| Research Article | ||
Open Vet. J.. 2026; 16(3): 1523-1533 Open Veterinary Journal, (2026), Vol. 16(3): 1523-1533 Research Article A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial propertiesNguyen Phuong Thuy1* and Vuong Tuan Phong21School of Agriculture and Aquaculture, Tra Vinh University, Vinh Long, Vietnam 2College of Engineering and Technology, Tra Vinh University, Vinh Long, Vietnam *Corresponding Author: Nguyen Phuong Thuy. School of Agriculture and Aquaculture, Tra Vinh University, Submitted: 13/11/2025 Revised: 27/01/2026 Accepted: 09/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: The global antimicrobial resistance (AMR) crisis in poultry production has intensified the search for sustainable alternatives, specifically host-adapted probiotics. The Vietnamese CV Super M duck (Anas platyrhynchos domesticus) is an economically vital breed; however, its gastrointestinal tract (GIT) remains an underexplored reservoir for specialized microbial communities that may offer superior protective benefits compared to terrestrial poultry strains. Aim: This study aimed to isolate and characterize lactic acid bacteria (LAB) from the GIT of healthy CV Super M ducks and evaluate their in vitro probiotic potential and antimicrobial efficacy against prominent avian pathogens. Methods: With a sample size of 40 ducks, 60 LAB isolates were obtained through biochemical analysis. Neutralized cell-free supernatants (nCFSs) were used for screening to evaluate antimicrobial activity against Salmonella enterica, Escherichia coli, and Staphylococcus aureus. The most potent isolate, CV12, was evaluated for in vitro tolerance to simulated gastric acid (pH 2.0) and intestinal conditions (0.60% bile salt). Molecular characterization was performed using 16S rRNA gene sequencing. Results: Neutralized cell-free supernatants screening results demonstrated broad-spectrum antimicrobial activity: 37 isolates (61.67%), 33 isolates (55.00%), and 32 isolates (53.33%) inhibited S. enterica, E. coli, and S. aureus, respectively. Isolate CV12 is a highly promising strain, exhibiting excellent in vitro tolerance and high viability under gastric acid (4.45 ± 0.07 Log CFU/ml) and bile salt conditions (4.52 ± 0.14 Log CFU/ml). 16S rRNA sequencing identified CV12 as a member of the genus Companilactobacillus, exhibiting 98.42% similarity to Companilactobacillus farciminis. Notably, this identifies level falls below the common 98.7% species-identify threshold, indicating that CV12 may represent a novel species-level lineage specifically adapted to the GIT. Conclusion: Companilactobacillus sp. CV12 is a host-adapted strain with significant probiotic and antibacterial potential. This study provides the first evidence that the GIT of CV Super M ducks harbors unique, resilient microflora capable of serving as targeted probiotics. Further in vivo trials and whole-genome safety evaluations are required to confirm the commercial viability of this drug. Keywords: Anas platyrhynchos domesticus, Antimicrobial activity, Companilactobacillus sp., Lactic acid bacteria (LAB), Probiotic. IntroductionAntibiotic resistance is a critical global crisis, mainly due to the indiscriminate use of antibiotics in livestock. This poses a serious threat to public health and global food security (Pérez-Sánchez et al., 2018; Patil et al., 2021). The global poultry industry is urgently looking for sustainable alternatives to maintain animal health and address this pressing issue. Probiotics are live microorganisms that confer health benefits, and their use represents a well-established, science-based strategy for promoting human health (Plaza-Diaz et al., 2019). Lactic acid bacteria (LAB) are a type of probiotic notable for their immune-enhancing properties, both inhibiting pathogenic bacteria and producing antibacterial compounds, contributing to the maintenance of intestinal and overall health (Reuben et al., 2019; Hidalgo et al., 2023; Khushboo et al., 2023). The remarkable efficacy of LAB has prompted scientists to search for novel, host-adapted LAB strains from diverse environments and animals to develop next-generation probiotics with superior health protection and support capabilities (Nallala et al., 2017; Krysiak et al., 2021; Rahimoon, 2023). The CV Super M duck (Anas platyrhynchos domesticus) is a predominant and economically important breed in Vietnam’s agricultural sector, particularly in the Mekong Delta, with a national population exceeding 85 million as of 2022. However, bacterial diseases pose a persistent threat to this industry (Ho et al., 2019). Key pathogens, including Salmonella spp. (causing Salmonellosis), avian pathogenic Escherichia coli (APEC, causing Colibacillosis), and Staphylococcus aureus (causing septicemia and bumblefoot), are responsible for conditions ranging from enteritis and high duckling mortality to poor growth rates and carcass condemnation (Abdelrahman and Amer, 2021; Abd El-Ghany, 2023; Sorour et al., 2023; Hussein et al., 2024). The rise of zoonotic, drug-resistant strains of these pathogens underscores the urgent need for targeted, duck-specific control measures that can replace conventional antibiotics without compromising animal health (Aguidissou et al., 2022; Hamed et al., 2024). The efficacy of probiotics is highly host-specific; consequently, when applied to waterfowl, strains derived from chickens often provide suboptimal protection (Xie et al., 2015; Alipin and Safitri, 2016). The CV Super M duck represents a unique reservoir for probiotic discovery due to its exceptional biological resilience and accelerated growth profile, achieving a marketable weight of 3.0–3.5 kg within a short period of 45–50 days. This intensive growth rate, coupled with high survival rates under industrial conditions, strongly indicates the presence of a highly efficient and robust native microflora. Furthermore, their semi-aquatic nature and adaptation to swamp environments, where they subsist on aquatic plants, invertebrates, and rice, expose them to a microbial landscape distinct from that of terrestrial poultry (West et al., 2022; Hemprabha and Arya, 2023). The specialized lifestyle of these waterfowl has driven the evolutionary co-adaptation of their gastrointestinal tract (GIT) microbiota, resulting in a community that is profoundly influenced by rearing conditions and access to water (Lyu et al., 2021; Ma et al., 2024). In particular, the well-developed duck cecum creates an anaerobic environment dominated by the Firmicutes and Bacteroidetes phyla, which is believed to facilitate the colonization of robust LAB strains with superior competitive and antimicrobial capabilities (Kamollerd et al., 2016; Risnaet al., 2022 ). Despite the CV Super M duck’s economically important and distinct biological advantages, its indigenous LAB remains a largely untapped resource in Vietnam. Preliminary studies have identified beneficial Lactobacillus spp., Enterococcus spp., and Pediococcus spp. strains in other duck breeds (Kamollerd et al., 2016; Herdian et al., 2018; Risna et al., 2022; Maquiné et al., 2024). However, a comprehensive characterization of the microflora specifically adapted to the Mekong Delta’s intensive rearing environments is currently lacking. Therefore, this study was conducted to test the hypothesis that the GIT of healthy Vietnamese CV Super M duck harbors unique LAB strains with elite antimicrobial properties. The primary objectives of this study were to isolate and identify LAB from duck GIT and subsequently evaluate their in vitro resilience to gastrointestinal stress and antagonistic activity against Salmonella spp., Escherichia coli, and S. aureus to identify a potent candidate for host-specific probiotic development. Materials and MethodsSample collectionA total of 40 healthy, market-age CV Super M ducks, with an equal sex ratio (20 males:20 females), were collected from commercial farms in Vinh Long Province, Vietnam. After the anesthetized and humanely slaughtered ducks, their GITs were collected aseptically. Four anatomical intestinal segments were isolated from each duck: the crop, gizzard, small intestine, and cecum. Isolation and phenotypic characterization of lactic acid bacteriaEach GIT section was surface sterilized with 70% (v/v) ethanol and rinsed with sterile distilled water to isolate endogenous bacteria. A 1 g sample from each section was homogenized in 9 ml of sterile saline (0.85% w/v) and serially diluted up to 10−7. Aliquots (100 µl) of each dilution were spread-plated in triplicate onto de Man, Rogosa and Sharpe (MRS) agar (Himedia, India). The medium was supplemented with 0.1% (w/v) CaCO3 to screen for acid production and 0.05% (w/v) bile salts to select gut-adapted isolates. Plates were anaerobically incubated at 37°C for 48 hours (Tavakoli et al., 2017). Eighty presumptive LAB colonies, identified by clear hydrolysis halos, were selected and purified by three successive streaking rounds on fresh MRS agar (Nurcahyo et al., 2019). Isolates were confirmed as LAB through Gram staining, microscopic examination, and a panel of biochemical tests, including catalase, urease, oxidase, and citrate use tests. Isolates were identified as Gram-positive and negative for catalase, urease, oxidase, and citrate use. Only Gram-positive, catalase-negative, and oxidase-negative isolates capable of fermenting glucose, lactose, and sucrose were advanced for study (Kandi, 2015). A total of 60 isolates were preserved in MRS broth supplemented with 30% (v/v) glycerol at −20°C (de Carvalho et al., 2021). Screening for antimicrobial activityThe antimicrobial capacity of the 60 LAB isolates was evaluated against three indicator pathogens: Escherichia coli ATCC 25922, S. aureus ATCC 25923, and S. enterica serovar Typhimurium ATCC 14028. Pathogens were cultured in Luria-Bertani (LB) broth (37°C, 24 hours), whereas LAB isolates were grown anaerobically in MRS broth (37°C, 24 hours). Cell-free spermatants (CFS) were obtained by centrifugation at 10,000 pm for 5 minutes at 4°C. To specifically evaluate non-acidic antimicrobial compounds (e.g., bacteriocins), the CFS was adjusted to pH 7.0 ± 0.2 using 1 M NaOH to create neutralized CFS (nCFS). The pH stability of the nCFS was validated by measuring the pH at the beginning and end of the assay (24 hours) to ensure that no significant fluctuations occurred. The antimicrobial activity was determined in triplicate using the agar well diffusion method. Indicator pathogens were adjusted to 106 CFU/ml, mixed with molten nutrient agar (Neogen, USA), and poured into plates. The wells (4 mm diameter) were filled with 100 µl of nCFS. Sterile MRS broth and neutralized MRS broth (nMRS, pH 7.0) were used as negative controls to confirm that neither the medium components nor the neutralization process exerted intrinsic antimicrobial effects. Ampicillin (10 µg) was used as a positive control. After incubation at 37°C for 24 hours, inhibition zones were measured. Based on Rossi et al. (2021), activity was classified into 3 groups: strong (>15 mm), moderate (10–15 mm), weak (<10 mm), and inactive (no inhibition zone). In vitro screening of probiotic propertiesA subset of 27 selected isolates was advanced for in vitro screening of probiotic properties. The isolates were cultured overnight in MRS broth (37°C, 120 rpm). Cells were harvested by centrifugation at 9,000 rpm for 5 minutes at 4°C, washed twice with sterile distilled water, and resuspended in fresh MRS broth. These cell suspensions were standardized to an OD of 0.5–0.7 at 600 nm, corresponding to approximately 103 CFU/ml (Reuben et al., 2021). For acid tolerance, 1 ml of the standardized cell suspension was inoculated into 9 ml of MRS broth adjusted to a pH of 2.0, 3.0, 4.0, and 6.5 (control) and incubated at 37°C for 4 hours (de Carvalho Maquiné et al., 2024). For bile salt tolerance, 1 ml of suspension was added to 9 ml of MRS broth supplemented with 0% (control), 0.20%, 0.40%, or 0.60% bile salts and incubated at 37°C for 4 hours (Tian et al., 2024). Both assays were incubated at 37°C for exactly 4 hours. This duration was chosen to simulate the maximum cumulative transit time in the duck digestive tract, which typically ranges from 2.5 to 5 hours from ingestion to the lower intestinal segments (Lyu et al., 2021; Ma et al., 2024). Survival was quantified using the spread-plate method on MRS agar after 24 hours of anaerobic incubation. All tolerance assays were performed in triplicate (Ramlucken et al., 2020). Antibiotic susceptibility testing was performedThe antibiotic susceptibility of the 27 selected LAB isolates was assessed using the Kirby-Bauer disk diffusion method (Sharma et al., 2024). Cultures were adjusted to a 0.5 McFarland standard and spread on MRS agar. Commercial antibiotic disks (Doxycycline 30 µg, Ampicillin 10 µg, Tetracycline 30 µg, Kanamycin 30 µg, Gentamicin 10 µg, Erythromycin 15 µg, Ciprofloxacin 5 µg, and Chloramphenicol 30 µg) were applied. Escherichia coli ATCC 25922 served as a quality control strain. The inhibition zones were measured after 24 hours at 37°C, and isolates were categorized as sensitive (≥20 mm), intermediate (15–19 mm), or resistant (≤14 mm) (Makzum et al., 2023). The assays were performed in triplicate. Molecular identification using 16S rRNA gene amplification and sequencingThe most promising isolate, CV12, was selected for species-level identification based on its superior antimicrobial spectrum and gastrointestinal resilience. Genomic DNA was extracted using the Wizard® Genomic DNA Purification Kit (Promega, USA). The 16S rRNA gene was amplified by polymerase chain reaction (PCR) using the universal primers 27F (5’-AGAGTTTGATCCTGGCTCAG-3’) and 1492R (5’-GGTTACCTTGTTACGACTT-3’) (Swacita et al., 2022). The PCR amplification was run on a Veriti thermocycler using GoTaq® Green Master Mix (Promega, USA) under the following conditions: initial denaturation at 94°C for 3 minutes; 30 cycles of 94°C for 45 seconds, 53°C for 60 seconds, and 72°C for 90 seconds; and a final extension at 72°C for 5 minutes. The resulting ~1,500 bp PCR product was first confirmed by 1% agarose gel electrophoresis (where Lane 3 represents the negative control using nuclease-free water), then purified with a QIAquick PCR Purification Kit (Qiagen, Germany), and finally sequenced at Next Gen Scientific Co., Ltd (Ho Chi Minh City, Vietnam). Statistical and bioinformatics analysesQuantitative data are presented as mean ± SD from triplicate assays. Statistical analysis was performed using the Statistical Package for the Social Sciences via one-way Analysis of Variance (ANOVA) followed by Tukey’s HSD post hoc test (p < 0.05). The 16S rRNA sequence was assembled using BioEdit and identified using the NCBI BLASTn tool. A species-level match was assigned to a type strain based on >99% identity. A phylogenetic tree was constructed using the neighbor-joining method with 1,000 bootstrap replicates in MEGA11 (Tamura et al., 2021). Ethical approvalAll animal procedures were approved by the institutional ethics committee (Approval No. 5887/QĐ-ĐHTV), following the guidelines described by Risa et al. (2020). ResultsIsolation and phenotypic characterization of LABsFrom the gastrointestinal tracts of 40 healthy CV Super M ducks, 80 presumptive LAB colonies were selected based on halo formation on selective MRS agar. After purification and a panel of biochemical tests, 60 isolates were confirmed as LAB (Table 1). Table 1. Morphological, physiological, and biochemical characteristics of the 60 selected LAB isolates.
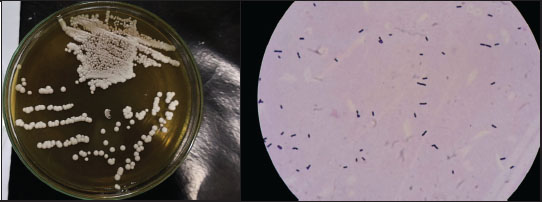
All 60 confirmed isolates were Gram-positive, non-motile, non-endospore-forming, and negative for catalase, urease, oxidase, and citrate use. All isolates were also fermented with glucose, sucrose, and lactose. Based on this profile, all 60 isolates were advanced for antimicrobial screening. Figure 1 shows representative colony and cellular morphologies for the lead candidate, isolate CV12.
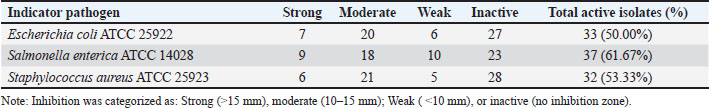
Fig. 1. Morphology of isolated CV12. (A) Colony morphology on MRS agar. (B) Gram staining revealing rod-shaped Gram-positive cells. Antimicrobial activity against avian pathogensThe antimicrobial capacity of the nCFS from all 60 LAB isolates was quantified against three indicator pathogens: S. enterica ATCC 14028, E. coli ATCC 25922, and S. aureus ATCC 25923. Significant strain-specific variation in inhibitory activity was observed (Table 2). Notably, while 53.33%–61.67% of the isolates demonstrated broad-spectrum activity, approximately 40% remained inactive against the tested pathogens. This lack of activity in a substantial subset of isolates indicates that antimicrobial efficacy in these duck-derived LAB is not solely dependent on common metabolites but likely relies on the production of specific bacteriocins or specialized antimicrobial peptides, which were not present in all strains. Table 2. Antimicrobial activity of 60 bacterial isolates against key pathogens.
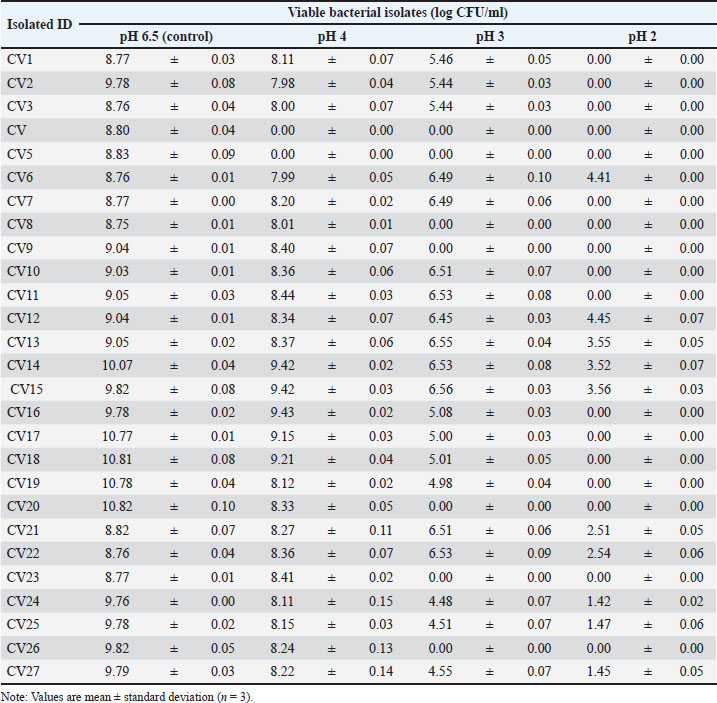
Among the active isolates, CV12 emerged as the most potent candidate, producing statistically significant (p < 0.05) zones of inhibition against S. aureus (19.7 ± 0.25 mm), E. coli (17.3 ± 0.21 mm), and S. enterica (17.7 ± 0.25 mm) compared with those of the other isolates and the neutralized MRS control. Tolerance to simulated gastrointestinal conditionsSurvival of a subset of 27 selected isolates was assessed under simulated gastric acid and intestinal bile salt conditions. Tolerance to simulated gastric acidAfter a 4-hour exposure to simulated gastric acid (pH 2.0), survival varied significantly (p < 0.05) among the isolates (Table 3). While 16 of the 27 isolates showed no viable cells at pH 2.0, isolate CV12 demonstrated exceptional resilience, maintaining a high viable count of 4.45 ± 0.07 Log CFU/ml. Isolate CV6 also showed strong survival (4.41 ± 0.00 Log CFU/ml), indicating that these strains are highly acid-tolerant. Table 3. Viability of the selected LAB isolates under acid stress (pH).
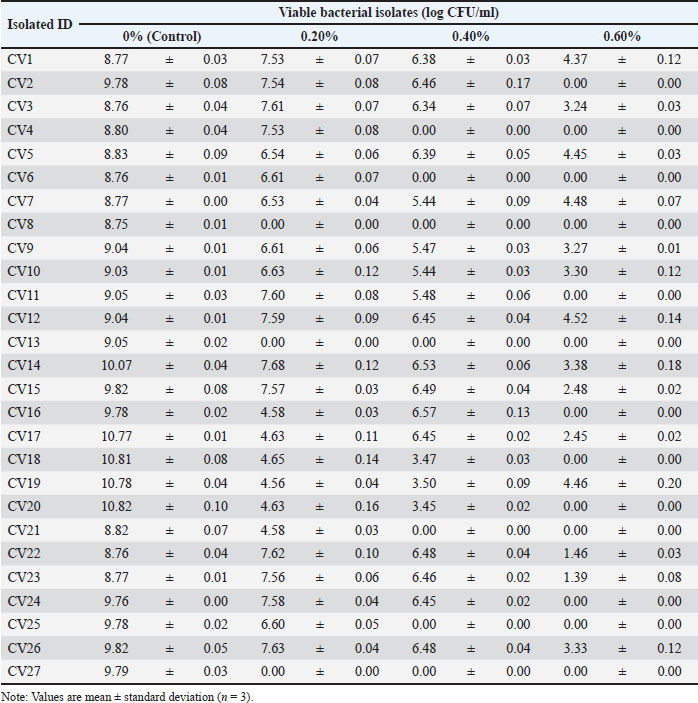
Tolerance to bile saltsWe assessed tolerance to intestinal conditions by a 4-hour exposure to a high concentration of 0.60% bile salts (Table 4). Isolate CV12 demonstrated high resilience, maintaining a viability of 4.52 ± 0.14 Log CFU/ml. Several other isolates, including CV5 (4.45 ± 0.03 Log CFU/ml) and CV7 (4.48 ± 0.07 Log CFU/ml), also survived this condition well. In contrast, 13 of the 27 isolates failed to survive at this concentration. Table 4. Viability of the selected LAB isolates under BS stress.
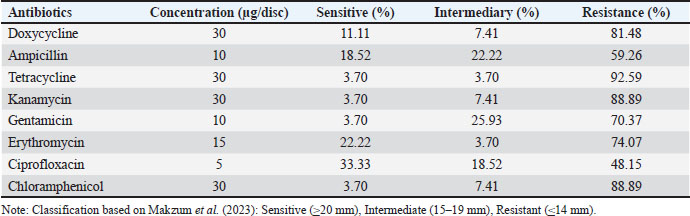
Antibiotic susceptibility profile (ASP) The antibiotic susceptibility of the 27 selected isolates was evaluated as a key safety parameter (Table 5). The most alarming finding was the high prevalence of resistance to TC (92.59% of isolates), kanamycin (88.89%), and chloramphenicol (88.89%). Only isolate CV12 and a few others showed sensitivity to ciprofloxacin (33.33%) and ampicillin (18.52%). Although high tetracycline resistance is a significant safety concern, such resistance in LAB is frequently intrinsic or associated with non-transmissible chromosomal genes rather than mobile genetic elements. This result necessitates mandatory whole-genome sequencing (WGS) in future studies to differentiate between intrinsic and acquired resistance before commercial development. Table 5. Antibiotic susceptibility of the 27 selected LAB isolates.
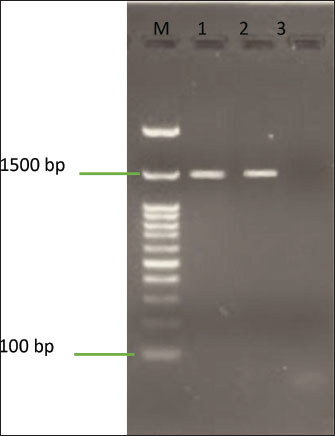
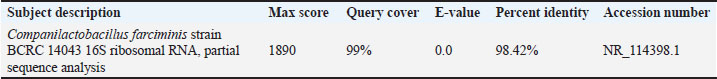
Molecular identification of CV12 as Companilactobacillus sp.CV12 was selected for molecular identification based on its superior combination of broad-spectrum antimicrobial activity and exceptional gastrointestinal resilience. The 16S rRNA gene was successfully amplified via PCR, yielding the expected ~1,500 bp product. The presence and correct size of this amplicon were confirmed by 1% agarose gel electrophoresis (Fig. 2). BLASTn analysis revealed that isolate CV12 shared a 98.42% sequence identity with C. farciminis strain BCRC 14043 (Table 6).
Fig. 2. Agarose gel electrophoresis of the 16S rRNA gene amplified from the CV12 isolate M: 100 bp DNA ladder; Lanes 1 and 2: PCR product from isolate CV12 showing the 1,500 bp amplicon; Lane 3: negative control. Table 6. Top BLASTn hit for the 16S rRNA gene sequence of isolate CV12.
A phylogenetic tree constructed using the neighbor-joining method confirmed that CV12 clusters were definitive within the Companilactobacillus clade (Fig. 3). The 16S rRNA gene sequence of the CV12 isolate has been deposited in the GenBank database under accession number PX890495.
Fig. 3. A phylogenetic tree was constructed using the neighbor-joining method to illustrate the evolutionary relationship of isolate CV12 (accession PX890495) with related Companilactobacillus type strains based on 16S rRNA gene sequences. The bootstrap values (from 1,000 replicates) are indicated at the nodes. The scale bar represents 0.005 nucleotide substitutions per site. DiscussionEcological niche and survival in waterfowl GITThe escalating global crisis of antimicrobial resistance (AMR) has necessitated an urgent transition toward sustainable, non-antibiotic alternatives in livestock production (Patil et al., 2021). In the context of Vietnamese aquaculture and poultry, the CV Super M duck (Anas platyrhynchos domesticus) represents an economically vital breed; however, its intensive rearing often leads to high pathogen pressure (Ho et al., 2019). This study investigated the GIT of these ducks as a specialized reservoir for host-adapted LAB. Our findings identify Companilactobacillus sp. CV12 as a uniquely resilient strain with potent antagonistic activity. However, its high antibiotic resistance profile presents a significant safety hurdle that must be addressed before commercial adoption. The ability to withstand the harsh physiological barriers of the host’s digestive system is a fundamental prerequisite for any probiotic candidate (Plaza-Diaz et al., 2019). The avian GIT is exhibits by rapid transit times and extreme fluctuations in chemical environments, particularly the high acidity of the proventriculus (pH 2.0–3.5) and the detergent-like properties of bile salts in the duodenum (Li et al., 2024). Our results demonstrated that the CV12 isolate maintained exceptional viability (4.45 ± 0.07 Log CFU/ml) even after 4 hours of exposure to pH 2.0 and 0.60% bile salts. This performance is notably superior to that of many terrestrial poultry-derived LAB. For example, previous studies on L. reuteri isolated from chickens have shown a sharp decline in viability (often >5 log reductions) when exposed to pH levels below 2.5 (Khurajog et al., 2023). The resilience of CV12 reflects a specialized evolutionary adaptation to the waterfowl GIT. Unlike chickens, ducks are semi-aquatic and consume a diet rich in aquatic plants and invertebrates, which exposes them to a broader array of environmental microbes and potentially more fluctuating gut pH levels (Ma et al., 2024). This environmental pressure likely selected for a “hardier” microflora capable of rapid metabolic adjustment, allowing CV12 to remain functionally active throughout the transit from the gizzard to the cecum (Lyu et al., 2021). Mechanisms of antimicrobial activity and broad-spectrum efficacyThe primary functional objective of this study was to identify strains capable of suppressing key avian pathogens. Using nCFS, we confirmed that the inhibitory effects of CV12 were not merely a byproduct of lactic acid production, which lowers pH and inhibits growth nonspecifically, but were instead driven by the secretion of specialized antimicrobial metabolites. CV12 demonstrated significant broad-spectrum inhibition against S. aureus (Gram-positive), E. coli, and S. enterica (Gram-negative). This is a critical finding because many LAB strains typically exhibit stronger activity against Gram-positive relatives while struggling to penetrate the LPS outer membrane of Gram-negative pathogens (Hidalgo et al., 2023). We hypothesize that the broad-spectrum efficacy of CV12 is linked to the production of Class II bacteriocins. These are ribosomally synthesized, heat-stable peptides that typically induce cell death by forming pores in the target cell membrane, leading to the leakage of essential intracellular ions and adenosine triphosphate (Simons et al., 2020). Within the genus Companilactobacillus, strains produce bacteriocins, such as farcimicin or acidocin, which exhibit stability across wide pH ranges (Jung et al., 2021). The ability of CV12 to inhibit E. coli and S. enterica suggests that its metabolites may act synergistically with other non-acidic compounds, such as reuterin or hydrogen peroxide, which can temporarily destabilize the Gram-negative outer membrane (Colautti et al., 2022). To confirm this hypothesis and move beyond speculation, future research must utilize liquid chromatography-mass spectrometry for precise peptide identification and WGS to map the Biosynthetic Gene Clusters (BGCs) responsible for these antimicrobial peptides (Shu et al., 2024). Taxonomic significance and host specificity of the speciesThe molecular characterization of CV12 revealed a 98.42% 16S rRNA gene sequence identity with C. farciminis. This taxonomic placement is highly significant. In microbial systematics, an identity threshold of 98.7% is commonly used to demarcate distinct species (Tamura et al., 2021). Therefore, the fact that CV12 falls below this threshold strongly indicates that it may represent a novel, previously undescribed species within the genus Companilactobacillus. Although Companilactobacillus species have been isolated from fermented sausages and some terrestrial livestock (Bampidis et al., 2023), this is the first documented isolation from the CV Super M duck in Vietnam’s Mekong Delta region. This discovery highlights the importance of “bioprospecting” within non-traditional animal hosts. The specialized physiology of waterfowl, including their well-developed cecum and unique semi-aquatic microbial exposure, creates an ecological niche that drives the evolution of host-specific microflora (Hemprabha and Arya, 2023). Strains such as CV12, which co-evolved with the duck host, are theoretically more likely to colonize the intestinal mucosa efficiently than commercial strains derived from cattle or humans (Miranda et al., 2021). This “host-adapted” advantage is a cornerstone of modern precision probiotics, as it ensures better persistence and immune modulation within the target species (Reuben et al., 2021). Safety hurdles: the dilemma of antibiotic resistanceDespite the promising functional data, the safety assessment of our isolates revealed a critical hurdle. The ASP showed alarming rates of resistance to tetracycline (92.59%), kanamycin (88.89%), and chloramphenicol (88.89%). The high prevalence of resistance in duck-derived LAB is likely a direct consequence of the long-term, indiscriminate use of these specific antibiotics in Vietnamese duck farming (Aguidissou et al., 2022). From a regulatory and safety perspective, this is a major concern. According to the European Food Safety Authority and the United States Food and Drug Administration Food and Drug Administration, any microorganism intended for use as a feed additive must not harbor transferable antibiotic resistance genes (Bampidis et al., 2021; FDA, 2022). While some LAB species possess “intrinsic” resistance meaning the resistance is a natural, non-transferable property of the species’ physiology (e.g., cell wall impermeability), the resistance observed here for tetracycline and chloramphenicol is frequently “acquired” and mediated by mobile genetic elements like plasmids or transposons (e.g., tet or cat genes) (Icer et al., 2023). If these genes are mobile, there is a severe risk of horizontal gene transfer to pathogenic bacteria within the duck gut or the broader environment, potentially exacerbating the AMR crisis we aim to solve (Nami et al., 2019). Therefore, the sensitivity of CV12 to ampicillin and ciprofloxacin is insufficient to guarantee safety. Before CV12 can be considered for commercial application, WGS must be performed to differentiate between intrinsic chromosomal mutations and acquired mobile resistance genes (Shu et al., 2024). Furthermore, the safety evaluation must be expanded to include phenotypic tests for hemolytic activity and gelatinase production to ensure that the strain does not possess opportunistic pathogenic traits (Tabanelli et al., 2024). ConclusionCompanilactobacillus sp. CV12 is a potent, host-adapted probiotic candidate capable of reducing antibiotic reliance in the Vietnamese duck industry. Its exceptional resilience and broad-spectrum activity demonstrate high efficacy within the waterfowl GIT. Our future research will address existing safety and application gaps: (1) Whole-genome sequencing to confirm the safety of its antibiotic resistance profile; (2) formulation development using microencapsulation to ensure shelf-stability; and (3) in vivo trials to validate growth performance and immune response in commercial duck flocks. AcknowledgmentsWe acknowledge the support of time and facilities from Tra Vinh University (TVU) for this study. Conflict of interestThe authors declare no conflict of interest. FundingNone. Authors’ contributionsNguyen Phuong Thuy: Conceptualization, methodology, investigation, validation, formal analysis, original draft, review, and editing. Vuong Tuan Phong: Investigation, resources, and data curation. Data availabilityAll data supporting this study’s findings are available within the manuscript. ReferencesAbdelrahman, M.A. and Amer, F. 2021. Characterization of toxin gene profiles and antibiotic resistance genes of methicillin resistant Staphylococcus aureus isolated from ducks. Adv. Anim. Vet. Sci. 9(8), 1150–1158. Abd El-ghany, W.A. 2023. An overview on riemerellosis: a worldwide emerging disease of ducks. Bulg. J. Vet. Med. 26(3), 309–324. Aguidissou, O.N.C., Akpo, Y., Adoko, A.M.J., AdoligbEa, C.M., Koutinhoui, B.G., Boko, C.K. and Farougou, S. 2022. Avian Salmonellosis and Colibacillosis: risk factors, antibiotic resistance, public health impact and biological control. Int. J. Poult. Sci. 21(3), 90–106. Alipin, K. and Safitri, R. The potential of indigenous lactic acid bacteria against Salmonella sp. In AIP Conf. Proc. 2016, 1744(1), p 20001. AIP Publishing, Melville, NY, USA. Bampidis, V., Azimonti, G., Bastos, M.D.L., Christensen, H., Dusemund, B., Durjava, M., Kouba, M., López., Alonso, M. and López Puente, S. 2021. Efficacy of the feed additive containing Companilactobacillus farciminis (formerly Lactobacillus farciminis) CNCM I-3740 (Biacton®) for chickens for fattening, turkeys for fattening and laying hens. EFSA. J. 19(5), 6627. Bampidis, V., Azimonti, G., Bastos, M.D.L., Christensen, H., Dusemund, B., Durjava, M., Kouba, M., López-Alonso, M., López Puente, S., Marcon, F., Mayo, B., Pechová, A., Petkova, M., Ramos, F., Sanz, Y., Villa, R.E., Woutersen, R., Galobart, J., Ortuño, J. and Brozzi, R. 2023. Efficacy of a feed additive consisting of Companilactobacillus farciminis CNCM I-3740 (Biacton®) for chickens and turkeys for fattening (ChemVet dk A/S). EFSA. J. 21(6), e08049. Colautti, A., Orecchia, E., Comi, G. and Iacumin, L. 2022. Lactobacilli, a weapon to counteract pathogens through the inhibition of their virulence factors. J. Bacteriol. 204(11), 272. De Carvalho Maquiné, L., Dos Santos Almeida Coelho, K.W., Da Silva Gomes, M.F., Vieira, J.R., Cavalcante, T.F., De Souza Carvalho, E.B., Rufino, J.P.F., De Oliveira, A.T., De Queiroz Costa Neto, P. and Pereira, J.O. 2024. In vitro probiotic potential of lactic acid bacteria isolated from the intestines of Muscovy ducks. Braz. J. Microbiol. 55(4), 4115–4128. De Carvalho, N.M., Oliveira, D.L., Dib Saleh, M.A., Pintado, M. and Madureira, A.R. 2021. Preservation of human gut microbiota inoculums for in vitro fermentations studies. Fermentation 7(1), 14. FDA. 2022. Notice to US food and drug administration of the conclusion that the intended use of Lacticaseibacillus rhamnosus IDCC 3201 is Generally Recognized as Safe. U.S. Food Drug Adm. AIBMR Life Sci, Inc. Hamed, R.I., Nabil, N.M., Tawakol, M.M. and AbouKhadra, S.H. 2024. An overview of current status of Salmonellosis in duck farms in Egypt. Egypt. J. Anim. Health 4(3), 182–189. Hasson, S.O., Jasim, A.M., Salman, S.A.K., Akrami, S., Saki, M. and Hassan, M.A. 2022. Evaluation of antibacterial and wound-healing activities of alcoholic extract of Boswellia carterii, in vitro and in vivo study. J. Cosmet. Dermatol. 21(11), 6199–6208. Hemprabha. and Arya, S. 2023. A review on behavior of Muscovy duck (Cairina moschata). Int. J. Biol. Sci. 14(2), 74–84. Herdian, H., Istiqomah, L., Damayanti, E., Suryani, A.E., Anggraeni, A.S., Rosyada, N. and Susilowati, A. 2018. Isolation of cellulolytic lactic-acid bacteria from Mentok (Anas moschata) Gastro-Intestinal tract. Trop. Anim. Sci. J. 41(3), 200–206. Hidalgo, V.M., Babot, J.D., Fernández, M.M., Perez Chaia, A., Audisio, C. and Apella, M.C. 2023. Characterization of lactic acid bacteria isolated from the poultry intestinal environment with anti-Salmonella activity in vitro. Braz. J. Microbiol. 54(1), 435–447. Ho, T.V.T., Doan, T.L.A. and Le, V.D. 2019. Escherichia coli infection in ducks in the Mekong Delta: bacterial isolation, serogroup distribution and antibiotic resistance. CTU. J. Innov. Sustain. Dev. 11(1), 24–29. Hussein, A., Eid, A., Hassaan, M., Mohamed, E., Elsohaby, I. and Shawky, M. 2024. Isolation and identification of multidrug resistance bacterial agents implicated in duck enteritis with first record of Salmonella enterica subspecies arizonae in Egypt. Open Vet. J. 14(1), 553–561. Icer, M.A., Özbay, S., Ağagündüz, D., Kelle, B., Bartkiene, E., Rocha, J.M.F. and Ozogul, F. 2023. The impacts of acidophilic lactic acid bacteria on food and human health: a review of the current knowledge. Foods 12(15), 2965. Jung, J.Y., Kang, H.K., Jin, H.M., Han, S.S., Kwon, Y.C., Eun, J.J., Kim, S.C., Seo, M.J., Ryu, B.G. and Chung, E.J. 2021. Companilactobacillus pabuli sp. nov., a lactic acid bacterium isolated from animal feed. Int. J. Syst. Evol. Microbiol. 71(3), 4670. Kamollerd, C., Surachon, P., Maunglai, P., Siripornadulsil, W. and Sukon, P. 2016. Assessment of probiotic potential of Lactobacillus reuteri MD5-2 isolated from ceca of Muscovy ducks. Korean J. Vet. Res. 56(1), 1–7. Kandi, V. 2015. Bacterial colony: first report of donut colony morphology among Diphtheroids isolated in blood. Cureus 7(11), 374. Khushboo, Karnwal, A. and Malik, T. 2023. Characterization and selection of probiotic lactic acid bacteria from different dietary sources for development of functional foods. Front. Microbiol. 14, 1170725. Khurajog, B., Disastra, Y., Lawwyne, L.D., Sirichokchatchawan, W., Niyomtham, W., Yindee, J., Hampson, D.J. and Prapasarakul, N. 2023. Selection and evaluation of lactic acid bacteria from chicken feces in Thailand as potential probiotics. PeerJ 11, e16637. Krysiak, K., Konkol, D. and Korczyński, M. 2021. Overview of the use of probiotics in poultry production. Animals 11(6), 1620. Li, X., Li, W., Zhao, L., Li, Y., He, W., Ding, K. and Cao, P. 2024. Characterization and assessment of native lactic acid bacteria from broiler intestines for potential probiotic properties. Microorganisms 12(4), 749. Lyu, W., Liu, X., Lu, L., Dai, B., Wang, W., Yang, H. and Xiao, Y. 2021. Cecal microbiota modulates fat deposition in Muscovy ducks. Front. Vet. Sci. 8, 609348. Ma, L., Lyu, W., Zeng, T., Wang, W., Chen, Q., Zhao, J., Zhang, G., Lu, L., Yang, H. and Xiao, Y. 2024. Duck gut metagenome reveals the microbiome signatures linked to intestinal regional, temporal development, and rearing condition. iMeta 3(4), e198. Makzum, S., Ghadam, P. and Ramezani, M. 2023. Isolation, functional evaluation of probiotic properties and molecular identification of strains isolated from Iranian poultry’s gut. Iran. J. Microbiol. 15(2), 267–275. Maquiné, L., Guimarães, C., Santos, A., Oliveira, A., Rufino, J., Silva Junior, J., Chaves, F., Mendonça, M., Costa Neto, P. and Pereira, J. 2024. Use of autochthonous lactic acid bacteria as probiotic additives for Muscovy ducks in housing. Braz. J. Poult. Sci. 26(3), 2024. Miranda, C., Contente, D., Igrejas, G., Câmara, S. P. A., Dapkevicius, M. de L. E. and Poeta, P. 2021. Role of exposure to lactic acid bacteria from foods of animal origin in human health. Foods 10, 2092. Nallala, V., Sadishkumar, V. and Jeevaratnam, K. 2017. Molecular characterization of antimicrobial Lactobacillus isolates and evaluation of their probiotic characteristics in vitro for use in poultry. Food. Biotechnol. 31(1), 20–41. Nami, Y., Vaseghi Bakhshayesh, R., Mohammadzadeh Jalaly, H., Lotfi, H., Eslami, S. and Hejazi, M.A. 2019. Probiotic properties of Enterococcus isolated from artisanal dairy products. Front. Microbiol. 10, 300. Nurcahyo, H., Dale, A. and Furqon, F.Y.A. Isolation and characterization of lactic acid bacteria (LAB) from small intestine content of duck (Anas sp.) as a probiotic candidate. J. Phys. Conf. Ser. 2019 1397(1), 12–43. Patil, S.S., Shinduja, R., Suresh, K.P., Phukan, S., Kumar, S., Sengupta, P.P., G. Amachawadi, R., Raut, A., Roy, P., Syed, A., Marraiki, N., Elgorban, A.M., Al-Harthi, H.F., Bahkali, A.H., Shivamallu, C. and Shiva Prasad, K. 2021. A systematic review and meta-analysis on the prevalence of infectious diseases of duck: a world perspective. Saudi. J. Biol. Sci. 28(9), 5131–5144. Pérez-Sánchez, T., Mora-Sánchez, B. and Balcázar, J.L. 2018. Biological approaches for disease control in aquaculture: advantages, limitations and challenges. Trends. Microbiol. 26(11), 896–903. Plaza-Diaz, J., Ruiz-Ojeda, F.J., Gil-Campos, M. and Gil, A. 2019. Mechanisms of action of probiotics. Adv. Nutr. 10, S49–S66. Rahimoon, M.M. 2023. Ameliorative effect of probiotics used in animals: a comprehensive review. Pure. Appl. Biol. 13(2), 179–193. Ramlucken, U., Lalloo, R., Roets, Y., Moonsamy, G., Van Rensburg, C.J. and Thantsha, M.S. 2020. Advantages of Bacillus-based probiotics in poultry production. Livest. Sci. 241, 104215. Reuben, R.C., Roy, P.C., Sarkar, S.L., Alam, R.U. and Jahid, I.K. 2019. Isolation, characterization, and assessment of lactic acid bacteria toward their selection as poultry probiotics. BMC. Microbiol. 19, 1–20. Reuben, R.C., Sarkar, S.L., Roy, P.C., Anwar, A., Hossain, M.A. and Jahid, I.K. 2021. Prebiotics, probiotics and postbiotics for sustainable poultry production. World’s Poult. Sci. 77(4), 825–882. Risa, Y.K., Harimurti, S. and Widodo, W. 2020. Screening for probiotic of lactic acid bacteria isolated from the digestive tract of a native Aceh duck (Anas platyrhynchos). Biodiversitas 21(7), 3125–3131. Risna, Y. K., Harimurti, S., Wihandoyo, W., Widodo, W. and Sukarno, A. S. 2022. Aktivitas Antibakteri Bakteri Asam Laktat (BAL) yang Diisolasi dari Saluran Pencernaan Itik Lokal Asal Aceh terhadap Salmonella pullorum dan Escherichia coli. J. Agripet 22, 169–174. Rossi, E., La Rosa, R., Bartell, J.A., Marvig, R.L., Haagensen, J.A.J., Sommer, L.M., Molin, S. and Johansen, H.K. 2021. Pseudomonas aeruginosa adaptation and evolution in patients with cystic fibrosis. Nat. Rev. Microbiol. 19(5), 331–342. Sharma, H., Upadhyay, A.K., Singh, N.K., Nagpal, A., Dhial, K. and Kumari, R. 2024. Antibiotic resistance profile and safety assessment of Lactobacillus acidophilus and Lactobacillus plantarum isolated from milk. J. Sci. Res. Rep. 30(7), 938–945. Shu, H., He, X., Hong, Z., Dong, K., Zou, Y., Cao, M., Wang, R., Xu, Y., Liao, L., Zuo, H. and Pei, X. 2024. Screening and genome analysis of potential probiotic lactic acid bacteria with broad-spectrum antibacterial activity from Sichuan sun-dried vinegar grains (Cupei). LWT 202, 116288. Simons, A., Alhanout, K. and Duval, R.E. 2020. Bacteriocins, antimicrobial peptides from bacterial origin: overview of their biology and their impact against multidrug-resistant bacteria. Microorganisms 8(5), 639. Sorour, H., Badr, H., Abdelaty, M., Roshdy, H., Mohammed, A. and AbdelRahman, M. 2023. Virulence range and new pathological pictures of Salmonella enteritidis and Salmonella typhimurium isolated from ducklings in experimental infected chicks. J. Appl. Vet. Sci. 8(1), 45–56. Swacita, I.B.N., Suardana, I.W., Sudisma, I.G.N. and Wihadmadyatami, H. 2022. Molecular identification of lactic acid bacteria SR6 strain and evaluation of its activity as an anticancer in T47D cell line. Vet. World. 15(6), 1583–1588. Tabanelli, G., Barbieri, F., Baños, A., Madero, J.M.G., Daza, M.V.B., Cortimiglia, C., Milani, G., Bassi, D., Gardini, F. and Montanari, C. 2024. Companilactobacillus alimentarius: an extensive characterization of strains isolated from spontaneous fermented sausages. Int. J. Food Microbiol. 410, 110489. Tamura, K., Stecher, G. and Kumar, S. 2021. MEGA11: molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 38(7), 3022–3027. Tavakoli, M., Hamidi-Esfahani, Z., Hejazi, M., Azizi, M. and Abbasi, S. 2017. Characterization of probiotic abilities of Lactobacilli isolated from Iranian Koozeh traditional cheese. Pol. J. Food Nutr. Sci. 67(1), 41–48. Tian, C., Wang, L., Liu, M., Liu, J., Qiu, M. and Chen, Y. 2024. Isolation and identification of chicken-derived lactic acid bacteria: in vitro probiotic properties and antagonistic effects against Salmonella pullorum, Staphylococcus aureus, and Escherichia coli. Microorganisms 12(4), 795. West, A.K., Xu, E.M., Nelson, M.D., Hart, T.R., Stricker, E.M., Cones, A.G., Martin, G.M., Strickland, K., Lambert, D.L., Burman, L., Zhu, B.H. and Schneider, E.R. 2022. Quantitative evaluation of tactile foraging behavior in Pekin and Muscovy ducks. Front. Physiol. 13, 921657. Xie, Z.L., Bai, D.P., Xie, L.N., Zhang, W.N., Huang, X.H. and Huang, Y.F. 2015. Intestinal lactic acid bacteria from Muscovy duck as potential probiotics that alter adhesion factor gene expression. Genet. Mol. Res. 14(4), 12262–12275. | ||
| How to Cite this Article |
| Pubmed Style Thuy NP, Phong VT. A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Vet. J.. 2026; 16(3): 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 Web Style Thuy NP, Phong VT. A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. https://www.openveterinaryjournal.com/?mno=296316 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.11 AMA (American Medical Association) Style Thuy NP, Phong VT. A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Vet. J.. 2026; 16(3): 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 Vancouver/ICMJE Style Thuy NP, Phong VT. A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 Harvard Style Thuy, N. P. & Phong, . V. T. (2026) A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Vet. J., 16 (3), 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 Turabian Style Thuy, Nguyen Phuong, and Vuong Tuan Phong. 2026. A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Veterinary Journal, 16 (3), 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 Chicago Style Thuy, Nguyen Phuong, and Vuong Tuan Phong. "A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties." Open Veterinary Journal 16 (2026), 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 MLA (The Modern Language Association) Style Thuy, Nguyen Phuong, and Vuong Tuan Phong. "A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties." Open Veterinary Journal 16.3 (2026), 1523-1533. Print. doi:10.5455/OVJ.2026.v16.i3.11 APA (American Psychological Association) Style Thuy, N. P. & Phong, . V. T. (2026) A novel Companilactobacillus isolate from the CV Super M duck (Anas platyrhynchos domesticus) with probiotic and antimicrobial properties. Open Veterinary Journal, 16 (3), 1523-1533. doi:10.5455/OVJ.2026.v16.i3.11 |