| Case Report | ||
Open Vet J. 2023; 13(3): 388-393 Open Veterinary Journal, (2023), Vol. 13(3): 388–393 Case Report Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dogThomas Dulaurent*, Laure Steun and Pierre-François IsardCentre Hospitalier Vétérinaire Saint-Martin, Allonzier-la-Caille, France *Corresponding Author: Thomas Dulaurent. Centre Hospitalier Vétérinaire Saint-Martin, Allonzier-la-Caille, France. Email: t.dulaurent [at] chvsm.com. Submitted: 19/12/2022 Accepted: 16/02/2023 Published: 23/03/2023 © 2023 Open Veterinary Journal
AbstractBackground: Cat scratches and ocular foreign bodies are frequent reasons for consultation in veterinary ophthalmology. Case Description: We here present an unusual case combining trauma to the cornea and lens by a cat scratch with retention of the claw in the anterior chamber. Management consisted of the removal of the claw, reconstruction of the cornea, and mechanized ablation of the lens by phacoemulsification with implantation of an artificial lens. Conclusion: The progression during the follow-up period was considered satisfactory, with positive responses to visual tests and intraocular pressure within the norms. Only dyscoria and a tear of the endothelium and Descemet membrane, secondary to the trauma, remained. Keywords: Claw, Cornea, Endothelium, Lens, Wound. IntroductionThe trauma of the ocular surface by a cat scratch (CS) is a common reason for veterinary ophthalmology consultation (Davidson et al., 1991; Spiess et al., 1996; Denis, 2002; Paulsen and Kass, 2012). In dogs, this situation occurs most often in adolescent subjects, following their introduction into a new home where a cat is already in residence (Paulsen and Kass, 2012). The young dog, by its naive attitude and exploratory behavior, goes to meet the house cat, which is a territorial animal and potentially aggressive by nature. The deleterious effects of CS are related to trauma (Davidson et al., 1991; Spiess et al., 1996; Doi et al., 1999; Denis, 2002; Sylvester et al., 2002; Williams et al., 2002; Paulsen and Kass, 2012; Borkar et al., 2021), the septic risk (Tanasescu et al., 1996; Oehler et al., 2009; Weinberg and Branda, 2010; Veraldi and Minuti, 2018), and the immune response involved (Wilcock and Peiffer, 1987; Streilein, 2003; Caspi, 2008; Perez et al., 2013). The claws of cats are indeed naturally sharp, in connection with predation. The lesions are often severe and correspond to a sharp trauma. They range from a simple tear of the conjunctiva to corneal lacerations sometimes associated with tears of the anterior capsule of the lens with the release of lens material into the anterior chamber AC (Davidson et al., 1991; Paulsen and Kass, 2012). Ocular foreign bodies (FB) are quite common in veterinary ophthalmology (Slatter and Bryan, 1972; Schmidt et al., 1975; Bussanich and Rootman, 1981; Grahn et al., 1995; Cullen and Grahn, 2005; Sandmeyer et al., 2007; Belknap, 2015; Lew et al., 2015; Tetas Pont et al., 2016). The diagnosis of intraocular FB is often more difficult to establish than surface FB because of their sometimes marginal location (iridocorneal angle) and the often intense inflammatory reaction that masks them (fibrinous-hemorrhagic exudate) (Tetas Pont et al., 2016). As with CS trauma, lesions associated with ocular FB are variable and range from benign conjunctivitis to more serious lesions that can involve the cornea, lens, and posterior segment (Tetas Pont et al., 2016). Although both situations (CS to the eye and ocular FB) are very common and have been described in case reports, cases series, and textbooks (Root, 2010), the combination of a CS and the retention of the claw into the eye has, to the authors’ knowledge, never been published. The clinical case presented here describes the management of an unusual case of laceration of the cornea and lens by a CS, with retention of the claw in the AC after trauma in a Bichon. Case DetailsAn 8-year-old castrated male Bichon was presented for emergency ophthalmology consultation for evaluation of a FB located in the AC of the right eye following a walk that same afternoon. The dog was not kept on a leash and was free to go where it wanted. General reviewThe general examination did not reveal any anomaly. Eye examinationThe responses to visual tests were positive on the left. The menace response was positive on the right, while the dazzle reflex was variable and difficult to interpret as a result of eye pain due to blepharospasm of moderate intensity. The left eye examination did not reveal any abnormalities. The right eye examination revealed the presence of sparse epiphora. A curvilinear, white, FB was present in the AC. Close examination with a biomicroscope revealed that it was a cat’s claw, for which the point of the corneal penetration was located in the upper quadrant. The latter was filled by a fibrin clot. Discrete corneal edema was present, as well as a uveal reaction manifested by tight myosis precluding observation of the lens (Fig. 1). The intraocular pressure was 7 mmHg, measured by rebound tonometry. Given the nature of the signs, FB extraction surgery was undertaken, associated with the perioperative exploration of the lens. Surgical procedureInduction was achieved by intravenous injection of propofol after tranquilization by intravenous administration of valium. Anesthesia was then maintained by the administration of a mixture of oxygen and isoflurane via the respiratory route after endotracheal intubation. Analgesia was achieved by intravenous injection of morphine, supplemented by local instillation of oxybuprocaine hydrochloride. After positioning the patient in a supine position, with the head held on a vacuum cushion, the surgical site was prepared for the procedure by six consecutive cleanings using a solution of 1% diluted povidone-iodine.
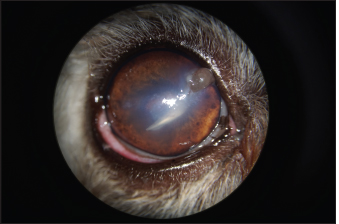
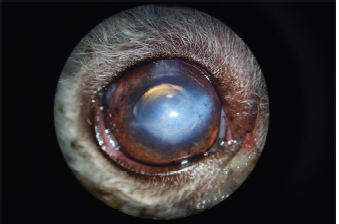
Fig. 1. Appearance of the eye at admission. Note the broken claw in the AC, corneal edema, fibrin clogging the entry point, and uveal reaction materialized by myosis.
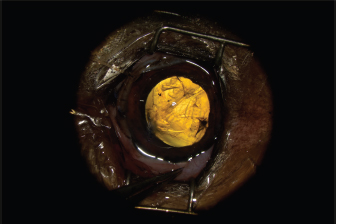
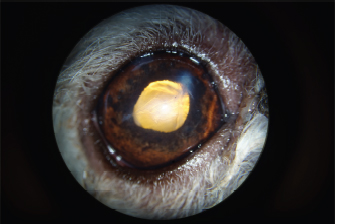
Fig. 2. Appearance of the eye after claw excision, corneal reconstruction, and pharmacological mydriasis. The tear of the anterior capsule is large and complex. The architecture of the lens is turned upside down. The AC was stabilized by intracameral injection of a viscoelastic gel (hyaluronic acid 1.6%, Ophteis Bio) by a perforating keratotomy port at 12 o’clock. As the base of the claw was crenelated, it was not possible to remove it through the site of penetration, which is why another perforating keratotomy port was made at 10 o’clock. The claw was grasped with Bonn’s pliers and then gently removed. The entry point of the claw and the 10 o’clock port were sutured by single stitches using polyglactin 9/0 (Vicryl 9/0). The pupil was then dilated by injection of 1 ml of epinephrine at a concentration of 1 mg/ml into the AC. The anterior lens was ruptured, and a heterogeneous traumatic cataract had already developed (Fig. 2). The lens was, therefore, removed by phacoemulsification via the port at 12 o’clock. After removal of the masses by irrigation/aspiration, the tear of the anterior capsule was shaped before injection of an implant (PFI 4X, Medicontur). The keratotomy port at 12 o’clock was sutured by three single stitches with Vicryl 9/0 after the removal of residual viscoelastic material by suction irrigation. At this stage of the surgery, a tear of the endothelium and Descemet membrane was visible, following the most likely trajectory of the claw into the AC (Fig. 3). Water bubbles were already forming in the corneal stroma. An injection of 0.25 μg of tissue plasminogen activator (Actilyse®, BOEHRINGER INGELHEIM) was performed in the AC at the end of the intervention to limit postoperative fibrinous collection. The claw was sent to the laboratory for bacteriological analysis. Postoperative medical treatmentThe prescribed medical treatment consisted of the administration of local (tobramycin eye drops, Tobrex®, ALCON, TiD) and general (amoxicillin and clavulanate, Bioclamox®, Axience, 12.5 mg/kg PO BiD, 10 days) antibiotics and systemic steroidal anti-inflammatory drugs (prednisolone, Megasolone®, DOPHARMA, 0.5 mg/kg PO SiD, 1 week), combined with local administration of a cycloplegic (atropine eye drops 1%, ALCON, TiD 1, week).
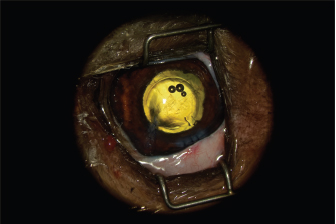
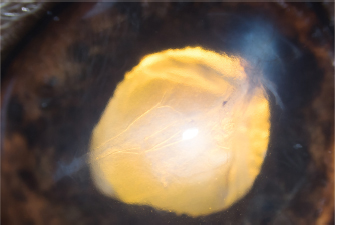
Fig. 3. Appearance of the eye after implantation. The endothelium and Descemet membrane are torn, which causes early formation of water compartments in the corneal stroma. Follow-upThe bacteriological analysis revealed contamination of the claw with Pasteurella spp., which is sensitive to tobramycin. The follow-up visit at 1 week showed significant degradation of the corneal edema, which then occupied the entire corneal surface. The pupillary light reflex was not discernible and the keratotomy wounds and the point of entry of the claw appeared excessively edematous (Fig. 4). The patient’s comfort was slightly improved, with the persistence of discrete blepharospasm increased by the examination. The intraocular pressure was measured to be 10 mmHg by rebound tonometry. It was decided to continue the medical treatment without any modifications. The follow-up visit at 3 weeks revealed marked improvement in the corneal transparency, with the persistence of edema at the ports, the point of penetration of the claw, and the tear area of the endothelium and Descemet membrane. The patient’s comfort was satisfactory, with the almost total disappearance of the blepharospasm (Fig. 5). The intraocular pressure was measured to be 8 mmHg by rebound tonometry. The medical treatment consisted of local instillation of dexamethasone (Maxidex®, ALCON, TiD) for 3 months.
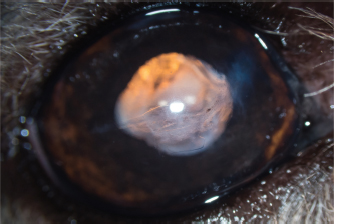
Fig. 4. Appearance of the eye 1 week after the procedure. Note the corneal edema masking the AC, the ciliary injection, and the swelling of the keratotomy port at 12 o’clock.
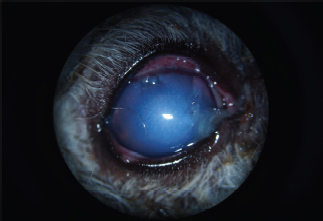
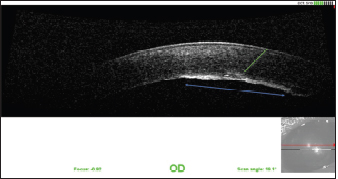
Fig. 5. Appearance of the eye 3 weeks after the procedure. The intensity of corneal edema has decreased. The tear of the endothelium and Descemet membrane is still visible. The various follow-up visits revealed improvement in comfort, with the disappearance of the blepharospasm. The corneal edema was greatly improved. The visual tests were positive and the intra ocular pressure (IOP) was stable. Six months after the surgery, the observable sequelae were the persistence of discrete corneal edema in the area of the point of penetration of the claw as well as dyscoria (Fig. 6). The tear area of the endothelium and Descemet membrane was still visible at high magnification (Fig. 7). One year after the surgery, the vision tests were positive. The cornea had fibrosis at the point of impact and opposite the endothelium and Descemet membrane tear, which was still visible (Fig. 8). The pupil was slightly deformed and the capsule of the lens was partially fibrosed. Optical coherence tomography (OCT) of the cornea revealed fibrous metaplasia of the endothelium and probable duplication of the Descemet membrane. The intraocular pressure was measured to be 13 mmHg by rebound tonometry (Fig. 9).
Fig. 6. Appearance of the eye 6 months after the procedure. The edema is reduced. The cornea is fibrosed at the point of impact.
Fig. 7. High magnification of Figure 6 showing the endothelium and Descemet tear and the associated corneal edema. DiscussionThe unusual nature of this case lies in the combination of a scratch of the cornea and lens by a cat, and in the retention of the claw in the AC after the trauma. Claws naturally carry many bacteria, which are likely to inoculate in the eye at the time of the trauma (Escherichia coli, Pseudomonas aeruginosa, β-hemolytic streptococci, Pasteurella multocida, Bartonella henselae, Moraxella spp., Staphylococcus spp., Streptococcus spp.), which can cause bacterial endophthalmitis (Oehler et al., 2009; Weinberg and Branda, 2010; Veraldi and Minuti, 2018). In our case, the claw carried Pasteurella spp., a ubiquitous microorganism, although no sign of infection was yet visible as the patient was presented only a few hours after the trauma. The retention of the claw in the AC represented a reservoir of germs, increasing the risk of endophthalmitis in the short term. Organic FB (e.g., plant spines, needles, bark, porcupine spines) are more often associated with bacterial endophthalmitis than non-organic FB (e.g., glass, plastic, metal, etc.) (Lit and Young, 2002). In our case, the claw entered the AC through a fairly small corneal entry point. The most likely scenario is that the claw broke at the beginning of the scratch and not at the end, minimizing the corneal laceration. However, by entering the AC, the claw lacerated the endothelium, the Descemet membrane, and the anterior capsule of the lens, also disorganizing the anterior cortex. When the lens is injured during trauma, the immune system may come into contact with lens proteins that were initially sequestered (Wilcock and Peiffer, 1987; Streilein, 2003; Caspi, 2008; Paulsen and Kass, 2012; Perez et al., 2013). The resulting phacoclastic uveitis is often severe and associated with inflammation involving intralenticular neutrophils and perilenticular fibroplasia (Wilcock and Peiffer, 1987; Paulsen and Kass, 2012). Here, the diagnosis of a FB was easy due to its large size and the surprisingly weak corneal edema and uveal reaction. When FB is located only on the surface of the cornea, ablation by hydropulsion may be sufficient (Labelle et al., 2014). When FB is penetrating, management often requires surgery consisting of removal of the FB and reconstruction of the cornea (Lew et al., 2015; Tetas Pont et al., 2016). The guidelines for the management of anterior capsule tears are, however, contradictory. Some authors recommend surgical management for tears greater than 1.5 mm, especially when lens material has been expelled into the AC after the trauma (Davidson et al., 1991). The main argument for an interventionist attitude is the risk of developing phacoclastic uveitis or septic implantation syndrome (Davidson et al., 1991; Bell et al., 2013). This syndrome results in severe and uncontrollable endophthalmitis, of slow progression, occurring a few weeks or months after eye trauma and rupture of the anterior capsule, with inoculation of microorganisms (bacteria, fungi) (Bell et al., 2013). The study Paulsen and Kass (2012), regarding the management of 77 cases of corneal laceration associated with a tear of the anterior capsule, showed that medical management alone had a better long-term prognosis than surgical management of the cornea and lens, provided that the corneal wound offers good apposition, the AC is reconstituted by the second aqueous humor (AH) without iridocoele, and the Seidel test is negative. The authors refer to the healing capacity of the anterior capsule by fibrin effusion and fibrous metaplasia of the capsule, again allowing sequestration of the lens material initially released in the AC, even after a few months. This conclusion, which is rather counterintuitive, had no influence on the management of our case since the claw was broken in the AC after the trauma, thus requiring a surgical approach anyway. The decision to perform phacoemulsification was dictated by the large and complex tearing of the anterior capsule and the disorganization of the anterior cortex, even though the lens material still appeared to be in place.
Fig. 8. Appearance of the eye 1 year after the surgery. The cornea has a slight degree of fibrosis at the point of entry of the claw and at the separation of the endothelium and Descemet membrane.
Fig. 9. OCT of the cornea 1 year after the surgery. Note the fibrous metaplasia of the endothelium (blue arrow) and probable duplication of the Descemet membrane (green arrow). The tear of the endothelium and Descemet membrane caused significant corneal edema by imbibition of the stroma by the AH, visible at the follow-up visit at 1 week. The various follow-up visits showed favorable progression of the edema, with an almost total disappearance at 9 months after the surgery. The endothelial phase of corneal healing normally begins 24 hours after trauma (Whitley and Hamor, 2021). In adult dogs, endothelial cells are incapable of mitosis (Befanis et al., 1981). The endothelium heals mainly by rearrangement of the cytoskeleton of the cells that compose it. The latter then occupy a larger surface area and migrate by sliding to fill the deficit related to the trauma. The density of endothelial cells decreases after trauma. Endothelial cells undergo fibrous metaplasia and produce a new Descemet membrane after a few weeks (Whitley and Hamor, 2021). Duplication of the Descemet membrane may occur after perforating trauma to the cornea (Kafarnik et al., 2009). In our case, endothelial compensation allowed the maintenance of corneal deturgescence during the follow-up period, thus ensuring the maintenance of satisfactory vision for the patient. The tear of the endothelium and Descemet membrane nevertheless remained visible by slit lamp at the check-up visit at 1 year. The case presented here describes the management of a broken cat’s claw in the AC of a dog after a laceration of the cornea. The corneal and lens lesions were treated surgically and successfully medicated. The prognosis was improved by early patient management. However, this case has some limitations. The most important one is the duration of the follow-up period, which was quite short. The wound to the corneal endothelium could probably have further consequences in the future, such as endothelial decompensation and subsequent chronic and intractable corneal edema. Close monitoring is thus required in the future, to better document the progression of such a corneal wound. Conflict of interestThe authors declare that there is no conflict of interest. ReferencesBefanis, P.J., Peiffer, R.L. and Brown, D. 1981. Endothelial repair of the canine cornea. Am. J. Vet. Res. 42, 590–595. Belknap, E.B. 2015. Corneal emergencies. Top. Companion Anim. Med. 30, 74–80. Bell, C.M., Pot, S.A. and Dubielzig, R.R. 2013. Septic implantation syndrome in dogs and cats: a distinct pattern of endophthalmitis with lenticular abscess. Vet. Ophthalmol. 16, 180–185. Borkar, D. and Avendano, P. 2021. Cat-inflicted eye injury in a child. J. Med. Cases. 12, 209–212. Bussanich, M.N. and Rootman, J. 1981. Intraocular foreign body in a dog. Can. Vet. J. 22, 207–210. Caspi R. 2008. Autoimmunity in the immune privileged eye: pathogenic and regulatory T cells. Immun. Res. 42, 41–50. Cullen, C.L. and Grahn, B.H. 2005. Diagnostic ophthalmology. Right corneal foreign body, secondary ulcerative keratitis, and anterior uveitis. Canadian Vet. J. 46, 1054–1055. Davidson, M.G., Nasisse, M.P., Jamieson V.E., English, R.V. and Olivero, D.K. 1991. Traumatic anterior lens capsule disruption. J. Am. Anim. Hosp. Assoc. 27, 410–414. Denis, H.M. 2002. Anterior lens capsule disruption and suspected malignant glaucoma in a dog. Vet. Ophthalmol. 5, 79–83. Doi, M., Ikeda, T., Yasuhara, T., Koizumi, K. and Nakamura, T. 1999. A case of bacterial endophthalmitis following perforating injury caused by a cat claw. Ophthal. Surg. Lasers 30, 315–316. Grahn, B.H., Szentimrey, D., Pharr, J.W., Farrow, C.S. and Fowler, D. 1995. Ocular and orbital porcupine quills in the dog: a review and case series. Can. Vet. J. 36, 488–493. Kafarnik, C., Murphy, C.J. and Dubielzig, R.R. 2009. Canine duplication of Descemet’s membrane. Vet. Pathol. 46, 464–473. Labelle, A.L., Psutka, K., Collins, S.P. and Hamor, R.E. 2014. Use of hydropulsion for the treatment of superficial corneal foreign bodies: 15 cases (1999-2013). J. Am. Vet. Med. Assoc. 244, 476–479. Lew, M., Lew, S., Drazek, M. and Pomianowski, A. 2015. Penetrating eye injury in a dog: a case report. Vet. Med. 60, 213–221. Lit, E.S. and Young, L.H. 2002. Anterior and posterior segment intraocular foreign bodies. Int. Ophthalmol. Clin. 42, 107–120. Oehler, R.L., Velez, A.P., Mizrachi, M., Lamarche, J. and Gompf, S. 2009. Bite-related and septic syndromes caused by cats and dogs. Lancet Infect. Dis. 9, 439–447. Paulsen, M.E. and Kass, P.H. 2012. Traumatic corneal laceration with associated lens capsule disruption: a retrospective study of 77 clinical cases from 1999 to 2009. Vet. Ophthalmol. 15, 355–368. Perez, V.L., Saeed, A.M., Tan, Y., Urbieta, M. and Cruz-Guilloty, F. 2013. The eye: a window to the soul of the immune system. J. Autoimmun. 45, 7–14. Root, T. 2010. Eye Trauma. https://timroot.com Available via https://timroot.com/trauma/ (Accessed 1 February 2023). Sandmeyer, L.S., Bowen, G. and Grahn, B.H. 2007. Diagnostic ophthalmology. Anterior uveitis, cataract, retinal detachment, and an intraocular foreign body. Can. Vet. J. 48, 975–976. Schmidt, G.M., Dice, P.F. and Koch, S.A. 1975. Intraocular lead foreign bodies in four canine eyes. J. Small Anim. Pract. 16, 33–39. Slatter, D.H. and Bryan, G.M. 1972. An unusual foreign body in the anterior chamber of a dog. Vet. Med. Small Anim. Clin. 67, 775–778. Spiess, B.M., Fuhli, M.R. and Bollinger, J. 1996. Eye injuries from cats claw at dog (in German). Schweizer Archiv fur Tierheilkunde. 138, 429–433. Streilein, J.W. 2003. Ocular immune privilege: therapeutic opportunities from an experiment of nature. Nat. Rev. Immunol. 3, 879–889. Sylvester, D.A., Burnstine, R.A. and Bower, J.R. 2002. Cat-inflicted corneal laceration: a presentation of two cases and a discussion of infection-related management. J. Pediat. Ophthalmol. Strabismus. 39, 114–117. Tanasescu, S., Joly, P., Le Corvaisier-Pieto, C., Dujardin, F., Latouche, J.B. and Lauret, P. 1996. Acute necrotizing cutaneous streptococcal infection following bites or scratch by dog or cat. Ann. Dermatol. Vénéréol. 123, 804–806. Tetas Pont, R., Matas Riera, M., Newton, R., and Donaldson, D. 2016. Corneal and anterior segment foreign body trauma in dogs: a review of 218 cases. Vet. Ophthalmol. 19, 386–397. Veraldi, S. and Minuti, A. 2018. Severe streptococcal infection following cat scratch. Wounds. 30, E57–E59. Weinberg, A.N. and Branda, J.A. 2010. Case records of the Massachusetts general hospital. Case 31-2010. A 29-year-old woman with fever after a cat bite. N. Eng. J. Med. 363, 1560–1568. Whitley, R.D. and Hamor, R.E. 2021. Diseases and surgery of the canine cornea and sclera. In Veterinary ophthalmology, 6th ed. Eds., Gelatt K.N., Ben-Schlomo G., Gilger CG, Hendrix D.V.H., Kern T.J., and Plummer C.E. Ames, IO: Wiley-Blackwell, pp: 1082–1172. Wilcock, B.P. and Peiffer, R.L. 1987. The pathology of lens-induced uveitis in dogs. Vet. Pathol. 24, 1549–1553. Williams, C.P., Sleep, T.J., and Morris, R.J. 2002. Lateral rectus muscle avulsion by a cat scratch. J. Am. Assoc. Pediatr. Ophthalmol. Strabismus. 6, 397–399. | ||
| How to Cite this Article |
| Pubmed Style Dulaurent T, Steun L, Isard P. Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Vet J. 2023; 13(3): 388-393. doi:10.5455/OVJ.2023.v13.i3.17 Web Style Dulaurent T, Steun L, Isard P. Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. https://www.openveterinaryjournal.com/?mno=97032 [Access: July 04, 2025]. doi:10.5455/OVJ.2023.v13.i3.17 AMA (American Medical Association) Style Dulaurent T, Steun L, Isard P. Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Vet J. 2023; 13(3): 388-393. doi:10.5455/OVJ.2023.v13.i3.17 Vancouver/ICMJE Style Dulaurent T, Steun L, Isard P. Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Vet J. (2023), [cited July 04, 2025]; 13(3): 388-393. doi:10.5455/OVJ.2023.v13.i3.17 Harvard Style Dulaurent, T., Steun, . L. & Isard, . P. (2023) Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Vet J, 13 (3), 388-393. doi:10.5455/OVJ.2023.v13.i3.17 Turabian Style Dulaurent, Thomas, Laure Steun, and Pierre-francois Isard. 2023. Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Veterinary Journal, 13 (3), 388-393. doi:10.5455/OVJ.2023.v13.i3.17 Chicago Style Dulaurent, Thomas, Laure Steun, and Pierre-francois Isard. "Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog." Open Veterinary Journal 13 (2023), 388-393. doi:10.5455/OVJ.2023.v13.i3.17 MLA (The Modern Language Association) Style Dulaurent, Thomas, Laure Steun, and Pierre-francois Isard. "Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog." Open Veterinary Journal 13.3 (2023), 388-393. Print. doi:10.5455/OVJ.2023.v13.i3.17 APA (American Psychological Association) Style Dulaurent, T., Steun, . L. & Isard, . P. (2023) Eye trauma from a cat scratch with retention of the claw in the anterior chamber in a dog. Open Veterinary Journal, 13 (3), 388-393. doi:10.5455/OVJ.2023.v13.i3.17 |