| Research Article | ||
Open Vet. J.. 2026; 16(3): 1859-1867 Open Veterinary Journal, (2026), Vol. 16(3): 1859-1867 Research Article A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogsSaddam Khalid Hummadi, Hiba Abdulaziz Shekho and Montaser Mohamad Helal*Department of Surgery and Obstetrics, Faculty of Veterinary Medicine, Tikrit University, Tikrit, Iraq *Corresponding Author: : Montaser M. Helal. Department of Surgery and Obstetrics, Faculty of Veterinary Medicine, Tikrit University, Tikrit, Iraq. Email:Montaser.helal1981 [at] tu.edu.iq Submitted: 29/11/2025 Revised: 27/01/2026 Accepted: 06/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Castrating stray dogs using a low-cost, easily performed, and less invasive method can help address public health issues. Aim: This study assessed the effectiveness of a single intratesticular dose of zinc gluconate in inducing sterilization in adult dogs. Methods: 18 adult dogs were divided into 3 groups. The GΙ (zinc gluconate) had an injection volume of 0.2–1 ml per testis; the GП (surgical), in which the testicular blood supply was tied off by double transfixing ligatures; and the GШ (control), in which the testicle was injected with normal saline. Results: Clinically, there were scrotal ulcerations in the GΙ and testicular swelling, surgical incision dehiscence, and infection in the GП, while in the GШ, testicular edema was the only finding. Testicular diameter values were dramatically (p ≤ 0.05) reduced at day 30 in GП (1.48 ± 0.13) compared with GΙ (2.49 ± 0.14) and GШ (3.10 ± 0.13). The serum testosterone level was significantly (p ≤ 0.05) lower in animals of GП (1.17 ± 0.26) at day 30 compared to animals of GΙ (2.55 ± 0.23) and GШ (3.53 ± 0.28). Histopathological sections in GΙ revealed atrophy of the tubuli recti epithelium and degeneration of the seminiferous tubules despite the presence of Leydig cell clusters. While in GП, the main characteristic findings were seminiferous tubule degeneration, Leydig cell atrophy, and vacuole degeneration of the basal and Sertoli epithelium. Conclusion: The injection of buffered zinc gluconate into the testicles caused tissue loss that produced sperm with little impact on testosterone production and had no effect on the general health of animals. Keywords: Castration, Dogs, Surgical, Testicular blood vessels, Zinc gluconate. IntroductionThe spread of floating dogs is a major health problem in civilian communities, as it is mainly responsible for the transmission of many fatal diseases, such as rabies, in humans and animals (Janaa and Samantab, 2007). Uncontrolled breeding in dogs leads to unwanted births and an increased dog population, resulting in public health problems (Slater, 2001). Castration is one of the important strategies used in controlled dog populations (Garcia et al., 2012). It also reduces injuries caused by dog biting by altering the dogs’ contribution and behaviors (Messam et al., 2008) and manages unwanted births (Marston et al., 2004). The time and cost of the surgical castration procedure (Looney et al., 2008), as well as animal management and postoperative care, are required for infection control, making surgical castration an inappropriate method for use in a wide range (Jana et al., 2005). Non-surgical contraceptives are more effective and safer methods that lead to irreversible infertility without adverse effects on animals (Oliveira et al., 2012). Many of these methods, such as immune-contraceptive, hormonal treatments, and chemical intra-epididymis or intra-testicular injections, have become the alternative methods used in the control population of dogs (Kutzler and Wood, 2006). Chemical sterilization is a simple and economical nonsurgical method that can be used in males (Chatterjee et al., 2009; Abd et al., 2025). It has less pain and stress effects and reduces the chance of hernia and hemorrhage, making it the best method for contraception in dogs (Canpolat et al., 2016). Many chemical materials, such as cadmium chloride, glycerol, ferrous sulfate, ferric chloride, and zinc gluconate (Kumar et al., 2018), are used in chemical contraceptives in dogs through intra-testicular, epididymal, or vassal injections to cause a lack of spermatozoa in semen and thus infertility (Kutzler and Wood, 2006). Most of these substances have some disadvantages, such as a short period of sterilizing action and inflammatory reaction (Chatterjee et al., 2009). In 2003, the United States Food and Drug Administration used Zinc gluconate in 2003 for a chemical contraceptive in dogs through intra-testicle injection to cause sclerosis of the testes, then sterility (Masseia and Miller, 2013). Zinc gluconate can be used in adult and young dogs by injecting this material through the disinfectant skin without the need for general anesthesia (Max et al., 2015). Some adverse effects, such as ulceration of the tissue, local pain, lack of appetite, lethargy, and vomiting, may be associated with the use of zinc gluconate as a chemical sterilization in dogs (Oliveira et al., 2012). This study aimed to compare the effectiveness of intratesticular administration of zinc gluconate as a chemical contraceptive method in dogs with that of surgical castration. Materials and MethodsExperimental animalsIn this in vivo investigation, 18 mixed-breed healthy adult male dogs, aged 3–6 years and weighing 20 ± 3 kg, were enrolled. The dogs were placed in standard cages under the same conditions, with a standard pellet diet. The dogs were randomly divided into 3 equal groups. The first group was the zinc gluconate group, the second was the surgical group, and the third was the control group. The animals were acclimated to their environment for 2 weeks. Experimental protocolPreparation of an injectable zinc gluconate solutionZinc gluconate was provided as a 50 mg powder (Solarbio Life Science, China( dissolved and neutralized to pH 7.0 using phosphate buffer saline with a concentration of 13.1 mg/ml, filtered, and sterilized by autoclave before injection. Dosing was correlated with testicular width, and the width of each testicle was measured with a caliper (Fig. 1). Volumes of solution for testes measuring more than 27 mm in width were injected by 1.0 ml/testicle (1 ml is the highest dose). The volume injected per testis ranged from 0.2 to 1 ml (Fagundes et al., 014).
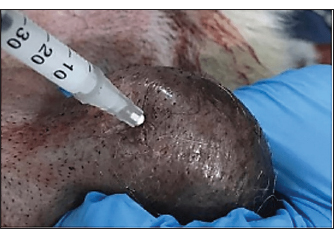
Fig. 1. Measurement of the width of each testicle with a caliper. Administration of ZnG or normal salineDogs in the first group (zinc gluconate) were not anesthetized but were intravenously sedated with medetomidine 1 mg/ml (Chemisch-pharmazeutisches Labor, Germany) at a dose rate of 0.02 mg/kg body weight (Siegenthaler et al., 2020). The scrotum was cleaned using 10% povidone iodine before intratesticular injection. Injection was performed very slowly using a 1.0 ml insulin syringe with a 28-gauge needle. Injections that are too rapid may stimulate contraction of the seminiferous tubules, causing pressure pain and leakage of the drug from the injection site. Leakage may lead to infection and ulceration. The needle was inserted parallel to the testis, into the dorsocranial aspect of the testis, near the epididymis head (Rafatmah et al., 2019) (Fig. 2). The testicle was injected with normal saline in the third group (control) using the same procedure as in the first group.
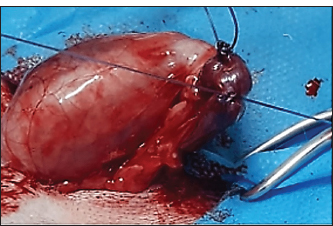
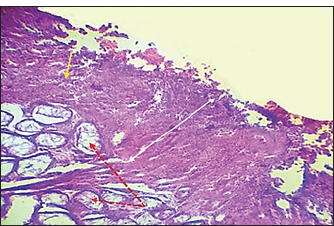
Fig. 2. Insertion of the needle to the dorsocranial side of the testis, close to the epididymis head, and parallel to the testis. Surgical procedureDogs in the second group (surgical group) were fasted for 12 hours before the operation and had free access to water. Then, they were anesthetized using a combination of ketamine hydrochloride 10% (Alfasan, Holland) at a dose rate of 10 mg/kg BW and xylazine hydrochloride 2% (2% xylo-Germany) at a dose rate of 2 mg/kg BW administered via the intramuscular route (Gebremedhin et al., 2018). The animal was placed in a dorsal position. The scrotum and the area directly in front of the scrotum were prepared for aseptic surgery. A single incision of approximately 2 cm in length was made directly in front of the testicles and scrotum. Each testicle is pushed up and through the single incision, and the testicular blood supply (pampiniform plexus and arterial supply) is visualized by double transfixing ligatures using 2–0 Vicryl suture (Fig. 3). The testicle is returned to the scrotum after the vessels are ligated. The incision is closed in multiple layers. Internal sutures are placed to close the subcutaneous tissue, and then a second layer of sutures brings the skin edges together.
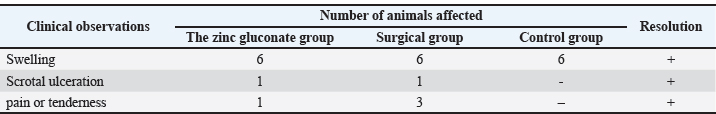
Fig. 3. Visualization of the testicular blood supply (pampiniform plexus and arterial supply) tied off by double transfixing ligatures. Clinical assessmentsThe dogs’ general behavior, as well as scrotal changes, gait observation, and assessment of systemic parameters, such as body temperature, respiratory rates, and heart rate, were assessed. Additionally, the testes width was measured. Evaluation of the testosterone levelsFor testosterone concentration measurement, blood samples were collected from the cephalic vein into plain tubes under aseptic conditions. From all dogs before sterilization (day 0) and after the next 30 days. Serum was harvested after centrifugation (at 3,000 rpm for 5 minutes at room temperature) (Domosławska and Zdunczyk, 2020) and stored at −20°C until testosterone concentration was evaluated. Testosterone was assayed using commercial enzyme immunoassay kits (testosterone ELISA kit AA-E-1300, LDN®, Germany). Histopathological assessmentAll dogs were castrated under anesthesia for microscopic examination, and dissected testes were collected from 18 dogs (6 dogs for each group) at 30 days post-sterilization. The testicles were halved and fixed in 10% neutral buffered formalin, and tissue sections at 4–5 μm thick were routinely prepared and stained with hematoxylin and eosin (Carriel et al., 2017). Statistical analysisThe SAS program was used to perform statistical analysis (Cary, 2018). The mean±standard error (M ± SE) was used to express all data observations, and one-way analysis of variance was used to examine the differences between the two groups. The means were compared using the least significant difference test. A p-value less than or equal to 0.05 was considered significant. Ethical approvalThe ethical clearance committee of the College of Veterinary Medicine, Tikrit University, Iraq, approved the animal experimentation. ResultsDuration of the techniquesThe injection of testicle by zinc gluconate or normal saline consumed approximately 90 seconds for each animal, whereas the surgical procedure, ligating of vessels, required approximately 12 minutes for each animal. Clinical observationsAfter sterilization, the systemic parameters, body temperature, respiratory rate, and heart rate of all dogs in the first group (Zinc gluconate group) were within reference range. No signs of either biting or licking the scrotum were observed, and testicular swelling was reported during 1–2 days after intratesticular injection. No conspicuous testicular pain or tenderness was observed, except for one dog suffering from scrotal ulceration, which was treated surgically by scrotal ablation. In the second group (surgical group), no intraoperative complications were observed. Post-surgery, a slight increase in rectal temperature and a reduction in food intake that lasted for 3 days were observed in three dogs. Testicular swelling was apparent 12 hours after surgery and gradually reduced within 3–5 days after the operation. All surgical incisions healed normally within 10 days post-surgery, except for one dog that showed dehiscence and infection. In the third group, no adverse signs or symptoms were observed, with the exception of testicle edema (Table 1). Table 1. Main clinical observations after intratesticular injection or surgery.
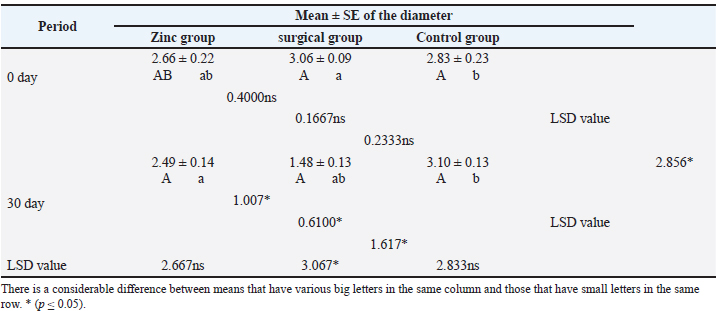
Assessment of the testicular diameterTable 2 shows the mean estimation of testicular diameters. The values of testicular diameter were dramatically (p ≤ 0.05) reduced at day 30 in the surgical group (1.48 ± 0.13) compared with the zinc gluconate group (2.49 ± 0.14) and the control group (3.10 ± 0.13) at the same period. Table 2. Testicular diameter of dogs at day 30th of the study.
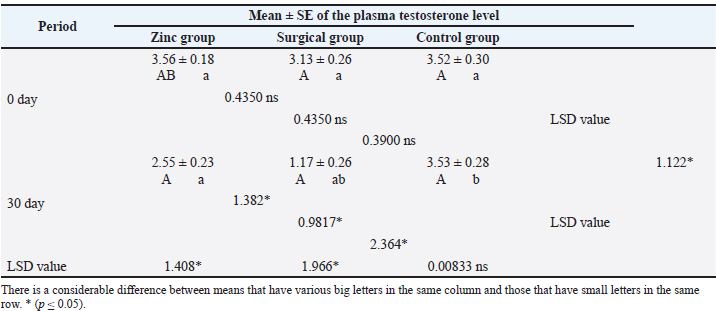
Evaluation of testosterone levelsThe baseline serum testosterone concentration (Table 3) was significantly (p ≤ 0.05) declined in animals of the surgical group (1.17 ± 0.26) at day 30 than in the zinc gluconate group (2.55 ± 0.23) and control group (3.53 ± 0.28) at the same time. Conversely, there was a significant (p ≤ 0.05) decline in testosterone levels in the zinc gluconate group compared with the control group. Table 3. Evaluation of testosterone levels (ng/ml) in the treated and control groups.
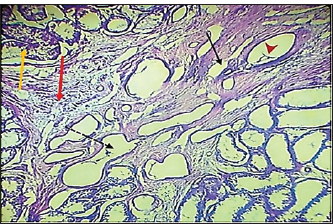
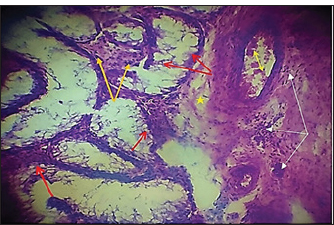
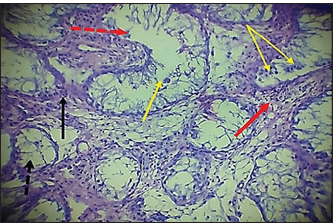
Histopathological evaluationThe zinc gluconate groupIn the GI group, 30 days post-injection, the testes section showed atrophy of the tubuli recti with dilatation of others, desquamation of the tubuli recti epithelium, infiltration of inflammatory cells, and degeneration of seminiferous tubules (Fig. 4). Fibroblast proliferation was observed around the thickened walls of blood vessels (Fig. 5). Other sections showed complete seminiferous tubule stroma necrosis with necrosis in tunica albuginea (Fig. 6). Despite the presence of Leydig cell clusters, some sections displayed germinal cell exfoliation in the lumen of seminiferous tubules accompanied by edematous interstitial tissue and inflammatory cell infiltration (Fig. 7).
Fig. 4. Histopathological section of testes from a dog in the zinc gluconate group at 30 days post-intrasticular injection, showing atrophy of the tubuli recti (black arrow) with dilatation of other (dotted black arrow) , disequilibrium of the tubuli recti epithelium (red head), infiltration of granulation inflammatory cells (red arrow), and degeneration of seminiferous tubules (yellow arrow), (H & E stain 10X).
Fig. 5. Histopathological section of testes from dogs in the zinc gluconate group at 30 days post-intratestinal injactin, showing degeneration of the tubule in the testis recti (black dotted arrow), granuloma (black arrow), congestion (red arrow), thickening wall of blood vessels (red dotted arrow), and proliferation of fibroblasts (yallow arrow),(H & E stain 10X).
Fig. 6. Histopathological section of testes from a dog of the zinc gluconate group at 30 days post-intrasticular injection, showing complete necrosis of seminiferous tubules (red stars) and stroma (black arrow), with necrosis in tunica albuginea(red dotted arrow)(H & E stain 10X).
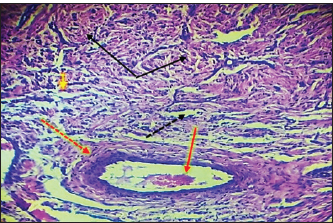
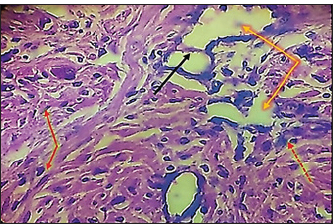
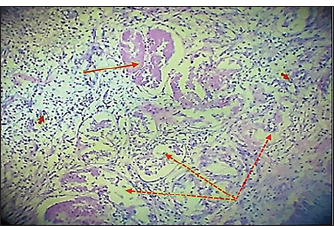
Fig. 7. Histopathological section of testes from a dog in the zinc gluconate group at 30 days post-intrasticular injection, showing exfoliation of germ cells (red arrows), infiltration of inflammatory cells (white arrows), Leydig cell clusters (yellow arrow), edema (star), and congestion (yellow head arrow) (H & E stain 10X). Surgical groupIn the surgical group (GII), the main characteristic findings at 30 days post-surgery were degeneration of seminiferous tubules, multinucleated spermatid, infiltration of inflammatory cells, atrophy of Leydig cells, and vacuole degeneration of basal and Sertoli epithelium (Fig. 8). Furthermore, there was loss of the straight shape of the tubuli recti, disequimentaion of the tubuli recti epithelium, and proliferation of myoid cells (Fig. 9). Other sections showed coagulative necrosis and ghost tubules in the rete testis (Figs. 10 and 11).
Fig. 8. Histopathological section of testes from dogs in the surgical group at 30 days post-surgery, showing degeneration of seminferous tubules (red dotted arrow), multinucleated spermatid (yellow arrows), infiltration of inflammatory cells (red arrow), atrophy of Leydig cells (black arrow), and vacuole degeneration of basel and sertoli epithelium (black dotted arrow) (H & E stain 10X).
Fig. 9. Histopathological section of testes from a dog in the surgical group at 30 days post-surgery, showing loss of the straight shape of the tubuli recti (yellow arrow), disequilibrium of the tubuli recti epithelium (black arrow), proliferation of myoid cells (red arrow), and infiltration of inflammatory cells (red dotted arrow) (H & E stain 40X).
Fig. 10. Histopathological section of testes from a dog in the surgical group at 30 days post-surgery, showing coagulative necrosis (red arrow), ghost tubules in the rete testis (red dotted arrow), and infiltration of inflammatory cells (red head arrow) (H & E stain 10X).
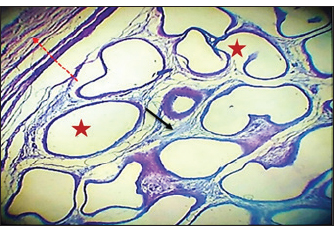
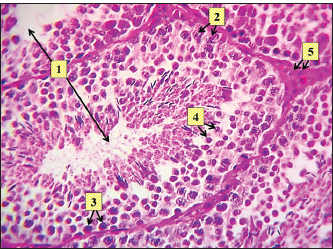
Fig. 11. Histopathological section of testes from a dog in the surgical group at 30 days post-surgery, showing tunica albuginea thickening (white arrows with two heads), inflammatory cell infiltration (yellow arrow), and vacuolar degeneration (red dotted arrow) (hematoxylin and eosin stain 4X). Control groupIn the control group, the histopathological testicular section expresses interstitial septa that surround the seminiferous tubules and contain Leydig cells. Various stages of spermatogenesis and Sertoli cells were also recognized (Fig. 12).
Fig. 12. Histopathological section of testes from a dog of the control group at 30 days post-injection, showing seminiferous tubule (1) containing Sertoli cells (2), spermatogonia (3), and spermatids (4), and identifying interstitial tissue including Leydig cells (5) (H & E stain X 10). DiscussionAlthough most castration-related consequences are minor and easily resolved, life-threatening complications can develop (Kilcoyne, 2013). To prevent surgical complications, the current study was designed to evaluate the effectiveness of a single intratesticular injection dose of zinc gluconate. Additionally, the present study may provide valuable information for a less invasive, successful substitute for surgical sterilization in the control of the stray dog population, in addition to addressing associated public health concerns. Castration with the injection of zinc gluconate needed a shorter time (90 seconds for each animal) to perform when compared with the surgical procedure, which can be performed within 12 minutes/testis (Hamed et al., 2023). The short time of the zinc gluconate injection techniques is important when trying to castrate a stray dog population, in which the injection is done rapidly, also with low cost (more economically), and easily performed, and does not require a specialized surgeon. In the present study, the animals of the GI group showed systemic parameters: body temperature, respiratory rate, and heart rate, within the reference range. Testicular swelling was reported during 1–2 days after intratesticular injection. Laku et al. (2021)indicated that the resolution of post-castration–associated swelling and inflammation shows the repair of injured tissue and the interruption of spermatogenesis. No conspicuous testicular pain or tenderness was observed. This result is in line with earlier studies on chemical castration (Brito et al., 2011; Oliveira et al., 2013), except for one dog in the current study suffering from scrotal ulceration, which may be attributed to the missed intertesticular injection of a chemical agent as explained by Cedillo et al. (2006). A gradual injection method must be applied to prevent medication leakage from the injected site, which may lead to scrotal ulceration. In the surgical group (GII), there were no intraoperative complications. Post-surgery, there was a slight increase in rectal temperature and a reduction in food intake that lasted for 3 days in three dogs. Testicular swelling was apparent 12 hours after surgery and gradually reduced within 3–5 days after the operation. These findings are attributed to acute inflammation, which leads to congestion and edema with signs of testicle swelling (Schwarz et al., 2023). There was no significant (p ≤ 0.05) reduction in testicular diameter observed in the zinc gluconate-treated group (2.49 ± 0.14) compared with the control group (3.10 ± 0.13) at day 30 post intra-testicular injection, as previously reported (Mohamed et al., 2024). Several researchers, such as Garde et al. (2016), have demonstrated the irreversible fibrosis of the epididymis and seminiferous tubules induced by zinc gluconate, which may explain the permanent sterilization in animals of GI in the current study, with no significant reduction in testicular diameter compared to the control group. However, the values of testicular diameter were dramatically (p ≤ 0.05) reduced at day 30 in the surgical group (1.48 ± 0.13) compared with the ZG group. This may be attributed to the interruption of testicular blood flow, which is the primary pathway for the regulation and interchange of all necessary nutrients, oxygen, regulatory hormones, and secretory products to and from the testes (Samir et al., 2021). The baseline concentration of serum testosterone was significantly (p ≤ 0.05) declined in animals of the surgical group (GII) (1.17 ± 0.26) at day 30 compared with animals of the zinc gluconate group (GI) (2.55 ± 0.23) and control group (3.53 ± 0.28) at the same time. Conversely, there was a significant (p ≤ 0.05) decline in testosterone levels in the zinc gluconate group compared with the control group. This finding is consistent with another investigation by Hamed et al. (2021), who showed that serum testosterone levels were not significantly decreased in donkeys treated with zinc gluconate from 15 to 60 days post-intra-testicular administration. Oliveira et al. (2013) found no discernible difference in testosterone levels between the treatment and control groups at any time after receiving an intratesticular injection of zinc gluconate as a permanent contraceptive method in male cats. Rafatmah et al. (2019) confirmed the findings of the current study by indicating the minimal toxicity of zinc gluconate on Leydig cells, which consequently leads to a minimal reduction in the levels of circulating testosterone compared to normal levels. Furthermore, the source of testosterone was not completely eradicated in the chemical castration protocols (Kutzler and Wood, 2006). This may preserve some preferred behaviors of the owners, such as guarding (Palestrini et al., 2021). In the current study, the histopathological evaluation of the testes in the GI group showed atrophy of the tubuli recti with dilatation of others, desquamation of the epithelium, infiltration of inflammatory cells, and degeneration of the seminiferous tubules. Proliferation of fibroblasts around the thickened walls of blood vessels. Complete necrosis of the seminiferous tubule stroma with necrosis in the tunica albuginea. Despite the presence of Leydig cell clusters, some sections displayed germinal cell exfoliation in the lumen of seminiferous tubules, accompanied by edematous interstitial tissue and inflammatory cell infiltration. Similar histopathological findings were reported by Hamed et al. (2023), who administered zinc gluconate as a chemical castration in male donkeys intra-testicularly. These alterations to the testicle structure probably lead to sterility, which is consistent with the findings of Fagundes et al. (2014), who reported that zinc gluconate promotes inflammation and necrosis of the testicles, as observed in autoimmune orchitis. In the surgical group (GII), the main characteristic findings at 30 days post-surgery were degeneration of seminiferous tubules, multinucleated spermatid, infiltration of inflammatory cells, atrophy of Leydig cells, and vacuole degeneration of basal and Sertoli epithelium. Furthermore, the tubuli recti lose their straight shape, the tubuli recti epithelium disequilibrates, and myoid cells proliferate. The other section showed coagulative necrosis and ghost tubules in the rete testis. This hardly changes in testicular tissue, which may be due to ischemia resulting from ligation of the testicular blood supply, as explained by (González and Ciancio, 2015; Spaska et al., 2024). ConclusionIn conclusion, the injection of buffered zinc gluconate into the testicles of adult male dogs resulted in the deterioration of spermatogenesis through the degeneration of seminiferous tubules, with little impact on testosterone production, and did not affect the general health of the animals. This indicates that it can be used as an alternative method for surgical castration in male dogs, particularly in cases where owners may be hesitant to pursue invasive procedures. AcknowledgmentsThe College of Veterinary Medicine/University of Tikrit supported this study. We thank all dog owners for their collaboration. Conflict of interestThe authors have no conflicts of interest to disclose. FundingNo grant was received for this study, and it has been self-funded. Authors' contributionsSaddam K. Humadi was in charge of animal care, clinical parameters, and blood sample collection, while Montaser M. Helal served as the corresponding author of the article. Hiba A. Shekho was in charge of research management and study observations. Each author contributed equally to the writing of the article. Data availabilityAll data are provided in the revised manuscript. ReferencesAbd, A.A., Al-Juhaishi, O.A. and Jumma, Q.S. 2025. Effects of sulpiride on the reproductive system of male rats after puberty. World. Vet. J. 15(1), 4248. Brito, L.F.C., Sertich, P.L., Rives, W., Knobbe, M., Piero, F.D. and Stull, G.B. 2011. Effects of intratesticular zinc gluconate treatment on testicular dimensions, echodensity, histology, sperm production, and testosterone secretion in American black bears (Ursus americanus). Theriogenology 75(8), 1444–1452. Canpolat, I., Karabulut, E. and Eroksuz, Y. 2016. Chemical castration of adult and non-adult male dogs with sodium chloride solution. JAVS 9(12), 09–11. Carriel, V., Campos, A., Alaminos, M., Raimondo, S. and Geuna, S. 2017. Staining methods for normal and regenerative myelin in the nervous system. Methods. Mol. Biol. (Clifton. NJ). 1560, 207–218. Cary, N. 2018. Statistical analysis system, User’s guide. Statistical. Version 9.1th ed; SAS. Institute Inc. USA. Cedillo, V., Pino, F. and Monroy, O. 2006. Results of the massive sterilization project using gluconate neutralized with arginine in male dogs living in the States of Hidalgo, Mexico and Puebla. In XVII Rabies in the Americas, Brasılia, Brazil RITA; 2016. p. 15–20. pp: 15–20. Chatterjee, K., Ali, K.M., De, D., Mallick, C. and Ghosh, D. 2009. Induction of chemosterilization by single intratesticular calcium chloride injection in stray dogs. Res. J. Ani. Vet. Sci. 4, 22–29. Domosławska, A. and Zdunczyk, S. 2020. Clinical and spermatological findings in male dogs with acquired infertility: a retrospective analysis. Andrologia 52(11), e13802. Fagundes, A.K.F., Oliveira, E.C.S., Tenorio, B.M., Melo, C.C.S., Nery, L.T.B., Santos, F.A.B., Alves, L.C., Douglas, R.H. and Silva, V.A. 2014. Injection of a chemical castration agent, zinc gluconate, into the testes of cats results in the impairment of spermatogenesis: a potentially irreversible contraceptive approach for this species?. Theriogenology 81(2), 230–236. Garcia, R., Calderón, N. and Ferreira, F. 2012. Consolidação de diretrizes internacionais de manejo de populações caninas em áreas urbanas e proposta de indicadores para seu gerenciamento. RPDSP 32(2), 140–144. Garde, E., Pérez, G.E., Vanderstichel, R., Dalla Villa, P.F. and Serpell, J.A. 2016. Effects of surgical and chemical sterilization on the behavior of free-roaming male dogs in Puerto Natales, Chile. Prev. Vet. Med. 123, 106–120. Gebremedhin, Y., Negash, G. and Fantay, H. 2018. Clinical Evaluation of Anesthetic Combinations of Xylazine-Ketamine, Diazepam- Page 2 of 9 Ketamine and Acepromazine-Ketamine in Dogs of Local Breed in Mekelle, Ethiopia. SOJ Vet Sci., 4(2), 1–9. González, J. and Ciancio, G. 2015. Vascular diseases of the testis. PanVas. Med. 3855, 3855–3886. Hamed, M.A., Amin, Y.A., Mohamed, R.H., El-Adl, M., Bazeed, S.M., Elnegiry, A.A., Shawki, H.H. and Al-Lethie, A.L.A. 2023. Evaluation of chemical castration using intra-testicular injection of zinc gluconate into the testis of the male donkey versus surgical castration: antimullerian hormone as an endpoint marker. BMC Vet. Res. 19(1), 140. Hamed, M., Soliman, H. A. M., Badrey, A. E. A., and Osman, A. G. M. 2021. Microplastics induced histopathological lesions in some tissues of tilapia (Oreochromis niloticus) early juveniles. Tissue and Cell, 71, 101512. Jana, K. and Samanta, P.K. 2007. Sterilization of male stray dogs with a single intratesticular injection of calcium chloride: a dose dependent study. Contraception 75, 390–400. Jana, K., Samanta, P.K. and Ghosh, D. 2005. Evaluation of single intratesticular injection of calcium chloride for nonsurgical sterilization of male Black Bengal goats (Capra hircus): a dose dependent study. Anim. Reprod. Sci. 86, 89–108. Kilcoyne, I. 2013. Equine castration: a review of techniques, complications and their management. EVE 25(9), 476–482. Kutzler, M. and Wood, A. 2006. Non-surgical methods of contraception and sterilization. Theriogenology 66(3), 514–525. Kumar, R., Soni, N., Kumar, S. and Pandey, A.K. 2018. Chemical control of fertility in male dogs: a review. Int. J.Curr. Microbiol. App. Sci., 7(7), 1760–1773. Laku, D., Mohammed, A. and Bukar, M.M. 2021. Effectiveness of castration with burdizzo, calcium chloride and olive oil in Sahel bucks. Sahel J. Vet. Sci. 18(1), 13–20. Looney, A., Bohling, M.W., Bushby, P.A., Howe, L.M. and Scarlett, J.M. 2008. The Association of Shelter Veterinarians veterinary medical care guidelines for spay-neuter programs. JAVMA 233(1), 74–86. Marston, L.C., Bennett, P.C. and Coleman, G.J. 2004. What happens to shelter dogs? An analysis of data for 1 year from three Australian shelters. J. Appl. Anim. Welf. Sci. 7(1), 27–47. Massei, G. and Miller, L.A. 2013. Nonsurgical fertility control for managing freeroaming dog populations: a review of products and criteria for field applications. Theriogenology 80, 829–838. Max, A., Jurka, P., Dobrzyński, A. and Rijsselaere, T. 2015. Non-surgical contraception in male dogs and cats. Acta. Sci. Pol. Zootechnica. 14(1), 3–14. Messam, L.L.M., Kass, P.H., Chomel, B.B. and Hart, L.A. 2008. The human-canine environment: a risk factor for nonplay bites?. Vet. J. 177(2), 205–215. Mohamed, A., Fathi, M., El-Shahat, K.H., Shamaa, A.A., Bahr, M.M. and El-Saied, M.A. 2024. Chemical castration in dogs using calcium chloride: effects on testicular hemodynamics and semen characteristic and serum levels of testosterone. BMC. Vet. Res. 20(1), 511. Oliveira, E.C.S., Fagundes, A.K.F., Melo, C.C.S., Nery, L.T.B., Rêvoredo, R.G., Andrade, T.F.G., Oliveira-Esquerre, K., Kastelic, J.P. and Silva, V.A. 2013. Intratesticular injection of a zinc-based solution for contraception of domestic cats: a randomized clinical trial of efficacy and safety. Vet. J. 197(2), 307–310. Oliveira, E.C.S., Moura, M.R.P., De Sá, M.J.C., Silva, V.A., Kastelic, J.P., Douglas, R.H. and Marques, A.P. 2012. Permanent contraception of dogs induced with intratesticular injection of a Zinc Gluconate-based solution. Theriogenology 77(6), 1056–1063. Palestrini, C., Mazzola, S.M., Caione, B., Groppetti, D., Pecile, A.M., Minero, M. and Cannas, S. 2021. Influence of gonadectomy on canine behavior. Animals 11(2), 553. Rafatmah, D., Mogheiseh, A. and Eshghi, D. 2019. Chemical sterilization with intratesticular administration of zinc gluconate in adult dogs: a preliminary report. Basic Clin. Androl. 29, 1–9. Samir, H., Radwan, F. and Watanabe, G. 2021. Advances in applications of color Doppler ultrasonography in the andrological assessment of domestic animals: a review. Theriogenology 161, 252–261. Schwarz, L., Dürlinger, S., Martin, V., Weißenböck, H., Brunthaler, R., Rümenapf, T., Auer, A., Loncaric, I., Zimpernik, I., Reisinger, N., Behler-Wöchtl, B., Scarlet, D., Althouse, G., Kuster, C., Kauffold, J., Segales, J., Laitat, M., Thilmant, P., Grahofer, A. and Ladinig, A. 2023. A single, episodic event of unilateral/bilateral scrotal swelling in a group of adult boars at an Austrian boar stud. Porcine Health Manag. 9(1), 17. Siegenthaler, J., Pleyers, T., Raillard, M., Spadavecchia, C. and Levionnois, O.L. 2020. Effect of Medetomidine, Dexmedetomidine, and Their Reversal with Atipamezole on the Nociceptive Withdrawal Reflex in Beagles. Animals 10(7), 1240. Slater, M.R. 2001. The role of veterinary epidemiology in the study of free-roaming dogs and cats. Prev. Vet. Med. 48(4), 273–286. Spaska, A., Grytsuliak, B. and Dolynko, N. 2024. Pathological and vascular changes in the rat testicle after experimental trauma. Clin. Exp. Reprod. Med. 52(2), 178. | ||
| How to Cite this Article |
| Pubmed Style Hummadi SK, Shekho HA, Helal MM. A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Vet. J.. 2026; 16(3): 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 Web Style Hummadi SK, Shekho HA, Helal MM. A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. https://www.openveterinaryjournal.com/?mno=300297 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.41 AMA (American Medical Association) Style Hummadi SK, Shekho HA, Helal MM. A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Vet. J.. 2026; 16(3): 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 Vancouver/ICMJE Style Hummadi SK, Shekho HA, Helal MM. A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 Harvard Style Hummadi, S. K., Shekho, . H. A. & Helal, . M. M. (2026) A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Vet. J., 16 (3), 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 Turabian Style Hummadi, Saddam Khalid, Hiba Abdulaziz Shekho, and Montaser Mohamad Helal. 2026. A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Veterinary Journal, 16 (3), 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 Chicago Style Hummadi, Saddam Khalid, Hiba Abdulaziz Shekho, and Montaser Mohamad Helal. "A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs." Open Veterinary Journal 16 (2026), 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 MLA (The Modern Language Association) Style Hummadi, Saddam Khalid, Hiba Abdulaziz Shekho, and Montaser Mohamad Helal. "A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs." Open Veterinary Journal 16.3 (2026), 1859-1867. Print. doi:10.5455/OVJ.2026.v16.i3.41 APA (American Psychological Association) Style Hummadi, S. K., Shekho, . H. A. & Helal, . M. M. (2026) A comparative study of surgical and chemical castration using zinc gluconate as a contraceptive agent in male dogs. Open Veterinary Journal, 16 (3), 1859-1867. doi:10.5455/OVJ.2026.v16.i3.41 |