| Research Article | ||
Open Vet. J.. 2026; 16(3): 1661-1677 Open Veterinary Journal, (2026), Vol. 16(3): 1661-1677 Research Article Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in ratsSohaila G. Fahmy1, Zeinab Al-Amgad2, Ibrahim F. Rehan3,4,5*, Fatma A. Mahmoud6, Rawia M. Ibrahim7<, František Zigo8, Silvia Ondrašovičová9 and Motamed E. Mahmoud11Department of Animal Husbandry and Behavior, Faculty of Veterinary Medicine, Sohag University, Sohag, Egypt 2Medical Technical College, Al-Farahidi University, Baghdad, Iraq 3Department of Animal Behavior and Husbandry, Faculty of Veterinary Medicine, Menoufia University, Menoufia, Egypt 4Department of Pathobiochemistry, Faculty of Pharmacy, Meijo University Yagotoyama, Nagoya-shi, Japan 5Institute of Genetics and Animal Biotechnology, Polish Academy of Sciences, Warsaw, Poland 6Department of Animal, Poultry and Wildlife Behavior and Management, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt 7Clinical Laboratory Diagnosis, Department of Internal Medicine, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt 8Department of Animal Nutrition and Husbandry, University of Veterinary Medicine and Pharmacy, Košice, Slovakia 9Department of Biology and Physiology, University of Veterinary Medicine and Pharmacy, Košice, Slovakia *Corresponding Author: Ibrahim F. Rehan. Department of Animal Behavior and Husbandry, Faculty of Veterinary Medicine, Menoufia University, Menoufia, Egypt. Email: ibrahim.rehan [at] vet.menofia.edu.eg Submitted: 21/09/2025 ;Revised: 31/01/2026 Accepted: 15/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
AbstractBackground: Dexamethasone, a synthetic analogue of betamethasone, exerts vast actions to mediate anti-inflammatory and immunosuppressive effects. Despite failing to exhibit obvious adverse involvements during its protective therapy, the unique role of dexamethasone affects an array of organs implicated in preliminary histopathological findings. Aim: This prospective study aimed to verify the alterations in the liver, brain, kidney, and spleen of pregnant rats treated with low-versus high-dexamethasone doses. Methods: Overall, 72 Wistar rats weighing 250 ± 20 g were divided into the control pregnant group, which was subdivided into two subgroups (14 and 21 days); the pregnant group, which was ultimately subdivided into two groups and then orally administered diluted concentrations of dexamethasone at 625 and 125 × 10 µg/kg body weight (BW); each of these concentrations was subdivided into two subgroups (14 and 21 days); the control non-pregnant group, which was subdivided into two subgroups (14 and 21 days); and the non-pregnant group, which was sub grouped into two groups, was orally administered the same mentioned concentrations of dexamethasone. Results: Histological sections from the liver, brain, kidney, and spleen were further stained with hematoxylin and eosin and then examined under a microscope to evaluate tissue integrity and histopathological lesions. Dexamethasone exhibited significant adverse effects on total organ weights. Significant histopathological variations in dexamethasone-treated tissues. Therefore, liver changes included hepatocyte necrosis and interstitial inflammation. Structural brain changes involving neuronal necrosis and perineuronal vacuolations. Despite kidney lesions, which included dilated tubules and necrosis with sloughing of the renal epithelium, the spleen exhibited lymphoid follicle depletion and hemorrhage of red pulp. Strong, unlimited alterations (+++) of the tissues were observed in the group that received 125 × 10 µg/kg BW dexamethasone. Additionally, the lowest concentration group, which received 625 µg/kg BW dexamethasone, showed non-negligible changes (++) compared with the controls. Conclusion: These findings suggest that the harmful effects of dexamethasone on organ architecture are dose-dependent. Therefore, dexamethasone is not decidedly contraindicated during pregnancy, but strict consideration and incessant monitoring are required during application, irrespective of the healthcare supplier. Keywords: Dexamethasone, Histopathology, Lesions, Pregnancy, Rats. IntroductionGCs are widely used in pharmacological practices. In fact, glucocorticoid drugs are potent medicines that can treat inflammation and several health problems. Unfortunately, substantial evidence exists that corticosteroid therapy can induce severe adverse effects on the structures of different organs (Hammadi et al., 2022). Hence, numerous clinical studies have been conducted to evaluate the severity of glucocorticoid drug side effects on various organs (Gloor et al., 2001). Dexamethasone is among the most popular synthetic glucocorticoids used with multiple pharmacologic properties, such as immunomodulatory and anti-inflammatory properties (Provan et al., 2010). Although dexamethasone is generally well-sustained, it is closely associated with the development of a broad range of metabolic diseases (Mohan et al., 2013). Prolonged dexamethasone therapy has been associated with pronounced harmful effects. Earlier studies have confirmed the harmful effects of dexamethasone on several body systems, such as the effects on rat body weights (Wafula et al., 2005). Dexamethasone is a disadvantageous medication attributed to an increased tendency to several diseases (Mesripour et al., 2021). Low and high exposure doses of dexamethasone induce abortion in pregnant females (Zone et al., 1995), in addition to pathological changes in the organ structures of the liver and kidney of female and male mice (Kathim and Al-Hillo, 2023). The liver is a vital organ that converts medications into forms that can be easily eliminated from the body. Hepatic toxicity occurs by the production of toxic metabolites and the covalent combining of the drugs to liver proteins (Mohan and Sharma, 2017). In the same context, the brain is the leading center of the nervous system. It primarily controls language, motor function, and other cognitive functions (Cuffari, 2020). The kidney is an essential target site for the effects of drugs in both animals and humans (Oaks et al., 2004). On the other hand, the spleen is a crucial organ for immunological responses, which support immunity and blood filtration (Lewis et al., 2019). Dexamethasone, like other glucocorticoids, can trigger oxidative stress processes and lipid peroxidation. In addition, dexamethasone is known to cause hepatotoxicity and liver damage (Yavroyan et al., 2025). Recently, the neurological adverse implications have been detected in individuals who received dexamethasone treatment (Marshall and Morriss-Kay, 2004). Kidney damage is a consequence of early-life overexposure, which impairs kidney development and decreases the number of glomeruli, which may contribute to hypertension in delayed life (Figueroa et al., 2005). Dexamethasone is used experimentally to induce immunosuppression (Jeklova et al., 2008). Immunosuppression, whether due to pharmacological treatments or infection, can exert significant alterations in splenic architecture (Vidya Vijayan et al., 2017). In rat models, dexamethasone has been shown to diminish spleen weights and sizes via the induction of lymphoid atrophy and lymphocytic apoptosis (Dehghani et al., 2021). This prospective study aimed to verify the alterations in the most leading organs of the body, such as the liver, brain, kidney, and spleen, of pregnant female rats treated with variable concentrations of dexamethasone, focusing on the histological aspect and organ weight profile. Therefore, this study aimed to model the potential maternal and fetal effects of repeated exposure during gestation. Materials and MethodsExperimental animalsThe current procedure was conducted at the Laboratory Animals Unit of Sohag University, Egypt. Pregnant female Wistar rats, within 4 months old, on the seventh day of gestation, were purchased from the Experimental Animals House. Therefore, this work was set in accordance with the Animal Ethics Committee of Sohag University with a code number: 2020, 1019 Dec 01. These rats, weighing around 250 ± 20 g, were reared in a suitable environment at 25◦C ± 2◦C surrounding temperature, 12:12 hours dark/light cycle, and 40%–50% humidity. The rats were continuously served a standard commercial diet and fresh water. Dexamethasone dosing solutionTo determine the ideal doses of dexamethasone for the survival of rats, the Wistar rats were administered different dilutions of dexamethasone as follows: 625 (Benediktsson et al., 1993; Ortiz et al., 2001; Costa-Silva et al., (2007)), 125 × 10 µg/kg body weight (BW) (Manojlivić et al., 1998). Dexamethasone is administered orally via drinking water via a syringe to ensure continuous and non-invasive delivery. Dexamethasone was selected because it is a clinically relevant synthetic glucocorticoid that is frequently administered during pregnancy to promote fetal organ maturation and manage maternal conditions. Animal groupingExperimental groups included 72 female Wistar rats exactly aged 4 months under random classification to the four principal groups, which were illustrated as follows; first) control pregnant animals (n=12) which subdivided into two subgroups; A1 after 14 days (n=6) and A2 after 21 days (n=6); second) pregnant animals received 625 (B1) & 125 × 10 (B2) µg dexamethasone/kg BW, expressing two sub-groups (n=12 for each) each concentration subdivided into two subgroups (b114 and b214) after 14 days and (b121), and (b221) after 21 days (n=6 for each); third) control non-pregnant animals (n=12) which subdivided into two subgroups; (C114) after 14 days (n=6) and (C121) after 21 days (n=6); and fourth) non-pregnant animals received (D1) 625 & (D2) 125 × 10 µg dexamethasone/kg BW, expressing two sub-groups (n=12 for each) each concentration subdivided into two subgroups (D114 and D214) after 14 days and (D121 and D221) after 21 days (n=6 for each). The experimental study is illustrated in Figure 1.
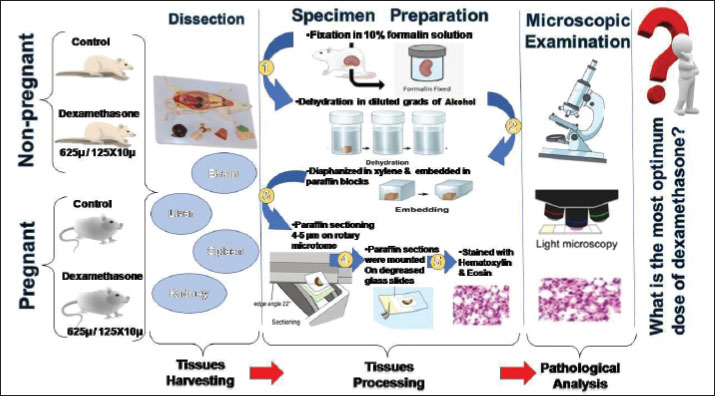
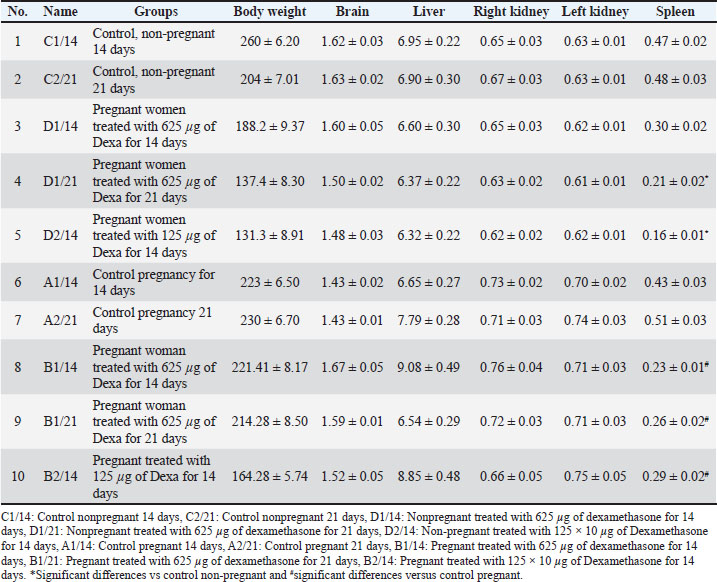
Fig. 1. Schematic of the experimental study. EuthanasiaAfter the experiment was completed, rats were euthanized under humane killing through cervical dislocation (Cheng, 2021) for tissue collection and sampling to be processed for histopathological analyses. Tissues harvestingThe abdominal region is excised precisely via a midline approach after 14 or 21 days of pregnancy (depending on the number of days planned for each group) to extract the liver, spleen, and kidney. However, the brain was revealed through midsagittal skull surgery. The tissues of each rat were immediately removed; their weights were recorded and then trimmed into tiny fragments not exceeding 1 cm in diameter for soaking in the buffered 10% formaldehyde for histopathological approaches. Tissues processingFor full histological visualization, liver, spleen, kidney, and brain specimens were perfectly fixed in 10% formalin solution and subjected to the entire histological procedure through dehydration in diluted grades of ethanol after washing with tap water, diaphanized in xylene, and embedded in paraffin blocks. Paraffin sections were created using a rotary microtome for thin slices of 4–5 μm thickness. Sections were carefully placed on standard laboratory slides for subsequent staining. The sequential slides were stained with hematoxylin and eosin to visualize the histological lesions using digital morphometric monitoring through a Leitz Dialux 20 analysis microscope (Bancroft and Gamble, 2002). Histopathological scoringThe histological data of the liver, brain, kidney, and spleen-stained sections were displayed as follows: no lesions (), few (+), and moderate (++), while (+++) indicated strong lesions. A semi-quantitative scoring assay was employed to differentiate the severity and degree of histological changes based on Gibson-Corley et al. (2013). Statistical analysisThe SPSS software was used for data analysis to identify the effect of dexamethasone in each group on the body weight parameter expressed by Mean values ± SEM. However. Dunnett’s post hoc test after one-way analysis of variance was performed to compare the significance of the means between groups. A minimum of significance was set at p ≤ 0.05. Ethical approvalThe study was performed according to the Animal Ethics Committee regulations at the Faculty of Veterinary Medicine, Sohag University, Egypt it is (Approval number; 2020, 1019 Dec 01). ResultsContrariwise to the other induced dilutions, the ideal dose for dexamethasone therapy with no observable outcomes was 625 μg/kg BW for 7 and 14 days, followed by 125 × 10 μg/kg BW for 14 days. Low-dose dexamethasone (625 μg/kg BW) for both 14 and 21 days was patent associated with moderate alterations compared with high-dose dexamethasone (125 × 10 µg). Therefore, in this, the deteriorations in the organs’ weights and their histological architectures pointed in by the two chosen doses with a detailed discussion. Comparison of the weights of organsComparison of the liver weightAs shown in Table 1, the livers of the non-pregnant groups treated with dexamethasone, compared with the control non-pregnant group, recorded the lowest dose (625 µg) recorded the least rise in liver weight, and no significant differences in terms of statistics. In contrast to the control pregnant group, the livers of the dexamethasone-treated pregnant groups in Table 1 showed that the lowest dose (625 µg) had the least increase in liver weight, with no significant differences. Table 1. Live body weights and postmortem internal organ weights of pregnant and non-pregnant rats.
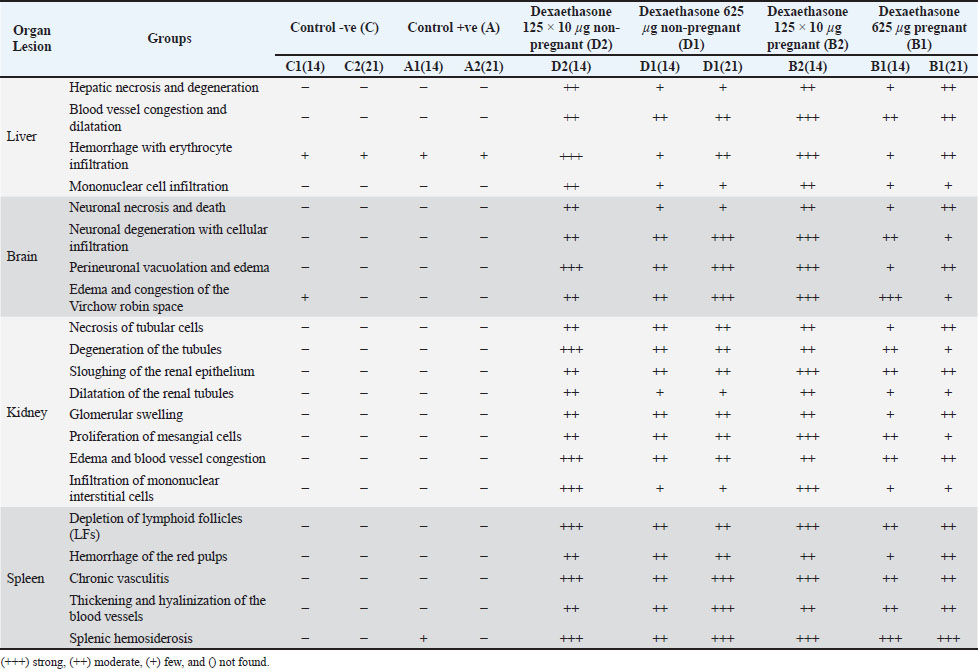
Comparison of brain weightIn contrast to the control non-pregnant group, the brains of the non-pregnant groups treated with dexamethasone in Table 1 showed that the lowest dose (625 µg) had the least decrease in brain weight, with no significant differences. The brain findings of the dexamethasone-treated pregnant groups in Table 1 showed that the lowest dosage (625 µg) had the least increase in brain weight, with no significant differences.Comparison of the kidney weightLeft kidneyIn contrast to the control non-pregnant group, the dose of 125 × 10 µg had the least amount of weight in the left kidneys of the non-pregnant groups treated with dexamethasone, according to the data given in Table 1. However, the results for the left kidney of dexamethasone treatment for pregnant groups revealed that, in contrast with the control pregnant group, the lowest dose (625 µg) had the least increase in left kidney weight and no significant difference. Right kidneyTable 1 shows that treating nonpregnant groups with a dexamethasone dose (125 × 10 µg) resulted in the least decline in right kidney weight, with no significant changes compared with the control group. Table 1 shows that the lowest dose of dexamethasone (625 µg) resulted in the least increase in right kidney weight compared with the control pregnant group. Comparison of the spleen weightTable 1 reveals that the lowest dose (625 µg) of dexamethasone resulted in the least decrease in spleen weight, with significant differences from the control non-pregnant group. Table 1 demonstrates that the dose of dexamethasone, 125 × 10 µg, led to the smallest decrease in spleen weight, differing significantly from the pregnant control group. Histopathological findingsMacroscopic resultsMacroscopic liver The leaves of the pregnant control groups A1(14) and A2(21) and the non-pregnant control groups C1(14) and C2(21) showed normal appearance within normal sizes, colors, and consistency. Pregnant animals of groups B1(14) and B1(21) received dexamethasone at 625 µg, respectively, and displayed congestion of the hepatic capsules. Pregnant animals in groups B2(14) and B2(21) received dexamethasone at 125 × 10 µg, respectively, and displayed congestion of the hepatic capsules. Non-pregnant animals in group D1(14) who received dexamethasone at 625 µg showed pinpoint areas of congestion, and those in group D1(21) who received dexamethasone at 625 µg, showed severe congestion with spots of reddish coloration. Non-pregnant animals in group D2(14) received dexamethasone at 125 × 10 µg, they appeared very congested with darkish red coloration. Macroscopic brain The brains of the pregnant control groups A1(14) and A2(21) appeared normal, and those of the non-pregnant control groups C1(14) and C2(21) appeared normal. Pregnant animals in group B1 (14) revealed darkish red spots of congested blood vessels, and those in group B1 (21) received dexamethasone at 625 µg, respectively, and exhibited pale coloration of the brain parenchyma with red spots. Pregnant animals of group B2(14) received dexamethasone at 125 × 10 µg, respectively, and revealed darkish red spots of congested blood vessels, and B2(21) received dexamethasone at 125 × 10 µg, respectively, and exhibited pale coloration of brain parenchyma with red spots. Non-pregnant animals in group D1(14) received dexamethasone at 625 µg. Congestion with a darkish red coloration of the cerebrum and D1(21) receiving dexamethasone at 625 µg showed congestion of blood vessels. Non-pregnant animals in group D2(14) received dexamethasone at 125 × 10 µg, demonstrating darkish red spots of congested blood vessels. Macroscopic kidney The kidneys of the pregnant control groups A1(7) and A2(14) showed normal sizes, colors, and consistency, whereas those of the non-pregnant control groups C1(7) and C2(14) appeared normal. Pregnant animals in group B1 (14) displayed congestion of renal tissues, and B1(21) received dexamethasone at 625 µg, respectively, and displayed pinpoints of congestion at the renal capsules. Pregnant animals in group B2(14) received dexamethasone at 125 × 10 µg, respectively, and displayed congestion of renal tissues, and those in group B2(21) received dexamethasone at 125 × 10 µg, respectively, and displayed pinpoints of congestion at the renal capsules. In addition, nonpregnant animals in groups D1(21) and D1(21) received dexamethasone at 625 µg congestion of the cortex, and D1(21) received dexamethasone at 625 µg, showing darkish red coloration of congestion of the cortex and medulla. Non-pregnant animals in group D2(14) who received dexamethasone at 125 × 10 µg showed congestion of the renal medulla. Macroscopic spleenThe spleens of the pregnant control groups [A1(14) and A2(21)] appeared normal, whereas those of the non-pregnant control groups: C1(14) showed normal coloration and consistency, and those of C2(21) showed normal discoloration with normal texture. Pregnant animals of group B1(14) displayed intensive congestion of the spleen with darkish coloration, and those of group B1(21) who received dexamethasone at 625 µg, respectively, displayed remarkable congestion of the spleen with a darkish red coloration. Pregnant animals in group B2(14) received dexamethasone at 125 × 10 µg, respectively, and displayed intensive congestion of the spleen with darkish coloration, and those in group B2(21) received dexamethasone at 125 × 10 µg, respectively, and displayed remarkable spleen congestion with darkish red coloration. In addition, nonpregnant animals of group D1(14) received dexamethasone at 625 µg, oozed blood when cut, and group D1(21) received dexamethasone at 625 µg, and showed darkish red coloration, comprising severe congestion of splenic tissues. Non-pregnant animals of group D2(14) received dexamethasone at 125 × 10 µg, they showed darkish red coloration, comprising an intensive degree of spleen congestion. Histopathological lesionsLight microscopic screening of the examined tissues verified that dexamethasone significantly manifests destructive alterations and changes in the architecture of the liver, brain, kidney, and spleen. The histopathological scoring data ( Table 2) confirmed that dexamethasone, accompanied by marked histological damage, varies in severity. Histopathological lesions were dose- and time-dependent changes; thus, the most common lesions were cytoplasmic degeneration, cell necrosis, extensive vasocongestion, and prominent inflammation of the different target organs, comparable with those of the normal architecture of the control groups. Table 2. Score of histopathological lesions of controls and dexamethasone-exposed groups for 14 and 21 days classified according to severity into severe (+++), moderate (++), mild (+), and not observed (-).
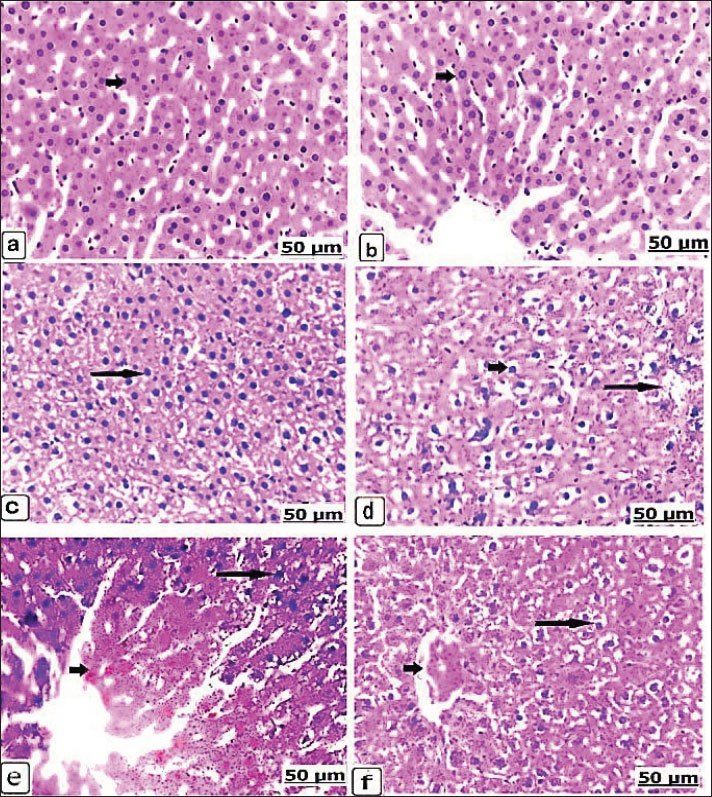
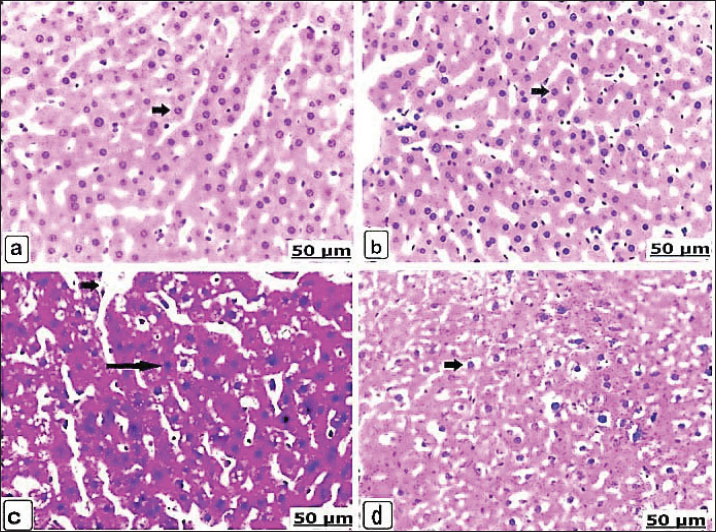
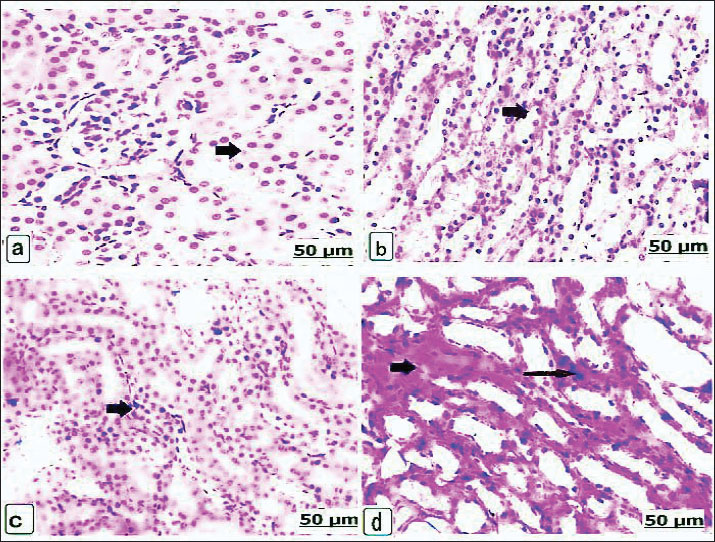
Microscopic liverAfter 14 days, the microscopic sections of the liver from the control groups of non-pregnant and pregnant rats showed normal histological arrangements of the hepatic cords enclosed with intact hepatocytes (Fig. 2a,b). The liver of the non-pregnant group received 625 µg dexamethasone, showed hypertrophy of the hepatocytes with large, condensed nuclei (Fig. 2c). Pregnant animals, while receiving the same dose, manifested necrosis and vacuolar degeneration of the hepatocytes with interstitial hemorrhage (Fig. 2d). By an increasing dexamethasone concentration to 125 × 10 µg, the histological changes become more severe; however, the non-pregnant animals recorded severe hemorrhage with red blood cell infiltration, in addition to hepatic necrosis with acidophilic cells (Fig. 2e). Therefore, pregnant animals showed vacuolar degeneration of the hepatocytes with congestion and central vein dilatation (Fig. 2f). The stained microscopic liver sections after 21 days showed normal hepatic parenchyma not only in the control non-pregnant but also in the control pregnancy (Fig. 3a,b). When the non-pregnant group, when received 625 µg of dexamethasone for 21 days, congestion with dilatation of the blood vessels, liver necrosis, and acidophilia were observed (Fig. 3c). Similarly, after the same concentration of dexamethasone was administered, pregnant rats exhibited hepatic vacuolation with scattered lesions (Fig. 3d).
Fig. 2. (a–f). Light photomicrograph of the liver of the control and experimental groups for 14 days, stained with hematoxylin and eosin. Control non-pregnant C1(14) showing normally arranged hepatocytes and intact vasculature (arrow) (a) Pregnant control A1(14) showing normally organized hepatic cords (arrow) (b). Group D1(14) received 625 μg of dexamethasone, showing hypertrophied hepatocytes with enlarged and condensed nuclei (arrow) (c). Group B1(14) received 625 μg of dexamethasone (d). Necrosis and vacuolar degeneration of the hepatocytes (short arrow) with interstitial hemorrhage (long arrow). Group D2(14) received 125 × 10 μg of dexamethasone and showed severe hemorrhage with red blood cell infiltration (short arrow) and hepatic necrosis with acidophilic cells (long arrow) (e). Group B2(14) received 125 × 10 μg of dexamethasone, showing hepatocyte vacuolar degeneration (long arrow) with congestion and central vein dilatation (short arrow) (f).
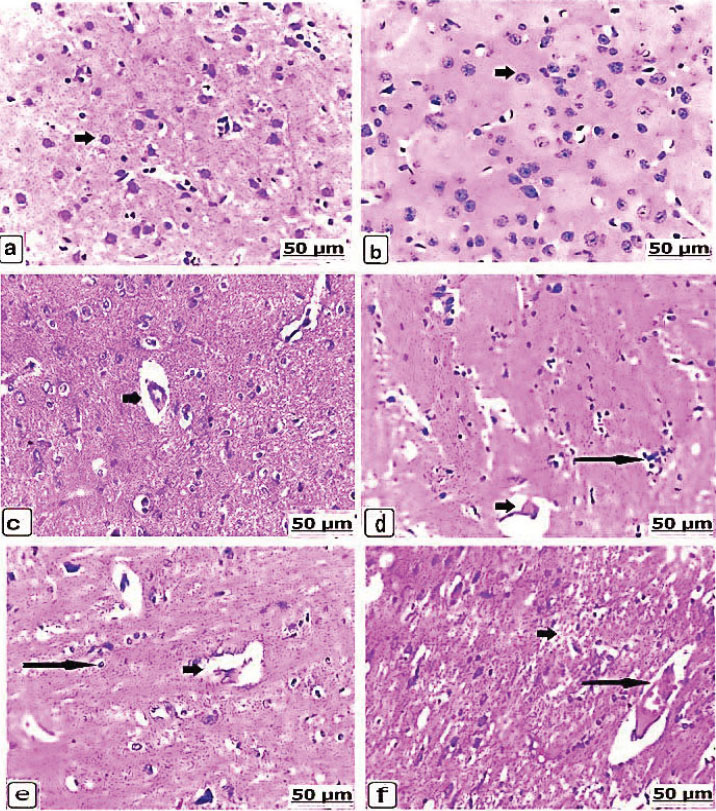
Fig. 3. (a–d). Light photomicrograph of the liver of the control and experimental groups for 21 days, stained with hematoxylin and eosin. Control non-pregnant C2(21) and control pregnant A2(21) showing normal hepatic parenchyma (a and b) (arrows). Group D1 (21) received 625 μg of dexamethasone for 21 days, showing congestion with blood vessel dilatation (short arrow) and hepatic necrosis with acidophilia (long arrow) (c). Group B1(21) received 625 μg of dexamethasone for 21 days, and scattered hepatic vacuolation was observed (arrow) (d). Microscopic brainLight sections of the control non-pregnant and pregnant groups after 14 days displayed intact neurons of the brain parenchyma, which were fundamentally normally arranged (Fig. 4a,b). The brains from the non-pregnant animals received 625 µg of dexamethasone exhibited dilated and expanded Virchow-Robin space (Fig. 4c), whereas the brain lesions of the pregnant cases were neuronal necrosis with focal gliosis, in addition to clear expansion and congestion of Virchow-Robin space (Fig. 4d). In the same context, the non-pregnant group after administration of 125 × 10 µg dexamethasone resulted in an extensive degree of neuronal necrosis with vacuolation and edema in Virchow-Robin space (Fig. 4e), and the pregnant animals suffered thrombotic vasculitis, demyelination, and necrosis of neurons (Fig. 4f). After 21 days, the brains of the controls from non-pregnant and pregnant rats exhibited normal architectures of the neurons (Fig. 5a,b). However, a dose of 625 µg dexamethasone administered to nonpregnant female rats for 21 days resulted in neuronal necrosis, perineuronal vacuolation, and dilatation of Virchow-Robin space (Fig. 5c). In addition to thrombotic vasculitis, the lesions of the brain were also manifested as perineuronal vacuolation of the pregnant animals (Fig. 5d).
Fig. 4. (a–f). Light photomicrographs of the brains of the control and experimental groups after 14 days of exposure, stained with H&E. Control non-pregnant C1(14) showing normally arranged neurons (arrow) (a). Control pregnant A1(14) showing normal brain tissue parenchyma (arrow) (b). Group D1(14) received 625 µg of dexamethasone, showing dilated and expanded Virchow-Robin space (arrow) (c). Group B1(14) received 625 µg of dexamethasone, showing neuronal necrosis with focal gliosis (long arrow) and clear expansion and congestion of Virchow-Robin space (short arrow) (d). Group D2(14) received 125 × 10 µg of dexamethasone, showing neuronal necrosis (long arrow) with vacuolation and edema in the Virchow-35 Robin space (short arrow) (e). Group B2(14) received 125 × 10 µg of dexamethasone, showing 36 cases of thrombotic vasculitis (long arrow) and demyelination and necrosis of neurons (short arrow) (f).
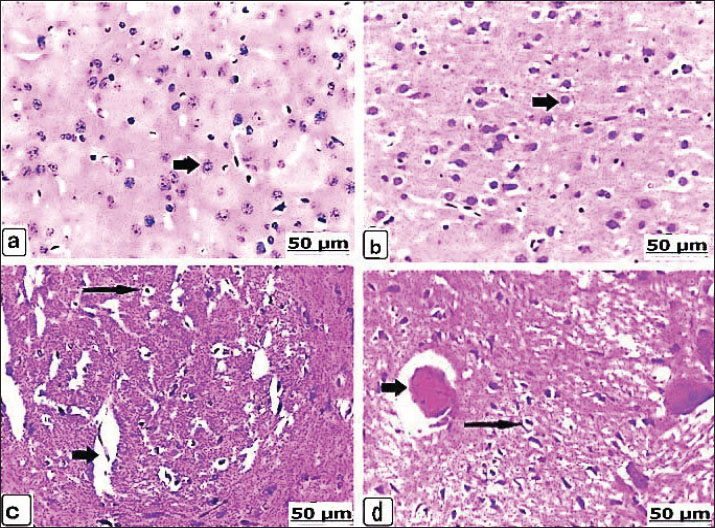
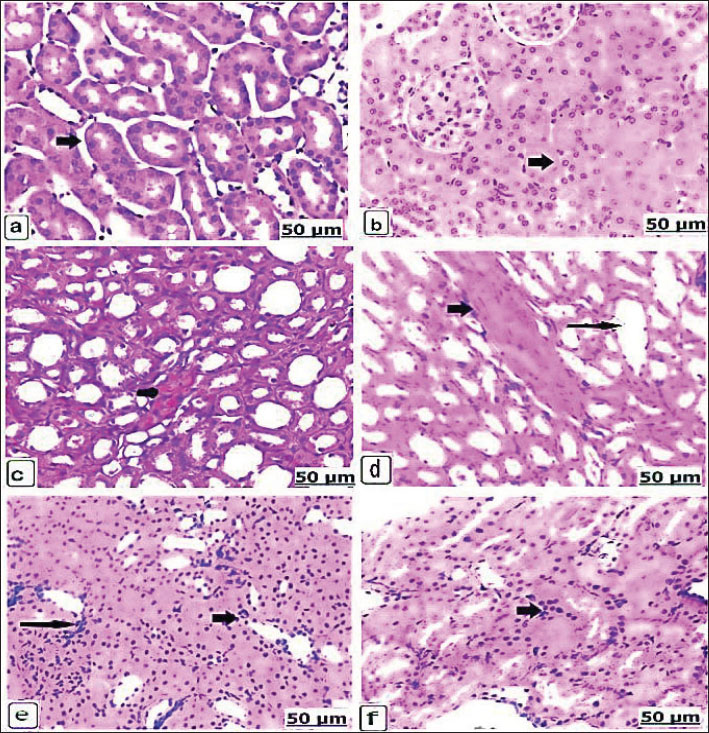
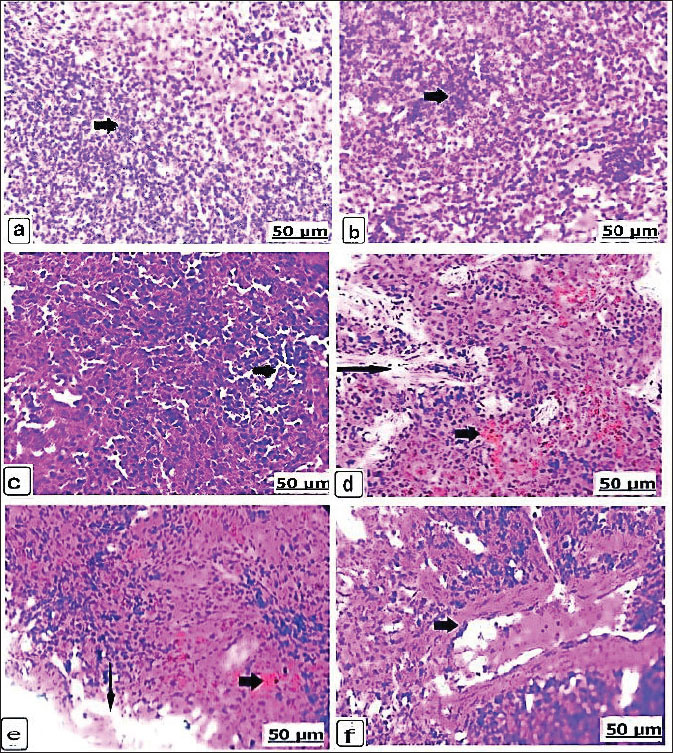
Fig. 5. (a–d). Light photomicrographs of the brains of the control and experimental groups for 21 days, stained with hematoxylin and eosin. Control non-pregnant C2(21) and control pregnant A2(21) showing normal neuron architecture (a and b). Group D1(21) received 625 μg of dexamethasone for 21 days, showing neuronal necrosis with perineuronal vacuolation (long arrow) and edema and dilatation in the Virchow robin space (short arrow) (c). Group B1(21) received 625 μg of dexamethasone for 21 days, showing perineuronal vacuolation (long arrow) and thrombotic vasculitis (short arrow) (d). Microscopic kidneyAfter 14 days, H&E staining of the kidneys of the control groups showed intact kidney tubules with normal organization (Fig. 6a,b). While there was edema and congestion of the blood vessels of the kidneys of the non-pregnant rats who received 625 µg dexamethasone (Fig. 6c), this congestion became extensive with thickening of blood vessels when the pregnant rats were administered a similar dose. In addition, necrosis of renal tissues with edema and congestion of the interstitial blood vessels, as well as hypercellularity of the glomeruli, was found (Fig. 6d). When the non-pregnant group was exposed to 125 × 10 µg of dexamethasone, severe renal tubule necrosis and interstitial fibrosis expressed (Fig. 6e), and sharp renal tissue necrosis occurred in the pregnant group (Fig. 6f). After 21 days, Kidney histology revealed the kidney structures of the normal renal tubules of the control non-pregnant (Fig. 7a); in parallel, the control pregnancy showed an ideal arrangement of the kidney tubules and collecting duct (Fig. 7b). Kidney lesions after receiving 625 µg dexamethasone showed interstitial fibrosis of the renal tubules with marked deposition of the fibrous tissues, besides renal necrosis beside edema, and congestion of the interstitial blood vessels, in non-pregnant and pregnant females (Fig. 7c,d).
Fig. 6. (a–f). Light photomicrograph of the kidneys of the control and experimental groups for 14 days, stained with H& E.: control non-pregnant C1(14) showing intact kidney tubules (arrow). Pregnant control A1(14) showing normal organization of the renal tubules (arrow) (b). Group D1(14) received 625 μg of dexamethasone, showing congested blood vessels (arrow) (c). Group B1(14) received 625 μg of dexamethasone, showing extensive congestion with thickening of blood vessels (short arrow) and renal collecting tubule expansion (long arrow) (d). Group D2(14) received 125 × 10 μg of dexamethasone showing severe necrosis of the renal tubules (arrow) and interstitial fibrosis (long arrow) (e). Group B2(14) received 125 × 10 μg of dexamethasone, showing sharp renal tissue necrosis (arrow) (f).
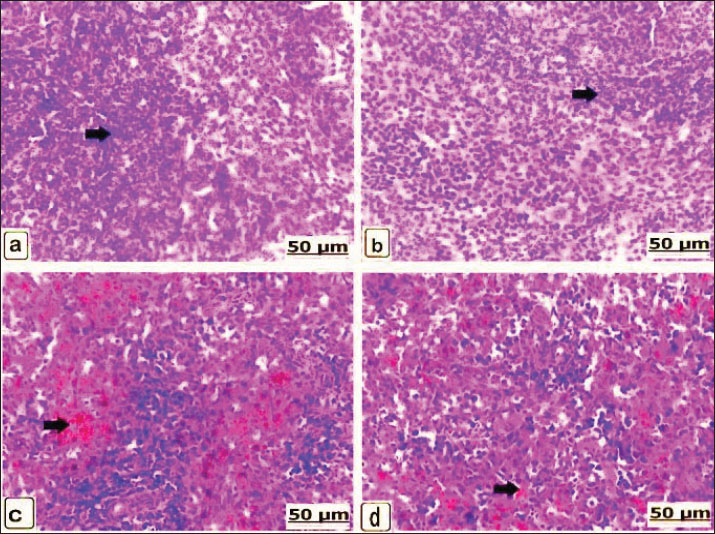
Fig. 7. (a–d). Light photomicrograph of the kidneys of the control and experimental groups stained with H&E for 21 days. Control non-pregnant C2(21) showed normal renal tubules (arrow) (a); likewise control pregnant A2(21) showed an ideal arrangement of collecting duct and tubules (arrow) (b). Group D1 (21) received 625 μg of dexamethasone for 21 days, showing interstitial renal tubule fibrosis (arrow) (c). Group B1(21) received 625 μg of dexamethasone for 21 days, showing edema and congestion of the interstitial blood vessels (short arrow) (d). Microscopic spleenAfter 14 days, the control spleen of the non-pregnant groups showed the normal structure of white and red pulps (Fig. 8a), and the splenic tissues of the pregnant control group showed the ideal organization of the white and red pulps (Fig. 8b). Conversely, the non-pregnant rats received 625 µg of dexamethasone, showing gradual depletion and reduction of lymphoid follicles (Fig. 8c), whereas the pregnant groups showed intensive red blood cell infiltration with remarkable splenic trabeculae protrusion correlated with intense hemosiderosis and white pulp lymphocyte depletion (Fig. 8d). Additionally, the non-pregnant spleen received 125 × 10 µg of dexamethasone and exhibited observable hemorrhage and hemosiderosis in the red pulp characterized by mixed infiltration of red blood cells and hemosiderin, as well as lymphocytic depletion of the lymphoid pulps (Fig. 8e). These lesions were observed in pregnant rats that received 125 × 10 µg dexamethasone; moreover, vasculitis with thickening and inflammation of the blood vessel wall was observed (Fig. 8f). After 21 days, non-pregnant animals in the control group had normally arranged splenic pulps (Fig. 9a), similar to the control spleen of pregnant animals (Fig. 9b). Non-pregnant spleen, when administerted 625 µg dexamethasone, displayed a remarkable degree of hemorrhage characterized by erythrocyte infiltration of the red pulp, as well as a scattered form of hemosiderosis (Fig. 9c), and brown particles of hemosiderin also appeared in the pregnant rat’s spleen of the same category dose (Fig. 9d).
Fig. 8. (a–f). Light photomicrographs of the spleens of the control and experimental groups stained with H&E for 14 days. Control non-pregnant C1(14) and control pregnant A1(14) showing normal white and red pulps (a and b) (arrows). Group D1(14) received 625 μg of dexamethasone, showing gradual reduction of lymphoid cells (arrow) (c). Group B1(14) received 625 μg of dexamethasone, showing intensive blood cell infiltration with remarkable splenic trabeculae protrusion (long arrow) correlated with significant hemosiderosis (short arrow) (d). Group D2(14) received 125 × 10 μg of dexamethasone and showed noticeable hemosiderosis (short arrow) and depleted lymphoid pulps with mononuclear cell infiltration (long arrow) (e). Group B2(14) received 125 × 10 μg of dexamethasone, showing vasculitis and fibrosis of the blood vessel wall with collagen fibers and lymphocytes (arrow) (f).
Fig. 9. (a–d). Light photomicrograph of the spleen of the control and experimental groups stained with H&E for 21 days. Control non-pregnant C2 (21) and control pregnant A2 (21) showing normally arranged splenic pulps (a and b) (arrows). Group D1 (21) received 625 μg of dexamethasone for 14 days, showing severe hemosiderin infiltration and red pulp depletion (arrow) (c). Group B1 (21) received 625 μg of dexamethasone for 14 days, showing brown hemosiderosis particles (arrow) (d). DiscussionDexamethasone is not only a robust glucocorticoid with numerous health benefits but also has intense negative effects (Eken et al., 2006). Dexamethasone is charged in pregnancy-associated adverse implications (Xiao et al., 2024). It was also found that dexamethasone manifested tissue injury with consequent morphological impairments in Wistar rats’ cells owing to pro-oxidants (Owu et al., 2020). This study elicits the influence of dexamethasone on the tissues of pregnant Wistar rats, focusing on organ weight and histomorphology. Here, rats orally drink various doses of dexamethasone in drinking water to identify the safest and most effective dexamethasone dose during pregnancy. Dexamethasone confers great dose-dependent outcomes on structural organs and histology. The dose of dexamethasone a rat would receive from a veterinarian was 0.527 μg/kg BW according to English et al. (1975). In contrast to the non-pregnant control group, the weights of the brain and spleen in the non-pregnant group that received 625 µg of dexamethasone showed the least decrease in weight, while the weights of the liver showed the least increase. Compared with the non-pregnant control group, the kidney weights of the non-pregnant group that received 125 × 10 µg of dexamethasone exhibited the least decrease. Glucocorticoid drugs can interfere with the development of many tissues, serving a great influence on organs such as the kidneys, liver, and lungs (Martins et al., 2003). According to Wafula et al. (2005), dexamethasone remarkably induced a significant decline in the organ weights, notably the splenic tissues. Pregnant Albino rats, when subcutaneously administered 1 mg/kg dexamethasone, produced harmful impacts on skeletal muscle fibers, muscle weight, and muscle Deoxyribonucleic acid. Dexamethasone also decreased cell proliferation in various tissues (Ranta et al., 2006). In this context, the pregnant group that received 625 µg of dexamethasone showed the least increase in liver and kidney weights compared with the pregnant control group. Similarly, the pregnant group that received 125 × 10 µg of dexamethasone showed the least increase in brain weights and the smallest decrease in spleen weights compared to the non-pregnant control group. Increased liver weights in mice administered ascending doses of dexamethasone were detected by Ashraf and Ilyas (2015). Besides these, the same results were confirmed by Hammadi et al. (2022), who recorded a significant increase in liver and kidney tissue weights after a period of dexamethasone injection, compared with the body weights of the control group. The various histological changes observed in discrete organs involving the liver, brain, kidney, and spleen were analyzed after the administration of dexamethasone according to the cross-section. In general, glucocorticoid treatment in rats is associated with abnormal histomorphology and increased cell apoptosis. The degree of histological damage was mild and transformed to severe with degenerative changes at higher doses (Chen et al., 2019). Dexamethasone administration was accompanied by extensive cellular defects (Dehghani et al., 2021). In the group administered 625 µg of dexamethasone, the liver showed intensive necrosis and vacuolar degeneration of the hepatocytes with interstitial hemorrhage, which agreed with the findings of the previous study (Hammadi et al., 2022). Another study showed that dexamethasone-treated rats exhibited varying histological changes, including mononuclear cell infiltrates, sinusoidal disruption, and congestion (Hussein et al., 2014). The possible mechanism may be due to the alteration of dexamethasone excitation in lipid metabolism or the motivation of oxidative cell stress (Kumar et al., 2015). It has been documented as a major cause of dexamethasone-related liver injury and extreme free radical formation (Hasona et al., 2017). These changes may be due to the dexamethasone provokes cellular apoptosis, cell death, and decreased proliferation in a variety of distinct cells (Ranta et al., 2006). Moreover, in the brain, 625 µg of dexamethasone showed neuronal necrosis and thrombotic vasculitis, as well as demyelination and necrosis of Purkinje cell layers. These findings evidenced that dexamethasone could intensify brain injury and cerebral edema (Duffy et al., 2014). Dexamethasone could elicit an inflammatory effect associated with neurotoxicity induced under considerable conditions (Munhoz et al., 2006). In the kidney, the same group showed severe renal tissue necrosis and glomerular hypercellularity with extensive edema and interstitial blood vessel congestion (Sanchez-Aceves et al., 2024). Additionally, Liu et al. (2008) mentioned that dexamethasone administration accelerates histopathological lesions in the rats’ kidney, resulting in fibrosis and necrosis of the convoluted tubule epithelium and atrophied glomeruli. These histological changes are probably due to increased monocyte chemoattractant protein-1 gene expression in the kidney, which was promoted after dexamethasone treatment. In the spleen, the same group showed intensive red blood cell infiltration with remarkable splenic trabeculae protrusion correlated with lymphocyte depletion of the white pulp (Madlool et al., 2024). Considering the similar findings of those who reported that the administration of dexamethasone pathologically affected the internal organs of female Wistar rats (Schäfer et al., 2005; Wei et al., 2016). Histological effects include damage to the lymphoid follicular areas of the white pulps and fibrin aggregation within red pulp zones (Dehghani et al., 2021). The cytotoxic and immunosuppressive effects of dexamethasone are a prospective explanation for the splenic lesions (Wafula et al. 2025). In dose-dependent effects, increasing doses of dexamethasone lead to increased reactive oxygen species formation by myeloid cells within the spleen pulps, causing apoptosis and cell death (Swildens et al., 2022). Dexamethasone is a glucocorticoid with significant therapeutic prospects but may induce adverse organ effects and be associated with clinical changes when the pregnant female is treated; however, it may trigger adverse impacts in the fetus. This is a limitation of our investigation, and this possibility calls for further study in the future and for exploring early treatment and prevention strategies. ConclusionChronic pathologies have consequences in vivo in a dose-dependent category, as evidenced by an increase in the level of dexamethasone during pregnancy. It indicates a potential crosstalk between dexamethasone use in pregnancy and increased risks, probably contributing to hormonal changes and other factors that may be involved in the mechanism for these unfavorable outcomes. Dexamethasone, a dose-dependent medication, should be used when medically necessary during pregnancy. AcknowledgmentsThe authors thank the Vet-Med Faculty at Sohag and Menofia Universities for their cooperation. The referees’ efforts to evaluate this article are greatly appreciated. Conflict of interestThe authors declare that there are no conflicts of interest in this article’s research, authorship, or publishing. FundingThis work was supported by the Cultural and Educational Grant Agency of the Ministry of Education, Research, Development and Youth of the Slovak Republic under the project KEGA no. 001UVLF-4/2025 --Innovation in physiology education for foreign students Authors’ contributionsSGF, IFR, and MEM jointly developed the hypothesis and concept of the study, participated in the experimental procedure, and contributed to the chemistry and materials, as well as techniques, of the manuscript. IFR, FAM, IRM, FZ, SO, and ZA-A were all involved in the analyses of this study and scientific paper. All authors participated in the experimental analysis and helped with the rewriting of the manuscript. The final manuscript was read and approved by all authors. Data availabilityThe data supporting their conclusions are available from the authors of this manuscript upon request. ReferencesAshraf, M. and Ilyas, M.S. 2015. Effects ofdexamethasone on liver histology of adult albino mice. Pak. Postgrad. Med. J. 26, 109–113. Bancroft, J.D. and Gamble, M. 2002. Theory and practice of histological techniques, 5th ed. Philadelphia, PA: Churchill Livingstone, pp: 125–38. Benediktsson, R., Lindsay, R.S., Noble, J., Seckl, J.R. and Edwards, C.R. 1993. Glucocorticoid exposure in utero: a new model for adult hypertension. Lancet 341, 339–341. Chen, Y., Huang, L.F. and Zhu, J.X. 2019. Dose-related histopathology and bone remodeling characteristics of glucocorticoid-induced knee articular cartilage and subchondral bone in rats. Exp. Ther. Med. 17, 4492–4498. Cheng. and Z. 2021. Modified cervical dislocation, a better way in laboratory rat euthanasia. J. Mod. J. Med. Biol. 1, 1–3. Costa-Silva, J.H., Lyra, M.M., Lima, C.R., Arruda, V.M., Araújo, A.V., Ribeiro, A.R., Arruda, A.C., Fraga, M.C.C.A., Lafayette, S.S.L. and Wanderley, A.G. 2007. A toxicological evaluation of the effect of Carapa guianensis Aublet on pregnancy in Wistar rats. J. Ethnopharmacol. 112, 122–126; doi: 10.1016/j.jep.2007.07.016 Cuffari. 2020. The anatomy of the human brain. Manchester, UK: News Medical Life Science. Dehghani, F., Hossieni, S.A., Noorafshan, A., Panjehshahin, M.R. and Esmaeilpour, T. 2021. Selenium effect on quantitative structural changes in dexamethasone-induced immunodeficiency rat models. Iran. J. Med. Sci. 46, 128–135. Duffy, B.A., Chun, K.P., Ma, D., Lythgoe, M.F. and Scott, R.C. 2014. Dexamethasone intensifies cerebral edema and brain injury following lithium-pilocarpine-induced status epilepticus. Neurobiol. Dis. 63, 229–236. Eken, H., Ozturk, H., Ozturk, H. and Buyukbayram, H. 2006. Dose-related effects of dexamethasone on rat liver damage due to bile duct ligation. World J. Gastroenterol. 12, 5379–5383. English, J., Chakraborty, J. and Marks, V. 1975. The metabolism of dexamethasone in the rat: effect of phenytoin. Steroid. Biochem. 6, 65–68. Figueroa, J.P., Rose, J.C., Massmann, G.A., Zhang, J. and Acuña, G. 2005. Alterations in fetal kidney development and arterial blood pressure elevations in young adult sheep after clinical antenatal glucocorticoid doses. Pediatr. Res. 58, 510–515. Gibson-Corley, K.N., Olivier, A.K. and Meyerholz, D.K. 2013. Principles for valid histopathologic scoring in research. Pathol 50, 1007–1015. Gloor, B., Uhl, W., Tcholakov, O., Roggo, A., Muller, C.A., Worni, M. and Büchler, M.W. 2001. Hydrocortisone treatment of early SIRS in acute experimental pancreatitis. Dig. Dis. Sci. 46, 2154–2161. Hammadi, J.N., Adel Kareem, D., Majeed, A.A. and Abbas, B.A. 2022. Effect of long-term treatment with dexamethasone on liver and kidney histopathology and blood biochemistry in male rabbits (Lepus cuniculus). Razi Inst. 77, 333–343. Hasona, N.A., Alrashidi, A.A., Aldugieman, T.Z., Alshdokhi, A.M. and Ahmed, M.Q. 2017. Vitis vinifera extract reduces dexamethasone-induced hepatic and renal dysfunction in albino rats. Toxics 5, 11. Hussein, A.J., Majeed, M.F. and Abbas, A.S. 2014. Histopathological study of some organs after long-term dexamethasone treatment in male rabbits. Science 30, 39. Jeklova, E., Leva, L., Jaglic, Z. and Faldyna, M. 2008. Dexamethasone-induced immunosuppression: a rabbit model. Immunopathol 122, 231–240. Kathim, A.S. and Al-Hillo, M.F. 2019. Biochemical changes of dexamethasone on liver and kidney functions in laboratory mice Mus musculus L. Bionatura 8(1), 1–7. Kumar, V.H., Im, N.N., Huilgol, S.V., Yendigeri, S.M. and Ch, R. 2015. Dose-dependent hepatic and endothelial changes in rats treated with dexamethasone. J. Clin. Diagn. Res. 9, FF08–FF10. Lewis, S.M., Williams, A. and Eisenbarth, S.C. 2019. Structure and function of the immune system in the spleen. Sci. Immunol. 4, 6085. Liu, Y., Van Goor, H., Havinga, R., Baller, J.F., Bloks, V.W., Van Der Leij, F.R., Sauer, P.J.J., Kuipers, F., Navis, G. and De Borst, M.H. 2008. Neonatal administration of dexamethasone causes progressive renal damage due to the induction of an early inflammatory response. Am. J. Physiol. Renal Physiol. 294, F768–F776. Madlool, Z.S., Kadhum, E.H., Nasar, D.M. and Mohammed, D.H. 2024. Physiological and histological effects of dexamethasone: a literature review. Int. J. Pharm. Sci. 2, 620–629. Manojlivić, M., Hristić, M., Kalafatić, D., Plećaš, B. and Ugrešić, N. 1998. The influence of dexamethasone treatment of pregnant rats on the development of chromaffin tissue in their offspring during the fetal and neonatal period. J. Endocrinol. Invest. 21, 211–218; doi:10.1016/j.jendo.2018.01.013 Marshall, J.C. and Morriss-Kay, G.M. 2004. Functional anatomy of the human brain. J. Anat. 205, 415. Martins, J.P., Monteiro, J.C. and Paixão, A.D. 2003. Renal function in adult rats subjected to prenatal dexamethasone. Clin. Exp. Pharmacol. Physiol. 30(1-2), 32–37. Mesripour, A. and Rakhshankhah, P. 2021. A synbiotic mixture ameliorates depressive behavior induced by dexamethasone or water avoidance stress in a mouse model. Turkish J. Pharm. Sci. 18(1), 21. Mohan, D. and Sharma, S. 2017. Histopathological alterations in liver of mice exposed to different doses of diclofenac sodium. Int. J. Anim. Vet. Sci. 11, 738–742. Mohan, K., Kecova, H., Hernandez-Merino, E., Kardon, R.H. and Harper, M.M. 2013. Retinal ganglion cell damage in a blast-mediated traumatic brain injury experimental rodent model. Invest. Ophthalmol. Vis. Sci. 54, 3440–3450. Munhoz, C.D., Lepsch, L.B., Kawamoto, E.M., Malta, M.B., Lima, L.S., Avellar, M.C.W., Sapolsky, R.M. and Scavone, C. 2006. Chronic unpredictable stress intensifies lipopolysaccharide-induced activation of nuclear factor-κB in the frontal cortex and hippocampus via glucocorticoid secretion. J. Neurosci. 26, 3813–3820. Oaks, J.L., Gilbert, M., Virani, M.Z., Watson, R.T., Meteyer, C.U., Rideout, B.A., Shivaprasad, H.L., Ahmed, S., Iqbal Chaudhry, M.J., Arshad, M., Mahmood, S., Ali, A. and Ahmed Khan, A. 2004. Diclofenac residues as the cause of vulture population decline in Pakistan. Nature 427, 630–633. Ortiz, L.A., Quan, A., Weinberg, A. and Baum, M. 2001. Effect of prenatal dexamethasone on renal development in rats. Kidney Int. 59, 1663–1669. Owu, D.U., Okon, I.A., Ufot, U.F. and Beshel, J.A. 2020. Cardiac and renal protective effect of vitamin E against dexamethasone-induced oxidative stress in Wistar rats. Niger. J. Physiol. Sci. 35, 52–60. Provan, D., Stasi, R., Newland, A.C., Blanchette, V.S., Bolton-Maggs, P., Bussel, J.B., Chong, B.H., Cines, D.B., Gernsheimer, T.B., Godeau, B., Grainger, J., Greer, I., Hunt, B.J., Watson, S., Young, J. and Kuter, D.J. 2010. International consensus report on primary immune thrombocytopenia investigation and management. Blood 115, 168–186. Ranta, F., Avram, D., Berchtold, S., Düfer, M., Drews, G., Lang, F. and Ullrich, S. 2006. Dexamethasone induces cell death in insulin-secreting cells, an effect reversed by exendin-4. Diabetes 55, 1380–1390. Sanchez-Aceves, L.M., Pérez-Alvarez, I., Onofre-Camarena, D.B., Gutiérrez-Noya, V.M., Rosales-Pérez, K.E., Orozco-Hernández, J.M., Hernández-Navarro, M.D., Flores, H.I. and Gómez-Oliván, L.M. 2024. Prolonged exposure to dexamethasone, a synthetic glucocorticoid, induces brain damage via oxidative stress and apoptotic response in adult Danio rerio. Chemosphere 364(no. 143012), 143012. Schäfer, S.C., Wallerath, T., Closs, E.I., Schmidt, C., Schwarz, P.M., Förstermann, U. and Lehr, H.A. 2005. Dexamethasone suppresses eNOS and CAT-1 and induces oxidative stress in mouse resistance arterioles. Heart Circ. Physiol. 288, H436–H444. Swildens, K.X., Sillevis Smitt, P.A.E., Van Den Bent, M.J., French, P.J. and Geurts, M. 2022. The effect of dexamethasone on the microenvironment and efficacy of checkpoint inhibitors in glioblastoma: a systematic review. Neurooncol. Adv. 4, 87; doi:10.1016/j.nonc.2018.01.007 Vidya Vijayan, K.K., Karthigeyan, K.P., Tripathi, S.P. and Hanna, L.E. 2017. Pathophysiology of CD4⁺ T-cell depletion in patients with HIV-1 and HIV-2 infections. Front. Immunol. 8, 580. Wafula, G.N., Chege, B.M., Cheruiyot, I., Butt, F. and Munguti, J. 2005. Protective effects of curcumin on spleen parenchyma in dexamethasone-treated rats. Mediterr. J. Pharm. Pharm. Sci. 5, 87–96. Wei, Y., Ji, X.B., Wang, Y.W., Wang, J.X., Yang, E.Q., Wang, Z.C. and Hou, M. 2016. High-dose dexamethasone versus prednisone for treatment of adult immune thrombocytopenia: a prospective multicenter randomized trial. Blood 127, 296–302. Xiao, H., He, B., Liu, H., Chen, Y., Xiao, D. and Wang, H. 2024. Dexamethasone exposure during pregnancy triggers metabolic syndrome in offspring via IGF1 epigenetic alteration. Cell. Communication. Signaling. 22, 62. Yavroyan, Z., Hakobyan, N., Hovhannisyan, A., Grigoryan, A., Karapetyan, A., Abgaryan, T. and Gevorgyan, E. 2025. Lipid peroxidation level and histological changes in rat liver after cisplatin and dexamethasone action. Indian. J. Biochem. Biophys. 62, 480–489. Zone, M., Wanke, M., Rebuelto, M., Loza, M., Mestre, J., Duchene, A. and Concannon, P. 1995. Termination of pregnancy in dogs by oral administration of dexamethasone. Theriogenology 43(2), 487–494.
| ||
| How to Cite this Article |
| Pubmed Style Fahmy SG, Al-amgad Z, Rehan IF, Mahmoud FA, Ibrahim RM, Zigo F, Ondrašovičová S, Mahmoud ME. Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Vet. J.. 2026; 16(3): 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 Web Style Fahmy SG, Al-amgad Z, Rehan IF, Mahmoud FA, Ibrahim RM, Zigo F, Ondrašovičová S, Mahmoud ME. Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. https://www.openveterinaryjournal.com/?mno=285502 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.24 AMA (American Medical Association) Style Fahmy SG, Al-amgad Z, Rehan IF, Mahmoud FA, Ibrahim RM, Zigo F, Ondrašovičová S, Mahmoud ME. Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Vet. J.. 2026; 16(3): 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 Vancouver/ICMJE Style Fahmy SG, Al-amgad Z, Rehan IF, Mahmoud FA, Ibrahim RM, Zigo F, Ondrašovičová S, Mahmoud ME. Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 Harvard Style Fahmy, S. G., Al-amgad, . Z., Rehan, . I. F., Mahmoud, . F. A., Ibrahim, . R. M., Zigo, . F., Ondrašovičová, . S. & Mahmoud, . M. E. (2026) Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Vet. J., 16 (3), 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 Turabian Style Fahmy, Sohaila G., Zeinab Al-amgad, Ibrahim F. Rehan, Fatma A. Mahmoud, Rawia M. Ibrahim, František Zigo, Silvia Ondrašovičová, and Motamed E. Mahmoud. 2026. Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Veterinary Journal, 16 (3), 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 Chicago Style Fahmy, Sohaila G., Zeinab Al-amgad, Ibrahim F. Rehan, Fatma A. Mahmoud, Rawia M. Ibrahim, František Zigo, Silvia Ondrašovičová, and Motamed E. Mahmoud. "Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats." Open Veterinary Journal 16 (2026), 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 MLA (The Modern Language Association) Style Fahmy, Sohaila G., Zeinab Al-amgad, Ibrahim F. Rehan, Fatma A. Mahmoud, Rawia M. Ibrahim, František Zigo, Silvia Ondrašovičová, and Motamed E. Mahmoud. "Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats." Open Veterinary Journal 16.3 (2026), 1661-1677. Print. doi:10.5455/OVJ.2026.v16.i3.24 APA (American Psychological Association) Style Fahmy, S. G., Al-amgad, . Z., Rehan, . I. F., Mahmoud, . F. A., Ibrahim, . R. M., Zigo, . F., Ondrašovičová, . S. & Mahmoud, . M. E. (2026) Comparable organs weights and histopathological changes after administration of dexamethasone during pregnancy in rats. Open Veterinary Journal, 16 (3), 1661-1677. doi:10.5455/OVJ.2026.v16.i3.24 |