| Research Article | ||
Open Veterinary Journal, (2024), Vol. 14(1): 470-480 Original Research Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogensSally S. Fathy1, Esmat I. Awad2, Salah F. A. Abd-El Aal2, Eman N. Abdelfatah2 and Asmaa B. M. B. Tahoun2*1Directorate of Veterinary Medicine in Dakahlia, Ministry of Agriculture, Egypt 2Food Control Department, Faculty of Veterinary Medicine, Zagazig University, Zagazig City, Egypt *Corresponding Author: Asmaa B.M.B. Tahoun. Food Control Department, Faculty of Veterinary Medicine, Zagazig University, Zagazig City, Egypt. Email: abbadr [at] vet.zu.edu.eg © 2024 Open Veterinary Journal
AbstractBackground: Bacillus cereus and Yersinia enterocolitica are implicated in foodborne diseases that have major effects on human health; therefore, it is considered universal public health disorders. Essential oils and essential oils nano emulsions have a sufficient antibacterial performance against a variety of bacteria, especially multi-drug resistant bacteria. Probiotics showed several health benefits via moderating the GIT microbiota and their metabolites. Aim: The study was designed to evaluate the biocontrol ability of cinnamon essential oil (CEO) nano emulsion and probiotics as natural antibacterial additives and reveal their bactericidal mechanism. Methods: 250 random samples (50 raw milk, 50 rice pudding, 50 kariesh cheese, 50 yogurt, and 50 ice cream) were purchased separately from different areas in Mansoura city, Egypt, and exposed to bacteriological analysis. Results: Bacillus cereus was found with the highest mean value of 66 × 107 ± 1.3 × 108 CFU/g in raw milk and the lowest mean value of 28 × 107 ± 2.6 × 107 CFU/g in kariesh cheese while Y. enterocolitica was found in 64% of the total inspected samples with the highest incidence (84%) in yogurt. The toxinogenic potential of the tested pathogens has been evaluated by multiplex PCR pointing nhe A and ces genes for B. cereus isolates while targeting in Y. enterocolitica 16s rRNA, and YST gene. Different concentrations (0.17%, 0.25%, 0.5%, 0.8%, 1%, 1.5%, and 2%) of cinnamon oil nano emulsion were employed in this study. CEO nano emulsion had the highest reduction rate at a concentration of 1.5% in the case of B. cereus and 2% in the case of Y. enterocolitica. Among different types of probiotics, the best one which showed inhibitory potential against B. cereus and Y. enterocolitica was L. plantarum. Conclusion: Lactobacillus plantarum and CEO nano emulsion at a concentration of 2% have the highest reduction rate against Y. enterocolitica, while L. plantarum and CEO nano emulsion at a concentration of 1.5% has the best antibacterial effect against B. cereus. In conclusion, more attention is required for both safety and quality in dairy products through the application of natural additives such as essential oils and probiotics. Keywords: Essential oils, Cinnamon oil, Bacillus cereus, Y. enterocolitica, L. plantarum. IntroductionBacillus cereus is considered one of the utmost common food-borne pathogens in foodstuffs (Rahnama et al., 2023). They were mentioned as “aerobic endospore-forming micro-organisms” which broadly blow out over the world (Zhou et al., 2023). The pathogenicity of this organism rests on frequent exogenic enzyme creation, the capability of forming biofilms, and the existence of toxin-encoding genes. It is related to a food poisoning syndrome characterized by diarrhea and gastrointestinal disturbances (Li et al., 2023). Bacillus cereus ranked as the fourth greatest reason for foodborne diseases by EFSA and ECDC in the European Union (Elafify et al., 2023). Yersinia enterocolitica is involved in different human disorders such as gastro-enteritis, sepsis, appendicitis, and lymphadenitis (Ahmed et al., 2023). Yersiniosis as a gastrointestinal contagion comes afterward campylobacteriosis and salmonellosis (Zadernowska et al., 2014) demonstrated by lymphadenitis, diarrhoea, appendicitis, and arthritis (Tavassoli et al., 2019). Yersinia enterocolitica resists numerous antibacterial agents and is considered one of the pathogenic drug-resistant bacteria with a public health importance (Sadek et al., 2014; Bonardi et al., 2018). Food additives that are used in the food industry to prolong shelf life may cause intoxication, and severe diseases as cancer. To avoid this problem, the application of natural extracts from herbs to limit the growth of micro-organisms might be a practical solution (El-Sayed and El-Sayed, 2021) Lactic acid bacteria are commonly reflected as probiotics which have great health importance via moderating the metabolites and microbiota (Fang et al., 2023). Therefore, they were used as preservatives in the food chain (Hamad et al., 2022). Lactobacillus plantarum has an aggressive influence against food micro-organisms (Tian et al., 2022). Finally, efficient foods or probiotic-supplemented food received more attention because of the awareness of healthy and beneficial products targeting good wellbeing (Ali et al., 2023; Hussain et al., 2023). The number of cells per milliliter and its therapeutic doses determine the probiotic potential (Begum et al., 2017). A probiotic food should not exceed 1.0 × 106 cfu/g of alive microbes. In accumulation to being a biopreservative, EOs mentioned as generally recognized as safe due to their effective antibacterial role in the food industry (Es et al., 2017). They can inhibit spoilage and pathogenic bacteria through their phenolic constituents and polypeptides (Bakhtiary et al., 2018) and improve nutritive value and organoleptic properties (Kholif and Olafadehan, 2021). Among them, cinnamon essential oil (CEO) has been used as an antifungal and antimicrobial agent (Paudel et al., 2019) due to the presence of cinnamic acid, eugenol, and cinnamaldehyde (Siripatrawan, 2016). It has broad-spectrum antimicrobial activities; in addition, it is characterized by its special aroma and medicinal properties (Nazzaro et al., 2013). Nano emulsions are biphasic dispersions of water and oil either water in oil microemulsions or oil in water microemulsions or (O/W), alleviated by an emulsifier. The exceptional features of these nanostructures diverge from their parent structures chemically, biologically, and physically. This potential request of nanotechnology is to prolong shelf-life, nutritional additives, coloring, flavoring, and food safety via using antimicrobial ingredients for food (Oladipo et al., 2020). Therefore, this study examined the preservative action of CEO nano emulsion and probiotics and how those could affect pathogenic bacteria. Materials and MethodsCollection of samplesTwo hundred and fifty samples of raw milk, yogurt, kariesh cheese, ice cream, and rice budding (50 each) were purchased randomly from different markets at localities in Dakahlia Governorate, Egypt, from September 2022 to March 2023. In sterile containers, samples were collected and transferred immediately to an ice box for isolation and examination. Microbiological analysisPreparation of samples Samples collected were 25 ml of milk and 25 g of yogurt, kareish cheese, rice pudding, and ice cream then homogenized in a sterile jar containing 225 ml of tryptone soya broth. Kareish cheese was mixed with 2% sodium citrate to accelerate homogenization (Salfinger, 2015). Isolation and quantification of B. cereus It was done through duplicates of BcSAB plates with polymyxin B supplement first then add the egg yolk emulsion. Incubated at 35°C for 24–48 hours. Suspected colonies of B. cereus appeared crenate, about 5 mm in diameter, and had a unique turquoise to peacock blue color. Then the recovered colonies were counted and calculated as the total B. cereus count. Three to five typical colonies were purified on TSA slants and incubated at 35°C for 24 hours (Oxoid, 2002). Identification of Y. enterocolitica Pre-enrichment of the sample (1 ml) by 10 ml of PBS supplemented with cefsulodin and novobiocin is necessary. Tubes were incubated at room temperature for 5–7 days. Streaking on selective CIN agar plates. Then incubated at 37°C for 48 hours. Characteristic colonies appeared as bull's eyes (Drake et al., 2018). Molecular confirmation of isolate By following manufacturer instructions, the DNA of Bacillus and Yersinia isolates have been extracted by QIAamp DNA Mini Kit. Amplification of the specific bacterial targets was done by PCR using DreamTaq™ Green PCR Master Mix (2×), Emerald Amp GT PCR master mix (TaKaRa, Japan), respectively (Koua et al., 2014; Owusu-Kwarteng et al., 2017) (Table 1). Table 1. Oligonucleotide sequences of the primers used in the present study.
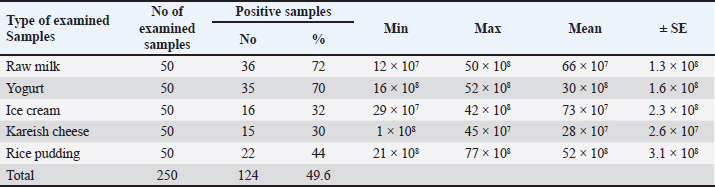
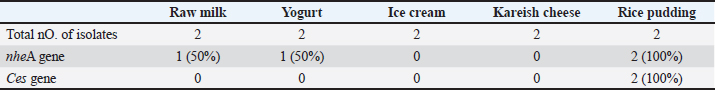
Preparation of essential oils nano emulsion Colloidal nano suspension from cinnamon seeds was prepared by ethanolic extract using solvent evaporation/micelles formation with some modification (Singh et al., 2023). CEO nano emulsions with concentrations of 0.17%, 0.25%, 0.5%, 0.8%, 1%, 1.5%, and 2% (v/v) for each were purchased from Nakaa Nanotechnology Network. Experiments and interpretation were done according to previous studies (NCCLS 2012; Kaskatep et al., 2016). Antimicrobial activity analysis Disc diffusion was done by using Mueller-Hinton agar following the guidelines of CLSI (2015, 2017). Then bacteria were spread on the plate with 1.5 × 107 CFU/ml density according to Lahtinen et al. (2007. Ampicillin (AM 10 µg), ciprofloxacin (Cip), amoxicillin-clavulanic acid (AMC 20/10 µg, and erythromycin (E15) discs were tested. Antibacterial activity of different probiotic strainPreparation of probiotic Lactobacillus plantarum, L. rhamnosus, and L. acidophillus were evaluated via agar well diffusion method following previous studies (Hamad et al., 2023). Wells (6 mm) were bored on the plates and 100 µl free supernatant containing antibacterial activity was added, ampicillin was used as a positive control, while the nutrient broth was utilized as a negative control. Incubation was done for 24 hours at 37°C and the inhibition zone was measured and a clear zone was noted in mm referring to the antibacterial influence of different probiotic strains. Antibacterial activity of CEO nano emulsion Different concentrations of CEO nano emulsion were presented in wells that were swabbed before with overnight cultures of bacteria. The test was done on triplicates as probiotic inoculation (Hulankova, 2022). Statistical analysisData were represented as Mean ± SD. ANOVA was done with SPSS and statistical significance was estimated at p < 0.05 (Feldman e t al., 2003). Ethical approvalNot required for this study. ResultsOccurrence of B. cereus in the examined samplesBacillus cereus was found in milk, yogurt, ice cream, kareish cheese, and rice pudding samples by 72%, 70%, 32%, 30%, and 44%, respectively (Table 2). Although nheA and ces genes were detected at 100% of rice pudding, nheA gene were found only at 50% of yogurt and raw milk as mentioned in Tables 3–5 and Figure 1. Table 2. Prevalence of B. cereus in the examined samples.
Table 3. Molecular identification of certain B. cereus among the examined samples.
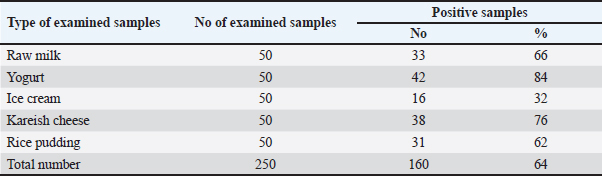
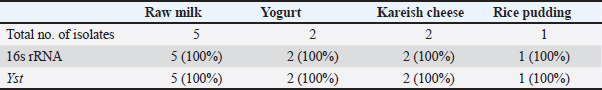
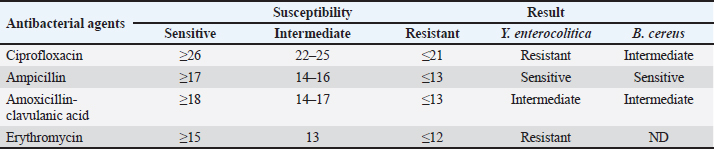
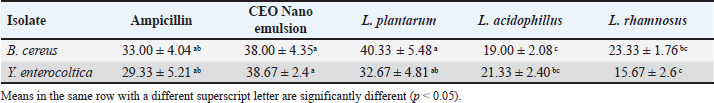
Occurrence of Y. enterocolitica and virulence genesYersinia enterocolitica was found in 160 samples out of 250 (64%) with the highest incidence of 84% followed by 76%, 66%, 62%, and 32% in yogurt, kariesh chees, raw milk, rice pudding, and ice cream, respectively, as shown in Table 4. Both 16s rRNA and yst gene were detected in 100% of tested isolates as illustrated in Table 5 and Figures 2 and 3. Antibacterial susceptibility against B. cereus and Y. enterocoltica isolatesBacillus cereus isolates were highly sensitive to ampicillin, middle sensitive to Amoxicillin-clavulanic and ciprofloxacin with no effect of erythromycin, while, Y. enterocolitica was highly sensitive to ampicillin, intermediate to Amoxicillin-clavulanic, and resistant to ciprofloxacin and erythromycin as depicted in Table 6. Antibacterial activity of probioticBacillus cereus showed high sensitivity to L. plantarum, intermediate L. acidophillus then L. rhamnosus with 40.33 ± 5.48, while in Y. enterocoltica L. plantarum showed the strongest antibacterial activity with DIZs 38 mm, followed by L. acidophillus and L. rhamnosus (Table 7). Table 4. Prevalence of Yersinia spp. in the examined samples.
Table 5. Molecular identification of Y. enterocolitica among the examined samples.
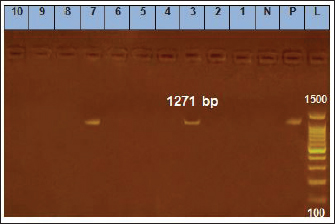
Fig. 1. Amplification of B. cereus isolates on agarose gel using ces (1,271 bp). Lane (1–10) lane ladder (100–1,500) B. cereus lanes 3 and 7 show positive results while other lanes show no expression.
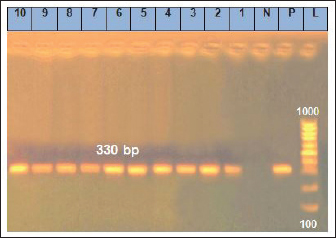
Fig. 2. Amplification of Y. enterocoltica isolates on agarose gel using 16srRNA (330 bp). Lane (1–10) Y. enterocoltica isolates shows positive results. Antibaterial activity of CEO nano emulsion against B. cereus and Y. enterocoltica isolatesBacillus cereus shows high sensitivity for 1.5% CEO nano emulsion reaching 38.00 ± 4.35, while Y. enterocoltica showed inhibition at 2% with 38.67 ± 2.4. p < 0.05 showed a significant decrease in values (Mean ± SE) in Table 7 which assumed that L. plantarum, 1.5% CEO, and 2% have a more powerful effect against the examined isolate. DiscussionDairy foods contaminated by contagious microorganisms are considered the most critical matter of human-being health and safety worldwide. Spoilage of dairy foodstuffs is highly risky because of the ability of raw milk and its dairy products to accelerate progression. Bacillus cereus is recognized as a significant pathogenic microorganism that is characterized by diarrheal and emetic syndrome (Radmehr et al., 2020; Han et al., 2023). Thus, B. cereus is a significant microbe that threatens human beings whether their toxins are present or not. In comparison with our findings, Hefny et al. (2020) and Osama et al. (2020) isolated B. cereus from 40% of tested Kareish cheese while (Heikal and Al-wakeel, 2014; Ibrahim et al., 2015) failed to isolate B. cereus in their examined cheese samples. In opposite to our result, Adam et al. (2021) and Fetouh et al. (2022) indicated that the occurrence of B. cereus in yogurt samples was at 8.0%, and 4.0%, respectively, while it could not be detected in yogurt samples by Tirloni et al. (2017). The absence of growth or even low incidence observed might be owing to the low pH < 5. Lower results were reported by Owusu-Kwarteng et al. (2017) who found that B. cereus is isolated at 47.00% from raw cow milk in Ghana. Also, Mohamed et al. (2016) declared that B. cereus was found in 60.00% of raw cow milk samples. Abraha et al. (2017) reported that B. cereus rate exists at 8.80% in raw cow milk samples in Ethiopia.
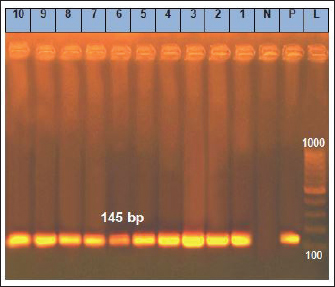
Fig. 3. Amplification of Y. enterocoltica isolates on agarose gel using YST (145bp). Lane (1–10) Y. enterocoltica isolates show positive results. Table 6. Antimicrobial sensitivity test for the isolated B. cereus and Y. enterocolitica.
Table 7. Antibacterial activities of different probiotic cell free supernatant (CFS) and CEO nano emulsion against B. cereus and Y. enterocolitica according to control positive inhibition zone detected by mm.
Although B. cereus was detected at 32% in ice cream, a higher rate (62.7%) was defined by Messelhäusser et al. (2010). Consequently, variation in B. cereus rate is owing to different bacterial loads and poor hygiene standards during the preparation and production process. Bacillus cereus is recurrently isolated from raw milk because of pasteurization process resistance (Tirloni et al., 2017). According to our result, B. cereus was isolated at 22 (44%) of the examined rice pudding and this disagreed with Morsy et al. (2022) who isolated B. cereus at 84% in rice pudding samples. The same findings were defined before (Mohamed et al., 2016; Amin, 2017; El-Sherif et al., 2021). While, higher results were also reported (Hussein et al., 2015; El-Karamani, 2017; El-Zmakan and Mubarak, 2017; El-Sherif et al., 2021). Our study discussed the mechanism of food contamination with B. cereus. Certainly, the main source of contamination is poor sanitation and cross contamination. Overall, the raw ingredients and the post-pasteurization contamination might increase the prevalence of B. cereus. This concluded that firm cleaning administration must be approved to control B. cereus with the aim of assuring high quality and safety products. According to our result, nheA gene was found in 50% of B. cereus isolated from each raw milk and yogurt. Higher occurrence rates were recorded by Eltokhy et al. (2021) who detected nheA in all isolates. Fetouh et al. (2022) stated that non-hemolytic (nheA) and cytotoxic K (cytK) genes were involved in food poisoning. Hefny et al. (2020) who failed to detect ces gene, detected cytk in 47% of all isolated B. cereus while, nhe and hbl were detected at 52% and 33%, respectively. Bianco et al. (2020) detected sich cytotoxic genes in 35%–100% of their isolated strains. Bacillus cereus revealed high sensitivity to ampicillin followed by amoxicillin, clavulanic, and ciprofloxacin with high resistance toward erythromycin and this might be due to the misuse of antibiotics in dairy farms. This finding differs from Ahmed et al. (2023) who said that all examined isolates were 100% sensitive to erythromycin, 100% resistant to tetracycline, and intermediate sensitive to ciprofloxacin. EFAS stated that probiotics to be used in food stuff; it should not have antimicrobial resistance genes (EFSA, 2007). Yersinia spp., is capable of growing in both anaerobic and aerobic situations. Nguyen et al. (2019) concluded that Y. pseudotuberculosis, Y. enterocolitica, and Y. pestis are pathogenic and transferred through contaminated milk. Yersiniosis is stated as a zoonotic syndrome with a public health importance. EFSA (2009) revealed that Y. enterocolitica in the whole European Union is considered one of the most informed zoonoses. Yersinia enterocolitica proliferated in the lower small intestine and upper large intestine, pusjing the organism establishment (Chlebicz and Śliżewska, 2018). Consequently, Yersiniosis causes gastric infection because of the improper handling and cooking of animal-origin foods during preparation (Tavassoli et al., 2018). Yersinia was detected in 64% (160 out of 250) of the examined products. This result was classified as following: 33(66%) raw milk, 42(84%) yogurt samples, 16(32%) ice cream samples, 38 (76%) kariesh cheese samples, and 31(62%) from rice pudding. However, Gṻven et al. (2010) isolated Y. enterocolitica from raw milk at 1.33%, while Darwish et al. (2015) isolated it at 38.5%. Hassan and Afify (2007) secluded the lowest incidence at 2% in Kareish cheese, while Basyoni and Elsheikh (2005) isolated it by 16.67%, and Basha et al. (2008) isolated it at 14%. A higher incidence of Y. enterocolitica was reported in kariesh cheese and yogurt at 60% and 84%, respectively. This might be due to poor hygiene during manufacture. Basha et al. (2008) confirmed that gathering Kareish cheese samples from shops and street retailers could clarify the poor quality. In small-scale ice cream, Gṻven et al. (2010) recorded the lowest incidence of Y. enterocolitica at 2.67% and Khalifa et al. (2007) could isolate it at 12.5%. Nearly similar results have been recorded by AlShammary and Madi (2016) who isolated it with a percentage of 30%. Being psychrotrophic bacteria elevates their prevalence in ice cream which affect badly on cold food chain. Similar results were recorded by Harakeh et al. (2012), and Ali and Al-Samarai (2020) who isolated Y. enterocolitica at 12%, and 9.75%; respectively. Yersinia enterocolitica was isolated at 5.78%, 5.3%, and 3.3%, respectively (Ali et al., 2015; Ozdemir and Arslan, 2015). While, higher incidence was recorded by Darwish et al. (2015) and Ahmed et al. (2019) at 46%, and 22%, respectively, whereas Zeinhom and Abdel-Latef (2014) could not isolate it. PCR was able to identify pathogenic strain genes in the isolates. We focused on virulence-associated genes of 10 isolates of Y. enterocolitica. These genes were 16S rRNA for detection and yst for virulence. PCR results declared that genes were found in all isolates. Younis et al. (2019) detected tetA and blaTEM genes in all eight isolates. On the other hand, it is remarkable that the YST gene cannot be detected (Peruzy et al., 2017). Yersinia enterocolitica revealed high resistance to ciprofloxacin, and erythromycin. Meanwhile, they were intermediate sensitive to AMC. Moreover, they were highly sensitive to ampicillin. While Abdelwahab et al. (2021) reported that Y. enterocolitica was highly sensitive to norfloxacin and meropenem (79.0% for each). Then, gentamycin was recovered at 68.4% and middle sensitive to doxycycline by 63.2%. These results demonstrated that Y. enterocolitica had multiple antibiotic resistances. Thus, we tried to demonstrate other alternatives as natural probiotics and essential oils. The misapplication of the antibacterial agents could raise the bacterial resistance. As B. cereus and Y. enterocolitica were examined against several antibiotics. All tested isolates showed high resistance levels. Probiotics that are applied in some foodstuffs and pharmaceutics are regarded to be safe, and they should not have transportable antibiotic-resistance genes (EFSA, 2007). Montassier et al. (2021) stated that antibacterial potential is a critical feature to assess the use of probiotics in the food industry (Aditya et al., 2020). According to the current study, L. plantarum is characterized by powerful activity against B. cereus and Y. enterocolitica strains. Lactobacillus plantarum (MK850930) showed inhibition zones of 40 mm in B. cereus and 38 mm in Y. enterocolitica. Ahmed et al. (2023) said that L. plantarum has an antimicrobial effect toward B. cereus EMCC1006. These studies disagreed with Yusra and Likaa (2013) who reported that in the case of B. cereus, L. plantarum MIC was 0.07 ml. Approaches of probiotics might successfully inhibit pathogens such as L. monocytogenes (Wu et al., 2022), other studies stated that L. casei IMAU60214 was effective against Escherichia coli (Rocha-Ramírez et al., 2023). Similarly, L. fermentum LBF433 and L. casei LBC 237 have sufficient action against Salmonella (Lando et al., 2023). Our study disclosed that L. plantarum had a significant inhibitory action on foodborne pathogens such as Y. enterocolitica and B. cereus. The main effect of the essential oils was attributed to their antimicrobial properties, and the ability to dissolve the cytoplasmic membrane of the bacterial cells (Kaskatep et al., 2016; Ferrari et al., 2012). One of the approaches to deal with these hydrophobic compounds is by dispersing them in nano emulsion delivery system (Singh et al., 2023). Dávila-Rodríguezet et al. (2019) reported that one of Eos disadvantages on food is its flavor effect in food products. To solve these issues, CEO can be encapsulated in nano emulsions to increase the stability, solubility, and potential activity of CEO (Akhavan et al., 2018). Paudel et al. (2019) stated that CEO has a proper source of antifungal and antimicrobial compounds. This activity is basically linked to eugenol, cinnamaldehyde, and cinnamic acid (Siripatrawan, 2016). In comparison with Eos nonencapsulation, nanoemulsions were extra active against bacteria, needing less than 50% EOs to decrease 5-log bacterial count. Numerous studies discussed the effectiveness of CEO against food-borne pathogens (Cava-Roda et al., 2010; Aliakbarlu et al., 2013). CEO nano emulsion has effective power on food-borne diseases. In our study, B. cereus was found to be delicate to CEO nano emulsion 0.5% with IZD 34 mm, Y. enterocolitica sensitive to CEO nano emulsion 2%. Gupta et al. (2008) reported that cinnamon oil is very supportive against Bacillus spp. with MIC 1.25%. Azadi et al. (2023) concluded that CEO nano emulsion have a notifiable activity toward Staphylococcus aureus and B. cereus more than S. typhimurium and E. coli (O157:H7). Sharma et al. (2022) stated that the CEO was the most operative as an antimicrobial agent. It is recognized by the presence of cinnamaldehyde. Cinnamaldehyde is an effective natural antioxidant that prevents stomach ulcers, preventing both strains of Helicobacter pylori, also it can be inhibited through the growth of yeast, molds, and bacteria (Basak et al., 2021). Oregano, basil, rosemary, and thyme were branded as the most favorable active antimicrobial EOs against Y. enterocolitica recently which deserve further studies (Durofil et al., 2022). It was found that Y. enterocolitica was sensitive to CEO nano emulsion 2% in our study. Hulankova (2022) concluded that Y. enterocolitica and Y. pseudotuberculosis are multi resistant bacteria against cinnamon oil by MICs (median 414 and 207 μg /ml, respectively). Koua et al. (2014) referred to the importance of micro-organisms not to acquire resistance toward essential oils. CEO contains cinnamaldehyde (68.79%) which was proven to have high antibacterial activity against G + ve bacteria (Es et al., 2017). Yersinia enterocolitica was the most sensitive bacteria to Cinnamomum cassia EOs with a zone of inhibition (16.67 mm). Klūga et al. (2021) examined 14 types of Eos against Y. enterocolitica and found extreme action against pathogenic microbes. ConclusionEntero-pathogenic Yersinia and B. cereus showed a great resistance level toward some antibiotics. Other approaches were used effectively against Y. enterocolitica and B. cereus such as L. plantarum which have a functional inhibition potential. CEO was effective against both Yersinia and Bacillus spp. Therefore, EOs and probiotics can be hopeful alternate inhibitors for these multiresistance strains. AcknowledgmentsAll appreciation to the Food Control Department, Faculty of Veterinary Medicine, Zagazig University. Conflict of interestThe authors declare that there is no conflict of interest. FundingNo external funding has been provided. Authors contributionsAll authors contributed to his study. All authors read and approved the final manuscript. Data availabilityAll data are provided in the manuscript. ReferencesAbdelwahab, A. M., El-Tawab, A., Awad, A., Abdallah, F. and Maarouf, A. A. 2021. Phenotypic and genotypic studies on antibiotic resistant Yersinia enterocolitica isolated from milk and milk products in Kaliobia, Egypt. Benha Vet. Med. J. 40(2), 149–153. Adam, A. H., Aly, S. A. and Saad, M. F. 2021. Evaluation of microbial quality and safety of selected dairy products with special focus on toxigenic genes of Bacillus cereus. Mljekarstvo 71(4), 257–268. Aditya, A., Peng, M., Young, A. and Biswas, D. 2020. Antagonistic mechanism of metabolites produced by Lactobacillus casei on lysis of enterohemorrhagic Escherichia coli. Front. Microbiol. 11, 574422 Ahmed, H. A., Tahoun, A. B., Abou Elez, R. M., Abd El-Hamid, M. I. and Abd Ellatif, S. S. 2019. Prevalence of Yersinia enterocolitica in milk and dairy products and the effects of storage temperatures on survival and virulence gene expression. Int. dairy. J. 94, 16–21. Ahmed, M.A., Elsisy, S.F. and Selim, A.O. 2023. Genotypic and phenotypic variation of Yersinia enterocolitica isolated from different sources. J. Adv. Vet. Res. 13(6), 1197–1202. Ali, A., Javaid, M. T., Tazeddinova, D., Khan, A., Mehany, T. and Djabarovich, T. A. 2023. Optimization of spray dried yogurt and its application to prepare functional cookies. Front. Nutr. 10, 1186469. Al-Nabulsi, A. A., Awaisheh, S. S., Osaili, T. M., Olaimat, A. N., Rahahaleh, R. J., Al-Dabbas, F. M., Al-Kharabsheh, L. A., Gyawali, R. and Ibrahim, S. A. 2015. Inactivation of Cronobacter sakazakii in reconstituted infant milk formula by plant essential oils. J. Appl. Bot. Food. Qual. 88(1), 97–101. Al-Shammary, A. and Madi, H. 2016. Prevalence of Yersinia enterocolitica in ice-creams in Baghdad. Med. Sci. 5 (6), 2277–8160. Amin, W. F. 2017. Occurrence of Bacillus Cereus in some milk-based desserts. Assiut. Vet. Med. J. 64 (156), 41––46. Angmo, K., Kumari, A. and Bhalla, T. C. 2016. Probiotic characterization of lactic acid bacteria isolated from fermented foods and beverage of Ladakh. LWT 66, 428–435. Azadi, A., Rafieian, F., Sami, M. and Rezaei, A. 2023. Fabrication, characterization and antimicrobial activity of chitosan/tragacanth gum/polyvinyl alcohol composite films incorporated with cinnamon essential oil nanoemulsion. Int. J. Biol. Macromolecul. 245, 125225. Bakhtiary, F., Sayevand H. R., Khaneghah A. M., Haslberger A. G. and Hosseini H. 2018. Antibacterial efficacy of essential oils and sodium nitrite in vacuum processed beef fillet. Appl. Food. Biotechnol. 5(1), 1–10. Basak, S., Singh, J. K., Morri, S. and Shetty, P. H. 2021. Assessment and modelling the antibacterial efficacy of vapours of cassia and clove essential oils against pathogens causing foodborne illness. LWT 150, 112076. Basha, O., El Shaboury, F. and Fayed, A. 2008. Some studies on the occurrence of Yersinia microorganisms in raw milk and some soft cheeses sold in Alex. Govern. Assiut. Vet. Med. J. 54 (116), 133–134. Basyoni, S. and Elsheikh, N. 2005. Incidence of Yersinia in raw milk and kariesh cheese with special reference to Yersinia enterocolitica. KFS. Vet. Medi. J. 3(1), 43–52. Begum, P. S., Madhavi, G., Rajagopal, S., Viswanath, B., Razak, M. A. and Venkataratnamma, V. 2017. Probiotics as functional foods: potential effects on human health and its impact on neurological diseases. Int. J. Nut. Pharm. Neuro. Dis. 7, 23–33. Bianco, A., Capozzi, L., Miccolupo, A., Iannetti, S., Danzetta, M. L., Del Sambro, L., Caruso, M. and Santagada, G., Parisi, A. 2020. Multi-locus sequence typing and virulence profile in Bacillus cereus sensu lato strains isolated from dairy products. Ital. J. Food. Saf. 9, 8401. Bonardi, S., Le Guern, A.S., Savin, C., Pupillo, G., Bolzoni, L., Cavalca, M. and Pongolini, S. 2018. Detection, virulence and antimicrobial resistance of Yersinia enterocolitica in bulk tank milk in Italy. Int. Dairy. J. 84, 46–53. Chlebicz, A. and Śliżewska, K. 2018. Campylobacteriosis, salmonellosis, yersiniosis, and listeriosis as zoonotic foodborne diseases: a review. Int. J. Env. Res. Public. Health. 15(5), 863. CLSI, 2015. Methods for antimicrobial dilution and disk susceptibility testing of infrequently isolated or fastidious bacteria. 3rd ed. CLSI guideline M45. Wayne, PA: Clinical and Laboratory Standards Institute. CLSI, 2017. Clinical and Laboratory Standards Institute (M11) performance standards for antimicrobial susceptibility testing. 27th informational supplement. Wayne, PA: CLSI. Darwish, S., Asfour, H. and Allam, H. 2015. Incidence of Yersinia enterocolitica and Yersinia pseudotuberculosis in raw milk samples of different animal species using conventional and molecular methods. Alex. Vet. Sci. J. 44, 174–185. Dietrich, R., Jessberger, N., Ehling-Schulz, M., M¨ artlbauer, E. and Granum, P. E. 2021. The food poisoning toxins of Bacillus cereus. Toxins 13 (2), 98. Donsì, F. and Ferrari, G. 2016. Essential oil nano-emulsions as antimicrobial agents in food J. Biotechnol. 233, 106–120. Drake, F. N., Davis, S., Khatiwada, J. and Williams, L. 2018. Identification and antimicrobial susceptibility of Yersinia enterocolitica found in chitterlings, raw milk and swine fecal samples. Adv. Microbiol. 8 (10), 804–820. Durofil, A., Maddela, N.R., Naranjo, R. A., Radice, M. 2022. Evidence on antimicrobial activity of essential oils and herbal extracts against Yersinia enterocolitica—A review. Food. Biosci. 47, 101712. EFSA Panel on Contaminants in the Food Chain (CONTAM). 2009. Scientific opinion on arsenic in food. EFSA. J. 7(10), 1351. Elafify, M., Alsayeqh, A. F., Aljasir, S. F., Tahon, A. B., Aly, S., Saad, M. F. Darwish, W.S. and Abdellatif, S. S. 2023. Occurrence and characterization of toxigenic Bacillus cereus in dairy products with an inactivation trial using D-Tryptophan and ascorbic acid in the rice pudding. LWT 175, 114485. Ehling-Schulz, M., Gui nebretiere, M., Monthán, A., Berge, O., Fricker, M. and Svensson, B. 2006. Toxin gene pro¢ling of enterotoxicandemetic Bacillus cereus. FEMS Microbiol. Lett. 260, 232–240. El-Karamani, Y. M. F. 2017. Studies on Bacillus cereus group in some milk-based foods. PhD. Thesis, Fac. Vet. Med. Assiut University, Assiut, Egypt. El-Sayed, H. S. and El-Sayed, S. M. 2021. A modern trend to preserve white soft cheese using nano-emulsified solutions containing cumin essential oil. Environ. Nanotechnol. Monitor. Manag. 16, 100499. El-Sherif., Walaa, M., El Hendy, A. H. M., Elnisr, N. A. and Wahba, Nahed, M. 2021. Studying the effect of chitosan on Bacillus cereus producing cereulide toxin in milk and some dairy desserts. J. Microbiol. Biotech. Food. Sci. 10, 5. Eltokhy, H., Abdelsamei, H., El Barbary, H. and Nassif, M. 2021. Prevalence of some pathogenic bacteria in dairy products. Benha. Vet. Medical. J. 40(2), 51–55. El-Zmakan, M. A. and Mubarak, A. G. 2017. Detection of B. cereus and some of its virulence genes in some dairy desserts and children diarrhea. Alex. J. Vet. Sci. 53, 28–38. Es, I., Khaneghah, A.M. and Akbariirad, H. 2017. Global regulation of essential oils. Essential oils in food processing: chemistry, safety and applications. Wiley, vol. 327, p: 38. Fang, F., Li, Y., Lu, X., Wu, K., Zhou, L., Sun, Y. and GAO, J. 2023. Effect of potential postbiotics derived from food-isolated Lactobacillus parabuchneri on different enterotypes of human gut microbiome. LWT 182, 114782. Feldman, D., Hoffman, R. and Simpson, J. 2003. The solution for data analysis and presentation graphics, 2nd ed. Berkeley, CA: Abacus Landcripts. Fetouh, M., Ibrahim, E., ElBarbary, H. and Maarouf, A. 2022. Isolation and genotypic identification of some spoilage and pathogenic microbes from yogurt. Benha. Vet. Med. J. 43(1), 123–128. Fois, F., Piras, F., Torpdahl, M., Mazza, R., Ladu, D. and Consolati, S.G., 2018. Prevalence, bioserotyping and antibiotic resistance of pathogenic Yersinia enterocolitica detected in pigs at slaughter in sardinia. Int. J. Food. Microbiol. 283, 1–6. Gupta, C., Garg, A. P., Uniyal, R. C. and Kumari, A. 2008. Comparative analysis of the antimicrobial activity of cinnamon oil and cinnamon extract on some food-borne microbes. Afr. J. Microbiol. Res. 2(9), 247–51. Gṻven, A., Sezer, Ç. Aydin, B., Oral, N. and Vatansever, L. 2010. Incidence and pathogenicity of Yersinia enterocolitica isolates from foods in Turkey. Kafkas. Universitesi. Veteriner. Fakultesi. 16, 107–112. Hamad, G. M., Omar, S. A., Mostafa, A. G. M., Cacciotti, I., Saleh, S. M. and Allam, M. G., 2022. Binding and removal of polycyclic aromatic hydrocarbons in cold smoked sausage and beef using probiotic strains. Food. Res. Int. 161, 111793. Hamad, G., Amer, A., Kirrella, G., Mehany, T., Elfayoumy, R. A. and Elsabagh, R. 2023. Evaluation of the prevalence of Staphylococcus aureus in chicken fillets and its bio-control using different seaweed extracts. Foods 12(1), 20. Han, A., Yoon, J. H., Choi, Y. S., Bong, Y., Jung, G. and Moon, S. K. 2023. Toxigenic diversity of Bacillus cereus isolated from fresh produce and effects of various factors on the growth and the cytotoxicity of B. cereus. Food Sci. Biotech. 1, 11. Hassan, G.M. and Afify, S.I. 2007. Occurrence of some pathogenic microorganisms in Kariesh cheese and their public health significance. Beni-Suef. Vet. Med. J. 18 (1), 142–150. Hefny, A., Mohamed, H. M., Etokhy, E. I. and Abd El-Azeem, M. W. 2020. Characterization of Bacillus cereus isolated from raw milk and milk products. J. Vet. An. Res. 3, 205. Heikal, G. I. and Al-wakeel, S. A. 2014. Bacteriological hazard of white cheese processed in some small rimitive lants (dairy shops) in Tata city. Beha. Vet. J. 26 (1), 185–194. Hulankova, R. 2022. Higher Resistance of Yersinia enterocolitica in comparison to Yersinia pseudotuberculosis to antibiotics and cinnamon, oregano and thyme essential oils. Pathogens 11(12), 1456. Hulankova, R. 2022. The influence of liquid medium choice in determination of minimum inhibitory concentration of essential oils against pathogenic bacteria. Antibiotics 11, 150 Hussain, M., Akhtar, S., Khalid, N., Azam, M., Iqbal, M. W., Ismail, T. and Korma, S. A. 2023. Hydrolysis microstructural profiling and utilization of Cyamopsis tetragonoloba in yoghurt. Fermentation 9 (1), 45. Hussein, M. F., Sadek, O. A. and El-Taher, S. G 2015. Occurrence of Bacillus Cereus and Staphylococcus aureus organisms in some dairy desserts. Assiut. Vet. Med. J. 61(145), 160–165. Ibrahim, G. A., Sharaf, O. M. and El-khalek, A. B. A. 2015. Microbiological quality of commercial raw milk, domiati cheese and Kareish cheese. Middle. East. J. Appl. Sci. 5(1), 171–176. Kaskatep, B., Kiymaci, M. E, Suzuk, S., Erdem, S. A., Cesur, S. and Yildiz, S. 2016. Antibacterial effects of cinnamon oil against carbapenem resistant nosocomial Acinetobacter baumannii and Pseudomonas aeruginosa isolates. Ind. Crops. Prod. (81), 191–194. Khalifa, N. Abou El roos, N. and Sharaf, E. 2007. Public health importance of Y. enterocolitica in ice cream peddlery in Kaliobia. Alexandria. Vet. Sci. J. 26(1), 51–54. kholif, A. E. and Olafadehan, O. A. 2021. Essential oils and phytogenic feed additives in ruminant diet: chemistry, ruminal microbiota and fermentation, feed utilization and productive performance. Phytochem. Rev. 20(6), 1087–1108. Klūga, A., Terentjeva, M., Vukovic, N. L. and Kačániová, M. 2021. Antimicrobial activity and chemical composition of essential oils against pathogenic microorganisms of freshwater fish. Plants (Basel) 10, 1265. Koua, A., Solange, K. E., Thomas, D. A., Ger main, K. T., Mireille, D., Sébastien, A. L. and Marcellin, D. K. 2014. Characterization of Yersinia spp. strains isolated from pigs in Apidjan, Côte d’Ivoire, West Africa. Afr. J. Microbiol. Res. 18(18), 1909–1915. Lahtinen, S. J., Jalonen, L., Ouwehand, A. C. and Salminen. 2007. Specific Bifidobacterium strains isolated from elderly subjects inhibit growth of Staphylococcus aureus. Inter. J. Food. Microbiol. 117, 125–128. Lando, V., Valduga, N. Z. and Moroni, L. S. 2023. Functional characterization of Lactobacilli strains with antimicrobial activity against Salmonella spp. and cell viability in fermented dairy product. Biocatal. Agr. Biotechnol. 47, 102605 Li, Y., Wang, M., Li, Y., Hong, B., Kang, D. and Ma, Y. 2023. Two novel antimicrobial peptides against vegetative cells, spores and biofilm of Bacillus cereus. Food. Control. 149, 109688. Madeira, J. P., Alpha-Bazin, B., Armengaud, J. and Duport, C. 2015. Time dynamics of the Bacillus cereus exoproteome are shaped by cellular oxidation. Front. Microbiol. 6, 342. Markey, B., Leonard, F., Archambault, M., Cullinane, A. and Maguire, D. 2013Elsevier Health Sciences. Mastrodonato, A.C., Favier, G.I., Lucero strada, C.S., Vidal, R. and Escudero, M.E., 2018. Bioserotypes, virulence genes, antimicrobial susceptibility and genomic diversity of Yersinia enterocolitica isolates from Argentina and Chile. J. Food. Saf. 38, 1249. Messelhäusser, U., Kämpf, P., Fricker, M., Ehling-Schulz, M., Zucker, R., Wagner, B., Busch, U. and Höller, C. 2010. Prevalence of Emetic Bacillus cereus in different ice creams in Bavaria. J. Food. Prot. 73, 395–399. Mohamed, A. S., Mohamed, E. A., Alnakip, M. E. A., Salah, F. and Abd-El Aal, S. F. 2016. Occurrence of Bacillus cereus in raw milk and some dairy products in Egypt. Jpn. J. Vet. Res. 64, S95–102. Montassier, E., Vald´es-Mas, R., Batard, E., Zmora, N., Dori-Bachash, M. and Suez, J. 2021. Probiotics impact the antibiotic resistance gene reservoir along the human GI tract in a person-specific and antibiotic-dependent manner. Nat. Microbiol. 6 (8), 1043–1054. Morsy, B., El-Kholy, A. and Meshref, A. 2022. Monitoring of Bacillus cereus in rice pudding. New. Valley. Vet. J. 2(2), 53–57. NCCLS. 2012. Performance standards for antimicrobial disc susceptibility test. Tentative standard M02-A11 (32), 12th ed. Wayne, PA: NCCLS. Nazzaro, F., F. Fratianni, L. D. Martino, R. Coppola and Feo, V. D. 2013. Effect of essential oils on pathogenic bacteria. Pharmaceuticals 6(12), 1451–1474. Nguyen, S. V., Muthappa, D. M., Hurley, D., Donoghue, O., McCabe, E., Anes, J. and Fanning, S. 2019. Yersinia hibernica sp. nov., isolated from pig-production environments. Inter. J. Syst. Evol. Microbiol. 69(7), 2023–2027. Oladipo, I. C., Ishola, O. S. and Oladipo, I. C. 2020. Appropriation of nanoparticle as food additive: a possibility. Europ. J. Adv. Res. Bio. Life Sci. 8, 1. Osama, R., Ahmed, M., Abdulmawjood, A. and Al-Ashmawy, M. 2020. Prevalence and antimicrobial resistance of Bacillus cereus in milk and dairy products. Man. Vet. Med. J. 21(2), 11–18. Owusu-Kwarteng, J., Wuni, A., Akabanda, F., Tano-Debrah, K. and Jespersen, L. 2017. Prevalence, virulence factor genes and antibiotic resistance of Bacillus cereus sensu lato isolated from dairy farms and traditional dairy products. BMC. Microbiol. 17(1), 9–16. Oxoid. 2002. Oxoid Limited © 2001-2009. Cambridge, UK: Themo Fisher Scientific. Özdemir, F. and Arslan, S. 2015. Genotypic and phenotypic virulence characteristics and antimicrobial resistance of Yersinia spp. isolated from meat and milk products. J. Food. Sci. 80(6), 1306–1313. Paudel, S. K., Bhargava, K. and Kotturi, H. 2019. Antimicrobial activity of cinnamon oil nano emulsion against Listeria monocytogenes and Salmonella spp. on melons. LWT, 111, 682–687. Radmehr, B., Zaferanloo, B., Tran, T., Beale, D. J. and Palombo, E, A. 2020. Prevalence and characteristics of Bacillus cereus group isolated from raw and pasteurised milk. Curr. Microbiol. 77(10), 3065–3075 Rahimi, E., Sepehri, S., Dehkordi, F. S., Shaygan, S. and Momtaz, H. 2014. Prevalence of Yersinia species in traditional and commercial dairy products in Isfahan Province, Iran Jundishapur J. Microbiol. 7(4), 1–6. Rahnama, H., Azari, R., Yousefi, M. H., Berizi, E., Mazloomi, S. M., Hosseinzadeh, S. and Conti, G. O. 2023. A systematic review and meta-analysis of the prevalence of Bacillus cereus in foods. Food. Control. 143, 109250. Rocha-Ramírez, L. M., Hern´ andez-Chinas, ˜ U., Moreno-Guerrero, S. S., RamírezPacheco, A. and Eslava, C. A. 2023. In vitro effect of the cell-free supernatant of the Lactobacillus casei strain IMAU60214 against the different pathogenic properties of diarrheagenic Escherichia coli. Microorganisms 11(5), 1324 Sadek, O. A., Sayed, S. M., El Berbawy, S. M., Mansy, M.F. and Hussien, M.F. 2014. Some antibiotic resistant bacteria of public health hazards isolated from raw milk sold in some Assiut city markets. Assiut. University. Bull. Environ. Res. 17(1), 97–107. Salfinger, Y. and Tortorello, M. L. 2015. Compendium of methods for 453 the microbiological examination of foods. 5th ed. Washington, DC: American Public Health 454 Association Washington. Serrano Cardona, L. and Munoz ˜ Mata, E. 2013. Paraninfo digital. Early Hum Dev 83(1), 1–11. Sharma, S., Mulrey, L., Byrne, M., Jaiswal, A. K. and Jaiswal, S. 2022. Encapsulation of essential oils in nanocarriers for active food packaging. Foods 11(15), 2337. Singh, P., Kaur, G. and Singh, A. 2023. Physical, morphological and storag e stability of clove oil nanoemulsion based delivery system. Food. Sci. Techno. Int. 29(2), 156–167. Singh, Y., Meher, J. G., Raval, K., Khan, F. A., Chaurasia, M., Jain, N. K. and Chourasia, M. K. 2017. Nanoemulsion: concepts, development and applications in drug delivery. J. Control. 252, 28–49. Siripatrawan, U. 2016. Active food packaging from chitosan incorporated with plant polyphenols. In Nanotechnology in the agri-food Industry. Eds., F. Grumezescu. London, UK: Academic Press, pp: 465–507. Tavassoli, M., Jamshidi,A., Movafagh,F. and Afshari, A. 2019. Virulence characteristics of Yersinia enterocolitica isolated from dairy products in the Northeast of Iran. J. Human. Environ. Health. Prom. 5(2), 72–78. Tian, L., Liu, R., Zhou, Z., Xu, X., Feng, S., Kushmaro, A. and Sun, Q. 2022. Probiotic characteristics of lacti planti Bacillus plantarum N-1 and its cholesterol-lowering effect in hypercholesterolemic rats. Probiotics Antimicrob. Proteins. 14(2), 337–348. Tirloni, E., Bernardi, C., Celandroni, F., Mazzantini, D., Massimino, M. and Stella, S. 2023. Prevalence, virulence potential, and growth in cheese of Bacillus cereus strains isolated from fresh and short-ripened cheeses sold on the Italian market. Microorganisms 11(2), 521 Wanger, A. 2007. Yersinia Manual of cl microbiol, 9th ed. Washington, DC: ASM Press, pp: 692–693. Wu, M., Dong, Q., Ma, Y., Yang, S., Aslam, M. Z. and Liu, Y. 2022. Potential antimicrobial activities of probiotics and their derivatives against Listeria monocytogenes in food field: a review. Food. Res. Int. 160, 111733. Ye, Q. H., Wu, Q. P., Hu, H. J., Zhang, J. M. and Huang, H. X. 2016. Prevalence and characterization of Yersinia enterocolitica isolated from retail frozen foods in China. Food. Control. 61, 20–27. Zadernowska, A., Chajęcka-Wierzchowska, W. and ŁaniewskaTrokenheim, Ł. 2014. Yersinia enterocolitica: a dangerous, but often ignored, foodborne pathogen. Food. Rev. Int. 30(1), 53–73. Zeinhom, M. M. and Abdel-Latef, G. K. 2014. Public health risk of some milk borne pathogens. Beni-Suef university. J. Basic App. Sci. 3(3), 209–215. Zhou, Z., Lan, X., Zhu, L., Zhang, Y., Chen, K. and Zhang, W. 2023. Portable dualaptamer microfluidic chip biosensor for Bacillus cereus based on aptamer tailoring and dumbbell-shaped probes. J. Hazard. Mater. 445, 30545. | ||
| How to Cite this Article |
| Pubmed Style Fathy SS, Awad EI, Aal SFA, Abdelfatah EN, Tahoun AB. Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Vet J. 2024; 14((1) (Zagazig Veterinary Conference)): 470-480. doi:10.5455/OVJ.2024.v14.i1.43 Web Style Fathy SS, Awad EI, Aal SFA, Abdelfatah EN, Tahoun AB. Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. https://www.openveterinaryjournal.com/?mno=182100 [Access: July 03, 2025]. doi:10.5455/OVJ.2024.v14.i1.43 AMA (American Medical Association) Style Fathy SS, Awad EI, Aal SFA, Abdelfatah EN, Tahoun AB. Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Vet J. 2024; 14((1) (Zagazig Veterinary Conference)): 470-480. doi:10.5455/OVJ.2024.v14.i1.43 Vancouver/ICMJE Style Fathy SS, Awad EI, Aal SFA, Abdelfatah EN, Tahoun AB. Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Vet J. (2024), [cited July 03, 2025]; 14((1) (Zagazig Veterinary Conference)): 470-480. doi:10.5455/OVJ.2024.v14.i1.43 Harvard Style Fathy, S. S., Awad, . E. I., Aal, . S. F. A., Abdelfatah, . E. N. & Tahoun, . A. B. (2024) Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Vet J, 14 ((1) (Zagazig Veterinary Conference)), 470-480. doi:10.5455/OVJ.2024.v14.i1.43 Turabian Style Fathy, Sally S., Esmat I. Awad, Salah F.a. Abd-el Aal, Eman N. Abdelfatah, and Asmaa B.m.b. Tahoun. 2024. Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Veterinary Journal, 14 ((1) (Zagazig Veterinary Conference)), 470-480. doi:10.5455/OVJ.2024.v14.i1.43 Chicago Style Fathy, Sally S., Esmat I. Awad, Salah F.a. Abd-el Aal, Eman N. Abdelfatah, and Asmaa B.m.b. Tahoun. "Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens." Open Veterinary Journal 14 (2024), 470-480. doi:10.5455/OVJ.2024.v14.i1.43 MLA (The Modern Language Association) Style Fathy, Sally S., Esmat I. Awad, Salah F.a. Abd-el Aal, Eman N. Abdelfatah, and Asmaa B.m.b. Tahoun. "Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens." Open Veterinary Journal 14.(1) (Zagazig Veterinary Conference) (2024), 470-480. Print. doi:10.5455/OVJ.2024.v14.i1.43 APA (American Psychological Association) Style Fathy, S. S., Awad, . E. I., Aal, . S. F. A., Abdelfatah, . E. N. & Tahoun, . A. B. (2024) Inhibitory effect of some probiotic strains and essential oils on the growth of some foodborne pathogens. Open Veterinary Journal, 14 ((1) (Zagazig Veterinary Conference)), 470-480. doi:10.5455/OVJ.2024.v14.i1.43 |