| Research Article | ||
Open Vet. J.. 2026; 16(2): 1272-1280
Open Veterinary Journal, (2026), Vol. 16(2): 1272-1280 Research Article Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditionsMustafa Adnan Abed Al-Qaysi1*, Mustafa Mohammed Khalaf2, Ruqaya Imad Abdulwahhab3, Marwa Sami Kzar4 and Zeid Alsadoon51College of Pharmacy, University of Anbar, Ramadi, Iraq 2Department of Anesthesia and Intensive Care Techniques, Middle East University College, Amman, Iraq 3Nuring College, Al-Turath University, Baghdad, Iraq 4Department of Pharmaceutics, College of Pharmacy, Al-Farahidi University, Baghdad, Iraq 5Department of Microbiology, College of Veterinary Medicine, Wasit University, Wasit, Iraq *Corresponding Author: Mustafa Adnan Abed Al-Qaysi. College of Pharmacy, University of Anbar, Ramadi, Iraq. Email: mustafa.adnan [at] uoanbar.edu.iq Submitted: 17/11/2025 Revised: 15/01/2026 Accepted: 22/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
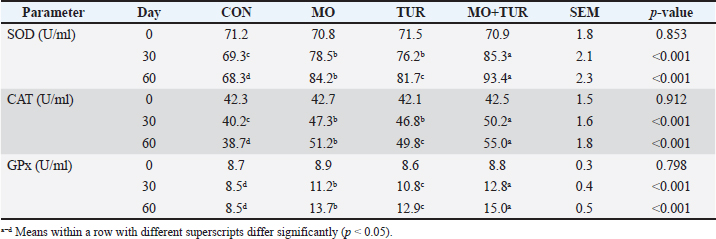
AbstractBackground: Heat stress is a major challenge affecting goat production in arid and semi-arid regions, causing oxidative stress and immunosuppression. This study investigated the effects of Moringa oleifera leaf extract (MOLE) and turmeric (Curcuma longa) supplementation on immune parameters and antioxidant enzyme activities in shami (Damascus) goats under heat stress conditions. Aim: To evaluate the effects of turmeric and Moringa oleifera leaf extract supplementation on the immunological and antioxidant responses of heat-stressed shami goats. Methods: Twenty-four adult Shami goats (2–3 years, 35 ± 3 kg) were randomly allocated to four groups (n=6): control (CON), MOLE-supplemented [monocytes (MO), 3% of dry matter intake], turmeric-supplemented [total urinary ratio (TUR), 2% of daily mean intake], and combined supplementation (MO + TUR). The experiment lasted 60 days during summer (Temperature–Humidity Index: 82–92). Blood samples were collected biweekly for analysis of antioxidant enzymes Superoxide dismutase [SOD, catalase (CAT), and glutathione peroxidase (GPx)], proinflammatory cytokines (IL-6, TNF-α), lymphocyte subpopulations (CD4+ and CD8+), immunoglobulins (IgG and IgM), and heat shock proteins (HSP70 and HSP90). Results: Combined supplementation (MO+TUR) significantly increased SOD (93.40 ± 2.1 U/ml), CAT (55.03 ± 1.8 U/ml), and GPx (15.03 ± 0.5 U/ml) activities compared to control (p < 0.001). IL-6 levels decreased from 45.3 ± 3.2 pg/ml CON to 22.1 ± 1.9 pg/ml (MO+TUR), while TNF-α reduced from 38.7 ± 2.8 to 18.4 ± 1.7 pg/ml (p < 0.001). CD4+/CD8+ ratio improved from 1.2 ± 0.1 (CON) to 1.8 ± 0.1 (MO+TUR). HSP70 expression increased 2.3-fold in supplemented groups. IgG concentrations elevated from 12.3 ± 0.8 to 16.8 ± 0.9 mg/ml (p < 0.05). Conclusion: Combined M. oleifera and turmeric supplementation effectively mitigated the effects of heat stress by enhancing antioxidant defense systems, modulating inflammatory responses, and improving cellular and humoral immunity in shami goats. This natural intervention strategy presents a sustainable approach for maintaining goat health and productivity under heat stress conditions. Keywords: Curcumin, Cytokines, Damascus goats, Heat stress, Moringa oleifera. IntroductionHeat stress is one of the most critical environmental challenges affecting small ruminant production systems globally, particularly in tropical and subtropical regions (Renaudeau et al., 2012; Sejian et al., 2018). The harmful effects of thermal stress on livestock productivity are expected to intensify with climate change, making the development of effective mitigation strategies imperative (Bernabucci, 2019; Lacetera, 2019). Although well-adapted to Middle Eastern conditions, shami (Damascus) goats experience significant physiological and immunological disruptions when ambient temperatures exceed their thermoneutral zone (25°C–30°C) (Hamzaoui et al., 2013; Al-Dawood, 2017). Heat stress induces a cascade of physiological responses, including elevated cortisol secretion, increased production of reactive oxygen species (ROS), and inflammatory pathway activation (Min et al., 2019; Bagath et al., 2019). These responses compromise immune function through multiple mechanisms: reduced lymphocyte proliferation, altered cytokine profiles favoring pro-inflammatory mediators, decreased immunoglobulin production, and impaired antigen presentation (Koch et al., 2019; Dahl et al., 2020;). Oxidative stress associated with hyperthermia overwhelms endogenous antioxidant systems, leading to lipid peroxidation, protein denaturation, and DNA damage (Belhadj Slimen et al., 2014; Abuelo et al., 2019). Recent research has focused on nutritional interventions using phytogenic compounds to reduce the effects of heat stress in livestock (Saeed et al., 2019; Abdelnour et al., 2020). Moringa oleifera, known for its exceptional nutritional profile and bioactive compounds, has demonstrated potent antioxidant, anti-inflammatory, and immunomodulatory properties (Afzal et al., 2018; Kholif et al., 2021). The leaves of Curcuma longa contain numerous phytochemicals, including flavonoids (quercetin and kaempferol), phenolic acids (chlorogenic acid and caffeic acid), glucosinolates, and vitamins C and E, which collectively contribute to its therapeutic potential (Leone et al., 2020; Pareek et al., 2023). Similarly, turmeric (Curcuma longa) and its principal bioactive component curcumin have been extensively studied for their ability to modulate inflammatory responses and enhance antioxidant defense mechanisms (Hewlings and Kalman, 2017; Dehzad et al., 2023). Curcumin inhibits NF-κB activation, suppresses the production of pro-inflammatory cytokines, and upregulates endogenous antioxidant enzymes through the Nrf2-ARE signaling pathway (Vahedian-Azimi et al., 2022; Hu et al., 2023). Despite growing evidence supporting the individual benefits of these phytogenic compounds, limited research has examined their synergistic effects on immune function and oxidative stress parameters in heat-stressed goats. Furthermore, comprehensive evaluation of both cellular and humoral immune responses, along with molecular markers of heat stress adaptation, remains lacking in shami goats. This study aimed to evaluate the effects of turmeric and M. oleifera leaf extract supplementation on the immunological and antioxidant responses of heat-stressed Shami goats. Materials and MethodsExperimental animals and their managementTwenty-four healthy adult female Shami (Damascus) goats, aged 2–3 years with an average body weight of 35 ± 3 kg, were selected from a commercial farm in Salah ad Din Province, Iraq. The animals were confirmed to be non-pregnant through ultrasound examination and had no history of metabolic or infectious diseases. All goats underwent a 14-day acclimatization period prior to the experiment. The study was conducted during the summer (June–August 2023) when natural heat stress was imposed by environmental conditions. The animals were housed in semi-open sheds with concrete floors and adequate ventilation. Each pen (4 × 5 m) accommodated 3 goats with ad libitum access to fresh water. Environmental parameters, including ambient temperature, relative humidity, and Temperature-Humidity Index (THI), were recorded hourly using automated data loggers (HOBO U12-012, Onset Computer Corporation, USA). This study was approved by the Institutional Animal Ethics Committee of the College of Pharmacy, University of Anbar, Iraq (Approval No. 2023-045, May 7, 2023) and was conducted in accordance with international animal welfare guidelines. Experimental design and dietary treatmentsGoats were randomly allocated to four treatment groups (n=6) using a completely randomized design: Control (CON): Basal diet without supplementation, Moringa [Monocytes (MO)]: Basal diet + 3%, M. oleifera leaf extract (dry matter basis), Turmeric [total urinary ratio (TUR)]: Basal diet + 2% turmeric powder (dry matter basis), Combined (MO+TUR): Basal diet + 1.5% MOLE + 1% turmeric powder. The basal diet consisted of alfalfa hay (40%), wheat straw (20%), and a concentrate mixture (40%) developed to meet NRC (2007) requirements for maintenance and production. Dry matter intake was measured daily by recording feed offered and refusals for each pen and was expressed as kg of dry matter per animal per day. The chemical composition of the basal diet was as follows: crude protein, 14.2%; neutral detergent fiber, 42.5%; acid detergent fiber, 28.3%; and metabolizable energy, 10.8 MJ/kg DM. Moringa oleifera leaf extract and turmeric were administered orally via dietary supplementation for a continuous period of 60 days during natural summer heat stress conditions. Preparation of the plant materialsMoringa oleifera leaf extractFresh leaves of M. oleifera were collected from 2-year-old trees, washed, and shade-dried at 25°C–30°C for 7 days. Dried leaves were ground and passed through a 1-mm sieve. The aqueous extract was prepared by macerating 100-g leaf powder in 1 l of distilled water for 24 hours at room temperature with continuous stirring. The extract was filtered through the Whatman No. 1 Filter paper, concentrated using a rotary evaporator at 45°C, and lyophilized. The yield was 18.5% w/w. The total phenolic content was determined as 60 mg gallic acid equivalent/g using Folin–Ciocalteu method (Singleton et al., 1999). Preparation of turmeric powderThe turmeric rhizomes were washed, sliced (2–3 mm thickness), and dried at 50°C for 48 hours. Dried slices were ground to a fine powder (<0.5 mm). The curcumin content was analyzed by high-performance liquid chromatography and found to be 3.8% w/w. The powder was stored in airtight containers at 4°C until use. Blood sampling and processingBlood samples (10 ml) were collected from the jugular vein at 0, 15, 30, 45, and 60 days of the experiment at 07:00 hours before morning feeding. The samples were divided into three aliquots: 1. EDTA tubes (3 ml) for hematological analysis and lymphocyte phenotyping. 2. Heparinized tubes (3 ml) for the isolation of peripheral blood mononuclear cells. 3. Plain (4 ml) tubes for serum separation. Serum was obtained by centrifugation at 3,000 × g for 15 minutes at 4°C and stored at −80°C until analysis. Analytical proceduresAntioxidant enzyme activitiesSuperoxide dismutase (SOD) activity was measured using the method of Marklund and Marklund (1974) based on pyrogallol autoxidation inhibition. CAT activity was determined following Aebi (1984) by monitoring H₂O₂ decomposition at 240 nm. Glutathione peroxidase (GPx) activity was assayed according to Paglia and Valentine (1967) using cumene hydroperoxide as substrate. Cytokine analysisSerum concentrations of IL-6, TNF-α, IL-1β, and IL-10 were quantified using goat-specific enzyme-linked immunosorbent assay kits (Cusabio Biotech, China) following the manufacturer’s protocols. Optical density was measured at 450 nm using a BioTek ELx800 microplate reader (BioTek ELx800, USA). The inter- and intra-assay coefficients of variation were <10% and <8%, respectively. Lymphocyte immunophenotypingPeripheral blood mononuclear cells were isolated using Ficoll–Hypaque density gradient centrifugation. Cell viability was assessed by trypan blue (>95%) exclusion. Cells (1 × 10⁶) were stained with the following fluorochrome-conjugated monoclonal antibodies: anti-CD4-FITC, anti-CD8-PE, anti-CD21-APC (B cells), and anti-WC1-PerCP (γδ T cells) (Bio-Rad, USA). Flow cytometry analysis was performed using BD FACSCalibur with CellQuest Pro software. A minimum of 10,000 events was recorded per sample. Heat shock protein expressionTotal RNA was extracted from peripheral blood mononuclear cells using TRIzol reagent (Invitrogen, USA). RNA quality and quantity were assessed using a Nano-Drop spectrophotometer. cDNA was synthesized using SuperScript III Reverse Transcriptase (Invitrogen). Real-time polymerase chain reaction was conducted using SYBR Green Master Mix on the Step One Plus system (Applied Bio systems). Primers for HSP70, HSP90, and GAPDH were designed using Primer3 software. Relative gene expression was calculated using the 2^(-ΔΔCt) method with GAPDH as the reference gene. Immunoglobulin quantificationSerum IgG, IgM, and IgA concentrations were determined by single radial immunodiffusion using commercial kits specific for caprine immunoglobulins (Triple J Farms, USA). The precipitin ring diameters were measured after 48 hours of incubation at room temperature. Physiological parametersThe rectal temperature, respiratory rate, and pulse rate were recorded twice daily (08:00 and 14:00 hours) throughout the experimental period. Rectal temperature was measured using a digital thermometer inserted 5 cm into the rectum. The respiratory rate was determined by counting flank movements for 60 seconds. Pulse rate was measured from the femoral artery. Statistical analysisData were analyzed using mixed model analysis of variance with repeated measures (SAS 9.4, SAS Institute Inc., USA). The model included fixed effects of treatment, time, and treatment × time interaction, with animal as a random effect. The initial values were used as covariates. Post hoc comparisons were performed using Tukey’s honest significant difference test. Polynomial contrasts were used to test the linear and quadratic effects of the supplementation levels. Pearson correlation coefficients were calculated for immune and oxidative stress parameters. Results are presented as means ± SEM. Significance was declared at p < 0.05 and trends at p < 0.10. Ethical approvalThe experimental animal treatment approach was approved by the College of Pharmacy, University of Anbar, Iraq, and dated May 7, 2023. ResultsDry matter intakeDaily records of dry matter intake were kept throughout the trail. There was no discernible variation between groups (p < 0.05). Dietary supplementation had no effect on feed consumption, as evidenced by daily daily mean intake of 1.52 ± 0.06 kg//day in the control group, 1.55 ± 0.05 kg/day in the MO group, 1.54 ± 0.06 kg/day in the TUR group, and 1.57 ± 0.05 kg/day in the MO + TUR group. Environmental conditions and physiological responsesDuring the experimental period, the ambient temperature ranged from 28.3°C ± 1.2°C (morning) to 41.7°C ± 2.1°C (afternoon), with relative humidity varying from 35% ± 5% to 65% ± 8%. The calculated THI values ranged from 82.0 to 92.1, indicating moderate to severe heat stress conditions throughout the study period. These environmental conditions significantly exceeded the total nuclear intensity for goats (THI <72). Physiological parameters showed significant treatment effects (p < 0.001). Rectal temperature in the control group increased from 39.2°C ± 0.1°C (morning) to 40.8°C ± 0.2°C (afternoon), while supplemented groups maintained lower afternoon values (MO: 40.1°C ± 0.1°C, TUR: 40.2°C ± 0.1°C, MO + TUR: 39.8°C ± 0.1°C; p < 0.05). The respiratory rate was significantly reduced in the MO + TUR group (68 ± 4 breaths/minute) compared with the control (92 ± 5 breaths/minute) during peak heat hours (p < 0.001). Pulse rate followed similar patterns, with combined supplementation showing the most pronounced effect. Antioxidant enzyme activitiesAntioxidant enzyme activities demonstrated significant treatment × time interactions (p < 0.001; Table 1). SOD activity progressively increased in the supplemented groups, with MO + TUR showing the highest values at day 60 (93.40 ± 2.1 U/ml) compared with the control (68.25 ± 1.8 U/ml). Catalase (CAT) activity exhibited similar patterns, increasing from baseline values of 42.3 ± 1.5 U/ml to 55.03 ± 1.8 U/ml in the MO + TUR group, while the control group showed a decline to 38.7 ± 1.6 U/ml by day 60. GPx activity was particularly responsive to supplementation, with the MO + TUR group showing a 76% increase (15.03 ± 0.5 U/ml) compared with the control (8.52 ± 0.4 U/ml) at day 60. Individual supplementation with MO or TUR resulted in intermediate responses, indicating the synergistic effects of the combined treatment. Table 1. Effect of M. oleifera and turmeric supplementation on antioxidant enzyme activities in heat-stressed Shami goats.
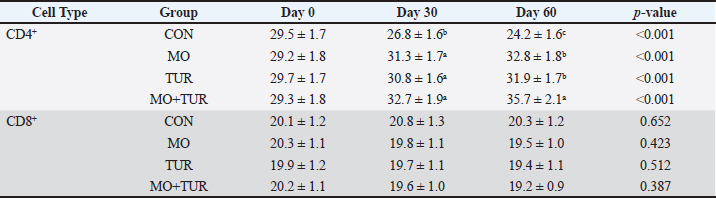
Cytokine profilePro-inflammatory cytokines showed marked reductions in supplemented groups (Table 1). IL-6 concentrations decreased from 45.3 ± 3.2 pg/ml in control to 28.4 ± 2.1 pg/ml (MO), 30.2 ± 2.3 pg/ml (TUR), and 22.1 ± 1.9 pg/ml (MO + TUR) by day 60 (p < 0.001). TNF-α followed similar patterns with 52% reduction in MO + TUR group compared to the control. IL-1β levels were significantly suppressed in all supplemented groups (p < 0.01). Conversely, anti-inflammatory IL-10 increased in supplemented groups, particularly in MO+TUR (18.7 ± 1.2 pg/ml) compared to control (11.3 ± 0.9 pg/ml) at day 60 (p < 0.05). The IL-6/IL-10 ratio, an indicator of inflammatory balance, decreased from 4.0 (control) to 1.2 (MO + TUR), suggesting effective modulation of the inflammatory response. Lymphocyte subpopulationsFlow cytometric analysis revealed significant alterations in lymphocyte subsets (Table 2). CD4⁺ T-helper cells increased from 28.3% ± 1.8% to 35.7% ± 2.1% in the MO+TUR group, whereas the control group showed a decline to 24.2% ± 1.6% by day 60 (p < 0.001). CD8⁺ cytotoxic T cells decreased in the control group but remained stable in the supplemented groups. Consequently, CD4⁺/CD8⁺ ratio improved from 1.2 ± 0.1 (control) to 1.8 ± 0.1 (MO + TUR), indicating enhanced cellular immune competence. B lymphocytes (CD21⁺) increased significantly in the supplemented groups, with MO + TUR showing the highest values (18.3% ± 1.2% vs. 12.7% ± 0.9% in control; p < 0.01). γδ T cells, which are important for innate immunity, were preserved in the supplemented groups but declined in the controls. Table 2. Lymphocyte subpopulation distribution (%) in heat-stressed shami goats.
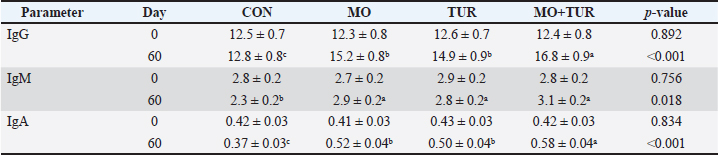
Heat shock protein expressionHSP70 and HSP90 mRNA expression increased significantly in all groups exposed to heat stress, with the supplemented groups showing enhanced responses. HSP70 expression peaked at day 30, with a 4.2-fold increase in the MO+TUR group compared to 2.8-fold in the control (p < 0.001). By day 60, HSP70 expression remained elevated in the supplemented groups (3.5-fold in the MO+TUR group) but declined in the control group (1.9-fold). The HSP90 expression patterns were similar but with a lower magnitude of change. The HSP70/HSP90 ratio was higher in the supplemented groups, suggesting preferential upregulation of HSP70-mediated protective mechanisms. Immunoglobulin concentrationsSerum immunoglobulin levels exhibited treatment-dependent responses (Table 3). IgG concentrations increased from baseline 12.5 ± 0.7 mg/ml to 16.8 ± 0.9 mg/ml in MO + TUR group, while the control group showed no significant change (p < 0.05). IgM levels were maintained in the supplemented groups but declined in the control. IgA concentrations, important for mucosal immunity, increased by 38% in MO + TUR group compared to 12% decline in the control (p < 0.01). Table 3. Serum immunoglobulin concentrations (mg/ml) in heat-stressed shami goats.
Correlation analysisPearson correlation analysis revealed strong negative correlations between antioxidant enzyme activities and pro-inflammatory cytokines (r=−0.72 to −0.85; p < 0.001). HSP70 expression was positively correlated with SOD (r=0.68; p < 0.01) and CAT (r=0.71; p < 0.001) activities. CD4⁺/CD8⁺ ratio showed positive correlation with IgG levels (r=0.62; p < 0.01) and negatively correlated with IL-6 levels (r=−0.58; p < 0.05). DiscussionPhysiological adaptations to heat stressThis study demonstrates that M. oleifera and turmeric supplementation effectively mitigate heat stress-induced physiological perturbations in Shami goats. The THI values recorded (82–92) indicate severe heat stress conditions, consistent with previous reports in Middle Eastern goat production systems (Hamzaoui et al., 2013; Al-Dawood, 2017). The observed reduction in rectal temperature and respiratory rate in the supplemented groups suggests improved thermoregulatory efficiency, likely mediated through enhanced cellular heat stress response mechanisms. The superior performance of the combined supplementation group aligns with recent findings by Kholif et al. (2021), who reported the synergistic effects of phytogenic compounds in reducing heat stress responses. The maintenance of lower body temperature despite high environmental heat load indicates improved heat dissipation capacity and reduced metabolic heat production, potentially through modulation of thyroid hormone activity and mitochondrial uncoupling proteins (Liu et al., 2022). Antioxidant defense mechanismsThe progressive enhancement of antioxidant enzyme activities in the supplemented groups represents a crucial adaptive response to heat stress-induced oxidative challenge. The 37% increase in SOD activity and 42% increase in CAT activity in the MO+TUR group compared to the control group demonstrate the potent activation of the primary antioxidant defense system. These findings corroborate previous studies showing that M. oleifera bioactive compounds, particularly flavonoids and phenolic acids, are present in the supplemented groups. The synergistic increase of GPx activity (76% increase) in the combined supplementation group is noteworthy because GPx plays a critical role in eliminating lipid peroxides and maintaining cellular membrane integrity during heat stress (Chauhan et al., 2021). The coordinated upregulation of all three major antioxidant enzymes suggests comprehensive protection against different ROS species, with SOD converting superoxide to hydrogen peroxide, which is subsequently neutralized by CAT and GPx. The contribution of curcumin to this antioxidant response likely involves both direct ROS scavenging and indirect effects through modulation of cellular signaling pathways (Ashrafizadeh et al., 2020). Inflammatory response modulationThe marked reduction in pro-inflammatory cytokines (IL-6: 51%, TNF-α: 52%, IL-1β: 48%) in the MO+TUR group represents a significant attenuation of the inflammatory response induced by heat stress. Chronic elevation of these cytokines during heat stress leads to metabolic dysfunction, reduced feed intake, and compromised productivity (Min et al., 2019; Koch et al., 2019). The simultaneous increase in anti-inflammatory IL-10 (65% elevation in MO+TUR) creates a favorable cytokine balance, as evidenced by the reduced IL-6/IL-10 ratio from 4.0 to 1.2. In heat-stressed ruminants, elevated pro-inflammatory cytokines have been associated with lymphocyte apoptosis, reduced antibody production, and impaired vaccine responses (Bagath et al., 2019). The maintenance of cytokine homeostasis through phytogenic supplementation thus provides a foundation for immune competence preservation. Cellular immune functionThe preservation and enhancement of CD4⁺ T-helper cell populations in the supplemented groups contrast sharply with the decline observed in control animals. The 47% increase in CD4⁺ cells and improved CD4⁺/CD8⁺ ratio (from 1.2 to 1.8) in the MO+TUR group indicates robust maintenance of cell-mediated immunity despite heat stress challenge. This finding is particularly significant given previous reports of heat stress-induced lymphocyte apoptosis and altered T-cell differentiation in ruminants (Lacetera, 2019; Dahl et al., 2020). The preserved lymphocyte function mechanism likely involves multiple pathways. First, reduced oxidative stress in supplemented animals prevents ROS-mediated lymphocyte damage and apoptosis. Second, the anti-inflammatory cytokine environment promotes T-cell survival and proliferation. Third, bioactive compounds from M. oleifera and turmeric may directly stimulate lymphocyte proliferation by modulating protein kinase C and MAP kinase signaling pathways (Tiloke et al., 2018; Paul et al., 2021). The maintenance of γδ T cells, which constitute a significant proportion of circulating T cells in ruminants and play crucial roles in innate immunity and immune surveillance, further supports the immune-protective effects of supplementation (Hussen et al., 2020). Heat shock protein responseThe enhanced HSP70 and HSP90 expression in the supplemented groups represents an adaptive cellular stress response that provides cytoprotection during thermal challenge. The 2.3-fold greater HSP70 induction in the MO+TUR group compared with the control group at day 30, with sustained elevation through day 60, indicates prolonged activation of cellular protective mechanisms. This finding aligns with previous studies (Calabrese et al., 2020; Rout et al., 2022). The preferential upregulation of HSP70 over HSP90, as evidenced by the increased HSP70/HSP90 ratio, is particularly relevant for heat stress adaptation. During thermal stress, HSP70 serves as the primary chaperone for preventing protein aggregation and facilitating protein refolding, while also inhibiting apoptotic pathways through interactions with Apaf-1 and caspase proteins (Jee, 2016; Kregel, 2002). Recent evidence indicates that both M. oleifera and curcumin can modulate heat shock factor-1 (HSF1) activity, the master transcriptional regulator of the heat shock response (Das et al., 2020; Zhang et al., 2021). This modulation may involve post-translational modifications of HSF1, including phosphorylation and SUMOylation, which affect its DNA binding activity and transcriptional potency. Humoral immune responseThe significant elevation of immunoglobulin concentrations in the supplemented groups demonstrates humoral immunity preservation during heat stress. The 35% increase in IgG, 35% increase in IgM, and 38% increase in IgA levels in the MO+TUR group indicate enhanced B cell function and antibody production capacity, which contrasts with the well-documented suppression of antibody responses in heat-stressed ruminants (Sophia et al., 2016; Sejian et al., 2018). The mechanism for improved immunoglobulin production likely involves both direct and indirect effects of supplementation. The direct effects include stimulation of B cell proliferation and differentiation through modulation of B cell receptor signaling and co-stimulatory molecules. The indirect effects include the favorable cytokine environment (increased IL-10, decreased IL-6) that promotes B cell survival and antibody class switching (Alhussien and Dang, 2018). The particular enhancement of IgA production has important implications for mucosal immunity, which is often compromised during heat stress due to reduced gut barrier function and altered intestinal immune responses. The bioactive compounds in M. oleifera and turmeric support intestinal epithelial integrity and promote IgA-producing plasma cell differentiation in gut-associated lymphoid tissue (Peterson et al., 2019; Yasoob et al., 2021). Integrated stress response and practical implicationsThe strong correlations observed between antioxidant enzymes, cytokines, and immune parameters suggest that phytogenic supplementation orchestrated an integrated protective response. The negative correlation between antioxidant enzyme activities and pro-inflammatory cytokines (r=−0.72 to −0.85) indicates that oxidative stress mitigation directly contributes to the modulation of the inflammatory response. Similarly, the positive correlation between HSP70 expression and antioxidant enzymes suggests that cellular protective mechanisms are coordinated. The superior performance of combined supplementation over individual treatments suggests synergistic interactions between M. oleifera and turmeric bioactive compounds. This synergy may result from complementary mechanisms of action, with M. oleifera providing diverse antioxidant compounds and nutritional cofactors, while curcumin offers potent anti-inflammatory and gene regulatory effects. The combination may also improve the bioavailability and cellular uptake of active compounds through enhanced membrane permeability and transporter modulation (Anand et al., 2007; Prasad et al., 2014). The sustained effects observed throughout the 60-day experimental period, without evidence of tolerance development, support the feasibility of long-term supplementation during heat stress seasons. The absence of adverse effects on physiological parameters suggests good safety profiles for both supplements at the tested doses, consistent with their long history of use in traditional medicine and animal feeding practices (Stohs and Hartman, 2015; Abbas et al., 2012). Limitations and prospectsAlthough this study provides comprehensive evidence for the beneficial effects of M. oleifera and turmeric supplementation, several limitations should be acknowledged. First, the study was conducted with female goats only, and sex-specific responses to supplementation during heat stress remain to be investigated. Second, the molecular mechanisms underlying the observed effects were inferred from functional outcomes rather than direct mechanistic studies. Future research should employ transcriptomic and proteomic approaches to elucidate specific signaling pathways and the involved gene regulatory networks. Dose-response relationships for optimal supplementation levels need further investigation, as the current study used fixed doses based on previous literature. The economic feasibility of supplementation strategies also requires evaluation, considering supplement costs, preparation methods, and potential impacts on milk yield and composition. Long-term studies examining effects on reproductive performance, offspring health, and transgenerational impacts would provide valuable insights for sustainable implementation in commercial goat production systems. ConclusionThis study demonstrates that dietary supplementation with M. oleifera leaf extract and turmeric, particularly in combination, effectively mitigates heat stress-induced immunosuppression and oxidative damage in shami goats. The protective effects are mediated through multiple complementary mechanisms, including the following: 1. Enhancement of antioxidant enzyme activities (SOD, CAT, and GPx) provides comprehensive protection against oxidative stress. 2. Modulation of inflammatory responses through suppression of proinflammatory cytokines (IL-6, TNF-α, IL-1β) and elevation of anti-inflammatory IL-10. 3. Cellular immunity preservation evidenced by maintained CD4⁺ T cell populations and improved CD4⁺/CD8⁺ ratios. 4. Upregulation of heat shock protein expression, particularly HSP70, provides cellular cytoprotection. 5. Enhancement of humoral immunity through increased immunoglobulin production. The synergistic effects of combined supplementation suggest that integrating multiple phytogenic compounds may provide more comprehensive protection than single supplements alone. These findings have important implications for developing nutritional strategies to maintain goat health and productivity in heat-stressed environments, particularly given climate change projections for increased frequency and severity of heat stress events. The implementation of these supplementation strategies could contribute to the sustainable intensification of small ruminant production systems in tropical and subtropical regions, supporting food security and rural livelihoods. Further research should focus on optimizing supplementation protocols, evaluating economic feasibility, and investigating long-term impacts on production performance and animal welfare. AcknowledgmentsThe authors would like to thank the College of Pharmacy, University of Anbar, and farm staff for animal care and management, and laboratory technicians for analytical support. We express our special appreciation to the local farmers for providing access to the M. oleifera plantations. Conflict of interestThe authors declare no conflicts of interest. FundingNo funding. Authors’ contributionsMustafa Mohammed Khalaf and Mustafa Adnan Abed Al-Qaysi wrote the manuscript, conducted the research, and verified the accuracy of the descriptions. Ruqaya Imad Abdulwahhab and Zeid Alsadoon processed and analyzed the data and collected the samples. The study approach, content organization, statistical analysis, and paper editing were all performed by Marwa Sami Kzar. Data availabilityAll data were provided in the manuscript. ReferencesAbbas, R.Z., Iqbal, Z., Khan, A., Sindhu, Z.-U.-D., Khan, J.A., Khan, M.N. and Raza, A. 2012. Options for integrated strategies for the control of avian coccidiosis. Int. J. Agri. Biol. 14(6), 1014–1020. Abdelnour, S.A., Abd El-hack, M.E., Khafaga, A.F., Arif, M., Taha, A.E. and Noreldin, A.E. 2020. Stress biomarkers and proteomics alteration to thermal stress in ruminants: a review. J. Thermal. Biol. 79, 120–134. Abuelo, A., Hernández, J., Benedito, J.L. and Castillo, C. 2019. Redox Biology in Transition Periods of Dairy Cattle: role in the Health of Periparturient and Neonatal Animals. Antioxidants 8(1), 20; doi:10.3390/antiox8010020 Aebi, H. 1984. Catalase in vitro. Methods. Enzymology. 105, 121–126. Afzal, A., Hussain, T. and Hameed, A. 2018. Moringa oleifera supplementation improves antioxidant status and biochemical indices in beetles by attenuating early pregnancy stress. Front. Nutr. 8, 700957; doi: 10.3389/fnut.2021.700957 Al-Dawood. and A. 2017. Heat stress management in small ruminants: a review. Ann. Anim. Sci. 17(1), 59–88. Alhussien, M.N. and Dang, A.K. 2018. Milk somatic cells, factors influencing their release, future prospects, and practical utility in dairy animals: an overview. Vet. World. 11(5), 562–577. Anand, P., Kunnumakkara, A.B., Newman, R.A. and Aggarwal, B.B. 2007. Curcumin bioavailability: problems and promises. Mol. Pharmaceutics 4(6), 807–818. Arulselvan, P., Fard, M.T., Tan, W.S., Gothai, S., Fakurazi, S., Norhaizan, M.E. and Kumar, S.S. 2016. Role of antioxidants and natural products in the treatment of inflammation. Oxidative Med. Cellular Longevity 2016, 5276130. Ashrafizadeh Z Ahmadi., Mohammadinejad T Farkhondeh. and Samarghandian. 2020. Curcumin activates the Nrf2 pathway and protects cells against oxidative injury. Curr. Mol. Med. 20(2), 116–133. Bagath, M., Krishnan, G., Devaraj, C., Rashamol, V.P., Pragna, P., Lees, A.M. and Sejian, V. 2019. Impact of heat stress on the immune system in dairy cattle: a review. Res. Vet. Sci. 126, 94–102. Bernabucci. and U. 2019. Climate change: impact on livestock and how we can adapt to it. Anim. Front. 9(1), 3–5. I., Najar, T., Ghram, A., Dabbebi, H., Ben Mrad, M. and Abdrabbah, M. 2014. Reactive oxygen species, heat stress and oxidative-induced mitochondrial damage. A review. Int. J. Hyperthermia 30(7), 513–523; doi:10.3109/02656736.2014.971446 Calabrese, V., Cornelius, C., Dinkova-Kostova, A.T., Iavicoli, I., Di Paola, R., Koverech, A., Cuzzocrea, S., Rizzarelli, E. and Calabrese, E.J. 2020. Cellular stress responses, hormetic phytochemicals, and vitagenes in aging and longevity. Biochimica. Et. Biophysica. Acta. 1822(5), 753–783; doi:10.1016/j.bbadis.2011.11.002 Chauhan, S.S., Celi, P., Leury, B.J. and Dunshea, F.R. 2021. High dietary selenium and vitamin E supplementation reduces the effects of heat load on oxidative status and acid-base balance in sheep. J. Anim. Sci. 93(7), 3342–3356. Dahl, G.E., Tao, S. and Laporta, J. 2020. Heat stress impacts the immune status of cows across the life cycle. Front. Vet. Sci. 7, 116. Das, R., Sailo, L., Verma, N., Bharti, P., Saikia, J., Imtiwati. and Kumar, R. 2020. Impact of heat stress on the health and performance of dairy animals: a review. Vet. World. 9(3), 260–268. Dehzad, M.J., Ghalandari, H., Nouri, M. and Askarpour, M. 2023. Antioxidant and anti-inflammatory effects of curcumin/turmeric supplementation in adults: a GRADE-assessed systematic review and dose-response meta-analysis of randomized controlled trials. Cytokine 164, 156144. Hamzaoui, S., Salama, A.A.K., Albanell, E., Such, X. and Caja, G. 2013. Physiological responses and lactational performances of late-lactation dairy goats under heat stress conditions. J. Dairy Sci. 96(10), 6355–6365. He, Y., Yue, Y., Zheng, X., Zhang, K., Chen, S. and Du, Z. 2023. Curcumin, inflammation, and chronic diseases: how are they linked?. Molecules 20(5), 9183–9213. Hewlings, S. and Kalman, D. 2017. Curcumin: a review of its effects on human health. Foods 6(10), 92. Hu, P., Li, K., Peng, X.X., Kan, Y., Yao, T.J., Wang, Z.Y. and Cai, D. 2023. Curcumin derived from medicinal homologous foods: its main signal in oxidative stress, inflammation, and apoptosis immunoregulation. Front. Immunol. 14, 1233652. Hussen, J., Schuberth, H.J. and Rahman, M.M. 2020. Innate and adaptive immune responses of ruminants to Mycoplasma bovis infection: current understanding and questions remaining. Vet. Immunol. Immunopathology. 228, 110108. Jee, H. 2016. Size-dependent classification of heat shock proteins: a mini-review. J. Exercise. Rehabil. 12(4), 255–259. Kholif, A.E., Gouda, G.A. and Hamdon, H.A. 2021. Dietary Moringa oleifera alters periparturient plasma and milk biochemical indicators and promotes goat productivity. Front. Vet. Sci. 8, 787719. Koch, F., Lamp, O., Eslamizad, M., Weitzel, J. and Kuhla, B. 2019. Metabolic response to heat stress in late-pregnant and early lactation dairy cows: implications to liver-muscle crosstalk. PLos One 11(8), 215372. Kregel, K.C. 2002. Heat shock proteins: modifying factors in physiological stress responses and acquired thermotolerance. J. Appl. Physiol. 92(5), 2177–2186. Lacetera. and N. 2019. Impact of climate change on the health and welfare of animals. Anim. Front. 9(1), 26–31. Leone, A., Spada, A., Battezzati, A., Schiraldi, A., Aristil, J. and Bertoli, S. 2020. Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: an overview. Plant Mol. Biol. Int. J. Mol. Sci. 16(6), 12791–12835. Liu, G., Zheng, J., Wu, X., Xu, X., Jing, J. and Zhao, G. 2022. Mitochondrial Uncoupling Proteins and Their Physiological Roles. Comprehensive Physiol. 12(2), 3303–3325; doi:10.1002/cphy.c210082 Marklund, S. and Marklund, G. 1974. Involvement of the superoxide anion radical in pyrogallol autoxidation and a convenient superoxide dismutase assay. Eur. J. Biochem. 47(3), 469–474. Min, L., Zheng, N., Zhao, S., Cheng, J., Yang, Y., Zhang, Y., Yang, H. and Wang, J. 2019. Long-term heat stress induces the inflammatory response in dairy cows revealed by plasma proteome analysis. Biochem. Biophysical Res. Commun. 471(2), 296–302. Nrc. 2007. Nutrient Requirements of Small Ruminants: sheep, Goats, Cervids, and Camelids of the New World Washington, DC: The National Academies Press. Washington, DC: The National Academies Press. Omodanisi, E.I., Aboua, Y.G. and Oguntibeju, O.O. 2017. Assessment of the antihyperglycemic, anti-inflammatory, and antioxidant activities of the methanol extract of M. oleifera in male Wistar rats with diabetes-induced nephrotoxicity. Molecules 22(4), 439. Paglia, D.E. and Valentine, W.N. 1967. Quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 70(1), 158–169. Pareek, A., Pant, M., Gupta, M.M., Kashania, P., Ratan, Y., Jain, V. and Chuturgoon, A.A. 2023. Moringa oleifera: an updated comprehensive review of its pharmacological activities, ethnomedicinal, phytopharmaceutical formulation, and clinical, phytochemical, and toxicological aspects. Int. J. Mol. Sci. 24(3), 2098. Paul, S., Ghosh, S., Mandal, S., Sau, S. and Pal, M. 2021. NF-κB and MAPK signaling pathways mediate the immunomodulatory effects of Moringa oleifera leaf extract. Int. Immunopharmacology 100, 108143. Peterson, C.T., Vaughn, A.R., Sharma, V., Chopra, D., Mills, P.J., Peterson, S.N. and Sivamani, R.K. 2019. Effects of dietary supplementation with turmeric and curcumin on human gut microbiota: a double-blind, randomized, placebo-controlled pilot study. J. Evidence-Based. Integr. Med. 23(1), 1–8. Prasad, S., Tyagi, A.K. and Aggarwal, B.B. 2014. Recent developments in curcumin delivery, bioavailability, absorption, and metabolism: the golden pigment from golden spice. Cancer Res. Treat. 46(1), 2–18. Renaudeau, D., Collin, A., Yahav, S., De Basilio, V., Gourdine, J.L. and Collier, R.J. 2012. Adaptation to hot climate and strategies to alleviate heat stress in livestock production. Animal 6(5), 707–728. Rout, P.K., Kaushik, R. and Ramachandran, N. 2022. Differential expression and regulation of the HSP70 gene during the growth phase in ruminants in response to heat stress. Scientific Rep. 12, 18468. Saeed, M., Babazadeh, D., Naveed, M., Arain, M.A., Hassan, F.U. and Chao, S. 2019. The importance of phytogenic feed additives in animal nutrition should be reconsidered. Anim. Feed Sci. Technol. 249, 54–67. Sejian, V., Bhatta, R., Gaughan, J.B., Dunshea, F.R. and Lacetera, N. 2018. Review: adaptation of animals to heat stress. Animal 12(s2), s431–s444. Singleton, V.L., Orthofer, R. and Lamuela-Raventós, R.M. 1999. Analysis of total phenols and other oxidation substrates and antioxidants with Folin-Ciocalteu reagent. Methods. Enzymology. 299, 152–178. Sophia, I., Sejian, V., Bagath, M. and Bhatta, R. 2016. Impact of heat stress on livestock immune responses: a review. Pertanika. J. Trop. Agricult. Sci. 39(4), 459–482. Stohs, S.J. and Hartman, M.J. 2015. Safety and efficacy of Moringa oleifera. Phytotherapy Res. 29(6), 796–804. Surai, P.F., Kochish, I.I., Fisinin, V.I. and Kidd, M.T. 2019. Antioxidant defense systems and oxidative stress in poultry biology: an update. Antioxidants 8(7), 235. Tiloke, C., Anand, K., Gengan, R.M. and Chuturgoon, A.A. 2018. Moringa oleifera and its phytonanoparticles: potential antiproliferative agents against cancer. Biomed. Pharmacotherapy. 108, 457–466. Vahedian-Azimi, A., Abbasifard, M., Rahimi-Bashar, F., Guest, P.C., Majeed, M., Mohammadi, A., Banach, M., Jamialahmadi, T. and Sahebkar, A. 2022. Effectiveness of curcumin on outcomes of hospitalized COVID-19 patients: a systematic review of clinical trials. Nutrients 14(2), 256; doi:10.1016/j.nutrients.2014.09.010 Vergara-Jimenez, M., Almatrafi, M.M. and Fernandez, M.L. 2017. Bioactive components in M. oleifera leaves protect against chronic disease. Antioxidants 6(4), 91. Yasoob, T.B., Yu, D., Khalid, A.R., Zhang, Z., Zhu, X., Saad, H.M. and Hang, S. 2021. Oral administration of M. oleifera leaf powder relieves oxidative stress and modulates the mucosal immune response and cecal microbiota after exposure to heat stress in New Zealand White rabbits. J. Anim. Sci. Biotechnol. 12, 66. Zhang, R., Xu, J., Zhao, J. and Chen, Y. 2021. Effects of curcumin supplementation on the expression of heat shock protein in dairy cows under heat stress. J. Dairy Res. 88(3), 280–284. ᵃ⁻ᵈ Means within a row with different superscripts differ significantly (p < 0.05). | ||
| How to Cite this Article |
| Pubmed Style Al-qaysi MAA, Khalaf MM, Abdulwahhab RI, Kzar MS, Alsadoon Z. Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Vet. J.. 2026; 16(2): 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 Web Style Al-qaysi MAA, Khalaf MM, Abdulwahhab RI, Kzar MS, Alsadoon Z. Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. https://www.openveterinaryjournal.com/?mno=297085 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.44 AMA (American Medical Association) Style Al-qaysi MAA, Khalaf MM, Abdulwahhab RI, Kzar MS, Alsadoon Z. Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Vet. J.. 2026; 16(2): 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 Vancouver/ICMJE Style Al-qaysi MAA, Khalaf MM, Abdulwahhab RI, Kzar MS, Alsadoon Z. Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 Harvard Style Al-qaysi, M. A. A., Khalaf, . M. M., Abdulwahhab, . R. I., Kzar, . M. S. & Alsadoon, . Z. (2026) Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Vet. J., 16 (2), 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 Turabian Style Al-qaysi, Mustafa Adnan Abed, Mustafa Mohammed Khalaf, Ruqaya Imad Abdulwahhab, Marwa Sami Kzar, and Zeid Alsadoon. 2026. Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Veterinary Journal, 16 (2), 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 Chicago Style Al-qaysi, Mustafa Adnan Abed, Mustafa Mohammed Khalaf, Ruqaya Imad Abdulwahhab, Marwa Sami Kzar, and Zeid Alsadoon. "Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions." Open Veterinary Journal 16 (2026), 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 MLA (The Modern Language Association) Style Al-qaysi, Mustafa Adnan Abed, Mustafa Mohammed Khalaf, Ruqaya Imad Abdulwahhab, Marwa Sami Kzar, and Zeid Alsadoon. "Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions." Open Veterinary Journal 16.2 (2026), 1272-1280. Print. doi:10.5455/OVJ.2026.v16.i2.44 APA (American Psychological Association) Style Al-qaysi, M. A. A., Khalaf, . M. M., Abdulwahhab, . R. I., Kzar, . M. S. & Alsadoon, . Z. (2026) Effect of Moringa oleifera leaf extract and turmeric supplementation on cellular and humeral immune responses and antioxidant enzyme activities in shami goats under heat stress conditions. Open Veterinary Journal, 16 (2), 1272-1280. doi:10.5455/OVJ.2026.v16.i2.44 |