| Research Article | ||
Open Vet. J.. 2026; 16(3): 1687-1694 Open Veterinary Journal, (2026), Vol. 16(3): 1687-1694 Research Article Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat modelMarwa Sabah Majed1, Fatema Ali Al-Kafhage1, Hiba Alameri1,3*, Amaal Sahib Al-Zughaibi1, Amna Mohammed Hamza2 and Roaa Noori Ali11College of Veterinary Medicine, Kerbala University, Karbala, Iraq 2Department of Pathological Analysis, College of Applied Medical Sciences, University of Karbala, Karbala, Iraq 3Department of Radiology, College of Health and Medical Techniques, Al-Zahraa University for Women, Karbala, Iraq *Corresponding Author: Hiba Alameri. College of Veterinary Medicine, Kerbala University, Karbala, Iraq. Email:hiba.abdulkreem [at] uokerbala.edu.iq Submitted: 21/09/2025 Revised: 11/02/2026 Accepted: 24/02/2026 Published: 31/03/2026 © 2026 Open Veterinary Journal
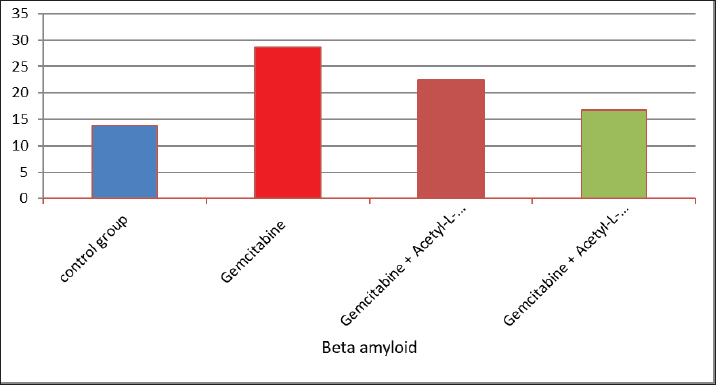
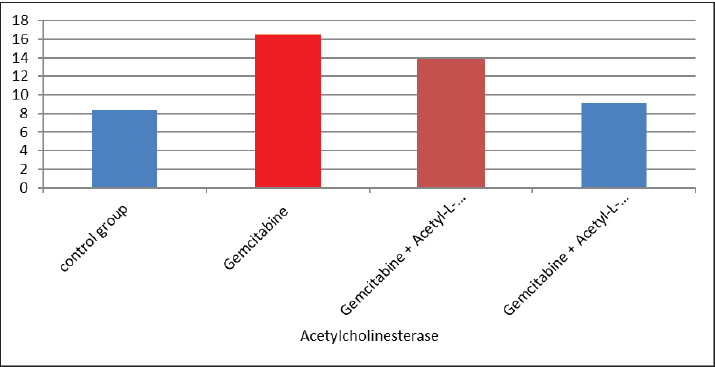
AbstractBackground: The acetyl ester of L-carnitine (ALC) is essential for cellular energy homeostasis and intermediate metabolism. It demonstrates several well-established biological characteristics, such as antioxidant activity, intracellular membrane stability, mitochondrial function improvement, and neuroprotective and neurotrophic effects. Given that oxidative stress, mitochondrial malfunction, and neuronal membrane damage have been linked to gemcitabine-induced neurotoxicity, ALC may have a preventive role against these negative consequences. Its capacity to boost mitochondrial metabolism and act as an antioxidant points to a potential mechanism for reducing damage to the central nervous system (CNS) caused by gemcitabine. Thus, using an experimental model of gemcitabine-induced neurotoxicity, we explored the possible neuroprotective benefits of ALC. Aim: This study aimed to examine the neuroprotective effects of two distinct dosages of ALC on cognitive impairment in male rats caused by gemcitabine injection. Cognitive function was assessed using well-known behavioral tests, such as the Morris water maze or novel object identification, to offer objective measures of learning and memory. This study aimed to elucidate the possible function of ALC in reducing gemcitabine-related CNS toxicity by evaluating behavioral performance in addition to biochemical indicators of oxidative stress and neuronal damage. It is anticipated that the results of this study will shed light on the dose-dependent effectiveness of ALC in maintaining cognitive function after chemotherapy. Methods: The experiment was conducted using 24 adult male rats, which were randomly assigned to four equal groups (n=6 per group). Treatments were administered once daily for 28 consecutive days. Group 1 (G1–Control): Rats received a subcutaneous injection of normal saline and served as the negative control. Group 2 (G2–Gemcitabine 25 mg/kg): Rats received 25 mg/kg of Gemcitabine intraperitoneally once daily. Group 3 (G3– ALC 50 mg/kg): Rats received 50 mg/kg of ALC intraperitoneally once daily. Group 4 (G4–Gemcitabine + ALC): Rats received a combination of Gemcitabine (25 mg/kg bw) and ALC (50 mg/kg bw), both administered intraperitoneally once daily. All treatments were administered at the same time each day to ensure consistency, and the animals were monitored throughout the study period to assess their health status and minimize stress. Results: Rats receiving ALC in addition to gemcitabine demonstrated a significant reduction in gemcitabine-induced adverse effects (p < 0.05) compared with the group treated with gemcitabine alone. This protective effect was mainly seen in characteristics associated with the function of the central nervous system, that ALC plays a neuroprotective role. Conclusion: The fourth experimental group (G4) had considerably lower levels of β-amyloid and acetylcholinesterase activity than the second (G2) and third (G3) groups. Furthermore, brain sections from the G4 group showed significantly fewer β-amyloid plaques than those from the G2 and G3 groups, according to Congo red staining histological analysis. Keywords: Acetyl-L-carnitine, Acetylcholinesterase, Beta amyloid, Congo red, Gemcitabine. IntroductionCancer treatment is frequently associated with adverse effects, as the toxicity of chemotherapeutic agents can reduce life expectancy and significantly impair patients’ quality of life. Gemcitabine is a potent cytotoxic antimetabolite that is widely used for treating various solid tumors, including pancreatic, lung, breast, and bladder cancers. Gemcitabine primarily exerts its anticancer effects by inhibiting DNA synthesis during the S phase of the cell cycle. Accurate depiction of medication indications and mechanisms of action is crucial, even though chemotherapy-induced toxicity is a significant therapeutic problem. Thus, the toxicological effects of gemcitabine administration are the main topic of this investigation, with a focus on the systemic effects (Maleki et al., 2025). White blood cell malignancies, including non-Hodgkin lymphoma, acute myeloid leukemia, also known as acute lymphocytic leukemia, and long-term myelogenous leukemia, are the main conditions treated with gemcitabine, a class of chemotherapeutic medication (Larson et al., 2024). Its effects are specific to the S-shaped cellular cycle. Significant chromosomal damage causes chromosomal abnormalities. Mitosis has the greatest effect in cells with rapid division that need to duplicate DNA (Yi et al., 2022) reactive oxygen species (ROS) are produced, and their concentrations are decreased because the function of enzymes known as antioxidants in the liver tissues is inhibited by gemcitabine. However, since this combination is known to undergo significant hepatic metabolism, the resulting increase in ROS may contribute to brain tissue injury (Tauffenberger and Magistretti, 2021). The liver, kidney, and brain all naturally produce the amino acid L- alongside the necessary amino acids methionine and lysine (Davidova et al., 2022). Since the chemical originates after flesh, its scientific designation is derived from the Latin carnus, which means meat Taub (2023). Acetyl-L-carnitine (ALC) is essential for energy utilization because it moves toxic compounds through organelles and prevents these contaminants from building up inside the cell’s tissues. It additionally carries long-chain fatty acids to the mitochondria Javan et al. (2025). ALC is essential for the oxidation of fatty acids and the conversion of energy; thus, it might safeguard the cell membranes from damage caused by oxidative stress (Kıran et al., 2023). ALC might inhibit these chemical reactions in cells by numerous pathways—preservation of mitochondrial function (Huseynova, 2025) and reduction of ROS generation at various intracellular sites (Virmani and Cirulli, 2022). Protein Kinase B is among the most common neurotransmitters in the human nervous system. Ace is a neurotransmitter used as a signaling molecule by neurons with cholinergic activity (Zulfugarova et al., 2025). The activity of cholinesterase is crucial to stop chemicals released from acetylcholine neurons from reaching the nervous system, skeletal muscle, or gland that is being activated (Ashraf, 2023). Acetylcholinesterase (AChE) quickly breaks down acetylcholine into choline and acetic acid, rendering it ineffective. The brain’s nervous system needs oxygen to function properly (Guliyeva et al., 2025). Because ace is inhibited, acetylcholine can accumulate, overstimulating cholinergic junctions and organs controlled by cholinergic neurons (Lakshmanan, 2021). Materials and MethodsIn this study, 24 male white albino rats weighing between 260 and 300 g were used. Individuals were housed in well-maintained cardboard cages that included proper ventilation, light structure, and temperature (about 30℃ ± 5℃) for 12 hours each day. The trial was conducted during a 28-day period. Twenty-four adult male rats were randomly divided into four experimental groups, each consisting of six animals. Six male rats in Group I (G1, control group) were administered intraperitoneal injections of normal saline once a day during the experiment. Six male rats in Group II (G2) received 25 mg/kg body weight of intraperitoneal gemcitabine. Six male rats in Group III (G3) were administered intraperitoneal doses of gemcitabine (25 mg/kg body weight) and ALC (50 mg/kg body weight). Six male rats in Group IV (G4) were administered gemcitabine (25 mg/kg body weight) and ALC (50 mg/kg body weight) intraperitoneally for 4 consecutive days. Blood samples were taken from each rat separately and appropriately labeled to prevent sampling bias. In compliance with institutional ethical guidelines for animal care and use, the animals were put to sleep before blood collection using a compassionate and authorized anesthetic approach instead of being exposed to chloroform. Blood was drawn via heart puncture using sterile needles in an aseptic setting after anesthesia was administered. Each animal provided approximately 5 ml of blood. The leftover blood was split into two parts, and 1 ml was set aside for whole blood analysis. After transferring approximately 2 ml into gel tubes, the mixture was left to coagulate for 30 minutes at room temperature. Study parametersMeasurement of serum β-Amyloid (Aβ₁–₄₂) A commercial rat ELISA kit (Elabscience Biotechnology Comapny Ltd., China) was used to measure serum β-amyloid (Aβ₁–₄₂) concentrations according to the manufacturer’s instructions. A quantitative sandwich enzyme immunoassay method serves as the foundation of the assay. Results were expressed in pg/ml, and the sensitivity and detection range of the kit were within the manufacturer’s limits. Every sample was examined twice to guarantee analytical precision and repeatability. Serum AChE measurementA rat AChE enzyme-linked immunosorbent assay kit (Elabscience Biotechnology Company Ltd., China) was used to test serum AChE levels in accordance with the manufacturer’s instructions. A microplate reader was used to detect the absorbance, and the standard calibration curve was used to determine the concentrations. The kit’s stated units were used to express the results. Every measurement was conducted twice, and assay performance was verified using the manufacturer’s quality control methods. Histopathological analysis (Congo red staining)Brain tissues were quickly removed and preserved for 24–48 hours following animal sacrifice in 10% neutral buffered formalin. Samples were dehydrated, cleaned, and embedded in paraffin following conventional histological methods. To find β-amyloid buildup, coronal slices (4–5 µm thick) were taken and stained with Congo red. The hippocampus and cerebral cortex, two brain areas linked to cognitive performance, were histologically examined. An investigator who was blinded to the experimental groups examined the amyloid deposits under light microscopy. Statistical analysisSPSS software (version 24.0; IBM Corp., USA) was used to analyze the data. Every outcome is presented as mean ± standard error. One-way analysis of variance was used to compare the various experimental groups. When a statistically significant difference was found, Tukey’s test was used in post-hoc multiple comparison analysis to find differences between individual groups. A statistically significant p-value was defined as <0.05. Ethical approvalThis work was approved by the ethics committee of the College of Veterinary Medicine, Kerbala University (UOK.VET. PH.2025). ResultsThe results of the present investigation indicate that the Gemcitabine group had a significant (p ≤ 0.05) raised level of blood samples beta-amyloid when compared with the remainder of the categories, whereas the Gemcitabine + ALC (25 mg/kg/bw) group showed a significant (p ≤ 0.05) improve when contrasted to the comparison group and the combination of Gemcitabine + ALC (300 mg/kg/bw) organization. However, Fig. 1 shows that there was no statistically significant difference (p > 0.05) in serum-amyloid levels between the control group and the group treated with gemcitabine + ALC (300 mg/kg bw). The present research also demonstrates that the Gemcitabine category had a significant (p ≤ 0.05) improvement in blood samples AChE when compared with each of the remaining organizations, whereas the gemcitabine + ALC (25 mg/kg/bw) category showed an important (p ≤ 0.05) increase when compared with the group that received no treatment and the gemcitabine + ALC (300 mg/kg/bw) organization (Fig. 1).
Fig. 1. Effect of gemcitabine, gemcitabine + ALC (25 mg/kg/bw), and gemcitabine + acetyl-L-incarnadine (300 mg/kg/bw) on serum beta-amyloid levels. The present research also demonstrates that the Gemcitabine category had a significant (p ≤ 0.05) improvement in blood samples AChE when compared with each of the remaining organizations, whereas the gemcitabine + ALC (25 mg/kg/bw) category showed an important (p ≤ 0.05) increase when compared with the group that received no treatment and the gemcitabine + ALC (300 mg/kg/bw) organization (Fig. 2).
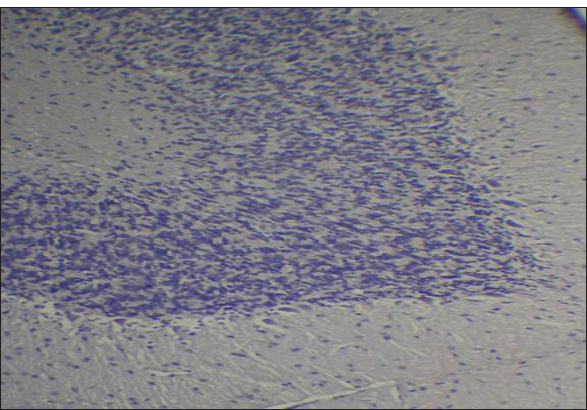
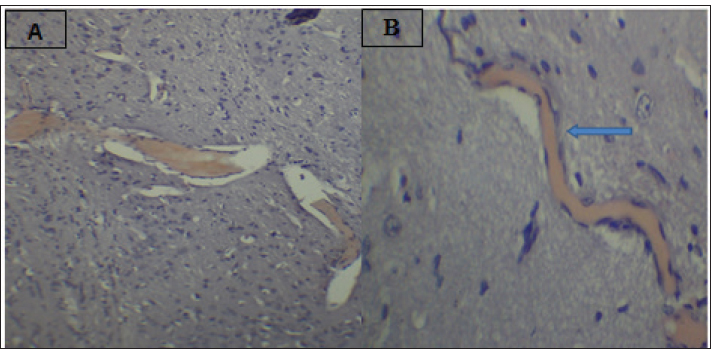
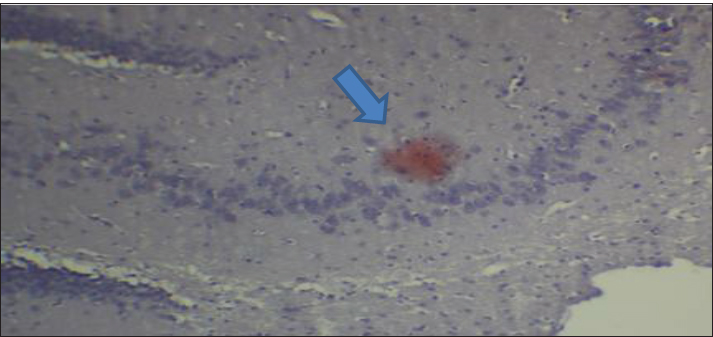
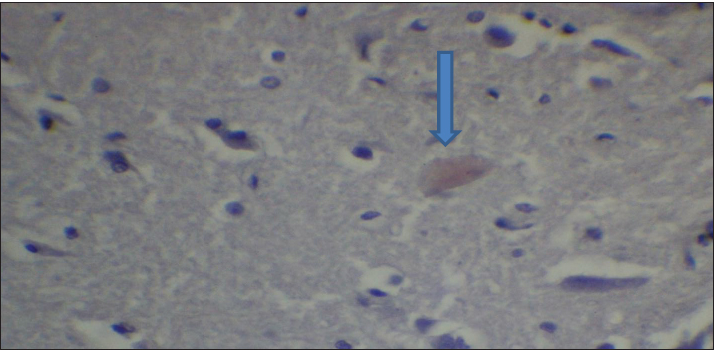
Fig. 2. Effect of gemcitabine, gemcitabine + ALC (25 mg/kg/bw), and gemcitabine + ALC (300 mg/kg/bw) on serum AChE. Histological resultsAccording to the histological investigation, the central nervous system did not contain any beta clusters. The experimental beta-amyloid turns red because it is stained using the pigment Congo red, indicating that the central nervous system is secure from beta-amyloid buildup in the absence of treatment (Fig. 3). The gemcitabine group showed an increase in beta-amyloid accumulation in the rat’s brain, which is stained in red color as shown in Figure 4. However, compared to the gemcitabine group, rats administered gemcitabine plus ALC (25 mg/kg/bw) showed a significant reduction in the buildup of beta-amyloid in the rat’s brain, as depicted in Figure 5. Furthermore, as illustrated in Figure 6, rats treated with gemcitabine + ALC (300 mg/kg/bw) exhibit a significant reduction in the buildup of beta-amyloid in their brains when compared to rats treated with gemcitabine + ALC (25 mg/kg/bw).
Fig. 3. Photomicrograph of the control group’s brain section showing the normal histological architecture without the accumulation of beta-amyloid (congo red, 4×).
Fig. 4. Photomicrograph of the brain section of the gemcitabine group showing the accumulation of beta-amyloid plaques (congo red) (A 4×, B 10×).
Fig. 5. Photomicrograph of the brain section of the Gemcitabine + ALC (25 mg/kg/bw) group showing the accumulation of beta-amyloid plaques (congo red) (4×).
Fig. 6. Photomicrograph of the brain section of the Gemcitabine + ALC (300 mg/kg/bw) group showed the accumulation of beta plaques (red) (4×). DiscussionGemcitabine increases the production of ROS, including superoxide radicals, which can lead to oxidative stress. The build-up of these reactive species may contribute to cellular toxicity by increasing protein oxidation, reducing enzyme activity, and compromising cell membrane integrity through lipid peroxidation. Moreover, oxidative stress may facilitate the production of secondary reactive chemicals that worsen cellular damage. Although these mechanisms offer a reasonable explanation for the toxicity caused by gemcitabine, care should be taken when interpreting them because the precise molecular pathways may differ based on the kind of tissue and the settings of the experiment. This investigation found that the fatty acid aldehydes, such as the substance and 4-hydroxynonenal, this investigation found indicating that the Gemcitabine-treated group had significantly greater variations in the bloodstream beta-amyloid levels than the remainder of the participants (Metselaar et al., 2024). The effects of gemcitabine are exclusive to the S-shaped cellular lifecycle. Significant chromosome damage causes chromosomal abnormalities. This drug thus inhibits the capacity of cells with quick division that need replication of DNA to repair individuals, potentially leading to the death of certain nerve cells (Morgillo and Marovino, 2021). Beta-amyloid may have increased in individuals who just received a combination of dying nerve cells generate a lot of amyloids in a type of beta-amyloid (Yin et al., 2021; Goel et al., 2022). ALC, and these assists in minimizing the damaging effects of oxidative stress with tissue and additionally assists in eliminating the chemical toxicology associated with gemcitabine, triggered an important decrease within the concentration of the protein beta-a within animals the fact that switched the package. Nevertheless, it has been found that a concentration of 300 mg/kg/bw was more effective in terms of effectiveness and efficiency compared with the 50 mg/kg/bw dose (Madkour et al., 2022; Wang et al., 2024). Brain beta-amyloid peptide (Abeta) may affect ache stages because ache concentrations are elevated in the vicinity of beta-amyloid plaque. Such data piqued our curiosity about the potential for the enzyme AchE as well as amyloidosis level to fluctuate within tandem, meaning that if beta-amyloid plaques rise, which is this will affect ache, which might ultimately result in cerebral suppression (Mahdi et al., 2021; Bansal et al., 2025). Histological evaluation demonstrated that the group receiving treatment had less beta-amyloid buildup and smaller beta-amyloid deposits compared to this drug category. Amyloid beta precursor amino acids are present in a variety of cellular membrane types and discharge. Amyloids quickly enter the plasma and cerebrospinal fluid. Deposits are lumps of strands and are aggregated from internalized β-amyloids, which pile on over each other as well as compress forming β-pleated or β-folded shapes (Gottwald and Röcken, 2021; Whitfield et al., 2023). Congo red pigment is frequently used to examine amyloidosis because it binds to amyloid fibrils in several components. Additionally, this appears red due to bonds (Antimonova et al., 2024). There was a notable increase in beta-amyloid levels in the Gemcitabine category resulting from the fact that Gemcitabine increases cellular stress from oxidative damage, which in turn causes an increase in beta-amyloid synthesis (Tamagno et al., 2021). Additionally, because ALC is administered, the nervous system produces less beta-amyloid due to lowering the quantity of ROS within the central nervous system, which aids their brain resulting in less beta-amyloid and, consequently, the elimination of the fluorescent red color from the tissues, as changes depicted in figures (Morid et al., 2023; Mateus et al., 2023). ConclusionAccording to the research findings, ALC reduces the negative effects of gemcitabine by primarily lowering the body’s concentrations of beta-amyloid and the buildup of the protein beta-lesions. AcknowledgmentThe authors thank the laboratory staff and colleagues for their technical support and valuable assistance throughout this study. The authors also thank the ethics committee for approving the research protocol and the institution for providing the necessary facilities. Conflict of interestThe authors declare that there is no conflict of interest. FundingNone. Authors’ contributionsMarwa Sabah Majed: Materials and Methods. Fatema Ali Al Kafhage: Results. Hiba Alameri: Discussion. Amaal Sahib Al-Zughaibi contributed to the statistical analysis of the study data. Amna Mohammed Hamza contributed to the writing and preparation of the manuscript. Roaa Noori Ali was responsible for conducting the experimental work and collecting the samples. All authors read and approved the final manuscript. Data availabilityAll data were presented in the study. ReferencesAntimonova, O.I., Polyakov, D.S., Grudinina, N.A., Masterova, K.V. and Shavlovsky, M.M. 2024. Antibodies to Congo red dye as a promising tool for detection and destruction of amyloid. Cell. Tissue. Biol. 18(6), 721–727. Ashraf, G.M. 2023. Neurochemical systems and signaling: from molecules to networks. Boca Raton, FL: CRC Press. Bansal , S., Yadav, M., Bisht, P., Bansal, D., Tushir, S. and Rathore, D. 2025. Β-Amyloid pathways in Alzheimer's disease: mechanisms and therapeutic targets. CNS Neurol. Disord-Drug. Targets. Davidová, M., Sharma, S., McMeel, D. and Loizides, F. 2022. Co-De| GT: The gamification and tokenisation of more-than-human qualities and values. Sustainability 14(7), 3787. Goel, P., Chakrabarti, S., Goel, K., Bhutani, K., Chopra, T. and Bali, S. 2022. Neuronal cell death mechanisms in Alzheimer’s disease: an insight. Front. Mol. Neurosci. 15, 937133. Gottwald, J. and Röcken, C. 2021. The amyloid proteome: a systematic review and proposal of a protein classification system. Crit. Rev. Biochem. Mol. Biol. 56(5), 526–542. Guliyeva, A., Abbasova, M. and Gadzhiev, A. 2025. Blood leukocyte formula in rats exposed to electromagnetic radiation during prenatal development. Adv. Biol. Earth. Sci. 10(3), 511–518; doi:10.62476/abes.103511 Huseynova, L. (2025). Pyruvate kinase modulation in the brain under stress factors: structural, developmental, and molecular perspectives. Adv. Biol. Earth Sci. 10(3), 488–498; doi:10.62476/abes.103488 Javan, M., Ebrahimi, M., Madatli, A., Madatli, F. and Karimova, R. (2025). Acetylation and sirtuins: molecular mechanisms driving metabolic flexibility. Adv. Biol. Earth Sci. 10(3), 547–584; doi:10.62476/abes.103547 Kıran, T.R., Otlu, O. and Karabulut, A.B. 2023. Oxidative stress and antioxidants in health and disease. J. Lab. Med. 47(1), 1–11. Lakshmanan, M. 2021. Cholinoceptor agonists and anticholinesterase agents. In Introduction to basics of pharmacology and toxicology: volume 2: essentials of systemic pharmacology: from principles to practice. Singapore: Springer, 2, pp: 3–24. Larson, A.C., Doty, K.R. and Solheim, J.C. 2024. The double life of a chemotherapy drug: immunomodulatory functions of gemcitabine in cancer. Cancer. Med. 13(10), e7287. Madkour, H., Ahmed, S. and Mohammed, W. 2022. L-carnitine ameliorates methotrexate-induced ovarian dysfunction in female rats. Records Pharm. Biomed. Sci. 6(3), 14–27. Mahdi, S.S., Albazi, W. and Hussain Al-aameli, M. 2021. The beneficial effect of glutathione in protecting the central nervous system from D-galactose-induced damage. In 1st International Ninevah Conference on Medical Sciences (INCMS 2021), Amsterdam, The Netherlands: Atlantis Press, pp 70–74. Maleki, N., Amiruddin, R., Moawad, A.W., Yordanov, N., Gkampenis, A., Fehringer, P. and Taherzadeh, M. 2025. Analysis of the MICCAI brain tumor segmentation--metastases (BraTS-METS) 2025 lighthouse challenge: brain metastasis segmentation on pre-and post-treatment MRI. arXiv preprint arXiv:2504.12527. Mateus, F.G., Moreira, S., Martins, A.D., Oliveira, P.F., Alves, M.G. and Pereira, M.D.L. 2023. L-carnitine and male fertility: is supplementation beneficial?. J. Clin. Med. 12(18), 5796. Metselaar, D.S., Meel, M.H., Goulding, J.R., Du Chatinier, A., Rigamonti, L., Waranecki, P., Geisemeyer, N., De Gooijer, M.C., Breur, M., Koster, J., Veldhuijzen Van Zanten, S.E.M., Bugiani, M., Franke, N.E., Reddy, A., Wesseling, P., Kaspers, G.J.L. and Hulleman, E. 2024. Gemcitabine therapeutically disrupts essential SIRT1-mediated p53 repression in atypical teratoid/rhabdoid tumors. Cell Rep. Med. 5(9). Morgillo, A. and Marovino, E. 2021. Chemotherapy neurotoxicity: pharmacological and toxicological aspects. Int. J. Curr. Res. 13(12), 19916–19922. Morid, O.F., Menze, E.T., Tadros, M.G. and George, M.Y. 2023. L-Carnitine modulates cognitive impairment induced by doxorubicin and cyclophosphamide in rats; insights to oxidative stress, inflammation, synaptic plasticity, liver/brain, and kidney/brain axes. J. Neuroimmune. Pharmacol. 18(3), 310–326. Tamagno, E., Guglielmotto, M., Vasciaveo, V. and Tabaton, M. 2021. Oxidative stress and -amyloid in Alzheimer’s disease which comes first: the chicken or the egg?. Antioxidants 10(9), 1479. Taub, L. 2023. Ancient Greek and Roman science: a very short introduction (Vol. 731). Oxford, UK: Oxford University Press, p: 731. Tauffenberger, A. and Magistretti, P.J. 2021. Reactive oxygen species: beyond their reactive behavior. Neurochem. Res. 46(1), 77–87. Virmani, M.A. and Cirulli, M. 2022. The role of L-carnitine in mitochondria, prevention of metabolic inflexibility and disease initiation. Int. J. Mol. Sci. 23(5), 2717. Wang, W., Pan, D., Liu, Q., Chen, X. and Wang, S. 2024. L-carnitine in the treatment of psychiatric and neurological manifestations: a systematic review. Nutrition 16(8), 1232. Whitfield, C.J., Cavaliere, E., Baulch, H.M., Clark, R.G., Spence, C., Shook, K.R. and Wolfe, J.D. 2024. An integrated assessment of impacts to ecosystem services associated with prairie pothole wetland drainage quantifying wide-ranging losses. Facets 9, 1–15. Yi, E., Chamorro González, R., Henssen, A.G. and Verhaak, R.G.W. 2022. Extrachromosomal DNA amplifications in cancer. Nat. Rev. Genet. 23(12), 760–771. Yin, X., Qiu, Y., Zhao, C., Zhou, Z., Bao, J. and Qian, W. 2021. The role of amyloid-beta and tau in the early pathogenesis of Alzheimer’s disease. Med. Sci. Monitor. Int. Med. J. Exp. Clin. Res. 27, e933084-1. Zulfugarova, P.A., Humbatov, S.I. and Mekhtiev, A.A. 2025. Molecular mechanisms of adaptation to desynchronization stress. Adv. Biol. Earth Sci. 10(3). | ||
| How to Cite this Article |
| Pubmed Style Majed MS, Al-kafhage FA, Alameri H, Al-zughaibi AS, Hamza AM, Ali RN. Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Vet. J.. 2026; 16(3): 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 Web Style Majed MS, Al-kafhage FA, Alameri H, Al-zughaibi AS, Hamza AM, Ali RN. Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. https://www.openveterinaryjournal.com/?mno=285394 [Access: March 31, 2026]. doi:10.5455/OVJ.2026.v16.i3.26 AMA (American Medical Association) Style Majed MS, Al-kafhage FA, Alameri H, Al-zughaibi AS, Hamza AM, Ali RN. Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Vet. J.. 2026; 16(3): 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 Vancouver/ICMJE Style Majed MS, Al-kafhage FA, Alameri H, Al-zughaibi AS, Hamza AM, Ali RN. Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Vet. J.. (2026), [cited March 31, 2026]; 16(3): 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 Harvard Style Majed, M. S., Al-kafhage, . F. A., Alameri, . H., Al-zughaibi, . A. S., Hamza, . A. M. & Ali, . R. N. (2026) Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Vet. J., 16 (3), 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 Turabian Style Majed, Marwa Sabah, Fatema Ali Al-kafhage, Hiba Alameri, Amaal Sahib Al-zughaibi, Amna Mohammed Hamza, and Roaa Noori Ali. 2026. Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Veterinary Journal, 16 (3), 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 Chicago Style Majed, Marwa Sabah, Fatema Ali Al-kafhage, Hiba Alameri, Amaal Sahib Al-zughaibi, Amna Mohammed Hamza, and Roaa Noori Ali. "Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model." Open Veterinary Journal 16 (2026), 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 MLA (The Modern Language Association) Style Majed, Marwa Sabah, Fatema Ali Al-kafhage, Hiba Alameri, Amaal Sahib Al-zughaibi, Amna Mohammed Hamza, and Roaa Noori Ali. "Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model." Open Veterinary Journal 16.3 (2026), 1687-1694. Print. doi:10.5455/OVJ.2026.v16.i3.26 APA (American Psychological Association) Style Majed, M. S., Al-kafhage, . F. A., Alameri, . H., Al-zughaibi, . A. S., Hamza, . A. M. & Ali, . R. N. (2026) Acetyl-L-carnitine protective role in gemcitabine-induced neurotoxicity in an experimental rat model. Open Veterinary Journal, 16 (3), 1687-1694. doi:10.5455/OVJ.2026.v16.i3.26 |