| Short Communication | ||
Open Vet. J.. 2026; 16(3): 1925-1931 Open Veterinary Journal, (2026), Vol. 16(3): 1925-1931 Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbumin-sensitized mouse modelAhmad Al Athamneh1*, Omar Al Haj1, Anas Khaleel2 and Shigeru Katayama31Department of Nutrition, Faculty of Pharmacy and Medical Sciences, University of Petra, Amman, Jordan 2Department of Clinical Pharmacy and Pharmacy Practice, Faculty of Pharmacy and Medical Sciences, University of Petra, Amman, Jordan 3Department of Agriculture, Graduate School of Science and Technology, Shinshu University, Minamiminowa Kamiina, Japan *Corresponding Author: Ahmad Al Athamneh. Department of Nutrition, Faculty of Pharmacy and Medical Sciences, University of Petra, Amman, Jordan. Email: Ahmad.Alathamneh [at] uop.edu.jo Submitted: 12/11/2025 Revised: 27/01/2026 Accepted: 08/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
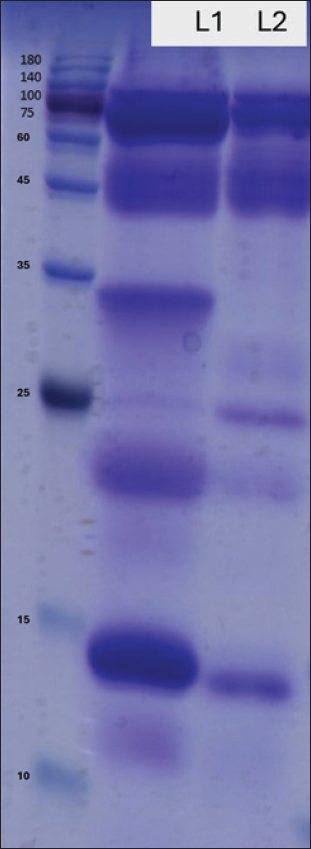
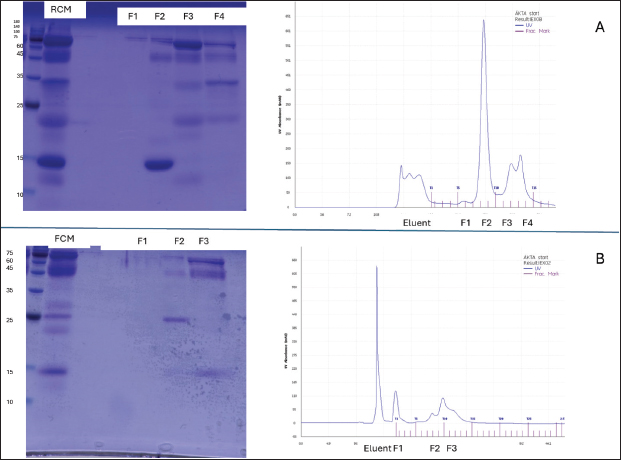
AbstractBackground: Camel milk is widely consumed in regions with an increasing incidence of allergic diseases. Fermentation can reshape milk protein and peptide profiles, which may influence immunoreactivity. Aim: This study aimed to provide preliminary evidence on whether raw camel milk (RCM) and fermented camel milk (FCM) modulate allergic responses in an ovalbumin (OVA)-sensitized mouse model and to contextualize any in vivo effects with sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and size-exclusion chromatography (SEC) profiles. Methods: Female BALB/c mice (n=21) were assigned to control (n=6), sham (n=5), raw camel milk (RCM; n=5), or fermented camel milk (FCM; n=5), sensitized, and challenged with ovalbumin. Clinical allergy scores and serum immunoglobulins were assessed (total IgE in all groups; OVA-specific IgE/IgA/IgG2a in sham, RCM, and FCM groups). Milk samples were subjected to SDS-PAGE and SEC. Results: Fermentation attenuated protein bands in the ~15–35 kDa region and shifted early SEC fractions relative to RCM. In vivo, total IgE differed across groups, driven primarily by lower values in non-sensitized controls; no BH–FDR-adjusted pairwise differences were detected between Sham and RCM/FCM. Antigen-specific immunoglobulins exhibited modest or non-significant changes; clinical scores were numerically lower but small in magnitude. Conclusion: Fermentation produced reproducible compositional shifts in camel milk, alongside group-level differences in total IgE that were primarily explained by non-sensitized controls rather than significant differences among sensitized groups. Although the effects on clinical scores and antigen-specific antibodies were limited in this small experiment, the data support further work integrating quantitative proteomics/peptidomics and cytokine profiling to clarify the mechanisms. Keywords: Allergy, Camel milk, Fermentation, Immunoglobulin E, Murine model. IntroductionFood allergy is a growing public health concern, most often driven by IgE-mediated mechanisms that can precipitate acute and sometimes severe reactions (Johansson, 2004; Sicherer and Sampson, 2018; Wong, 2024). Although prevalence varies by region and setting, contemporary surveys and reviews point to a substantial burden in children and continued increases worldwide (Gupta et al., 2018; Wong, 2024). The ovalbumin (OVA)–sensitized BALB/c mouse remains a well-established model for dissecting allergen-specific immune responses and evaluating dietary or immunological interventions (Li et al., 2000; Zhang et al., 2021). Camel milk has long been consumed for nutrition and health, and modern analyses have described its bioactive profile, which includes lactoferrin, immunoglobulins, lactoperoxidase, peptides, and other components with antioxidant and anti-inflammatory potential (Mati et al., 2017; Swelum et al., 2021; Behrouz et al., 2022; Ho et al., 2022). Fermentation further reshapes the protein landscape and may introduce probiotic effects relevant to mucosal immunity. In OVA-based models, fermented dairy or defined probiotics have reduced symptom scores and OVA-specific IgE while shifting responses away from a Th2-dominant profile, supporting the plausibility of diet-derived immunomodulation in food allergy (Vélez et al., 2015). Beyond dairy per se, processing or modifying dietary proteins can influence allergenicity and downstream immunoglobulin patterns. In murine systems, compared with intact proteins, phosphorylated or hydrolyzed allergens have attenuated allergic reactions, lowered specific IgE, increased IgA, and engaged T follicular helper (Tfh)–linked mechanisms (Katayama et al., 2018; Al Athamneh et al., 2020). Against this backdrop, we conducted a preliminary evaluation of raw camel milk (RCM) and fermented camel milk (FCM) in an OVA-sensitized mouse model, focusing on clinical scores and serum immunoglobulins (total IgE, OVA-specific IgE, IgA, and IgG2a). Materials and MethodsAnimals and study designTwenty one female BALB/c mice (5 weeks old; Oriental Yeast Co., Tokyo, Japan) were housed under controlled conditions (22°C ± 2°C; 55% ± 10% humidity; 12-hour light/dark cycle) with standard milk-free chow and water ad libitum. Mice were randomly assigned to four groups: control (n=6), sham (n=5), RCM (n=5), and FCM (n=5). The OVA-sensitized BALB/c model was used as a standard platform for food allergy studies (Zhang et al., 2021). Preparation and administration of camel milkRaw camel milk was collected in Jordan under sterile conditions. The starter cultures of Streptococcus thermophilus, Lactobacillus bulgaricus, and Lactobacillus helveticus (1% each) were incubated at 43°C to pH 4.42 (≈11 hours), then freeze-dried and stored at −80°C until use. From day 1, RCM or FCM was incorporated into the diet at 5% (w/w) and continued until the end of the study; the control and sham groups received the base diet only. A 5% (w/w) inclusion level was selected as a moderate dietary supplementation dose intended to preserve diet palatability and overall macronutrient balance in a short-term feeding design. Protein characterization of camel milk samplesThe protein profiles of RCM and FCM were examined by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) (15% resolving, 5% stacking; samples reduced with 1% β-mercaptoethanol and heated at 95°C for 5 minutes) and stained with Coomassie Brilliant Blue R-250. Complementary fractionation was performed by chromatography on a size-exclusion chromatography column, and fractions were assessed by SDS-PAGE as needed to visualize distribution differences after fermentation. Sensitization, challenge, and clinical scoringThe sham, RCM, and FCM groups were sensitized intraperitoneally with ovalbumin (OVA; Sigma-Aldrich, Tokyo, Japan) emulsified with alum adjuvant (In vivo Gen, France) on days 3 and 17 (phosphate-buffered saline (PBS) was administered to the control group). Mice were challenged orally with OVA (10 mg per mouse) on days 31, 33, and 35. Forty minutes after the final challenge, clinical symptoms were graded on a scale of 0–3 (0=no symptoms; 1=scratching/itching; 2=periorbital/perioral edema, piloerection, reduced activity, and faster breathing; 3=wheezing or labored breathing with cyanosis, tremor/convulsions, or minimal response to stimulus). This time point and scale were pre-specified. Two independent observers blinded to group allocation assessed clinical scores. Serum collection and immunoglobulin assaysAfter the last challenge, the mice were euthanized by CO2 inhalation. Blood was collected, and serum was isolated for enzyme-linked immunosorbent assay (ELISA) quantification of total IgE, OVA-specific IgE, IgA, and IgG2a using matched capture/detection antibodies (Pierce, Rockford, IL, USA). OVA-specific assays used OVA-coated plates (100 µg ml 1) with otherwise identical procedures. Assays were performed according to the manufacturer’s instructions using appropriate blanks and standards. Where absolute quantification could not be robustly calibrated across plates, results are presented as relative ELISA units as indicated in the corresponding figure axes/legends. Antigen-specific assays (OVA-IgE, OVA-IgA, and OVA-IgG2a) were run for Sham, RCM, and FCM; these endpoints were not assayed in the Control group. Limited serum volume or technical issues led to unequal sample sizes for some ELISAs; the actual n per group is stated in the figure legends. The effect sizes (Cliff’s d or Hedges’ g) were calculated where applicable. Statistical analysis of the dataThe analyses were two-sided (α=0.05). The primary test for each endpoint was the Kruskal–Wallis test because group sizes were small and clinical scores were ordinal, followed, when applicable, by pairwise Mann–Whitney U tests with a Benjamini–Hochberg false-discovery rate (BH-FDR) adjustment within each endpoint. For ELISA endpoints, we additionally report one-way analysis of variance (Welch-robust) p-values as a supplementary check where sample sizes permitted. Data are presented as individual animals (dots) with mean ± SEM. No animals were excluded; available data were used for analyses. Ethical approvalAll procedures were approved by the Animal Experiment Committee of Shinshu University (Approval No. 0221; August 9, 2024) and conducted in accordance with institutional guidelines and ARRIVE 2.0. Results and DiscussionProtein profiling of camel milk samplesSDS–PAGE revealed qualitative differences between RCM and FCM, with lighter bands in the ~15–35 kDa range after fermentation and comparatively stable high-molecular mass features (Fig. 1). The ~15–35 kDa window is consistent with the approximate size range of several milk protein fractions; however, the present SDS–PAGE/SEC data are descriptive and do not support definitive band assignment. Chromatographic profiling with matched fraction gels revealed a redistribution of eluting protein fractions in FCM relative to RCM (Fig. 2).
Fig. 1. SDS–PAGE of camel milk proteins (representative of n=3 independent gels). Representative gel comparing RCM and FCM. Molecular-mass markers (kDa) indicated. Bands in the ~15–35 kDa range appear less intense after fermentation, whereas higher-mass bands remain comparatively unchanged. Experimental details (gel 15%, 5% load, reducing conditions) are described in the Methods section.
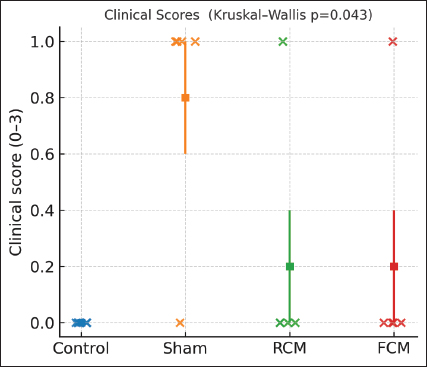
Fig. 2. Chromatographic profiling of camel milk proteins (representative of n=3 independent injections). (A) RCM elution profile and collected fractions with the corresponding SDS–PAGE lanes (fractions labeled under chromatograms). (B) FCM elution profile and collected fractions with the corresponding SDS–PAGE lanes (fractions labeled under chromatograms). Fermentation altered the distribution of protein fractions relative to RCM. The column, buffer, and flow parameters are described in the Methods section. Clinical symptomsClinical scores differed across groups following the final oral OVA challenge (Kruskal–Wallis p=0.043; ANOVA p=0.027), but no pairwise comparison remained significant after BH–FDR adjustment (all q ≥ 0.05). Both milk-treated groups showed numerically lower scores than Sham (Fig. 3).
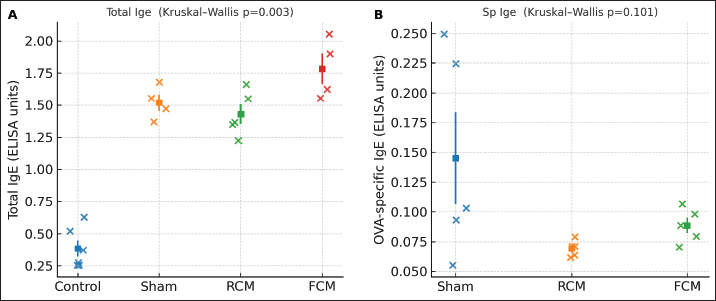
Fig. 3. Clinical scores after the oral OVA challenge Individual mice (dots) with mean ± SEM, (n) per group shown by dots. Overall difference: Kruskal–Wallis test, p=0.043 (ANOVA, p=0.027). No pairwise comparison remained significant after BH–FDR (all q ≥ 0.05). Group order: Control, Sham, RCM, and FCM. Serum IgE levelsTotal IgE levels varied across groups (Kruskal–Wallis p=0.003; ANOVA p < 0.0001). After BH-FDR adjustment, the control was lower than each challenged cohort (control vs. sham, q=0.019; control vs. RCM, q=0.019; control vs. FCM, q=0.019; Fig. 4A), whereas no significant difference was observed in the sham versus RCM/FCM contrast after adjustment. In contrast, OVA-specific IgE showed no significant differences among the assayed groups (Sham, RCM, FCM: Kruskal–Wallis p=0.101; ANOVA, p=0.087; all pairwise q ≥ 0.05; Fig. 4B).
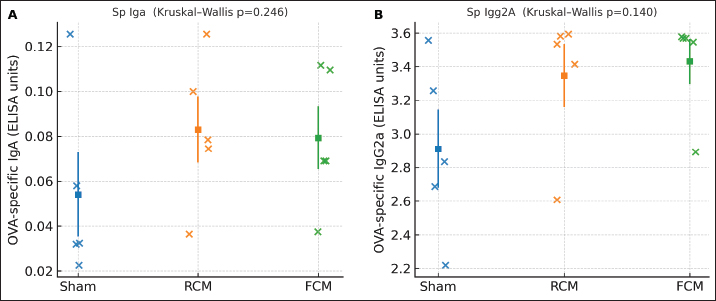
Fig. 4. Serum IgE. (A) Total IgE (control, n=6; sham, n=4; RCM, n=5; FCM, n=4). Dots show individual mice with mean ± SEM. Overall difference: Kruskal–Wallis test, p=0.003 (ANOVA p < 0.0001). Significant BH–FDR contrasts: Control versus Sham q=0.019; Control versus RCM q=0.019; Control versus FCM q=0.019. Notably, no sham versus. RCM/FCM contrast remained significant after BH-FDR, indicating that the apparent separation in total IgE is largely attributable to the control (non-sensitized) group rather than a robust treatment effect within sensitized mice. (B) OVA-specific IgE (groups shown: Sham, RCM, FCM; n=5 each). Dots with mean ± SEM. Overall NS (Kruskal–Wallis p=0.101; ANOVA p=0.087); no BH–FDR-significant pairwise differences. Antigen-specific antibodies against IgA and IgG2aOVA-specific IgA (Fig. 5A) and OVA-specific IgG2a (Fig. 5B) did not differ significantly across the groups on the overall non-parametric test (Kruskal–Wallis p=0.246 and p=0.140, respectively), and no pairwise contrast survived the BH–FDR test (all q ≥ 0.05).
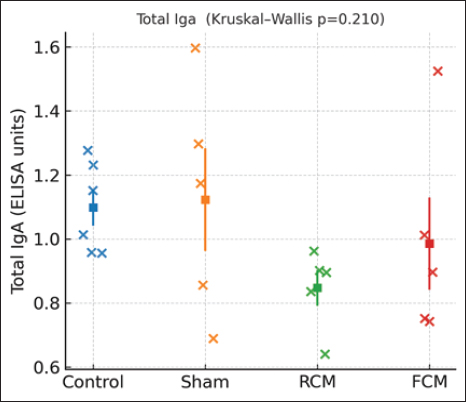
Fig. 5. Antigen-specific isotypes. (A) OVA-specific IgA (groups shown: Sham, RCM, FCM; n=5 each). Dots with mean ± SEM. Overall NS (Kruskal–Wallis p=0.246); no BH–FDR-significant pairwise differences. (B) OVA-specific IgG2a (groups shown: Sham, RCM, FCM; n=5 each). Dots with mean ± SEM. Overall NS (Kruskal–Wallis p=0.140); no BH–FDR-significant pairwise differences. Total IgA levelTotal serum IgA showed no detectable group differences (Kruskal–Wallis p=0.210; all Benjamini–Hochberg false discovery rate (BH–FDR)-adjusted pairwise q ≥ 0.05) (Fig. 6).
Fig. 6. Total IgA. Individual mice (dots) with mean ± SEM for control, sham, RCM, and FCM groups (n per group shown). Y-axis: ELISA units. No significant differences were detected (overall Kruskal–Wallis p=0.210; all pairwise Mann–Whitney U comparisons BH–FDR-adjusted q ≥ 0.05). Group order is Control → Sham → RCM → FCM. Integrated discussion and limitationsFermentation reshaped the camel-milk protein landscape (Figs. 1 and 2). However, the immunological readouts in this OVA model were modest: clinical scores were numerically lower in the milk-treated groups than in the sham groups on central tendency, but pairwise contrasts did not survive multiple-testing correction (Fig. 3). Among the serological endpoints, total IgE differed across groups; however, this pattern was statistically driven by the non-sensitized control cohort, and no BH-FDR-adjusted pairwise differences were detected between the sham and RCM/FCM groups (Fig. 4A). OVA-specific IgE, IgA, and IgG2a showed no significant differences between the groups (Figs. 4–6). These patterns indicate biochemical changes without a clearly separable immunological signature between RCM and FCM under this design. Previous studies have shown that processing food proteins can dampen allergenicity and tilt responses toward mucosal protection—phosphorylation and controlled hydrolysis reduced IgE levels and improved outcomes in sensitized mice (Katayama et al., 2018; Al Athamneh et al., 2020). Camel milk itself carries bioactive proteins and peptides with immunomodulatory potential, and fermentation can further alter epitope exposure and peptide release (Swelum et al., 2021; Behrouz et al., 2022). Contemporary in vivo data in allergic airways also suggest that camel milk mitigates Th2-skewed inflammation and airway hyperreactivity (Rakhmatulina et al., 2025). Probiotic fermented dairy interventions in OVA-sensitized mice have been reported to attenuate allergic outcomes and shift responses away from Th2-dominant profiles, supporting the plausibility of diet-derived immunomodulation in food allergy (Cross and Gill, 2001; Vélez et al., 2015). However, the present data do not provide strong evidence of a treatment effect on antigen-specific readouts. Three factors are likely to limit resolution here. First, the sample size (n≈5/group) reduces power after FDR correction. Second, assay scope: cytokines or Tfh/GC readouts were not retained in the final analysis, though these pathways often track shifts in IgE/IgA after protein processing (Katayama et al., 2018; Al Athamneh et al., 2020). Third, measurement scale: several ELISAs were plotted as ELISA units rather than calibrated concentrations, which can compress the dynamic range. These are actionable in a follow-up: (i) predefine a primary endpoint (e.g., clinical score and OVA-specific IgE), power for it, and control multiplicity; (ii) export calibrated concentrations (ng/ml) and add mechanistic panels (Th2/Th1/Tfh cytokines, class-switch markers); and (iii) fractionate FCM (as in Fig. 2) to test which fractions carry tolerogenic activity (Swelum et al., 2021; Behrouz et al., 2022). With these refinements, we can more decisively test whether fermentation confers an immunological advantage over raw camel milk. ConclusionFermentation altered camel-milk protein profiles, and clinical scores were numerically lower in milk-treated mice than in Sham mice; however, no pairwise comparisons remained significant after BH–FDR adjustment, and antigen-specific serology did not differ significantly between RCM and FCM at this sample size. The findings of this study align with the literature on processing-mediated hypoallergenicity and camel-milk immunomodulation while highlighting the limitations of the power and assay scope (Katayama et al., 2018; Al Athamneh et al., 2020; Swelum et al., 2021; Behrouz et al., 2022; Rakhmatulina et al., 2025). A powered study using calibrated enzyme-linked immunosorbent assays and mechanistic readouts is warranted to determine whether fermentation yields a clinically meaningful immunological advantage. AcknowledgmentsThe authors would like to acknowledge the efforts of the Faculty of Pharmacy and Medical Sciences of the University of Petra. The authors also extend their gratitude to the Deanship of Scientific Research and Graduate Studies for their continuous administrative and academic support. The authors thank Shinshu University (Nagano, Japan) for hosting the study and providing access to laboratory facilities and instrumentation. They are especially grateful to Prof. Shigeru Katayama and the members of the Food Chemistry Laboratory for their technical assistance, chromatography support, and constructive discussions. Conflict of interestThe authors have no conflicts of interest related to this work. FundingThis work was supported by the University of Petra Research Fund (grant number 3/4/2024). Authors’ contributionsConceptualization: A.A., S.K. Methodology: A.A., A.Ka., O.A., S.K. Investigation: A.A., O.A., A.Kh., A.Ka. Formal analysis: A.A. Resources: S.K. Visualization: A.A. Writing—original draft: A.A. Writing—review & editing: All authors. Supervision: S.K. Funding acquisition: A.A. Data availabilityAll data supporting the findings are included within the manuscript and figures; upon reasonable request, additional raw data are available from the corresponding author. ReferencesAl Athamneh, A.M., Suzuki, Y., Nakamura, S. and Katayama, S. 2020. Hydrolysate of highly digestible phosphorylated buckwheat major allergen Fag e 2 attenuates allergic reactions in Fage 2-sensitized mice. Japanese J. Food Chem. Saf. 27(2), 67–75. Available via https://www.jstage.jst.go.jp/article/jjfcs/27/2/27_67/_pdf Behrouz, S., Saadat, S., Memarzia, A., Sarir, H., Folkerts, G. and Boskabady, M.H. 2022. The antioxidant, anti-inflammatory and immunomodulatory effects of camel milk. Front. Immunol. 13, 855342; doi:10.3389/fimmu.2022.855342 Cross, M.L. and Gill, H.S. 2001. Can immunoregulatory lactic acid bacteria be used as dietary supplements to limit allergies?. Int. Arch. Allergy Immunol. 125(2), 112–119; doi:10.1159/000053804 Ho, T.M., Zou, Z. and Bansal, N. 2022. Camel milk: a review of its nutritional value, heat stability, and potential food products. Food. Res. Int. 153, 110870; doi:10.1016/j.foodres.2021.110870 Johansson, S. 2004. Revised nomenclature for allergy for global use: report of the Nomenclature Review Committee of the World Allergy Organization, October 2003. J. Allergy Clin. Immunol. 113(5), 832–836; doi:10.1016/j.jaci.2003.12.591 Katayama, S., Yamaguchi, D., Suzuki, Y., Athamneh, A.M.A., Mitani, T., Satoh, R., Teshima, R., Mine, Y. and Nakamura, S. 2018. Oral immunotherapy with a phosphorylated hypoallergenic allergen ameliorates allergic responses more effectively than intact allergen in a murine model of buckwheat allergy. Mol. Nutr. Food Res. 62(21), e1800303; doi:10.1002/mnfr.201800303 Gupta, R.S., Warren, C.M., Smith, B.M., Blumenstock, J.A., Jiang, J., Davis, M.M. and Nadeau, K.C. 2018. The public health impact of parent-reported childhood food allergies in the United States. Pediatrics 142(6), e20181235; doi:10.1542/peds.2018-1235 Li, X.M., Serebrisky, D., Lee, S.Y., Huang, C.K., Bardina, L., Schofield, B.H., Stanley, J.S., Burks, A.W., Bannon, G.A. and Sampson, H.A. 2000. A murine model of peanut anaphylaxis: t- and B-cell responses to a major peanut allergen mimic human responses. J. Allergy Clin. Immunol. 106(1 Pt 1), 150–158; doi:10.1067/mai.2000.107395 Mati, A., Senoussi-Ghezali, C., Si Ahmed Zennia, S., Almi-Sebbane, D., El-Hatmi, H. and Girardet, J.M. 2017. Dromedary camel milk proteins, a source of peptides having biological activities—a review. Int. Dairy J. 73, 25–37; doi:10.1016/j.idairyj.2016.12.001 Rakhmatulina, A., Kenenbay, S., Abuova, A., Kizatova, M., Ibraikhan, A., Makhmudov, F., Mukashev, A., Aitbaeva, A., Abilkaiyr, Z., Ibadullayeva, G., Chomanov, U., Murzabulatov, A., Azimova, S., Kulpiisova, A., Bayantassova, S., Aralbayev, N., Imanbayeva, N., Dikhanbayeva, F., Burambayeva, N., Smagulova, N., Issimov, A. and White, P. 2025. The effect of camel milk on house dust mite allergen–induced asthma model in BALB/c mice. PLos One. 20(6), 327504; doi:10.1371/journal.pone.0327504 Sicherer, S.H. and Sampson, H.A. 2018. Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J. Allergy. Clin. Immunol. 141(1), 41–58; doi:10.1016/j.jaci.2017.11.003 Swelum, A.A., El-Saadony, M.T., Abdo, M., Ombarak, R.A., Hussein, E.O.S., Suliman, G., Alhimaidi, A.R., Ammari, A.A., Ba-Awadh, H., Taha, A.E., El-Tarabily, K.A. and Abd El-hack, M.E. 2021. Nutritional, antimicrobial and medicinal properties of camel’s milk: a review. Saudi J. Biol. Sci. 28(6), 3126–3136; doi:10.1016/j.sjbs.2021.02.057 Vélez, E.M.M., Duarte-Martínez, C.T., Galeano, P., Bermúdez-Humarán, L.G., Hernández-Mendoza, A., Mendoza-Hernández, G., García, H.S. and Vallejo-Cordoba, B. 2015. Probiotic fermented milk consumption modulates the allergic process induced by ovalbumin in mice. Br. J. Nutr. 114(4), 566–576; doi:10.1017/S0007114515001981 Wong, G.W.K. 2024. Food allergies around the world. Front. Nutr. 11, 1373110; doi:10.3389/fnut.2024.1373110 Zhang, Y., Li, J., Wang, Y., Xu, J., Wu, Y. and Wang, X. 2021. Effective model of food allergy in mice sensitized with ovalbumin and Freund’s adjuvant. Bull. Exp. Biol. Med. 171(3), 352–356; doi:10.1007/s10517-021-05271-y. | ||
| How to Cite this Article |
| Pubmed Style Al-athamneh A, Al-haj O, Khaleel A, Katayama S. Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 Web Style Al-athamneh A, Al-haj O, Khaleel A, Katayama S. Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. https://www.openveterinaryjournal.com/?mno=296075 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.47 AMA (American Medical Association) Style Al-athamneh A, Al-haj O, Khaleel A, Katayama S. Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 Vancouver/ICMJE Style Al-athamneh A, Al-haj O, Khaleel A, Katayama S. Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 Harvard Style Al-athamneh, A., Al-haj, . O., Khaleel, . A. & Katayama, . S. (2026) Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 Turabian Style Al-athamneh, Ahmad, Omar Al-haj, Anas Khaleel, and Shigeru Katayama. 2026. Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 Chicago Style Al-athamneh, Ahmad, Omar Al-haj, Anas Khaleel, and Shigeru Katayama. "Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model." doi:10.5455/OVJ.2026.v16.i3.47 MLA (The Modern Language Association) Style Al-athamneh, Ahmad, Omar Al-haj, Anas Khaleel, and Shigeru Katayama. "Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model." doi:10.5455/OVJ.2026.v16.i3.47 APA (American Psychological Association) Style Al-athamneh, A., Al-haj, . O., Khaleel, . A. & Katayama, . S. (2026) Fermentation modifies camel-milk protein fractions (SDS-PAGE/SEC) and associates with lower IgE in an ovalbuminsensitized mouse model. doi:10.5455/OVJ.2026.v16.i3.47 |