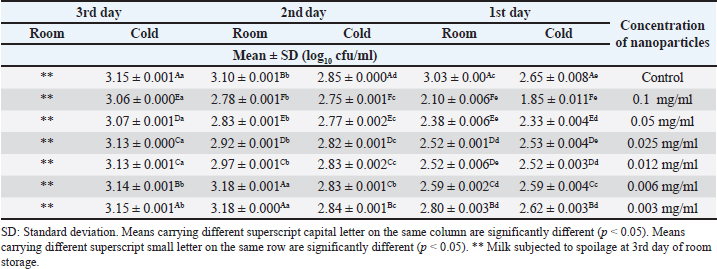
Summarized results in Table 4 showed the effects of different concentrations of various nanoparticles on E. coli inoculated into UHT milk stored at cold storage (4°C ± 1°C) on the 3rd day. Data revealed that the most effective concentration of zinc oxide, MgO, and CaO nanoparticles was 0.1 mg/ml with mean counts of 3.06 ± 0.000, 2.82 ± 0.002, and 2.74 ±0.002 (log10 cfu/ml). Results in Table 5 point out the impact of the storage temperature and zinc oxide nanoparticles concentrations on E. coli inoculated into UHT milk stored over 3 days. DiscussionInorganic metal oxide nanoparticles such as ZnO, CaO, and MgO gain great attention as they are not only constant under strict process, but also their antimicrobial role gains clear importance for their probable application in the dairy branch (Vasilache et al., 2011; Husen, 2017). The obtained results of the current study revealed that the highest concentration of ZnO evolved the best results. ZnO nanoparticles with concentration (0.012 mg/ml) also exhibited a high inhibition zone 2.23 ± 0.153 which is in line with other reports (Vasilache et al., 2011; Bai et al., 2015). They found that 15 μg/ml zinc oxide nanoparticles had a significant effect against E. coli. Zinc oxide nanostructures are global antimicrobial compounds and are regarded bio-safe and nontoxic. The antibacterial efficacy was previously examined using agar well diffusion assay at higher concentrations (compared to our work) (256, 128, 64, 32 mg/ml) of ZnO NPs. The findings of the study revealed a higher reduction in E. coli counts (12.66 ± 0.66 mm) (Navale et al., 2015). In contrast, higher concentrations of ZnO (0.5 and 2 mM) did not exhibit an inhibitory zone on E. coli, according to Mirhosseini and Firouzabadi (2013). In contrast, the inhibition zone against E. coli for 5 and 10 mM of ZnO was 12 mm. The outcomes of our experiment matched those of Abdulrahman and Nssaif (2016). Table 4. Impact of different concentrations of various nanoparticles on viability of E. coli inoculated in UHT milk stored at (4˚C ± 1˚C) and (25˚C ± 1˚C) on the 3rd day.
Table 5. Impact of storage temperature and zinc oxide nanoparticles concentrations on viability of E.coli inoculated in UHT milk stored over 3 days.
Because of their stability, surface characteristics, and structure, MgO nanoparticles play a significant role as antibacterial materials in dairy applications (He et al., 2016). MIC of 500 μg/ml was the antibacterial activity of MgO nanoparticles against E. coli. Because of the existence of oxygen vacancy defects at the surface of the nanoparticles, it was hypothesized that the mechanism of MgO nanoparticles' antibacterial effect would include lipid peroxidation and ROS (Krishnamoorthy et al., 2012). Our results contradicted those of Kumari et al. (2023), who reported that calcium oxide nanoparticles exhibited the maximum inhibition zone against E. coli. To assess the antibacterial activity of CaO NPs against a variety of bacterial strains, including E. coli, the Agar well diffusion method was utilized. A 16.6 mm zone of inhibition was observed for E. coli at doses of CaO NPs (1 mg/ml) (Anantharaman et al., 2016). As the nanoparticle concentration rose, the zone of inhibition progressively widened. It is likely that Sawai (2003) stated that as the quantity of MgO nanoparticles increased, so did their activity against E. coli. It has been demonstrated that inorganic metal oxide nanoparticles, like ZnO, are extremely hazardous to E. coli (Hu et al., 2009). When used at low concentrations, ZnO nanoparticles dramatically prevent E. coli from growing (Hirota et al., 2010). Researchers looked at the mechanism underlying ZnO nanoparticles' antibacterial effectiveness against E. coli. The chemical interactions between hydrogen peroxide and membrane proteins, as well as the chemical interactions between other unknown chemical species produced as a result of ZnO nanoparticles existing within the lipid bi-layer, are found to be the primary mechanisms of such antibacterial efficacy. Under the circumstances of this investigation, it is discovered that the impact of direct physical interactions between nanoparticles and biological cells is comparatively minimal (Zhang et al., 2010). Both the size and concentration of the nanoparticles affect how effective they are against germs. It has been observed that nanoparticle concentration matters more than particle size (Vasilache et al., 2011). The antibacterial role of MgO nanoparticles was almost >3 mg/ml concentration required to observe the destruction effect on E. coli (Jin and He, 2011). These results are extensively higher than our experiment as we demonstrated that the application of 0.1 mg/ml of MgO nanoparticles gave the best significant effect against E.coli compared to control. From the analysis of the antibacterial activity of MgO nanoparticles, it is revealed that 7.5 μg/ml of a dose is sufficient for killing E. coli (Maji et al., 2020) this is parallel to our findings which approved that usage of MgO nanoparticles with concentration (0.006 mg/ml) decreased the count of E. coli from 2.65 ± 0.008 to 2.58 ± 0.002 (log10 cfu/ml) significantly. Likely, ZnO and MgO nanostructures cause a major reduction in E. coli number in the broth medium. A wide group of researchers found that nano-sized ZnO reveals diverse morphologies and exhibits strong antibacterial activity over a broad spectrum of bacterial species (Raghupathi et al., 2011). The highest E. coli bacterial count was seen at the lowest doses of zinc oxide and MgO nanoparticles, which was significantly different from the control. E. coli is susceptible to the antibacterial activity of MgO nanoparticles (Makhluf et al., 2005). The antimicrobial activity of MgO and CaO nanoparticles is dependent on a variety of parameters, including nanoparticle size, shape, biodegradation, high-temperature stability, high surface charge, and surface adsorption ability, all of which can be extremely harmful to biological systems (Dizaj et al., 2014). Cold storage significantly plays a great antimicrobial role in E. coli count compared to room temperature storage. These results are in the same line with data reported before (Lo’Ay and Ameer, 2019) who said that most nanoparticles’ effect was more pronounced at lower temperatures. In general, it has been found that the antibacterial activity of ZnO NPs is increased at lower temperatures. This is likely due to the fact that ROS are more stable at lower temperatures. Additionally, the cell membrane of bacteria is more susceptible to damage at lower temperatures (Qu et al., 2010; Sirelkhatim et al., 2015). Cold storage affects E. coli microbial count compared to the room temperature. Also, most concentrations of ZnO nanoparticles affect E. coli count at cold storage significantly compared to the room temperature. This means that ZnO nanoparticles play a synergistic role with cold storage on E. coli count on the 2nd day of storage. All concentrations of MgO nanoparticles have a great effect on E. coli count together with the aid of cold storage. The storage period can have a vital role in the activity of zinc oxide nanoparticles and in the viability of E. coli due to agglomeration, nanoparticles tend to agglomerate or form clusters over time especially if they are not stored under proper conditions which can reduce their surface area and hinder their reactivity. Surface oxidation, some nanoparticles such as metal-based ones may undergo surface oxidation when exposed to air during storage for an extended period of time; this oxide layer can alter the nanoparticles properties and affect its activity. Contamination and storage conditions might lead to contaminants in the end products. The stability of certain types of nanoparticles is influenced by storage conditions such as temperature and humidity levels, if stored improperly; these particles may degrade or lose their desired properties over time. Particle size distribution changes over long periods of storage, there might be changes in particle size distribution due to various reasons including agglomeration leading to change in catalytic performance (Mukha et al., 2013; Abdou et al., 2012). ConclusionNanoparticles became among the most commonly used preservatives due to their specific chemical and physical features, and their antibacterial properties against milk-containing pathogens as E. coli. According to our study, it can be concluded that ZnO, MgO, and CaO nanoparticles are effective antibacterial agents for E. coli. Conflict of interestThe authors declare that there is no conflict of interest. FundingThis research received no external funding. Author's contributionsAll authors participated in lab work, data analysis, and writing, revising, and approving the manuscript. Data availabilityData of the current study are available upon request. ReferencesAbbas, Z.M.A., Mohsin, I.H. and Ahmade, N. 2020. The biological activity of Zinc oxide and copper oxide nanoparticles against Staphylococcus aurous and Escherichia coli bacteria. Solid State Technol. 63(6), 12957–12968. Abdou, E.S., Osheba, A. and Sorour, M. 2012. Effect of chitosan and chitosan-nanoparticles as active coating on microbiological characteristics of fish fingers. Int. J. Appl. 2(7), 158–169. Abdulrahman, N.B.A. and Nssaif, Z.M. 2016. Antimicrobial activity of zinc oxide, titanium dioxide and silver nanoparticles against mithicillin-resistant Staphylococcus aureus Isolates. Tikrit J. Pure Sci. 21(3), 49–53. Ali, H.M., Babar, H., Shah, T.R., Sajid, M.U., Qasim, M.A., and Javed, S. 2018. Preparation techniques of TiO2 nanofluids and challenges: a review. Appl. Sci. 8(4), 587. Anantharaman, A., Ramalakshmi, S. and George, M. 2016. Green synthesis of calcium oxide nanoparticles and its applications. Int. J. Eng. Res. Afr. 6(10), 27–31. Anicˇić, N., Vukomanović, M., Koklicˇ, T. and Suvorov, D. 2018. Fewer defects in the surface slows the hydrolysis rate, decreases the ROS generation potential, and improves the non-ROS antimicrobial activity of MgO. Small 14(26), 1800205. Bai, X., Li, L., Liu, H., Tan, L., Liu, T. and Meng, X., 2015. Solvothermal synthesis of ZnO nanoparticles and anti-infection application in vivo. ACS Appl Mater Interfaces. 7(2), 1308–1317. Balouiri, M., Sadiki, M. and Ibnsouda, S.K. 2016. Methods for in vitro evaluating antimicrobial activity: a review. J. Pharm. Anal. 6(2), 71–79. Bertu, W.J., Dapar, M., Gusi, A.M., Ngulukun, S.S., Leo, S. and Jwander, L.D. 2010. Prevalence of brucella antibodies in marketed milk in Jos and environs. Afr. J. Food Sci. 4(2), 62–64. Boey, A., Leong, S.Q., Bhave, S. and Ho, H.K. 2021. Cerium oxide nanoparticles alleviate hepatic fibrosis phenotypes in vitro. Int. J. Mol. Sci. 22(21), 11777. Dizaj, S.M., Lotfipour, F., Barzegar-Jalali, M., Zarrintan, M.H. and Adibkia, K. 2014. Antimicrobial activity of the metals and metal oxide nanoparticles. Mater. Sci. Eng. C. 44, 278–284. Hasan, A., Reza, A., Rume, F.I., Meher, M.M., Parvej, M.S. and Anower, A.M. 2018. Prevalence and antibiotic resistance pattern of Escherichia coli isolated from raw dairy milk. TURJAF. 6(9), 1108–1113. He, Y., Ingudam, S., Reed, S., Gehring, A., Strobaugh, T.P. and Irwin, P. 2016. Study on the mechanism of antibacterial action of magnesium oxide nanoparticles against foodborne pathogens. J. Nanobiotechnol. 14, 1–9. Hirota, K., Sugimoto, M., Kato, M., Tsukagoshi, K., Tanigawa, T. and Sugimoto, H. 2010. Preparation of zinc oxide ceramics with a sustainable antibacterial activity under dark conditions. Ceram Int. 36(2), 497–506. Hu, X., Cook, S., Wang, P. and Hwang, H.M. 2009. In vitro evaluation of cytotoxicity of engineered metal oxide nanoparticles. Sci Total Environ. 407(8), 3070–3072. Husen, A. 2017. Gold nanoparticles from plant system: synthesis, characterization and their application. In: Ghorbanpour, M., Manika, K., Varma, A. (eds) Nanoscience and Plant–Soil Systems. Soil Biology, vol 48. Springer, Cham. https://doi.org/10.1007/978-3-319-46835-8_17. Ibrahim, A.H., Ali, M.E., Ahmed, M.F. and Abdelkhalek, A. 2022. Prevalence and characterization of Escherichia coli in raw milk and some dairy products at Mansoura City. J. Adv. Vet. Res. 12(4), 363–370. Jin, T. and He, Y. 2011. Antibacterial activities of magnesium oxide (MgO) nanoparticles against foodborne pathogens. J. Nanopart. Res. 13(12), 6877–6885. Krishnamoorthy, K., Manivannan, G., Kim, S.J., Jeyasubramanian, K. and Premanathan, M. 2012. Antibacterial activity of MgO nanoparticles based on lipid peroxidation by oxygen vacancy. J. Nanopart. Res. 14, 1–10. Kumari, M., Sarkar, B. and Mukherjee, K. 2023. Nanoscale calcium oxide and its biomedical applications: a comprehensive review. Biocatal. Agric. Biotechnol. 47, 102506. Lo’Ay, A.A. and Ameer, N.M. 2019. Performance of calcium nanoparticles blending with ascorbic acid and alleviation internal browning of ‘Hindi Be-Sennara’mango fruit at a low temperature. Sci. Hortic. 25, 199–207. Magaldi, S., Mata-Essayag, S., De Capriles, C.H., Pérez, C., Colella, M.T., Olaizola, C. and Ontiveros, Y. 2004. Well diffusion for antifungal susceptibility testing. IJID. 8(1), 39–45. Maji, J., Pandey, S. and Basu, S. 2020. Synthesis and evaluation of antibacterial properties of magnesium oxide nanoparticles. B Mater. Sci. 43(1), 1–10. Makhluf, S., Dror, R., Nitzan, Y., Abramovich, Y., Jelinek, R. and Gedanken, A. 2005. Microwave-assisted synthesis of nanocrystalline MgO and its use as a bacteriocide. Adv. Funct. 15(10), 1708–1715. Mirhosseini, M. and Firouzabadi, F.B. 2013. Antibacterial activity of zinc oxide nanoparticle suspensions on food-borne pathogens. Int. J. Dairy Technol. 66(2), 291–295. Mirhosseini, M. and Afzali, M. 2016. Investigation into the antibacterial behavior of suspensions of magnesium oxide nanoparticles in combination with nisin and heat against Escherichia coli and Staphylococcus aureus in milk. Food Control. 68, 208–215. Mukha, I.P., Eremenko, A.M., Smirnova, N.P., Mikhienkova, A.I., Korchak, G.I., Gorchev, V.F. and Chunikhin, A.Y. 2013. Antimicrobial activity of stable silver nanoparticles of a certain size. Appl. Biochem. Micro. 49, 199–206. Navale, G.R., Thripuranthaka, M., Late, D.J. and Shinde, S.S. 2015. Antimicrobial activity of ZnO nanoparticles against pathogenic bacteria and fungi. JSM Nanotechnol. Nanomed. 3(1), 1033. Nazoori, E.S. and Kariminik, A. 2018. In vitro evaluation of antibacterial properties of zinc oxide nanoparticles on pathogenic prokaryotes. JABR. 5(4), 162–165. Nguyen, N.Y.T., Grelling, N., Wetteland, C.L., Rosario, R. and Liu, H. 2018. Antimicrobial activities and mechanisms of magnesium oxide nanoparticles (nMgO) against pathogenic bacteria, yeasts, and biofilms. Sci. Rep. 8(1), 16260. Pietroiusti, A., Magrini, A. and Campagnolo, L. 2016. New frontiers in nanotoxicology: gut microbiota/microbiome-mediated effects of engineered nanomaterials. Toxicol. Appl. Pharm. 299, 90–95. Qu, F., Xu, H., Wei, H., Lai, W., Xiong, Y., Xu, F. and Wang, Y.A. 2010. Effects of pH and temperature on antibacterial activity of silver nanoparticles. In 2010 3rd International Conference on Biomedical Engineering and Informatics, IEEE, pp: 2033–2037. Raghupathi, K.R., Koodali, R.T. and Manna, A.C. 2011. Size-dependent bacterial growth inhibition and mechanism of antibacterial activity of zinc oxide nanoparticles. Langmuir. 27(7), 4020–4028. Saeed, S.A.B.A.H.A.T. and Tariq, P.E.R.W.E.E.N. 2005. Antibacterial activities of Mentha piperita, Pisum sativum and Momordica charantia. Pak. J. Bot. 37(4), 997. Sawai, J. 2003. Quantitative evaluation of antibacterial activities of metallic oxide powders (ZnO, MgO and CaO) by conductimetric assay. J. Microbiol. Methods 54(2), 177–182. Siddiqi, K.S., Husen, A. and Rao, R.A. 2018. A review on biosynthesis of silver nanoparticles and their biocidal properties. J. Nanobiotechnology 16(1), 1–28. Siddiqui, S. and Alrumman, S.A., 2021. Influence of nanoparticles on food: an analytical assessment. J. King Saud Univ. Sci. 33(6), 101530. Sirelkhatim, A., Mahmud, S., Seeni, A., Kaus, N.H.M., Ann, L.C., Bakhori, S.K.M. and Mohamad, D. 2015. Review on zinc oxide nanoparticles: antibacterial activity and toxicity mechanism. Nano Lett. 7, 219–242. Stankic, S., Suman, S., Haque, F. and Vidic, J. 2016. Pure and multi metal oxide nanoparticles: synthesis, antibacterial and cytotoxic properties. J. Nanobiotechnology. 14(1), 1–20. Tang, Z.X. and Lv, B.F. 2014. MgO nanoparticles as antibacterial agent: preparation and activity. Braz. J. Chem. Eng. 31(3), 591–601. Valgas, C., Souza, S.M.D., Smânia, E.F. and Smânia Jr, A. 2007. Screening methods to determine antibacterial activity of natural products. Braz. J. Microbiol. 38, 369–380. Vasilache, V., Popa, C., Filote, C., Cretu, M.A. and Benta, M. 2011, February. Nanoparticles applications for improving the food safety and food processing. In 7th International Conference on Materials Science and Engineering–BRAMAT Braşov, ebruary, pp: 24-26. Yohannes, G. 2018. Isolation, identification and antimicrobial susceptibility testing of Escherichia coli isolated from selected dairy farms in and around Mekelle, Tigray, Ethiopia. J. Dairy Vet. Anim. Res. 7, 287–291. Zeinhom, M.M. and Abdel-Latef, G.K. 2014. Public health risk of some milk borne pathogens. Beni-Seuf Univ. J. Appl. Sci. 3(3), 209–215. Zhang, L., Jiang, Y., Ding, Y., Daskalakis, N., Jeuken, L., Povey, M., O’neill, A.J., and York, D. W. 2010. Mechanistic investigation into antibacterial behaviour of suspensions of ZnO nanoparticles against E. coli. J. Nanopart. Res. 12, 1625–1636. | ||
| How to Cite this Article |
| Pubmed Style Nazem AM, Shaala EKA, Awad SA. Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Vet J. 2024; 14((1) (Zagazig Veterinary Conference)): 545-552. doi:10.5455/OVJ.2024.v14.i1.49 Web Style Nazem AM, Shaala EKA, Awad SA. Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. https://www.openveterinaryjournal.com/?mno=182429 [Access: July 12, 2025]. doi:10.5455/OVJ.2024.v14.i1.49 AMA (American Medical Association) Style Nazem AM, Shaala EKA, Awad SA. Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Vet J. 2024; 14((1) (Zagazig Veterinary Conference)): 545-552. doi:10.5455/OVJ.2024.v14.i1.49 Vancouver/ICMJE Style Nazem AM, Shaala EKA, Awad SA. Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Vet J. (2024), [cited July 12, 2025]; 14((1) (Zagazig Veterinary Conference)): 545-552. doi:10.5455/OVJ.2024.v14.i1.49 Harvard Style Nazem, A. M., Shaala, . E. K. A. & Awad, . S. A. (2024) Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Vet J, 14 ((1) (Zagazig Veterinary Conference)), 545-552. doi:10.5455/OVJ.2024.v14.i1.49 Turabian Style Nazem, Ashraf M., Eman K. Abo Shaala, and Sameh A. Awad. 2024. Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Veterinary Journal, 14 ((1) (Zagazig Veterinary Conference)), 545-552. doi:10.5455/OVJ.2024.v14.i1.49 Chicago Style Nazem, Ashraf M., Eman K. Abo Shaala, and Sameh A. Awad. "Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk." Open Veterinary Journal 14 (2024), 545-552. doi:10.5455/OVJ.2024.v14.i1.49 MLA (The Modern Language Association) Style Nazem, Ashraf M., Eman K. Abo Shaala, and Sameh A. Awad. "Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk." Open Veterinary Journal 14.(1) (Zagazig Veterinary Conference) (2024), 545-552. Print. doi:10.5455/OVJ.2024.v14.i1.49 APA (American Psychological Association) Style Nazem, A. M., Shaala, . E. K. A. & Awad, . S. A. (2024) Application of some inorganic metal oxide nanoparticles to control E. coli in raw milk. Open Veterinary Journal, 14 ((1) (Zagazig Veterinary Conference)), 545-552. doi:10.5455/OVJ.2024.v14.i1.49 |