| Review Article | ||
Open Vet. J.. 2026; 16(3): 1402-1425 Open Veterinary Journal, (2026), Vol. 16(3): 1402-1425 Review Article Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicineThamer J. Shihab1* and Eman Hashim Yousif21Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq 2Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Baghdad, Baghdad, Iraq *Corresponding Author: Thamer J. Shihab. Department of Pathology and Poultry Diseases, College of Veterinary Medicine, University of Tikrit, Tikrit, Iraq. Email: dr.thamer.vet [at] tu.edu.iq Submitted: 24/10/2025 Revised: 05/02/2026 Accepted: 17/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractRecombinant Listeriolysin O (rLLO), a cholesterol-dependent cytolysin (CDC) derived from Listeria monocytogenes, has gained increasing attention as a novel immunological adjuvant for next-generation vaccine development. rLLO enhances both innate and adaptive immune responses by facilitating controlled endosomal membrane permeabilization, promoting cytosolic antigen delivery, and enabling efficient antigen cross-presentation, leading to robust CD8⁺ T-cell activation. These immunological properties are particularly advantageous for vaccines targeting intracellular pathogens and malignancies. This narrative review synthesizes current experimental, preclinical, and translational evidence on the application of rLLO across diverse vaccine platforms in human and veterinary medicine, including DNA-, protein-, nanoparticle-, and vector-based formulations. Studies addressing infectious diseases, such as tuberculosis, HIV, dengue, and listeriosis, as well as cancer immunotherapy models, are critically examined. Emerging strategies involving fusion proteins, detoxified rLLO mutants, and recombinant delivery systems are highlighted to illustrate advances in formulation design and immunogenic optimization. Despite promising preclinical outcomes, several challenges remain, including the need for comprehensive safety evaluation, dosing and delivery strategy optimization, and regulatory pathway clarification. Addressing these limitations through standardized toxicological assessment and large-scale clinical and veterinary trials is essential for the successful translation of rLLO-based vaccine components. Overall, rLLO represents a versatile and potent immunological tool with significant potential to advance vaccine design in human and veterinary applications. Keywords: Listeria monocytogenes, Vaccine development, Immune response, CD8+ T cells. IntroductionThe current global burden of infectious illnesses and the complications associated with cancer treatment make it even more important to improve vaccination performance (Yusuf et al., 2023). When it comes to complicated illnesses such as cancer and intracellular infections, traditional vaccination techniques struggle to produce robust and long-lasting immune responses from time to time. This has prompted extensive investigations into the development of novel vaccination platforms and efficient adjuvants for immune modulation. Listeria monocytogenes is a potent inducer of host cell-mediated immunity, particularly strong CD8⁺ T-cell responses, owing to its intracellular lifestyle and efficient antigen presentation (Pham et al., 2025). Listeriolysin O (LLO) is the most important Listeria monocytogenes (LM) virulence protein (Obaidi et al., 2010; Ibrahim et al., 2017). LLO is a thiol-activated cholesterol-dependent pore-forming toxin and the major LM virulence factor (Hernández-Flores and Vivanco-Cid, 2015). This pore-forming toxin exhibits optimum activity at acidic pH levels, associating with the phagosome milieu, and is closely regulated to prevent host cell damage upon cytosolic release (Banerji et al., 2021). Although L. monocytogenes induces strong antigen-specific T-cell immune responses, its intrinsic virulence and cytotoxicity limit its direct application as a live bacterial vaccine vector. Consequently, safer alternatives are required to harness its potent immunomodulatory properties (Hamon et al., 2012; Sun and Liu, 2013). Recombinant LLO (rLLO), recombinantly expressed from L. monocytogenes, preserves essential immunostimulatory functions, including controlled pore-forming activity, while removing the broader bacterial context responsible for pathogenicity (Liang et al., 2022). Recent studies have highlighted the significance of LLO in modulating host immune responses and influencing antigen presentation pathways (Hamon et al., 2007; Hamon and Cossart, 2011; Khitam et al., 2018; Eldridge and Hamon, 2021; Cong et al., 2024), including apoptosis and pyroptosis, and by altering host gene expression through histone modifications (Hamon et al., 2007; Hamon and Cossart, 2011; Eldridge and Hamon, 2021). Furthermore, the chaperone protein PrsA2 has been recognized as vital for the proper secretion, stability, and folding of LLO during infection, underscoring the complexity of its regulation (Agbavor et al., 2024). The field of vaccine improvement is rapidly evolving, mostly with the use of recombinant proteins and innovative adjuvants (Asokan et al., 2025; Frosth et al., 2025). Improved understanding of intracellular antigen delivery and immune activation pathways has enabled the development of more effective and safer vaccine adjuvants, including pore-forming proteins such as rLLO (Rea et al., 2001). Current vaccine strategies increasingly employ live recombinant bacteria or viral vectors that mimic natural infections and exploit their intrinsic adjuvant properties to enhance immune activation (Gupta and Pellett, 2023). Future research should focus on boosting existing adjuvants and developing new ones that can effectively stimulate a robust immune response without undesirable side effects (Ibrahim et al., 2012; Nascimento and Leite, 2012). Occurrences in mRNA vaccine formulation represent a promising avenue for future vaccine applications (Lukeman et al., 2025). The adaptability of mRNA technology facilitates rapid updates to vaccines in response to evolving infectious diseases, as demonstrated by the current success of mRNA vaccines against COVID-19 (Wang et al., 2023). These adjuvants could promote the stimulation and proliferation of cytotoxic T lymphocytes (CTLs), thereby advancing the overall efficacy of immunotherapeutic methods (Zhao et al., 2023). Moreover, recent approaches have demonstrated that incorporating double-stranded RNA or protein antigens into lipid nanoparticles can function as an effective adjuvant strategy to enhance immune responses (Nguyen et al., 2012; Wang et al., 2023; Lai et al., 2025). Furthermore, clinical trials and studies will provide valuable insights that can guide future vaccine formulations and applications, potentially revolutionizing the landscape of preventive disease and cancer treatment (Wang et al., 2023; Lukeman et al., 2025). Recombinant LLO (rLLO) lacks the bacterial context but retains its pore-forming and immune‐stimulatory properties (Yin et al., 2010). In the last decade, interest in using rLLO as a vaccine component has grown. rLLO could act as a natural adjuvant, stimulating CD4+ and CD8+ responses (Peng et al., 2007). The use of recombinant LLO in vaccine development presents significant opportunities for improving immune responses against various pathogens. Extra recent work has intensive on manufacturing rLLO (mutated or fused forms) that can be coupled as a vaccine adjuvant or carrier, supplying a mechanism for the efficient delivery of antigens and boosting both humoral and cellular immune responses (Hernández-Flores and Vivanco-Cid, 2015). Despite the wide-ranging investigational research on Listeriolysin-O, the immunological mechanisms, safety profile, and translational importance of rLLO as a vaccine adjuvant remain fragmented across diverse experimental platforms (Xia et al., 2025). Accordingly, this narrative review aims to critically synthesize and integrate current experimental, preclinical, and translational evidence regarding the immunological mechanisms, safety profile, and vaccine applications of rLLO. This review summarizes mechanistic insights related to innate and adaptive immune activation, delivery system strategies, and reported immunological outcomes across diverse vaccine platforms. This review compares findings from human and veterinary disease models and highlights underrepresented veterinary data to delineate the emerging role of rLLO as a versatile immunological adjuvant for next-generation vaccine development. Materials and MethodsStudy designThis narrative review synthesizes experimental, preclinical, and translational evidence on the application of rLLO as a vaccine component or immunological adjuvant in human and veterinary medicine. A comprehensive literature search was conducted across multiple scientific databases, and relevant studies were qualitatively analyzed and integrated to provide a thematic narrative synthesis. No original experimental data were generated. Literature search strategy and data sourcesA comprehensive literature search was conducted in PubMed, Scopus, Web of Science, and Google Scholar to identify peer-reviewed studies relevant to rLLO-based vaccine research. Controlled vocabulary and free-text keywords were combined, including “Listeriolysin O”, “recombinant Listeriolysin O”, “rLLO”, “Listeria-based vaccine”, and “vaccine adjuvant.” These terms were supplemented with disease-specific keywords (HIV, tuberculosis (TB), dengue, and listeriosis) to capture applications in both human and veterinary contexts. Recent publications were prioritized, and seminal studies were included where appropriate. Inclusion and exclusion criteriaStudies that satisfied the following obligations were encompassed:
Studies were ignored if they:
Review articles were used selectively for contextual background and conceptual frameworks but were not relied upon as primary sources for experimental outcomes unless they presented novel insights directly relevant to the review objectives. Study selection and data extractionThe headlines and abstracts of all retrieved records were initially screened for relevance. Worthy studies then underwent full-text assessment. After the selected articles were published, key information was extracted and systematically organized, including:
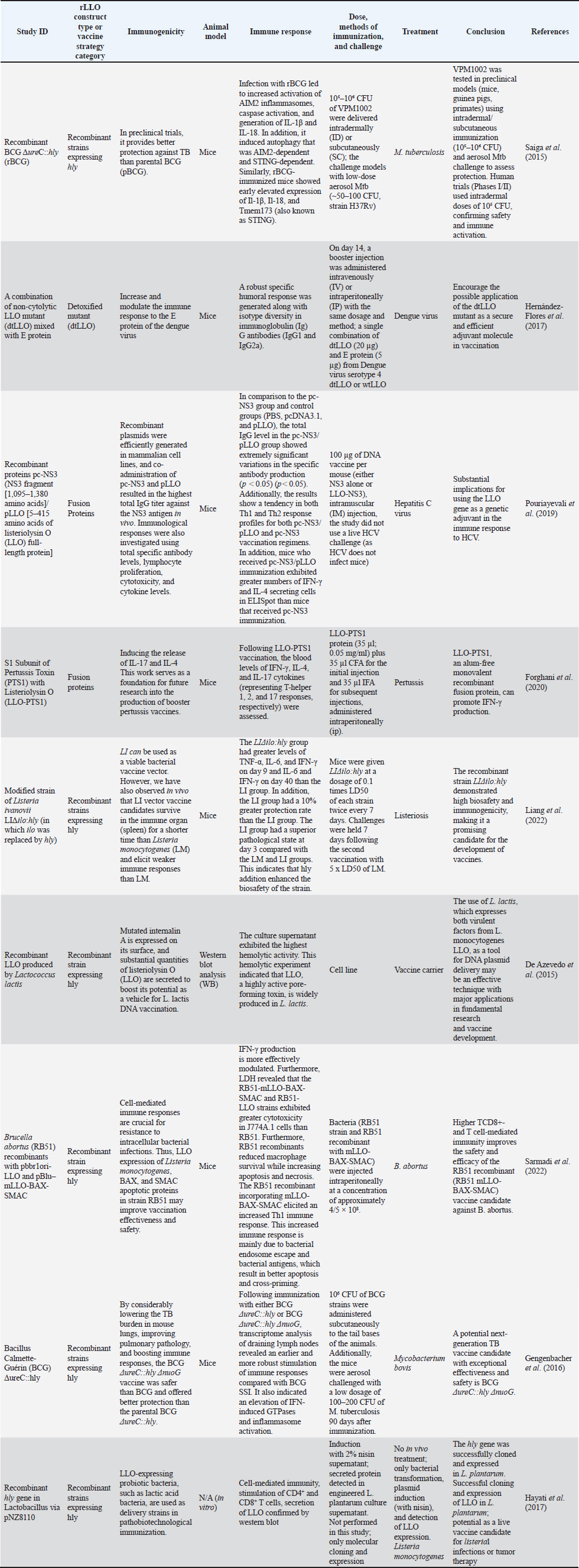
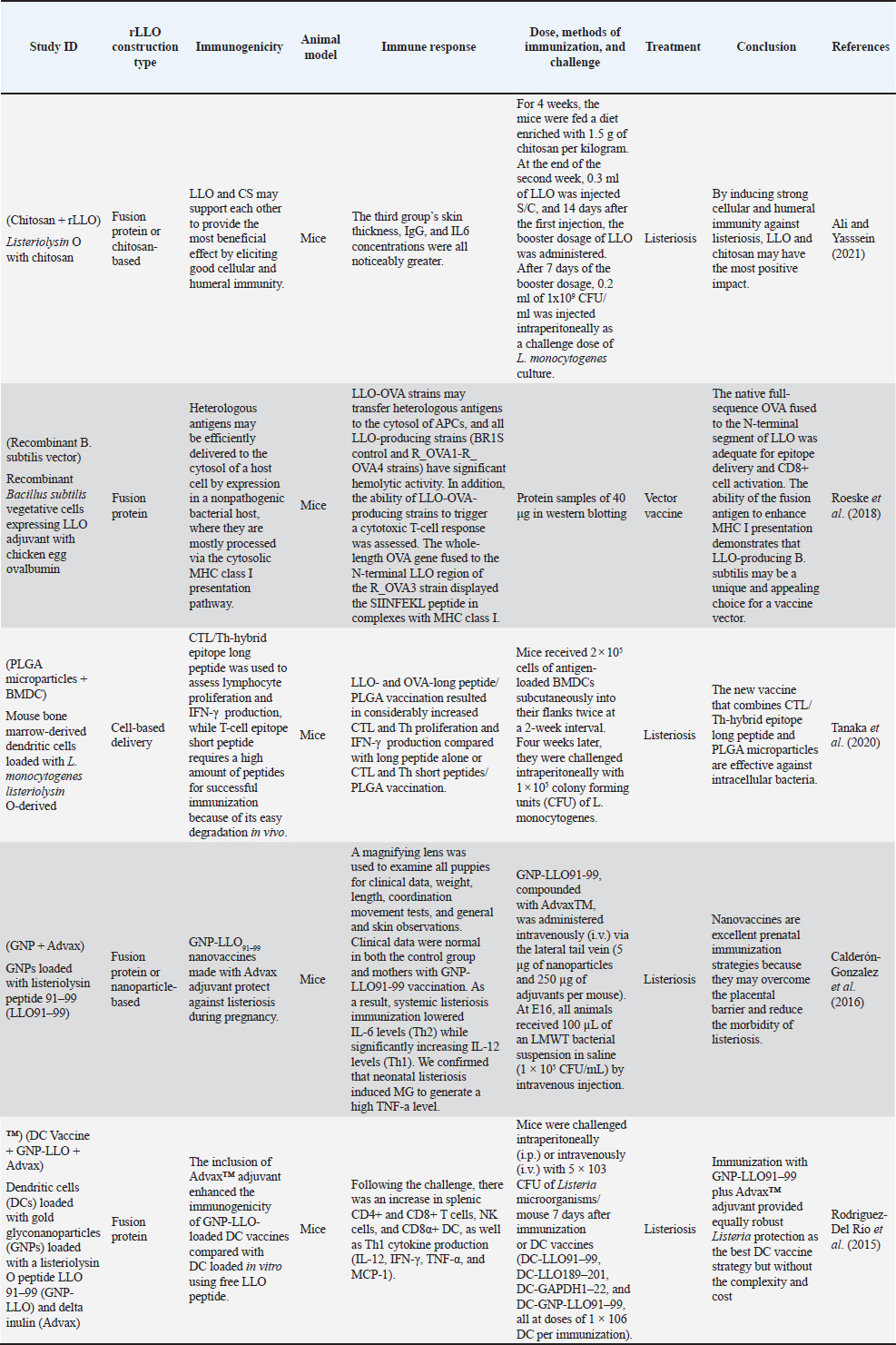
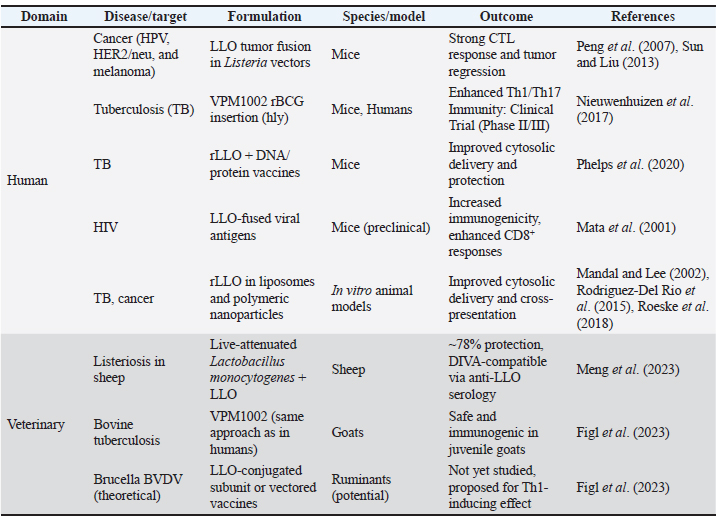
These data formed the basis for the narrative synthesis and the comparative summary (Tables 4 and 5), which highlight representative applications of rLLO across different vaccine platforms. Table 4. rLLO as an immunological adjuvant in experimental vaccines targeting bacterial and viral antigens in human and veterinary models.
Table 5. Delivery systems and formulation strategies using rLLO in experimental vaccines against listeriosis and other pathogens.
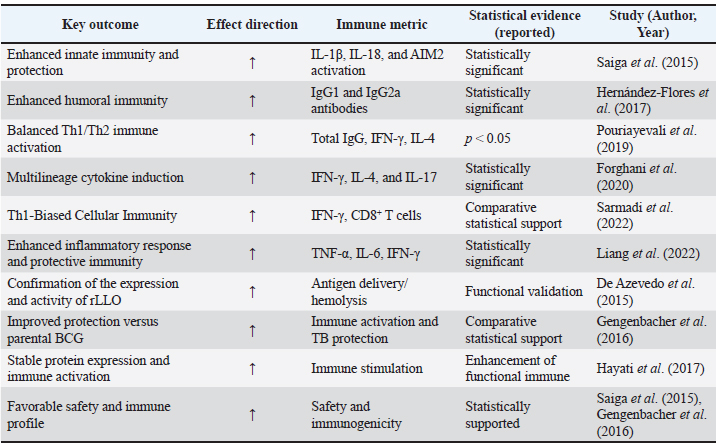
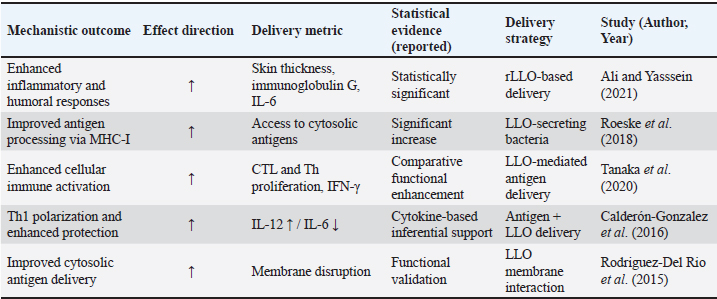
Data synthesisAs a result of the substantial heterogeneity detected across the included studies, mainly in terms of rLLO construct types, vaccine platforms, animal models, immunization routes, dosing regimens, and immunological endpoints (Tables 4 and 5), a qualitative narrative synthesis approach was accepted. Data were extracted, categorized, and thematically integrated to highlight mechanistic insights, translational relevance, and initial trends in rLLO-based vaccine development. Additionally, the statistically reported results and inferential immunological data derived from these research papers were systematically briefed in Tables 6 and 7 to support claims related to next-generation immunization, rather than performing a quantitative meta-analysis. Table 6. Immunological and protective outcomes of rLLO-based experimental vaccine studies.
Table 7. Functional and mechanistic evidence supporting rLLO-mediated antigen delivery systems.
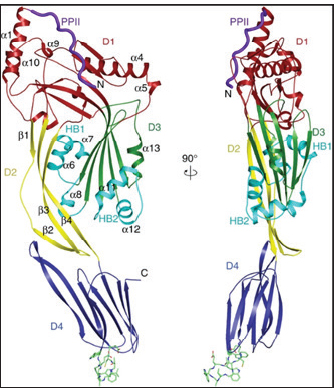
Structure of LLOLLO belongs to the CDC family of pore-forming toxins that disrupt host cell membranes and facilitate the intracellular survival of L. monocytogenes (Fig. 1) (Dubail et al., 2001; Hernández-Flores and Vivanco-Cid, 2015; Banerji et al., 2021). The LLO molecule is organized into four distinct structural domains (D1–D4) (Köster et al., 2014). Domain 1 (D1) contains a five-stranded β-sheet and is surrounded by six α-helices) PEST-like sequence (amino acids 39–51), enriched in proline, glutamate, serine, and threonine residues, which is dispensable for hemolytic activity but essential for phagosomal escape, intracellular stability, and virulence regulation (Hamon et al., 2012; Hotze and Tweten, 2012; Hernández-Flores and Vivanco-Cid, 2015). Domain 2 (D2) functions as a flexible hinge connecting D1 and D4, which consists of four β-strands (Köster et al., 2014). It is connected to D4 through a glycine linker and contains the immunodominant CD8⁺ T-cell epitope LLO₉₁–₉₉, highlighting its importance in adaptive immune recognition (Dubail et al., 2001; Wallecha et al., 2013). Domain 3 (D3) is formed by a five-stranded antiparallel β-sheet, which is surrounded by six α-helices (Köster et al., 2014). It acts as a pH sensor, with key acidic residues (D208, E247, and D320) regulating LLO activation under acidic phagosomal conditions (Schuerch et al., 2005), thereby ensuring controlled pore formation and minimizing host cytotoxicity (Köster et al., 2014; Nguyen et al., 2019). Domain 4 (D4) has eight β-sheets, which are organized forming a β-sandwich structure that mediates binding to cholesterol-rich host membranes through a conserved undecapeptide motif, representing a critical step in oligomerization, pore formation, and cytolytic activity (Köster et al., 2014; Banerji et al., 2021). Collectively, the coordinated functions of these domains link the LLO structure to membrane disruption, immune modulation, and intracellular pathogenic mechanisms (Ilangumaran Ponmalar et al., 2021).
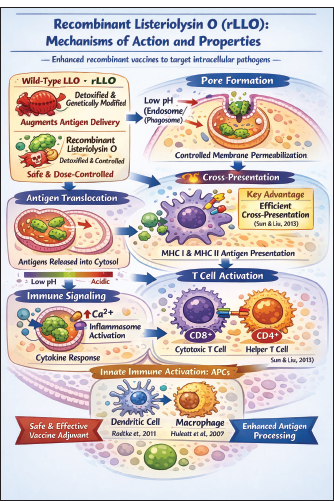
Fig. 1. Crystal structure of LLO (domains D1–D4). The undecapeptide in D4 is inserted into membranes. The key functional motifs (e.g., cholesterol-binding loop) are highlighted in red (Köster et al., 2014). LLO is a potent immunogen capable of inducing robust CD4⁺ and CD8⁺ T-cell responses (Singh et al., 2005; Sun and Liu, 2013; Wallecha et al., 2013; Phelps et al., 2020). Several immunodominant epitopes, including LLO₉₁–₉₉, LLO₁₈₉–₂₀₁, and LLO₂₁₅–₂₂₆, have been shown to elicit strong antigen-specific T-cell responses in experimental models (Singh et al., 2005; Sun and Liu, 2013; Hernández-Flores and Vivanco-Cid, 2015). Notably, the immunogenicity of LLO is independent of its cytotoxicity; dtLLO, which lacks pore-forming ability, still effectively stimulates immune responses (Phelps et al., 2020). This property has been harnessed in vaccine development, where dtLLO serves as an adjuvant, enhancing antigen presentation and promoting dendritic cell maturation, thereby augmenting both innate and adaptive immunity (Wallecha et al., 2013). The structural domains of LLO are intricately associated with its role in pathogenesis and its capability to modulate the host immune response, making it a focal point in the study of bacterial virulence and vaccine design. rLLO: mechanism of action and propertiesRecombinant protein vaccines represent an important enhancement in existing vaccinology because they are designed to provoke targeted immune responses against defined antigens, particularly those related to intracellular pathogens (Huleatt et al., 2007). These vaccines depend on extremely purified recombinant proteins or pathogen-derived subunits, permitting particular immune targeting while maintaining promising safety profiles. Within this context, rLLO has attracted significant attention as a vaccine component and immunological adjuvant due to its strong immunogenic potential and capacity to augment antigen delivery and processing (Li et al., 2021). LLO is a non-enzymatic, thiol-activated, CDC produced by L. monocytogenes. Its primary biological function is to facilitate bacterial escape from the phagosome through pore formation in host cell membranes, a process essential for intracellular survival and pathogenicity (Beauregard et al., 1997; Hamon et al., 2012; Li et al., 2022). In contrast, rLLO used in vaccine platforms is typically genetically modified, detoxified, or carefully dose-controlled to reduce cytotoxicity while preserving immunologically relevant functions. Experimental studies have demonstrated that rLLO retains the ability to transiently permeabilize endosomal or phagosomal membranes without causing extensive host cell damage, supporting its suitability for safe vaccine and adjuvant applications (Köster et al., 2014; Phelps et al., 2020). Mechanistically, rLLO exerts its activity through pH-dependent and transient pore formation within endosomal and phagosomal membranes of APCs, including dendritic cells and macrophages (Radtke et al., 2011). This controlled membrane permeabilization enables the translocation of co-delivered antigens from endosomal compartments into the cytosol, thereby facilitating access to cytosolic processing pathways and enhancing major histocompatibility complex (MHC) class I cross-presentation (Portnoy et al., 1988; Hiltbold et al., 1996). Consequently, rLLO promotes the efficient activation of CD8⁺ T lymphocytes, which is critical for protective immunity against intracellular pathogens (Hiltbold et al., 1996). In addition to its membrane-permeabilizing activity, LLO and rLLO can activate innate immune signaling pathways in APCs. LLO has been shown to stimulate macrophages and dendritic cells via Toll-like receptor (TLR) 4 (TLR4), inducing lipid raft aggregation, NF-κB activation, and pro-inflammatory cytokine production (Kayal et al., 1999; Nguyen et al., 2019). Furthermore, transient pore formation by rLLO induces controlled increases in intracellular Ca²⁺ levels and activates inflammasome-associated signaling without causing irreversible membrane damage or widespread cytolysis, further supporting its favorable safety profile (Tsuchiya et al., 2005; Nguyen et al., 2019). Collectively, these mechanisms underpin the capacity of rLLO to function as an effective facilitator of intracellular antigen delivery, cross-presentation, and immunostimulation (Fig. 2).
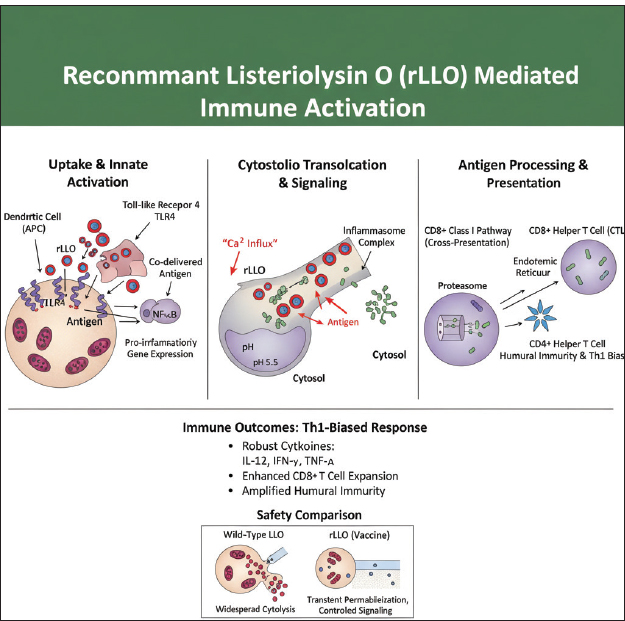
Fig. 2. AI-generated schematic illustration depicting the immunological mechanisms mediated by rLLO. rLLO mediates immune activationBased on the aforementioned molecular mechanisms, rLLO-containing vaccine formulations elicit a coordinated activation of innate and adaptive immune responses (Wira et al., 2005). Antigen delivery into the cytosol of antigen-presenting cells promotes dendritic cell maturation and enhances their capacity to prime T lymphocytes. rLLO has been shown to engage TLR signaling pathways, particularly TLR4, leading to the downstream activation of transcription factors, such as NF-κB and the initiation of proinflammatory gene expression (Stierschneider and Wiesner, 2023). As a consequence of these signaling events, rLLO-based vaccines induce robust cytokine responses characterized by increased production of interleukin-12 (IL-12), interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α). This cytokine milieu favors a Th1-biased immune response, which is particularly desirable for vaccines targeting intracellular pathogens and malignancies (Rodriguez-Del Rio et al., 2015). Furthermore, enhanced cross-presentation resulting from rLLO-mediated antigen delivery leads to the efficient activation and expansion of CD8⁺ cytotoxic T cells, thereby strengthening cell-mediated immunity. In addition to shaping cellular immune responses, rLLO exhibits adjuvant-like properties that amplify humoral immunity when administered in combination with vaccine antigens. By improving antigen availability and immune cell activation, rLLO enhances both the magnitude and breadth of the immune response. Antigen delivery strategies involving LLO fusion proteins have been shown to unmask subdominant epitopes, thereby broadening the repertoire of antigen-specific T-cell responses (Singh et al., 2005). Picard et al. (2015) reported efficient MHC class I antigen presentation in antigen libraries incorporating cLLO. However, the specific contribution of cLLO to MHC-I presentation cannot be definitively determined because no comparison was performed with libraries lacking cLLO. Similarly, rLLO has been successfully employed to direct DNA- and protein-based vaccine antigens into the cross-presentation pathway, effectively functioning as an endosomolytic carrier to improve cytosolic antigen delivery (Carrero et al., 2009) (Fig. 3).
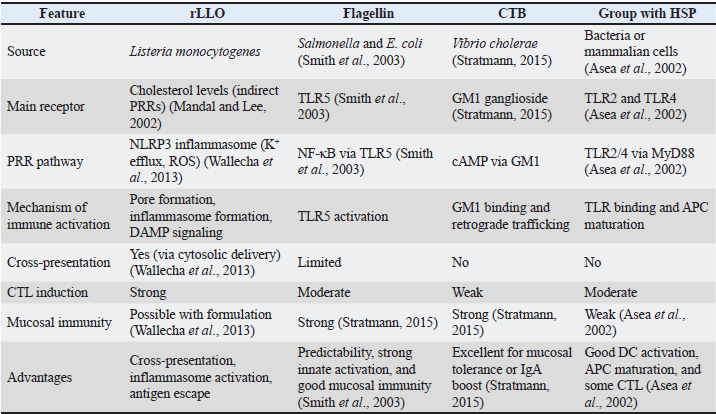
Fig. 3. Enhanced cross-presentation and cytokine signaling induced by rLLO-antigen fusion proteins Key mechanistic features of rLLOMembrane perforation and cytosolic antigen deliveryAt acidic pH, rLLO is quickly incorporated into the host cell membrane to form pores. This crucial mechanism results in the disruption and lysis of phagosomes and endosomes, traditionally responsible for the internalization of pathogens or antigens. The immediate effect of membrane perforation is the efficient delivery of the antigen to the cell cytosol. The process significantly increases vaccine efficacy by enabling the processing and presentation of antigens through the MHC class I pathway (Del Valle et al., 2021), which primarily presents cytoplasmic, viral, and tumor-associated antigens. Antigen loading via the TAP-dependent pathway is essential for effective CD8⁺ T-cell priming and the development of CTL responses, which are critical for cellular immunity. Accordingly, rLLO-based antigen chimeras have been shown to enhance MHC class I presentation and CD8⁺ T-cell activation, supporting their potential to promote CTL responses in experimental vaccine platforms (Mantel et al., 2022). Innate activationBeyond antigen delivery, rLLO contributes to the activation of innate immune responses. Its pore-forming activity induces transient increases in intracellular calcium (Ca²⁺) levels, likely as a consequence of membrane permeabilization, which can contribute to downstream signaling events, including inflammasome activation (Lee et al., 2012). In addition, LLO has been reported to activate TLR4-dependent signaling pathways, leading to enhanced pro-inflammatory cytokine production in APCs (Cheng et al., 2020). Immune activation mechanisms by rLLO: implications for innate and adaptive immunity and vaccine developmentrLLO is a powerful immunological adjuvant that can improve the effectiveness of vaccines against intracellular pathogens by activating both innate and adaptive immune responses through inflammasome activation and improved antigen presentation. Innate immune responsesAntigen-presenting cell activationrLLO enhances the activation and functional maturation of antigen-presenting cells, particularly dendritic cells and macrophages. Pouriayevali et al. (2019) demonstrated that exposure to LLO-containing formulations resulted in increased expression of APC activation markers and increased production of pro-inflammatory cytokines, indicating enhanced innate immune stimulation. This activation state is associated with improved antigen processing and presentation capacity, thereby creating a favorable immunological environment for initiating adaptive immune responses. These effects were observed without inducing excessive cytotoxicity, supporting the suitability of rLLO as an immunostimulatory component in vaccine platforms (Pouriayevali et al., 2019). Inflammatory cytokine inductionThe liberation of key pro-inflammatory cytokines like TNF-α, IL-6, and IL-12, is induced, and these molecules act as messengers, coordinating the immune response (Pouriayevali et al., 2019). Nucleotide-binding domain, leucine-rich repeat, and pyrin domain-containing protein 3 (NLRP3) Inflammasome activationThrough its pore-forming activity, LLO-mediated membrane perturbation has been shown to induce potassium (K⁺) efflux and reactive oxygen species (ROS) generation during L. monocytogenes infection. These events have been implicated in the activation of the NLRP3 inflammasome and subsequent IL-1β production (Ayna et al., 2012). Phagosome escape mimicryrLLO helps deliver antigens into the cell’s main compartment (cytosol) in cellular models. This effectively mimics how L. monocytogenes naturally infects cells, making it a powerful tool for immune activation (Wan et al., 2015). Adaptive immune responsesrLLO also plays a pivotal role in training the adaptive immune system, providing long-lasting, specific protection (Sun and Liu, 2013). CD4⁺ T cell activationWhen processed inside endosomes, rLLO can be present on MHC II molecules, leading to helper T cell activation. These CD4+ T cells are essential for developing strong antibody responses and play a crucial role in preparing CTLs for action (Roche and Furuta, 2015). CD8⁺ T cell primingrLLO disrupts the phagosome, allowing antigens to access the cytosol. This enables cross-presentation on MHC I molecules, significantly boosting CTL priming. These "killer" T cells are vital for eliminating infected cells (Ito and Seishima, 2010). Adjuvant propertiesrLLO acts as an effective adjuvant, enhancing the immune response to co-administered antigens. It has already been used in DNA vaccines, peptide vaccines, and recombinant viral vectors (Huleatt et al., 2007; Kim et al., 2015). Standard adjuvants, such as alum, CpG oligonucleotides, and bacterial flagellin, stimulate the immune system through well-characterized pattern recognition receptors (PRRs), such as TLR4, TLR9, and NOD-like receptors. In contrast, rLLO promotes immunity via a well-defined mechanism involving the creation of transient pores in cholesterol-rich host cell membranes. This activity initiates NLRP3 inflammasome activation via potassium efflux, ROS generation, and phagosomal disruption and enables cytosolic antigen release, promoting cross-presentation via MHC class I. This twofold role in both innate and adaptive immunity is relatively rare among protein-based adjuvants (Sun and Liu, 2013; Wallecha et al., 2013). Although the interaction of rLLO with cholesterol is well understood, the precise immune-sensing receptors remain unclear. In contrast to adjuvant proteins such as flagellin (TLR5), lipopolysaccharide (TLR4), and CpG DNA (TLR9), which occupy definite PRRs, rLLO's immune stimulation likely depends on secondary mechanisms, including damage-associated molecular patterns (DAMPs), inflammasome priming (Lamkanfi and Dixit, 2014), and cytosolic sensing pathways such as absent in melanoma-2 (AIM-2) or stimulator of interferon genes (STING), though direct indication is motionless deficient (Mandal and Lee, 2002; Wallecha et al., 2013). Additionally, while adjuvants, such as cholera toxin B subunit (CTB) and heat shock proteins (HSPs), bind to GM1 gangliosides and TLR2/TLR4 and correspondingly encourage APC development without membrane damage, rLLO imitates ordinary intracellular bacterial invasion, disrupting membranes and facilitating cytosolic antigen delivery. This increases CD8⁺ T cell responses, which is particularly appreciated in vaccine approaches against intracellular pathogens and tumors (Sun and Liu, 2013; Wallecha et al., 2013). Despite these advantages, rLLO remains underexplored compared with conventional adjuvants, particularly regarding its receptor specificity, immune profile in neonates, and safety in immunocompromised hosts. Defining its molecular targets and comparing them with other immune activators is essential for optimizing its use in next-generation vaccines (Peng et al., 2007). Table 1 shows the comparative characteristics of rLLO and other adjuvant proteins. Table 1. Comparative characteristics between rLLO and other protein-based adjuvants.
rLLO applications in vaccine developmentHuman applicationsCancer immunotherapy Recombinant LLO has been widely explored in cancer vaccine platforms, especially due to its ability to facilitate cytosolic antigen delivery and promote CD8⁺ T cell responses. Listeria-based vectors expressing LLO-tumor antigen fusion proteins, such as HPV E7 or HER2/neu—have demonstrated therapeutic efficacy in cervical, prostate, and breast cancer murine models (Peng et al., 2007). These vectors enhance MHC I presentation and cytotoxic lymphocyte priming, which are crucial for tumor clearance (Sun and Liu, 2013) (Table 2). Table 2. Summary of rLLO applications in vaccinology.
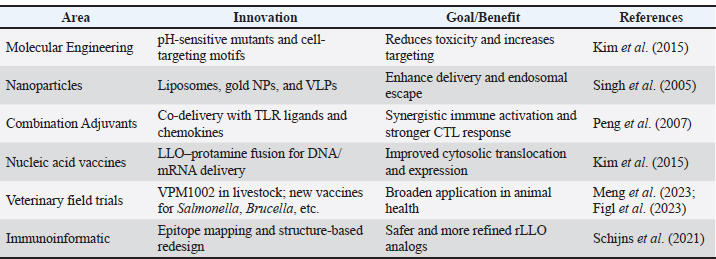
Infectious disease vaccinesTuberculosis The most sophisticated application of rLLO is the recombinant BCG strain VPM1002, which substitutes Listeria's hly gene (encoding LLO) for ureC. Th1/Th17 immunity and antigen export are improved by LLO-mediated phagosomal disruption. VPM1002 is undergoing phase II/III clinical trials and has demonstrated improved protection in mice and immunogenicity in humans (Nieuwenhuizen et al., 2017). Additionally, rLLO enhances the presentation of antigens in DNA and protein subunit TB vaccines, thereby enhancing their protective effectiveness (Phelps et al., 2020; Imad Abd-AlAziz et al., 2023), Table 2. Human immunodeficiency virus LLO-fused HIV antigens have improved CD8 T cell responses in preclinical models. These results imply that rLLO may serve as a carrier or adjuvant in viral subunit vaccines intended to boost cellular immunity (Mata et al., 2001) (Table 2). Nanoparticle and liposomal delivery systemsrLLO was successfully integrated into nanoparticles and liposomes to improve endosomal escape and antigen presentation (Table 2): - LLO-functionalized liposomes fuse with host membranes to release cargo directly into the cytoplasm (Rodriguez-Del Rio et al., 2015; Roeske et al., 2018). - Polymeric nanoparticles coloaded with rLLO and antigen improve cytosolic access and CTL priming (Mandal and Lee, 2002). Veterinary applicationsrLLO has also been used in livestock vaccines, particularly for listeriosis and TB. Listeriosis in sheepA live-attenuated L. monocytogenes strain (ΔactA, ΔplcB, ΔorfX) expressing LLO conferred 78% protection in sheep and allowed for differentiating infection from vaccinated animals (DIVAs) via anti-LLO serology (Meng et al., 2023) (Table 2). TB in goats and cattleThe VPM1002 strategy has been translated into veterinary contexts, demonstrating safety and immunogenicity in juvenile goats, thereby supporting its potential application in bovine TB vaccination (Figl et al., 2023). Probiotic and mucosal delivery platforms in animal modelsRecombinant probiotic systems expressing LLO have also been investigated as oral or mucosal vaccine platforms in animal models. Lactococcus lactis and Lactobacillus strains engineered to express LLO demonstrated stable antigen expression, effective mucosal immune stimulation, and enhanced antigen delivery, indicating the feasibility of non-invasive livestock vaccination strategies (De Azevedo et al., 2015; Hayati et al., 2017). Other potential applicationsAlthough these applications remain theoretical, rLLO has been proposed as a potential adjuvant for veterinary vaccines targeting diseases that require strong Th1-biased immune responses, such as brucellosis and bovine viral diarrhea virus (BVDV), based on immunological rationale and translational evidence (Figl et al., 2023). Future directions in rLLO-based vaccine researchThese prospective applications position rLLO as a viable immunostimulant and a customizable platform for next-generation vaccines targeting cancer, infectious diseases, and animal diseases. The integration of molecular engineering, advanced delivery systems, and computational design will be essential for translating preclinical advances into clinical and field-level success (Table 3). Table 3. Emerging directions for rLLO vaccine research.
Molecular engineering of rLLOEfforts to develop engineered rLLO variants with improved safety and specificity are underway. For example, pH-sensitive LLO mutants selectively activate pore formation under acidic endosomal conditions, thereby reducing off-target effects in neutral extracellular environments. In addition, rLLO fusion with cell-targeting ligands or nanoparticle scaffolds enables localized delivery to APCs, enhancing immunological precision (Kim et al., 2015). Nanoparticle and biomaterial integrationThe incorporation of rLLO into nanoparticle-based platforms, including gold nanoparticles, liposomes, and virus-like particles (VLPs), has shown promise in improving antigen stability, endosomal escape, and cytosolic delivery (Table 3), while simultaneously harnessing rLLO’s natural membrane-disrupting properties and reducing systemic toxicity (Singh et al., 2005). Combination of adjuvant strategiesrLLO can interact with other immunostimulants, such as TLRs ligands or cytokines, as granulocyte-macrophage colony-stimulating factor (Cao et al., 2024). For example, DNA vaccines co-delivering rLLO and chemokines have shown superior tumor immunity in murine models (Peng et al., 2007). These groupings could tailor the immune response more precisely, enhancing both humoral and cell-mediated responses (Wang and Xu, 2020). Applications in gene and mRNA vaccine platformsThe global shift toward nucleic acid vaccine technologies, rLLO, is being explored to enhance the intracellular delivery of DNA and mRNA vaccines (Lukeman et al., 2025). In addition, an LLO–protamine fusion protein has been shown to improve DNA transfection efficiency both in vitro and in vivo, providing a foundation for future mRNA vaccine designs that exploit rLLO-mediated endosomal escape (Kim et al., 2015). Veterinary expansion and field testingField trials of VPM1002-based vaccines for bovine TB are underway in veterinary medicine (Kaufmann et al., 2014). Beyond TB, rLLO could be integrated into vaccines for other economically important livestock diseases, such as Salmonella or Eimeria, in poultry, where Th1-skewed immunity is advantageous because it promotes cell-mediated immune responses and IFN-γ production that are critical for controlling intracellular pathogens (Figl et al., 2023; Yao et al., 2023). The demonstrated safety and immunogenicity of rLLO-based platforms in multiple animal systems support their potential expansion to livestock species such as swine, poultry, and ruminants for vaccines targeting intracellular pathogens (De Azevedo et al., 2015; Hayati et al., 2017; Figl et al., 2023; Yao et al., 2023). To date, no rLLO-based adjuvant has been formally approved for use in human or veterinary vaccines; however, rLLO-containing platforms, such as the recombinant BCG vaccine VPM1002, are currently undergoing clinical evaluation (Kaufmann et al., 2014). Rational design via immunoinformaticsAdvances in immunoinformatics and structural biology have enabled the rational design of rLLO variants (Ostuni et al., 2021). Computational tools are being used to optimize T cell epitopes, reduce immunopathology, and enhance stability (Suleman et al., 2022). Such epitope-guided engineering can yield safer, more effective adjuvants with tailored immune signatures (Schijns et al., 2021) (Table 3). Experimental use of rLLO in vaccine platformsTables 4 and 5 summarize key studies evaluating rLLO as an adjuvant or delivery enhancer in vaccines targeting bacterial and viral pathogens across both human and veterinary models, highlighting rLLO constructs, immune responses, immunization strategies, and experimental outcomes that collectively demonstrate its role in enhancing cellular and humoral immunity and facilitating antigen delivery in DNA-, protein-, and live-attenuated vaccine platforms. Statistical overview of rLLO-based vaccine and delivery system studiesThe statistical and inferential evidence summarized in Tables 6 and 7 collectively demonstrate a consistent trend toward enhanced cellular and Th1-biased immune responses in rLLO-based vaccine strategies. Experimental vaccine studies (Table 6) frequently reported statistically significant increases in cytokine production, CD8⁺ T-cell activation, and protective efficacy compared with conventional formulations. Complementarily, delivery system–oriented studies (Table 7) provided functional and inferential support by demonstrating improved antigen delivery and immune polarization. Together, these findings substantiate the role of rLLO as a key component in next-generation immunization platforms without necessitating quantitative meta-analysis. Safety, cytotoxicity, and regulatory considerationsRegardless of the show's potential immunological capability of rLLO, more than a few safety- and regulation-correlated challenges must be considered. Wild-type LLO is a member of the CDC family and has potent pore-forming and cytolytic activity, which initiates its function as a crucial virulence factor of L. monocytogenes (Vadia and Seveau, 2004; Hamon et al., 2012). This integral cytotoxicity promotes safety concerns for vaccine applications if membrane destruction is not tightly controlled. Although recombinant and detoxified variants of LLO have been developed to reduce cytotoxic effects, LLO-mediated membrane permeabilization and calcium influx can still induce inflammasome activation, apoptosis, and proinflammatory signaling in a dose-dependent manner (Seveau et al., 2004; Nguyen et al., 2019). Preclinical research commonly describes suitable safety profiles for rLLO-based vaccine platforms; nevertheless, heterogeneity in delivery systems, antigen fusion strategies, dosing regimens, and administration routes confuses direct comparisons across studies and underscores the need for homogeneous toxicological estimation (Kim et al., 2015; Rodriguez-Del Rio et al., 2015). From a regulatory perspective, additional challenges include manufacturing consistency, long-term stability, and the classification of rLLO as either a functional antigen carrier or an immunological adjuvant, all of which may influence regulatory trials for agreement (Del Giudice et al., 2018). At the same time, these deliberations highlight the importance of rigorous safety evaluation and clear regulatory frameworks for the extensive implementation of rLLO-based vaccine components in clinical or veterinary settings. Limitations of the reviewDespite a comprehensive literature search across multiple databases, this narrative review has limitations. Restricting sources to peer-reviewed English-language articles may have resulted in the omission of relevant findings published in other languages or gray literature. Furthermore, heterogeneity in study designs, experimental models, and immunological endpoints precluded quantitative synthesis, necessitating a qualitative narrative approach. These constraints should be considered when interpreting the findings, and future research with standardized methodologies should be encouraged. ConclusionrLLO has great potential as a multifunctional adjuvant and delivery enhancer in vaccine development. rLLO induces innate and adaptive immune responses, including CD8⁺ T cell activation, to attack intracellular infections and tumors. This is accomplished by phagosomal escape, NLRP3 inflammasome activation, and cytosolic antigen delivery. Compared with distinctive adjuvants, rLLO has distinct advantages, as proven in a variety of investigational models targeting bacterial, viral, and tumor antigens in both human and animal contexts. Furthermore, its combination with delivery systems, such as nanoparticles, liposomes, and DNA vectors, boosts its immunogenic capacity. However, issues persist. More research is desirable to control the particular immunological receptors occupied in rLLO identification, its safety profile in susceptible individuals, and dosage optimization. To wholly use rLLO's capabilities, extra study into molecular engineering, combination tactics, and clinical evaluation is essential. In conclusion, rLLO is a practical approach for next-generation vaccines, with the potential to increase immunogenicity, broaden protection, and increase vaccine efficacy across species and disease conditions. AcknowledgmentsNone. Conflict of interestThe authors have no conflicts of interest to declare. FundingThis analysis did not obtain any specific grant from community, commercial, or not-for-profit agencies. Authors' contributionsThamer J. Shihab and Eman Hashim Yousif drafted the major and minor sections of this review. The authors have reviewed and approved the final version of the manuscript. Data availabilityThis narrative review does not include any original datasets. The statistical results presented in Tables 6 and 7 were taken from peer-reviewed, previously published studies that were referenced throughout the text. All underlying data may be found in the cited corresponding open-access papers. ReferencesAgbavor, C., Zimnicka, A., Kumar, A., George, J.L., Torres, M., Prehna, G., Alonzo, F., Durrant, J.D., Freitag, N.E. and Cahoon, L.A. 2024. The chaperone PrsA2 regulates listeriolysin O the secretion, stability, and folding of listeriolysin O during Listeria monocytogenes infection. mBio 15(7), e00743-24; doi:10.1128/mbio.00743-24 Ali, H.I. and Yasssein, S.N. 2021. Immunotherapeutic effect of chitosan and listeriolysin O on Listeria monocytogenes infection in mice. Iraqi. J. Vet. Sci. 35(Suppl. I–III), 149–155; doi:10.33899/ijvs.2021.132332.2082 Asea, A., Rehli, M., Kabingu, E., Boch, J.A., Baré, O., Auron, P.E., Stevenson, M.A. and Calderwood, S.K. 2002. Novel signal transduction pathway utilized by extracellular HSP70: role of Toll-like receptor 2 and TLR4. J. Biol. Chem. 277(17), 15028–15034. Asokan, S., S, S., Jacob, T., Jisha, M.S. and Vijayan, S. 2025. Biogenic ZnO nanoparticle-coated endotracheal tubes: a study on their biocompatibility and antibacterial activity. Next Nanotechnol. 8, 100274; doi:10.1016/j.nxnano.2025.100274 Ayna, G., Krysko, D.V., Kaczmarek, A., Petrovski, G., Vandenabeele, P. and Fésüs, L. 2012. ATP release from dying autophagic cells and their phagocytosis are crucial for inflammasome activation in macrophages. PLos One. 7(6), e40069; doi:10.1371/journal.pone.0040069 Banerji, R., Karkee, A., Kanojiya, P. and Saroj, S.D. 2021. Pore-forming toxins of foodborne pathogens. Compr. Rev. Food. Sci. Food. Saf. 20(3), 2265–2285; doi:10.1111/1541-4337.12712 Beauregard, K.E., Lee, K.D., Collier, R.J. and Swanson, J.A. 1997. PH-dependent perforation of macrophage phagosomes by listeriolysin O from Listeria monocytogenes. J. Exp. Med. 186(7), 1159–1163; doi:10.1084/jem.186.7.1159 Calderón-Gonzalez, R., Terán-Navarro, H., Frande-Cabanes, E., Ferrández-Fernández, E., Freire, J., Penadés, S., Marradi, M., García, I., Gómez-Román, J., Yañez-Díaz, S. and Álvarez-Domínguez, C. 2016. Pregnancy vaccination with gold glyco-nanoparticles carrying Listeria monocytogenes peptides protects against listeriosis. Nanomaterials 6(8), 151; doi:10.3390/nano6080151 Cao, L.M., Yu, Y.F., Li, Z.Z., Zhong, N.N., Wang, G.R., Xiao, Y. and Bu, L.L. 2024. Adjuvants for cancer mRNA vaccines in the era of nanotechnology: strategies, applications, and future directions. J. Nanobiotechnol. 22(1), 308; doi:10.1186/s12951-024-02590-6 Carrero, J.A., Vivanco-Cid, H. and Unanue, E.R. 2009. Listeriolysin O is strongly immunogenic on multiple MHC backgrounds. J. Immunol. 182, 42; doi:10.4049/jimmunol.182.Supp.78.42 Cheng, C., Sun, J., Yu, H., Ma, T., Guan, C., Zeng, H., Zhang, X., Chen, Z. and Song, H. 2020. Listeriolysin O pore-forming activity is required for ERK1/2 phosphorylation. Front. Immunol. 11, 1146; doi:10.3389/fimmu.2020.01146 Cong, Z., Xiong, Y., Lyu, L., Fu, B., Guo, D., Sha, Z., Yang, B. and Wu, H. 2024. The relationship between Listeria infections and host immune responses. Biomed. Pharmacother. 171, 116129; doi:10.1016/j.biopha.2023.116129 De Azevedo, M.S.P., Santos Rocha, C., Pereira, V.B., De Oliveira Junior, A.F., De Sousa, C.S., Azevedo, V., LeBlanc, J.G., Chatel, J.M. and Miyoshi, A. 2015. Prospective uses of recombinant Lactococcus lactis expressing listeriolysin O. Genet. Mol. Res. 14(4), 18485–18493; doi:10.4238/2015.December.23.26 Del Giudice, G., Rappuoli, R. and Didierlaurent, A.M. 2018. Correlates of adjuvanticity: a review on adjuvants in licensed vaccines. Semin. Immunol. 39, 14–21; doi:10.1016/j.smim.2018.05.001 Del Valle, A., Acosta-Rivero, N., Laborde, R.J., Cruz-Leal, Y., Cabezas, S., Luzardo, M.C. and Lanio, M.E. 2021. Sticholysin II shows similar immunostimulatory properties to listeriolysin O, stimulating dendritic cells and MHC-I-restricted T cell responses of heterologous antigen. Toxicon 200, 38–47; doi:10.1016/j.toxicon.2021.07.003 Dubail, I., Autret, N., Beretti, J.L., Kayal, S., Berche, P. and Charbit, A. 2001. Functional assembly of two membrane-binding domains in listeriolysin O, the cytolysin of Listeria monocytogenes. Microbiology 147(10), 2679–2688; doi:10.1099/00221287-147-10-140279 Eldridge, M.J.G. and Hamon, M.A. 2021. Histone H3 deacetylation promotes host cell viability for efficient infection by Listeria monocytogenes. PLos Pathog. 17(12), e1010173; doi:10.1371/journal.ppat.1010173 Figl, J., Köhler, H., Wedlich, N., Liebler-Tenorio, E.M., Grode, L., Parzmair, G., Krishnamoorthy, G., Nieuwenhuizen, N.E., Kaufmann, S.H.E. and Menge, C. 2023. Safety and immunogenicity of recombinant BCG VPM1002 in goats. Int. J. Mol. Sci. 24(6), 5509; doi:10.3390/ijms24065509 Forghani, H., Jamshidi Makiani, M., Zarei Jaliani, H., Zahraei, S.M., Namayandeh, S.M. and Khani, P. 2020. Toward an alum-free mono-component monovalent pertussis vaccine. Iran. J. Immunol. 17(2), 111–120; doi:10.22034/iji.2020.86199.1749 Frosth, S., Reddick, D., Righetti, F., Bjerketorp, J., Jacobsson, K., Henriques-Normark, B., Jacobson, M., Guss, B., Wood, T., Frykberg, L., Flock, J.I. and Waller, A. 2025. Sow vaccination with a novel recombinant protein vaccine protects piglets against Streptococcus suis infection. Vaccine 53, 127077; doi:10.1016/j.vaccine.2025.127077 Gengenbacher, M., Nieuwenhuizen, N., Vogelzang, A., Liu, H., Kaiser, P., Schuerer, S., Lazar, D., Wagner, I., Mollenkopf, H.J. and Kaufmann, S.H.E. 2016. Deletion of nuoG improves BCG protection. mBio 7(3), 679; doi:10.1128/mBio.00679-16 Gupta, S. and Pellett, S. 2023. Recent developments in vaccine design: from live vaccines to recombinant toxin vaccines. Toxins 15(9), 563; doi:10.3390/toxins15090563 Hamon, M.A. and Cossart, P. 2011. K⁺ efflux is required for histone H3 dephosphorylation by Listeria monocytogenes listeriolysin O and other pore-forming toxins. Infect. Immun. 79(7), 2839–2846; doi:10.1128/IAI.01243-10 Hamon, M.A., Batsché, E., Régnault, B., Tham, T.N., Seveau, S., Muchardt, C. and Cossart, P. 2007. Histone modifications induced by a family of bacterial toxins. Proc. Natl. Acad. Sci. USA. 104(33), 13467–13472; doi:10.1073/pnas.0702729104 Hamon, M.A., Ribet, D., Stavru, F. and Cossart, P. 2012. Listeriolysin O: the Swiss army knife of Listeria. Trends. Microbiol. 20(8), 360–368; doi:10.1016/j.tim.2012.04.006 Hayati, M., Hosseinzadeh, S., Tabatabaee, S.M., Hosseini, S.M.H. and Derakhshandeh, A. 2017. Cloning and expression of listeriolysin O in Lactobacillus plantarum. Int. J. Enteric Pathog. 5(4), 115–120; doi:10.15171/ijep.2017.27 Hernández-Flores, K.G. and Vivanco-Cid, H. 2015. Biological effects of listeriolysin O. Biomed. Res. Int. 2015, 360741; doi:10.1155/2015/360741 Hernández-Flores, K.G., Calderón-Garcidueñas, A.L., Mellado-Sánchez, G., Ruiz-Ramos, R., Sánchez-Vargas, L.A., Thomas-Dupont, P., Izaguirre-Hernández, I.Y., Téllez-Sosa, J., Martínez-Barnetche, J., Wood, L., Paterson, Y., Cedillo-Barrón, L., López-Franco, O. and Vivanco-Cid, H. 2017. Evaluation of the safety and adjuvant effect of a detoxified listeriolysin O mutant on the humoral response to dengue virus antigens. Clin. Exp. Immunol. 188(1), 109–126; doi:org/10.1111/cei.12906 Hiltbold, E.M., Safley, S.A. and Ziegler, H.K. 1996. The presentation of class I and class II epitopes of listeriolysin O is regulated by intracellular localization and by intercellular spread of Listeria monocytogenes. J. Immunol. 157(3), 1163–1175. Hotze, E.M. and Tweten, R.K. 2012. Membrane assembly of the cholesterol-dependent cytolysin pore complex. Biochimica Et Biophysica Acta (BBA). -. Biomembranes. 1818(4), 1028–1038; doi:10.1016/j.bbamem.2011.07.036 Huleatt, J.W., Jacobs, A.R., Tang, J., Desai, P., Kopp, E.B., Huang, Y., Song, L., Nakaar, V. and Powell, T.J. 2007. Vaccination with recombinant fusion proteins incorporating Toll-like receptor ligands induces rapid cellular and humoral immunity. Vaccine 25(4), 763–775; doi:10.1016/j.vaccine.2006.08.028 Ibrahim, I.Z., Al-Wan, M.J. and Nahi, Y.Y. 2012. Study the influence of some Listeria monocytogenes antigens on the side effects of mitomycin C. Iraqi J. Vet. Med. 36(0E), 347–354; doi:org/10.30539/iraqijvm.v36i0E.442 Ibrahim, Z.I., Abdul-Mounam, M.A.W. and Alsufi, L.A.M. 2017. Study of ultrastructure changes in the liver of mice post infection with Listeria monocytogenes. Iraqi J. Vet. Med. 41(1), 55–159; doi:org/10.30539/iraqijvm.v41i1.99 Ilangumaran Ponmalar, I., Ayappa, K.G. and Basu, J.K. 2021. Bacterial protein listeriolysin O induces nonmonotonic dynamics because of lipid ejection and crowding. Biophys. J. 120(15), 3040–3049; doi:10.1016/j.bpj.2021.06.014 Imad Abd-AlAziz, S., Thalij, K.M. and Zakari, M.G. 2023. Inhibitory susceptibility of synthetic selenium nanoparticles and some conjugate nutritional compounds in inhibition of some bacterial isolates causing food poisoning. Tikrit J. Agric. Sci. 23(4), 26–34; doi: 10.25130/tjas.23.4.3 Ito, H. and Seishima, M. 2010. Regulation of the induction and function of cytotoxic T lymphocytes by natural killer T cell. J. Biomed. Biotechnol. 2010, 641757; doi:10.1155/2010/641757 Kaufmann, S.H., Cotton, M.F., Eisele, B., Gengenbacher, M., Grode, L., Hesseling, A.C. and Walzl, G. 2014. The BCG replacement vaccine VPM1002: from drawing board to clinical trial. Expert. Rev. Vaccines. 13(5), 619–630; doi:10.1586/14760584.2014.905746 Kayal, S., Lilienbaum, A., Poyart, C., Memet, S., Israel, A. and Berche, P. 1999. Listeriolysin O-dependent activation of endothelial cells during infection with Listeria monocytogenes: activation of NF-κB and upregulation of adhesion molecules and chemokines. Mol. Microbiol. 31(6), 1709–1722; doi:10.1046/j.1365-2958.1999.01305.x Khitam S.S, Alhtheal E.D. and Azhar J.B. 2018. Effect of zinc oxide nanoparticles prepared from zinc sulphate (ZnSO₄) against Gram-negative and Gram-positive microorganisms in vitro. Iraqi J. Vet. Med. 42(1), 18–22; doi:10.30539/iraqijvm.v42i1.25 Kim, N.H., Provoda, C. and Lee, K.D. 2015. Design and characterization of novel recombinant listeriolysin O–protamine fusion proteins for enhanced gene delivery. Mol. Pharm. 12(2), 342–350; doi:10.1021/mp500592j Köster, S., Van Pee, K., Hudel, M., Leustik, M., Rhinow, D., Kühlbrandt, W., Chakraborty, T. and Yildiz, O. 2014. Crystal structure of listeriolysin O reveals molecular details of oligomerization and pore formation. Nat. Commun. 5, 3690; doi:10.1038/ncomms4690 Lai, D.C., Nguyen, T.N., Poonsuk, K., Mcvey, D.S. and Vu, H.L.X. 2025. Lipid nanoparticle-encapsulated DNA vaccine encoding African swine fever virus p54 antigen elicits robust immune responses in pigs. Vet. Microbiol. 305, 110508; doi:10.1016/j.vetmic.2025.110508 Lamkanfi, M. and Dixit, V.M. 2014. Mechanisms and functions of inflammasomes. Cell 157(5), 1013–1022; doi:10.1016/j.cell.2014.04.00 Lee, G.S., Subramanian, N., Kim, A.I., Aksentijevich, I., Goldbach-Mansky, R., Sacks, D.B., Germain, R.N., Kastner, D.L. and Chae, J.J. 2012. The calcium-sensing receptor regulates the NLRP3 inflammasome through Ca²⁺ and cAMP. Nature 492(7427), 123–127; doi:10.1038/nature11588 Li, L., Yan, X., Xia, M., Shen, B., Cao, Y., Wu, X., Sun, J., Zhang, Y. and Zhang, M. 2021. Nanoparticle/nanocarrier formulation as an antigen: the immunogenicity and antigenicity of itself. Mol. Pharmaceutics 19(1), 148–159; doi:10.1021/acs.molpharmaceut.1c00704 Li, R., Liang, Q., Tian, S., Zhang, Y., Liu, S., Ou, Q., Chen, Z. and Wang, C. 2022. Hemolysin function of Listeria is related to biofilm formation: transcriptomics analysis. Vet. Res. 53(1), 113; doi:10.1186/s13567-022-01124-y Liang, Q., Li, R., Liu, S., Zhang, Y., Tian, S., Ou, Q., Chen, Z. and Wang, C. 2022. Recombinant Listeria ivanovii strain expressing listeriolysin O might be a potential antigen carrier for vaccine construction. Front. Microbiol. 13, 962326; doi:10.3389/fmicb.2022.962326 Lukeman, H., Chan, E. and Triccas, J. 2025. Targeting tuberculosis with LNP-mRNA vaccines: opportunities, challenges and future directions. Microbiol. Aust. 46(2), 48–51; doi:10.1071/MA25017 Mandal, M. and Lee, K.D. 2002. Listeriolysin O-liposome-mediated cytosolic delivery of macromolecule antigen in vivo. Biochimica Et Biophysica Acta (BBA) - Mol. Cell. Biol. Lipids. 1563(1–2), 7–17; doi:10.1016/S0005-2736(02)00380-4 Mantel, I., Sadiq, B.A. and Blander, J.M. 2022. Spotlight on TAP and its vital role in antigen presentation and cross-presentation. Mol. Immunol. 142, 105–119; doi:10.1016/j.molimm.2021.11.013 Mata, M., Yao, Z.J., Zubair, A., Syres, K. and Paterson, Y. 2001. Evaluation of a recombinant Listeria monocytogenes expressing an HIV protein that protects mice against viral challenge. Vaccine 19(11), 1435–1445; doi:10.1016/S0264-410X(00)00355-7 Meng, F., Zhu, T., Chen, C., Yao, H., Zhang, R., Li, J., Chen, X., Huang, J., Pan, Z., Jiao, X. and Yin, Y. 2023. A live attenuated DIVA vaccine affords protection against Listeria monocytogenes challenge in sheep. Microb. Pathog. 181, 106204; doi:10.1016/j.micpath.2023.106204 Nascimento, I.P. and Leite, L.C.C. 2012. Recombinant vaccines and the development of new vaccine strategies. Braz. J. Med. Biol. Res. 45(12), 1102–1111; doi:10.1590/S0100-879X2012007500142 Nguyen, B.N., Peterson, B.N. and Portnoy, D.A. 2019. Listeriolysin O: a phagosome-specific cytolysin revisited. Cell. Microbiol. 21(3), e12988; doi:10.1111/cmi.12988 Nguyen, D.N., Mahon, K.P., Chikh, G., Kim, P., Chung, H., Vicari, A.P., Love, K.T., Goldberg, M., Chen, S., Krieg, A.M., Chen, J., Langer, R. and Anderson, D.G. 2012. Lipid-derived nanoparticles for immunostimulatory RNA adjuvant delivery. Proc. Natl. Acad. Sci. USA. 109(14), E797–E803; doi:10.1073/pnas.1121423109 Nieuwenhuizen, N.E., Kulkarni, P.S., Shaligram, U., Cotton, M.F., Rentsch, C.A., Eisele, B., Grode, L. and Kaufmann, S.H.E. 2017. The recombinant Bacille Calmette–Guérin vaccine VPM1002: ready for clinical efficacy testing. Front. Immunol. 8, 1147; doi:10.3389/fimmu.2017.01147 Obaidi, F.A.A., Abdul-Nabi, B.S. and Al-Shididi, S.M. 2010. Saccharomyces cerevisiae cell wall effects on immunological response of broiler chicks infected with Listeria monocytogenes. Iraqi. J. Vet. Med. 34(1), 1; doi:10.30539/iraqijvm.v34i1.665 Ostuni, A., Monné, M., Crudele, M.A., Cristinziano, P.L., Cecchini, S., Amati, M., De Vendel, J., Raimondi, P., Chassalevris, T., Dovas, C.I. and Bavoso, A. 2021. Design and structural bioinformatic analysis of polypeptide antigens useful for the SRLV serodiagnosis. J. Virol. Methods 297, 114266; doi:10.1016/j.jviromet.2021.114266 Peng, X., Treml, J. and Paterson, Y. 2007. Adjuvant properties of listeriolysin O protein in a DNA vaccination strategy. Cancer. Immunol. Immunother. 56(6), 797–806; doi:10.1007/s00262-006-0226-5 Pham, H.G., Tran, K.N., Gomelsky, L., Roy, T., Gigley, J.P. and Gomelsky, M. 2025. Robust inducible gene expression in intracellular Listeria monocytogenes in vitro ACS. Synth. Biol. 14(5), 1397–1404; doi:10.1021/acssynbio.5c00131 Phelps, C.C., Vadia, S., Boyaka, P.N., Varikuti, S., Attia, Z., Dubey, P., Satoskar, A.R., Tweten, R. and Seveau, S. 2020. A listeriolysin O subunit vaccine is protective against Listeria monocytogenes. Vaccine 38(36), 5803–5813; doi:10.1016/j.vaccine.2020.07.002 Picard, M.D., Bodmer, J.L., Gierahn, T.M., Lee, A., Price, J., Cohane, K., Clemens, V., Devault, V.L., Gurok, G., Kohberger, R., Higgins, D.E., Siber, G.R., Flechtner, J.B. and Geisler, W.M. 2015. Resolution of Chlamydia trachomatis infection is associated with a distinct T cell response profile. Clin. Vaccine Immunol. 22(11), 1206–1218; doi:10.1128/CVI.00431-15 Portnoy, D.A., Jacks, P.S. and Hinrichs, D.J. 1988. Role of hemolysin for the intracellular growth of Listeria monocytogenes. J. Exp. Med. 167(4), 1459–1471; doi:10.1084/jem.167.4.1459 Pouriayevali, M.H., Bamdad, T., Sadat, S.M., Sadeghi, S.A., Sabahi, F., Mahdavi, M. and Aghasadeghi, M.R. 2019. Listeriolysin O immunogenetic adjuvant enhances potency of hepatitis C virus NS3 DNA vaccine. IUBMB. Life 71(10), 1645–1652; doi:10.1002/iub.2082 Radtke, A.L., Anderson, K.L., Davis, M.J., Dimagno, M.J., Swanson, J.A. and O'Riordan, M.X. 2011. Listeria monocytogenes exploits cystic fibrosis transmembrane conductance regulator (CFTR) to escape the phagosome. Proc. Natl. Acad. Sci. USA. 108(4), 1633–1638; doi:10.1073/pnas.1013262108 Rea, D., Johnson, M.E., Havenga, M.J.E., Melief, C.J.M. and Offringa, R. 2001. Strategies for improved antigen delivery into dendritic cells. Trends. Mol. Med. 7(3), 91–94; doi:10.1016/S1471-4914(01)01948-7 Roche, P.A. and Furuta, K. 2015. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 15(4), 203–216; doi:10.1038/nri3818 Rodriguez-del Rio, E., Marradi, M., Calderon-Gonzalez, R., Frande-Cabanes, E., Penadés, S., Petrovsky, N. and Alvarez-Dominguez, C. 2015. A gold glyco-nanoparticle carrying a listeriolysin O peptide and formulated with Advax delta inulin adjuvant induces robust T-cell protection against Listeria infection. Vaccine 33(12), 1465–1473; doi:10.1016/j.vaccine.2015.01.067 Roeske, K., Stachowiak, R., Jagielski, T., Kamiński, M. and Bielecki, J. 2018. Delivery of chicken egg ovalbumin to dendritic cells by listeriolysin O-secreting vegetative Bacillus subtilis. J. Microbiol. Biotechnol. 28(1), 122–135; doi:10.4014/jmb.1709.09009 Saiga, H., Nieuwenhuizen, N., Gengenbacher, M., Koehler, A.B., Schuerer, S., Moura-Alves, P., Wagner, I., Mollenkopf, H.J., Dorhoi, A. and Kaufmann, S.H.E. 2015. The recombinant BCG ΔureC: :hly vaccine targets the AIM2 inflammasome to induce autophagy and inflammation. J. Infect. Dis. 211(11), 1831–1841; doi:10.1093/infdis/jiu675 Sarmadi, M., Gheibi, A., Khanahmad, H., Khorramizadeh, M.R., Hejazi, S.H., Zahedi, N., Mianesaz, H. and Kashfi, K. 2022. Design and characterization of a recombinant Brucella abortus RB51 vaccine that elicits enhanced T cell-mediated immune response. Vaccines 10(3), 388; doi:10.3390/vaccines10030388 Schijns, V., Majhen, D., Van Der Ley, P., Thakur, A., Summerfield, A., Berisio, R., Nativi, C., Fernández-Tejada, A., Alvarez-Dominguez, C., Gizurarson, S., Zamyatina, A., Molinaro, A., Rosano, C., Jakopin., Gursel, I. and McClean, S. 2021. Rational vaccine design in times of emerging diseases: critical choices of immunological correlates of protection, vaccine antigen and immunomodulation. Pharmaceutics 13(4), 501; doi:10.3390/pharmaceutics13040501 Schuerch, D.W., Wilson-Kubalek, E.M. and Tweten, R.K. 2005. Molecular basis of listeriolysin O pH dependence. Proc. Natl. Acad. Sci. USA. 102(35), 12537–12542; doi:10.1073/pnas.0500558102 Seveau, S., Bierne, H., Giroux, S., Prévost, M.C. and Cossart, P. 2004. Role of lipid rafts in E-cadherin- and HGF-R/Met-mediated entry of Listeria monocytogenes into host cells. J. Cell Biol. 166(5), 743–753; doi:10.1083/jcb.200404148 Singh, R., Dominiecki, M.E., Jaffee, E.M. and Paterson, Y. 2005. Fusion to listeriolysin O and delivery by Listeria monocytogenes enhances the immunogenicity of HER-2/neu and reveals subdominant epitopes in mice. J. Immunol. 175(6), 3663–3673; doi:10.4049/jimmunol.175.6.3663 Smith, K.D., Andersen-Nissen, E., Hayashi, F., Strobe, K., Bergman, M.A., Barrett, S.L., Cookson, B.T. and Aderem, A. 2003. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat. Immunol. 4(12), 1247–1253; doi:10.1038/ni1011 Stierschneider, A. and Wiesner, C. 2023. Shedding light on the molecular and regulatory mechanisms of TLR4 signaling in endothelial cells under physiological and inflamed conditions. Front. Immunol. 14, 1264889; doi:10.3389/fimmu.2023.1264889 Stratmann. 2015. Cholera toxin subunit B as adjuvant: an accelerator in protective immunity and a brake in autoimmunity. Vaccines 3(3), 579–599; doi:10.3390/vaccines3030579 Suleman, M., Rashid, F., Ali, S., Sher, H., Luo, S., Xie, L. and Xie, Z. 2022. Immunoinformatic-based design of immune-boosting multiepitope subunit vaccines against monkeypox virus and validation through molecular dynamics and immune simulation. Front. Immunol. 13, 1042997; doi:10.3389/fimmu.2022.1042997 Sun, R. and Liu, Y. 2013. Listeriolysin O as a strong immunogenic molecule for development of new anti-tumor vaccines. Hum. Vaccin. Immunother. 9(5), 1058–1068; doi:10.4161/hv.23529 Tanaka, K., Enomoto, N., Uehara, M., Furuhashi, K., Sakurai, S., Yasui, H., Karayama, M., Hozumi, H., Suzuki, Y., Fujisawa, T., Inui, N., Nakamura, Y., Nagata, T. and Suda, T. 2020. Development of a novel T cell-oriented vaccine using CTL/Th-hybrid epitope long peptide and biodegradable microparticles against an intracellular bacterium. Microbiol. Immunol. 64(10), 666–678; doi:10.1111/1348-0421.12828 Tsuchiya, K., Kawamura, I., Takahashi, A., Nomura, T., Kohda, C. and Mitsuyama, M. 2005. Listeriolysin O-induced membrane permeation mediates persistent interleukin-6 production in Caco-2 cells during Listeria monocytogenes infection in vitro. Infect. Immun. 73(7), 3869–3877; doi:10.1128/IAI.73.7.3869-3877.2005 Vadia, S., Arnett, E., Haghighat, A.C., Wilson-Kubalek, E.M., Tweten, R.K. and Seveau, S. 2011. The pore-forming toxin listeriolysin O mediates a novel entry pathway of L. monocytogenes into human hepatocytes. PLoS pathog. 7(11), e1002356; doi:org/10.1371/journal.ppat.1002356 Wallecha, A., Wood, L., Pan, Z.K., Maciag, P.C., Shahabi, V. and Paterson, Y. 2013. Listeria monocytogenes-derived listeriolysin O has pathogen-associated molecular pattern-like properties independent of its hemolytic ability. Clin. Vaccine. Immunol. 20(1), 77–84; doi:10.1128/CVI.00502-12 Wan, Y., Moyle, P.M. and Toth, I. 2015. Endosome escape strategies for improving the efficacy of oligonucleotide delivery systems. Curr. Med. Chem. 22(29), 3326–3346; doi:10.2174/0929867322666150727112704 Wang, Y.S., Kumari, M., Chen, G.H., Hong, M.H., Yuan, J.P.Y., Tsai, J.L. and Wu, H.C. 2023. MRNA-based vaccines and therapeutics: an in-depth survey of current and upcoming clinical applications. J. Biomed. Sci. 30(1), 84; doi:10.1186/s12929-023-00948-8 Wang, Z.B. and Xu, J. 2020. Better adjuvants for better vaccines: progress in adjuvant delivery systems, modifications, and adjuvant–antigen codelivery. Vaccines 8(1), 128; doi:10.3390/vaccines8010128 Wira, C.R., Fahey, J.V., Sentman, C.L., Pioli, P.A. and Shen, L. 2005. Innate and adaptive immunity in the female genital tract: cellular responses and interactions. Immunol. Rev. 206(1), 306–335; doi:10.1111/j.0105-2896.2005.00287.x Xia, J., Feng, Y., Xu, J., Deng, S., Chen, M., Han, Y. and Cheng, C. 2025. Novel amino acid residues in listeriolysin O drive phagosome escape and pathogenicity in Listeria monocytogenes. Virulence 16(1), 2580098; doi:10.1080/21505594.2025.2580098 Yao, Y., Zhang, Z. and Yang, Z. 2023. The combination of vaccines and adjuvants to prevent the occurrence of high incidence of infectious diseases in bovine. Front. Vet. Sci. 10, 1243835; doi:10.3389/fvets.2023.1243835 Yin, Y., Zhang, C., Dong, H., Niu, Z., Pan, Z., Huang, J. and Jiao, X. 2010. Protective immunity induced by an LLO-deficient Listeria monocytogenes. Microbiol. Immunol. 54(4), 175–183; doi:10.1111/j.1348-0421.2010.00211.x Yusuf, K., Sampath, V. and Umar, S. 2023. Bacterial infections and cancer: exploring this association and its implications for cancer patients. Int. J. Mol. Sci. 24(4), 3110; doi:10.3390/ijms24043110 Zhao, T., Cai, Y., Jiang, Y., He, X., Wei, Y., Yu, Y., and Tian, X. 2023. Vaccine adjuvants: mechanisms and platforms. Signal Transduct. Target. Ther. 8(1), 283; doi:10.1038/s41392-023-01557-7 | ||
| How to Cite this Article |
| Pubmed Style Shihab TJ, Yousif EH. Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Vet. J.. 2026; 16(3): 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 Web Style Shihab TJ, Yousif EH. Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. https://www.openveterinaryjournal.com/?mno=292418 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.1 AMA (American Medical Association) Style Shihab TJ, Yousif EH. Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Vet. J.. 2026; 16(3): 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 Vancouver/ICMJE Style Shihab TJ, Yousif EH. Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 Harvard Style Shihab, T. J. & Yousif, . E. H. (2026) Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Vet. J., 16 (3), 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 Turabian Style Shihab, Thamer J., and Eman Hashim Yousif. 2026. Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Veterinary Journal, 16 (3), 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 Chicago Style Shihab, Thamer J., and Eman Hashim Yousif. "Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine." Open Veterinary Journal 16 (2026), 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 MLA (The Modern Language Association) Style Shihab, Thamer J., and Eman Hashim Yousif. "Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine." Open Veterinary Journal 16.3 (2026), 1402-1425. Print. doi:10.5455/OVJ.2026.v16.i3.1 APA (American Psychological Association) Style Shihab, T. J. & Yousif, . E. H. (2026) Recombinant listeriolysin-O as a platform for next-generation vaccines: Mechanisms, immunological evidence, and translational applications in human and veterinary medicine. Open Veterinary Journal, 16 (3), 1402-1425. doi:10.5455/OVJ.2026.v16.i3.1 |