Open Veterinary Journal, (2026), Vol. 16(3): 1534-1555
Research Article
10.5455/OVJ.2026.v16.i3.12
Effects of probiotic formulations on gastrointestinal tract morphological changes and gut microbiota in Hu lambs
Ting Xu1, Siling Ding1, Ye Xu1, Siyu Yang1, Yian Yao1, Pengcheng Wan2, Guoqing Shi2, Yimin Wang3,
Feng Guan and Junfang Jiang4*
1College of Life Sciences, China Jiliang University, Hangzhou, China
2Research Institute of Animal Husbandry and Veterinary, Xinjiang Agricultural Reclamation Academy, Shihezi, China
3Hangzhou Caiyang Animal Husbandry Co., Ltd, Hangzhou, China
4Institute of Animal and Veterinary Science, Academy of Zhejiang Agriculture Science, Hangzhou, China
*Corresponding Author: Junfang Jiang. Institute of Animal and Veterinary Science, Academy of Zhejiang Agriculture Science, Hangzhou, China. Email: jiangjunfang1031 [at] sina.cn
Submitted: 29/10/2025 Revised: 25/01/2026 Accepted: 05/02/2026 Published: 31/03/2026
© 2026 Open Veterinary Journal
This is an Open Access article distributed under the terms of the Creative Commons Attribution-Non Commercial-No Derivatives License (http://creativecommons.org/licenses/by-nc-nd/4.0/), which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited, and is not altered, transformed, or built upon in any way.
Abstract
Background: Gut microbiota plays an important role in the digestive system of ruminants, affecting the health status of Hu sheep and their development and production traits. Factors such as gender, age, diet, and probiotic formulations may influence their compositions.
Aim: This study aimed to investigate the morphological changes in the gastrointestinal tract and gut microbiota dynamics during postnatal development of Hu lambs and evaluate the effects of probiotic supplementation.
Methods: During the 60-day experiment, rumen, duodenum, jejunum, and ileum samples were collected at 2 hours, 20 days, and 55 days after birth. High-throughput 16S rRNA gene sequencing was employed to analyze microbial diversity, and morphological analysis was conducted to compare differences in the digestive tract surface tissues.
Results: The results indicated that the length of the rumen papillae and the height of the villi in the jejunum and ileum continued to increase with increasing age. The overall diversity of the gut microbiota exhibited a gradual upward trend. The microbial diversity indices were higher in the experimental group than in the control group at corresponding time points (p > 0.05). The number of beneficial bacteria, such as Ruminococci, increased, whereas the number of potentially pathogenic bacteria, such as Escherichia spp., decreased.
Conclusion: Probiotic supplementation promotes early gastrointestinal tract development, enhances the colonization of beneficial bacteria, and improves microbial diversity in Hu lambs.
Keywords: Hu lambs, Probiotic formulations, 16S rRNA gene sequencing, Gut microbiota, GIT morphology.
Introduction
Sheep are a pivotal species in global animal husbandry, being distributed worldwide and serving as a significant economic resource for herders. With the evolution and scaling of the livestock industry, sheep farming has become a ornerstone of this sector. Prolificacy is a critical determinant in the development of meat sheep farming (Fu et al., 2022). Hu sheep are renowned for their high fertility, early sexual maturity, continuous estrous cycles, adaptability to hot and humid climates, and suitability for intensive housing systems (Guo et al., 2023). This breed has one of the highest fertility rates globally, with lambing rates exceeding 280% (Guan et al., 2007; Li et al., 2022), making it a primary choice for meat sheep farming and crossbreeding programs. However, Hu sheep are known for their low carcass yield and inferior meat quality (Kong et al., 2023).
In sheep breeding, feed expenses can constitute up to 70% of total costs (Zhang et al., 2017a), while diseases such as colds, pneumonia, and diarrhea in lambs, along with milk fever and mastitis in ewes, are prevalent (Zhao et al., 2024a). Therefore, enhancing feed conversion efficiency and immune competence is crucial for sheep breeding’s economic gains. Genetic predisposition and gut microbiota influence these factors. Nutrient absorption, energy metabolism, and immune regulation in animals are intricately linked to the gut microbiota (Shen et al., 2017; Abecia et al., 2018; Arshad et al., 2021). The gut microbiota is a vital component of ruminant growth and health. The interaction between the gut microbiota and the host intestinal environment plays a key role in digestion, metabolism, immunity, and disease prevention (Cholewińska et al., 2020a; Newbold and Ramos-Morales, 2020; Zhou et al., 2023). Given the substantial intake of crude fiber by ruminants, the breakdown of cellulose depends on cellulolytic bacteria within the gut microbiota (Wang et al., 2017). As a key site for digestion and absorption, the gut microbiota exerts a substantial influence on the health and performance of the host (Ma et al., 2022).
In the animal husbandry industry, probiotic formulations have been widely employed as alternatives to antibiotics, offering non-toxicity, non-residue, and non-pathogenicity (Dong et al., 2024). Previous studies have demonstrated that feeding probiotic formulations enhances immune parameters and growth performance in Hu sheep pre- and post-weaning (60 days of age) (Yao et al., 2022). The addition of probiotic formulations to the diet of Hu sheep significantly increases the carcass yield and feed conversion ratio, enhances immunity and antioxidant capacity, and regulates gut microbial homeostasis (Wang et al., 2024b). Moreover, feeding probiotics to ruminants has been shown to improve nutrient intake and digestibility, milk production, immunity, reproduction, and feed conversion efficiency (Kulkarni et al., 2022). Furthermore, feeding probiotic formulations significantly increases total blood protein, globulin, and weight gain in lambs (Devyatkin et al., 2021), as well as enhances daily weight gain, feed digestibility, and body immunity in weaned goats (Lu et al., 2023). Probiotic formulations primarily improve the structure and function of microbial communities and influence animal behaviors such as feed intake in ruminants (Schofield et al., 2018). Probiotic formulations adhere to the inner wall of the digestive tract, forming colonization resistance in the intestine. Over time, a stable microbial community is established, which regulates the structure and quantity of intestinal microbiota, thereby influencing the immune system and growth performance of the host (Wang et al., 2022a).
Currently, commercial probiotic formulations primarily include probiotics, prebiotics, and synbiotics, which are widely used in sheep farming and offer various beneficial functions (Yao et al., 2022; Yao et al., 2023), thereby improving sheep breeding efficiency. In Hu lambs, commercially available probiotic formulations designed with slow-release encapsulation technology and containing multiple probiotics can enhance immunity and promote rapid growth during the short-term growth phase (Yao et al., 2022). However, the effects of such probiotic formulations on the digestive system microorganisms of lambs and the alteration of gut microbiota composition remain unclear. Therefore, this experiment provides a theoretical basis for elucidating the developmental processes of the gastrointestinal tract (GIT), the basic characteristics of the gut microbiota, and the effects of probiotic formulations in the feeding of Hu lambs.
Materials and Methods
Experimental material
The probiotic formulation was purchased from Qingdao Zhengtai United Nutritional Technology Co., Ltd. (product name “Duyikang”), provided in a cream formulation with a volume of 60 ml per tube. The labeled ingredients included Bacillus licheniformis (≥ 3.7 × 107 CFU/g), Bacillus subtilis (≥ 3.7 × 107 CFU/g), Enterococcus faecalis (≥ 6.0 × 107 CFU/g), and Lactobacillus rhamnosus (≥ 3.7 × 107 CFU/g) as the main probiotics, along with prebiotics such as colostrum extract, medium-chain triglycerides, trace elements, and vitamins.
Laboratory animals and sample collection
This experiment was conducted over a 60-day period from October to December 2022 at Hangzhou Caiyang Herding Co., Ltd. (Zhejiang, China). Fifteen lambs with similar birth times and weights were selected from 56 newly born male Hu lambs that were twin-born and randomly assigned to five groups (n=3). The initial body weight of all lambs was recorded within 2 hours after birth, before any experimental treatment. The average birth weight was 3.54 ± 0.83 kg, with no significant difference (p > 0.05) among the groups. The groups were as follows: one blank control group (BG00), two control groups (CG20 and CG55), and two experimental groups (TG20 and TG55).
The GIT contents of lambs in Blank Group were sampled within 2 hours after birth. Lambs in TG20 and TG55 were orally administered 10 ml of the probiotic formulations within 2 hours after birth, whereas lambs in CG20 and CG55 did not receive the probiotic formulations. All lambs in the Treatment Group (TG) and Control Group (CG) groups were suckled naturally under identical conditions, with supplemental lamb milk substitute provided from approximately 30 days of age. Free feeding was allowed, and all experimental lambs were tagged using ear tags with wool staining for identification.
All experimental procedures involving Hu sheep were conducted in accordance with the China Council on Animal Care guidelines. At the corresponding time of experimental design, namely 0 day, 20 days, and 55 days after lamb birth, no food was fed on the day of sample collection. All animals from each group were euthanized using captive bolt stunning. Physical disruption methods are often followed by exsanguination as an adjunctive method to ensure humane euthanasia. Trained personnel carried out the slaughter procedures in accordance with standard animal welfare protocols. Immediately after slaughter and dissection, the sampled organs were rinsed clean with ice-cold sterile phosphate-buffered saline. Intestinal segments were defined as follows: the proximal third (duodenum), mid-third (jejunum), and distal third (ileum) of the small intestine were defined. For each target segment (duodenum, jejunum, and ileum), a section approximately 3 cm in length was ligated proximally and distally using sterile suture material. The isolated segment was carefully excised using sterile surgical scissors. The luminal contents within this isolated segment were gently expressed into sterile collection tubes. One section of each sample was snap-frozen in liquid nitrogen and then stored at −80°C until analysis. The remaining portion was fixed in 10% neutral buffered formalin for histological analysis. Samples from the rumen, duodenum, jejunum, and ileum contents were collected from lambs in CG20 and TG20, as well as those in CG55 and TG55, at 20 and 55 days of age, respectively, based on the rate and timing of weight gain influenced by feeding the probiotic formulations (Yao et al., 2022). For BG00, GIT contents were similarly collected from the same anatomical sites within 2 hours after birth, snap-frozen in liquid nitrogen, and stored at −80°C.
Morphological observations
Tissue samples from the rumen, duodenum, jejunum, and ileum were collected following the GIT content sampling strategy. Morphological analysis was performed using hematoxylin-eosin staining and light microscopy. The methodology followed (Wang et al., 2022b), with the following procedure: tissue samples were fixed in paraffin, and the embedded paraffin blocks were sectioned into 4 μm-thick cross-sections. The sections were stained with hematoxylin and eosin and observed under a light microscope (Eclipse Ci, Nikon, Melville, NY). Photographs of the tissues were captured using an imaging system (Digital Sight DS-Fi2, Nikon, Melville, NY, USA). The length and width of rumen papillae, muscularis propria depth, villus height, crypt depth, and muscularis propria thickness were measured in the duodenum, jejunum, and ileum. Subsequently, the villus height to crypt depth (V/C) ratio was calculated.
DNA extraction and polymerase chain reaction (PCR) amplification of gut microbiota
Total genomic Deoxyribonucleic Acid (DNA) was extracted from samples collected from different organs using a Bacterial Genome Extraction Kit (Tiangen Biotech Co., Ltd., Beijing, China) according to the manufacturer’s instructions. DNA concentration and purity were determined using a NanoDrop 2,000 spectrophotometer (Thermo Scientific, Wilmington, United States), and DNA integrity was assessed using 2% agarose gel electrophoresis.
Polymerase chain reaction amplification targeting the hypervariable V3-V4 region of the 16S rRNA gene was performed using the primers 515F (5′-GTGYCAGCMGCCGCGGGTAA-3′) and 806R (5′-GGACTACHVGGGTWTCTAAT-3′) (Caporaso et al., 2011; Zhao et al., 2024b). Positive PCR products were purified using the GeneJET® Gel Extraction Kit (Thermo Scientific), and sequencing libraries were constructed with the Ion Plus Fragment Library Kit (Thermo Scientific). The library quality was evaluated using a Qubit 2.0 fluorometer (Thermo Scientific).
16S ribosomal RNA sequencing and analysis
Sequencing was performed on the Ion S5TM XL platform, generating single-end reads of 407–412 base pairs. Sequencing data were processed by splitting sequences based on barcode information, followed by removal of barcode and primer sequences using the cutadapt (v1.9) software (Martin, 2011). The overlapping region sequences were merged using FLASH (v1.2.8) (Magoč and Salzberg, 2011). Sequencing reads were subjected to quality control using a window scanning method implemented in trim, with discarded sequences shorter than 100 bp after truncation. The chimeric sequences were identified and removed using Vsearch (v2.3.4). DADA2 was employed via QIIME2 for length filtering and denoising to obtain Amplicon sequence variant (ASV) feature sequences and abundance tables (Schloss et al., 2009; Callahan et al., 2016). Taxonomic annotation was performed on the ASV sequence files using the SILVA database (Release 138, https://www.arb-silva.de/documentation/release138/, annotation threshold: --minimum_confidence 0.7) and the NT-16S database (Release 20230718, annotation threshold: --minimum_ident 90 --minimum_cov 80 --maximum_e 1e-5) (Quast et al., 2013). Subsequently, the abundance of taxa at each taxonomic level across all samples was calculated based on the ASV abundance table.
Based on the ASV feature sequences and abundance data, α-diversity and β-diversity analyses were performed. Comparative analyses of species abundance were conducted between the control and experimental groups, with a significance threshold of p < 0.05. Additionally, the LDA effect size (LEfSe, LDA scores > 3) enabled the identification of important bacteria between groups.
Statistical analysis
The experimental data were organized using Excel 2021, and SPSS 23.0 was used for statistical analyses. A one-way analysis of variance was used for comparisons among multiple groups. The Least Significant Difference method was employed to determine statistical significance, with p < 0.05 considered indicative of a significant difference. Results are expressed as mean ± standard deviation.
Ethical approval
All experimental procedures involving Hu sheep were conducted in accordance with the China Council on Animal Care guidelines. The experimental animal management committee of China Jiliang University approved the protocols (approval number: 2022-014). The ethical approval was approved on August 1, 2022.
Results
Changes in GIT morphological structure and effects of probiotic formulations
The GIT is the primary organ responsible for nutrient absorption in animals, and its developmental status directly impacts the efficiency of nutrient absorption. Histological and morphological analyses revealed that the length and width of the rumen papillae, as well as the thickness of the rumen musculature, were significantly greater at 55 days of age than at 20 days (p < 0.05), consistent with the growth of the lambs. In the duodenum, the villi height was higher at birth, gradually decreased, and subsequently increased with age. Similarly, over time, the depth of the crypts increased, the V/C ratio decreased, and the muscularis propria thickness increased.
In the control group, the villus height in the duodenum at both 20 and 55 days of age was significantly lower than that at birth. The crypt depth and muscularis propria thickness were significantly greater at 55 days compared with those at 20 days and at birth. The V/C ratio in control lambs at both 20 and 55 days of gestation was significantly lower than that at birth. In the jejunum, the villus height at 55 days was significantly higher than that at 20 days and at birth. The crypt depth and muscularis propria thickness at 55 days were also significantly greater than those at 20 days and at birth. The V/C ratio at birth and 55 days was significantly higher than that at 20 days. In the ileum, the villus height and crypt depth at 55 days were significantly greater than those at 20 days and birth, while the muscularis propria thickness was also significantly higher at 55 days compared to earlier stages. However, no significant differences in V/C were observed among the three control groups.
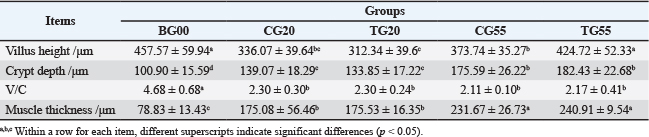
Comparative analysis between the experimental and control groups indicated no significant difference (p > 0.05) in the length of the rumen papillae at 20 days. However, at 55 days, the length of the rumen papillae in the experimental group increased significantly (p < 0.05). The rumen morphology data are summarized in Table 1, with representative images shown in Figure S1. No significant difference (p > 0.05) was observed in the width of the rumen papillae or the thickness of the muscularis layer between the experimental and control groups at the same age. In the duodenum, no significant difference in villus height (p > 0.05) was observed between the experimental and control groups at 20 days. However, at 55 days, the villus height in the experimental group was significantly greater (p < 0.05). The duodenal morphology is depicted in Figure 1 and Table 2. Additionally, no significant differences (p > 0.05) were observed in crypt depth, V/C, or muscularis layer thickness in the duodenum between the experimental and control groups at any age. Similarly, no significant differences (p > 0.05) were found in villus height, crypt depth, V/C, or muscularis layer thickness in the histomorphology of the jejunum and ileum.
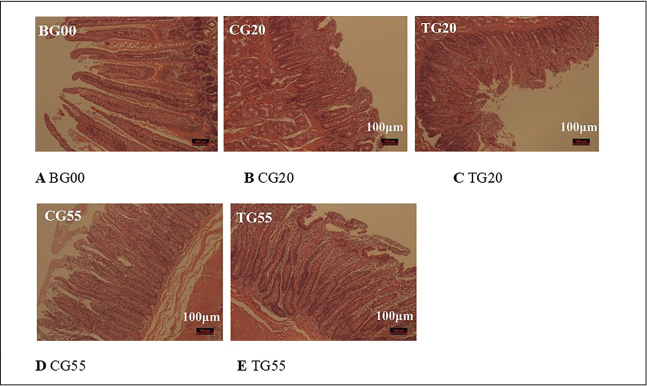
Fig. 1. Effect of probiotic formulations on duodenal morphology (10×). Representative photomicrographs (10× magnification) of duodenal sections from Hu sheep, stained with hematoxylin and eosin (H&E), illustrating villus height, crypt depth, and overall muscle thickness integrity. Images correspond to (A) BG00: Blank group at birth (0 days), before any treatment. (B) CG20: control group at 20 days of age, without probiotic treatment. (C) TG20: probiotic-treated group at 20 days of age. (D) CG55: Control group at 55 days of age. (E) TG55: Probiotic-treated group at 55 days of age. Morphological comparisons demonstrate the postnatal development of the duodenum and the structural influence of early probiotic intervention.
Table 1. Effect of probiotic formulations on rumen tissue morphology in Hu sheep.
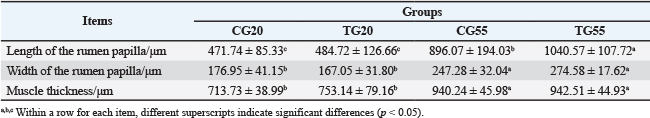
Table 2. Effect of probiotic formulations on duodenal morphology in Hu sheep.
Changes in gut microbiota diversity in control lambs
High-throughput sequencing of the microbiota from the rumen, duodenum, jejunum, and ileum contents of the BG00, CG20, and CG55 groups revealed plateaued Shannon curves at sequencing depths exceeding 5,000 reads (Fig. S2). This indicated that the sequencing depth and data volume of the 16S rRNA V3-V4 gene region were sufficient for diversity analysis. A Venn diagram was used to visualize the specific and shared ASVs among the three groups. The Venn diagram illustrates the distribution of unique and shared ASVs among the three developmental groups (Fig. 2). A consistent trend was observed across all four intestinal sites: total ASV richness was lowest in the newborn group (BG00) and highest in the weaned group (CG55), reflecting a progressive expansion of the microbial community with postnatal growth. This increase in ASV count indicates that the lamb’s gut microbiota undergoes substantial diversification during early development. Furthermore, the relatively small core microbiome shared across all stages, compared to the considerable number of stage-specific ASVs, indicates that gut ecosystem maturation is characterized by microbial succession, not merely accumulation.
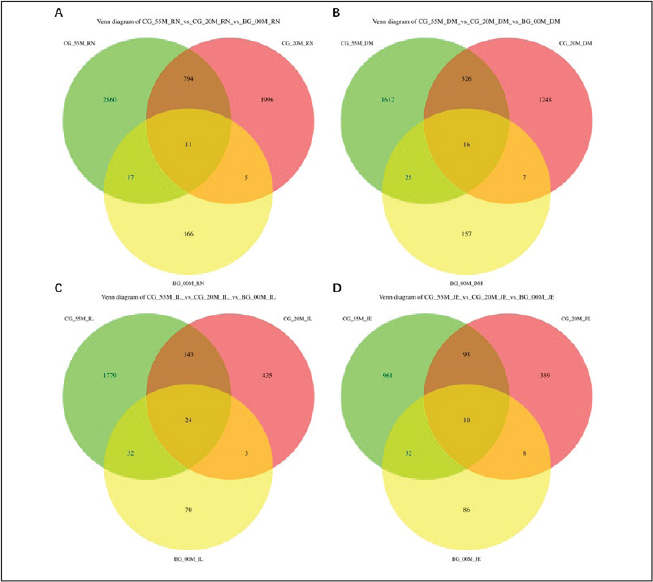
Fig. 2. Effect of probiotic formulations on duodenal morphology (10×). Representative photomicrographs (10× magnification) of duodenal sections from Hu sheep, stained with hematoxylin and eosin (H&E), illustrating villus height, crypt depth, and overall muscle thickness integrity. Images correspond to (A) BG00: Blank group at birth (0 days), before any treatment. (B) CG20: control group at 20 days of age, without probiotic treatment. (C) TG20: probiotic-treated group at 20 days of age. (D) CG55: Control group at 55 days of age. (E) TG55: Probiotic-treated group at 55 days of age. Morphological comparisons demonstrate the postnatal development of the duodenum and the structural influence of early probiotic intervention.
Comparison of α diversity in different gastrointestinal regions at various growth stages (Table 3) showed that the Chao1 and Shannon indices displayed an upward trend with age in the rumen. The Chao1 index increased significantly at 20 days compared with that at birth (p < 0.05), and both the Chao1 and Shannon indices were significantly higher at 55 days compared with that at birth (p < 0.05; Fig. S3). The Chao1 index in the duodenum increased with age, but the change was not significant. In the jejunum, the α diversity indices followed a clear “V” pattern, being higher at birth and 55 days and lowest at 20 days, although the differences were not significant. In the ileum, the Chao1 index showed a continuous increase, with significant differences at 20 and 55 days compared with that at birth (p < 0.05). However, the Simpson and Shannon indices exhibited a "V-shaped" pattern, with significantly higher values at birth and 55 days than at 20 days. These findings indicate an overall increasing trend in the diversity and abundance of gut microbiota as lambs grow.
Table 3. Changes in α-diversity of gut microbiota of Hu lambs at 0, 20, and 55 days of age.
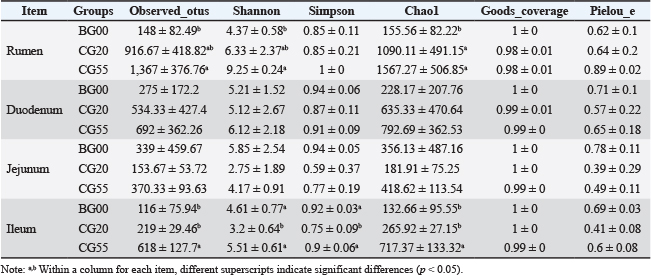
The results of β diversity analysis using principal coordinates analysis (PCoA) (Fig. S4) demonstrated distinct separations among the three control groups at birth, 20 days, and 55 days. Samples were aggregated within each group, showing significant differences in gut microbiota composition among the three stages. This indicates that the gut microbiota structure undergoes substantial changes, with increased diversity during lamb growth.
Gut microbiota biomarkers in control lambs
To analyze changes in bacterial microbiota during lamb growth at both the phylum and genus levels, LEfSe was used to identify significantly different bacterial taxa (LDA scores > 3) across the control groups. The BG00 group was enriched with 20 bacterial taxa in the rumen, including 1 phylum and 5 genera (Fig. 3). The CG20 group exhibited enrichment in 28 bacterial taxa, comprising 2 phyla and 12 genera, whereas the CG55 group exhibited enrichment in 40 bacterial taxa, including 1 phylum and 16 genera. The BG00 group was enriched in 26 bacterial taxa in the duodenum, encompassing 2 phyla and 5 genera. The CG20 group was enriched in 8 bacterial taxa, including 4 genera, and the CG55 group was enriched in 14 bacterial taxa, involving 6 genera. In the ileum, the BG00 group was enriched in 18 bacterial taxa, including 5 genera. The CG20 group exhibited enrichment in 6 bacterial taxa, comprising 4 genera, whereas the CG55 group showed enrichment in 25 bacterial taxa, encompassing 2 phyla and 7 genera.
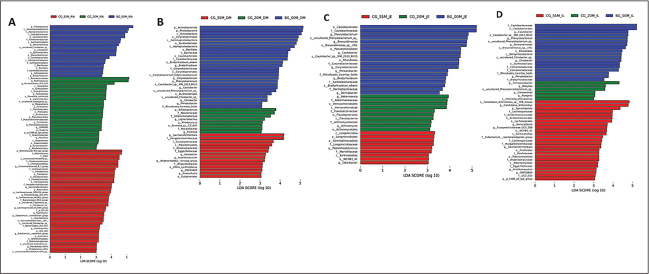
Fig. 3. Venn diagrams of the ASV composition in the gut microbiota of Hu lambs at 0, 20, and 55 days of age. Venn diagrams illustrate the overlap and uniqueness of ASVs in the microbiota from (A) rumen, (B) duodenum, (C) jejunum, and (D) ileum contents of Hu lambs at 0 (BG00), 20 (CG20), and 55 (CG55) days of age. Each circle represents an age group. The overlapping areas indicate the number of ASVs shared between groups, whereas the non-overlapping sections represent ASVs unique to a specific group. This visualization reveals the patterns of microbial community similarity and specificity across different gut sites as lambs aged.
A comparative analysis of the gut microbiota from birth to 55 days of age identified 33 genera with significant differences in the rumen, 16 in the duodenum, 11 in the jejunum, and 16 in the ileum. The BG00 group exhibited the lowest number of genera in the rumen, indicating that the bacterial microbiota of Hu lambs is relatively established but limited early in life. As lambs matured, the relative abundance of Ruminococcus, Prevotellaceae, Megasphaera, Fibrobacterota, Richenellaceae_RC9_gut_group, Lachnospiraceae, Christensenellaceae_R-7_group, and Ligilactobacillus increased, whereas the relative abundance of Caulobacter and Citrobacter decreased. The relative abundance of Firmicutes, Bacteroidetes, Prevotella, and Christensenellaceae_R-7_group increased in the duodenum. In the jejunum, Firmicutes and Akkermansia were increased, with a concomitant decrease in Proteobacteria. Proteobacteria and Actinobacteria decreased in the ileum, whereas Firmicutes, Prevotella, and Christensenellaceae_R-7_group increased.
Effect of probiotic formulations on the diversity of gut microbiota in Hu lambs
Following the administration of probiotic formulations, ASV visualization analysis of 16S rRNA sequencing Venn diagrams for gastrointestinal contents in the TG20 and TG55 groups indicated that the number of ASVs in the rumen, duodenum, jejunum, and ileum increased with the growth of Hu lambs (Fig. S5). The diversity indices in the experimental groups were higher than those in the control groups at corresponding time points, although the differences were not statistically significant (p > 0.05). PCoA based on Bray-Curtis distance demonstrated less pronounced separation of gut microbiota in the TG groups compared to the CG groups over the same period, with lower levels of within-group aggregation.
Effects of probiotic formulations on gut microbiota structure and composition in Hu lambs
A comparative analysis of microbial communities in the rumen, duodenum, jejunum, and ileum of the experimental and control groups at 20 and 55 days revealed increased microbial diversity indices and relative abundances at both the phylum and genus levels (Figs. 4 and 5). However, these increases were not statistically significant. Firmicutes were identified as the dominant phyla across all gastrointestinal sites in both the experimental and control groups at 20 and 55 days of age. Variations in the microbial communities were evident among the gastrointestinal sites. Firmicutes and Bacteroidetes were predominant in the rumen of all groups. Firmicutes remained dominant in the duodenum and jejunum, whereas Firmicutes and Proteobacteria were major phyla in the ileum at 20 days, with Proteobacteria declining by 55 days as Firmicutes became predominant.
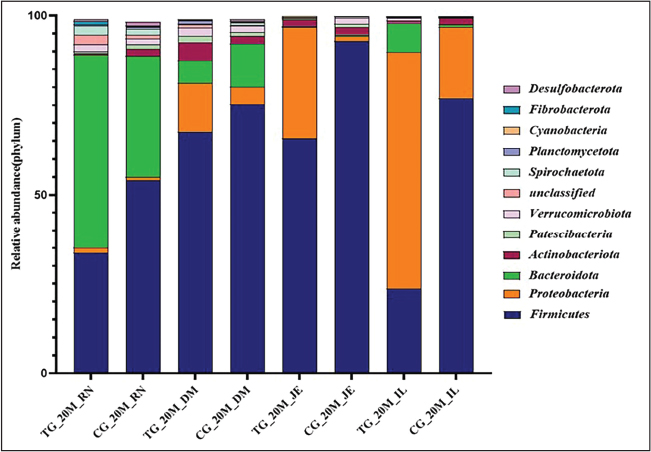
Fig. 4. Changes in the relative abundances of 20-day-old microbiota in the treatment and control groups at the phylum level. Stacked bar charts show the relative abundance profile of the bacterial communities at the phylum level in 20-day-old Hu lambs across different gut sites, comparing probiotic-treated groups (TG20) with their corresponding control groups (CG20). Only the dominant bacterial phyla with an average relative abundance of >1% are shown. The color legend identifies each phylum, allowing for a visual comparison of how the treatment influenced the overall microbial community structure in young lambs.
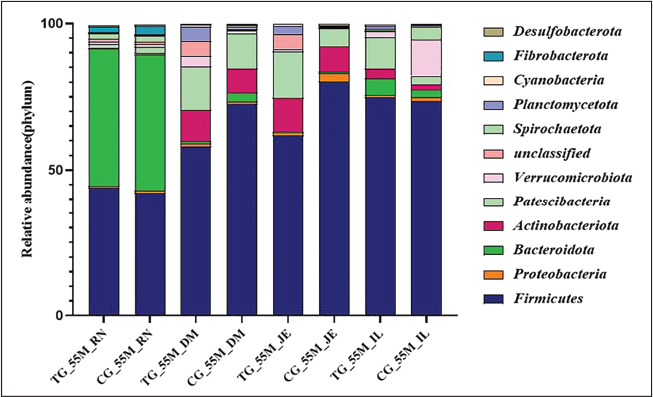
Fig. 5. Changes in the relative abundance of the 55-day-old microbiota in the treatment and control groups at the phylum level Stacked bar charts show the relative abundance profile of the bacterial communities at the phylum level in 55-day-old Hu lambs across different gut sites, comparing probiotic-treated groups (TG55) with their corresponding control groups (CG55). Only the dominant bacterial phyla with an average relative abundance of >1% are shown. The color legend identifies each phylum, allowing for a visual comparison of how the treatment influenced the overall microbial community structure in young lambs.
At the genus level, microbial species varied across organs and groups, with no significant differences observed (Figs. 6 and 7). In the rumen, the relative abundance of Rikenellaceae_RC9_gut_group, Succiniclasticum, and Prevotella increased in the experimental group, whereas that of Muribaculaceae_unclassified decreased. In the duodenum, the experimental group exhibited increased relative abundance of Lactobacillus, Christensenellaceae_R-7_group, Prevotella, and Rikenellaceae_RC9_gut_group, while Clostridium_sensu_stricto_1 decreased. In the jejunum, the experimental group demonstrated increases in Lactobacillus and Ruminococcus, with declines in Romboutsia, Clostridium_sensu_stricto_1, Sarcina, and Candidatus_Arthromitus. Lactobacillus, Candidatus_Saccharimonas, and Lachnospiraceae_UCG_002 showed increased relative abundance in the ileum in the experimental group (Figs. S6 and S7).
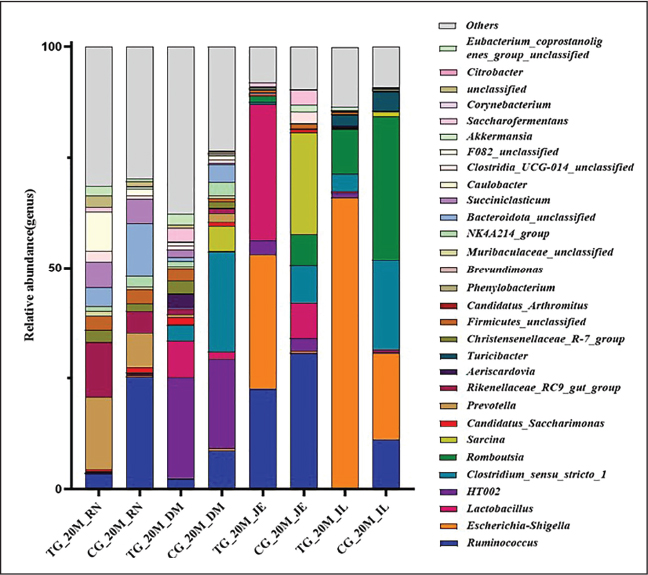
Fig. 6. Changes in the relative abundance of 20-day-old microbiota in the treatment and control groups at the genus level Stacked bar charts display the relative abundance of gut microbiota at the genus level in 20-day-old lambs across groups. Genera with low average abundance are grouped as "Others." The figure highlights the distribution of key functional genera or genera showing significant differences in abundance between the treatment and control groups.
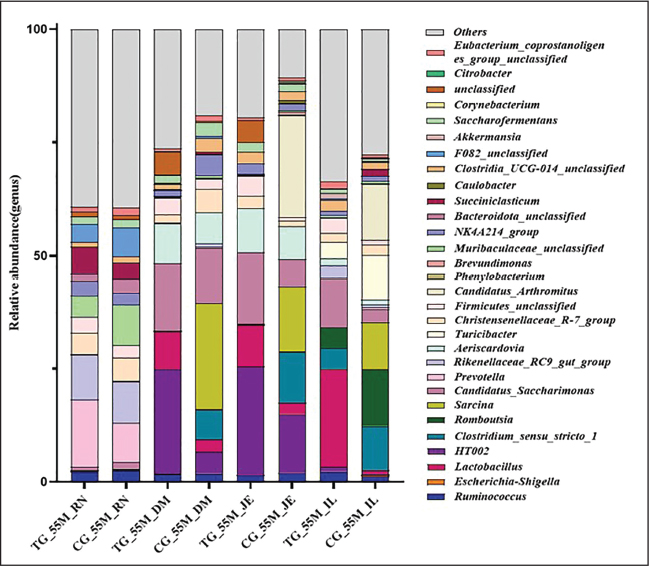
Fig. 7. Changes in the relative abundance of 55-day-old microbiota in the treatment and control groups at the genus level Stacked bar charts display the relative abundance of gut microbiota at the genus level in 55-day-old lambs across groups. Genera with low average abundance are grouped as "Others." The figure can be used to assess the long-term impact of probiotic intervention on the colonization or proliferation of specific bacterial genera during gut microbiota maturation.
Discussion
Hu sheep, a renowned, prolific indigenous breed in China, provides valuable insights into ovine growth, development, and physiological processes. The prolificacy of this breed not only enhances sheep farming productivity but also necessitates additional investments in rearing multiple lambs. These investments include artificial feeding, fostering, and the development and application of various multifunctional protein materials. Additionally, probiotic formulations and other management strategies are employed to increase lambs’ survival and growth rates from multiple births. Probiotics commonly used in animal husbandry, such as Lactobacillus, Enterococcus, Bacillus, and Bifidobacterium, exert beneficial effects on animal growth (Zamojska et al., 2021). Combined probiotics are generally more effective than single strains (Ma et al., 2023; Wang et al., 2024a), as they exhibit synergistic or cooperative effects (Ouwehand et al., 2018; Sugiharto et al., 2018). Specifically, B. licheniformis, which is included in probiotic supplements, promotes growth, enhances antioxidant and immune functions, and increases the population of beneficial intestinal bacteria in sheep ((Jia et al., 2018). Commercial probiotic mixtures containing B. licheniformis have been shown to improve growth rates, bolster immune function, and enhance intestinal microbiota diversity, thereby supporting metabolic homeostasis (Devyatkin et al., 2021).
The interaction between the gut microbiota and the host is essential for the development of the host’s gastrointestinal, immune, metabolic, and neurological systems (Liu et al., 2022). Numerous studies have demonstrated that early microbial colonization significantly influences long-term health across an organism’s life cycle (Tamburini et al., 2016; Krajewska-Włodarczyk 2017; Taylor et al., 2021). The larval stage is critical for the establishment and maturation of microbiota, with the composition of gastrointestinal microbiota playing a crucial role in growth and health (Laustsen et al., 2021). This study analyzed the intestinal microbiota of Hu lambs from birth to weaning and identified significant increases in beneficial bacterial populations along with growth and diversification. Furthermore, the administration of probiotic formulations significantly enhanced the probiotic microbiota in the rumen, duodenum, jejunum, and ileum. These findings are consistent with the observed improvements in immunity and growth rates during the same period (Yao et al., 2022).
Microbiota analysis revealed that Firmicutes predominated in the rumen, duodenum, jejunum, and ileum, alongside Bacteroidetes and Proteobacteria, throughout the growth period. The diversity of microbiota increased with the growth of the lambs, consistent with the main probiotic components of the probiotic formulations used in this experiment. The formulation included B. licheniformis, B. subtilis, E. faecalis, and L. rhamnosus, all of which belong to the Firmicutes phylum. In the control group, microbial diversity exhibited a "V-shaped" pattern from birth to 55 days of age, with a decline at 20 days followed by an increase. This pattern is associated with the initial acquisition of microbiota exclusively from the dam. A reduction in microbial diversity occurs at 20 days, as the lambs have not yet established their distinct microbiota. The original microbial populations declined, and by 55 days of age, lambs gradually developed their own microbial communities through environmental exposure and maternal milk intake, resulting in significant differences across the three periods.
The introduction of sustained-release strains resulted in a convergence of microbial community types across different organs and between the ages of 20 and 55 days following the administration of probiotic formulations. This reduced inter-group differences and led to lower intra-group clustering, suggesting that the additive had consistent effects. Consequently, the study confirms that feeding probiotic formulations can enhance the population of dominant microbial species in GIT of Hu lambs, thereby ensuring the colonization of beneficial microbes and improving immune function and growth rates.
Morphological analysis revealed significant differences in gastrointestinal tissue structure across different stages in the control group of Hu lambs from birth to 55 days of age. Notably, the length and width of the rumen papillae, as well as the rumen muscle layer, were significantly greater at 55 days of age than at 20 days of age. The increase in rumen papillae dimensions is believed to enhance the absorption efficiency of volatile fatty acids (VFAs), thereby improving feed conversion rates and promoting growth and development (Na and Guan, 2022). These findings reveal significant improvements in rumen fermentation performance as Hu lambs mature.
In the duodenum, the V/C ratio decreased significantly from birth to 20 and 55 days of age. This reduction is likely associated with the initial need for maternal milk absorption in newborn lambs. The absorptive surface area of the intestine is determined by the height of the villus, with taller villi correlating with greater absorptive capacity. Villus height, crypt depth, and V/C ratio reflect the capacity of the small intestine for nutrient digestion and absorption, while intestinal muscularis thickness indicates rhythmic contractile capability (Zhang et al., 2024a).
Administration of probiotic formulations significantly increased the length of rumen papillae in 55-day-old lambs (p < 0.05), with a concurrent increase in width, although early changes were less pronounced. This may be related to the dietary transition from a milk-based diet to concentrate feeds such as cornmeal and subsequently to pelleted feed. Probiotics ingested and colonized gradually exerted their effects through probiotic formulations by breaking down complex compounds like fiber, leading to the production of VFAs that promote the growth of rumen papillae (Na and Guan, 2022). Consequently, this enhanced the growth performance of lambs, consistent with findings that lambs fed probiotic formulations exhibited higher relative growth rates after 30 days of age.
Probiotic formulations also significantly increased duodenal villus height in 55-day-old lambs (p < 0.05), potentially enhancing nutrient absorption and contributing to the observed growth improvements. The formulation had minimal impact on intestinal tissue changes in 20-day-old lambs, possibly due to the colonization and effects of probiotics being influenced by postnatal dietary structure and digestive tract development. The observed maturation of the gut microbiota and mucosal structure during early life aligns with and may functionally underpin the improvements in systemic growth performance indicators (including enhanced ADG in later stages and elevated blood protein profiles) reported for Hu lambs receiving the same probiotic intervention (Yao et al., 2022).
In evaluating microbial diversity, α diversity represents the diversity within a specific environment or ecosystem, encompassing species richness and evenness. In contrast, β diversity reflects the similarity or dissimilarity between individuals or groups, where greater distances between samples indicate more pronounced differences in microbial community structures. Together, α and β diversities define a given environmental community’s overall diversity or biological heterogeneity.
In this study, analyses of α and β diversities revealed that microbial diversity in both the rumen and duodenum of Hu lambs was lowest at birth, intermediate at 20 days of age, and highest at 55 days of age. However, in the jejunum and ileum, α diversity indices at 20 days of age were lower than those observed at birth. This may be attributable to the transitional nature of postpartum microbial maturation, the singularity of a milk-based diet, and the decline of initial microbial communities as the intestinal environment develops (Amat et al., 2021; Ahearn-Ford et al., 2022). Microbial diversity had surpassed birth levels by 55 days of age, indicating that gastrointestinal microbial diversity increases dynamically rather than linearly with age. Significant changes were observed in the gastrointestinal microbial community structure, suggesting that enhanced microbial diversity is correlated with healthy growth in Hu lambs (Yao et al., 2022).
Furthermore, the microbial community structures in the rumen, duodenum, jejunum, and ileum of lambs were significantly different at 20 days of age compared with those at both birth and 55 days of age. These findings indicate that substantial changes in the gut microbiota occur from birth to weaning, after which the community structure gradually stabilizes. Supplementation with probiotic formulations was found to increase both α and β diversities in the gut microbiota of Hu lambs, although the effect was not pronounced, consistent with the observations by Wang et al. (2021b). This reflects that probiotic supplementation can modulate the gut microbiota of lambs. However, because our microbiota analysis was resolved only to the genus level, the colonization status of the administered probiotic species remains unconfirmed. The observed changes may indicate a modulatory effect, or a period longer than 30 days may be required for the probiotics to exert a more stable influence on the microbial community.
Certain additives can alter the community structure and diversity of rumen microorganisms (Zhang et al., 2017b; Bi et al., 2018; Zhang et al., 2024b), likely influenced by the composition and dosage of probiotic formulations. Different components of probiotic formulations have varying effects on gut microbiota, with high doses of probiotics inducing rapid structural changes. However, the slow-release composite probiotic formulations used in this study were designed primarily to enhance the immunity of weak lambs and improve the colonization of beneficial gut bacteria, aligning largely with the expected outcomes.
As lambs mature, notable changes occur in both microbial composition and relative abundance, particularly in the proliferation of beneficial bacterial species within different GIT regions. Ruminococcus and Lachnospiraceae are fibrolytic bacteria capable of degrading cellulosic polysaccharides, producing beneficial metabolic byproducts, and playing critical roles in feed digestion and absorption (Cheng et al., 2024). Through its cellulolytic and enzyme-binding regions, Succiniclasticum can degrade crystalline cellulose, which other rumen bacteria cannot process, thereby enhancing feed digestion and absorption (Bai et al., 2023; Jin et al., 2023).
The Christensenellaceae_R-7_group has been shown to improve growth performance, enhance intestinal metabolism, and increase protein and fiber breakdown efficiency in animals (Beaumont et al., 2017; Manor et al., 2018). Prevotella species are associated with starch, protein, and polysaccharide degradation and utilization in the rumen (Paz et al., 2018). They play crucial roles in maintaining the balance of the digestive tract, reducing systemic inflammatory responses, and improving digestive capabilities. Megasphaera elsdenii is notable for its ability to use accumulated lactic acid, thereby preventing acidosis—a significant concern in ruminant nutrition. Akkermansia contributes to gastrointestinal homeostasis and metabolic balance (Clarke et al., 2014). Rikenellaceae, which is commonly found in mammalian GIT, plays a significant role in the digestion of coarse fiber, and its abundance is closely linked to the type of feed consumed (Tavella et al., 2021).
This study demonstrated that the composition and abundance of beneficial bacterial genera increase as Hu lambs grow. Moreover, the genera enriched in the treatment group supplemented with probiotic formulations overlapped with those in the control group that increased over time. These included Rikenellaceae RC9_gut_group, Prevotella, and Fibrobacter in the rumen; Christensenellaceae_R-7_group and Prevotella in the duodenum; Lactobacillus and Ruminococcus in the jejunum; and Lactobacillus and Lachnospiraceae_UCG_002 in the ileum. These findings indicate that probiotic formulations positively influence the formation and stabilization of the gut microbiota in lambs.
Extensive research has indicated that Firmicutes and Bacteroidetes are the predominant microbial phyla in the digestive tracts of ruminants, contributing to host metabolism, lipid regulation, and enhanced energy conversion efficiency (Turnbaugh et al., 2006; Schwiertz et al., 2010; Szeligowska et al., 2021; Wang et al., 2021b; Du et al., 2020). Firmicutes, primarily involved in cellulose breakdown, are essential for protein and carbohydrate digestion (Spence et al., 2006; Nicholson et al., 2012; Zhao et al., 2023). Gram-positive bacteria play significant roles in maintaining gut microbiota balance and boosting immunity (Wang et al., 2018; Kwon et al., 2019; Zhang et al., 2022). For instance, Clostridiales species are involved in nutrient metabolism and absorption, providing energy to intestinal epithelial cells (Shan et al., 2022).
The inclusion of Lactobacillus in feed promotes the production of adhesins that colonize the intestinal mucosa, thereby helping maintain microbiota balance and supporting weight gain in ruminants (Du et al., 2019). The findings of this study further substantiate the role of Firmicutes in enhancing ruminant metabolic efficiency and overall health.
Firmicutes are the most abundant phylum in the GIT of Hu lambs, accounting for 40%–90% of the relative abundance (Zhao et al., 2016; Álvarez Narváez et al., 2024). In this study, the relative abundance of Firmicutes in the jejunum of the 55-day-old control group was 80.35%, which is consistent with the findings of Wang et al. (2021a) on the bacterial microbiota characteristics in different intestinal segments of Qinghai semi-fine wool sheep. These results show that the primary microbial structure of lamb GIT forms rapidly after birth and gradually stabilizes. Supplementation with probiotic formulations increased the relative abundance of Firmicutes and Bacteroidetes, albeit without statistical significance. This finding indicates that the preparation may enhance microbial populations without disrupting the existing balance or that such effects may require a longer duration or higher dosage to become clear (Ley et al., 2005).
The gut microbiota of ruminants plays a crucial role in digestion, metabolism, nutritional compensation, and growth, with higher microbial diversity and richness being positively correlated with improved animal health (Yang et al., 2015). Characterization of the Hu sheep rumen microbiota revealed a highly complex and diverse community, with Bacteroidetes and Firmicutes identified as the dominant and core phyla (�holewi44;ska et al., 2020b). In the ileum, an increase in dominant phyla has been associated with reduced diarrhea in animals (Katoh et al., 2023), suggesting that probiotic formulations may be particularly effective in lambs exhibiting diarrheal symptoms (Guo et al., 2024).
In summary, significant changes in the gut microbiota of Hu lambs were observed between birth and 20 and 55 days of age, indicating an increase in microbial diversity and stability as the lambs matured. Due to their unique physiological functions, the rumen and intestines harbored distinct microbial communities; the rumen was dominated by fiber-degrading bacteria, while the small intestine primarily hosted short-chain fatty acid-producing bacteria. Supplementation with probiotic formulations after birth was shown to enhance the gut microbiota abundance, potentially offering probiotic benefits.
Conclusion
Probiotic formulations represent an effective alternative to antibiotics in animal husbandry, playing a vital role in promoting animal health and productivity while exerting beneficial effects on growth. This study demonstrated that supplementation with probiotic formulations sustainably enhanced the diversity of gut microbiota, increased the abundance of beneficial bacteria, and improved digestive function, thereby elevating the health status and growth rate of Hu lambs.
Acknowledgments
Not applicable.
Conflict of interest
We have no competing interests.
Funding
This work was supported by grants from the Zhejiang team science and technology correspondent pairing service plan (Hu sheep, 2025R22A58C03), the National Natural Science Foundation of China (31672394), the Zhejiang Science and Technology Major Program on Agricultural New Variety Breeding (2021C02068-6), the Zhejiang Province Agricultural Major Technology Collaborative Promotion Plan (2023ZDXT16-3), the Project for Zhejiang Province Basic Public Welfare Research Program (LTGN24C050001), the China Agriculture Research System (CARS-39-07), the Young Science and Technology Top Talent Program of Tianshan Talent Training Program in Xinjiang Province (2022TSYCCX0124), the Xinjiang Agriculture Research System (XJARS-09-26), and the Project of Corps Science and Technology in Key Areas (2024AB017).
Authors’ contributions
T.X. and G.S. conceived and designed the study, performed the microbiological analyses, and wrote and revised the manuscript. Y.Y. and S.Y. performed the data analysis and visualization. S.D. and Y.X. performed the animal experiments. Y.W., P.W., and J.J. supervised the project and acquired funding and participated in the experiment. F.G. provided direction and supervision for the study and reviewed and edited the final manuscript. All authors have read and approved the published version of the manuscript.
Data availability
The sequence datasets generated during the current study are available in the GSA repository (https://ngdc.cncb.ac.cn/gsa) under the accession number CRA023269. All other data are included in this article.
References
Abecia, L., Martínez-Fernandez, G., Waddams, K., Martín-García, A.I., Pinloche, E., Creevey, C.J., Denman, S.E., Newbold, C.J. and Yáñez-Ruiz, D.R. 2018. Analysis of the rumen microbiome and metabolome to study the effect of an Antimethanogenic treatment applied in early life of kid goats. Front. Microbiol. 9, 2227; doi:10.3389/fmicb.2018.02227
Ahearn‐Ford, S., Berrington, J.E. and Stewart, C.J. 2022. Development of the gut microbiome in early life. Exp. Physiol. 107(5), 415–421; doi:10.1113/ep089919
Álvarez Narváez, S., Beaudry, M.S., Norris, C.G., Bartlett, P.B., Glenn, T.C. and Sanchez, S. 2024. Improved equine fecal microbiome characterization using target enrichment by hybridization capture. Animals (Basel). 14(3), 445; doi:10.3390/ani14030445
Amat, S., Holman, D.B., Schmidt, K., McCarthy, K.L., Dorsam, S.T., Ward, A.K., Borowicz, P.P., Reynolds, L.P., Caton, J.S., Sedivec, K.K. and Dahlen, C.R. 2021. Characterization of the Microbiota Associated With 12-Week-Old Bovine Fetuses Exposed to Divergent in utero Nutrition. Front. Microbiol. 12, 771832; doi:10.3389/fmicb.2021.771832
Arshad, M.A., Hassan, F.U., Rehman, M.S., Huws, S.A., Cheng, Y. and Din, A.U. 2021. Gut microbiome colonization and development in neonatal ruminants: strategies, prospects, and opportunities. Anim. Nutr. 7(3), 883–895; doi:10.1016/j.aninu.2021.03.004
Bai, T., Pu, X., Guo, X., Liu, J., Zhao, L., Zhang, X., Zhang, S. and Cheng, L. 2023. Effects of Dietary Nonfibrous Carbohydrate/Neutral Detergent Fiber Ratio on Methanogenic Archaea and Cellulose-Degrading Bacteria in the Rumen of Karakul Sheep: a 16S rRNA Gene Sequencing Study. Appl. Environ. Microbiol. 89(1), 129122; doi:10.1128/aem.01291-22
Beaumont, M., Portune, K.J., Steuer, N., Lan, A., Cerrudo, V., Audebert, M., Dumont, F., Mancano, G., Khodorova, N., Andriamihaja, M., Airinei, G., Tomé, D., Benamouzig, R., Davila, A.M., Claus, S.P., Sanz, Y. and Blachier, F. 2017. Quantity and source of dietary protein influence metabolite production by gut microbiota and rectal mucosa gene expression: a randomized, parallel, double-blind trial in overweight humans. Am. J. Clin. Nutr. 106(4), 1005–1019; doi:10.3945/ajcn.117.158816
Bi, Y., Zeng, S., Zhang, R., Diao, Q. and Tu, Y. 2018. Effects of dietary energy levels on rumen bacterial community composition in Holstein heifers under the same forage to concentrate ratio condition. BMC. Microbiol. 18(1), 69; doi:10.1186/s12866-018-1213-9
Callahan, B.J., Mcmurdie, P.J., Rosen, M.J., Han, A.W., Johnson, A.J.A. and Holmes, S.P. 2016. DADA2: high-resolution sample inference from Illumina amplicon data. Nat. Methods 13(7), 581–583; doi:10.1038/nmeth.3869
Caporaso, J.G., Lauber, C.L., Walters, W.A., Berg-Lyons, D., Lozupone, C.A., Turnbaugh, P.J., Fierer, N. and Knight, R. 2011. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA. 108, 4516–4522; doi:10.1073/pnas.1000080107
Cheng, Y., Zhang, H., Zhang, J., Duan, H., Yin, Y., Li, Y. and Mao, S. 2024. Effects of fermented rice husk powder on growth performance, rumen fermentation, and rumen microbial communities in fattening Hu sheep. Front. Vet. Sci. 11, 1503172; doi:10.3389/fvets.2024.1503172
Cholewińska, P., Czyż, K., Nowakowski, P. and Wyrostek, A. 2020a. The microbiome of the digestive system of ruminants—a review. Anim. Health Res. Rev. 21(1), 3–14; doi: 10.1017/s1466252319000069
Cholewińska, P., Wołoszyńska, M., Michalak, M., Czyż, K., Rant, W. and Janczak, M. 2020b. Evaluation of changes in the levels of Firmicutes and Bacteroidetes Phyla of sheep feces depending on the breed. Animals. (Basel). 10(10), 1901; doi:10.3390/ani10101901
Clarke, S.F., Murphy, E.F., O'Sullivan, O., Lucey, A.J., Humphreys, M., Hogan, A., Hayes, P., O'Reilly, M., Jeffery, I.B., Wood-Martin, R., Kerins, D.M., Quigley, E., Ross, R.P., O'Toole, P.W., Molloy, M.G., Falvey, E., Shanahan, F. and Cotter, P.D. 2014. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 63(12), 1913–1920; doi:10.1136/gutjnl-2013-306541
Devyatkin, V., Mishurov, A. and Kolodina, E. 2021. Probiotic effect of Bacillus subtilis B-2998D, B-3057D, and Bacillus licheniformis B-2999D complex on sheep and lambs. J. Adv. Vet. Anim. Res. 8(1), 146–157; doi:10.5455/javar.2021.h497
Dong, P., Xie, Z., Yin, M. and Chen, X. 2024. Mechanisms of microecological preparations and their application in the healthy farming of cattle and sheep. Mod. J. Anim. Husbandry. Vet. Med. 2, 88–91; doi:10.20154/j.cnki.issn1672-9692.2024.02.019
Du, R., Jin, Y., Wang, B., Luo, Y., Bao, L., Zhao, L. and Su, L. 2020. Effects of probiotics on gastrointestinal microbiota, metabolites, and meat quality in Sunit Sheep. Food. Sci. 41(11), 14–21; doi:10.7506/spkx1002-6630-20190714-181
Du, R., Wang, B., Luo, Y., Wang, Y., Li, Q., Han, L., Zhao, L. and Jin, Y. 2019. Advances in research on probiotic regulation of gastrointestinal microbiota and improvement of meat quality. Microbiol. China. 46(09), 2378–2385.
Fu, X., Yang, Y., Yan, Z., Liu, M. and Wang, X. 2022. Transcriptomic study of Spermatogenesis in the testis of Hu sheep and tibetan sheep. Genes (Basel). 13(12), 2212; doi:10.3390/genes13122212
Guan, F., Liu, S.R., Shi, G.Q. and Yang, L.G. 2007. Polymorphism of FecB gene in nine sheep breeds or strains and its effects on litter size, lamb growth and development. Anim. Reprod. Sci. 99(1-2), 44–52; doi:10.1016/j.anireprosci.2006.04.048
Guo, H., Cui, J., Li, Q., Liang, X., Li, J., Yang, B., Kalds, P., Chen, Y. and Yang, Y. 2024. A multi-omic assessment of the mechanisms of intestinal microbes used to treat diarrhea in early-weaned lambs. mSystems 9(2), 95323; doi:10.1128/msystems.00953-23
Guo, R., Wang, H., Meng, C., Gui, H., Li, Y., Chen, F., Zhang, C., Zhang, H., Ding, Q., Zhang, J., Zhang, J., Qian, Y., Zhong, J. and Cao, S. 2023. Efficient and Specific Generation of MSTN-Edited Hu Sheep Using C-CRISPR. Genes. (Basel). 14(6), 1216; doi:10.3390/genes14061216
Jia, P., Cui, K., Ma, T., Wan, F., Wang, W., Yang, D., Wang, Y., Guo, B., Zhao, L. and Diao, Q. 2018. Influence of dietary supplementation with Bacillus licheniformis and Saccharomyces cerevisiae as alternatives to monensin on growth performance, antioxidant, immunity, ruminal fermentation and microbial diversity of fattening lambs. Sci. Rep. 8(1), 16712; doi:10.1038/s41598-018-35081-4
Jin, Y., Huang, Y., Luo, H., Wang, L., Chen, B., Zhang, Y., Deng, K., Zhao, N. and Lai, A. 2023. Effects of replacing hybrid giant napier with sugarcane bagasse and fermented sugarcane bagasse on growth performance, nutrient digestibility, rumen fermentation characteristics, and rumen microorganisms of Simmental crossbred cattle. Front. Microbiol. 14, 1236955; doi:10.3389/fmicb.2023.1236955
Katoh, T., Yamada, C., Wallace, M.D., Yoshida, A., Gotoh, A., Arai, M., Maeshibu, T., Kashima, T., Hagenbeek, A., Ojima, M.N., Takada, H., Sakanaka, M., Shimizu, H., Nishiyama, K., Ashida, H., Hirose, J., Suarez-Diez, M., Nishiyama, M., Kimura, I., Stubbs, K.A., Fushinobu, S. and Katayama, T. 2023. A bacterial sulfoglycosidase highlights mucin O-glycan breakdown in the gut ecosystem. Nat. Chem. Biol. 19(6), 778–789; doi:10.1038/s41589-023-01272-y
Kong, L., Yue, Y., Li, J., Yang, B., Chen, B., Liu, J. and Lu, Z. 2023. Transcriptomics and metabolomics reveal improved performance of Hu sheep on hybridization with Southdown sheep. Food Res. Int. 173, 113240; doi:10.1016/j.foodres.2023.113240
Krajewska-Włodarczyk, M. 2017. The role of the human microbiom in the pathogenesis of rheumatoid arthritis - a literature review. Wiad. Lek. 70(4), 798–803; doi.
Kulkarni, N.A., Chethan, H.S., Srivastava, R. and Gabbur, A.B. 2022. Role of probiotics in ruminant nutrition as natural modulators of health and productivity of animals in tropical countries: an overview. Trop. Anim. Health. Prod. 54(2), 110; doi:10.1007/s11250-022-03112-y
Kwon, W.Y., Cha, B.S., Kim, S., Hwang, S.H., Kim, J.M., Kalimuthu, K., Park, H.G. and Park, K.S. 2019. Fluorescence polarization-based detection of cancer-related mutations using target-initiated rolling circle amplification. Analyst 144(14), 4149–4152; doi:10.1039/c9an00429g
Laustsen, L., Edwards, J.E., Hermes, G.D.A., Lúthersson, N., Van Doorn, D.A., Okrathok, S., Kujawa, T.J. and Smidt, H. 2021. Free Faecal Water: analysis of Horse Faecal Microbiota and the Impact of Faecal Microbial Transplantation on Symptom Severity. Animals. (Basel). 11(10), 2776; doi:10.3390/ani11102776
Ley, R.E., Bäckhed, F., Turnbaugh, P., Lozupone, C.A., Knight, R.D. and Gordon, J.I. 2005. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA. 102(31), 11070–11075; doi:10.1073/pnas.0504978102
Li, Y., Chen, Z., Fang, Y., Cao, C., Zhang, Z., Pan, Y. and Wang, Q. 2022. Runs of Homozygosity Revealed Reproductive Traits of Hu Sheep. Genes. (Basel). 13(10), 1848; doi:10.3390/genes13101848
Liu, Z., Hu, Y., Pan, S. and Shi, S. 2022. Early colonization of gut microbiota in animals and its effects on host health. Chin. J. Anim. Nutr. 34(05), 2831–2838; doi:10.3969/j.issn.1006
Lu, J., Chen, Z., Chen, P., Li, Z., Wan, Y., Song, Y., Wang, F. and Zhang, Y. 2023. Dietary potential probiotics and enzymes complex modulates the performance and rumen microbiota in weaned goats. J. Appl. Microbiol. 134(2), 79; doi:10.1093/jambio/lxac079
Ma, W., Li, W., Yu, S., Bian, H., Wang, Y., Jin, Y., Zhang, Z., Ma, Q. and Huang, L. 2023. Immunomodulatory effects of complex probiotics on the immuno-suppressed mice induced by cyclophosphamide. Front. Microbiol. 14, 1055197; doi:10.3389/fmicb.2023.1055197
Ma, Y., Yang, X., Hua, G., Deng, X., Xia, T., Li, X., Feng, D. and Deng, X. 2022. Contribution of gut microbiomes and their metabolomes to the performance of Dorper and Tan sheep. Front. Microbiol. 13, 1047744; doi:10.3389/fmicb.2022.1047744
Magoč, T. and Salzberg, S.L. 2011. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21), 2957–2963; doi:10.1093/bioinformatics/btr507
Manor, O., Zubair, N., Conomos, M.P., Xu, X., Rohwer, J.E., Krafft, C.E., Lovejoy, J.C. and Magis, A.T. 2018. A Multi-omic Association Study of Trimethylamine N-Oxide. Cell. Rep. 24(4), 935–946; doi:10.1016/j.celrep.2018.06.096
Martin, M. 2011. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnetjournal 17(1), 10–12; doi:10.14806/ej.17.1.200
Na, S.W. and Guan, L.L. 2022. Understanding the role of rumen epithelial host-microbe interactions in cattle feed efficiency. Anim. Nutr. 10, 41–53; doi:10.1016/j.aninu.2022.04.002
Newbold, C.J. and Ramos-Morales, E. 2020. Review: ruminal microbiome and microbial metabolome: effects of diet and ruminant host. Animal 14(S1), s78–s86; doi:10.1017/s1751731119003252
Nicholson, J.K., Holmes, E., Kinross, J., Burcelin, R., Gibson, G., Jia, W. and Pettersson, S. 2012. Host-gut microbiota metabolic interactions. Science 336(6086), 1262–1267; doi:10.1126/science.1223813
Ouwehand, A.C., Invernici, M.M., Furlaneto, F.A.C. and Messora, M.R. 2018. Effectiveness of Multistrain Versus Single-strain Probiotics: current Status and Recommendations for the Future. J. Clin. Gastroenterol. 52(Suppl 1), S35–S40.
Paz, H.A., Hales, K.E., Wells, J.E., Kuehn, L.A., Freetly, H.C., Berry, E.D., Flythe, M.D., Spangler, M.L. and Fernando, S.C. 2018. Rumen bacterial community structure impacts feed efficiency in beef cattle. J. Anim. Sci. 96(3), 1045–1058.
Quast, C., Pruesse, E., Yilmaz, P., Gerken, J., Schweer, T., Yarza, P., Peplies, J. and Glöckner, F.O. 2013. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic. Acids. Res. 41, D590–D596; doi:10.1093/nar/gks1219
Schloss, P.D., Westcott, S.L., Ryabin, T., Hall, J.R., Hartmann, M., Hollister, E.B., Lesniewski, R.A., Oakley, B.B., Parks, D.H., Robinson, C.J., Sahl, J.W., Stres, B., Thallinger, G.G., Van Horn, D.J. and Weber, C.F. 2009. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 75(23), 7537–7541; doi:10.1128/aem.01541-09
Schofield, B.J., Lachner, N., Le, O.T., Mcneill, D.M., Dart, P., Ouwerkerk, D., Hugenholtz, P. and Klieve, A.V. 2018. Beneficial changes in rumen bacterial community profile in sheep and dairy calves as a result of feeding the probiotic Bacillus amyloliquefaciens H57. J. Appl. Microbiol. 124(3), 855–866; doi:10.1111/jam.13688
Schwiertz, A., Taras, D., Schäfer, K., Beijer, S., Bos, N.A., Donus, C. and Hardt, P.D. 2010. Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver. Spring). 18(1), 190–195; doi:10.1038/oby.2009.167
Shan, C., Zhai, H., Li, J., Wang, Y., Yu, H.M., Wu, Y.Q., Liu, E. and Liu, Y. 2022. Effects of clostridium butyricum on short-chain fatty acid content and microbial diversity in broiler chickens. China Anim. Husbandry Vet. Med. 49(11), 4239–4251; doi:10.16431/j.cnki.1671-7236.2022.11.014
Shen, H., Lu, Z., Xu, Z., Chen, Z. and Shen, Z. 2017. Associations among dietary non-fiber carbohydrate, ruminal microbiota and epithelium G-protein-coupled receptor, and histone deacetylase regulations in goats. Microbiome 5(1), 123; doi:10.1186/s40168-017-0341-z
Spence, C., Wells, W.G. and Smith, C.J. 2006. Characterization of the primary starch utilization operon in the obligate anaerobe Bacteroides fragilis: regulation by carbon source and oxygen. J. Bacteriol. 188(13), 4663–4672; doi:10.1128/jb.00125-06
Sugiharto, S., Isroli, I., Yudiarti, T. and Widiastuti, E. 2018. The effect of supplementation of multistrain probiotic preparation in combination with vitamins and minerals to the basal diet on the growth performance, carcass traits, and physiological response of broilers. Vet. World. 11(2), 240–247.
Szeligowska, N., Cholewińska, P., Czyż, K., Wojnarowski, K. and Janczak, M. 2021. Inter and intraspecies comparison of the level of selected bacterial phyla in cattle and sheep based on feces. BMC. Vet. Res. 17(1), 224; doi:10.1186/s12917-021-02922-w
Tamburini, S., Shen, N., Wu, H.C. and Clemente, J.C. 2016. The microbiome in early life: implications for health outcomes. Nat. Med. 22(7), 713–722; doi:10.1038/nm.4142
Tavella, T., Rampelli, S., Guidarelli, G., Bazzocchi, A., Gasperini, C., Pujos-Guillot, E., Comte, B., Barone, M., Biagi, E., Candela, M., Nicoletti, C., Kadi, F., Battista, G., Salvioli, S., O’Toole, P.W., Franceschi, C., Brigidi, P., Turroni, S. and Santoro, A. 2021. Elevated gut microbiome abundance of Christensenellaceae, Porphyromonadaceae and Rikenellaceae is associated with reduced visceral adipose tissue and healthier metabolic profile in Italian elderly. Gut Microbes 13(1), 1–19; doi:10.1080/19490976.2021.1880221
Taylor, S.L., Simpson, J.L. and Rogers, G.B. 2021. The influence of early-life microbial exposures on long-term respiratory health. Paediatr. Respir. Rev. 40, 15–23; doi:10.1016/j.prrv.2021.05.003
Turnbaugh, P.J., Ley, R.E., Mahowald, M.A., Magrini, V., Mardis, E.R. and Gordon, J.I. 2006. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444(7122), 1027–1031; doi:10.1038/nature05414
Wang, H., Liu, Y. and Wang, Z. 2024a. Effects of complex probiotics on growth performance, serum biochemical indicators, and slaughter performance in Hu lambs. Feed Res. 47(10), 1–6; doi:10.13557/j.cnki.issn1002-2813.2024.10.001
Wang, H., Yu, Z., Gao, Z., Li, Q., Qiu, X., Wu, F., Guan, T., Cao, B. and Su, H. 2022a. Effects of compound probiotics on growth performance, rumen fermentation, blood parameters, and health status of neonatal Holstein calves. J. Dairy Sci. 105(3), 2190–2200; doi:10.3168/jds.2021-20721
Wang, J., Fan, H., Han, Y., Zhao, J. and Zhou, Z. 2017. Characterization of the microbial communities along the gastrointestinal tract of sheep by 454 pyrosequencing analysis. Asian-Australas. J. Anim. Sci. 30(1), 100–110; doi:10.5713/ajas.16.0166
Wang, L., Lv, Z., Ning, X., Yue, Z., Wang, P., Liu, C., Jin, S., Li, X., Yin, Q., Zhu, Q. and Chang, J. 2024b. The effects of compound probiotics on production performance, rumen fermentation and microbiota of Hu sheep. Front. Vet. Sci. 11, 1440432; doi:10.3389/fvets.2024.1440432
Wang, L., Qi, W., Mao, S., Zhu, W. and Liu, J. 2022b. Effects of whole corn high-grain diet feeding on ruminal bacterial community and epithelial gene expression related to VFA absorption and metabolism in fattening lambs. J. Anim. Sci. 100(3), 56; doi:10.1093/jas/skac056
Wang, X., Hu, L., Liu, H., Xu, T., Zhao, N., Zhang, X., Geng, Y., Kang, S. and Xu, S. 2021a. Characterization of the bacterial microbiota across the different intestinal segments of the Qinghai semi-fine wool sheep on the Qinghai-Tibetan Plateau. Anim. Biosci. 34(12), 1921–1929; doi:10.5713/ab.20.0809
Wang, X., Zhang, Z., Wang, X., Bao, Q., Wang, R. and Duan, Z. 2021b. The impact of host genotype, intestinal sites and probiotics supplementation on the gut microbiota composition and diversity in sheep. Biol. (Basel). 10(8), 769; doi:10.3390/biology10080769
Wang, Y., Li, A., Jiang, X., Zhang, H., Mehmood, K., Zhang, L., Jiang, J., Waqas, M., Iqbal, M. and Li, J. 2018. Probiotic Potential of Leuconostoc pseudomesenteroides and Lactobacillus Strains Isolated From Yaks. Front. Microbiol. 9, 2987; doi:10.3389/fmicb.2018.02987
Yang, T., Santisteban, M.M., Rodriguez, V., Li, E., Ahmari, N., Carvajal, J.M., Zadeh, M., Gong, M., Qi, Y., Zubcevic, J., Sahay, B., Pepine, C.J., Raizada, M.K. and Mohamadzadeh, M. 2015. Gut dysbiosis is linked to hypertension. Hypertension 65(6), 1331–1340; doi:10.1161/hypertensionaha.115.05315
Yao, Y., Hu, Q., Yu, K., Hu, X., Yu, R., Wang, S. and Guan, F. 2022. Effects of complex microecological preparations on growth performance and blood protein indicators in Hu lambs. Chin. J. Anim. Sci. 58(11), 265–270; doi:10.19556/j.0258-7033.20211026-01
Yao, Y., Sun, R., Hu, Q., Yu, K., Jiang, J., Jiang, Y. and Guan, F. 2023. Advances in research on microecological preparations and their applications in sheep farming. China Feed. 05, 16–23. doi: 10.15906/j.cnki.cn11-2975/s.2022040023-02.
Zamojska, D., Nowak, A., Nowak, I. and Macierzyńska-Piotrowska, E. 2021. Probiotics and postbiotics as substitutes of antibiotics in farm animals: a review. Animals. (Basel). 11(12), 3431; doi:10.3390/ani11123431
Zhang, C., Lu, W., Liu, H., Shen, L., Zhu, M., Zhou, T., Zhang, L., Xiao, D. and Chen, L. 2024a. Rumen microbiota transplantation alleviates gossypol diet-induced reproductive, liver, and intestinal damage in male mice. Animals. (Basel). 14(15), 2206; doi:10.3390/ani14152206
Zhang, H., Lang, X., Zhang, Y. and Wang, C. 2022. Distribution of bacteria in different regions of the small intestine with Zanthoxylum bungeanum essential oil supplement in small-tailed Han sheep. Front. Microbiol. 13, 1062077; doi:10.3389/fmicb.2022.1062077
Zhang, J., Li, X., Sha, Y., Wang, Z., Qi, S., Zhang, X., Zhao, S. and Jiao, T. 2024b. Effects of Steviol Glycosides on growth performance, ruminal fermentation and microbial diversity of Hu sheep. Animals. (Basel). 14(13), 1991; doi:10.3390/ani14131991
Zhang, J., Shi, H., Wang, Y., Li, S., Cao, Z., Ji, S., He, Y. and Zhang, H. 2017a. Effect of dietary forage to concentrate ratios on dynamic profile changes and interactions of ruminal microbiota and metabolites in Holstein Heifers. Front. Microbiol. 8, 2206; doi:10.3389/fmicb.2017.02206
Zhang, X., Wang, W., Mo, F., La, Y., Li, C. and Li, F. 2017b. Association of residual feed intake with growth and slaughtering performance, blood metabolism, and body composition in growing lambs. Sci. Rep. 7(1), 12681; doi:10.1038/s41598-017-13042-7
Zhao, H., Jiang, F., Gu, H., Gao, H., Zhang, J., Zhang, M. and Zhang, T. 2023. Seasonal variation of gut microbial composition and metabolism in Tibetan Antelopes in Hoh Xil National Nature Reserve. Animals. (Basel). 13(22), 13; doi:10.3390/ani13223569
Zhao, J., Zhao, X., Gao, J., Bai, B., Niu, J., Yang, Y., Zhao, G., Wang, Z., Xu, Z., Wang, J., Cheng, Y. and Hao, L. 2024. Ensiled diet improved the growth performance of Tibetan sheep by regulating the rumen microbial community and rumen epithelial morphology. J. Anim. Sci. 102, skae173; doi: 10.1093/jas/skae173
Zhao, P., Wang, H., Bian, X., Wen, X., Zhang, X. and Hua, S. 2024. Comparative experiment of different compulsory supplemental feeding methods for Hu lambs. J. Anim. Sci. Vet. Med. 43(02), 17–20; doi:10.3969/j.issn.1004-6704.2023.05.027
Zhao, Y., Li, B., Bai, D., Huang, J., Shiraigo, W., Yang, L., Zhao, Q., Ren, X., Wu, J., Bao, W. and Dugarjaviin, M. 2016. Comparison of Fecal Microbiota of Mongolian and Thoroughbred Horses by High-throughput Sequencing of the V4 Region of the 16S rRNA Gene. Asian-Australas. J. Anim. Sci. 29(9), 1345–1352; doi:10.5713/ajas.15.0587
Zhou, X., Qiao, K., Wu, H. and Zhang, Y. 2023. The impact of food additives on the abundance and composition of gut microbiota. Molecules 28(2), 631; doi:10.3390/molecules28020631
Supplementary Materials
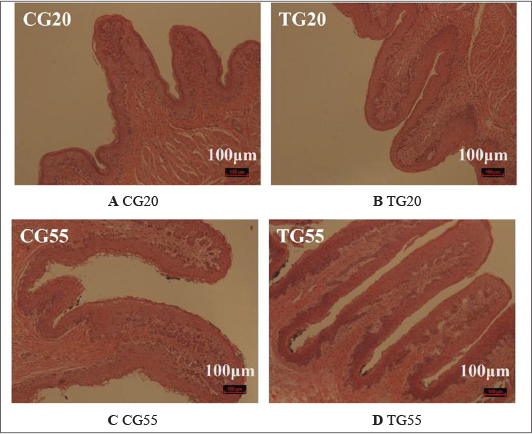
Fig. S1. Effect of probiotic formulations on rumen morphology (10×) Images correspond to (A) CG20: control group at 20 days of age, without probiotic treatment. (B) TG20: probiotic-treated group at 20 days of age. (C) CG55: Control group at 55 days of age. (D) TG55: Probiotic-treated group at 55 days of age.
Fig. S2. Rarefaction curves of rumen, duodenum, jejunum and ileum in Hu lambs at 0, 20 and 55 days of age. (A) Observed_otus rarefaction curves. (B) Shannon index rarefaction curves.
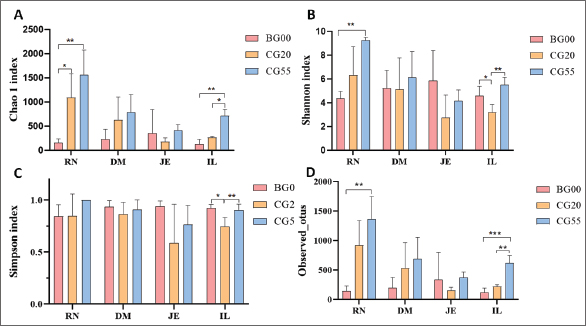
Fig. S3. Dynamic changes in α diversity metrics of the gut microbiota of Hu lambs at 0, 20 and 55 days of age. (A) Chao1 index; (B) Shannon index; (C) Simpson index; (D) Observed_otus. All values are reported as mean ± SD, in which * indicates statistical significance (p < 0.05).
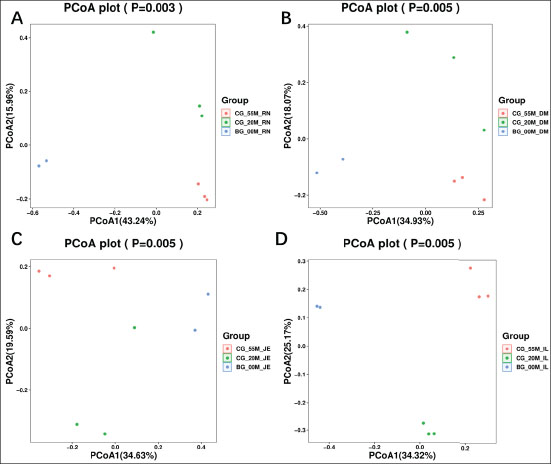
Fig. S4. PCoA of the gut microbiota of Hu lambs at 0, 20 and 55 days of age. (A) Rumen. (B) Duodenum. (C) Jejunum. (D) Ileum.
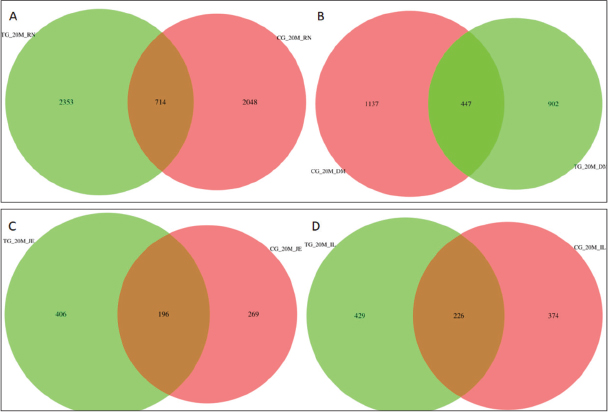
Fig. S5. Venn diagrams of the composition of ASVs in gut microbiota of Hu lambs in the 20-day-old treatment and control groups. (A) Rumen. (B) Duodenum. (C) Jejunum. (D) Ileum.
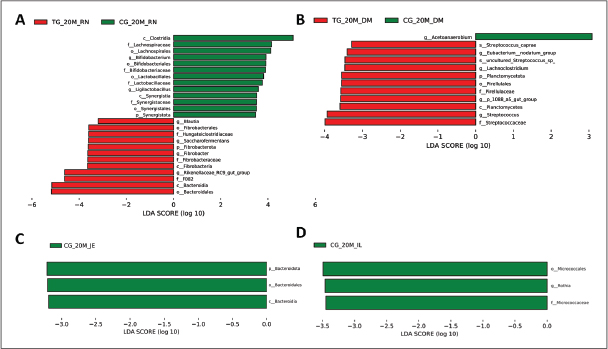
Fig. S6. Distribution of linear discriminant analysis (LDA) scores for Hu sheep at 20 days of age. The ordinate indicates the taxon with significant differences between groups; the abscissa corresponds to the logarithmic LDA scores based on taxonomic analysis. Differential microbial species in the GIT microbiota of Hu sheep between two groups. (A) Rumen. (B) Duodenum. (C) Jejunum. (D) Ileum.
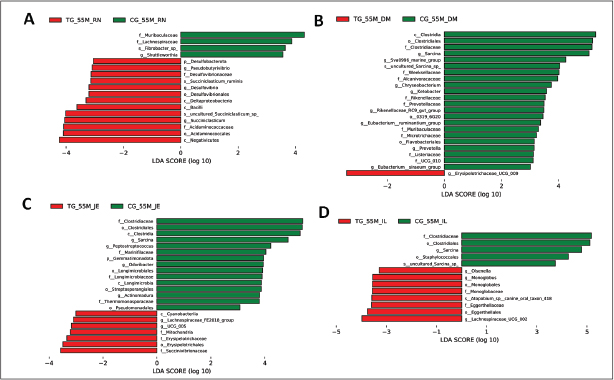
Fig. S7. Distribution of linear discriminant analysis (LDA) scores for Hu sheep at 55 days of age. The ordinate indicates the taxon with significant differences between groups; the abscissa corresponds to the logarithmic LDA scores based on taxonomic analysis. Differential microbial species in the GIT microbiota of Hu sheep between two groups. (A) Rumen. (B) Duodenum. (C) Jejunum. (D) Ileum.

























