| Research Article | ||
Open Vet. J.. 2026; 16(3): 1877-1884 Open Veterinary Journal, (2026), Vol. 16(3): 1877-1884 Research Article Role of inflammatory cytokines in tissue remodeling and disease progressionWasman Jaber Alyahya* and Rawayh Muslim AlbaghlanyDepartment of Medical Microbiology, College of Dentistry, University of Basrah, Basrah, Iraq *Corresponding Author: Wasman Jaber Alyahya. Department of Medical Microbiology, College of Dentistry, University of Basrah, Basrah, Iraq. Email:wasman.naser [at] uobasrah.edu.iq Submitted: 15/12/2025 Revised: 11/02/2026 Accepted: 21/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
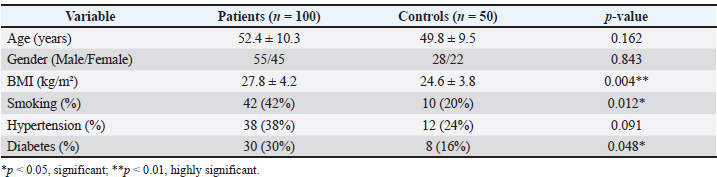
AbstractBackground: The persistence of the activation of pro-inflammatory cytokines, which facilitates the pathological tissue remodeling by disrupting the extracellular matrix (ECM), has been identified as a risk factor for chronic inflammation. A disproportion in the proinflammatory cytokines and anti-inflammatory cytokines enhances tissue destruction and disease etiology. Aim: This study aimed to assess the role of inflammatory cytokines and tissue remodeling markers in disease progression and determine the association of these factors with systemic inflammation and structural tissue changes. Methods: The study was a hospital-based case-control study that was conducted between February 20 and October 15, 2024, and involved 150 participants (100 with chronic inflammatory disease involving tissue remodeling and 50 healthy controls). The clinical and laboratory diagnoses were made. Serum samples of pro-inflammatory cytokines interleukin-6 [IL-6, tumor necrosis factor (TNF) a, IL-1 b, and C-reactive protein (CRP)], regulatory cytokines interleukin-10 (IL-10, TGFB), and tissue remodeling markers matrix metalloproteinases [MMP-2, MMP-9, and Tissue metalloproteinase inhibitors (TIMP)-1] were obtained by Enzyme-Linked Immunosorbent Assay. Results: Age and sex were not different between patients and controls, which demonstrated demographic homogeneity. However, patients were older, smoked more, and had more diabetes, which is evidence of the presence of inflammation-facilitating risk factors. The levels of pro-inflammatory cytokines (IL-6, TNF- 02, interleukin-1 beta) and CRP were significantly high in the patients, indicating active systemic inflammation. Conversely, the levels of anti-inflammatory cytokines (IL-10, transforming growth factor-beta), on the other hand, were decreased significantly. The tissue markers of remodeling were found to be more MMP-2 and MMP-9 with less TIMP-1, indicating dysregulated ECM turnover. Conclusion: Excess pro-inflammatory cytokine activity and the absence of regulatory signaling are features of chronic inflammatory diseases that cause dysregulated matrix remodeling. High levels of IL-6, TNF, and MMPs stimulate the degradation of the ECM, whereas low levels of IL-10 and TIMP-1 do not balance the effects of inflammation, increasing tissue destruction and disease progression. Keywords: Inflammatory cytokines, Tissue remodeling, Disease progression, Matrix metalloproteinases, Tissue remodeling. IntroductionChronic inflammation is a biological process involved in the initiation, development, and outcome of a broad range of human diseases. Although acute inflammation is a homeostatic reaction to eliminate harmful stimuli and facilitate tissue repair, a continuous or unregulated inflammatory response may result in pathological tissue remodeling with progressive organ dysfunction (Anna, 2022; Evgenii and Zhuravleva, 2022; Kiss, 2022; Gusev and Zhuravleva, 2022). In this regard, inflammatory cytokines are very important due to their central role in coordinating immune responses, controlling cell behavior, and modifying structural tissue alterations. The role of inflammatory cytokines in tissue remodeling is also important in understanding disease mechanisms and finding possible therapeutic targets (Henein, et al., 2022). Cytokines that are instrumental in the control of local and systemic inflammatory responses include interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and C-reactive protein (CRP). These mediators are produced by activated immune cells, endothelial cells, fibroblasts, and injured tissue cells in response to injury or infection (Sikora et al., 2023). Persistently high levels of pro-inflammatory cytokines have been reported to stimulate cell growth, apoptotic survival, angiogenesis, and breakdown of the extract-cellular matrix (ECM), which are associated with rearranged tissue structure and disease development. On the contrary, anti-inflammatory and regulatory cytokines, such as interleukin-10 (IL-10) and transforming growth factor-beta (TGF-β), are important in the regulation of immune activation and homeostasis of the tissues; however, their imbalance can also be involved in fibrosis and pathological remodeling (Atsu et al., 2023; Naif et al., 2025). Tissue remodeling is a dynamic process that entails the process of degradation and synthesis of ECM components in a coordinated manner. The matrix metalloproteinases (MMPs) in specific cases of MMP-2 and MMP-9 are zinc-dependent proteolytic enzymes that assist in tearing down collagen and other ECM proteins that are necessary for tissue restructuring (Wolosowicz et al., 2024). Tissue metalloproteinase inhibitors (TIMPs), including TIMP-1, strictly control them. An imbalance between TIMPs and MMPs may cause excessive degradation or accumulation of the ECM due to the inflammatory cytokines, which causes fibrosis, stiffening of tissues, and diffusion of normal organ functions (Trojanek, 2023). These mechanisms have been associated with many chronic diseases, including cardiovascular, fibrotic, inflammatory joint, and metabolic diseases (Diller and Tabor, 2022). A growing body of evidence shows that inflammatory cytokines are not only disease activity markers but, also active disease progressors. High concentrations of IL-6 and TNF-α have been linked to poor clinical progression in chronic inflammatory and degenerative illnesses, whereas modulations in IL-10 and TGF- β signaling have been linked to dysfunctional inflammatory resolution and abnormal tissue healing (Tylutka et al., 2024). In addition, the connection between tissue remodeling enzymes and cytokine networks demonstrates the complexity of the inflammatory signaling pathways and their long-term influence on the development of the disease (Lacina et al., 2024). Although large volumes of research have been conducted, the relative balance of pro-inflammatory and anti-inflammatory cytokines and how they relate to tissue remodeling markers is still not fully understood, especially when it comes to disease progression. Simultaneous assessment of these biomarkers can help gain a clear understanding of the mechanisms by which inflammation promotes structural tissue remodeling and leads to persistent disease conditions (Camargo and Brandão, 2025). The recent study will focus on the contribution of key inflammatory and regulatory cytokines in combination with the examination of matrix remodeling markers to understand the multiplicative influences on tissue remodeling and disease progression. These findings can be used to improve the risk stratification and design of specific anti-inflammatory and anti-fibrotic therapeutic measures. Materials and MethodsEthical approvalThe study was approved by the Human Ethics Committee of Al-Hussein Teaching Hospital, Iraq. Each of the procedures was performed based on the ethical principles of the Declaration of Helsinki. All participants provided written informed consent and/or were signed by legal guardians. The data of the participants were kept confidential, and their privacy was upheld. This research was planned as a hospital case-control study that was undertaken on February 20, 2024, and October 15, 2024. A total of 150 patients with chronic inflammatory diseases related to tissue remodeling and disease development, in addition to 50 age and sex-matched apparently healthy controls, were recruited. The target patients were those who had clinically confirmed diseases with active inflammation and ECM remodeling, including chronic inflammatory fibrosis-associated diseases, atherosclerotic cardiovascular disease, inflammatory joint diseases, and chronic metabolic-inflammatory diseases. The inclusion criteria included patients who had confirmed diagnoses of systemic inflammation using clinical assessment and laboratory results. Pregnancy, acute infections, autoimmune diseases not directly connected with tissue remodeling, malignancy, and chronic liver/kidney failure, recent surgery or trauma, and current intake of immunosuppressive or anti-inflammatory drugs that may affect the levels of cytokine or MMPs were also excluded from the study. The control group did not have inflammatory, metabolic, or chronic systemic illness. The diagnosis was made based on the standardized clinical examination with the additional support of laboratory tests and imaging examinations as needed based on each condition’s existing diagnostic criteria. Aseptic venous blood samples were collected, and serum was centrifuged to be analyzed using biomarkers. Commercial Enzyme-Linked Immunosorbent Assay kits were used to determine serum concentrations of pro-inflammatory (IL-6, TNF- α, IL-1 β, CRP), anti-inflammatory, and regulatory (IL-10, TGF- β) cytokines and tissue remodeling (MMP-2, MMP-9, TIMP-1) markers according to the manufacturer’s instructions. The number of patients selected in this study was based on the number of eligible patients who met the inclusion criteria within the spanned study period, which was more exploratory and hospital-based. There was no formal a priori calculation or power analysis of the sample size. Nevertheless, the analysis showed statistically significant differences and correlations in the main inflammatory and hematological parameters, and the sample size was sufficient to reveal the relevant effects. However, larger prospective studies that have been pre-power-calculated should verify and generalize these findings. Statistical analysisThe SPSS version 26 software was used to analyze the quantitative data. The mean standard deviation indicated continuous variables, whereas frequencies and percentages were used to represent categorical variables. An independent Student t-test was used to compare patients and controls in variables that were normally distributed, and the Mann–Whitney U test was used to compare variables with non-uniform distribution. The chi square test was used to analyze the categorical variables. Pearson or Spearman correlation coefficients were used to determine the relationships between inflammatory cytokines and inflammatory tissue remodeling markers. A p-value of 0.05 was considered statistically significant. ResultsSociodemographic and clinical characteristics of patients and controlsTable 1 depicts the sociodemographic and clinical factors of the patients compared with the healthy control group. The differences between the two groups were not statistically significant in relation to age or sex distribution (p > 0.05), indicating adequate homogeneity of the demographics. Conversely, the patients had significantly higher body mass index than the healthy control group (27.8 ± 4.2 vs. 24.6 ± 3.8 kg/m2, p=0.004). A significantly greater smoking rate and prevalence of diabetes (p < 0.05), which indicates that metabolic and lifestyle factors were more likely to promote inflammation in patients. Although the difference in the prevalence of hypertension was not statistically significant, it was comparatively greater in the patients, which might also contribute to the aggravation of inflammation and disease progression. Table 1. Demographic factors, body mass index, and major clinical risk factors.
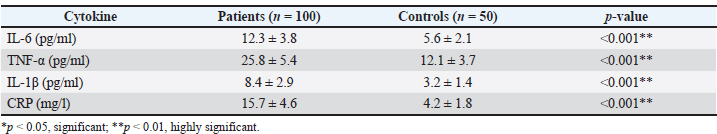
Pro-inflammatory cytokine levels in patients and healthy controlsTable 2 demonstrates that the inflammatory cytokine levels were highly increased in the patients compared with the healthy control group. The concentrations of IL-6, TNF-α, IL-1β, and CRP significantly increased, and they were greatly significant (p < 0.001). These outcomes show that patients have an active systemic inflammatory state, which is a major contributor to the activation of inflammatory pathways and tissue injury and remodeling. These improvements are also indicative of the central role of inflammatory cytokines in mediating disease progression and complicating the pathological alterations that occur during chronic inflammation. Table 2. Systemic inflammatory profile associated with disease progression.
Anti-inflammatory and regulatory cytokine levels in patients and healthy controlsTable 3 demonstrates that the levels of anti-inflammatory and immune-regulating cytokines in patients declined significantly relative to the healthy control sample. The reduction in the levels of IL-10 and TGF-β was significantly evident in the patients (p < 0.001), indicating that the immune-regulation mechanisms were impaired and the antibodies could not adequately suppress the excessive inflammatory reaction. Such disruption of pro-inflammatory and anti-inflammatory cytokines is a major factor in the maintenance of persistent inflammation, the facilitation of tissue damage, and the increase in remodeling processes, which directly lead to the progression of a disease and the exacerbation of its clinical manifestations. Table 3. Alterations in the levels of immune regulatory cytokines associated with disease progression.
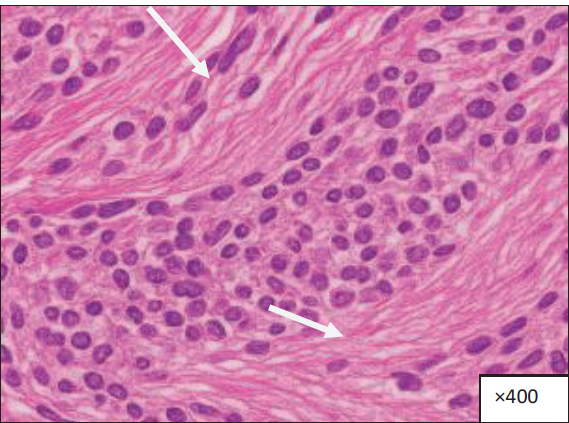
MMPs and tissue remodeling markers in patients and healthy controlTable 4 reveals a serious imbalance in the parameters of tissue remodeling in patients in relation to the healthy control condition. Patients recorded significant increases in the levels of ECM degrading enzymes MMP-9 and MMP-2 (p=0.001), which is a hyperactivation of the processes of tissue structural breakdown. On the other hand, there was a major reduction in the concentration of the tissue inhibitor of metalloproteinase enzymes (TIMP-1), indicating a lack of control of these enzymes. This disproportion between MMPs and TIMP-1 facilitates pathological tissue remodeling, accelerates the destruction of the tissue structure, and leads to disease development and clinical exacerbation (Figs. 1 and 2).
Fig. 1. Histological section of chronically inflamed tissue obtained from the affected organ (specify the organ name), stained with hematoxylin and eosin (H&E), at ×400 magnification. Significant chronic inflammatory alterations are shown by extensive deposits of mononuclear inflammatory cells, lymphocytes, and macrophages in the stromal compartment (marked by arrows). These cells are diffusely distributed throughout tissue structures, indicating chronic inflammatory activity. Stromal edema and disruption of normal tissue architecture is also associated with chronic inflammation and other histopathological features.
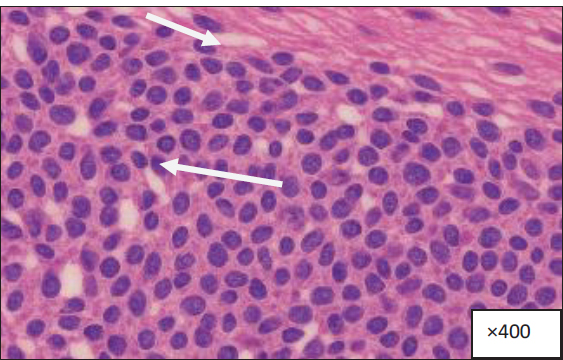
Fig. 2. Histological section of inflamed tissue obtained from the affected organ (specify the organ name), stained with hematoxylin and eosin (H&E), at ×400 magnification. Table 4. Imbalance between ECM degradation and inhibitory pathways.
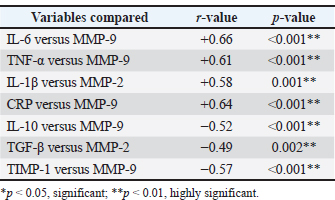
Association between systemic inflammatory cytokines and tissue remodeling markersIn Table 5, the correlation analysis revealed significant correlations between the tissue remodeling markers and the inflammatory mediators. There were positive correlations between pro-inflammatory cytokines (IL-6, TNF-α, IL-1β, and CRP) and MMP-9 and MMP-2, and higher levels of systemic inflammation were correlated with degradation of extra-cellular matrix. Conversely, anti-inflammatory cytokines (IL-10 and TGF-β) had negative associations with MMPs, which indicated the regulation of tissue components to limit tissue destruction. TIMP-1 had a negative relationship with MMP-9, indicating poor inhibitory activity over proteolytic activity in patients. Overall, these results confirm the existence of an inflammation-induced imbalance that is biased toward pathological tissue remodeling. Table 5. Correlation analysis between inflammatory and tissue remodeling parameters.
An inflammatory reaction is observed in the tissue section with significant lymphocyte and macrophage accumulation in both the interstitial and perivascular areas (marked by arrows). The inflammatory infiltrate is associated with vascular congestion, focal tissue edema, and partial distortion of the normal histological structure, indicating ongoing immune activation and tissue damage indicative of active inflammation. DiscussionRecent studies provide strong evidence that inflammatory cytokines play a key role in remodeling and disease progression in the body. This conclusion is based on the finding that pro-inflammatory, anti-inflammatory, and matrix-remodeling are very different between patients and healthy controls (Giacomini et al., 2021; Moldoveanu et al., 2024). The marginally significant differences in the age and sex distributions are also supportive of the sufficient alignment of cohorts and the reduction of possible demographic confounding, which support the argument that the observed biochemical changes can be explained primarily by inflammatory processes caused by diseases as opposed to inherent properties of the initial population (García-Donas et al., 2022; Allen et al., 2023). Clinically, patients had a very high BMI, smoking rates, and rates of diabetes compared to controls. These are well-known contributors of chronic low-grade inflammation and induce cytokine production (Musale et al., 2023). The secretion of IL-6 and TNF-α by adipose tissue, the induction of oxidative stress and endothelial damage by smoking, and the sustained inflammatory response and loss of tissue repair in diabetes are promoted by obesity. It is in these past research works that there is a consistent finding that these comorbidities work together with inflammatory pathways to speed up tissue remodeling and disease progression (Dilworth et al., 2021; Yadav et al., 2023). Therefore, their increased presence in the patient group probably leads to further dysregulation of cytokines and matrix degradation. The notable rise in proinflammatory cytokines, including IL-6, TNF- α, IL-1β, and CRP, was one of the major findings. These cytokines are commonly known to be agents of chronic inflammation and mediators of pathological remodeling of tissues. IL-6 is a central mediator of immune activation and fibroblast growth, angiogenesis, and acute phase, and high levels of IL-6 have been strongly linked to fibrosis, vascular remodeling, and poor outcomes in heart and metabolic diseases ( Libby, 2021; Li et al., 2022). TNF-α and IL-1β also enhance the twists of inflammatory pathways by encouraging the recruitment of leukocytes, endothelial malfunction, and proteolytic enzyme activation, which induces the acceleration of ECM breakdown (Zhao et al., 2021; Theofilis et al., 2021). The significant increase in CRP levels is not only an effective indicator of systemic inflammatory load, which sums up the downstream consequences of cytokine signaling, but it also concurs with the existing literature that shows that higher CRP levels correlate with disease severity and progression (Lazarut-Nistor and Mark, 2025; Pah et al., 2025). Conversely, the levels of anti-inflammatory and regulating cytokines, such as IL-10 and TGF-β, were greatly decreased in patients. IL-10 is a vital immunoregulatory cytokine that is important in restraining inflammation by reducing the production of pro-inflammatory cytokines and preventing the activation of immune cells. Reductions in IL-10 levels are found in a range of chronic inflammatory conditions and are related to the inability to resolve inflammation and continued tissue damage (Martinez-Espinosa et al., 2021; Huynh et al., 2023). Similarly, TGF-β has two functions in the immune system inhibition and the healing process. Although high local concentrations of TGF-25 can enhance fibrogenesis, sufficient levels of the protein are essential for maintaining immune tolerance and supporting tissue remodeling. It is possible that the low circulating concentrations of TGF-B found in this study indicate the impairment of regulatory control, which then allows pro-inflammatory signaling to prevail (Budi et al., 2021). Nevertheless, other studies have shown contradictory results, especially on the level of TGF- β with high levels being recorded in the advanced fibrotic diseases (Antar et al., 2023). The discrepancies can be attributed to variations in disease stage, tissue specificity, and sample type. During the early stages or active progression of the disease, the presence of inflammatory dominance and ECM degradation can be predominant, leading to decreased levels of circulating TGF-β, but in the late fibrosis (Qin et al., 2023), the compensatory overexpression of TGF- β within the tissues can be found. Other factors that cause different results in studies include variations in methodology, such as serum or tissue measurements and patient heterogeneity (Shi et al., 2022). The greatest evidence of active tissue remodeling in the present study is the substantial increase in MMP-9 and MMP-2 levels and the decrease in TIMP-1 levels in patients. MMPs are one of the most crucial enzymes involved in ECM degradation and structural reorganization, which are highly expressed by proinflammatory cytokines, including IL-6, TNF-α, and IL-1β (Caimi et al., 2021). Stimulated MMP activity promotes tissue remodeling, but uncontrolled MMP activity causes tissue structural damage, tissue integrity loss, and functional decline. The corresponding decrease in TIMP-1 indicates a lack of inhibition of MMP activity, and the balance shifts to over-degradation of the matrix. Analogous MMP/TIMP disproportions have been closely associated with disease progression in cardiovascular disease, chronic inflammatory conditions, and fibrotic diseases (Wolosowicz et al., 2024; Qi et al., 2025). Other conflicting reports have recorded high TIMP-1 average in specific chronic illnesses, especially in late fibrotic stages (Dimic-Janjic et al., 2023). This apparent paradox could indicate the effect of stage-specific remodeling in that early disease is defined by prevailing matrix degradation and later stages by compensatory inhibition and matrix deposition. Distinctions in underlying pathologic entities, comorbidity, and therapeutic exposure may also increase the expression patterns of MMP and TIMP (Chen et al., 2021). Limitations of the studyThis study involved patients with a heterogenous group of chronic inflammatory diseases to define similar inflammatory and tissue remodeling pathways. Nonetheless, subgroup analyses on each disease were not conducted because of the small sample sizes in each disease category, which could have minimized the power of the statistical analysis. Furthermore, the cross-sectional design does not allow causal interpretation, and the effects of unknown confounding variables cannot be avoided. Thus, the results can be interpreted as representing overall inflammatory processes and not as profile-specific conditions. Conducting future research using bigger, disease-stratified, and longitudinal cohorts. ConclusionThe findings suggest that the presence of high levels of pro-inflammatory cytokines and low levels of regulatory cytokines, and a scale effect of MMPs and TIMP-1 are related to the pathological remodeling of tissues and disease progression. The findings indicate a possible association between inflammatory activity and ECM dysregulation; however, because the study design was observational, causal associations are not possible. Therefore, the findings on this basis should be seen as indicative of associative biological processes and not necessarily direct treatment objectives. More longitudinal and mechanistic research is needed to confirm the causal relationship and determine the clinical significance of targeting inflammatory and tissue remodeling pathways. AcknowledgmentsThis research is supported by the Department of Medical Microbiology, College of Dentistry, University of Basrah. Conflict of interestThe authors declare there is no conflict of interest. FundingNone. Data availabilityThe datasets used and analyzed during the current study are available from the corresponding author on reasonable request. Authors' contributionsAuthors contributed equally in the designing, analysis, writing, and final version of the article. ReferencesAllen, J.B., Ludtka, C. and James, B.D. 2023. Sex as a biological variable in tissue engineering and regenerative medicine. Annu. Rev. Biomed. Eng. 25, 311–331; doi:10.1146/annurev-bioeng-092222-030857 Antar, S.A., Ashour, N.A., Marawan, M.E. and Al-Karmalawy, A.A. 2023. Fibrosis: types, effects, markers, mechanisms for disease progression, and its relationship with oxidative stress, immunity, and inflammation. Int. J. Mol. Sci. 24(4), 4004; doi:10.3390/ijms24044004 Atsu, P.M., Mowen , C. and Thompson, G.L. 2023. Enhanced cell viability and migration of primary bovine annular fibrosus fibroblast-like cells induced by microsecond pulsed electric field exposure. 8(40), 36815–36822; doi:10.1021/acsomega.3c03518 Budi, E.H., Schaub, J.R., Decaris, M., Turner, S. and Derynck, R. 2021. TGF-β as a driver of fibrosis: physiological roles and therapeutic opportunities. J. Pathol. 254(4), 358–373; doi:10.1002/path.5680 Caimi, G., Hopps, E., Montana, M., Urso, C., Carollo, C., Canino, B. and Lo Presti, R. 2021. The function of matrix metalloproteinase-9 (MMP-9) and its tissue inhibitor (TIMP-1) in several clinical conditions: results and analysis of our survey. Clin. Hemorhol. Microcircul. 78(4), 401–416; doi:10.3233/CH-201094 Chen, Y., Yan, H., Li, G. and Zhang, Y. 2021. Higher TGF-β1, TGF-β2, MMP-2, and TIMP-1 levels in the aqueous humor of patients with acute primary angle closure. Ophthalmic Res. 64(1), 62–67; doi:10.1159/000507762 Diller, R.B. and Tabor, A.J. 2022. The role of the extracellular matrix (ECM) in wound healing: a review. Biomimetics (Basel, Switzerland) 7(3), 87; doi:10.3390/biomimetics7030087 Dilworth, L., Facey, A. and Omoruy, F. 2021. Diabetes mellitus and its metabolic complications: the role of adipose tissues. Int. J. Mol. Sci. 22(14), 7644; doi:10.3390/ijms22147644 Dimic-Janjic, S., Hoda, M.A., Milenkovic, B., Kotur-Stevuljevic, J., Stjepanovic, M., Gompelmann, D., Jankovic, J., Miljkovic, M., Milin-Lazovic, J., Djurdjevic, N., Maric, D., Milivojevic, I. and Popevic, S. 2023. The usefulness of MMP-9, TIMP-1 and MMP-9/TIMP-1 ratio for diagnosis and assessment of COPD severity. Eur. J. Med. Res. 28, 127; doi:10.1186/s40001-023-01094-7 García-Donas, J.G., Paine, R.R., Bonicelli, A. and Kranioti, E.F. 2022. Age estimation for two Mediterranean populations: rib histomorphometry applied to forensic identification and bone remodeling research. Int. J. Legal Med. 136(5), 1469–1481; doi:10.1007/s00414-022-02812-2 Giacomini, E., Minetto, S., Li Piani, L., Pagliardini, L., Somigliana, E. and Viganò, P. 2021. Genetics and inflammation in endometriosis: improving knowledge for development of new pharmacological strategies. Int. J. Mol. Sci. 22(16), 9033; doi:10.3390/ijms22169033 Gusev, E. and Zhuravleva, Y. 2022. Inflammation: a new look at an old problem. Int. J. Mol. Sci. 23(9), 4596; doi:10.3390/ijms23094596 Henein, M.Y., Vancheri, S., Longo, G. and Vancheri, F. 2022. The role of inflammation in cardiovascular disease. Int. J. Mol. Sci. 23(21), 12906; doi:10.3390/ijms232112906 Huynh , T., Reed, C., Blackwell, Z., Phelps, P., Herrera, L.C.P., Almodovar, J., Zaharoff, D.A. and Wolchok, J. 2023. Local IL-10 delivery modulates the immune response and enhances repair of volumetric muscle loss muscle injury. Sci. Rep. 13(1), 1983; doi:10.1038/s41598-023-27981-x Kiss, A. 2022. Inflammation in focus: the beginning and the end. Pathol. Oncol. Res. 27, 1610136; doi:10.3389/pore.2021.1610136 Lacina, L., Kolář, M., Pfeiferová, L., Gál, P. and Smetana, K., Jr. 2024. Wound healing: insights into autoimmunity, aging, and cancer ecosystems through inflammation and IL-6 modulation. Front Immunol. 15, 1403570; doi:10.3389/fimmu.2024.1403570 Lazarut-Nistor, A. and Slevin, M. 2025. Beyond the biomarker: monomeric CRP as a driver of multisystem pathology in rheumatoid arthritis. Int. J. Mol. Sci. 26(17), 8227; doi:10.3390/ijms26178227 Li, Y., Zhao, J., Yin, Y., Li, K., Zhang, C. and Zheng, Y. 2022. The role of IL-6 in fibrotic diseases: molecular and cellular mechanisms. Int. J. Biol. Sci. 18(14), 5405–5414; doi:10.7150/ijbs.75876 Libby, P. 2021. Targeting inflammatory pathways in cardiovascular disease: the inflammasome, interleukin-1, interleukin-6 and beyond. Cells 10(4), 951; doi:10.3390/cells10040951 Martinez-Espinosa, I., Serrato, J.A. and Ortiz-Quintero, B. 2021. Role of IL-10-producing natural killer cells in the regulatory mechanisms of inflammation during systemic infection. Biomolecules 12(1), 4; doi:10.3390/biom12010004 Moldoveanu, C.A., Tomoaia-Cotisel, M., Sevastre-Berghian, A., Tomoaia, G., Mocanu, A., Pal-Racz, C., Toma, V.A., Roman, I., Ujica, M.A. and Pop, L.C. 2024. A review on current aspects of curcumin-based effects in relation to neurodegenerative, neuroinflammatory and cerebrovascular diseases. Molecules (Basel, Switzerland) 30(1), 43; doi:10.3390/molecules30010043 Musale, V., Wasserman, D.H. and Kang, L. 2023. Extracellular matrix remodeling in obesity and metabolic disorders. Life Metab. 2(4), load021; doi:10.1093/lifemeta/load021 Naif, N., Ahmed, E., Thamer, N. and Muhsein, O. 2025. The impact of insulin and adipokine biomarkers on breast cancer progression and patient outcomes. Bull. Pharm. Sci. Assiut Univ. 48(1), 511–522; doi:10.21608/bfsa.2025.339732.2360 Pah, A.M., Serban, S., Mateescu, D.M., Cotet, I.G., Muresan, C.O., Ilie, A.C., Buleu, F., Craciun, M.L., Crisan, S. and Avram, A. 2025. Systemic inflammatory biomarkers (interleukin-6, high-sensitivity C-reactive protein, and neutrophil-to-lymphocyte ratio) and prognosis in heart failure: a meta-analysis of prospective cohort studies. J. Clin. Med. 14(23), 8610; doi:10.3390/jcm14238610 Qi, S., Wang, S., Tan, Y., Pan, C. and Bi, X. 2025. Extracellular matrix (ECM)-regulated molecular switches: tissue inhibitors of metalloproteinases in synaptic formation and neuropathic diseases. Cell Mol. Neurobiol. 45(1), 100; doi:10.1007/s10571-025-01621-6 Qin, L., Liu, N., Bao, C.L., Yang, D.Z., Ma, G.X., Yi, W.H., Xiao, G.Z. and Cao, H.L. 2023. Mesenchymal stem cells in fibrotic diseases-the two sides of the same coin. Acta Pharmacol. Sin. 44(2), 268–287; doi:10.1038/s41401-022-00952-0 Shi, N., Wang, Z., Zhu, H., Liu, W., Zhao, M., Jiang, X., Zhao, J., Ren, C., Zhang, Y. and Luo, L. 2022. Research progress on drugs targeting the TGF-β signaling pathway in fibrotic diseases. Immunol. Res. 70(3), 276–288; doi:10.1007/s12026-022-09267-y Sikora, J.P., Karawani, J. and Sobczak, J. 2023. Neutrophils and the systemic inflammatory response syndrome (SIRS). Int. J. Mol. Sci. 24(17), 13469; doi:10.3390/ijms241713469 Siqueira, R.C. and Brandão, C.C. 2025. The role of cytokines in degenerative retinal diseases: a comprehensive review. Biomedicines 13(7), 1724; doi:10.3390/biomedicines13071724 Theofilis, P., Sagris, M., Oikonomou, E., Antonopoulos, A.S., Siasos, G., Tsioufis, C. and Tousoulis, D. 2021. Inflammatory Mechanisms contributing to endothelial dysfunction. Biomedicines 9(7), 781; doi:10.3390/biomedicines907078 Trojanek, J. 2023. The specific role of extracellular matrix metalloproteinases in the pathology and therapy of hard-to-heal wounds. Acta Biochim. Polonica. 70(4), 745–750; doi:10.18388/abp.2020_6934 Tylutka, A., Walas, Ł. and Zembron-Lacny, A. 2024. Level of IL-6, TNF, and IL-1β and age-related diseases: a systematic review and meta-analysis. Front. Immunol. 15, 1330386; doi:10.3389/fimmu.2024.1330386 Wolosowicz, M., Prokopiuk, S. and Kaminski, T.W. 2024. The complex role of matrix metalloproteinase-2 (MMP-2) in health and disease. Int. J. Mol. Sci. 25(24). 13691; doi:10.3390/ijms252413691 Yadav, U., Kumar, N. and Sarvottam, K. 2023. Role of obesity related inflammation in pathogenesis of peripheral artery disease in patients of type 2 diabetes mellitus. J. Diabetes Metab. Disord. 22(1), 175–88; doi:10.1007/s40200-023-01221-5 Zhao, H., Wu, L., Yan, G., Chen, Y., Zhou, M., Wu, Y. and Li, Y. 2021. Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduc. Target. Ther. 6(1), 263; doi:10.1038/s41392-021-00658-5 | ||
| How to Cite this Article |
| Pubmed Style Alyahya WJ, Albaghlany RM. Role of inflammatory cytokines in tissue remodeling and disease progression. Open Vet. J.. 2026; 16(3): 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 Web Style Alyahya WJ, Albaghlany RM. Role of inflammatory cytokines in tissue remodeling and disease progression. https://www.openveterinaryjournal.com/?mno=303345 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.43 AMA (American Medical Association) Style Alyahya WJ, Albaghlany RM. Role of inflammatory cytokines in tissue remodeling and disease progression. Open Vet. J.. 2026; 16(3): 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 Vancouver/ICMJE Style Alyahya WJ, Albaghlany RM. Role of inflammatory cytokines in tissue remodeling and disease progression. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 Harvard Style Alyahya, W. J. & Albaghlany, . R. M. (2026) Role of inflammatory cytokines in tissue remodeling and disease progression. Open Vet. J., 16 (3), 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 Turabian Style Alyahya, Wasman Jaber, and Rawayh Muslim Albaghlany. 2026. Role of inflammatory cytokines in tissue remodeling and disease progression. Open Veterinary Journal, 16 (3), 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 Chicago Style Alyahya, Wasman Jaber, and Rawayh Muslim Albaghlany. "Role of inflammatory cytokines in tissue remodeling and disease progression." Open Veterinary Journal 16 (2026), 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 MLA (The Modern Language Association) Style Alyahya, Wasman Jaber, and Rawayh Muslim Albaghlany. "Role of inflammatory cytokines in tissue remodeling and disease progression." Open Veterinary Journal 16.3 (2026), 1877-1884. Print. doi:10.5455/OVJ.2026.v16.i3.43 APA (American Psychological Association) Style Alyahya, W. J. & Albaghlany, . R. M. (2026) Role of inflammatory cytokines in tissue remodeling and disease progression. Open Veterinary Journal, 16 (3), 1877-1884. doi:10.5455/OVJ.2026.v16.i3.43 |