| Research Article | ||
Open Vet. J.. 2026; 16(2): 1072-1081
Open Veterinary Journal, (2026), Vol. 16(2): -1081 Research Article Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, VietnamManh Tuong Nguyen, Thi Lan Huong Pham, Ha Thi Thanh Nguyen, and Thanh Trung Nguyen*Department of Pharmacology, Toxicology, Internal Medicine and Diagnostics, Faculty of Veterinary Medicine, Vietnam National University of Agriculture, Hanoi, Vietnam *Corresponding Author: Thanh Trung Nguyen. Department of Pharmacology, Toxicology, Internal Medicine and Diagnostics, Faculty of Veterinary Medicine, Vietnam National University of Agriculture, Hanoi, Vietnam. Email: nguyenthanhtrung [at] vnua.edu.vn Submitted: 22/09/2025 Revised: 29/12/2025 Accepted: 12/01/2026 Published: 28/02/2026 © 2026 Open Veterinary Journal
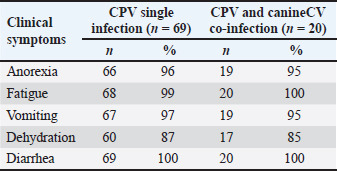
ABSTRACTBackground: Canine parvovirus (CPV) causes acute hemorrhagic gastroenteritis in dogs, whereas canine circovirus (CanineCV), an emerging virus, is associated with immunosuppression and gastrointestinal disease. However, data on CPV–CanineCV coinfections are limited in Vietnam. Aim: The aim of this study was to investigate the prevalence and clinical, hematological, biochemical, and pathological features of CPV–CanineCV coinfections in dogs with enteric disease in Hanoi, Vietnam. Methods: The fecal and blood samples from suspected CPV cases were tested using molecular diagnostics. Clinical and laboratory findings were compared between the CPV mono- and coinfected groups. Necropsies were performed on the deceased animals. Results: CPV infection was confirmed in 94.68% (89/94) of the cases, with 21.28% (19/89) showing CanineCV coinfection. Diarrhea, vomiting, and anorexia were observed in both groups, but coinfected dogs exhibited more severe symptoms, higher mortality, longer treatment, and worse recovery. The hematological findings included marked leukopenia, lymphopenia, and thrombocytopenia. Biochemical changes, such as hypoglycemia, hypoproteinemia, hypoalbuminemia, and mild liver enzyme elevations, along with notable hypokalemia and mild metabolic acidosis, were observed. Pathological findings showed a more extensive intestinal and systemic damage in patients with co-infections. Conclusion: CPV–CanineCV coinfection results in more severe disease than CPV alone. Routine Canine CV screening in CPV-positive dogs is recommended for better clinical management. Keywords: Circovirus, Coinfection, Dogs, Hanoi, Parvovirus. IntroductionAs society continues to progress, companion animals, particularly pet dogs, are receiving increased attention and care. The rising popularity of imported dog breeds has significantly contributed to the growth of the pet dog population. Despite notable advancements in veterinary medicine and the widespread adoption of vaccination programs, viral infectious diseases continue to pose a serious threat to canine health (Gibbs and Gibbs, 2013). Among these, canine parvovirus (CPV) remains a particular concern due to its high morbidity and mortality rates (Segalés et al., 2005; Decaro and Buonavoglia, 2012). In recent years, co-infections involving CPV and other viruses have been increasingly documented, with canine circovirus (CanineCV) emerging as a virus of growing concern (Nguyen Manh et al., 2021; Tuong et al., 2021). CanineCV is a relatively newly identified virus in dogs, and its pathogenic mechanisms and epidemiological patterns are currently poorly understood. Existing studies have primarily focused on its genetic characterization, identifying five genotypes—CanineCV-1 through CanineCV-5—distributed across various geographical regions (Liu et al., 2024). CanineCV-1 and CanineCV-3 have been detected in dogs suffering from CPV-associated diarrhea in Vietnam, indicating that the virus is actively circulating within the domestic canine population (Tuong et al., 2021). In 2002, the CPV-2c variant was first reported in Vietnam in 2002, marking the first detection of this strain in Asia (Nakamura et al., 2004). Since then, it has been identified in several other Asian countries, including China, India, Taiwan, Indonesia, Laos, and Thailand (Nandi et al., 2010; Geng et al., 2015; Chiang et al., 2016; Vannamahaxay et al., 2017; Charoenkul et al., 2019; Moon et al., 2020). These studies have primarily focused on the molecular characteristics of the VP2 gene, with CPV-2c full-length genome characterizations now available in Asia (Nguyen Manh et al., 2021). However, due to the relatively recent emergence of CanineCV, research on this virus in Vietnam remains limited—particularly in relation to its prevalence, pathogenic potential, and role in coinfections. A central unanswered question is whether CanineCV can exist asymptomatically in healthy dogs or contributes to disease severity when coinfecting with CPV. Such coinfections could intensify clinical symptoms and pathological damage, complicating the diagnosis, treatment, and disease control efforts (Balboni et al., 2022). Therefore, understanding the pathological outcomes of CPV and CanineCV co-infections is critical. Preliminary evidence demonstrates that dual infections may result in more complex and severe GI and systemic lesions than infections caused by either virus alone. However, the pathogenesis of CanineCV-associated disease and the potential synergistic effects of CanineCV and other pathogens have not been clarified. To help fill these gaps, this study aimed to investigate the pathological features of diarrheic enteritis in dogs co-infected with CPV and CanineCV in the Hanoi area. The findings of this study are expected to enhance the current understanding of viral coinfections in dogs and inform improvements in veterinary diagnostic, treatment, and prevention protocols in Vietnam. Materials and MethodsSample collection and nucleic acid extractionDogs exhibiting clinical signs of enteritis and diarrhea who were admitted to veterinary clinics in Hanoi were selected for sample collection. A total of 94 dog samples were collected from five veterinary clinics in Hanoi between August 2023 and October 2024. We obtained rectal swabs using sterile cotton swabs, which were immediately placed into 1.5 ml Eppendorf tubes containing 1 ml of phosphate-buffered saline (pH 7.4). The samples were kept refrigerated during transport to the laboratory and subsequently stored at 20°C until further analysis. Viral DNA was extracted using the Viral DNA/RNA Extraction Kit II (Geneaid Biotech, Taiwan) according to the manufacturer’s instructions. Fecal samples from all dogs with diarrhea were subjected to standard parasitological examination, including flotation and sedimentation techniques, to detect helminth eggs and protozoan cysts. Each dog’s deworming history was recorded based on owner-reported treatment. Detection of viral nucleic acids by polymerase chain reaction (PCR)The extracted viral nucleic acids were subjected to PCR for the detection of parvovirus using specific primers targeting the VP2 gene (Nguyen Manh et al., 2021) and the Rep gene for circovirus using the primer pair CanineCV-605F and CanineCV-1041R following protocols described in previous studies (Buonavoglia et al., 2000) (Supplementary Table 1). Thermal cycling was initiated at 94 °C for 7 minutes, followed by 35 cycles of denaturation at 94 °C for 30 seconds, annealing at 55 °C for 30 seconds, and extension at 72 °C for 1 minute. A final extension step was performed at 72 °C for 7 minutes. PCR products were separated on a 1.0% (w/v) agarose gel containing 0.5% (v/v) ethidium bromide and visualized using a UV transilluminator. Assessment of the clinical indicatorsDogs showing clinical signs of enteritis and diarrhea were initially screened for CPV using a commercial rapid test. Patients who were positive and receiving treatment were monitored daily until recovery or the end of therapy. After PCR confirmation of CPV and detection of co-infections, clinical data were compiled and analyzed using Microsoft Excel to compare CPV mono-infected dogs with those co-infected with CanineCV. A clinical scoring system was applied with slight modification to assess disease severity, incorporating symptoms such as coma, vomiting, dehydration, diarrhea, anorexia (scored by duration), and debilitation (Iris et al., 2010). Specifically, the clinical scoring system for dogs with enteritis and diarrhea assigns 1 point for coma, 1 point for vomiting, 3 points for dehydration, 5 points for diarrhea, 1, 6, or 12 points for anorexia lasting 1, 2, or 3 days, respectively, and 12 points for a moribund condition. Each symptom was weighted based on severity, with total scores classified as mild (<10), moderate (10–15), or severe (>15). Table 1. Clinical symptoms of dogs with diarrheal enteritis co-infected with parvovirus and circovirus.
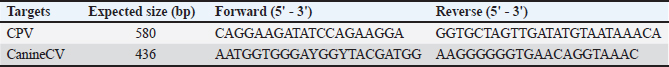
Supplementary Table 1. Primer sequences used in this study.
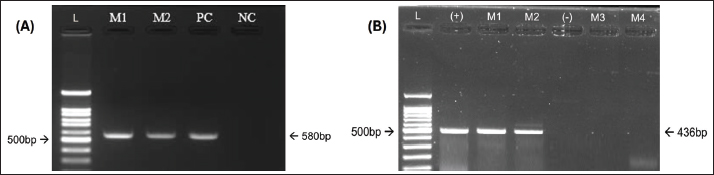
Hematological and biochemical analysis of the samplesFor each CPV-positive dog, 2 ml of blood was aseptically collected from the cephalic or saphenous vein using a 3 ml syringe and 25G butterfly needle. The sample was split into two tubes: one with ethylenediaminetetraacetic acid for hematology and one with heparin for biochemical and electrolyte analysis. Hematological parameters were measured using 1 ml of ethylenediaminetetraacetic acid blood on a GENVET-VH20 hematology analyzer (24 CBC parameters) (Genvet, China). Biochemical and electrolyte analyses were performed with 100 µl of heparinized blood using 16- and 13-parameter test plates, respectively, on the SMT-120V analyzer (Seamaty, Singapore). For deceased dogs, blood samples were collected during the terminal stage of the disease, generally within a few hours before death, to evaluate hematological changes associated with mortality. Assessment of treatment effectivenessTreatment outcomes were recorded at the end of the treatment period and entered into a spreadsheet for statistical analysis. The study participants were divided into two groups based on infection status: dogs with parvovirus mono-infection and dogs co-infected with parvovirus and circovirus. The mortality and recovery rates were calculated and compared between the two groups. Treatment effectiveness was further evaluated by measuring survival and recovery times. The survival time was defined as the interval from the start of treatment until death for dogs that did not survive. The recovery time was defined as the interval from the start of treatment until full clinical recovery for dogs that survived. Full recovery was determined based on the resolution of clinical symptoms and a white blood cell (WBC) count equal to or greater than 6.0 × 109/l, indicating normalization of hematological parameters (McCourt and Rizzi, 2022). Lesion assessmentLesions were assessed in both groups by examining specific characteristic changes in various organs. The intestinal wall was evaluated for thickening and discoloration. The presence of water, mucus, or blood was inspected in the intestinal lumen. Abdominal and thoracic lymph nodes were examined for edema and congestion. The heart was observed for the presence of pale streaks. These findings were recorded to compare lesion patterns between dogs with parvovirus mono-infection and those coinfected with parvovirus and circovirus. Statistical analysisResults are presented as means ± SD for continuous variables and as proportions for categorical variables. The Mann–Whitney U test was used to analyze continuous variables due to the small sample sizes and nonnormal data distribution. Categorical variables (e.g., mortality, recovery, and severity grades) were compared using the chi-square or Fisher’s exact tests. The time-to-event outcomes were assessed using Kaplan–Meier survival analysis with log-rank tests. All statistical analyses were performed using GraphPad Prism 4.3 (GraphPad, San Diego, CA, USA), and a p-value < 0.05 was considered statistically significant. Ethical approvalThe Experimental Animal Research Committee of Vietnam National University of Agriculture approved all experimental protocols (CARE-2023/15). ResultsCo-infection rate of parvovirus and circovirus in dogsOf the 94 dogs diagnosed with diarrheal enteritis, 89 (94.68%) tested positive for CPV using a rapid enzyme-linked immunosorbent assay test kit (Bionote, South Korea). Fecal samples from the CPV-positive dogs were further analyzed by PCR for both CPV (Fig. 1A) and canine circovirus (CanineCV) (Fig. 1B). PCR screening was only detected in dogs co-infected with CPV and was not found as a sole infection. Specifically, 19 of the 89 CPV-positive dogs were co-infected with CanineCV, yielding a co-infection rate of 21.28%, consistent with previous reports (Nguyen Manh et al., 2021). The most frequently observed clinical signs in affected dogs were vomiting, anorexia, lethargy, bloody diarrhea with pale mucous membranes, fever (>39.5°C), and dehydration affecting ≥5% of body weight. A comparison of clinical signs between dogs with CPV mono-infection (n=69) and those co-infected with CPV and CanineCV (n=20) is presented in Table 1.
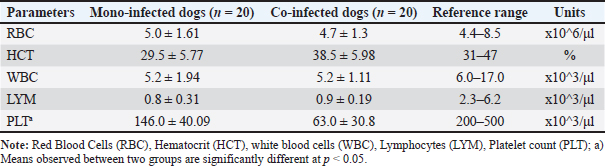
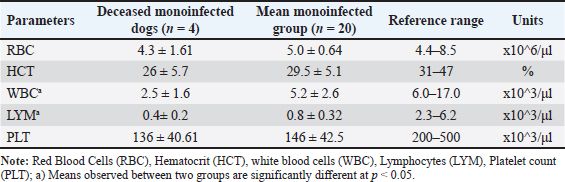
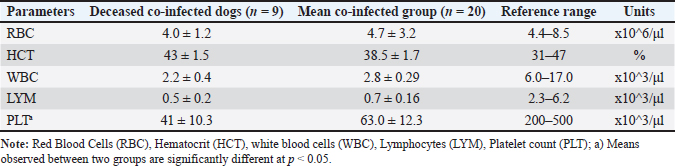
Fig. 1. Detection of CPV and Canine CV using PCR. (A) PCR amplification of CPV using primer pair A produced a bright, clearly defined band at approximately 580 bp, consistent with that of the positive control, indicating the presence of CPV in the sample. (B) PCR amplification of CanineCV using primer pair B generated a distinct band at approximately 436 bp, matching the positive control and confirming CanineCV co-infection in CPV-positive samples. L: DNA ladder (marker); M1–M4: test samples; PC or (+): positive control; NC or (−): negative control. All dogs in both groups exhibited diarrhea (100%). The prevalence of other clinical signs, such as anorexia, vomiting, fatigue, and dehydration, was also high and showed minimal variation between the two groups. These results indicate that the clinical manifestations of CPV mono-infection and CPV–CanineCV coinfection are largely indistinguishable. Therefore, clinical signs alone may not be sufficient for differentiating single and coinfections at the initial presentation. All examined fecal samples (n=94) were negative for common enteric parasites, including helminth eggs and protozoan cysts. All dogs had a documented history of recent deworming. These findings indicate that the observed enteritis and diarrhea were unlikely to be associated with parasitic infections, supporting the role of CPV and CanineCV coinfection in disease manifestation. Hematological findings in dogs with CPV mono-infection and CPV–CanineCV coinfectionA total of 40 dogs (20 with CPV mono-infection and 20 with CPV–CanineCV coinfection) were randomly selected for hematological evaluation (Table 2). The mean red blood cell (RBC) counts were within the normal range for both groups, although near the lower limit (mono-infected: 5.0 ± 1.61 × 106/μl; co-infected: 4.7 ± 1.3 × 106/μl; normal range: 4.4–8.5 × 106/μl), with no signs of anemia. hematocrit (HCT) values were slightly lower in mono-infected dogs (29.5 ± 5.77%) and closer to the normal range in co-infected dogs (38.5 ± 5.98%; normal: 31%–47%). WBC counts were decreased in both groups (5.2 ± 1.94 ×10³/μl in mono-infected; 5.2 ± 1.11 × 10³/μl in co-infected; normal: 6.0–17.0 × 10³/μl), reflecting leukopenia. Lymphocyte counts were markedly reduced in both groups (mono-infected: 0.8 ± 0.31 × 10³/μl; co-infected: 0.9 ± 0.19 × 10³/μl; normal: 2.3–6.2 × 10³/μl), consistent with CPV-induced lymphopenia. Thrombocytopenia was more severe in co-infected dogs (63.0 ± 30.8 × 10³/μl) compared to mono-infected dogs (146.0 ± 40.09 × 10³/μl), both below the normal range (200–500 × 10³/μl), suggesting that reduced platelet counts may serve as a hematological marker of co-infection. Hematological evaluation in dogs with CPV mono-infectionBlood parameters from four deceased dogs in the CPV mono-infected group were compared to the group’s mean values (n=20) to assess hematological changes associated with mortality (Table 3). Table 2. Hematological values in dogs with CPV mono-infection and CPV–CanineCV Co-infection.
Table 3. Hematological values in deceased dogs with CPV mono-infection.
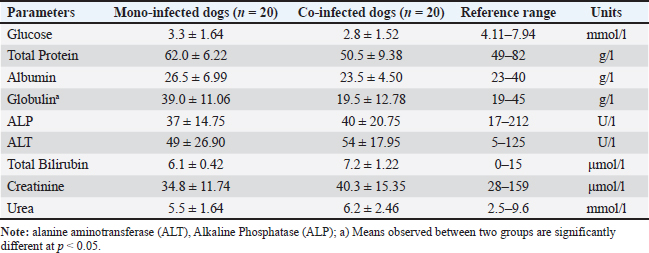
Non-survivors showed significant reductions in multiple indices. RBC counts averaged 4.3 × 106/μl, below both the group mean (5.0 × 106/μl) and the normal range (4.4–8.5 × 106/μl), indicating anemia. HCT was also reduced (26% vs. 29.5%; normal: 31%–47%). The WBC (2.5 × 10³/μl) and lymphocyte (LYM) counts (0.4 × 10³/μl) were markedly lower than the group averages and reference ranges, indicating severe immunosuppression. Platelet counts were mildly decreased (136 × 10³/μl vs. 146 × 10³/μl; normal: 200–500 × 10³/μl), consistent with fatal thrombocytopenia. Hematological evaluation in deceased dogs coinfected with CPV and CanineCVBlood parameters from eight deceased dogs with CPV and CanineCV co-infection were compared to the group average (n=20) to evaluate hematological changes linked to mortality in co-infected cases (Table 4). Non-survivors showed marked reductions in WBC (2.2 × 10³/μl vs. 2.8 × 10³/μl) and LYM counts (0.5 × 10³/μl vs. 0.7 × 10³/μl), both well below normal ranges, indicating severe immunosuppression. Platelet counts were drastically lower in deceased dogs (41 × 10³/μl vs. 63.0 × 10³/μl; normal: 200–500 × 10³/μl), suggesting a high risk of bleeding. RBC counts (4.0 × 106/μl) were mildly reduced, while HCT remained normal (43%), likely due to hemoconcentration. Compared with CPV mono-infected nonsurvivors, coinfected dogs had lower WBC and significantly lower platelet (PLT) counts, indicating more severe hematological compromise. Blood biochemical indices in mono- and co-infected dogsTo assess organ function and internal damage, blood biochemistry was analyzed in CPV mono-infected (n=20) and CPV–CanineCV co-infected dogs (n=20), with results compared to reference ranges (Alvarez and Whittemore, 2009). Co-infected dogs exhibited more pronounced biochemical abnormalities (Table 5). Glucose levels were lower in both groups, with coinfected dogs being more severely hypoglycemic (2.8 vs. 3.3 mmol/l). Total protein, albumin, and globulin levels were reduced in co-infected dogs, with markedly lower globulin levels (19.5 vs. 39.0 g/l), demonstrating suppressed humoral immunity. Liver enzymes [alkaline phosphatase (ALP), alanine transaminase (ALT)] and bilirubin levels were mildly elevated in coinfected dogs, indicating potential subclinical hepatic involvement. The levels of renal markers (creatinine and urea) were slightly higher in coinfected dogs but remained within normal ranges, demonstrating that renal function was largely preserved. Table 4. Hematological values in deceased dogs co-infected with CPV and Canine CV.
Table 5. Biochemistry values in mono-infected and co-infected dogs.
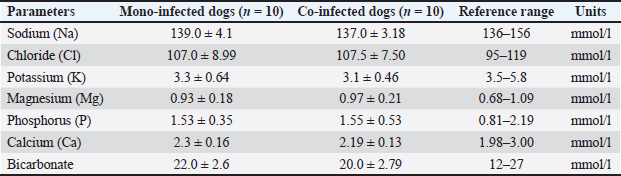
Electrolyte indices in mono- and co-infected dogsThe electrolyte profiles were assessed in CPV mono-infected (n=10) and CPV–CanineCV coinfected dogs (n=10), and the results are summarized in Table 6. Table 6. Electrolyte values in mono-infected and co-infected dogs.
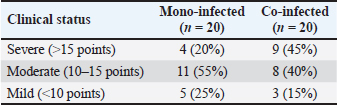
Sodium and chloride levels were within normal ranges in both groups, although sodium in co-infected dogs (137.0 mmol/l) was near the lower limit, demonstrating mild dehydration. Potassium levels were below normal in both groups, indicating hypokalemia, with co-infected dogs showing a greater deficit (3.1 vs. 3.3 mmol/l), likely due to gastrointestinal losses. The levels of magnesium, phosphorus, and calcium remained within normal limits, with minimal variation between the groups. Bicarbonate levels were also normal but slightly lower in coinfected dogs (20.0 vs. 22.0 mmol/l), demonstrating a possible trend toward mild metabolic acidosis. Clinical status at admission of the dog groupsClinical evaluations were performed on 40 dogs at admission, with classification into three severity categories based on clinical index scores: severe (>15 points), moderate (10–15 points), and mild (<10 points). The distribution of clinical severity between mono-infected (CPV only) and coinfected (CPV and CIV) groups is presented in Table 7. Table 7. Clinical status at admission of dog groups.
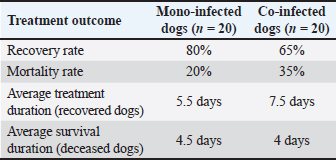
In the mono-infected group (n=20), 20% (4/20) of dogs were classified as severe, 55% (11/20) as moderate, and 25% (5/20) as mild. In contrast, the co-infected group (n=20) exhibited a significantly higher proportion of severe cases at 45% (9/20), moderate cases at 40% (8/20), and mild cases at 15% (3/20). Treatment response of the mono- and co-infected groupsTreatment outcomes for dogs infected solely with CPV (mono-infected) and those co-infected with CPV and CIV are summarized in Table 8. Both groups received the same therapeutic regimen. Table 8. Treatment response of mono-infected and co-infected groups.
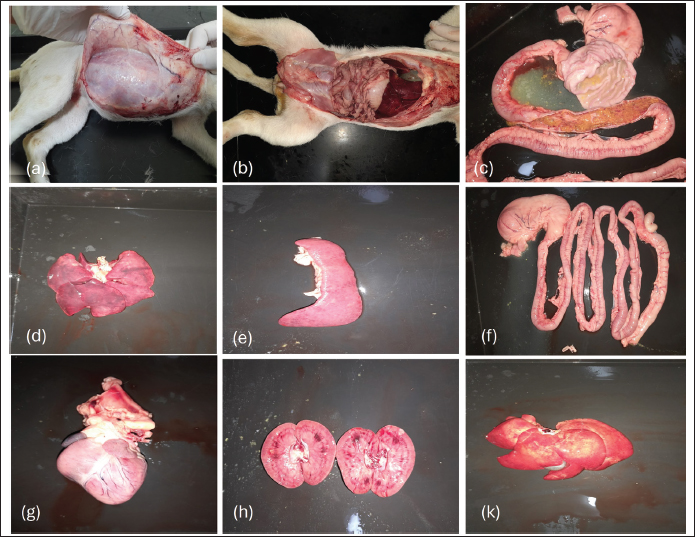
The recovery rate was higher in the mono-infected group at 80% (16/20) than in the co-infected group at 65% (13/20), indicating a more favorable response to treatment in dogs infected with CPV alone. Conversely, the mortality rate was higher in the co-infected group (35%, 7/20) than in the mono-infected group (20%, 4/20), reflecting the increased clinical severity associated with co-infection. Recovered dogs in the co-infected group required a longer average treatment duration of 7.5 days, compared to 5.5 days in the mono-infected group, suggesting a more protracted and complicated clinical course due to the combined viral effects. The average survival time among deceased dogs was marginally shorter in the co-infected group (4 days) than in the mono-infected group (4.5 days), although this difference was not statistically significant. Gross lesions in dogs with diarrheal enteritis co-infected with parvovirus and circovirusNecropsy examinations were conducted on two deceased dogs: one with CPV mono-infection and one with CPV and CanineCV coinfection. Gross pathological changes in various organs were evaluated to compare the lesions between the two groups (Fig. 2).
Fig. 2. Gross lesions in dogs with diarrheal enteritis co-infected with parvovirus and circovirus. In both cases (a, b), the small intestines exhibited severe inflammation, marked by pronounced congestion and scattered petechial hemorrhages on the serosal surface (c, f). Mild edema was noted in the liver and spleen, while the heart and kidneys exhibited focal degenerative changes (d, e, g, h, k). Severe small intestinal inflammation characterized by edematous mucosa, pronounced congestion, and scattered petechial hemorrhages on the intestinal surface were observed in both cases. The mucosal layer exhibited deep ulcerations with extensive necrosis, resulting in the loss of normal mucosal architecture. Notably, the coinfected dog exhibited more extensive lesions, including larger areas of necrosis with widespread edema and hemorrhage, surpassing the severity observed in the mono-infected dog. In both cases, the mesenteric lymph nodes were markedly enlarged and darkened, consistent with congestion and inflammatory response. Mild edema was also observed in the liver and spleen, whereas focal degenerative changes or mild edema were present in the kidneys. DiscussionTo the best of our knowledge, this is the first study to investigate the clinical, hematological, biochemical, electrolyte, and pathological features of co-infection with CPV and CanineCV in dogs in Hanoi. This comprehensive analysis provides new insights into the clinical importance of CanineCV in intensifying enteric disease in dogs already infected with CPV. This study reaffirms that CPV remains the predominant causative agent of diarrheal enteritis in the examined canine population, with an exceptionally high infection rate of 94.68%. Co-infection with CanineCV was identified in 21.28% of CPV-positive cases—a notably higher prevalence than that previously reported in similar studies conducted in Hanoi (Tuong et al., 2021). Although the pathogenesis of CanineCV-associated disease is not yet fully understood, growing evidence suggests that CanineCV may act synergistically with other pathogens, thereby exacerbating disease severity (Thaiwong et al., 2016; Zaccaria et al., 2016; Kotsias et al., 2019; Kotsias et al., 2019). The findings of this study support this hypothesis, as CanineCV was consistently co-detected with CPV-2c in dogs exhibiting clinical signs of enteritis, aligning with prior reports (Giraldo-Ramirez et al., 2020). This pattern of co-infection is particularly significant given that CPV-2c has been recognized as the predominant genotype circulating in Vietnam (Hoang et al., 2019; Nguyen Manh et al., 2021), consistent with our observations. Our data indicate that coinfection with CanineCV significantly worsens clinical outcomes. The mortality rate among coinfected dogs (35%) was nearly double that of those infected with CPV alone (20%). This underscores the potential role of CanineCV as a negative cofactor that amplifies CPV pathogenesis, likely through immunosuppressive mechanisms. Although CanineCV was not detected alone in our study, these findings align with earlier reports (Anderson et al., 2017) suggesting that CanineCV can act synergistically with CPV and may function as a primary pathogen in some cases. Although we cannot confirm single CanineCV infections in our dataset, the increasing recognition of its potential clinical relevance warrants further investigation in both diagnostic protocols and disease management strategies. Despite the relatively high prevalence of coinfection, the initial clinical signs in CPV mono-infected and CPV–CanineCV coinfected dogs were largely indistinguishable. Diarrhea, vomiting, anorexia, lethargy, and dehydration were universally observed, with 100% of cases presenting with diarrhea. This symptomatic overlap underscores the diagnostic limitations of clinical examination alone and highlights the need for molecular diagnostic tools to differentiate co-infections, which may otherwise be underestimated or mismanaged. Nevertheless, marked differences in clinical severity and disease outcomes were observed between the two groups. Co-infected dogs presented with significantly more severe illness at admission and experienced poorer treatment responses, including longer recovery times, higher mortality rates, and more frequent progression to critical conditions. These findings support the hypothesis that CanineCV acts synergistically with CPV, likely by contributing to immune dysfunction and worsening gastrointestinal pathology (Goddard and Leisewitz, 2010; Tuong et al., 2021). Its role in immunosuppression may compromise host defenses, thereby facilitating more aggressive CPV replication and tissue damage. The hematological findings further support this hypothesis. Dogs co-infected with CPV and CanineCV displayed more profound leukopenia, lymphopenia, and thrombocytopenia—particularly pronounced in deceased individuals, suggesting that co-infection significantly disrupts hematopoietic and immune functions (Dowgier et al., 2017; Faraji et al., 2022). The thrombocytopenia observed in co-infected dogs in this study is consistent with findings from other viral co-infections, such as CPV combined with immunosuppressive viruses, which can intensify hematological abnormalities. Severe thrombocytopenia, which may lead to increased bleeding tendencies, is of particular concern and may serve as a potential prognostic indicator of disease severity (Goddard et al., 2008; Iris et al., 2010; Mazzaferro, 2020). CanineCV may contribute to immunosuppression through several mechanisms, including interference with lymphocyte proliferation and function, as well as modulation of host immune responses, potentially increasing susceptibility to secondary infections and worsening clinical outcomes. Co-infected dogs exhibited more pronounced hypoalbuminemia, hypoproteinemia, and hypoglycemia, likely to reflect more severe gastrointestinal malabsorption, nutritional compromise, and hepatic dysfunction. Mild elevations in liver enzymes (ALT and ALP) and bilirubin levels indicate hepatic involvement, possibly due to systemic inflammatory responses or direct viral insult. Renal function markers remained within normal ranges, indicating that kidney function was largely preserved during the acute disease phase. Electrolyte analysis revealed that hypokalemia was common in both groups, but more severe in dogs co-infected with the disease. This disturbance likely resulted from prolonged GI fluid loss, particularly vomiting and diarrhea (Mylonakis et al., 2016). The observed mild decrease in bicarbonate levels in coinfected cases indicates the potential for metabolic acidosis, which is consistent with severe dehydration and systemic compromise (Mazzaferro, 2020). Gross pathological findings aligned with clinical and laboratory observations, revealing that coinfected dogs had more extensive intestinal necrosis, mucosal hemorrhage, and systemic organ involvement. Enlargement of mesenteric lymph nodes and mild pathological changes in the liver, spleen, and kidneys further confirmed the systemic impact of co-infection, likely mediated by immune suppression and widespread inflammatory damage. The sample size in this study was limited (n=89 CPV-positive, n=19 co-infected), which may affect the generalizability of the findings. Future multicenter studies with larger cohorts are warranted to validate these results and provide a more comprehensive understanding of CPV–CanineCV coinfections in the canine population. Although this study focused on molecular detection, the absence of histopathological and immunohistochemical evaluation represents a limitation. Such analyses would allow confirmation of lesions associated with CPV and CanineCV and direct visualization of viral antigen distribution in intestinal and lymphoid tissues. Future studies incorporating these methods are essential to strengthen causal inference regarding viral pathogenicity. Taken together, these findings emphasize that co-infection with CanineCV significantly worsens the course of CPV infection, leading to more severe immune suppression, greater tissue damage, and poorer treatment outcomes. The inability to distinguish coinfection from CPV mono-infection based on clinical signs alone reinforces the need for early molecular screening to identify high-risk cases. Early diagnosis would allow for the implementation of more intensive supportive care, improved fluid-electrolyte management, and the use of immunomodulatory therapies in severely affected dogs. This study provides foundational evidence for CanineCV’s exacerbating role in CPV-associated enteritis. Thrombocytopenia has emerged as a potential biomarker for disease severity and warrants further exploration in larger, prospective studies. There is also a pressing need to investigate the pathogenic mechanisms of CanineCV, especially its role in immune modulation and hematopoietic suppression. ConclusionIn summary, our findings indicate that CPV–CanineCV coinfections occur in clinical cases of enteritis; however, co-pathogenicity was not established. The observations highlight the need for increased diagnostic attention to coinfections and support further investigations to clarify the biological interactions and potential clinical impact of CPV and CanineCV. AcknowledgmentsWe would like to thank the owner of the dog for their permission and cooperation in providing samples and allowing the necropsy. Conflict of interestThe authors have no competing interests to declare. FundingThis research did not receive any specific grant from public, commercial, or not-for-profit funding agencies. Authors’ contributionsM.T.N. and T.T.N. collected the data, performed statistical analysis, and drafted the manuscript. T.T.N., T.L.H.P., M.T.N., and H.T.T.N. conceived and participated in the study design, wrote the manuscript, and reviewed the manuscript. All authors have read and approved the submitted version. Data availabilityData will be made available on reasonable request. ReferencesAlvarez, L. and Whittemore, J. 2009. Liver enzyme elevations in dogs: a diagnostic approach. Compendium (Yardley. PA). 31(9), 416–418. Anderson, A., Hartmann, K., Leutenegger, C.M., Proksch, A.L., Mueller, R.S. and Unterer, S. 2017. Role of canine circovirus in dogs with acute hemorrhagic diarrhea. Vet. Rec. 180(22), 542; doi:10.1136/vr.103926 Balboni, A., Terrusi, A., Urbani, L., Troia, R., Stefanelli, S.A.M., Giunti, M. and Battilani, M. 2022. Canine circovirus and canine adenovirus types 1 and 2 in dogs with parvovirus-induced enteritis. Vet. Res. Commun. 46(1), 223–232; doi:10.1007/s11259-021-09850-y Buonavoglia, D., Cavalli, A., Pratelli, A., Martella, V., Greco, G., Tempesta, M. and Buonavoglia, C. 2000. Antigenic analysis of isolated canine parvovirus strains in Italy. New Microbiol. 23(1), 93–96. Charoenkul, K., Tangwangvivat, R., Janetanakit, T., Boonyapisitsopa, S., Bunpapong, N., Chaiyawong, S. and Amonsin, A. 2019. Emergence of canine parvovirus type 2c in Thai domestic dogs and cats. Transbound. Emerg. Dis. 66(4), 1518–1528; doi:10.1111/tbed.13177 Chiang, S.Y., Wu, H.Y., Chiou, M.T., Chang, M.C. and Lin, C.N. 2016. Identification of a novel type 2c canine parvovirus in Taiwan. Virol. J. 13(1), 160; doi:10.1186/s12985-016-0620-5 Decaro, N. and Buonavoglia, C. 2012. Canine parvovirus: a review of epidemiological and diagnostic aspects, with an emphasis on type 2c. Vet. Microbiol. 155(1), 1–12; doi:10.1016/j.vetmic.2011.09.007 Dowgier, G., Lorusso, E., Decaro, N., Desario, C., Mari, V., Lucente, M.S., Lanave, G., Buonavoglia, C. and Elia, G. 2017. A molecular survey of selected viral enteropathogens revealed that canine circovirus has a limited role in the development of canine acute gastroenteritis. Vet. Microbiol. 204, 54–58; doi:10.1016/j.vetmic.2017.04.007 Faraji, R., Sadeghi, M., Mozhgani, S.H., Vasinioti, V., Ndiana, L.A., Desario, C., Beikpour, F. and Decaro, N. 2022. Canine circovirus detection in dogs infected with canine parvovirus. Acta Tropica 235, 106646; doi: 10.1016/j.actatropica.2022.106646 Geng, Y., Guo, D., Li, C., Wang, E., Wei, S., Wang, Z., Yao, S., Zhao, X., Su, M., Wang, X., Wang, J., Wu, R., Feng, L. and Sun, D. 2015. Co-circulation of the rare CPV-2c with unique Gln370Arg substitution, new CPV-2b with unique Thr440Ala substitution, and new CPV-2a with high prevalence and variation in Heilongjiang Province, Northeast China. PLos One. 10(9), 137288; doi:10.1371/journal.pone.0137288 Gibbs, S.E. and Gibbs, E.P. 2013. The historical, present, and future roles of veterinarians in One Health. Curr. Top. Microbiol. Immunol. 365, 31–47; doi:10.1007/82_2012_259 Giraldo-Ramirez S Rendon., -Marin DS Vargas., -Bermudez J Jaime. and Ruiz-Saenz, J. 2020. First detection and full genomic analysis of canine circovirus in dogs infected with CPV-2 in Colombia, South America. Scientific. Rep. 10(1), 17579; doi:10.1038/s41598-020-74630-8 Goddard, A. and Leisewitz, A.L. 2010. Canine parvovirus. Vet. Clinics North Amer. Small Anim. Pract. 40(6), 1041–1053; doi:10.1016/j.cvsm.2010.07.007 Goddard, A., Leisewitz, A.L., Christopher, M.M., Duncan, N.M. and Becker, P.J. 2008. Prognostic usefulness of blood leukocyte changes in canine parvoviral enteritis. J. Vet. Internal Med. 22(2), 309–316; doi:10.1111/j.1939-1676.2008.0073.x Hoang, M., Lin, W.H., Le, V.P., Nga, B.T.T., Chiou, M.T. and Lin, C.N. 2019. Molecular epidemiology of type 2 canine parvovirus in Vietnam from November 2016 to February 2018. Virol. J. 16(1), 52; doi:10.1186/s12985-019-1159-z Iris, K., Leontides, L.S., Mylonakis, M.E., Adamama-Moraitou, K., Rallis, T. and Koutinas, A.F. 2010. Factors affecting the occurrence, duration of hospitalization, and outcome of canine parvovirus infection. Res. Vet. Sci. 89(2), 174–178; doi:10.1016/j.rvsc.2010.02.013 Kotsias, F., Bucafusco, D., Nuñez, D.A., Lago Borisovsky, L.A., Rodriguez, M. and Bratanich, A.C. 2019. Genomic characterization of canine circovirus associated with fatal disease in dogs in South America. PLos One. 14(6), e0218735; doi:10.1371/journal.pone.0218735 Liu, Y., Qin, Y., Hu, Y., Chen, W., Han, Z., Yi, C., Bi, J., Huang, H., Li, Y., Zhang, X., Lan, T., Zheng, M. and Sun, W. 2024. Epidemiological and evolutionary analysis of canine circovirus (CCV) from 1996 to 2023. BMC Vet. Res. 20(1), 328; doi:10.1186/s12917-024-04186-6 Mazzaferro, E.M. 2020. Update on Canine Parvovirus Enteritis. Vet. Clinics. North. Amer. Small. Anim. Pract. 50(6), 1307–1325; doi:10.1016/j.cvsm.2020.07.008 McCourt, M. R., & Rizzi, T. E. (2022). Hematology of dogs. Schalm’s Veterinary Hematology. 969-982. https://doi.org/10.1002/9781119500537.ch108 Moon, B.Y., Jang, J., Kim, S.H., Kim, Y.H., Lee, H.K., So, B., Park, C.K. and Lee, K.K. 2020. Genetic characterization of canine parvovirus type 2c in domestic dogs in Korea. Transbound. Emerg. Dis. 67, 1645–1653; doi:10.1111/tbed.13501 Mylonakis, M.E., Kalli, I. and Rallis, T.S. 2016. Canine parvoviral enteritis: clinical diagnosis, treatment, and prevention. Vet. Med. (Auckland. NZ). 7, 91–100; doi:10.2147/vmrr.s80971 Nakamura, M., Tohya, Y., Miyazawa, T., Mochizuki, M., Phung, H.T., Nguyen, N.H., Huynh, L.M., Nguyen, L.T., Nguyen, P.N., Nguyen, P.V., Nguyen, N.P. and Akashi, H. 2004. Novel antigenic variant of canine parvovirus isolated from a Vietnamese dog. Arch. Virol. 149(11), 2261–2269; doi:10.1007/s00705-004-0367-y Nandi, S., Chidri, S., Kumar, M. and Chauhan, R.S. 2010. Occurrence of canine parvovirus type 2c in the dogs with hemorrhagic enteritis in India: a retrospective study. Res. Vet. Sci. 88(1), 169–171; doi:10.1016/j.rvsc.2009.05.018 Nguyen Manh, T., Piewbang, C., Rungsipipat, A. and Techangamsuwan, S. 2021. Molecular and phylogenetic analysis of Vietnamese canine parvovirus 2C originating from dogs reveals a new Asia-IV clade. Transboundary Emerg. Dis. 68(3), 1445–1453; doi:10.1111/tbed.13811 Segalés, J., Allan, G.M. and Domingo, M. 2005. Diseases caused by porcine circovirus. Anim. Health. Res. Rev. 6(2), 119–142; doi:10.1079/ahr2005106:10.1079/ahr2005106 Thaiwong, T., Wise, A.G., Maes, R.K., Mullaney, T. and Kiupel, M. 2016. Canine Circovirus 1 (CaCV-1) and Canine Parvovirus 2 (CPV-2): recurrent Dual Infections in a Papillon Breeding Colony. Vet. Pathol. 53(6), 1204–1209; doi: 10.1177/0300985816646430 Tuong, N.M., Piewbang, C., Rungsipipat, A. and Techangamsuwan, S. 2021. Detection and molecular characterization of two co-circulating canine circovirus genotypes in Vietnam. Vet. Q. 41(1), 232–241; doi:10.1080/01652176.2021.1967511 Vannamahaxay, S., Vongkhamchanh, S., Intanon, M., Tangtrongsup, S., Tiwananthagorn, S., Pringproa, K. and Chuammitri, P. 2017. Molecular characterization of canine parvovirus in Laos. Arch. Virol. 162(5), 1355–1361; doi:10.1007/s00705-016-3212-1 Zaccaria, G., Malatesta, D., Scipioni, G., Di Felice, E., Campolo, M., Casaccia, C., Savini, G., Di Sabatino, D. and Lorusso, A. 2016. Circovirus in domestic and wild carnivores: an important opportunistic agent?. Virology 490, 69–74; doi:10.1016/j.virol.2016.01.007 | ||
| How to Cite this Article |
| Pubmed Style Nguyen MT, Pham TLH, Nguyen HTT, Nguyen TT. Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Vet. J.. 2026; 16(2): 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 Web Style Nguyen MT, Pham TLH, Nguyen HTT, Nguyen TT. Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. https://www.openveterinaryjournal.com/?mno=285776 [Access: February 27, 2026]. doi:10.5455/OVJ.2026.v16.i2.27 AMA (American Medical Association) Style Nguyen MT, Pham TLH, Nguyen HTT, Nguyen TT. Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Vet. J.. 2026; 16(2): 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 Vancouver/ICMJE Style Nguyen MT, Pham TLH, Nguyen HTT, Nguyen TT. Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Vet. J.. (2026), [cited February 27, 2026]; 16(2): 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 Harvard Style Nguyen, M. T., Pham, . T. L. H., Nguyen, . H. T. T. & Nguyen, . T. T. (2026) Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Vet. J., 16 (2), 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 Turabian Style Nguyen, Manh Tuong, Thi Lan Huong Pham, Ha Thi Thanh Nguyen, and Thanh Trung Nguyen. 2026. Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Veterinary Journal, 16 (2), 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 Chicago Style Nguyen, Manh Tuong, Thi Lan Huong Pham, Ha Thi Thanh Nguyen, and Thanh Trung Nguyen. "Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam." Open Veterinary Journal 16 (2026), 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 MLA (The Modern Language Association) Style Nguyen, Manh Tuong, Thi Lan Huong Pham, Ha Thi Thanh Nguyen, and Thanh Trung Nguyen. "Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam." Open Veterinary Journal 16.2 (2026), 1072-1081. Print. doi:10.5455/OVJ.2026.v16.i2.27 APA (American Psychological Association) Style Nguyen, M. T., Pham, . T. L. H., Nguyen, . H. T. T. & Nguyen, . T. T. (2026) Clinicopathological profiling of diarrheic canine parvovirus–circovirus co-infections in Hanoi, Vietnam. Open Veterinary Journal, 16 (2), 1072-1081. doi:10.5455/OVJ.2026.v16.i2.27 |